Proteomic Profile of Saliva in Parkinson’s Disease Patients: A Proof of Concept Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Group Characteristics and Material Collection
2.2. Analytical Methods
2.2.1. Sample Preparation for Mass Spectrometry Analysis
2.2.2. Mass Spectrometry
2.2.3. Data Analysis
2.2.4. Quantitative MS Data Processing
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Emamzadeh, F.N.; Surguchov, A. Parkinson’s disease: Biomarkers, treatment, and risk factors. Front. Neurosci. 2018, 12, 612. [Google Scholar] [CrossRef]
- Rizzo, G.; Copetti, M.; Arcuti, S.; Martino, D.; Fontana, A.; Logroscino, G. Accuracy of clinical diagnosis of Parkinson disease: A systematic review and meta-analysis. Neurology 2016, 86, 566–576. [Google Scholar] [CrossRef] [PubMed]
- Goldman, J.G.; Andrews, H.; Amara, A.; Naito, A.; Alcalay, R.N.; Shaw, L.M.; Taylor, P.; Xie, T.; Tuite, P.; Henchcliffe, C. Cerebrospinal fluid, plasma, and saliva in the BioFIND study: Relationships among biomarkers and Parkinson’s disease features. Mov. Disord. 2018, 33, 282–288. [Google Scholar] [CrossRef]
- Chahine, L.M.; Beach, T.G.; Seedorff, N.; Caspell-Garcia, C.; Coffey, C.S.; Brumm, M.; Adler, C.H.; Serrano, G.E.; Linder, C.; Mosovsky, S. Feasibility and Safety of Multicenter Tissue and Biofluid Sampling for α-Synuclein in Parkinson’s Disease: The Systemic Synuclein Sampling Study (S4). J. Parkinson’s Dis. 2018, 8, 517–527. [Google Scholar] [CrossRef]
- Gelpi, E.; Navarro-Otano, J.; Tolosa, E.; Gaig, C.; Compta, Y.; Rey, M.J.; Martí, M.J.; Hernández, I.; Valldeoriola, F.; Reñé, R. Multiple organ involvement by alpha-synuclein pathology in Lewy body disorders. Mov. Disord. 2014, 29, 1010–1018. [Google Scholar] [CrossRef] [PubMed]
- Del Tredici, K.; Hawkes, C.H.; Ghebremedhin, E.; Braak, H. Lewy pathology in the submandibular gland of individuals with incidental Lewy body disease and sporadic Parkinson’s disease. Acta Neuropathol. 2010, 119, 703–713. [Google Scholar] [CrossRef]
- Hawkes, C.H.; Del Tredici, K.; Braak, H. Parkinson’s disease: A dual-hit hypothesis. Neuropathol. Appl. Neurobiol. 2007, 33, 599–614. [Google Scholar] [CrossRef] [PubMed]
- Savica, R.; Boeve, B.F.; Mielke, M.M. When do α-synucleinopathies start? An epidemiological timeline: A review. JAMA Neurol. 2018, 75, 503–509. [Google Scholar] [CrossRef] [PubMed]
- Paciotti, S.; Bellomo, G.; Gatticchi, L.; Parnetti, L. Are we ready for detecting α-synuclein prone to aggregation in patients? The case of “protein-misfolding cyclic amplification” and “real-time quaking-induced conversion” as diagnostic tools. Front. Neurol. 2018, 9, 415. [Google Scholar] [CrossRef]
- Masters, J.M.; Noyce, A.J.; Warner, T.T.; Giovannoni, G.; Proctor, G.B. Elevated salivary protein in Parkinson’s disease and salivary DJ-1 as a potential marker of disease severity. Parkinsonism Relat. Disord. 2015, 21, 1251–1255. [Google Scholar] [CrossRef]
- Cersosimo, M.G.; Raina, G.B.; Calandra, C.R.; Pellene, A.; Gutiérrez, C.; Micheli, F.E.; Benarroch, E.E. Dry mouth: An overlooked autonomic symptom of Parkinson’s disease. J. Parkinson’s Dis. 2011, 1, 169–173. [Google Scholar] [CrossRef] [PubMed]
- Figura, M.; Friedman, A. In search of Parkinson’s disease biomarkers—is the answer in our mouths? A systematic review of the literature on salivary biomarkers of Parkinson’s disease. Neurol. I Neurochir. Pol. 2020, 54, 14–20. [Google Scholar] [CrossRef] [PubMed]
- Vivacqua, G.; Latorre, A.; Suppa, A.; Nardi, M.; Pietracupa, S.; Mancinelli, R.; Fabbrini, G.; Colosimo, C.; Gaudio, E.; Berardelli, A. Abnormal salivary total and oligomeric alpha-synuclein in Parkinson’s disease. PLoS ONE 2016, 11, e0151156. [Google Scholar]
- Kang, W.; Chen, W.; Yang, Q.; Zhang, L.; Zhang, L.; Wang, X.; Dong, F.; Zhao, Y.; Chen, S.; Quinn, T.J. Salivary total α-synuclein, oligomeric α-synuclein and SNCA variants in Parkinson’s disease patients. Sci. Rep. 2016, 6, 28143. [Google Scholar] [CrossRef] [PubMed]
- Plotegher, N.; Gratton, E.; Bubacco, L. Number and Brightness analysis of alpha-synuclein oligomerization and the associated mitochondrial morphology alterations in live cells. Biochim. Biophys. Acta 2014, 1840, 2014–2024. [Google Scholar] [CrossRef] [PubMed]
