Is Developmental Dyslexia Due to a Visual and Not a Phonological Impairment?
Abstract
1. Introduction
2. Origin of the Magnocellular, Parvocellular, and Koniocellular Pathways in the Retina
3. Neural Wiring in the Visual Cortex
4. Two Visual Systems
5. Processing of Motion, Contrast, and Patterns in the Dorsal and Ventral Streams
6. Is DD Due to Impairment of M-Cells?
7. Is DD Due to an Impairment of Visual Processing?
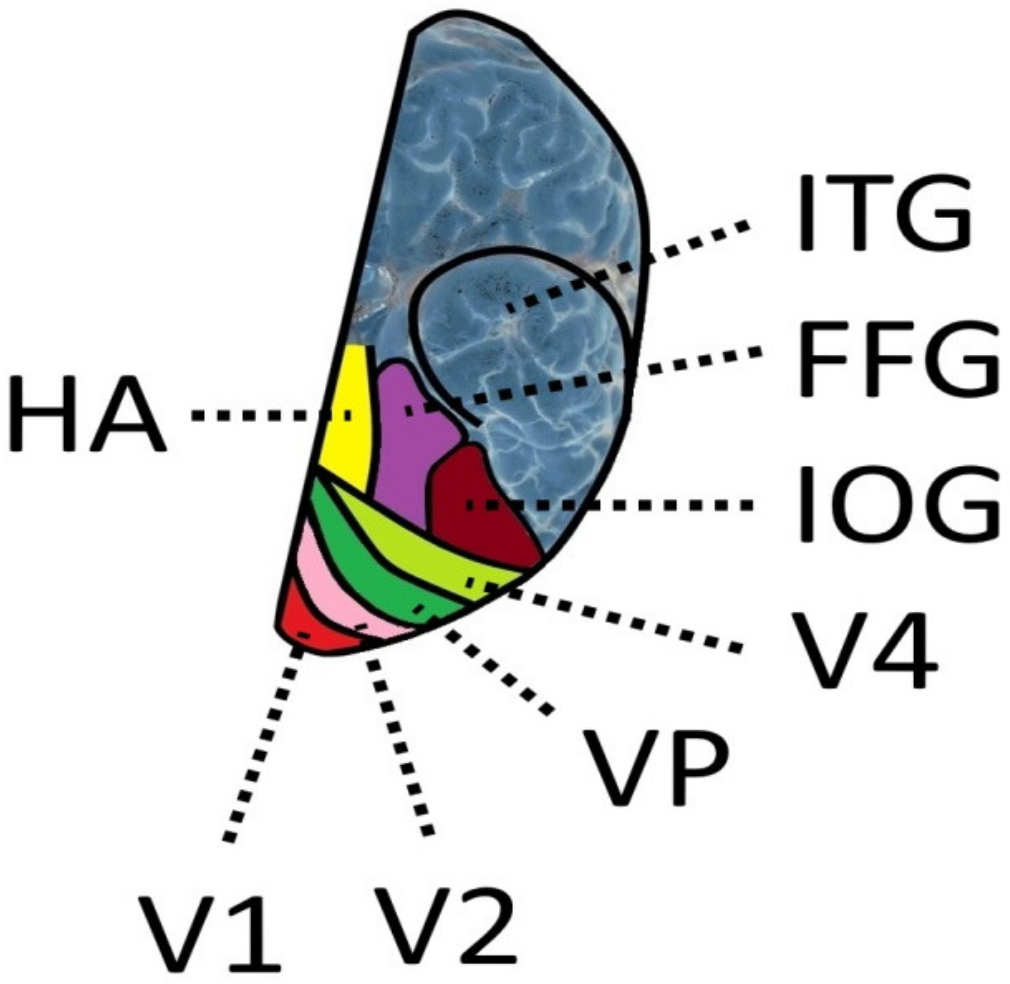
8. The Role of the Fusiform Gyrus and the Visual Word Form Area
8.1. Anatomical Background
8.2. Is There a Specialization for the Recognition of Words?
8.3. No Priority for Words over Pseudowords
8.4. The VWFA Processes All Kinds of Stimuli
8.5. A Hierarchy of Visual Processing
8.6. Reading after Damage to the VWFA
9. The Neurobiological Basis of Grapheme to Phoneme Association
10. Summary and Conclusions
Funding
Conflicts of Interest
Appendix A. Causation
Appendix B. Developmental Dyslexia and Hemianopic Reading Disturbance
Appendix C. Differences between Developmental Dyslexia and Simultanagnosia
References
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, DSM 5; APA: Washington, DC, USA; London, UK, 2013; p. 67. [Google Scholar]
- Wagner, R.K.; Torgesen, J.K. The nature of phonological processing and its causal role in the acquisition of reading skills. Psychol. Bull. 1987, 101, 192–212. [Google Scholar] [CrossRef]
- Bruce, D. An analysis of word sounds by young children. Br. J. Educ. Psychol. 1964, 34, 158–170. [Google Scholar] [CrossRef]
- Bryant, P.E.; MacLean, M.; Bradley, L.L.; Crossland, J. Rhyme and alliteration, phoneme detection, and learning to read. Dev. Psychol. 1990, 26, 429–438. [Google Scholar] [CrossRef]
- Torgesen, J.K.; Wagner, R.K.; Rashotte, C.A. Longitudinal studies of phonological processing and reading. J. Learn. Disabil. 1994, 27, 276–286. [Google Scholar] [CrossRef]
- McBride-Chang, C. What is phonological awareness? J. Educ. Psychol. 1995, 87, 179–192. [Google Scholar] [CrossRef]
- Yopp, H.K. A test for assessing phonemic awareness in young children. Read. Teach. 1995, 49, 20–29. [Google Scholar] [CrossRef]
- Torgesen, J.K.; Wagner, R.K.; Rashotte, C.A. Prevention and remediation of severe reading disabilities: Keeping the end in mind. Sci. Stud. Read. 1997, 1, 217–234. [Google Scholar] [CrossRef]
- Muter, V.; Hulme, C.; Snowling, M.J.; Taylor, S. Segmentation, not rhyming, predicts early progress in learning to read. J. Exp. Child. Psychol. 1998, 71, 3–27. [Google Scholar] [CrossRef]
- Ehri, L.C. Research on learning to read and spell: A personal historical perspective. Sci. Stud. Read. 1998, 2, 97–114. [Google Scholar] [CrossRef]
