Psychological and Physiological Signatures of Music Listening in Different Listening Environments—An Exploratory Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Experimental Procedure
2.3. Data Analyses
3. Results
3.1. Effects of Music Listening on Emotion Ratings
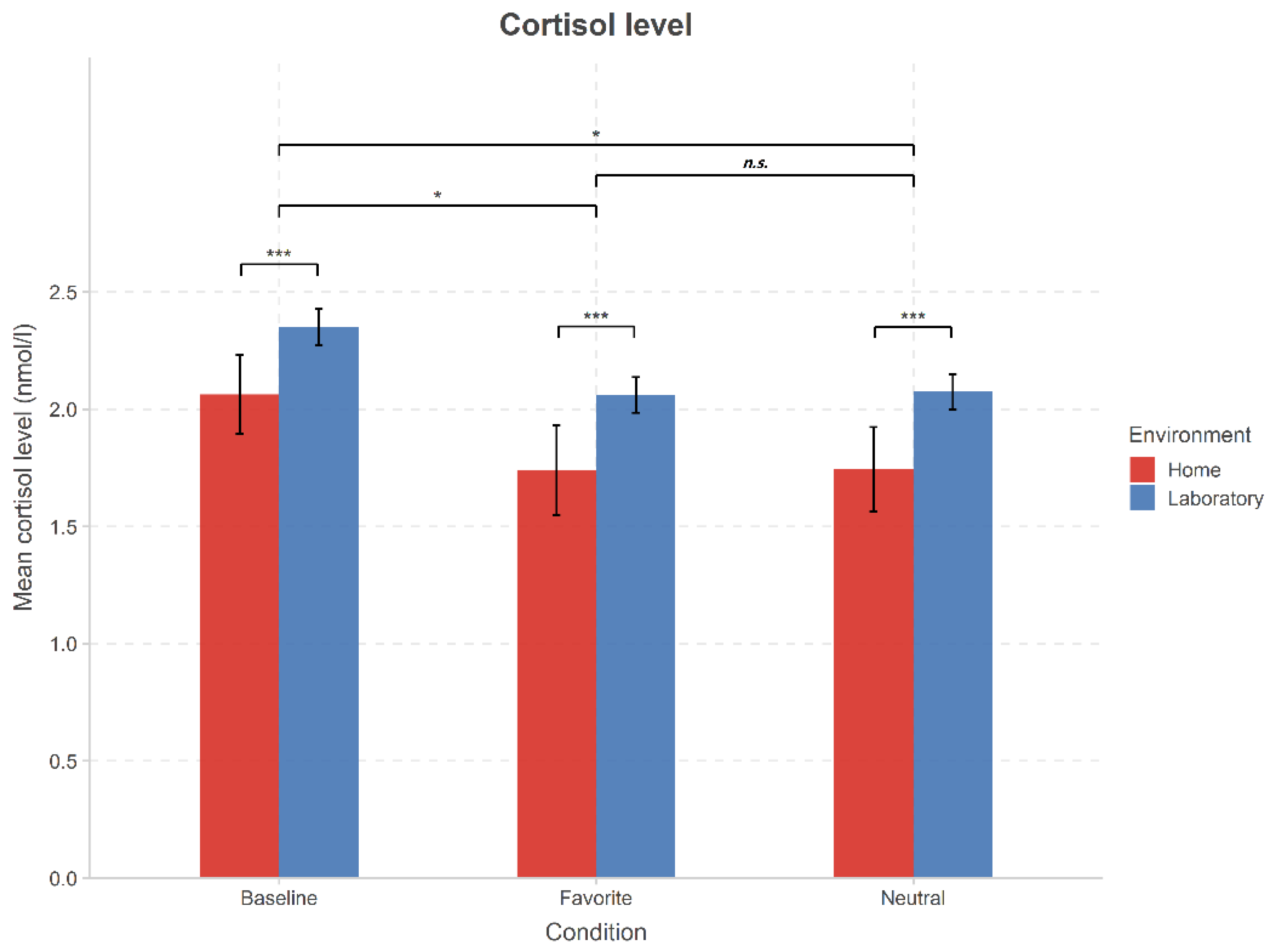
3.2. Effects of Music Listening on Cortisol Levels
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- IFPI Annual Report. 2020. Available online: https://www.ifpi.org/ifpi-issues-annual-global-music-report (accessed on 29 March 2021).
