The Body across the Lifespan: On the Relation between Interoceptive Sensibility and High-Order Body Representations
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Behavioral Testing
2.2.1. Assessment of the Interoceptive Sensibility
2.2.2. Assessment of Body Representations
Assessment of the Action-Oriented BR
Assessment of Non-Action Oriented BR
2.3. Statistical Analysis
2.3.1. Comparison Analyses among Age Groups
2.3.2. Correlation Analyses between BR and Interoceptive Sensibility
2.3.3. Moderation Analysis
3. Results
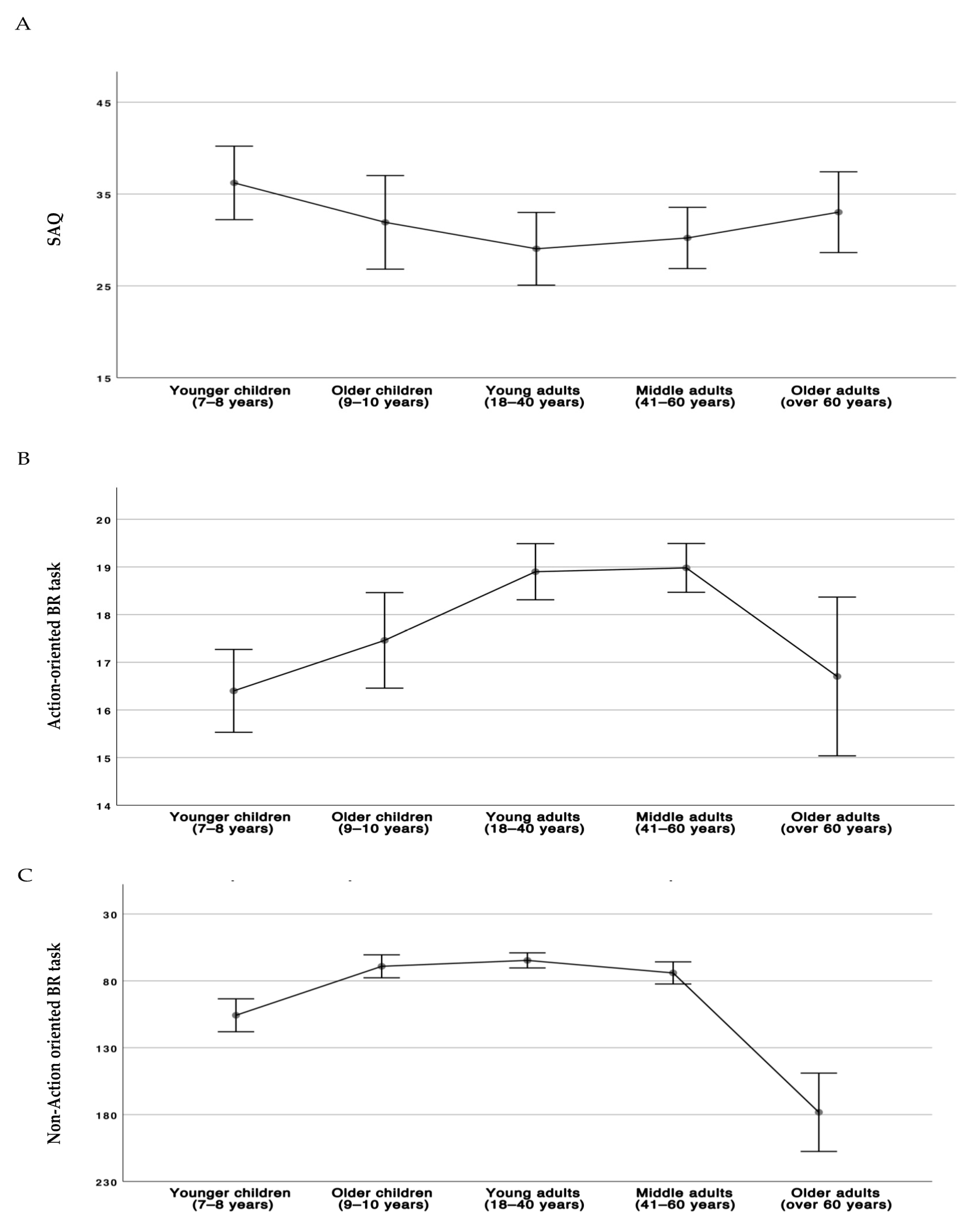
3.1. Comparison Analyses among Age Groups
3.2. Correlation Analyses between BR and Interoceptive Sensibility
3.3. Moderation Role of Interoceptive Sensibility in the Relation between Age and BR
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Azañón, E.; Tamè, L.; Maravita, A.; Linkenauger, S.A.; Ferrè, E.R.; Tajadura-Jiménez, A.; Longo, M. Multimodal contributions to body representation. Multisens. Res. 2016, 29, 635–661. [Google Scholar] [CrossRef]
- De Vignemont, F. Embodiment, ownership and disownership. Conscious. Cogn. 2011, 20, 82–93. [Google Scholar] [CrossRef] [PubMed]
- Karnath, H.O.; Baier, B. Right insula for our sense of limb ownership and self-awareness of actions. Brain Struct. Funct. 2010, 214, 411–417. [Google Scholar] [CrossRef] [PubMed]
- Palermo, L.; Di Vita, A.; Piccardi, L.; Traballesi, M.; Guariglia, C. Bottom-up and topdown processes in body representation: A study of brain-damaged and amputee patients. Neuropsychology 2014, 28, 772–781. [Google Scholar] [CrossRef]
- Boccia, M.; Raimo, S.; Di Vita, A.; Battisti, A.; Matano, A.; Guariglia, C.; Grossi, D.; Palermo, L. Topological and hodological aspects of body representation in right brain damaged patients. Neuropsychologia 2020, 148, 107637. [Google Scholar] [CrossRef]
- Di Vita, A.; Palermo, L.; Boccia, M.; Guariglia, C. Topological map of the body in post-stroke patients: Lesional and hodological aspects. Neuropsychology 2019, 33, 499–507. [Google Scholar] [CrossRef]
- Raimo, S.; Boccia, M.; Di Vita, A.; Iona, T.; Cropano, M.; Ammendolia, A.; Colao, R.; Angelillo, V.; Maiorino, A.; Guariglia, C.; et al. Body Representation Alterations in Patients with Unilateral Brain Damage. J. Int. Neuropsychol. Soc. 2021, 1–13. [Google Scholar] [CrossRef]
- Razmus, M. Body representation in patients after vascular brain injuries. Cogn. Process. 2017, 18, 359–373. [Google Scholar] [CrossRef]
- Schwoebel, J.; Coslett, H.B. Evidence for multiple, distinct representations of the human body. J. Cogn. Neurosci. 2005, 17, 543–553. [Google Scholar] [CrossRef]
- Di Vita, A.; Boccia, M.; Palermo, L.; Guariglia, C. To move or not to move, that is the question! Body schema and non-action oriented body representations: An fMRI meta-analytic study. Neurosci. Biobehav. Rev. 2016, 68, 37–46. [Google Scholar] [CrossRef]