- Goswami, U. Phonology, reading development, and dyslexia: A cross-linguistic perspective. Ann. Dyslexia 2002, 52, 139–163. [Google Scholar] [CrossRef]
- Goswami, U. Phonology, reading and reading difficulties. In Interdisciplinary Perspectives on Learning to Read. Culture, Cognition and Pedagogy; Hall, K., Goswami, U., Harrison, C., Ellis, S., Soler, J., Eds.; Routhledge: Abingdon, UK, 2010; pp. 103–116. [Google Scholar]
- Snowling, M.J. From language to reading and dyslexia. Dyslexia 2001, 7, 37–46. [Google Scholar] [CrossRef]
- Vellutino, F.R.; Fletcher, J.M.; Snowling, M.J.; Scanlon, D.M. Specific reading disability (dyslexia): What have we learned in the past four decades. J. Child. Psychol. Psychiatry 2004, 45, 2–40. [Google Scholar] [CrossRef] [PubMed]
- Ziegler, J.C.; Goswami, U. Reading acquisition, developmental dyslexia, and skilled reading across languages: A psycholinguistic grain size theory. Psychol. Bull. 2005, 131, 3–29. [Google Scholar] [CrossRef] [PubMed]
- Peterson, R.L.; Pennington, B.F. Developmental dyslexia. Lancet 2012, 26, 1997–2007. [Google Scholar] [CrossRef]
- Liberman, I.Y.; Shankweiler, D.; Fischer, F.W.; Carter, B. Explicit syllable and phoneme segmentation in the young child. J. Exp. Child. Psychol. 1974, 18, 201–212. [Google Scholar] [CrossRef]
- Kaminski, R.A.; Good, R.H. Toward a technology for assessing basic early literacy skills. Sch. Psychol. Rev. 1996, 25, 215–227. [Google Scholar] [CrossRef]
- Nation, K.; Hulme, C. Phonemic segmentation, not onset-rime segmentation, predicts early reading and spelling skills. Read. Res. Quart. 1997, 32, 154–167. [Google Scholar] [CrossRef]
- Tallal, P. Auditory temporal perception, phonics, and reading disabilities in children. Brain Lang. 1980, 9, 182–198. [Google Scholar] [CrossRef]
- Geiger, G.; Lettvin, J.Y. Peripheral vision in persons with dyslexia. N. Engl. J. Med. 1987, 316, 1238–1243. [Google Scholar] [CrossRef]
- Atkinson, J. Review of human visual development: Crowding and dyslexia. Vis. Vis. Dyslexia 1991, 13, 44–57. [Google Scholar]
- Galaburda, A.; Livingstone, M. Evidence for a magnocellular defect in developmental dyslexia. Ann. N. Y. Acad. Sci. 1993, 682, 70–82. [Google Scholar] [CrossRef]
- Tallal, P.; Miller, S.; Fitch, R.H. Neurobiological basis of speech: A case for the pre-eminence of temporal processing. Ann. N. Y. Acad. Sci. 1993, 682, 27–47. [Google Scholar] [CrossRef] [PubMed]
- Farah, M.; Stowe, R.M.; Levinson, K.L. Pholological dyslexia. Loss of a reading specific component of cognitive architecture? Cogn. Neuropsychol. 1996, 13, 849–868. [Google Scholar] [CrossRef]
