Heart Rate Variability and Decision-Making: Autonomic Responses in Making Decisions
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Instruments and Measures
2.2.1. Sociodemographic Interview
2.2.2. Blood Pressure and Heart Rate
2.2.3. Height and Weight
2.2.4. Heart Rate Variability
2.2.5. Global Cognitive Functioning
2.2.6. Iowa Gambling Task (IGT)
Apparatus
Stimuli
Procedure
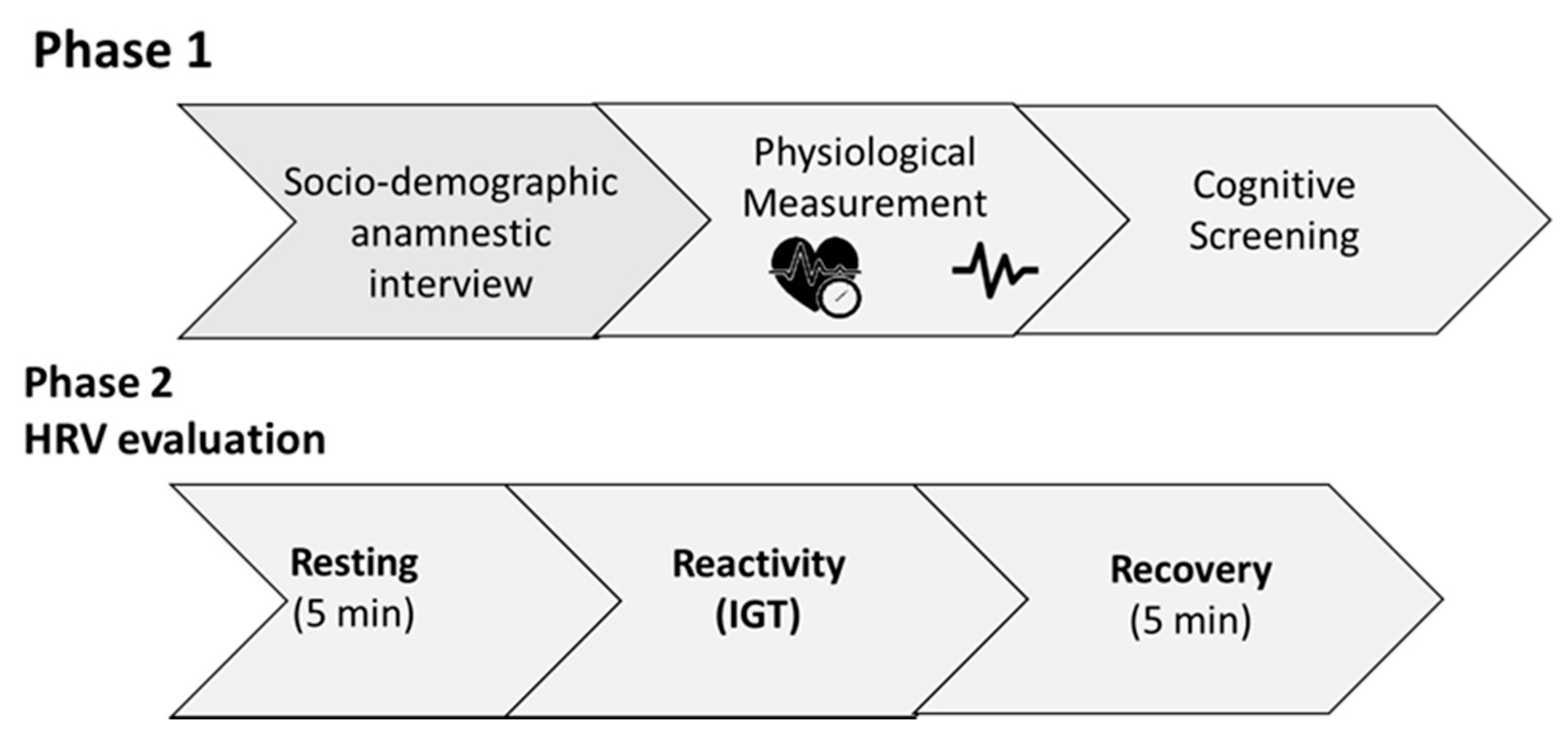
2.3. General Procedure
2.4. Data Analysis
3. Results
3.1. Resting, Reactivity, and Recovery Differences
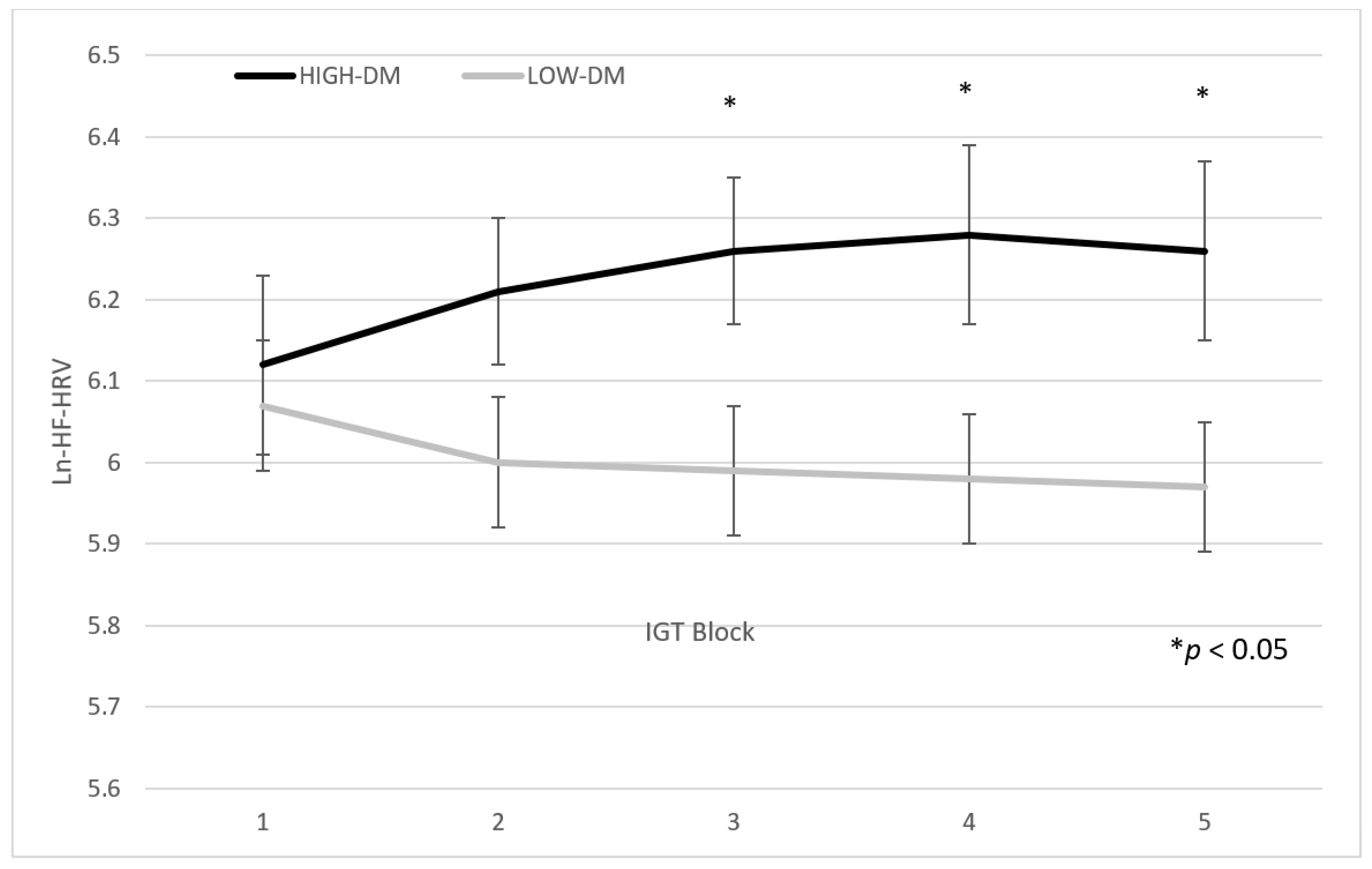
3.2. Differences in IGT Blocks
4. Discussion
Limitations and Future Perspectives
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Fellows, L.K. The Cognitive Neuroscience of Human Decision Making: A Review and Conceptual Framework. Behav. Cogn. Neurosci. Rev. 2004, 3, 159–172. [Google Scholar] [CrossRef]
- Gold, J.I.; Shadlen, M.N. The Neural Basis of Decision Making. Annu. Rev. Neurosci. 2007, 30, 535–574. [Google Scholar] [CrossRef]
- Gigerenzer, G.; Selten, R. Rethinking Rationality. In Bounded Rationality: The Adaptive Toolbox; Gigerenzer, G., Selten, R., Eds.; MIT Press: Cambridge, MA, USA, 2001; pp. 1–12. [Google Scholar]
- Mishra, S. Decision-Making under Risk: Integrating Perspectives from Biology, Economics, and Psychology. Personal. Soc. Psychol. Rev. 2014, 18, 280–307. [Google Scholar] [CrossRef]
- Thayer, J.F.; Lane, R.D. Claude Bernard and the Heart—Brain Connection: Further Elaboration of a Model of Neurovisceral Integration. Neurosci. Biobehav. Rev. 2009, 33, 81–88. [Google Scholar] [CrossRef]
- Damasio, A.R. The Somatic Marker Hypothesis and the Possible Functions of the Prefrontal Cortex. Philos. Trans. R. Soc. Lond. Ser. B Biol. Sci. 1996, 351, 1413–1420. [Google Scholar] [CrossRef]
- Bechara, A.; Damasio, A.R. The Somatic Marker Hypothesis: A Neural Theory of Economic Decision. Games Econ. Behav. 2005, 52, 336–372. [Google Scholar] [CrossRef]
- Bechara, A.; Damasio, H.; Tranel, D.; Damasio, A.R. Deciding Advantageously before Knowing the Advantageous Strategy. Science 1997, 275, 1293–1295. [Google Scholar] [CrossRef]
- Appelhans, B.M.; Luecken, L.J. Heart Rate Variability as an Index of Regulated Emotional Responding. Rev. Gen. Psychol. 2006, 10, 229–240. [Google Scholar] [CrossRef]