- Corradi-Dell’Acqua, C.; Tomasino, B.; Fink, G.R. What is the position of an arm relative to the body? Neural correlates of body schema and body structural description. J. Neurosci. 2009, 29, 4162–4171. [Google Scholar] [CrossRef]
- Adams, I.L.; Lust, J.M.; Wilson, P.H.; Steenbergen, B. Development of motor imagery and anticipatory action planning in children with developmental coordination disorder—A longitudinal approach. Hum. Mov. Sci. 2017, 55, 296–306. [Google Scholar] [CrossRef]
- Butson, M.L.; Hyde, C.; Steenbergen, B.; Williams, J. Assessing motor imagery using the hand rotation task: Does performance change across childhood? Hum. Mov. Sci. 2014, 35, 50–65. [Google Scholar] [CrossRef]
- Fontes, P.L.B.; Cruz, T.K.F.; Souto, D.O.; Moura, R.; Haase, V.G. Body representation in children with hemiplegic cerebral palsy. Child Neuropsychol. 2017, 23, 838–863. [Google Scholar] [CrossRef]
- Heron, M.; Slaughter, V. Toddlers’ categorization of typical and scrambled dolls and cars. Infant. Behav. Dev. 2008, 31, 374–385. [Google Scholar] [CrossRef]
- Raimo, S.; Iona, T.; Di Vita, A.; Boccia, M.; Buratin, S.; Ruggeri, F.; Iosa, M.; Guariglia, C.; Grossi, D.; Palermo, L. The development of body representations in school-aged children. Appl. Neuropsychol. Child. 2019, 1–13. [Google Scholar] [CrossRef]
- Slaughter, V.; Heron, M.; Sim, S. Development of preferences for the human body shape in infancy. Cognition 2002, 85, B71–B81. [Google Scholar] [CrossRef]
- Cholewiak, R.W.; Collins, A.A. Vibrotactile localization on the arm: Effects of place, space, and age. Percept. Psychophys. 2003, 65, 1058–1077. [Google Scholar] [CrossRef]
- Personnier, P.; Paizis, C.; Ballay, Y.; Papaxanthis, C. Mentally represented motor actions in normal aging: II. The influence of the gravito-inertial context on the duration of overt and covert arm movements. Behav. Brain Res. 2008, 186, 273–283. [Google Scholar] [CrossRef]
- Raimo, S.; Boccia, M.; Di Vita, A.; Cropano, M.; Guariglia, C.; Grossi, D.; Palermo, L. The Body Across Adulthood: On the Relation Between Interoception and Body Representations. Front. Neurosci. 2021, 15, 586684. [Google Scholar] [CrossRef]
- Sorrentino, G.; Franza, M.; Zuber, C.; Blanke, O.; Serino, A.; Bassolino, M. How ageing shapes body and space representations: A comparison study between healthy young and older adults. Cortex 2020, 136, 56–76. [Google Scholar] [CrossRef]
- Fuelscher, I.; Williams, J.; Hyde, C. Developmental improvements in reaching correction efficiency are associated with an increased ability to represent action mentally. J. Exp. Child Psychol. 2015, 140, 74–91. [Google Scholar] [CrossRef] [PubMed]
- Cardinali, L.; Serino, A.; Gori, M. Hand size underestimation grows during childhood. Sci. Rep. 2019, 9, 13191. [Google Scholar] [CrossRef]