- Sharon, R.; Bar-Joseph, I.; Frosch, M.P.; Walsh, D.M.; Hamilton, J.A.; Selkoe, D.J. The formation of highly soluble oligomers of alpha-synuclein is regulated by fatty acids and enhanced in Parkinson’s disease. Neuron 2003, 37, 583–595. [Google Scholar] [CrossRef]
- Bengoa-Vergniory, N.; Roberts, R.F.; Wade-Martins, R.; Alegre-Abarrategui, J. Alpha-synuclein oligomers: A new hope. Acta Neuropathol 2017, 134, 819–838. [Google Scholar] [CrossRef] [PubMed]
- Khurshid, Z.; Moin, S.F.; Khan, R.S.; Agwan, M.A.S.; Alwadaani, A.H.; Zafar, M.S. Human salivary protein extraction from RNAPro· SAL™, Pure· SAL™, and passive drooling method. Eur. J. Dent. 2017, 11, 385–389. [Google Scholar]
- Iorgulescu, G. Saliva between normal and pathological. Important factors in determining systemic and oral health. J. Med. Life 2009, 2, 303. [Google Scholar]
- Beach, T.G.; Adler, C.H.; Serrano, G.; Sue, L.I.; Walker, D.; Dugger, B.N.; Shill, H.A.; Driver-Dunckley, E.; Caviness, J.N.; Intorcia, A. Prevalence of submandibular gland synucleinopathy in Parkinson’s disease, dementia with Lewy bodies and other Lewy body disorders. J. Parkinson’s Dis. 2016, 6, 153–163. [Google Scholar] [CrossRef] [PubMed]
- Beach, T.G.; Adler, C.H.; Dugger, B.N.; Serrano, G.; Hidalgo, J.; Henry-Watson, J.; Shill, H.A.; Sue, L.I.; Sabbagh, M.N.; Akiyama, H. Submandibular gland biopsy for the diagnosis of Parkinson disease. J. Neuropathol. Exp. Neurol. 2013, 72, 130–136. [Google Scholar] [CrossRef]
- Adler, C.H.; Dugger, B.N.; Hentz, J.G.; Hinni, M.L.; Lott, D.G.; Driver-Dunckley, E.; Mehta, S.; Serrano, G.; Sue, L.I.; Duffy, A. Peripheral Synucleinopathy in Early Parkinson’s Disease: Submandibular Gland Needle Biopsy Findings. Mov. Disord. 2016, 31, 250–256. [Google Scholar] [CrossRef]
- Adler, C.H.; Dugger, B.N.; Hinni, M.L.; Lott, D.G.; Driver-Dunckley, E.; Hidalgo, J.; Henry-Watson, J.; Serrano, G.; Sue, L.I.; Nagel, T. Submandibular gland needle biopsy for the diagnosis of Parkinson disease. Neurology 2014, 82, 858–864. [Google Scholar] [CrossRef]
- Castagnola, M.; Scarano, E.; Passali, G.C.; Messana, I.; Cabras, T.; Iavarone, F.; Di Cintio, G.; Fiorita, A.; De Corso, E.; Paludetti, G. Salivary biomarkers and proteomics: Future diagnostic and clinical utilities. Acta Otorhinolaryngol. Ital. 2017, 37, 94–101. [Google Scholar] [CrossRef]
- Pappa, E.; Kousvelari, E.; Vastardis, H. Saliva in the “Omics” era: A promising tool in paediatrics. Oral Dis 2019, 25, 16–25. [Google Scholar] [CrossRef]
- Batista, T.B.D.; Chaiben, C.L.; Penteado, C.A.S.; Nascimento, J.M.C.; Ventura, T.M.O.; Dionizio, A.; Rosa, E.A.R.; Buzalaf, M.A.R.; Azevedo-Alanis, L.R. Salivary proteome characterization of alcohol and tobacco dependents. Drug Alcohol. Depend. 2019, 204, 107510. [Google Scholar] [CrossRef]
- Tierney, C.; Bazou, D.; Lê, G.; Dowling, P.; O’Gorman, P. Saliva-omics in plasma cell disorders- Proof of concept and potential as a non-invasive tool for monitoring disease burden. J. Proteom. 2021, 231, 104015. [Google Scholar] [CrossRef]
- Chiang, S.H.; Thomas, G.A.; Liao, W.; Grogan, T.; Buck, R.L.; Fuentes, L.; Yakob, M.; Laughlin, M.J.; Schafer, C.; Nazmul-Hossain, A. RNAPro• SAL: A device for rapid and standardized collection of saliva RNA and proteins. Biotechniques 2015, 58, 69–76. [Google Scholar] [CrossRef]
- Malinowska, A.; Kistowski, M.; Bakun, M.; Rubel, T.; Tkaczyk, M.; Mierzejewska, J.; Dadlez, M. Diffprot—software for non-parametric statistical analysis of differential proteomics data. J. Proteom. 2012, 75, 4062–4073. [Google Scholar] [CrossRef]
- Elias, J.E.; Gygi, S.P. Target-decoy search strategy for increased confidence in large-scale protein identifications by mass spectrometry. Nat. Methods 2007, 4, 207–214. [Google Scholar] [CrossRef]
- Elias, J.E.; Gygi, S.P. Target-decoy search strategy for mass spectrometry-based proteomics. In Proteome Bioinformatics; Springer: Berlin/Heidelberg, Germany, 2010; pp. 55–71. [Google Scholar]
- Rubel, T. Available online: http://proteom.ibb.waw.pl/mscan/ (accessed on 14 May 2021).
