Effects of Specific Virtual Reality-Based Therapy for the Rehabilitation of the Upper Limb Motor Function Post-Ictus: Randomized Controlled Trial
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Participants and Setting
2.3. Outcome Measures
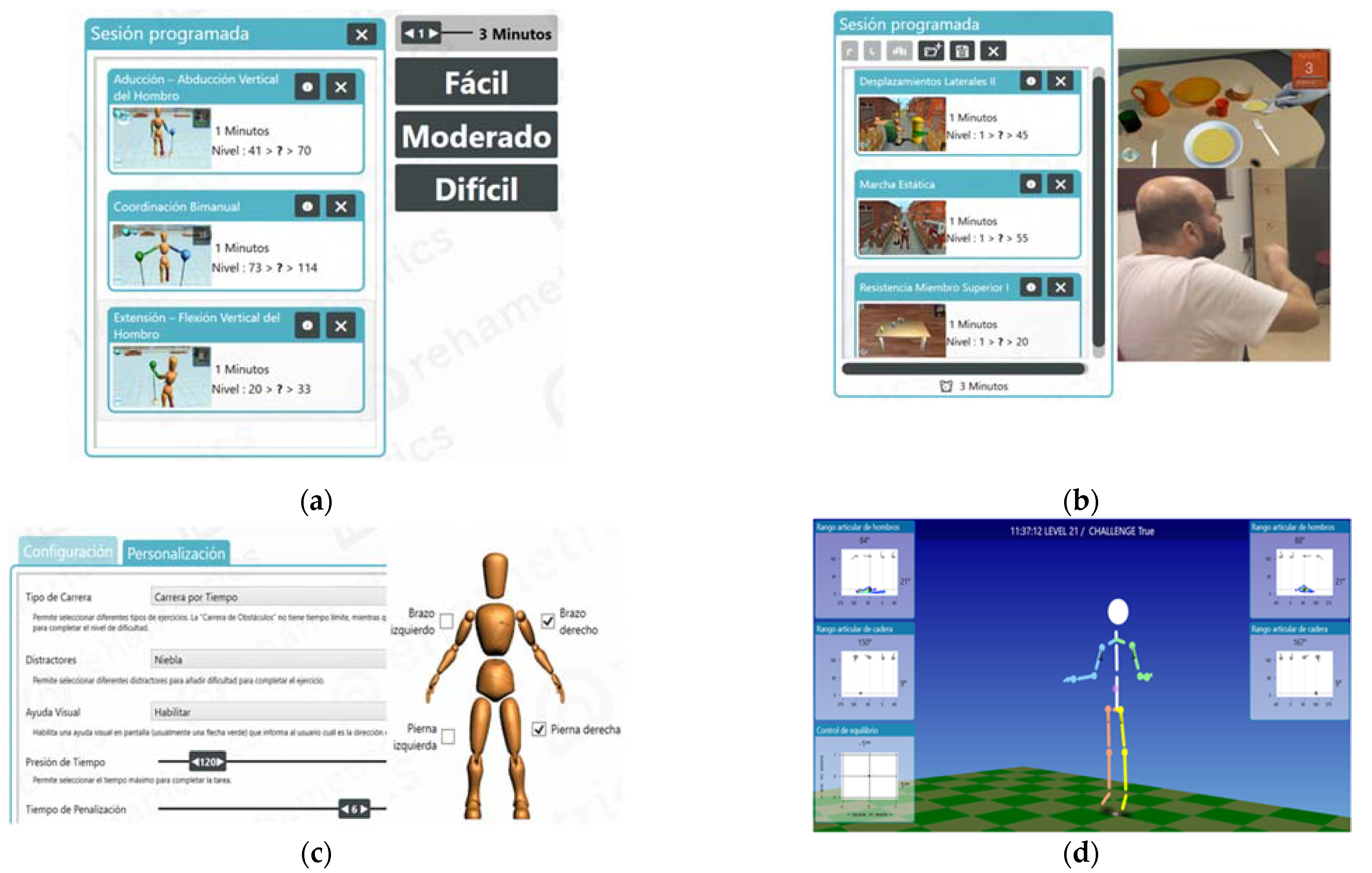
2.4. Intervention
2.5. Statistical Analysis
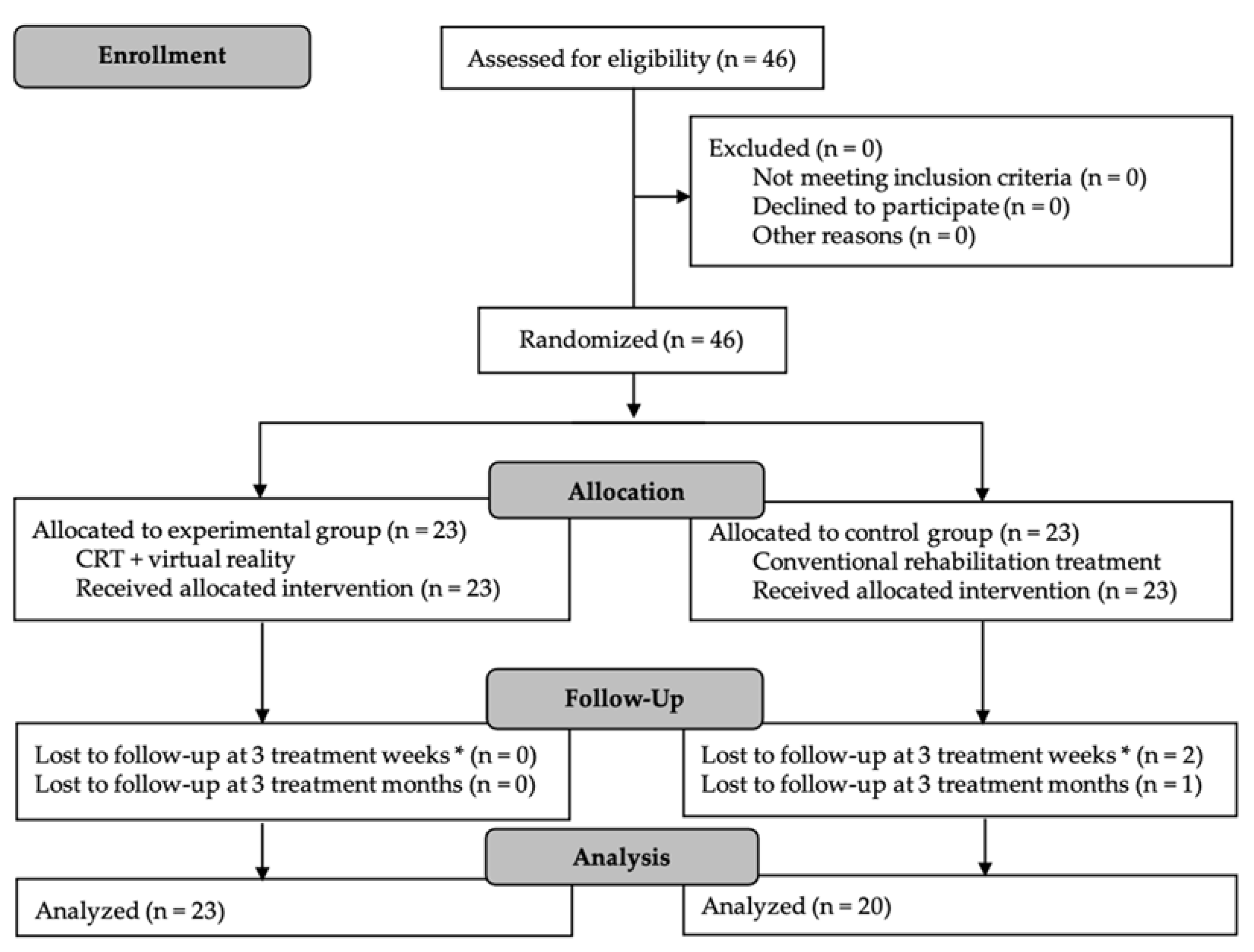
3. Results
4. Discussion
Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Mendis, S. Stroke Disability and Rehabilitation of Stroke: World Health Organization Perspective. Int. J. Stroke 2013, 8, 3–4. [Google Scholar] [CrossRef] [PubMed]
- Mathers, C.D.; Loncar, D. Projections of global mortality and burden of disease from 2002 to 2030. PLoS Med. 2006, 3, e442. [Google Scholar] [CrossRef] [PubMed]
- SAFE: The Economic Impact of Stroke. Available online: https://www.safestroke.eu/economic-impact-of-stroke/ (accessed on 14 March 2021).
- Duncan, J.; Van Wijck, F.; Pollock, A.; Ali, M. Outcome measures in post-stroke arm rehabilitation trials: Do existing measures capture outcomes that are important to stroke survivors, carers, and clinicians? Clin. Rehabil. 2019, 33, 737–749. [Google Scholar] [CrossRef] [PubMed]
- Bosomworth, H.; Rodgers, H.; Shaw, L.; Smith, L.; Aird, L.; Howel, D.; Wilson, N.; Alvarado, N.; Andole, S.; Cohen, D.L.; et al. Evaluation of the enhanced upper limb therapy programme within the Robot-Assisted Training for the Upper Limb after Stroke trial: Descriptive analysis of intervention fidelity, goal selection and goal achievement. Clin. Rehabil. 2020, 35, 119–134. [Google Scholar] [CrossRef]
- Schnabel, S.; Van Wijck, F.; Bain, B.; Barber, M.; Dall, P.; Fleming, A.; Kerr, A.; Langhorne, P.; McConnachie, A.; Molloy, K.; et al. Experiences of augmented arm rehabilitation including supported self-management after stroke: A qualitative investigation. Clin. Rehabil. 2020, 35, 288–301. [Google Scholar] [CrossRef]
- Crichton, S.L.; Bray, B.D.; McKevitt, C.; Rudd, A.G.; Wolfe, C.D.A. Patient outcomes up to 15 years after stroke: Survival, disability, quality of life, cognition and mental health. J. Neurol. Neurosurg. Psychiatry 2016, 87, 1091–1098. [Google Scholar] [CrossRef]