- de Haan, A.M.; Smit, M.; Van der Stigchel, S.; Keyner, S.A.; Dijkerman, H.C. Body representation does not lag behind in updating for the pubertal growth spurt. J. Exp. Child Psychol. 2018, 175, 48–66. [Google Scholar] [CrossRef]
- Saimpont, A.; Pozzo, T.; Papaxanthis, C. Aging affects the mental rotation of left and right hands. PLoS ONE 2009, 4, e6714. [Google Scholar] [CrossRef]
- Skoura, X.; Personnier, P.; Vinter, A.; Pozzo, T.; Papaxanthis, C. Decline in motor prediction in elderly subjects: Right versus left arm differences in mentally simulated motor actions. Cortex 2008, 44, 1271–1278. [Google Scholar] [CrossRef]
- Fanghella, M.; Era, V.; Candidi, M. Interpersonal Motor Interactions Shape Multisensory Representations of the Peripersonal Space. Brain Sci. 2021, 11, 255. [Google Scholar] [CrossRef]
- Palermo, L.; Piccardi, L.; Nori, R.; Giusberti, F.; Guariglia, C. The impact of ageing and gender on visual mental imagery processes: A study of performance on tasks from the Complete Visual Mental Imagery Battery (CVMIB). J. Clin. Exp. Neuropsychol. 2016, 38, 752–763. [Google Scholar] [CrossRef]
- Badoud, D.; Tsakiris, M. From the body’s viscera to the body’s image: Is there a link between interoception and body image concerns? Neurosci. Biobehav. Rev. 2017, 77, 237–246. [Google Scholar] [CrossRef]
- Khalsa, S.S.; Adolphs, R.; Cameron, O.G.; Critchley, H.D.; Davenport, P.W.; Feinstein, J.S.; Feusner, J.D.; Garfinkel, S.N.; Lane, R.D.; Mehling, W.E. Interoception and mental health: A roadmap. Biol. Psychiatry Cogn. Neurosci. Neuroimaging 2018, 3, 501–513. [Google Scholar] [CrossRef]
- Filippetti, M.L.; Tsakiris, M. Heartfelt embodiment: Changes in body-ownership and self-identification produce distinct changes in interoceptive accuracy. Cognition 2017, 159, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, K.; Garfinkel, S.N.; Critchley, H.D.; Seth, A.K. Multisensory integration across exteroceptive and interoceptive domains modulates self-experience in the rubber-hand illusion. Neuropsychologia 2013, 51, 2909–2917. [Google Scholar] [CrossRef] [PubMed]
- Tsakiris, M. The multisensory basis of the self: From body to identity to others. Q. J. Exp. Psychol. 2017, 70, 597–609. [Google Scholar] [CrossRef] [PubMed]
- Di Vita, A.; Cinelli, M.C.; Raimo, S.; Boccia, M.; Buratin, S.; Gentili, P.; Inzitari, M.T.; Iona, T.; Iosa, M.; Morelli, D. Body Representations in Children with Cerebral Palsy. Brain Sci. 2020, 10, 490. [Google Scholar] [CrossRef]
- Gori, M.; Del Viva, M.; Sandini, G.; Burr, D.C. Young children do not integrate visual and haptic form information. Curr. Biol. 2008, 18, 694–698. [Google Scholar] [CrossRef]
- Gori, M.; Giuliana, L.; Sandini, G.; Burr, D. Visual size perception and haptic calibration during development. Dev. Sci. 2012, 15, 854–862. [Google Scholar] [CrossRef]
- Besedeš, T.; Deck, C.; Sarangi, S.; Shor, M. Age Effects and Heuristics in Decision Making. Rev Econ. Stat. 2012, 94, 580–595. [Google Scholar] [CrossRef]