- Bakun, M.; Karczmarski, J.; Poznanski, J.; Rubel, T.; Rozga, M.; Malinowska, A.; Sands, D.; Hennig, E.; Oledzki, J.; Ostrowski, J. An integrated LC-ESI-MS platform for quantitation of serum peptide ladders. Application for colon carcinoma study. Proteom. Clin. Appl. 2009, 3, 932–946. [Google Scholar] [CrossRef]
- Youden, W.J. Index for rating diagnostic tests. Cancer 1950, 3, 32–35. [Google Scholar] [CrossRef]
- Kumari, S.; Goyal, V.; Kumaran, S.S.; Dwivedi, S.; Srivastava, A.; Jagannathan, N. Quantitative metabolomics of saliva using proton nmr spectroscopy in patients with parkinson’s disease and healthy controls. Neurol. Sci. 2020, 41, 1–10. [Google Scholar] [CrossRef]
- Masters, J.; Noyce, A.; Lynham, S.; Warner, T.; Giovannoni, G.; Proctor, G. A comprehensive characterisation of the salivary proteome of patients with Parkinson’s disease. In Proceedings of the 20th International Congress of Parkinson’s Disease and Movement Disorders, Berlin, Germany, 19–23 June 2016. [Google Scholar]
- Serrano, A.; Apolloni, S.; Rossi, S.; Lattante, S.; Sabatelli, M.; Peric, M.; Andjus, P.; Michetti, F.; Carrì, M.T.; Cozzolino, M. The S100A4 transcriptional inhibitor niclosamide reduces pro-inflammatory and migratory phenotypes of microglia: Implications for amyotrophic lateral sclerosis. Cells 2019, 8, 1261. [Google Scholar] [CrossRef] [PubMed]
- Qiu, X.; Ping, S.; Kyle, M.; Longo, J.; Chin, L.; Zhao, L.-R. S100 Calcium-Binding Protein A9 Knockout Contributes to Neuroprotection and Functional Improvement after Traumatic Brain Injury. J. Neurotrauma 2019, 37, 950–965. [Google Scholar] [CrossRef]
- Kaca-Oryńska, M.; Tomasiuk, R.; Friedman, A. Neuron-specific enolase and S 100B protein as predictors of outcome in ischaemic stroke. Neurol. I Neurochir. Pol. 2010, 44, 459–463. [Google Scholar] [CrossRef]
- Fardell, C.; Zettergren, A.; Ran, C.; Carmine Belin, A.; Ekman, A.; Sydow, O.; Bäckman, L.; Holmberg, B.; Dizdar, N.; Söderkvist, P. S100B polymorphisms are associated with age of onset of Parkinson’s disease. BMC Med. Genet. 2018, 19. [Google Scholar] [CrossRef]
- Papuć, E.; Rejdak, K. Increased Cerebrospinal Fluid S100B and NSE Reflect Neuronal and Glial Damage in Parkinson’s Disease. Front. Aging Neurosci. 2020, 12, 156. [Google Scholar] [CrossRef]
- Marenholz, I.; Heizmann, C.W. S100A16, a ubiquitously expressed EF-hand protein which is up-regulated in tumors. Biochem. Biophys. Res. Commun. 2004, 313, 237–244. [Google Scholar] [CrossRef]