- Mukherjee, D.; Patil, C.G. Epidemiology and the Global Burden of Stroke. World Neurosurg. 2011, 76, S85–S90. [Google Scholar] [CrossRef] [PubMed]
- Cheiloudaki, E.; Alexopoulos, E.C. Adherence to Treatment in Stroke Patients. Int. J. Environ. Res. Public Health 2019, 16, 196. [Google Scholar] [CrossRef] [PubMed]
- Turolla, A.; Dam, M.; Ventura, L.; Tonin, P.; Agostini, M.; Zucconi, C.; Kiper, P.; Cagnin, A.; Piron, L. Virtual reality for the rehabilitation of the upper limb motor function after stroke: A prospective controlled trial. J. Neuroeng. Rehabil. 2013, 10, 85. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, P.E.; Cailliet, R.; Kaplan, C.P. Rehabilitation of Stroke; Kaplan, P.E., Cailliet, R., Kaplan, C.P., Eds.; Elsevier Science, Butterworth-Heinemann: Burlington, MA, USA, 2003; p. 180. [Google Scholar]
- Buma, F.E.; Lindeman, E.; Ramsey, N.F.; Kwakkel, G. Review: Functional Neuroimaging Studies of Early Upper Limb Recovery After Stroke: A Systematic Review of the Literature. Neurorehabil. Neural Repair 2010, 24, 589–608. [Google Scholar] [CrossRef]
- Kwakkel, G.; Kollen, B.J.; Van der Grond, J.; Prevo, A.J.H. Probability of Regaining Dexterity in the Flaccid Upper Limb. Stroke 2003, 34, 2181–2186. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, M.; Bermúdez, S.; Duarte, E.; Verschure, P.F.M.J. Virtual reality based rehabilitation speeds up functional recovery of the upper extremities after stroke: A randomized controlled pilot study in the acute phase of stroke using the rehabilitation gaming system. Restor. Neurol. Neurosci. 2011, 29, 287–298. [Google Scholar] [CrossRef]
- Mekbib, D.B.; Han, J.; Zhang, L.; Fang, S.; Jiang, H.; Zhu, J.; Roe, A.W.; Xu, D. Virtual reality therapy for upper limb rehabilitation in patients with stroke: A meta-analysis of randomized clinical trials. Brain Inj. 2020, 34, 456–465. [Google Scholar] [CrossRef] [PubMed]