- Nagarajan, S.; Mahnke, H.; Salz, T.; Tallal, P.; Roberts, T.; Merzenich, M.M. Cortical auditory signal processing in poor readers. Proc. Natl. Acad. Sci. USA 1999, 96, 6483–6488. [Google Scholar] [CrossRef]
- Hornickel, J.; Kraus, N. Unstable representation of sound: A biological marker of dyslexia. J. Neurosci. 2013, 33, 3500–3504. [Google Scholar] [CrossRef] [PubMed]
- Biscaldi, M.; Fischer, B.; Hartnegg, K. Voluntary saccadic control in dyslexia. Perception 2000, 29, 509–521. [Google Scholar] [CrossRef]
- Hari, R.; Renvall, H. Impaired processing of rapid stimulus sequences in dyslexia. Trends Cogn. Sci. 2001, 5, 525–532. [Google Scholar] [CrossRef]
- Lorusso, M.L.; Facoetti, A.; Pesenti, S.; Cattaneo, C.; Molteni, M.; Geiger, G. Wider recognition in peripheral vision common to different subtypes of dyslexia. Vis. Res. 2004, 44, 2413–2424. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Roach, N.W.; Hogben, J.H. Attentional modulation of visual processing in adult dyslexia: A spatial cuing deficit. Psychol. Sci. 2004, 15, 650–654. [Google Scholar] [CrossRef]
- Buchholz, J.; Davis, A.A. Adults with dyslexia demonstrate space-based and object-based covert attention deficits: Shifting attention to the periphery and shifting attention between objects in the left visual field. Brain Cogn. 2005, 57, 30–34. [Google Scholar] [CrossRef]
- Daniel, A.; Abrams, D.A.; Nicol, T.; Zecker, S.; Kraus, N. Abnormal cortical processing of the syllable rate of speech in poor readers. J. Neurosci. 2009, 29, 7686–7693. [Google Scholar] [CrossRef]
- Menghini, D.; Finzi, A.; Benassi, M.; Bolzani, R.; Facoetti, A.; Giovagnoli, S.; Ruffino, M.; Vicari, S. Different underlying neurocognitive deficits in developmental dyslexia: A comparative study. Neuropsychologia 2010, 48, 863–872. [Google Scholar] [CrossRef]
- Lorusso, M.L.; Cantiani, C.; Molteni, M. Age, dyslexia subtype and comorbidity modulate rapid auditory processing in developmental dyslexia. Front. Hum. Neurosci. 2014, 8, 313. [Google Scholar] [CrossRef]
- Van Bergen, E.; de Jong, P.F.; Maassen, B.; Krikhaar, E.; Plakas, A.; van der Leij, A. IQ of four-year-olds who go on to develop dyslexia. J. Learn. Disabil. 2014, 47, 475–484. [Google Scholar] [CrossRef]
- Oliveira, E.P.; Neri, M.L.; Capelatto, L.L.; Guimarães, C.A.; Guerreiro, M.M. Rolandic epilepsy and dyslexia. Arq. Neuropsiquiatry 2014, 72, 826–831. [Google Scholar] [CrossRef] [PubMed]