- Noh, J.W.; Kim, J.; Park, J.; Kim, H.J.; Kwon, Y.D. Gender Difference in Relationship between Health-Related Quality of Life and Work Status. PLoS ONE. 2015, 10, e0143579. [Google Scholar] [CrossRef]
- Brown, L.A. Spatial-Sequential Working Memory in Younger and Older Adults: Age Predicts Backward Recall Performance within Both Age Groups. Front. Psychol. 2016, 7, 1514. [Google Scholar] [CrossRef]
- Choudhury, S.; Charman, T.; Bird, V.; Blakemore, S.J. Adolescent development of motor imagery in a visually guided pointing task. Conscious. Cogn. 2007, 16, 886–896. [Google Scholar] [CrossRef]
- Choudhury, S.; Charman, T.; Bird, V.; Blakemore, S.J. Development of action representation during adolescence. Neuropsychologia 2007, 45, 255–262. [Google Scholar] [CrossRef]
- Raven, J.C. Progressive Matrices 1947; Series A, AB; B.H.K. Levis & Co. Ltd.: London, UK, 1947. [Google Scholar]
- Belacchi, C.; Scalisi, T.G.; Cannoni, E.; Cornoldi, C. Manuale CPM. Coloured Progressive Matrices. Standardizzazione Italiana [CPM Handbook. Colored Progressive Matrices. Italian Standardization]; Giunti, O.S.: Florence, Italy, 2008. [Google Scholar]
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 5th ed.; American Psychiatric Association: Washington, DC, USA, 2013. [Google Scholar]
- Folstein, M.F.; Folstein, S.E.; McHugh, P.R. “Mini-mental state”. A practical method for grading the cognitive state of patients for the clinician. J. Psychiatr. Res. 1975, 12, 189–198. [Google Scholar] [CrossRef]
- Magni, E.; Binetti, G.; Bianchetti, A.; Rozzini, R.; Trabucchi, M. Mini-Mental State Examination: A normative study in Italian elderly population. Eur. J. Neurol. 1996, 3, 198–202. [Google Scholar] [CrossRef]
- Basso, A.; Capitani, E.; Laiacona, M. Raven’s coloured progressive matrices: Normative values on 305 adults normal controls. Funct. Neurol. 1987, 2, 189–194. [Google Scholar]
- Longarzo, M.; D’Olimpio, F.; Chiavazzo, A.; Santangelo, G.; Trojano, L.; Grossi, D. The relationships between interoception and alexithymic trait. The Self-Awareness Questionnaire in healthy subjects. Front. Psychol. 2015, 6, 1149. [Google Scholar] [CrossRef]
- Mehling, W.E.; Acree, M.; Stewart, A.; Silas, J.; Jones, A. The multidimensional assessment of interoceptive awareness, version 2 (MAIA-2). PLoS ONE 2018, 13, e0208034. [Google Scholar] [CrossRef]
- Parsons, L.M. Imagined spatial transformations of one’s hands and feet. Cogn. Psychol. 1987, 19, 178–241. [Google Scholar] [CrossRef]
- Daurat-Hmeljiak, C.; Stambak, M.; Berges, J. Il Test Dello Schema Corporeo. Una Prova di Conoscenza e Costruzione Dell’immagine del Corpo. [The Body Schema Test. A Test of Knowledge and Construction of Body Image]; Giunti, O.S.: Florence, Italy, 1978. [Google Scholar]
