Neurorehabilitation through Hippotherapy on Neurofunctional Sequels of Stroke: Effect on Patients’ Functional Independence, Sensorimotor/Cognitive Capacities and Quality of Life, and the Quality of Life of Their Caregivers—A Study Protocol
Abstract
:1. Introduction
2. Materials and Methods
2.1. Design
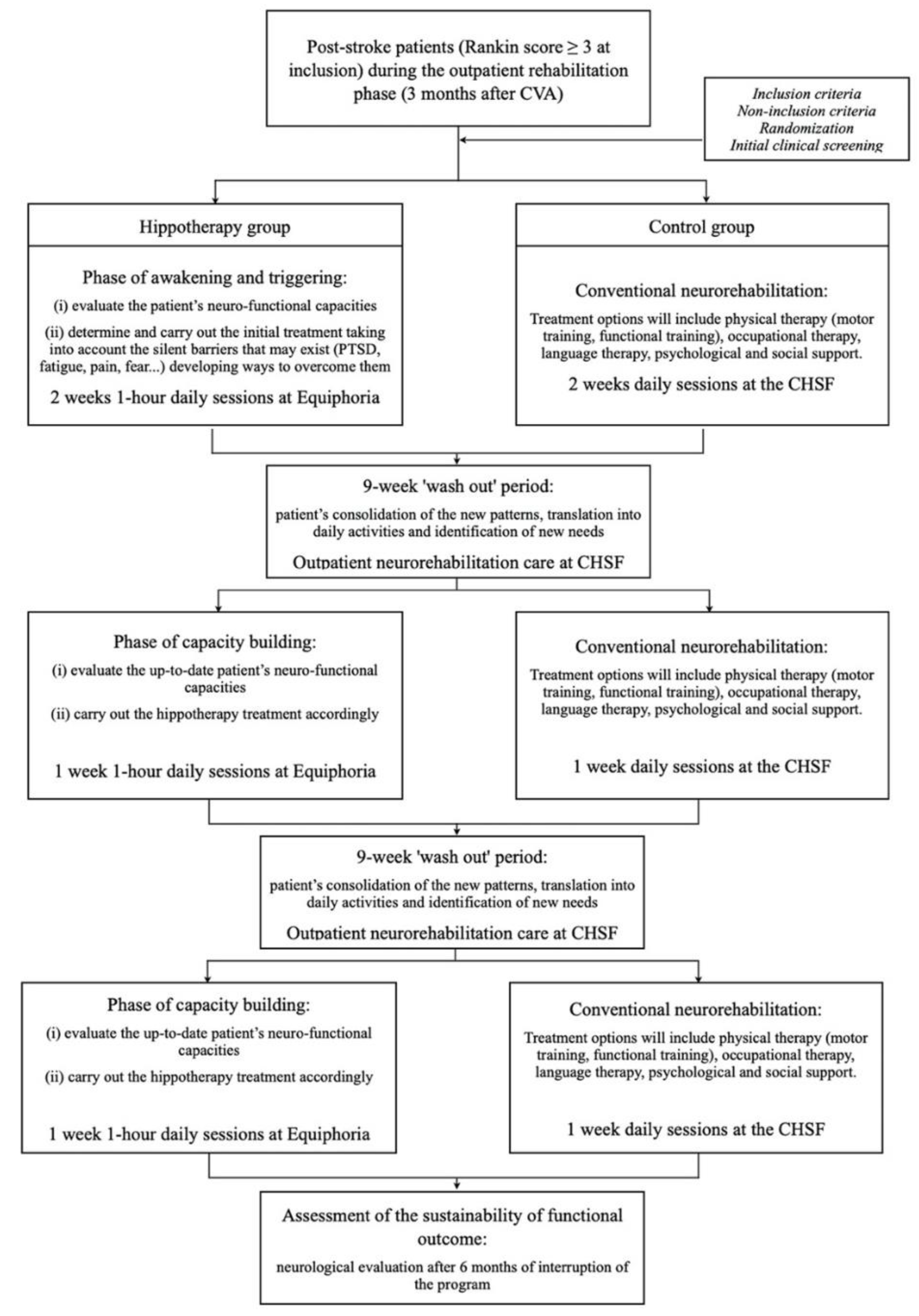
2.1.1. The Study’s Protocol
2.1.2. Primary Efficacy Endpoint
2.1.3. Secondary Efficacy Endpoint
2.1.4. Sample Size and Recruitment
2.2. Selection/Treatment of Subjects
2.2.1. Participants
2.2.2. Inclusion and Exclusion Criteria
- Age ≥ 18 years old;
- Ischemic or hemorrhagic acute stroke;
- Inclusion > 3 and ≤ 12 months post-stroke,
- Rankin score ≥ 3 and ≤ 4 at inclusion,
- Written informed consent,
- Affiliation to social security,
- Hip minimal abduction of 25 degrees bilateral with no history of hip dislocation and/or dysplasia,
- Certificate of non-contraindication.