- Berntson, G.G.; Thomas Bigger Jr, J.; Eckberg, D.L.; Grossman, P.; Kaufmann, P.G.; Malik, M.; Nagaraja, H.N.; Porges, S.W.; Saul, J.P.; Stone, P.H.; et al. Heart Rate Variability: Origins, Methods, and Interpretive Caveats. Psychophysiology 1997, 34, 623–648. [Google Scholar] [CrossRef]
- Reyes del Paso, G.A.; Langewitz, W.; Mulder, L.J.; Van Roon, A.; Duschek, S. The Utility of Low Frequency Heart Rate Variability as an Index of Sympathetic Cardiac Tone: A Review with Emphasis on a Reanalysis of Previous Studies. Psychophysiology 2013, 50, 477–487. [Google Scholar] [CrossRef]
- Thayer, J.F.; Lane, R.D. A Model of Neurovisceral Integration in Emotion Regulation and Dysregulation. J. Affect. Disord. 2000, 61, 201–216. [Google Scholar] [CrossRef]
- Thayer, J.F.; Hansen, A.L.; Saus-Rose, E.; Johnsen, B.H. Heart Rate Variability, Prefrontal Neural Function, and Cognitive Performance: The Neurovisceral Integration Perspective on Self-Regulation, Adaptation, and Health. Ann. Behav. Med. 2009, 37, 141–153. [Google Scholar] [CrossRef]
- Porges, S.W. Orienting in a Defensive World: Mammalian Modifications of Our Evolutionary Heritage. A Polyvagal Theory. Psychophysiology; W.W. Norton & Company: New York, NY, USA, 1995; Volume 32, pp. 301–318. [Google Scholar]
- Forte, G.; Favieri, F.; Casagrande, M. Heart Rate Variability and Cognitive Function: A Systematic Review. Front. Neurosci. 2019, 13, 710. [Google Scholar] [CrossRef] [PubMed]
- Colzato, L.S.; Jongkees, B.J.; de Wit, M.; van der Molen, M.J.W.; Steenbergen, L. Variable Heart Rate and a Flexible Mind: Higher Resting-State Heart Rate Variability Predicts Better Task-Switching. Cogn. Affect. Behav. Neurosci. 2018, 18, 730–738. [Google Scholar] [CrossRef]
- Thayer, J.F.; Åhs, F.; Fredrikson, M.; Sollers III, J.J.; Wager, T.D. A Meta-Analysis of Heart Rate Variability and Neuroimaging Studies: Implications for Heart Rate Variability as a Marker of Stress and Health. Neurosci. Biobehav. Rev. 2012, 36, 747–756. [Google Scholar] [CrossRef] [PubMed]
- Bechara, A.; Damasio, A.R.; Damasio, H.; Anderson, S.W. Insensitivity to Future Consequences Following Damage to Human Prefrontal Cortex. Cognition 1994, 50, 7–15. [Google Scholar] [CrossRef]
- Laborde, S.; Mosley, E.; Mertgen, A. Vagal Tank Theory: The Three Rs of Cardiac Vagal Control Functioning—Resting, Reactivity, and Recovery. Front. Neurosci. 2018, 12, 458. [Google Scholar] [CrossRef]