- Gori, S.; Seitz, A.R.; Ronconi, L.; Franceschini, S.; Facoetti, A. Multiple Causal Links between Magnocellular-Dorsal Pathway Deficit and Developmental Dyslexia. Cereb. Cortex 2016, 26, 4356–4369. [Google Scholar] [CrossRef] [PubMed]
- Hendren, R.L.; Haft, S.L.; Black, J.M.; White, N.C.; Hoeft, F. Recognizing Psychiatric Comorbidity with Reading Disorders. Front. Psychiatry 2018, 9, 101. [Google Scholar] [CrossRef]
- Cheng, D.; Xiao, Q.; Chen, Q.; Cui, J.; Zhou, X. Dyslexia and dyscalculia are characterized by common visual perception deficits. Dev. Neuropsychol. 2018, 43, 497–507. [Google Scholar] [CrossRef] [PubMed]
- Peters, L.; Bulthé, J.; Daniels, N.; de Beeck, H.O.; De Smedt, B. Dyscalculia and dyslexia: Different behavioral, yet similar brain activity profiles during arithmetic. Neuroimage Clin. 2018, 18, 663–674. [Google Scholar] [CrossRef]
- Moll, K.; Landerl, K.; Snowling, M.J.; Schulte-Körne, G. Understanding comorbidity of learning disorders: Task-dependent estimates of prevalence. J. Child. Psychol. Psychiatry 2019, 60, 286–294. [Google Scholar] [CrossRef]
- Maziero, S.; Tallet, J.; Bellocchi, S.; Jover, M.; Chaix, Y.; Jucla, M. Influence of comorbidity on working memory profile in dyslexia and developmental coordination disorder. J. Clin. Exp. Neuropsychol. 2020, 42, 660–674. [Google Scholar] [CrossRef]
- Heim, S.; Tschierse, J.; Amunts, K.; Wilms, M.; Vossel, S.; Willmes, K.; Grabowska, A.; Huber, W. Cognitive subtypes of dyslexia. Acta Neurobiol. Exp. 2008, 68, 73–82. [Google Scholar]
- Spirtes, P.; Glymour, C.; Scheines, R. Causation, Prediction, and Search; MIT Press: Cambridge, MA, USA, 1992. [Google Scholar]
- Spohn, W. Causation: An alternative. Br. J. Philos. Sci. 2006, 57, 93–119. [Google Scholar] [CrossRef]
- Pearl, J.; Glymour, M.; Jewell, N.P. Causal Inference in Statistics; Wiley: Chichester, UK, 2016. [Google Scholar]
- Pearl, J. Causality—Models, Reasoning and Inference; Cambridge University Press: Cambridge, UK; New York, NY, USA; Port Melbourne, Australia, 2018; pp. 316–320. [Google Scholar]
- Livingstone, M.S.; Rosen, G.D.; Drislane, F.W.; Galaburda, A.M. Physiological and anatomical evidence for a magnocellular defect in developmental dyslexia. Proc. Natl. Acad. Sci. USA 1991, 88, 7943–7947. [Google Scholar] [CrossRef] [PubMed]
- Eden, G.F.; Van Meter, J.W.; Rumsey, J.M.; Maisog, J.M.; Woods, R.P.; Zeffiro, T.A. Abnormal processing of visual motion in dyslexia revealed by functional brain imaging. Nature 1996, 382, 66–69. [Google Scholar] [CrossRef] [PubMed]
- Demb, J.B.; Boynton, G.M.; Heeger, D.J. Brain activity in visual cortex predicts individual differences in reading performance. Proc. Natl. Acad. Sci. USA 1997, 94, 13363–13366. [Google Scholar] [CrossRef] [PubMed]