- Major cognitive impairment affecting comprehension (Mini Mental State Examination test < 24 points);
- Global or sensory aphasia;
- Neurological or psychiatric co-morbidity (other than mild to moderate post-stroke depression);
- Evidence of uncontrolled seizures;
- Substance abuse;
- History of uncontrolled pain;
- History of allergic reactions to dust and/or horsehair, or severe asthma;
- Body weight ≥ 110 kg;
- Contraindications to physical activity;
- Inability or medical contraindication to travel to the Institute Equiphoria;
- History of therapeutic horse riding or hippotherapy during the last 6 months;
- Pregnant or lactating women;
- Patients participating in other biomedical research or in a period of exclusion.
2.3. Interventional Methods
2.3.1. Treated Group
- On the horse simulator (Racewood Ltd., Tarporley, UK): (i) 10 min of warm-up allowing patient’s familiarization, muscles’ warm-up, and nervous system facilitation (the Therapeutic Equine Simulator System TESS© has been used as valuable complement for hippotherapy in postural balance rehabilitation [12]); in some cases, the simulator can be used for the entire 1 h session depending on the patient’s needs;
- On the horse: (ii) 5 min passive and active mobilization of the lower limbs, passive and active stretching of the different muscle groups; (iii) 40 min work on global postural balance and fine-tuning of postural responses (eyes open and closed), work on upper limbs’ fine motor skills by manipulating objects, strengthening of different muscle groups, reinforcement of the body schema and body image [49], breathing techniques and visualizations; and (iv) 5 min relaxation with passive mobilizations and passive stretching, especially of the flexor muscles.
2.3.2. Control Group
2.3.3. Intensity and Effectiveness
2.3.4. Safety/Adverse Events
2.4. Data Collection and Analysis
2.4.1. Analysis Population Sets
2.4.2. Statistical Analysis
3. Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Johnson, W.; Onuma, O.; Owolabi, M.; Sachdev, S. Stroke: A global response is needed. Bull. WHO 2016, 94, 634. [Google Scholar] [CrossRef] [PubMed]
- Hankey, G.J. Stroke. Lancet 2017, 389, 641–654. [Google Scholar] [CrossRef]
- Luengo-Fernandez, R.; Paul, N.L.; Gray, A.M.; Pendlebury, S.T.; Bull, L.M.; Welch, S.J.; Cuthbertson, F.C.; Rothwell, P.M.; Oxford Vascular Study. Population-based study of disability and institutionalization after transient ischemic attack and stroke: 10-year results of the Oxford Vascular Study. Stroke 2013, 44, 2854–2861. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rice, D.; Janzen, S.; McIntyre, A.; Vermeer, J.; Britt, E.; Teasell, R. Comprehensive Outpatient Rehabilitation Program: Hospital-Based Stroke Outpatient Rehabilitation. J. Stroke Cerebrovasc. Dis. 2016, 25, 1158–1164. [Google Scholar] [CrossRef] [PubMed]
- Wissel, J.; Olver, J.; Sunnerhagen, K.S. Navigating the poststroke continuum of care. J. Stroke Cerebrovasc. Dis. 2013, 22, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Demain, S.; Wiles, R.; Roberts, L.; McPherson, K. Recovery plateau following stroke: Fact or fiction? Disabil. Rehabil. 2006, 28, 815–821. [Google Scholar] [CrossRef]
- Bernhardt, J.; Borschmann, K.; Boyd, L.; Carmichael, S.T.; Corbett, D.; Cramer, S.C.; Hoffmann, T.; Kwakkel, G.; Savitz, S.; Saposnik, G.; et al. Moving Rehabilitation Research Forward: Developing Consensus Statements for Rehabilitation and Recovery Research. Neurorehabil. Neural Repair. 2017, 31, 694–698. [Google Scholar] [CrossRef]
