Flow Preserving Endovascular Treatment of Traumatic Pseudoaneurysms of the Distal Anterior Cerebral Artery—Case Reports and Review of Literature
Abstract
:1. Introduction
2. Case Report
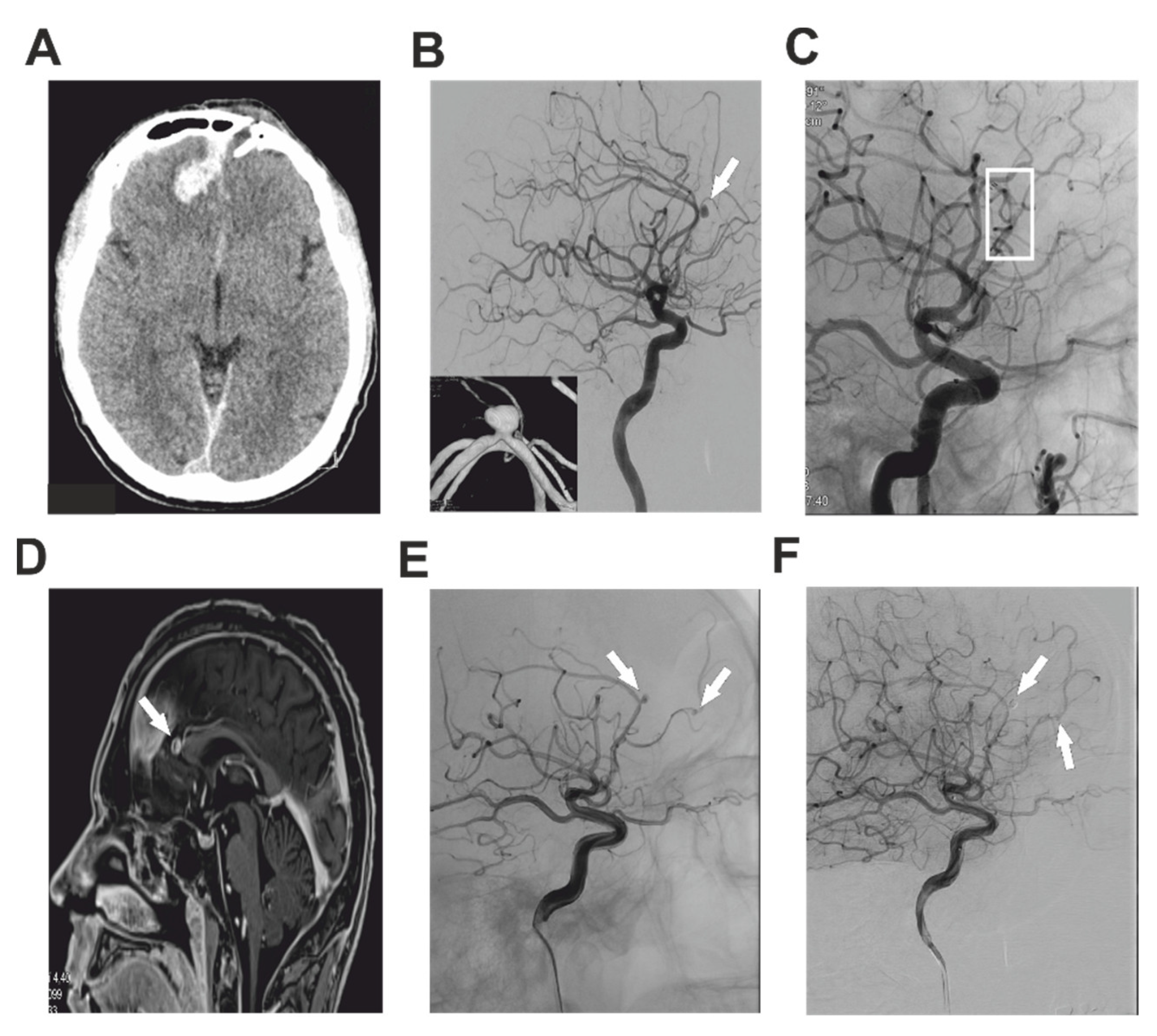
2.1. Case Report 1
2.2. Case Report 2
3. Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Larson, P.S.; Reisner, A.; Morassutti, D.J.; Abdulhadi, B.; Harpring, J.E. Traumatic intracranial aneurysms. Neurosurg. Focus 2000, 8, 1–6. [Google Scholar]
- Wang, W.; Li, M.-H.; Li, Y.-D.; Gu, B.-X.; Wang, J.; Zhang, P.-L.; Li, M. Treatment of traumatic internal carotid artery pseudoaneurysms with the Willis covered stent: A prospective study. J. Trauma Acute Care Surg. 2011, 70, 816–822. [Google Scholar] [CrossRef] [PubMed]
- Nakstad, P.; Nornes, H.; Hauge, H.N. Traumatic aneurysms of the pericallosal arteries. Neuroradiology 1986, 28, 335–338. [Google Scholar] [CrossRef] [PubMed]
- Fleischer, A.S.; Patton, J.M.; Tindall, G.T. Cerebral aneurysms of traumatic origin. Surg. Neurol. 1975, 4, 233–239. [Google Scholar] [PubMed]
- Thompson, J.R.; Harwood-Nash, D.C.; Fitz, C.R. Cerebral aneurysms in children. Am. J. Roentgenol. Radium Ther. Nucl. Med. 1973, 118, 163–175. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, D.J.; O’Dell, M.W.; Eversol, A. Delayed traumatic cerebral aneurysm after brain injury. Arch. Phys. Med. Rehabil. 1997, 78, 883–885. [Google Scholar] [CrossRef]
- Asari, S.; Nakamura, S.; Yamada, O.; Beck, H.; Sugatani, H. Traumatic aneurysm of peripheral cerebral arteries. Report of two cases. J. Neurosurg. 1977, 46, 795–803. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Smith, K.R.; Bardenheier, J.A. Aneurysm of the pericallosal artery caused by closed cranial trauma. J. Neurosurg. 1968, 29, 551–554. [Google Scholar]
- Sui, M.; Mei, Q.; Sun, K. Surgical treatment achieves better outcome in severe traumatic pericallosal aneurysm: Case report and literature review. Int. J. Clin. Exp. Med. 2015, 8, 1598–1603. [Google Scholar]
- Wewel, J.; Mangubat, E.Z.; Muñoz, L. Iatrogenic traumatic intracranial aneurysm after endoscopic sinus surgery. J. Clin. Neurosci. 2014, 21, 2072–2076. [Google Scholar] [CrossRef]
- Yoon, S.; Burkhardt, J.-K.; Lawton, M.T. Long-term patency in cerebral revascularization surgery: An analysis of a consecutive series of 430 bypasses. J. Neurosurg. JNS 2019, 131, 80–87. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Velz, J.; Esposito, G.; Regli, L. Traumatic Distal Anterior Cerebral Artery Aneurysms—Pathomechanism and Revascularisation Strategies. J. Stroke Cerebrovasc. Dis. 2021, 30, 105578. [Google Scholar] [CrossRef]