- Hayes, A.F. Introduction to Mediation, Moderation, and Conditional Process Analysis: A Regression—Based Approach; Guildford Press: New York, NY, USA, 2013. [Google Scholar]
- Kail, R.; Salthouse, T.A. Processing speed as a mental capacity. Acta Psychol. 1994, 86, 199–225. [Google Scholar] [CrossRef]
- Zelazo, P.D.; Craik, F.I.; Booth, L. Executive function across the life span. Acta Psychol. 2004, 115, 167–183. [Google Scholar] [CrossRef]
- Fuelscher, I.; Williams, J.; Wilmut, K.; Enticott, P.G.; Hyde, C. Modeling the Maturation of Grip Selection Planning and Action Representation: Insights from Typical and Atypical Motor Development. Front. Psychol. 2016, 7, 108. [Google Scholar] [CrossRef]
- Berlucchi, G.; Aglioti, S. The body in the brain: Neural bases of corporeal awareness. Trends Neurosci. 1997, 20, 560–564. [Google Scholar] [CrossRef]
- Tsakiris, M.; Jiménez, A.T.; Costantini, M. Just a heartbeat away from one’s body: Interoceptive sensitivity predicts malleability of body-representations. Proc. Biol. Sci. 2011, 278, 2470–2476. [Google Scholar] [CrossRef] [PubMed]
- Spaccasassi, C.; Frigione, I.; Maravita, A. Bliss in and Out of the Body: The (Extra) Corporeal Space is Impervious to Social Pleasant Touch. Brain Sci. 2021, 11, 225. [Google Scholar] [CrossRef] [PubMed]
- Ardizzi, M.; Ferri, F. Interoceptive influences on peripersonal space boundary. Cognition 2018, 177, 79–86. [Google Scholar] [CrossRef]
- Craig, A.D. How do you feel? Interoception: The sense of the physiological condition of the body. Nat. Rev. Neurosci. 2002, 3, 655–666. [Google Scholar] [CrossRef]
- Pitron, V.; Alsmith, A.; de Vignemont, F. How do the body schema and the body image interact? Conscious. Cogn. 2018, 65, 352–358. [Google Scholar] [CrossRef]
- Holmes, N.P.; Spence, C. The body schema and the multisensory representation(s) of peripersonal space. Cogn. Process. 2004, 5, 94–105. [Google Scholar] [CrossRef]

| Children Group Aged 7–8 Years | |||
| Action-oriented BR task | Non-action-oriented BR task | ||
| SAQ | rrho p | 0.21 0.096 | −0.02 0.852 |
| Action-oriented BR task | rrho p | - | −0.07 0.581 |
| Non-action-oriented BR task | rrho p | - | - |
| Children Group Aged 9–10 years | |||
| Action-oriented BR task | Non-action-oriented BR task | ||
| SAQ | rrho p | −0.40 * 0.015 | 0.36 * 0.028 |
| Action-oriented BR task | rrho p | - | −0.36 * 0.027 |
| Non-action-oriented BR task | rrho p | - | - |
| Healthy Participants Group Aged 18–40 | |||
| Action-oriented BR task | Non-action-oriented BR task | ||
| SAQ | rrho p | 0.11 0.456 | 0.02 0.897 |
| Action-oriented BR task | rrho p | - | −0.25 0.080 |
| Non-action-oriented BR task | rrho p | - | - |
| Healthy Participants Group Aged 41–60 | |||
| Action-oriented BR task | Non-action-oriented BR task | ||
| SAQ | rrho p | −0.01 0.924 | 0.10 0.487 |
| Action-oriented BR task | rrho p | - | −0.13 0.362 |
| Non-action-oriented BR task | rrho p | - | - |