- Bernhardt, J.; Hayward, K.S.; Kwakkel, G.; Ward, N.S.; Wolf, S.L.; Borschmann, K.; Krakauer, J.W.; Boyd, L.A.; Carmichael, S.T.; Corbett, D.; et al. Agreed definitions and a shared vision for new standards in stroke recovery research: The Stroke Recovery and Rehabilitation Roundtable taskforce. Int. J. Stroke 2017, 12, 444–450. [Google Scholar] [CrossRef]
- Corbett, D.; Jeffers, M.; Nguemeni, C.; Gomez-Smith, M.; Livingston-Thomas, J. Lost in translation: Rethinking approaches to stroke recovery. Prog. Brain Res. 2015, 218, 413–434. [Google Scholar] [CrossRef]
- Malá, H.; Rasmussen, C.P. The effect of combined therapies on recovery after acquired brain injury: Systematic review of preclinical studies combining enriched environment, exercise, or task-specific training with other therapies. Restor. Neurol. Neurosci. 2017, 35, 25–64. [Google Scholar] [CrossRef]
- Viruega, H.; Gaillard, I.; Briatte, L.; Gaviria, M. Inter-Day Reliability and Changes of Surface Electromyography on Two Postural Muscles Throughout 12 Weeks of Hippotherapy on Patients with Cerebral Palsy: A Pilot Study. Brain Sci. 2020, 10, 281. [Google Scholar] [CrossRef] [PubMed]
- Viruega, H.; Gaillard, I.; Carr, J.; Greenwood, B.; Gaviria, M. Short- and Mid-Term Improvement of Postural Balance after a Neurorehabilitation Program via Hippotherapy in Patients with Sensorimotor Impairment after Cerebral Palsy: A Preliminary Kinetic Approach. Brain Sci. 2019, 9, 261. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Galeote, A.; Bastien, L.; Viruega, H.; Gaviria, M. Neurological rehabilitation after severe traumatic brain injury, new tools new hopes: The hippotherapy approach. J. Neurol. Neurophysiol. 2014, 5, 231. [Google Scholar] [CrossRef]
- Sterba, J.A. Does horseback riding therapy or therapist-directed hippotherapy rehabilitate children with cerebral palsy? Dev. Med. Child Neurol. 2007, 49, 68–73. [Google Scholar] [CrossRef] [PubMed]
- Bronson, C.; Brewerton, K.; Ong, J.; Palanca, C.; Sullivan, S.J. Does hippotherapy improve balance in persons with multiple sclerosis: A systematic review. Eur. J. Phys. Rehabil. Med. 2010, 46, 347–353. [Google Scholar]
- Gabriels, R.L.; Pan, Z.; Dechant, B.; Agnew, J.A.; Brim, N.; Mesibov, G. Randomized Controlled Trial of Therapeutic Horseback Riding in Children and Adolescents with Autism Spectrum Disorder. J. Am. Acad. Child Adolesc. Psychiatry 2015, 54, 541–549. [Google Scholar] [CrossRef] [Green Version]
- Han, J.Y.; Kim, J.M.; Kim, S.K.; Chung, J.S.; Lee, H.C.; Lim, J.K.; Lee, J.; Park, K.Y. Therapeutic effects of mechanical horseback riding on gait and balance ability in stroke patients. Ann. Rehabil. Med. 2012, 36, 762–769. [Google Scholar] [CrossRef]
- McGibbon, N.H.; Benda, W.; Duncan, B.R.; Silkwood-Sherer, D. Immediate and long-term effects of hippotherapy on symmetry of adductor muscle activity and functional ability in children with spastic cerebral palsy. Arch. Phys. Med. Rehabil. 2009, 90, 966–974. [Google Scholar] [CrossRef]
- Shurtleff, T.L.; Standeven, J.W.; Engsberg, J.R. Changes in dynamic trunk/head stability and functional reach after hippotherapy. Arch. Phys. Med. Rehabil. 2009, 90, 1185–1195. [Google Scholar] [CrossRef]
- Kwon, J.Y.; Chang, H.J.; Lee, J.Y.; Ha, Y.; Lee, P.K.; Kim, Y.H. Effects of hippotherapy on gait parameters in children with bilateral spastic cerebral palsy. Arch. Phys. Med. Rehabil. 2011, 92, 774–779. [Google Scholar] [CrossRef]