- Naqvi, N.; Shiv, B.; Bechara, A. The Role of Emotion in Decision Making: A Cognitive Neuroscience Perspective. Curr. Dir. Psychol. 2006, 15, 260–264. [Google Scholar] [CrossRef]
- Bechara, A.; Damasio, H.; Damasio, A.R.; Lee, G.P. Different Contributions of the Human Amygdala and Ventromedial Prefrontal Cortex to Decision-Making. J. Neurosci. 1999, 19, 5473–5481. [Google Scholar] [CrossRef] [PubMed]
- Steingroever, H.; Wetzels, R.; Horstmann, A.; Neumann, J.; Wagenmakers, E.J. Performance of Healthy Participants on the Iowa Gambling Task. Psychol. Assess. 2013, 25, 180–193. [Google Scholar] [CrossRef]
- Bull, P.N.; Tippett, L.J.; Addis, D.R. Decision Making in Healthy Participants on the Iowa Gambling Task: New Insights from an Operant Approach. Front. Psychol. 2015, 6, 391. [Google Scholar] [CrossRef]
- Wehrwein, E.A.; Orer, H.S.; Barman, S.M. Overview of the Anatomy, Physiology, and Pharmacology of the Autonomic Nervous System. Compr. Physiol. 2016, 6, 1239–1278. [Google Scholar] [CrossRef]
- Malik, M. Heart Rate Variability: Standards of Measurement, Physiological Interpretation, and Clinical Use: Task Force of the European Society of Cardiology and the North American Society for Pacing and Electrophysiology. Ann. Noninvasive Electrocardiol. 1996, 1, 151–181. [Google Scholar] [CrossRef]
- Laborde, S.; Mosley, E.; Thayer, J.F. Heart Rate Variability and Cardiac Vagal Tone in Psychophysiological Research—Recommendations for Experiment Planning, Data Analysis, and Data Reporting. Front. Psychol. 2017, 8, 213. [Google Scholar] [CrossRef]
- Shaffer, F.; Ginsberg, J.P. An Overview of Heart Rate Variability Metrics and Norms. Front. Public Health 2017, 5, 258. [Google Scholar] [CrossRef]
- Tarvainen, M.P.; Niskanen, J.P.; Lipponen, J.A.; Ranta-Aho P., O.; Karjalainen, P.A. Kubios HRV—Heart Rate Variability Analysis Software. Comput. Methods Programs Biomed. 2014, 113, 210–220. [Google Scholar] [CrossRef]
- Folstein, M.F.; Folstein, S.E.; McHugh, P.R. “Mini-Mental state”: A Practical Method for Grading the Cognitive State of Patients for the Clinician. J. Psychiatr. Res. 1975, 12, 189–198. [Google Scholar] [CrossRef]