- He, Y.; Wang, L.; Ou, Y.; Wang, H.; Wang, S.; Zhang, P.; He, X.; Guo, D. Surgical treatment of traumatic distal anterior cerebral artery aneurysm: A report of nine cases from a single centre. Acta Neurochir. 2020, 162, 523–529. [Google Scholar] [CrossRef] [PubMed]
- Cohen, J.E.; Gomori, J.M.; Segal, R.; Spivak, A.; Margolin, E.; Sviri, G.; Rajz, G.; Fraifeld, S.; Spektor, S. Results of endovascular treatment of traumatic intracranial aneurysms. Neurosurgery 2008, 63, 476–486. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.H.; McCarthy, D.J.; Sheinberg, D.; Hanel, R.; Sur, S.; Jabbour, P.; Atallah, E.; Chalouhi, N.; Dumont, A.; Amenta, P.; et al. Pipeline Embolization Device for the Treatment of Intracranial Pseudoaneurysms. World Neurosurg. 2019, 127, e86–e93. [Google Scholar] [CrossRef] [PubMed]
- Grossberg, J.A.; Rebello, L.C.; Haussen, D.C.; Bouslama, M.; Bowen, M.; Barreira, C.M.; Belagaje, S.R.; Frankel, M.R.; Nogueira, R.G. Beyond Large Vessel Occlusion Strokes: Distal Occlusion Thrombectomy. Stroke 2018, 49, 1662–1668. [Google Scholar] [CrossRef]
- Park, H.; Jeong, Y.S.; Lee, S.H.; Jang, S.H.; Kwon, D.H.; Hong, J.H.; Sohn, S.I.; Yoo, J. Clinical prognosis of isolated anterior cerebral artery territory infarction: A retrospective study. BMC Neurol. 2021, 21, 171. [Google Scholar] [CrossRef]
- Moshayedi, P.; Giurgiutiu, D.V.; Ducruet, A.F.; Jankowitz, B.T.; Jadhav, A.P. Provocative Testing Prior to Anterior Cerebral Artery Fusiform Aneurysm Embolization. Interv. Neurol. 2018, 7, 36–41. [Google Scholar] [CrossRef]
- Cagnazzo, F.; Cappucci, M.; Dargazanli, C.; Lefevre, P.H.; Gascou, G.; Riquelme, C.; Bonafe, A.; Costalat, V. Treatment of Distal Anterior Cerebral Artery Aneurysms with Flow-Diverter Stents: A Single-Center Experience. AJNR. Am. J. Neuroradiol. 2018, 39, 1100–1106. [Google Scholar] [CrossRef]
- Kadyrov, N.A.; Friedman, J.A.; Nichols, D.A.; Cohen-Gadol, A.A.; Link, M.J.; Piepgras, D.G. Endovascular treatment of an internal carotid artery pseudoaneurysm following transsphenoidal surgery: Case report. J. Neurosurg. 2002, 96, 624–627. [Google Scholar] [CrossRef]
- Lee, C.-H.; Luo, C.-B. Pseudoaneurysm of the basilar artery presenting with epistaxis. Br. J. Neurosurg. 2020, 34, 475–476. [Google Scholar] [CrossRef] [PubMed]