- Silkwood-Sherer, D.J.; Killian, C.B.; Long, T.M.; Martin, K.S. Hippotherapy—An intervention to habilitate balance deficits in children with movement disorders: A clinical trial. Phys. Ther. 2012, 92, 707–717. [Google Scholar] [CrossRef] [PubMed]
- Uchiyama, H.; Ohtani, N.; Ohta, M. Three-dimensional analysis of horse and human gaits in therapeutic riding. Appl. Animal Behav. Sci. 2011, 135, 271–276. [Google Scholar] [CrossRef]
- Fisher, P.W.; Lazarov, A.; Lowell, A.; Arnon, S.; Turner, J.B.; Bergman, M.; Ryba, M.; Such, S.; Marohasy, C.; Zhu, X.; et al. Equine-Assisted Therapy for Posttraumatic Stress Disorder Among Military Veterans: An Open Trial. J. Clin. Psychiatry 2021, 82, 271–276. [Google Scholar] [CrossRef] [PubMed]
- Menor-Rodríguez, M.J.; Sevilla Martín, M.; Sánchez-García, J.C.; Montiel-Troya, M.; Cortés-Martín, J.; Rodríguez-Blanque, R. Role and Effects of Hippotherapy in the Treatment of Children with Cerebral Palsy: A Systematic Review of the Literature. J. Clin. Med. 2021, 10, 2589. [Google Scholar] [CrossRef] [PubMed]
- Maresca, G.; Portaro, S.; Naro, A.; Crisafulli, R.; Raffa, A.; Scarcella, I.; Aliberti, B.; Gemelli, G.; Calabrò, R.S. Hippotherapy in neurodevelopmental disorders: A narrative review focusing on cognitive and behavioral outcomes. Appl. Neuropsychol. Child. 2020, 1–8. [Google Scholar] [CrossRef]
- Moraes, A.G.; Neri, S.G.R.; Motl, R.W.; Tauil, C.B.; von Glehn, F.; Corrêa, É.C.; de David, A.C. Effects of hippotherapy on postural balance, functional mobility, self-perceived fatigue, and quality of life in people with relapsing-remitting multiple sclerosis: Secondary results of an exploratory clinical trial. Mult. Scler. Relat. Disord. 2021, 52, 102948. [Google Scholar] [CrossRef]
- Lee, N.; Park, S.; Kim, J. Hippotherapy and neurofeedback training effect on the brain function and serum brain-derived neurotrophic factor level changes in children with attention-deficit or/and hyperactivity disorder. J. Exerc. Nutr. Biochem. 2017, 21, 35–42. [Google Scholar] [CrossRef]
- Bunketorp-Käll, L.; Lundgren-Nilsson, Å.; Samuelsson, H.; Pekny, T.; Blomvé, K.; Pekna, M.; Pekny, M.; Blomstrand, C.; Nilsson, M. Long-Term Improvements After Multimodal Rehabilitation in Late Phase After Stroke: A Randomized Controlled Trial. Stroke 2017, 48, 1916–1924. [Google Scholar] [CrossRef]
- Bunketorp-Käll, L.; Pekna, M.; Pekny, M.; Blomstrand, C.; Nilsson, M. Effects of horse-riding therapy and rhythm and music-based therapy on functional mobility in late phase after stroke. NeuroRehabilitation 2019, 45, 483–492. [Google Scholar] [CrossRef] [Green Version]
- Pohl, P.; Carlsson, G.; Bunketorp-Käll, L.; Nilsson, M.; Blomstrand, C. A qualitative exploration of post-acute stroke participants’ experiences of a multimodal intervention incorporating horseback riding. PLoS ONE 2018, 13, e0203933. [Google Scholar] [CrossRef] [Green Version]
- Beinotti, F.; Correia, N.; Christofoletti, G.; Borges, G. Use of hippotherapy in gait training for hemiparetic post-stroke. Arq. Neuropsiquiatr. 2010, 68, 908–913. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Beinotti, F.; Christofoletti, G.; Correia, N.; Borges, G. Effects of horseback riding therapy on quality of life in patients post stroke. Top Stroke Rehabil. 2013, 20, 226–232. [Google Scholar] [CrossRef] [PubMed]