- Thomson, K.; Pollock, A.; Bugge, C.; Brady, M. Commercial Gaming Devices for Stroke Upper Limb Rehabilitation: A Systematic Review. Int. J. Stroke 2014, 9, 479–488. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, M.A.; Singh, D.K.A.; Mohd, N.A.; Hooi, K.; Ibrahim, N. Virtual Reality Games as an Adjunct in Improving Upper Limb Function and General Health among Stroke Survivors. Int. J. Environ. Res. Public Health 2019, 16, 5144. [Google Scholar] [CrossRef] [PubMed]
- Akinladejo, F.O. Virtual Environments in Physical Therapy. In Virtual Reality and Environments; Sík Lányi, C., Ed.; IntechOpen, University of Pannonia: Veszprém, Hungary, 2012; pp. 1–10. [Google Scholar] [CrossRef]
- Sveistrup, H. Motor rehabilitation using virtual reality. J. Neuroeng. Rehabil. 2004, 1, 10. [Google Scholar] [CrossRef]
- Laver, K.E.; George, S.; Thomas, S.; Deutsch, J.E.; Crotty, M. Virtual reality for stroke rehabilitation. Cochrane Database Syst. Rev. 2015, 2015, CD008349. [Google Scholar] [CrossRef]
- Laver, K.E.; Lange, B.; George, S.; Deutsch, J.E.; Saposnik, G.; Crotty, M. Virtual reality for stroke rehabilitation. Cochrane Database Syst. Rev. 2017, 11, CD008349. [Google Scholar] [CrossRef]
- Hruby, F.; Castellanos, I.; Ressl, R. Cartographic Scale in Immersive Virtual Environments. J. Cartogr. Geogr. Inf. 2020, 21, 399. [Google Scholar] [CrossRef]
- Liang, Z.; Zhou, K.; Gao, K. Development of Virtual Reality Serious Game for Underground Rock-Related Hazards Safety Training. IEEE Access 2019, 2019, 118639–118649. [Google Scholar] [CrossRef]
- O’Meara, D.; Korte, A.; Dickmann, F. Effects of Virtual Reality Locomotion Techniques on Distance Estimations. ISPRS Int. J. Geo-Inf. 2021, 10, 150. [Google Scholar] [CrossRef]
- Ruddle, R.A.; Volkova, E.; Bülthoff, H.H. Walking improves your cognitive map in enviroments that are large-scale and large in extent. ACM Trans. Comput. Hum. Interact. 2011, 18, 1–20. [Google Scholar] [CrossRef]
- ISRCTN—ISRCTN27760662: Effectiveness of Virtual Reality Devices in the Rehabilitation of Adults with Stroke. Available online: http://www.isrctn.com/ISRCTN27760662 (accessed on 8 January 2021).