- Measso, G.; Cavarzeran, F.; Zappalà, G.; Lebowitz, B.D.; Crook, T.H.; Pirozzolo, F.J.; Amaducci, L.A.; Massari, D.; Grigoletto, F. The Mini-Mental State Examination: Normative Study of an Italian Random Sample. Dev. Neuropsychol. 1993, 9, 77–85. [Google Scholar] [CrossRef]
- Bechara, A.; Tranel, D.; Damasio, H. Characterization of the Decision-Making Deficit of Patients with Ventromedial Prefrontal Cortex Lesions. Brain 2000, 123, 2189–2202. [Google Scholar] [CrossRef] [PubMed]
- Williams, B.; Mancia, G.; Spiering, W.; Agabiti Rosei, E.; Azizi, M.; Burnier, M.; Clement, D.L.; Coca, A.; de Simone, G.; Dominiczak, A.; et al. 2018 ESC/ESH Guidelines for the Management of Arterial Hypertension. Eur. Heart J. 2018, 39, 3021–3104. [Google Scholar] [CrossRef] [PubMed]
- Drucaroff, L.J.; Kievit, R.; Guinjoan, S.M.; Gerschcovich, E.R.; Cerquetti, D.; Leiguarda, R.; Cardinali, D.P.; Vigo, D.E. Higher Autonomic Activation Predicts Better Performance in Iowa Gambling Task. Cogn. Behav. Neurol. 2011, 24, 93–98. [Google Scholar] [CrossRef] [PubMed]
- Alacreu-Crespo, A.; Costa, R.; Abad-Tortosa, D.; Salvador, A.; Serrano, M.Á. Good Decision-Making Is Associated with an Adaptive Cardiovascular Response to Social Competitive Stress. Stress 2018, 21, 528–537. [Google Scholar] [CrossRef] [PubMed]
- Serra, L.; Bruschini, M.; Ottaviani, C.; Di Domenico, C.; Fadda, L.; Caltagirone, C.; Cercignani, M.; Carlesimo, G.A.; Bozzali, M. Thalamocortical Disconnection Affects the Somatic Marker and Social Cognition: A Case Report. Neurocase 2019, 25, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Bechara, A. Neurobiology of Decision-Making: Risk and Reward. Semin. Clin. Neuropsychiatry 2001, 6, 205–216. [Google Scholar] [CrossRef] [PubMed]
- Lane, R.D.; McRae, K.; Reiman, E.M.; Chen, K.; Ahern, G.L.; Thayer, J.F. Neural Correlates of Heart Rate Variability during Emotion. Neuroimage 2009, 44, 213–222. [Google Scholar] [CrossRef]
- Park, G.; Vasey, M.; VanBavel, J.J.; Thayer, J.F. Cardiac Vagal Tone Is Cor-Related with Selective Attention to Neutral Distractors under Load. Psychophysiology 2013, 50, 398–406. [Google Scholar] [CrossRef]
- Hansen, A.L.; Johnsen, B.H.; Thayer, J.F. Vagal Influence on Working Memory and Attention. Int. J. Psychophysiol. 2003, 48, 263–274. [Google Scholar] [CrossRef]