- Sung, Y.H.; Kim, C.J.; Yu, B.K.; Kim, K.M. A hippotherapy simulator is effective to shift weight bearing toward the affected side during gait in patients with stroke. NeuroRehabilitation 2013, 33, 407–412. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.N.; Lee, D.K. Effects of horse-riding exercise on balance, gait, and activities of daily living in stroke patients. J. Phys. Ther. Sci. 2015, 27, 607–609. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gaviria, M.; Celeghin, A.; Michael-Titus, A.T.; Pallier, P.N. Editorial: Brain Plasticity and Contribution of the Emotional Brain to Neural Remodelling After Injury. Front. Neurol. 2020, 11, 606271. [Google Scholar] [CrossRef] [PubMed]
- Bromberg, P.M. Standing in the Spaces: Essays on Clinical Process, Trauma, and Dissociation; Analytic Press: Hillsdale, NJ, USA, 1998; p. 376. ISBN 9780881633566. [Google Scholar]
- Chumney, D.; Nollinger, K.; Shesko, K.; Skop, K.; Spencer, M.; Newton, R.A. Ability of Functional Independence Measure to accurately predict functional outcome of stroke-specific population: Systematic review. J. Rehabil. Res. Dev. 2010, 47, 17–29. [Google Scholar] [CrossRef]
- Bonita, R.; Beaglehole, R. Recovery of motor function after stroke. Stroke 1988, 19, 1497–1500. [Google Scholar] [CrossRef] [Green Version]
- Kasner, S.E. Clinical interpretation and use of stroke scales. Lancet Neurol. 2006, 5, 603–612. [Google Scholar] [CrossRef]
- Fugl-Meyer, A.R.; Jääskö, L.; Leyman, I.; Olsson, S.; Steglind, S. The post-stroke hemiplegic patient. 1. a method for evaluation of physical performance. Scand. J. Rehabil. Med. 1975, 7, 13–31. [Google Scholar]
- Berg, K.O.; Wood-Dauphinee, S.L.; Williams, J.I.; Maki, B. Measuring balance in the elderly: Validation of an instrument. Can. J. Public Health 1992, 83 (Suppl. 2), S7–S11. [Google Scholar] [PubMed]
- Blum, L.; Korner-Bitensky, N. Usefulness of the Berg Balance Scale in stroke rehabilitation: A systematic review. Phys. Ther. 2008, 88, 559–566. [Google Scholar] [CrossRef] [PubMed]
- Hiengkaew, V.; Jitaree, K.; Chaiyawat, P. Minimal detectable changes of the Berg Balance Scale, Fugl-Meyer Assessment Scale, Timed “Up & Go” Test, gait speeds, and 2-minute walk test in individuals with chronic stroke with different degrees of ankle plantarflexor tone. Arch. Phys. Med. Rehabil. 2012, 93, 1201–1208. [Google Scholar] [CrossRef] [PubMed]
- Ware, J., Jr.; Kosinski, M.; Keller, S.D. A 12-Item Short-Form Health Survey: Construction of scales and preliminary tests of reliability and validity. Med. Care 1996, 34, 220–233. [Google Scholar] [CrossRef] [Green Version]
- Zarit, S.H.; Reever, K.E.; Bach-Peterson, J. Relatives of the impaired elderly: Correlates of feelings of burden. Gerontologist 1980, 20, 649–655. [Google Scholar] [CrossRef] [Green Version]
- Caro, C.C.; Costa, J.D.; Da Cruz, D.M.C. Burden and Quality of Life of Family Caregivers of Stroke Patients. Occup. Ther. Health Care 2018, 32, 154–171. [Google Scholar] [CrossRef]
- Rode, G.; Lacour, S.; Jacquin-Courtois, S.; Pisella, L.; Michel, C.; Revol, P.; Alahyane, N.; Luauté, J.; Gallagher, S.; Halligan, P.; et al. Long-term sensorimotor and therapeutical effects of a mild regime of prism adaptation in spatial neglect. A double-blind RCT essay. Ann. Phys. Rehabil. Med. 2015, 58, 40–53. [Google Scholar] [CrossRef]
- Alisar, D.C.; Ozen, S.; Sozay, S. Effects of Bihemispheric Transcranial Direct Current Stimulation on Upper Extremity Function in Stroke Patients: A randomized Double-Blind Sham-Controlled Study. J. Stroke Cerebrovasc. Dis. 2020, 29, 104454. [Google Scholar] [CrossRef]
- Pitron, V.; Alsmith, A.; De Vignemont, F. How do the body schema and the body image interact? Conscious Cogn. 2018, 65, 352–358. [Google Scholar] [CrossRef]