- Chanda, M.L.; Levitin, D.J. The neurochemistry of music. Trends Cogn. Sci. 2013, 17, 179–193. [Google Scholar] [CrossRef]
- Koelsch, S. A coordinate-based meta-analysis of music-evoked emotions. Neuroimage 2020, 223, 117350. [Google Scholar] [CrossRef] [PubMed]
- Carlson, E.; Saarikallio, S.; Toiviainen, P.; Bogert, B.; Kliuchko, M.; Brattico, E. Maladaptive and adaptive emotion regulation through music: A behavioral and neuroimaging study of males and females. Front. Hum. Neurosci. 2015, 9, 466. [Google Scholar] [CrossRef]
- Saarikallio, S.H.; Randall, W.M.; Baltazar, M. Music listening for supporting adolescents’ sense of agency in daily life. Front. Psychol. 2020, 10, 2911. [Google Scholar] [CrossRef] [PubMed]
- Scherer, K.R.; Zentner, M.R.; Schacht, A. Emotional states generated by music: An exploratory study of music experts. Music Sci. 2001, 5, 149–171. [Google Scholar] [CrossRef]
- Eerola, T.; Vuoskoski, J.K. A comparison of the discrete and dimensional models of emotion in music. Psychol. Music 2011, 39, 18–49. [Google Scholar] [CrossRef]
- Blood, A.J.; Zatorre, R.J. Intensely pleasurable responses to music correlate with activity in brain regions implicated in reward and emotion. Proc. Natl. Acad. Sci. USA 2001, 98, 11818–11823. [Google Scholar] [CrossRef]
- Salimpoor, V.; Benovoy, M.; Larcher, K.; Dagher, A.; Zatorre, R.T. Anatomically distinct dopamine release during anticipation and experience of peak emotion to music. Nat. Neurosci. 2011, 14, 257–262. [Google Scholar] [CrossRef]
- Laeng, B.; Eidet, L.M.; Sulutvedt, U.; Panksepp, J. Music chills: The eye pupil as a mirror to music’s soul. Conscious. Cogn. 2016, 44, 161–178. [Google Scholar] [CrossRef]
- Chabin, T.; Gabriel, D.; Chansophonkul, T.; Michelant, L.; Joucla, C.; Haffen, E.; Moulin, T.; Comte, A.; Pazart, L. Cortical patterns of pleasurable musical chills revealed by high-density EEG. Front. Neurosci. 2020, 14, 1114. [Google Scholar] [CrossRef] [PubMed]
- Saarikallio, S.; Erkkilä, J. The role of music in adolescents’ mood regulation. Psychol. Music 2007, 35, 88–109. [Google Scholar] [CrossRef]
- Saarikallio, S. Music as emotional self-regulation throughout adulthood. Psychol. Music 2011, 39, 307–327. [Google Scholar] [CrossRef]
- Linnemann, A.; Ditzen, B.; Strahler, J.; Doerr, J.M.; Nater, U.M. Music listening as a means of stress reduction in daily life. Psychoneuroendocrinology 2015, 60, 82–90. [Google Scholar] [CrossRef] [PubMed]
- Akelma, F.K.; Altınsoy, S.; Arslan, M.T.; Ergin, J. Effect of favorite music on postoperative anxiety and pain. Anaesthesist 2020, 69, 198–204. [Google Scholar] [CrossRef]
- Koelsch, S.; Jäncke, L. Music and the heart. Eur. Heart J. 2015, 36, 3043–3049. [Google Scholar] [CrossRef]
- Juslin, P.; Barradas, G.; Eerola, T. From sound to significance: Exploring the mechanisms underlying emotional reactions to music. Am. J. Psychol. 2015, 128, 281–304. [Google Scholar] [CrossRef]
- Groarke, J.M.; Hogan, M.J. Listening to self-chosen music regulates induced negative affect for both younger and older adults. PLoS ONE 2019, 14, e0218017. [Google Scholar] [CrossRef] [PubMed]
- Groarke, J.M.; Groarke, A.; Hogan, M.J.; Costello, L.; Lynch, D. Does listening to music regulate negative affect in a stressful situation? Examining the effects of self-selected and researcher-selected music using both silent and active controls. Appl. Psychol. Health Well-Being 2020, 12, 288–311. [Google Scholar] [CrossRef]
- Athanasopoulos, G.; Eerola, T.; Lahdelma, I.; Kaliakatsos-Papakostas, M. Harmonic organisation conveys both universal and culture-specific cues for emotional expression in music. PLoS ONE 2021, 16, e0244964. [Google Scholar] [CrossRef] [PubMed]
- Paszkiel, S.; Dobrakowski, P.; Łysiak, A. The impact of different sounds on stress level in the context of EEG, cardiac measures and subjective stress level: A pilot study. Brain Sci. 2020, 10, 728. [Google Scholar] [CrossRef] [PubMed]
- Azhari, A.; Rigo, P.; Bornstein, M.H.; Esposito, G. Where sounds occur matters: Context effects influence processing of salient vocalisations. Brain Sci. 2020, 10, 429. [Google Scholar] [CrossRef]
- Azhari, A.; Truzzi, A.; Rigo, P.; Bornstein, M.H.; Esposito, G. Putting salient vocalisations in context: Adults’ physiological arousal to emotive cues in domestic and external environments. Physiol. Behav. 2018, 196, 25–32. [Google Scholar] [CrossRef] [PubMed]