- Hansen, A.L.; Johnsen, B.H.; Sollers, J.J.; Stenvik, K.; Thayer, J.F. Heart Rate Variability and Its Relation to Prefrontal Cognitive Function: The Effects of Training and Detraining. Eur. J. Appl. Physiol. 2004, 93, 263–272. [Google Scholar] [CrossRef] [PubMed]
- Britton, A.; Singh-Manoux, A.; Hnatkova, K.; Malik, M.; Marmot, M.G.; Shipley, M. The Association between Heart Rate Variability and Cognitive Impairment in Middle-Aged Men and Women. The Whitehall II Cohort Study. Neuroepidemiology 2008, 31, 115–121. [Google Scholar] [CrossRef]
- Colzato, L.S.; Steenbergen, L. High Vagally Mediated Restingstate Heart Rate Variability Is Associated with Superior Action Cascading. Neuropsychologia 2017, 106, 1–6. [Google Scholar] [CrossRef]
- Davidson, R.J.; Jackson, D.C.; Kalin, N.H. Emotion, Plasticity, Context, and Regulation: Perspectives from Affective Neuroscience. Psychol. Bull. 2000, 126, 890. [Google Scholar] [CrossRef]
- Bechara, A.; Damasio, H.; Tranel, D.; Damasio, A.R. The Iowa Gambling Task and the Somatic Marker Hypothesis: Some Questions and Answers. Trends Cogn. Sci. 2005, 9, 159–162. [Google Scholar] [CrossRef]
- Guarino, A.; Favieri, F.; Boncompagni, I.; Agostini, F.; Cantone, M.; Casagrande, M. Executive Functions in Alzheimer Disease: A Systematic Review. Front. Aging Neurosci. 2019, 10, 437. [Google Scholar] [CrossRef]
- Guarino, A.; Forte, G.; Giovannoli, J.; Casagrande, M. Executive Functions in the Elderly with Mild Cognitive Impairment: A Systematic Review on Motor and Cognitive Inhibition, Conflict Control and Cognitive Flexibility. Aging Ment. Health 2019, 24, 1028–1045. [Google Scholar] [CrossRef] [PubMed]
- Bourdillon, N.; Schmitt, L.; Yazdani, S.; Vesin, J.M.; Millet, G.P. Minimal Window Duration for Accurate HRV Recording in Athletes. Front. Neurosci. 2017, 11, 456. [Google Scholar] [CrossRef]
- Overman, W.H.; Pierce, A. Iowa Gambling Task with Non-Clinical Participants: Effects of Using Real + Virtual Cards and Additional Trials. Front. Psychol. 2013, 4, 935. [Google Scholar] [CrossRef]
- Forte, G.; Casagrande, M. Effects of Blood Pressure on Cognitive Performance in Aging: A Systematic Review. Brain Sci. 2020, 10, 919. [Google Scholar] [CrossRef]
- Forte, G.; de Pascalis, V.; Favieri, F.; Casagrande, M. Effects of Blood Pressure on Cognitive Performance: A Systematic Review. J. Clin. Med. 2020, 9, 34. [Google Scholar] [CrossRef]