- Horowitz, M.B.; Kopitnik, T.A.; Landreneau, F.; Ramnani, D.M.; Rushing, E.J.; George, E.; Purdy, P.P.; Samson, D.S. Multidisciplinary approach to traumatic intracranial aneurysms secondary to shotgun and handgun wounds. Surg. Neurol. 1999, 51, 31–42, discussion 41–32. [Google Scholar] [CrossRef]
- Ventureyra, E.C.; Higgins, M.J. Traumatic intracranial aneurysms in childhood and adolescence. Case reports and review of the literature. Child’s Nerv. Syst. 1994, 10, 361–379. [Google Scholar] [CrossRef] [PubMed]
- Yuen, C.-M.; Kuo, Y.-L.; Ho, J.-T.; Liao, J.-J. Rapid regrowth of a successfully coiled traumatic pericallosal aneurysm. J. Clin. Neurosci. 2007, 14, 1215–1219. [Google Scholar] [CrossRef]
- Levine, N.B.; Tanaka, T.; Jones, B.V.; Crone, K.R. Minimally invasive management of a traumatic artery aneurysm resulting from shaken baby syndrome. Pediatr. Neurosurg. 2004, 40, 128–131. [Google Scholar] [CrossRef]
- Sim, S.Y.; Shin, Y.S.; Yoon, S.H. Endovascular internal trapping of traumatic pericallosal pseudoaneurysm with hydrogel-coated self-expandable coil in a child: A case report. Surg. Neurol. 2008, 69, 418–422. [Google Scholar] [CrossRef]
- Van Rooij, W.J.; Van Rooij, S.B. Endovascular treatment of traumatic pericallosal artery aneurysms. A case report. Interv. Neuroradiol. 2013, 19, 56–59. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Krůpa, P.; Krajina, A.; Lojík, M.; Adamkov, J.; Česák, T. Flow Preserving Endovascular Treatment of Traumatic Pseudoaneurysms of the Distal Anterior Cerebral Artery—Case Reports and Review of Literature. Brain Sci. 2022, 12, 634. https://doi.org/10.3390/brainsci12050634
Krůpa P, Krajina A, Lojík M, Adamkov J, Česák T. Flow Preserving Endovascular Treatment of Traumatic Pseudoaneurysms of the Distal Anterior Cerebral Artery—Case Reports and Review of Literature. Brain Sciences. 2022; 12(5):634. https://doi.org/10.3390/brainsci12050634
Chicago/Turabian StyleKrůpa, Petr, Antonín Krajina, Miroslav Lojík, Jaroslav Adamkov, and Tomas Česák. 2022. "Flow Preserving Endovascular Treatment of Traumatic Pseudoaneurysms of the Distal Anterior Cerebral Artery—Case Reports and Review of Literature" Brain Sciences 12, no. 5: 634. https://doi.org/10.3390/brainsci12050634
APA StyleKrůpa, P., Krajina, A., Lojík, M., Adamkov, J., & Česák, T. (2022). Flow Preserving Endovascular Treatment of Traumatic Pseudoaneurysms of the Distal Anterior Cerebral Artery—Case Reports and Review of Literature. Brain Sciences, 12(5), 634. https://doi.org/10.3390/brainsci12050634