- Dolan, D.; Jensen, H.J.; Mediano, P.A.M.; Molina-Solana, M.; Rajpal, H.; Rosas, F.; Sloboda, J.A. The improvisational state of mind: A multidisciplinary study of an improvisatory approach to classical music repertoire performance. Front. Psychol. 2018, 9, 1341. [Google Scholar] [CrossRef] [PubMed]
- Wuttke-Linnemann, A.; Nater, U.M.; Ehlert, U.; Ditzen, B. Sex-specific effects of music listening on couples’ stress in everyday life. Sci. Rep. 2019, 9, 4880. [Google Scholar] [CrossRef] [PubMed]
- Murphy, L.; Levy, D. Emotions at home. In International Encyclopedia of Housing and Home; Elsevier: Amsterdam, The Netherlands, 2012; pp. 75–79. [Google Scholar] [CrossRef]
- Luft, C.D.B.; Bhattacharya, J. Aroused with heart: Modulation of heartbeat evoked potential by arousal induction and its oscillatory correlates. Sci. Rep. 2015, 5, 15717. [Google Scholar] [CrossRef]
- Ferreri, L.; Mas-Herrero, E.; Zatorre, R.J.; Ripollés, P.; Gomez-Andres, A.; Alicart, H.; Olivé, G.; Marco-Pallarés, J.; Antonijoan, R.M.; Valle, M.; et al. Dopamine modulates the reward experiences elicited by music. Proc. Natl. Acad. Sci. USA 2019, 116, 3793–3798. [Google Scholar] [CrossRef]
- Wilkins, R.; Hodges, D.; Laurienti, P.; Steen, M.; Burdette, J. Network science and the effects of music preference on functional brain connectivity: From Beethoven to Eminem. Sci. Rep. 2014, 4, 6130. [Google Scholar] [CrossRef]
- Särkämö, T.; Tervaniemi, M.; Laitinen, S.; Forsblom, A.; Soinila, S.; Mikkonen, M.; Autti, T.; Silvennoinen, H.M.; Erkkilä, J.; Laine, M.; et al. Music listening enhances cognitive recovery and mood after middle cerebral artery stroke. Brain 2008, 131, 866–876. [Google Scholar] [CrossRef]
- Sihvonen, A.J.; Leo, V.; Ripollés, P.; Lehtovaara, T.; Ylönen, A.; Rajanaro, P.; Laitinen, S.; Forsblom, A.; Saunavaara, J.; Autti, T.; et al. Vocal music enhances memory and language recovery after stroke: Pooled results from two RCTs. Ann. Clin. Transl. Neurol. 2020, 7, 2272–2287. [Google Scholar] [CrossRef]
- Finn, S.; Fancourt, D. The biological impact of listening to music in clinical and nonclinical settings: A systematic review. Prog. Brain Res. 2018, 237, 173–200. [Google Scholar] [CrossRef]
- Khalfa, S.; Bella, S.D.; Roy, M.; Peretz, I.; Lupien, S.J. Effects of relaxing music on salivary cortisol level after psychological stress. Ann. N. Y. Acad. Sci. 2003, 999, 374–376. [Google Scholar] [CrossRef] [PubMed]
| Music Type | Median | Z | p | |||
|---|---|---|---|---|---|---|
| Lab | Home | Lab—Home | ||||
| High arousal | Baseline | 2.25 | 2.25 | 0 | −0.776 | 0.438 |
| Favorite | 2.5 | 2.375 | 0.25 | −1.494 | 0.135 | |
| Neutral | 2 | 2.25 | −0.25 | −2.138 | 0.033 | |
| Low arousal | Baseline | 2.5 | 2.25 | 0.25 | −2.989 | 0.003 |
| Favorite | 2.75 | 2.5 | 0.25 | −1.362 | 0.173 | |
| Neutral | 2.25 | 2.25 | 0.25 | −1.55 | 0.121 | |
| Positive valence | Baseline | 3 | 2.5 | 0.375 | −3.013 | 0.003 |
| Favorite | 3.25 | 3 | 0.25 | −1.712 | 0.087 | |
| Neutral | 2.75 | 2.5 | 0.125 | −1.348 | 0.178 | |
| Negative valence | Baseline | 1.5 | 1.75 | −0.125 | −2.856 | 0.004 |
| Favorite | 1.5 | 1.75 | 0 | −1.881 | 0.06 | |
| Neutral | 1.75 | 1.75 | 0 | −1.233 | 0.217 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tervaniemi, M.; Makkonen, T.; Nie, P. Psychological and Physiological Signatures of Music Listening in Different Listening Environments—An Exploratory Study. Brain Sci. 2021, 11, 593. https://doi.org/10.3390/brainsci11050593
Tervaniemi M, Makkonen T, Nie P. Psychological and Physiological Signatures of Music Listening in Different Listening Environments—An Exploratory Study. Brain Sciences. 2021; 11(5):593. https://doi.org/10.3390/brainsci11050593
Chicago/Turabian StyleTervaniemi, Mari, Tommi Makkonen, and Peixin Nie. 2021. "Psychological and Physiological Signatures of Music Listening in Different Listening Environments—An Exploratory Study" Brain Sciences 11, no. 5: 593. https://doi.org/10.3390/brainsci11050593
APA StyleTervaniemi, M., Makkonen, T., & Nie, P. (2021). Psychological and Physiological Signatures of Music Listening in Different Listening Environments—An Exploratory Study. Brain Sciences, 11(5), 593. https://doi.org/10.3390/brainsci11050593