- Liu, Y.; Zhang, R.; Xin, J.; Sun, Y.; Li, J.; Wei, D.; Zhao, A.Z. Identification of S100A16 as a novel adipogenesis promoting factor in 3T3-L1 cells. Endocrinology 2011, 152, 903–911. [Google Scholar] [CrossRef]
- Zhang, R.; Kan, J.B.; Lu, S.; Tong, P.; Yang, J.; Xi, L.; Liang, X.; Su, D.; Li, D.; Liu, Y. S100A16-induced adipogenesis is associated with up-regulation of 11 β-hydroxysteroid dehydrogenase type 1 (11β-HSD1). Biosci. Rep. 2019, 39. [Google Scholar] [CrossRef] [PubMed]
- Sheard, J.M.; Ash, S.; Mellick, G.D.; Silburn, P.A.; Kerr, G.K. Malnutrition in a sample of community-dwelling people with Parkinson’s disease. PLoS ONE 2013, 8, e53290. [Google Scholar]
- Sheard, J.M.; Ash, S.; Silburn, P.A.; Kerr, G.K. Prevalence of malnutrition in Parkinson’s disease: A systematic review. Nutr. Rev. 2011, 69, 520–532. [Google Scholar] [CrossRef] [PubMed]
- Schrank, B.R.; Aparicio, T.; Li, Y.; Chang, W.; Chait, B.T.; Gundersen, G.G.; Gottesman, M.E.; Gautier, J. Nuclear ARP2/3 drives DNA break clustering for homology-directed repair. Nature 2018, 559, 61–66. [Google Scholar] [CrossRef] [PubMed]
- Vasquez, V.; Mitra, J.; Hegde, P.M.; Pandey, A.; Sengupta, S.; Mitra, S.; Rao, K.; Hegde, M.L. Chromatin-bound oxidized α-synuclein causes strand breaks in neuronal genomes in in vitro models of Parkinson’s disease. J. Alzheimer’s Dis. 2017, 60, S133–S150. [Google Scholar] [CrossRef]
- Gonzalez-Hunt, C.P.; Sanders, L.H. DNA damage and repair in Parkinson’s disease: Recent advances and new opportunities. J. Neurosci. Res. 2020, 99, 180–189. [Google Scholar] [CrossRef]
- Baietti, M.F.; Zhang, Z.; Mortier, E.; Melchior, A.; Degeest, G.; Geeraerts, A.; Ivarsson, Y.; Depoortere, F.; Coomans, C.; Vermeiren, E. Syndecan–syntenin–ALIX regulates the biogenesis of exosomes. Nat. Cell Biol. 2012, 14, 677–685. [Google Scholar] [CrossRef]
- Grey, M.; Dunning, C.J.; Gaspar, R.; Grey, C.; Brundin, P.; Sparr, E.; Linse, S. Acceleration of α-synuclein aggregation by exosomes. J. Biol. Chem. 2015, 290, 2969–2982. [Google Scholar] [CrossRef]
- Yu, H.; Sun, T.; An, J.; Wen, L.; Liu, F.; Bu, Z.; Cui, Y.; Feng, J. Potential Roles of Exosomes in Parkinson’s Disease: From Pathogenesis, Diagnosis, and Treatment to Prognosis. Front. Cell Dev. Biol. 2020, 8. [Google Scholar] [CrossRef]
- Rani, K.; Mukherjee, R.; Singh, E.; Kumar, S.; Sharma, V.; Vishwakarma, P.; Bharti, P.S.; Nikolajeff, F.; Dinda, A.K.; Goyal, V. Neuronal exosomes in saliva of Parkinson’s disease patients: A pilot study. Parkinsonism Relat. Disord. 2019, 67, 21–23. [Google Scholar] [CrossRef]