- Favieri, F.; Casagrande, M. The Executive Functions in Overweight and Obesity: A Systematic Review of Neuropsychological Cross-Sectional and Longitudinal Studies. Front. Psychol. 2019, 10, 2126. [Google Scholar] [CrossRef]
- Geisler, F.C.M.; Vennewald, N.; Kubiak, T.; Weber, H. The Impact of Heart Rate Variability on Subjective Well-Being Is Mediated by Emotion Regulation. Personal. Individ. Differ. 2010, 49, 723–728. [Google Scholar] [CrossRef]
- Quintana, D.S.; Guastella, A.J.; Outhred, T.; Hickie, I.B.; Kemp, A.H. Heart Rate Variability Is Associated with Emotion Recognition: Direct Evidence for a Relationship Between the Autonomic Nervous System and Social Cognition. Int. J. Psychophysiol. 2010, 86, 168–172. [Google Scholar] [CrossRef]
- Kemp, A.H.; Quintana, D.S.; Kuhnert, R.L.; Griffiths, K.; Hickie, I.B.; Guastella, A.J. Oxytocin Increases Heart Rate Variability in Humans at Rest: Implications for Social Approach-Related Motivation and Capacity for Social Engagement. PLoS ONE 2012, 7, e44014. [Google Scholar] [CrossRef]
- Park, G.; Thayer, J.F. From the Heart to the Mind: Cardiac Vagal Tone Modulates Top-Down and Bottom-Up Visual Perception And Attention to Emotional Stimuli. Front. Psychol. 2014, 5, 278. [Google Scholar] [CrossRef]
- Schroeder, E.B.; Liao, D.; Chambless, L.E.; Prineas, R.J.; Evans, G.W.; Heiss, G. Hypertension, Blood Pressure, and Heart Rate Variability: The Atherosclerosis Risk in Communities (ARIC) Study. Hypertension 2003, 42, 1106–1111. [Google Scholar] [CrossRef] [PubMed]
- Schroeder, E.B.; Chambless, L.E.; Liao, D.; Prineas, R.J.; Evans, G.W.; Rosamond, W.D.; Heiss, G. Diabetes, Glucose, Insulin, and Heart Rate Variability: The Atherosclerosis Risk in Communities (ARIC) Study. Diabetes Care 2005, 28, 668–674. [Google Scholar] [CrossRef] [PubMed]
- Buccelletti, E.; Gilardi, E.M.A.N.; Scaini, E.; Galiuto, L.E.O.N.; Persiani, R.O.B.E.; Biondi, A.L.B.E.; Basile, F.L.O.R.; Silveri, N.G. Heart Rate Variability and Myocardial Infarction: Systematic Literature Review and Metanalysis. Eur. Rev. Med. Pharmacol. Sci. 2009, 13, 299–307. [Google Scholar]
| High DM (n = 79) | Low DM (n = 51) | F/X2 | p | η2 | |
|---|---|---|---|---|---|
| Sex | |||||
| Male | 33 | 18 | <1 | 0.46 | |
| Female | 46 | 33 | |||
| Age | 23.2 (2.4) | 23.5 (2.6) | <1 | 0.49 | 0.004 |