- Schulz, K.F.; Altman, D.G.; Moher, D.; CONSORT Group. CONSORT 2010 Statement: Updated guidelines for reporting parallel group randomised trials. BMC Med. 2010, 8, 18. [Google Scholar] [CrossRef]
- Liu, X.; Cruz, S.; Moher, D.; Calvert, M.J.; Denniston, A.K.; The SPIRIT-AI and CONSORT-AI Working Group. Reporting guidelines for clinical trial reports for interventions involving artificial intelligence: The CONSORT-AI extension. Nat. Med. 2020, 26, 1364–1374. [Google Scholar] [CrossRef]
- Roman, N.; Miclaus, R.; Repanovici, A.; Nicolau, C. Equal Opportunities for Stroke Survivors’ Rehabilitation: A Study on the Validity of the Upper Extremity Fugl-Meyer Assessment Scale Translated and Adapted into Romanian. Medicina 2020, 56, 409. [Google Scholar] [CrossRef]
- Kim, W.S.; Cho, S.; Baek, D.; Bang, H.; Paik, N.J. Upper Extremity Functional Evaluation by Fugl-Meyer Assessment Scoring Using Depth-Sensing Camera in Hemiplegic Stroke Patients. PLoS ONE 2016, 11, e0158640. [Google Scholar] [CrossRef] [PubMed]
- Winstein, C.J.; Stein, J.; Arena, R.; Bates, B.; Cherney, S.C.; Deruyter, F.; Eng, J.J.; Fisher, B.; Harvey, R.L.; Lang, C.E.; et al. Guidelines for Adult Stroke Rehabilitation and Recovery: A Guideline for Healthcare Professionals From the American Heart Association/American Stroke Association. Stroke 2016, 47. [Google Scholar] [CrossRef]
- Woodbury, M.L.; Velozo, C.A.; Richards, L.G.; Duncan, P.W.; Studenski, S.; Lai, S.M. Longitudinal Stability of the Fugl-Meyer Assessment of the Upper Extremity. Arch. Phys. Med. Rehabil. 2008, 89, 1563–1569. [Google Scholar] [CrossRef] [PubMed]
- Ferrer, B.M. Adaptación y Validación al Español de la Escala Fugl-Meyer en el Manejo de la Rehabilitación de Pacientes con Ictus. Ph.D. Thesis, Sevilla University, Sevilla, Spain, 2016. Available online: http://hdl.handle.net/11441/40335 (accessed on 9 January 2021).
- Brashear, A.; Zafonte, R.; Corcoran, M.; Galvez-Jimenez, N.; Gracies, J.M.; Gordon, M.F.; Mcafee, A.; Ruffing, K.; Thompson, B.; Williams, M.; et al. Inter- and intrarater reliability of the Ashworth Scale and the Disability Assessment Scale in patients with upper-limb poststroke spasticity. Arch. Phys. Med. Rehabil. 2002, 83, 1349–1354. [Google Scholar] [CrossRef]
- Duncan, P.W.; Bode, R.K.; Min, S.; Perera, S. Rasch analysis of a new stroke-specific outcome scale: The stroke impact scale11No commercial party having a direct financial interest in the results of the research supporting this article has or will confer a benefit upon the author(s) or upon any organization with which the author(s) is/are associated. Arch. Phys. Med. Rehabil. 2003, 84, 950–963. [Google Scholar] [CrossRef]
- Lin, K.C.; Fu, T.; Wu, C.Y.; Hsieh, Y.W.; Chen, C.L.; Lee, P.C. Psychometric comparisons of the Stroke Impact Scale 3.0 and Stroke-Specific Quality of Life Scale. Qual. Life Res. 2010, 19, 435–443. [Google Scholar] [CrossRef]
- Chou, C.Y.; Ou, Y.C.; Chiang, T.R. Psychometric comparisons of four disease-specific health-related quality of life measures for stroke survivors. Clin. Rehabil. 2015, 29, 816–829. [Google Scholar] [CrossRef] [PubMed]
- Vellone, E.; Savini, S.; Fida, R.; Dickson, V.V.; Melkus, G.D.; Carod-Artal, F.J.; Rocco, G.; Alvaro, R. Psychometric Evaluation of the Stroke Impact Scale 3.0. J. Cardiovasc. Nurs. 2015, 30, 229–241. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.C.; Lin, G.H.; Huang, Y.J.; Huang, S.L.; Chou, C.Y.; Chiang, H.Y.; Hsieh, C.L. Cross-Validation of the Factorial Validity of the Stroke Impact Scale 3.0 in Patients With Stroke. Am. J. Occup. Ther. 2021, 75, 7502205070. [Google Scholar] [CrossRef] [PubMed]
- Carmeli, E.; Peleg, S.; Bartur, G.; Elbo, E.; Vatine, J.J. HandTutorTM enhanced hand rehabilitation after stroke—A pilot study. Physiother. Res. Int. 2011, 16, 191–200. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Hernández, M.; Fernández-Panadero, C.; López-Martín, O.; Polonio-López, B. Hand Rehabilitation after Chronic Brain Damage: Effectiveness, Usability and Acceptance of Technological Devices: A Pilot Study. In Physical Disabilities—Therapeutic Implications; Tan, U., Ed.; IntechOpen, Cukurova University: Sarıçam/Adana, Turkey, 2017; pp. 57–72. [Google Scholar] [CrossRef]
- Chua, K.S.G.; Kuah, C.W.K. Innovating With Rehabilitation Technology in the Real World: Promises, Potentials, and Perspectives. Am. J. Phys. Med. Rehabil. 2017, 96, S150–S156. [Google Scholar] [CrossRef] [PubMed]
- Rehametrics|Cuantificando la Rehabilitación Física y Cognitiva. Available online: https://rehametrics.com/ (accessed on 9 January 2021).