- Fung, H.-C.; Scholz, S.; Matarin, M.; Simón-Sánchez, J.; Hernandez, D.; Britton, A.; Gibbs, J.R.; Langefeld, C.; Stiegert, M.L.; Schymick, J. Genome-wide genotyping in Parkinson’s disease and neurologically normal controls: First stage analysis and public release of data. Lancet Neurol. 2006, 5, 911–916. [Google Scholar] [CrossRef]
- Nalls, M.A.; Blauwendraat, C.; Vallerga, C.L.; Heilbron, K.; Bandres-Ciga, S.; Chang, D.; Tan, M.; Kia, D.A.; Noyce, A.J.; Xue, A. Identification of novel risk loci, causal insights, and heritable risk for Parkinson’s disease: A meta-analysis of genome-wide association studies. Lancet Neurol. 2019, 18, 1091–1102. [Google Scholar] [CrossRef]
- Nalls, M.A.; Pankratz, N.; Lill, C.M.; Do, C.B.; Hernandez, D.G.; Saad, M.; DeStefano, A.L.; Kara, E.; Bras, J.; Sharma, M. Large-scale meta-analysis of genome-wide association data identifies six new risk loci for Parkinson’s disease. Nat. Genet. 2014, 46, 989–993. [Google Scholar] [CrossRef] [PubMed]
- Lesage, S.; Drouet, V.; Majounie, E.; Deramecourt, V.; Jacoupy, M.; Nicolas, A.; Cormier-Dequaire, F.; Hassoun, S.M.; Pujol, C.; Ciura, S. Loss of VPS13C function in autosomal-recessive Parkinsonism causes mitochondrial dysfunction and increases PINK1/Parkin-dependent mitophagy. Am. J. Hum. Genet. 2016, 98, 500–513. [Google Scholar] [CrossRef]
- Van Stiphout, M.A.; Marinus, J.; van Hilten, J.J.; Lobbezoo, F.; de Baat, C. Oral Health of Parkinson’s Disease Patients: A Case-Control Study. Parkinson’s Dis. 2018, 2018. [Google Scholar] [CrossRef] [PubMed]
- Youdim, M.; Grünblatt, E.; Mandel, S. The Pivotal Role of Iron in NF-κB Activation and Nigrostriatal Dopaminergic Neurodegeneration: Prospects for Neuroprotection in Parkinson’s Disease with Iron Chelators. Ann. N. Y. Acad. Sci. 1999, 890, 7–25. [Google Scholar] [CrossRef] [PubMed]
- Dehmer, T.; Heneka, M.T.; Sastre, M.; Dichgans, J.; Schulz, J.B. Protection by pioglitazone in the MPTP model of Parkinson’s disease correlates with IκBα induction and block of NFκB and iNOS activation. J. Neurochem. 2004, 88, 494–501. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.-W.; Koppula, S.; Kumar, H.; Park, J.-Y.; Kim, I.-W.; More, S.V.; Kim, I.-S.; Han, S.-D.; Kim, S.-K.; Yoon, S.-H. α-Asarone attenuates microglia-mediated neuroinflammation by inhibiting NF kappa B activation and mitigates MPTP-induced behavioral deficits in a mouse model of Parkinson’s disease. Neuropharmacology 2015, 97, 46–57. [Google Scholar] [CrossRef]