- Pollock, A.; Baer, G.; Campbell, P.; Choo, P.L.; Forster, A.; Morris, J.; Pomeroy, V.M.; Langhorne, P. Physical rehabilitation approaches for the recovery of function and mobility following stroke. Cochrane Database Syst. Rev. 2014, 2014, CD001920. [Google Scholar] [CrossRef] [Green Version]
- Slade, A.; Tennant, A.; Chamberlain, M.A. A randomised controlled trial to determine the effect of intensity of therapy upon length of stay in a neurological rehabilitation setting. J. Rehabil. Med. 2002, 34, 260–266. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Königs, M.; Beurskens, E.A.; Snoep, L.; Scherder, E.J.; Oosterlaan, J. Effects of Timing and Intensity of Neurorehabilitation on Functional Outcome After Traumatic Brain Injury: A Systematic Review and Meta-Analysis. Arch. Phys. Med. Rehabil. 2018, 99, 1149–1159.e1. [Google Scholar] [CrossRef] [PubMed]
- Frasca, D.; Tomaszczyk, J.; McFadyen, B.J.; Green, R.E. Traumatic brain injury and post-acute decline: What role does environmental enrichment play? A scoping review. Front. Hum. Neurosci. 2013, 7, 31. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Khan, F.; Amatya, B.; Galea, M.P.; Gonzenbach, R.; Kesselring, J. Neurorehabilitation: Applied neuroplasticity. J. Neurol. 2017, 264, 603–615. [Google Scholar] [CrossRef] [PubMed]
- Viruega, H.; Gaviria, M. Functional Weight of Somatic and Cognitive Networks and Asymmetry of Compensatory Mechanisms: Collaboration or Divergency among Hemispheres after Cerebrovascular Accident? Life 2021, 11, 495. [Google Scholar] [CrossRef]
- Diaz Heijtz, R.; Kolb, B.; Forssberg, H. Can a therapeutic dose of amphetamine during pre-adolescence modify the pattern of synaptic organization in the brain? Eur. J. Neurosci. 2003, 18, 3394–3399. [Google Scholar] [CrossRef]
- Horn, G. Pathways of the past: The imprint of memory. Nat. Rev. Neurosci. 2004, 5, 108–120. [Google Scholar] [CrossRef]
- Kolb, B.; Muhammad, A. Harnessing the power of neuroplasticity for intervention. Front. Hum. Neurosci. 2014, 8, 377. [Google Scholar] [CrossRef] [Green Version]
- Lee, C.W.; Kim, S.G.; Yong, M.S. Effects of hippotherapy on recovery of gait and balance ability in patients with stroke. J. Phys. Ther. Sci. 2014, 26, 309–311. [Google Scholar] [CrossRef] [Green Version]
- Koroleva, E.S.; Tolmachev, I.V.; Alifirova, V.M.; Boiko, A.S.; Levchuk, L.A.; Loonen, A.J.M.; Ivanova, S.A. Serum BDNF’s Role as a Biomarker for Motor Training in the Context of AR-Based Rehabilitation after Ischemic Stroke. Brain Sci. 2020, 10, 623. [Google Scholar] [CrossRef]
- Delgado, M.; Ganea, D. Anti-inflammatory neuropeptides: A new class of endogenous immunoregulatory agents. Brain Behav. Immun. 2008, 22, 1146–1151. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Janura, M.; Peham, C.; Dvorakova, T.; Elfmark, M. An assessment of the pressure distribution exerted by a rider on the back of a horse during hippotherapy. Hum. Mov. Sci. 2009, 28, 387–393. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Witte, K.; Schobesberger, H.; Peham, C. Motion pattern analysis of gait in horseback riding by means of Principal Component Analysis. Hum. Mov. Sci. 2009, 28, 394–405. [Google Scholar] [CrossRef] [Green Version]
- Mayo, N.E.; Anderson, S.; Barclay, R.; Cameron, J.I.; Desrosiers, J.; Eng, J.J.; Huijbregts, M.; Kagan, A.; MacKay-Lyons, M.; Moriello, C.; et al. Getting on with the rest of your life following stroke: A randomized trial of a complex intervention aimed at enhancing life participation post stroke. Clin. Rehabil. 2015, 29, 1198–1211. [Google Scholar] [CrossRef]