- Grefkes, C.; Fink, G.R. Connectivity-based approaches in stroke and recovery of function. Lancet Neurol. 2014, 13, 206–216. [Google Scholar] [CrossRef]
- Saposnik, G.; Cohen, L.G.; Mamdani, M.; Pooyania, S.; Ploughman, M.; Cheung, D.; Shaw, J.; Hall, J.; Nord, P.; Dukelow, S.; et al. Efficacy and safety of non-immersive virtual reality exercising in stroke rehabilitation (EVREST): A randomised, multicentre, single-blind, controlled trial. Lancet Neurol. 2016, 15, 1019–1027. [Google Scholar] [CrossRef]
- Sampaio-Baptista, C.; Sanders, Z.B.; Johansen-Berg, H. Structural Plasticity in Adulthood with Motor Learning and Stroke Rehabilitation. Annu. Rev. Neurosci. 2018, 41, 25–40. [Google Scholar] [CrossRef]
- Martínez, M.C.; Parejo, M.T.; Laiz, N.M. Valoración del uso de las nuevas tecnologías en personas con esclerosis múltiple. REDIS 2018, 6, 149–171. [Google Scholar] [CrossRef]
- Abreu, J.; Rebelo, S.; Paredes, H.; Barroso, J.; Martins, P.; Reis, A.; EuricoVasco, A. Assessment of Microsoft Kinect in the Monitoring and Rehabilitation of Stroke Patients. In Recent Advances in Information Systems and Technologies; Rocha, Á., Correia, A.M., Adeli, H., Reis, L.P., Costanzo, S., Eds.; Springer International Publishing: Berlin/Heidelberg, Germany, 2017; pp. 167–174. [Google Scholar] [CrossRef]
- Ashwini, K.; Amutha, R.; Nagarajan, K.K.; Raj, S.A. Kinect based Upper Limb Performance Assessment in Daily Life Activities. In Proceedings of the International Conference on Wireless Communications Signal Processing and Networking (WiSPNET), Chennai, India, 21–23 March 2019; pp. 201–205. [Google Scholar] [CrossRef]
- Mousavi, H.; Khademi, M. A Review on Technical and Clinical Impact of Microsoft Kinect on Physical Therapy and Rehabilitation. J. Med. Eng. 2014, 2014, 846514. [Google Scholar] [CrossRef]
- Rodríguez-Hernández, M.; Criado-Álvarez, J.J.; Corregidor-Sánchez, A.I.; Martín-Conty, J.L.; Mohedano-Moriano, A.; Polonio-López, B. Effects of Virtual Reality-Based Therapy on Quality of Life of Patients with Subacute Stroke: A Three-Month Follow-Up Randomized Controlled Trial. Int. J. Environ. Res. Public Health 2021, 18, 2810. [Google Scholar] [CrossRef]
- Kim, J.H. Effects of a virtual reality video game exercise program on upper extremity function and daily living activities in stroke patients. J. Phys. Ther. Sci. 2018, 30, 1408–1411. [Google Scholar] [CrossRef]
- Norouzi-Gheidari, N.; Hernandez, A.; Archambault, P.S.; Higgins, J.; Poissant, L.; Kairy, D. Feasibility, Safety and Efficacy of a Virtual Reality Exergame System to Supplement Upper Extremity Rehabilitation Post-Stroke: A Pilot Randomized Clinical Trial and Proof of Principle. Int. J. Environ. Res. Public Health 2020, 17, 113. [Google Scholar] [CrossRef]
- Miclaus, R.; Roman, N.; Caloian, S.; Mitoiu, B.; Suciu, O.; Onofrei, R.R.; Pavel, E.; Neculau, A. Non-Immersive Virtual Reality for Post-Stroke Upper Extremity Rehabilitation: A Small Cohort Randomized Trial. Brain Sci. 2020, 10, 655. [Google Scholar] [CrossRef] [PubMed]
- Schuster-Amft, C.; Eng, K.; Suica, Z.; Thaler, I.; Signer, S.; Lehmann, I.; Schmid, L.; McCaskey, M.; Hawkins, M.; Verra, M.L.; et al. Effect of a four-week virtual reality-based training versus conventional therapy on upper limb motor function after stroke: A multicenter parallel group randomized trial. PLoS ONE 2018, 13, e0204455. [Google Scholar] [CrossRef] [PubMed]