- Xu, W.; Zheng, D.; Liu, Y.; Li, J.; Yang, L.; Shang, X. Glaucocalyxin B alleviates lipopolysaccharide-induced Parkinson’s disease by inhibiting TLR/NF-κB and activating Nrf2/HO-1 pathway. Cell. Physiol. Biochem. 2017, 44, 2091–2104. [Google Scholar] [CrossRef]
- Soós, J.; Engelhardt, J.I.; Siklós, L.; Havas, L.; Majtényi, K. The expression of PARP, NF-κB and parvalbumin is increased in Parkinson disease. Neuroreport 2004, 15, 1715–1718. [Google Scholar] [CrossRef]
- Chen, G.; Nie, S.; Han, C.; Ma, K.; Xu, Y.; Zhang, Z.; Papa, S.; Cao, X. Antidyskinetic effects of MEK inhibitor are associated with multiple neurochemical alterations in the striatum of hemiparkinsonian rats. Front Neurosci 2017, 11, 112. [Google Scholar] [CrossRef]
- Chen, W.-F.; Wu, L.; Du, Z.-R.; Chen, L.; Xu, A.-L.; Chen, X.-H.; Teng, J.-J.; Wong, M.-S. Neuroprotective properties of icariin in MPTP-induced mouse model of Parkinson’s disease: Involvement of PI3K/Akt and MEK/ERK signaling pathways. Phytomedicine 2017, 25, 93–99. [Google Scholar] [CrossRef]
- Lavin, P.J.; Gawel, M.J.; Das, P.K.; Alaghband-Zadeh, J.; Rose, F.C. Effect of Levodopa on Thyroid Function and Prolactin Release: A Study in Patients With Parkinson’s Disease. Arch. Neurol. 1981, 38, 759–760. [Google Scholar] [CrossRef] [PubMed]
- Eisler, T.; Thorner, M.; MacLeod, R.; Kaiser, D.; Calne, D. Prolactin secretion in Parkinson disease. Neurology 1981, 31, 1356. [Google Scholar] [CrossRef] [PubMed]
- Lawton, N.; MacDermot, J. Abnormal regulation of prolactin release in idiopathic Parkinson’s disease. J. Neurol. Neurosurg. Psychiatry 1980, 43, 1012–1015. [Google Scholar] [CrossRef] [PubMed]
- Tumilasci, O.R.; Cersosimo, M.; Belforte, J.E.; Micheli, F.E.; Benarroch, E.E.; Pazo, J.H. Quantitative study of salivary secretion in Parkinson’s disease. Mov. Disord. 2006, 21, 660–667. [Google Scholar] [CrossRef]
- Tiigimäe-Saar, J.; Tamme, T.; Rosenthal, M.; Kadastik-Eerme, L.; Taba, P. Saliva changes in Parkinson’s disease patients after injection of Botulinum neurotoxin type A. Neurol. Sci. 2018, 39, 871–877. [Google Scholar] [CrossRef] [PubMed]
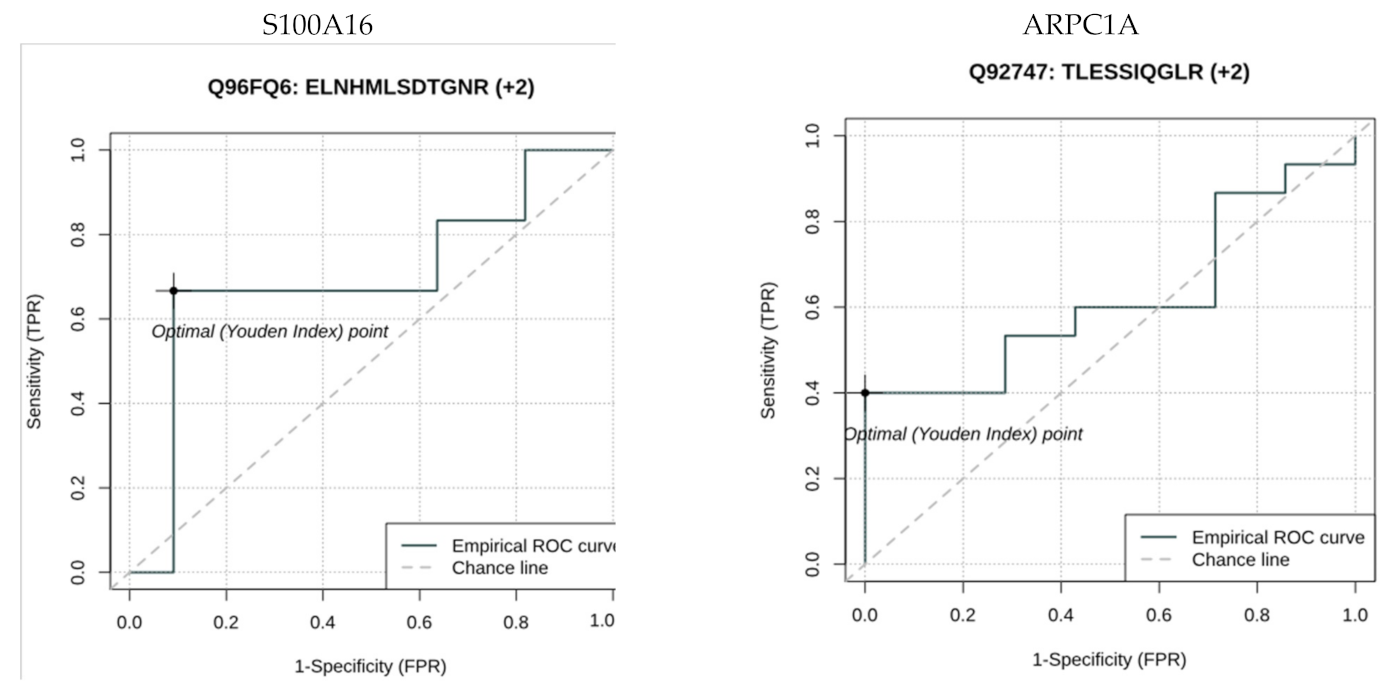
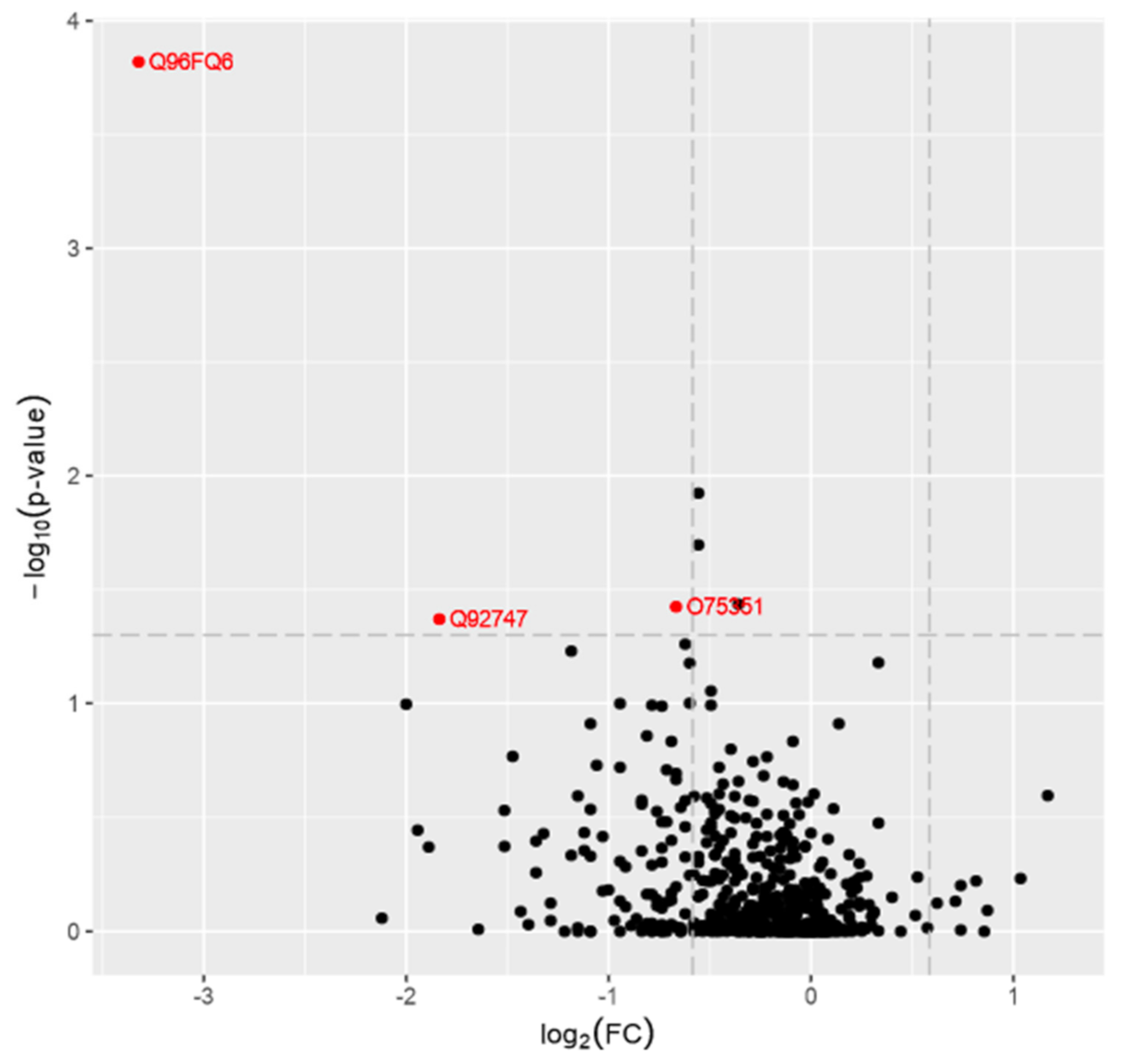
| PD Group | Healthy Control | p-Value | |
|---|---|---|---|
| Number of patients | 24 | 15 | |
| Sex M/F | 15/9 | 9/5 | ns |
| Age | 61.6 ± 8.2 | 60.9 ± 6.7 | ns |
| Levodopa dose | 1352 ± 763 | na | |
| LED | 1524 ± 786 | na | |
| UPDRS part III OFF | 34.6 ± 12.4 | na |
| Upstream Regulator | Molecule Type | Predicted Activation State | Activation z-Score | p-Value of Overlap | Target Molecules in Dataset |