- Duncan, F.; Kutlubaev, M.A.; Dennis, M.S.; Greig, C.; Mead, G.E. Fatigue after stroke: A systematic review of associations with impaired physical fitness. Int. J. Stroke 2012, 7, 157–162. [Google Scholar] [CrossRef]
- Gebruers, N.; Vanroy, C.; Truijen, S.; Engelborghs, S.; De Deyn, P.P. Monitoring of physical activity after stroke: A systematic review of accelerometry-based measures. Arch. Phys. Med. Rehabil. 2010, 91, 288–297. [Google Scholar] [CrossRef]
- Lincoln, N.B.; Brinkmann, N.; Cunningham, S.; Dejaeger, E.; De Weerdt, W.; Jenni, W.; Mahdzir, A.; Putman, K.; Schupp, W.; Schuback, B.; et al. Anxiety and depression after stroke: A 5 year follow-up. Disabil. Rehabil. 2013, 35, 140–145. [Google Scholar] [CrossRef]
- Gadidi, V.; Katz-Leurer, M.; Carmeli, E.; Bornstein, N.M. Long-term outcome poststroke: Predictors of activity limitation and participation restriction. Arch. Phys. Med. Rehabil. 2011, 92, 1802–1808. [Google Scholar] [CrossRef]
- Chevreul, K.; Durand-Zaleski, I.; Gouépo, A.; Fery-Lemonnier, E.; Hommel, M.; Woimant, F. Cost of stroke in France. Eur. J. Neurol. 2013, 20, 1094–1100. [Google Scholar] [CrossRef]
- Schmidt, A.; Heroum, C.; Caumette, D.; Le Lay, K.; Bénard, S. Acute Ischemic Stroke (AIS) patient management in French stroke units and impact estimation of thrombolysis on care pathways and associated costs. Cerebrovasc. Dis. 2015, 39, 94–101. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Viruega, H.; Imbernon, C.; Chausson, N.; Altarcha, T.; Aghasaryan, M.; Soumah, D.; Lescieux, E.; Flamand-Roze, C.; Simon, O.; Bedin, A.; et al. Neurorehabilitation through Hippotherapy on Neurofunctional Sequels of Stroke: Effect on Patients’ Functional Independence, Sensorimotor/Cognitive Capacities and Quality of Life, and the Quality of Life of Their Caregivers—A Study Protocol. Brain Sci. 2022, 12, 619. https://doi.org/10.3390/brainsci12050619
Viruega H, Imbernon C, Chausson N, Altarcha T, Aghasaryan M, Soumah D, Lescieux E, Flamand-Roze C, Simon O, Bedin A, et al. Neurorehabilitation through Hippotherapy on Neurofunctional Sequels of Stroke: Effect on Patients’ Functional Independence, Sensorimotor/Cognitive Capacities and Quality of Life, and the Quality of Life of Their Caregivers—A Study Protocol. Brain Sciences. 2022; 12(5):619. https://doi.org/10.3390/brainsci12050619
Chicago/Turabian StyleViruega, Hélène, Carole Imbernon, Nicolas Chausson, Tony Altarcha, Manvel Aghasaryan, Djibril Soumah, Edwige Lescieux, Constance Flamand-Roze, Olivier Simon, Arnaud Bedin, and et al. 2022. "Neurorehabilitation through Hippotherapy on Neurofunctional Sequels of Stroke: Effect on Patients’ Functional Independence, Sensorimotor/Cognitive Capacities and Quality of Life, and the Quality of Life of Their Caregivers—A Study Protocol" Brain Sciences 12, no. 5: 619. https://doi.org/10.3390/brainsci12050619
APA StyleViruega, H., Imbernon, C., Chausson, N., Altarcha, T., Aghasaryan, M., Soumah, D., Lescieux, E., Flamand-Roze, C., Simon, O., Bedin, A., Smadja, D., & Gaviria, M. (2022). Neurorehabilitation through Hippotherapy on Neurofunctional Sequels of Stroke: Effect on Patients’ Functional Independence, Sensorimotor/Cognitive Capacities and Quality of Life, and the Quality of Life of Their Caregivers—A Study Protocol. Brain Sciences, 12(5), 619. https://doi.org/10.3390/brainsci12050619