- Bevilacqua, R.; Maranesi, E.; Riccardi, G.R.; Donna, V.D.; Pelliccioni, P.; Luzi, R.; Lattanzio, F.; Pelliccioni, G. Non-Immersive Virtual Reality for Rehabilitation of the Older People: A Systematic Review into Efficacy and Effectiveness. J. Clin. Med. 2019, 8, 1882. [Google Scholar] [CrossRef] [PubMed]
- Saposnik, G.; Teasell, R.; Mamdani, M.; Hall, J.; McIlroy, W.; Cheung, D.; Thorpe, K.E.; Cohen, L.G.; Bayley, M. Effectiveness of Virtual Reality Using Wii Gaming Technology in Stroke Rehabilitation: A Pilot Randomized Clinical Trial and Proof of Principle. Stroke 2010, 41, 1477–1484. [Google Scholar] [CrossRef]
- Maier, M.; Rubio, B.; Duff, A.; Duarte, E.; Verschure, P.F.M.J. Effect of Specific Over Nonspecific VR-Based Rehabilitation on Poststroke Motor Recovery: A Systematic Meta-analysis. Neurorehabil. Neural Repair 2019, 33, 112–129. [Google Scholar] [CrossRef] [PubMed]
- Rand, D.; Weiss, P.L.; Katz, N. Training Multitasking in a Virtual Supermarket: A Novel Intervention after Stroke. Am. J. Occup. Ther. 2009, 63, 535–542. [Google Scholar] [CrossRef]
- Oh, Y.B.; Kim, G.W.; Han, K.S.; Won, Y.H.; Park, S.H.; Seo, J.H.; Ko, M.H. Efficacy of Virtual Reality Combined With Real Instrument Training for Patients With Stroke: A Randomized Controlled Trial. Arch. Phys. Med. Rehabil. 2019, 100, 1400–1408. [Google Scholar] [CrossRef] [PubMed]
- Ho, T.H.; Yang, F.C.; Lin, R.C.; Chien, W.C.; Chung, C.H.; Chiang, S.L.; Chou, C.-H.; Tsai, C.-K.; Lin, Y.-K.; Lee, J.-T. Impact of virtual reality-based rehabilitation on functional outcomes in patients with acute stroke: A retrospective case-matched study. J. Neurol. 2019, 266, 589–597. [Google Scholar] [CrossRef]
- Cano-Mañas, M.J.; Collado-Vázquez, S.; Rodríguez, J.; Muñoz, A.J.; Cano-de-la-Cuerda, R. Effects of Video-Game Based Therapy on Balance, Postural Control, Functionality, and Quality of Life of Patients with Subacute Stroke: A Randomized Controlled Trial. J. Healthc. Eng. 2020, 2020, 5480315. [Google Scholar] [CrossRef] [PubMed]
- Coomer, N.; Bullard, S.; Clinton, W.; Williams-Sanders, B. Evaluating the effects of four VR locomotion methods: Joystick, arm-cycling, point-tugging, and teleporting. In Proceedings of the SAP’18: 15th ACM Symposium on Applied Perception, Vancouver, BC, Canada, 10–11 August 2018; Grimm, C., Willemsen, P., Eds.; Association for Computing Machinery: New York, NY, USA, 2018; pp. 1–8. [Google Scholar]
- Cherep, L.A.; Lim, A.F.; Kelly, J.W.; Acharya, D.; Velasco, A.; Bustamante, E.; Ostrander, A.G.; Gilbert, S.B. Spatial cognitive implications of teleporting through virtual environments. J. Exp. Psychol. Appl. 2020, 26, 480–492. [Google Scholar] [CrossRef] [PubMed]
- Kelly, J.W.; Ostrander, A.G.; Lim, A.F.; Cherep, L.A.; Gilbert, S.B. Teleporting through virtual environments: Effects of path scale and environment scale on spatial updating. IEEE Trans. Vis. Comput. Graph. 2020, 26, 1841–1850. [Google Scholar] [CrossRef] [PubMed]
| Characteristics: Study Variables | Experimental Group (EG) | Control Group (CG) | Difference of Mean between Groups |
|---|---|---|---|
| (n = 23) | (n = 20) | (p-Value) | |
| Age | −0.9 (0.812) | ||
| Mean (SD) | 62.6 (13.5) | 63.6 (12.2) | 0.566 |
| Under 55 years (%) | 26.1 | 25 | |
| 55 to 70 years (%) | 30.4 | 45 | |
| Over 70 years (%) | 43.5 | 30 | |
| Sex (%) | |||
| Male | 78.3 | 85 | 0.571 |
| Female | 21.7 | 15 | |
| Main diagnostic (%) | 0.883 | ||
| Ischemic/thrombotic | 91.3 | 90 | |
| Hemorrhagic | 8.7 | 10 | |
| Middle cerebral artery lesion (%) | 60.9 | 55 | 0.697 |
| Location of the brain injury (%) | 0.832 | ||
| Right | 82.6 | 85 | |
| Left | 17.4 | 15 | |
| Time since diagnostic (days) * | |||