|---|---|---|---|---|---|
| PD98059 | chemical - kinase inhibitor | Activated | 2.737 | 9.13 × 10−5 | ANXA1⇓,CAMP⇓, FN1⇓, GRN⇓,ITGAM⇓, KRT19⇓, NQO1⇓, RETN⇓, S100A4⇓, S100A8⇓,SOD1⇓ |
| SRF | transcription regulator | Activated | 2.646 | 2.72 × 10−4 | CAMP⇓, ELANE⇓, ITGAM⇓, LTF⇓, MYH9⇓, S100A8⇓, S100A9⇓, SLPI⇓ |
| SB203580 | chemical - kinase inhibitor | Activated | 2.596 | 8.58 × 10−5 | ANXA1⇓, ANXA3⇓, CAMP⇓, FN1⇓, HMGB1⇑, ITGAM⇓, NQO1⇓, RETN⇓, SLPI⇓, TGM1⇓ |
| RICTOR | other | Activated | 2.236 | 1.11 × 10−2 | PSMA1⇓, PSMA5⇓, PSMB1⇓, PSMB3⇓, RPSA⇓ |
| GATA3 | transcription regulator | Activated | 2.216 | 1.22 × 10−3 | FN1⇓, PPL⇓, S100A8⇓, S100A9⇓, SLPI⇓, TGM1⇓ |
| mifepristone | chemical drug | Activated | 2.121 | 1.75 × 10−4 | ANXA1⇓, CAST⇓,GSTM1⇓, HSPD1⇓, ITGAM⇓, JUP⇓, LTF⇓, PSMA1⇓ |
| tert-butyl-hydroquinone | chemical reagent | Inhibited | −2.175 | 6.90 × 10−5 | GSS⇓, ITGAM⇓, ME1⇓, NQO1⇓, PSMA5⇓ |
| arsenic trioxide | chemical drug | Inhibited | −2.176 | 1.08 × 10−3 | FN1⇓, ITGAM⇓, ME1⇓, NQO1⇓, PDIA4⇓, S100A8⇓, VCP⇓ |
| IL17A | cytokine | Inhibited | −2.196 | 8.72 × 10−3 | CAMP⇓, MPO⇓, S100A12⇓, S100A8⇓, S100A9⇓ |
| SP1 | transcription regulator | Inhibited | −2.200 | 1.91 × 10−5 | CAMP⇓, CES1⇓, ELANE⇓, FN1⇓, GSS⇓, ITGAM⇓, KRT16⇓, KRT19⇓, KRT4⇓, PADI4⇑, SOD1⇓, TGM1⇓ |
| IL2 | cytokine | Inhibited | −2.213 | 3.35 × 10−2 | ANXA1⇓,FCGR3A/FCGR3B⇑,GRN⇓, HSPD1⇓, PSMB1⇓, S100A4⇓, S100A8⇓ |
| butyric acid | chemical - endogenous mammalian | Inhibited | −2.227 | 2.81 × 10−8 | ALDH1A1⇓, ANXA1⇓, ANXA5⇓, CAMP⇓, CEACAM5⇓, ELANE⇓, GRN⇓, HMGB1⇑, ITGAM⇓, KRT13⇓, MVP⇓, PCMT1⇑, TGM1⇓, TP53I3⇓ |
| curcumin | chemical drug | Inhibited | −2.236 | 7.96 × 10−3 | CAMP⇓, HMGB1⇑, ITGAM⇓, NQO1⇓, RETN⇓, SOD1⇓ |
| OSM | cytokine | Inhibited | −2.272 | 5.08 × 10−7 | ANXA1⇓, ANXA3⇓, CAMP⇓, CDA⇓, FN1⇓, GCA⇓, KLK13⇑, KRT16⇓, KRT19⇓, LRRFIP1⇓, S100A12⇓, S100A8⇓, S100A9⇓, SLPI⇓ |
| HSF1 | transcription regulator | Inhibited | −2.360 | 6.56 × 10−4 | CCT2⇓, CCT3⇑, FKBP4⇓, HMGB1⇑, HSPD1⇓, TCP1⇓ |
| 1,2-dithiol-3-thione | chemical reagent | Inhibited | −2.503 | 6.27 × 10−8 | CCT3⇑, GSTM1⇓, NQO1⇓, PDIA4⇓, PSMA1⇓, PSMA5⇓, PSMB1⇓, PSMB3⇓, SOD1⇓, VCP⇓ |
| EGF | growth factor | Inhibited | −2.516 | 1.16 × 10−5 | FN1⇓, ITGAM⇓, KRT16⇓, KRT19⇓, KRT5⇓, LTF⇓, PFKM⇓, PSMB1⇓, PSMB3⇓, S100A4⇓, S100A9⇓, TGM1⇓ |
| tetradecanoylphorbol acetate | chemical drug | Inhibited | −2.608 | 1.43 × 10−5 | ANXA1⇓, AZU1⇓, CES1⇓, ELANE⇓, GSTM1⇓, ITGAM⇓, LRRFIP1⇓, MPO⇓, MYH9⇓, NQO1⇓, RETN⇓, S100A14⇓, S100A8⇓, S100A9⇓, SLPI⇓, SOD1⇓, STATH⇑, TGM1⇓ |