| Years of education | 16.4 (1.8) | 16.1 (1.8) | <1 | 0.33 | 0.007 |
| BMI | 22.4 (3.2) | 22.6 (3.5) | <1 | 0.75 | 0.0001 |
| SBP | 116.7 (10.9) | 116.9 (11.1) | <1 | 0.93 | 0.0001 |
| DBP | 71.1 (7.6) | 72.5 (8.2) | 1.02 | 0.31 | 0.008 |
| HR | 74.3 (11.3) | 80.3 (13.9) | 6.9 | 0.009 | 0.05 |
| Smoke (cigarettes per die) | 3.3 (4.9) | 2.8 (5.4) | <1 | 0.54 | 0.003 |
| Coffee (per die) | 1.8 (1.5) | 1.7 (1.5) | <1 | 0.67 | 0.001 |
| MMSE score | 29.7 (0.5) | 29.6 (0.7) | 1.1 | 0.29 | 0.009 |
| SPM score | 49.16 (5.8) | 45.2 (8.30) | 7.99 | 0.006 | 0.07 |
| IOWA score | 0.20 (0.22) | −0.16 (0.13) | 103.52 | <0.001 | 0.44 |
| High DM (n = 79) | Low DM (n = 51) | F | p | η2 | |
|---|---|---|---|---|---|
| ln(HF baseline) | 6.40 (0.55) | 6.10 (0.54) | 9.50 | 0.003 | 0.06 |
| ln(HF total IOWA) | 6.23 (0.61) | 6.00 (0.63) | 3.75 | 0.04 | 0.02 |
| ln(HF recovery) | 6.30 (0.58) | 6.08 (0.56) | 4.61 | 0.03 | 0.03 |
| High DM (n = 79) | Low DM (n = 51) | F | p | η2 | |
|---|---|---|---|---|---|
| ln(HF-HRV) IGT first block | 6.12 (0.56) | 6.07 (0.71) | <1 | 0.73 | 0.001 |
| ln(HF-HRV) IGT second block | 6.21 (0.55) | 6.00 (0.70) | 3.34 | 0.07 | 0.02 |
| ln(HF-HRV) IGT third block | 6.26 (0.46) | 5.99 (0.63) | 7.80 | 0.006 | 0.05 |
| ln(HF-HRV) IGT fourth block | 6.28 (0.59) | 5.98 (0.69) | 6.94 | 0.009 | 0.05 |
| ln(HF-HRV) IGT fifth block | 6.26 (0.56) | 5.97 (0.70) | 6.53 | 0.01 | 0.05 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Forte, G.; Morelli, M.; Casagrande, M. Heart Rate Variability and Decision-Making: Autonomic Responses in Making Decisions. Brain Sci. 2021, 11, 243. https://doi.org/10.3390/brainsci11020243
Forte G, Morelli M, Casagrande M. Heart Rate Variability and Decision-Making: Autonomic Responses in Making Decisions. Brain Sciences. 2021; 11(2):243. https://doi.org/10.3390/brainsci11020243
Chicago/Turabian StyleForte, Giuseppe, Matteo Morelli, and Maria Casagrande. 2021. "Heart Rate Variability and Decision-Making: Autonomic Responses in Making Decisions" Brain Sciences 11, no. 2: 243. https://doi.org/10.3390/brainsci11020243
APA StyleForte, G., Morelli, M., & Casagrande, M. (2021). Heart Rate Variability and Decision-Making: Autonomic Responses in Making Decisions. Brain Sciences, 11(2), 243. https://doi.org/10.3390/brainsci11020243