| Baseline (pre-intervention) | 55.3 (34.3) | 54.2 (30.4) | 1.1 (0.909) |
| Post-intervention (3-week follow-up) | 75.3 (34.3) | 74.2 (30.4) | 1.1 (0.909) |
| Follow-up (3 months) | 162.3 (36.9) | 157.2 (36.1) | 5.1 (0.650) |
| Hemispatial neglect syndrome (%) | 13 | 10 | 0.756 |
| Presence of pain in extremities (%) | |||
| Baseline (pre-intervention) | 43.5 | 50 | 0.669 |
| Post-intervention (3-week follow-up) | 21.7 | 80 | 0 |
| Follow-up (3 months) | 82.6 | 100 | 0.05 |
| EuroQoL visual analog scale (EQ-VAS) * | |||
| Baseline | 29.1 (12.8) | 25.5 (5.1) | 0.241 |
| Post-intervention | 86.5 (7.1) | 57.0 (4.7) | 0 |
| Follow-up | 78.3 (10.7) | 58.5 (5.9) | 0 |
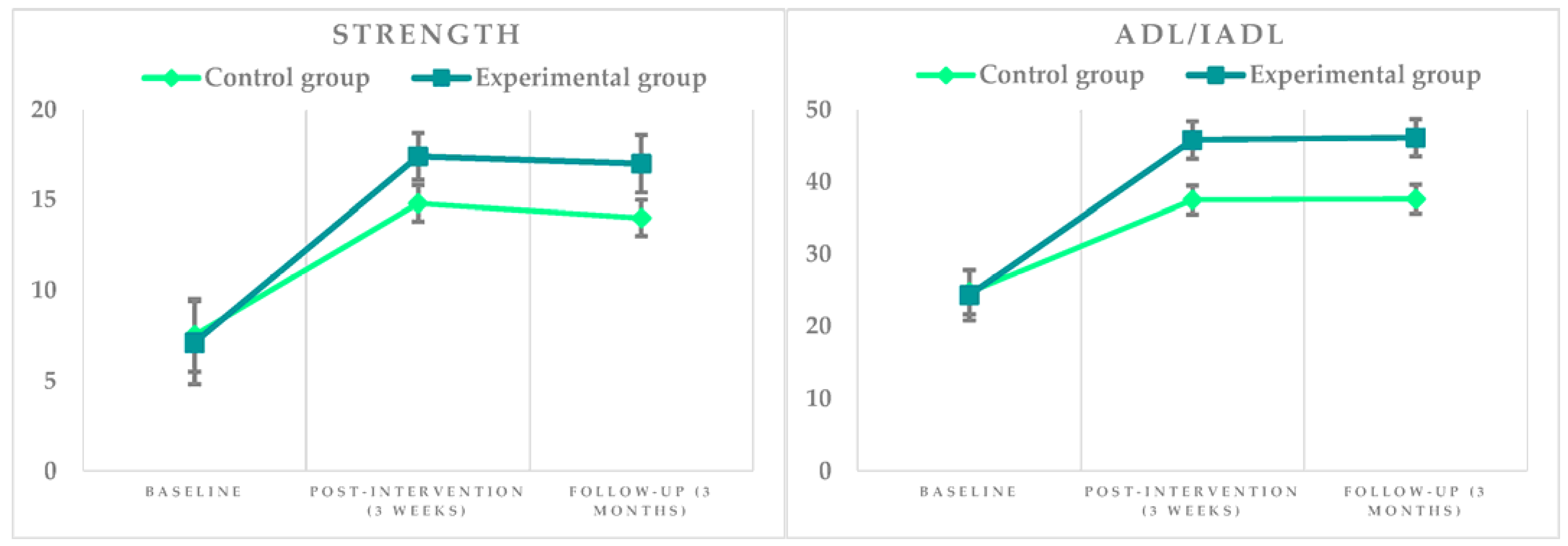
| FMA-UE (Total A. Upper extremity) * | |||
| Baseline | 12.6 (3.4) | 12.7 (3.3) | −0.1 (0.930) |
| Post-intervention | 30.1 (3.0) | 24.7 (3.7) | 5.4 (0.000) |
| Follow-up | 31.1 (4.3) | 26.9 (4.1) | 4.2 (0.002) |
| Dominance (change baseline/post-intervention) (%) | |||
| Right | 100.0/100.0 | 100.0/85.0 | 0.054 |
| Left | 0 | 0/15.0 |
| Intervention Group | Difference Baseline/Follow-Up | ||||||
|---|---|---|---|---|---|---|---|
| Modified Ashworth Scale (T-Score) | Baseline Mean (SD) | Post-Intervention Mean (SD) | Follow-Up Mean (SD) | Mean (CI95%) | ANOVA | ||
| F | p | η2 Parcial | |||||
| Experimental group | 1.30 (0.70) | 0.60 (0.50) | 0.87 (0.46) | 0.43 (0.07/0.79) * | |||
| Control group | 1.22 (0.67) | 1.05 (0.21) | 1.75 (0.44) | −0.45 (−0.85/−0.04) * | 12.7 | 0.001 | 0.237 |
| p | 0.670 | 0.001 | 0.000 | ||||
| SIS 3.0. Physical Domain. | Experimental Group (EG) | Control Group (CG) | Difference of Mean between Groups |
|---|---|---|---|
| Strength | (n = 23) | (n = 20) | (p-Value) |
| 1a. Of the arm that was most affected by your stroke | |||
| Baseline | 1.6 (0.7) | 1.7 (0.7) | −0.04 (0.838) |
| Post-intervention | 4.3 (0.5) | 3.4 (0.5) | 0.92 (0.000) |
| Follow-up | 4.3 (0.5) | 3.4 (0.5) | 0.95 (0.000) |
| 1b. Grip of your hand that was most affected by your stroke | |||
| Baseline | 1.3 (0.6) | 1.4 (0.6) | −0.04 (0.800) |
| Post-intervention | 4.2 (0.4) | 3.0 (0.0) | 1.22 (0.000) |
| Follow-up | 4.3 (0.5) | 3.0 (0.0) | 1.30 (0.000) |
| 1c. Of the leg that was most affected by your stroke | |||
| Baseline | 2.1 (1.0) | 2.3 (0.8) | −0.13 (0.623) |
| Post-intervention | 4.4 (0.5) | 4.2 (0.4) | 0.24 (0.086) |
| Follow-up | 4.2 (0.7) | 3.8 (0.4) | 0.37 (0.046) |
| 1d. Of the foot/ankle that was most affected by your stroke | |||
| Baseline | 2.0 (0.9) | 2.2 (0.8) | −0.13 (0.608) |
| Post-intervention | 4.4. (0.5) | 4.2 (0.4) | 0.24 (0.086) |
| Follow-up | 4.2 (0.7) | 3.8 (0.4) | 0.37 (0.046) |
| SIS 3.0. Physical Domain. | Experimental Group (EG) | Control Group (CG) | Difference of Mean between Groups |
|---|---|---|---|
| ADL/IADL | (n = 23) | (n = 20) | (p-Value) |
| 5a. Can you cut your food with a knife and fork? | |||
| Baseline | 1.5 (0.6) | 1.7 (0.6) | −0.17 (0.317) |
| Post-intervention | 4.6 (0.5) | 3.3 (0.5) | 1.28 (0.000) |
| Follow-up | 4.6 (0.5) | 3.3. (0.5) | 1.27 (0.000) |
| 5b. Can you dress the top part of your body? | |||
| Baseline | 1.7 (0.8) | 1.7 (0.6) | −0.04 (0.836) |
| Post-intervention | 4.9 (0.3) | 3.4 (0.5) | 1.49 (0.000) |
| Follow-up | 5.0 (0.0) | 3.4 (0.5) | 1.60 (0.000) |
| 5c. Can you bathe yourself? | |||
| Baseline | 1.6 (0.7) | 1.7 (0.6) | −0.09 (0.667) |
| Post-intervention | 4.5 (0.5) | 3.3 (0.5) | 1.14 (0.000) |
| Follow-up | 4.6 (0.5) | 3.4 (0.5) | 1.22 (0.000) |
| 5d. Can you clip your toenails? | |||
| Baseline | 1.3 (0.5) | 1.3 (0.6) | −0.09 (0.599) |