| Healthy Participants Group Aged over 60 | |||
| Action-oriented BR task | Non-action-oriented BR task | ||
| SAQ | rrho p | −0.68 ** <0.0001 | 0.67 ** <0.0001 |
| Action-oriented BR task | rrho p | - | −0.72 ** <0.0001 |
| Non-action-oriented BR task | rrho p | - | - |
| Children Group Aged 7–8 Years | |||
| Action-oriented BR task | Non-action-oriented BR task | ||
| SAQ | rrho p | 0.21 0.094 | 0.07 0.602 |
| Action-oriented BR task | rrho p | - | 0.11 0.412 |
| Non-action-oriented BR task | rrho p | - | - |
| Children Group Aged 9–10 years | |||
| Action-oriented BR task | Non-action-oriented BR task | ||
| SAQ | rrho p | −0.44 * 0.006 | 0.05 0.733 |
| Action-oriented BR task | rrho p | - | −.23 0.164 |
| Non-action-oriented BR task | rrho p | - | - |
| Healthy Participants Group Aged 18–40 years | |||
| Action-oriented BR task | Non-action-oriented BR task | ||
| SAQ | rrho p | −0.07 0.628 | −0.02 0.913 |
| Action-oriented BR task | rrho p | - | −0.07 0.644 |
| Non-action-oriented BR task | rrho p | - | - |
| Healthy Participants Group Aged 41–60 years | |||
| Action-oriented BR task | Non-action-oriented BR task | ||
| SAQ | rrho p | −0.07 0.624 | 0.11 0.431 |
| Action-oriented BR task | rrho p | - | −0.03 0.819 |
| Non-action-oriented BR task | rrho p | - | - |
| Healthy Participants Group over 60 years | |||
| Action-oriented BR task | Non-action-oriented BR task | ||
| SAQ | rrhop | −0.65 ** <0.0001 | 0.57 * 0.001 |
| Action-oriented BR task | rrho p | - | −0.63 ** <0.0001 |
| Non-action-oriented BR task | rrho p | - | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Raimo, S.; Di Vita, A.; Boccia, M.; Iona, T.; Cropano, M.; Gaita, M.; Guariglia, C.; Grossi, D.; Palermo, L. The Body across the Lifespan: On the Relation between Interoceptive Sensibility and High-Order Body Representations. Brain Sci. 2021, 11, 493. https://doi.org/10.3390/brainsci11040493
Raimo S, Di Vita A, Boccia M, Iona T, Cropano M, Gaita M, Guariglia C, Grossi D, Palermo L. The Body across the Lifespan: On the Relation between Interoceptive Sensibility and High-Order Body Representations. Brain Sciences. 2021; 11(4):493. https://doi.org/10.3390/brainsci11040493
Chicago/Turabian StyleRaimo, Simona, Antonella Di Vita, Maddalena Boccia, Teresa Iona, Maria Cropano, Mariachiara Gaita, Cecilia Guariglia, Dario Grossi, and Liana Palermo. 2021. "The Body across the Lifespan: On the Relation between Interoceptive Sensibility and High-Order Body Representations" Brain Sciences 11, no. 4: 493. https://doi.org/10.3390/brainsci11040493
APA StyleRaimo, S., Di Vita, A., Boccia, M., Iona, T., Cropano, M., Gaita, M., Guariglia, C., Grossi, D., & Palermo, L. (2021). The Body across the Lifespan: On the Relation between Interoceptive Sensibility and High-Order Body Representations. Brain Sciences, 11(4), 493. https://doi.org/10.3390/brainsci11040493