| NFkB (complex) | complex | Inhibited | −2.621 | 3.90 × 10−3 | ALDH7A1⇓, CAMP⇓, CAPNS1⇓, FN1⇓, HSPA9⇓, ITGAM⇓, KRT19⇓, PKP1⇓, SLPI⇓ |
| KLF4 | transcription regulator | Inhibited | −2.704 | 2.55 × 10−7 | ALDH1A1⇓, DSP⇓, FN1⇓, ITGAM⇓, KRT13⇓, KRT19⇓, PFKP⇓, PPL⇓, S100A14⇓, SLPI⇓, TGM1⇓ |
| CEBPA | transcription regulator | Inhibited | −2.755 | 1.11 × 10−5 | ANXA1⇓, CAMP⇓, ELANE⇓, ITGAM⇓, LTF⇓, MPO⇓, PPL⇓, RETN⇓, S100A8⇓, S100A9⇓, SOD1⇓ |
| Lipopolysaccharide | chemical drug | Inhibited | −2.951 | 9.57 × 10−9 | ANXA1⇓, ANXA3⇓, ANXA5⇓, AZU1⇓, CAMP⇓, CLIC3⇓, ELANE⇓, FN1⇓, GCA⇓, HMGB1⇑, ITGAM⇓, ITIH4⇓, KRT13⇓, KRT4⇓, LGALS3⇓, LRRFIP1⇓, LTF⇓, MPO⇓, MYH9⇓, NQO1⇓, ORM2⇓, PDIA4⇓, PFKP⇓, RETN⇓, S100A12⇓, S100A8⇓, S100A9⇓, SLPI⇓, SOD1⇓, TRIM29⇓ |
| PRL | cytokine | Inhibited | −3.071 | 1.07 × 10−6 | ANXA3⇓, ANXA5⇓, FN1⇓, GSTM1⇓, HSPD1⇓, KRT19⇓, KRT5⇓, PDIA4⇓, RPSA⇓, SOD1⇓ |
| NFE2L2 | transcription regulator | Inhibited | −3.263 | 2.58 × 10−8 | CCT3⇑, FN1⇓, GSS⇓, GSTM1⇓, HSPA9⇓, ME1⇓, NQO1⇓, PDIA4⇓, PSMA1⇓, PSMA5⇓, PSMB1⇓, PSMB3⇓, SOD1⇓, VCP⇓ |
| beta-estradiol | chemical - endogenous mammalian | Inhibited | −3.428 | 6.34 × 10−8 | ADK ⇓,ALDH7A1⇓, ANXA1⇓, ANXA3⇓, CAST⇓,CCT2⇓, DSP⇓, FN1⇓, HNRNPD⇓, HSPA9⇓, HSPD1⇓, ITIH4⇓, KRT13⇓, KRT16⇓, KRT19⇓, KRT4⇓, KRT5⇓, LGALS3⇓, LTF⇓, MPO⇓, PADI4⇑, PDIA4⇓, PPL⇓, PSMA1⇓, PSMB1⇓, S100A4⇓, S100A9⇓, SLPI⇓, TP53I3⇓, TRIM29⇓ |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Figura, M.; Sitkiewicz, E.; Świderska, B.; Milanowski, Ł.; Szlufik, S.; Koziorowski, D.; Friedman, A. Proteomic Profile of Saliva in Parkinson’s Disease Patients: A Proof of Concept Study. Brain Sci. 2021, 11, 661. https://doi.org/10.3390/brainsci11050661
Figura M, Sitkiewicz E, Świderska B, Milanowski Ł, Szlufik S, Koziorowski D, Friedman A. Proteomic Profile of Saliva in Parkinson’s Disease Patients: A Proof of Concept Study. Brain Sciences. 2021; 11(5):661. https://doi.org/10.3390/brainsci11050661
Chicago/Turabian StyleFigura, Monika, Ewa Sitkiewicz, Bianka Świderska, Łukasz Milanowski, Stanisław Szlufik, Dariusz Koziorowski, and Andrzej Friedman. 2021. "Proteomic Profile of Saliva in Parkinson’s Disease Patients: A Proof of Concept Study" Brain Sciences 11, no. 5: 661. https://doi.org/10.3390/brainsci11050661
APA StyleFigura, M., Sitkiewicz, E., Świderska, B., Milanowski, Ł., Szlufik, S., Koziorowski, D., & Friedman, A. (2021). Proteomic Profile of Saliva in Parkinson’s Disease Patients: A Proof of Concept Study. Brain Sciences, 11(5), 661. https://doi.org/10.3390/brainsci11050661