| Post-intervention | 3.7 (0.8) | 3.2 (0.4) | 0.51 (0.010) |
| Follow-up | 3.7 (0.8) | 3.2 (0.4) | 0.50 (0.013) |
| 5e. Can you get to the toilet on time? | |||
| Baseline | 3.2 (1.0) | 3.2 (0.8) | 0.04 (0.870) |
| Post-intervention | 4.8 (0.4) | 4.5 (0.5) | 0.26 (0.073) |
| Follow-up | 4.8 (0.4) | 4.6 (0.5) | 0.23 (0.109) |
| 5f. Can you control your bladder (not have an accident)? | |||
| Baseline | 5.0 (0.0) | 5.0 (0.2) | 0.04 (0.323) |
| Post-intervention | 5.0 (0.0) | 5.0 (0.2) | 0.05 (0.301) |
| Follow-up | 5.0 (0.0) | 5.0 (0.2) | 0.05 (0.289) |
| 5g. Can you control your bowels (not have an accident)? | |||
| Baseline | 5.0 (0.0) | 5.0 (0.2) | 0.04 (0.323) |
| Post-intervention | 5.0 (0.0) | 5.0 (0.2) | 0.05 (0.301) |
| Follow-up | 5.0 (0.0) | 5.0 (0.2) | 0.05 (0.289) |
| 5h. Can you do light household tasks/chores (e.g., dust, make the bed, take out garbage, do the dishes)? | |||
| Baseline | 1.7 (0.6) | 1.7 (0.4) | 0.00 (1.000) |
| Post-intervention | 4.7 (0.5) | 3.8 (0.4) | 0.93 (0.000) |
| Follow-up | 4.7 (0.5) | 3.8 (0.4) | 0.95 (0.000) |
| 5i. Can you go shopping? | |||
| Baseline | 1.7 (0.5) | 1.7 (0.4) | −0.09 (0.532) |
| Post-intervention | 4.4 (0.5) | 3.1 (0.4) | 1.34 (0.000) |
| Follow-up | 4.5 (0.5) | 3.1 (0.4) | 1.38 (0.000) |
| 5j. Can you do heavy household chores (e.g., vacuum, laundry, or yard work)? | |||
| Baseline | 1.7 (0.5) | 1.7 (0.4) | −0.09 (0.532) |
| Post-intervention | 4.3 (0.6) | 3.0 (0.4) | 1.21 (0.000) |
| Follow-up | 4.3 (0.6) | 3.1 (0.4) | 1.25 (0.000) |
| Intervention Group | Difference Baseline/Follow-Up | ||||||
|---|---|---|---|---|---|---|---|
| Baseline Mean (SD) | Post-Intervention Mean (SD) | Follow-Up Mean (SD) | Mean (CI95%) | ANOVA | |||
| F | p | η2 Parcial | |||||
| Global Recovery SIS 3.0 (T-Score) | |||||||
| Experimental group | 28.7 (12.5) | 86.5 (7.1) | 78.3 (10.7) | −49.5 (−56.4/−42.8) * | |||
| Control group | 26.1 (4.5) | 56.7 (4.8) | 59.0 (6.4) | −33.0 (−36.8/−29.1) * | 70.6 | 0.000 | 0.633 |
| p | 0.359 | 0.000 | 0.000 | ||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rodríguez-Hernández, M.; Polonio-López, B.; Corregidor-Sánchez, A.-I.; Martín-Conty, J.L.; Mohedano-Moriano, A.; Criado-Álvarez, J.-J. Effects of Specific Virtual Reality-Based Therapy for the Rehabilitation of the Upper Limb Motor Function Post-Ictus: Randomized Controlled Trial. Brain Sci. 2021, 11, 555. https://doi.org/10.3390/brainsci11050555
Rodríguez-Hernández M, Polonio-López B, Corregidor-Sánchez A-I, Martín-Conty JL, Mohedano-Moriano A, Criado-Álvarez J-J. Effects of Specific Virtual Reality-Based Therapy for the Rehabilitation of the Upper Limb Motor Function Post-Ictus: Randomized Controlled Trial. Brain Sciences. 2021; 11(5):555. https://doi.org/10.3390/brainsci11050555
Chicago/Turabian StyleRodríguez-Hernández, Marta, Begoña Polonio-López, Ana-Isabel Corregidor-Sánchez, José L. Martín-Conty, Alicia Mohedano-Moriano, and Juan-José Criado-Álvarez. 2021. "Effects of Specific Virtual Reality-Based Therapy for the Rehabilitation of the Upper Limb Motor Function Post-Ictus: Randomized Controlled Trial" Brain Sciences 11, no. 5: 555. https://doi.org/10.3390/brainsci11050555
APA StyleRodríguez-Hernández, M., Polonio-López, B., Corregidor-Sánchez, A.-I., Martín-Conty, J. L., Mohedano-Moriano, A., & Criado-Álvarez, J.-J. (2021). Effects of Specific Virtual Reality-Based Therapy for the Rehabilitation of the Upper Limb Motor Function Post-Ictus: Randomized Controlled Trial. Brain Sciences, 11(5), 555. https://doi.org/10.3390/brainsci11050555