- Stein, J.; Walsh, V. To see but not to read; the magnocellular theory of dyslexia. Trends Neurosci. 1997, 20, 147–152. [Google Scholar] [CrossRef]
- Stein, J.; Walsh, V. Impaired neural timing in developmental dyslexia—The magnocellular hypothesis. Dyslexia 1999, 5, 59–77. [Google Scholar] [CrossRef]
- Stein, J. The magnocellular theory of dyslexia. In Developmental Dyslexia, Early Precursors, Neurobehavioral Markers, and Biological Substrates; Benasich, A.A., Fitch, R.H., Eds.; P. H. Brooks: Baltimore, MD, USA; London, UK; Sydney, NSW, Australia, 2012. [Google Scholar]
- Stein, J. The current status of the magnocellular theory of developmental dyslexia. Neuropsychologia 2019, 130, 66–77. [Google Scholar] [CrossRef]
- Werth, R. Therapie von Lesestörungen durch Erkennen und Beheben der Ursachen. Ergother. Rehabil. 2006, 9, 6–11. [Google Scholar]
- Werth, R. Rapid improvement of reading performance in children with dyslexia by altering the reading strategy: A novel approach to diagnoses und therapy of reading deficiencies. Restor. Neurol. Neurosci. 2018, 36, 679–691. [Google Scholar] [CrossRef]
- Werth, R. What causes Dyslexia? Identifying the causes and effective compensatory therapy. Restor. Neurol. Neurosci. 2019, 37, 591–608. [Google Scholar] [CrossRef]
- Werth, R. Dyslexic readers improve without training when using a computer-guided reading strategy. Brain Sci. 2021, 11, 526. [Google Scholar] [CrossRef] [PubMed]
- Klische, A. Leseschwächen Gezielt Beheben. Individuelle Diagnose und Therapie mit dem Programm Celeco; Tectum: Marburg, Germany, 2007. [Google Scholar]
- Bringmann, A.; Syrbe, S.; Görner, K.; Kacza, J.; Francke, M.; Wiedemann, P.; Reichenbach, A. The primate fovea: Structure, function and development. Prog. Retin. Eye Res. 2018, 66, 49–84. [Google Scholar] [CrossRef]
- Rodieck, R.W.; Binmoeller, K.F.; Dineen, J. Parasol and midget ganglion cells of the human retina. J. Comp. Neurol. 1985, 233, 115–132. [Google Scholar] [CrossRef]
- Dacey, D.M. The mosaic of midget ganglion cells in the human retina. J. Neurosci. 1993, 13, 5334–5355. [Google Scholar] [CrossRef]
- Yan, W.; Peng, Y.R.; van Zyl, T.; Regev, A.; Shekhar, K.; Juric, D.; Sanes, J.R. Cell Atlas of The Human Fovea and Peripheral Retina. Sci. Rep. 2020, 10, 9802. [Google Scholar] [CrossRef]
- Masri, R.A.; Grünert, U.; Martin, P.R. Analysis of parvocellular and magnocellular visual Pathways in Human Retina. J. Neurosci. 2020, 40, 8132–8148. [Google Scholar] [CrossRef]
- Soto, F.; Hsiang, J.C.; Rajagopal, R.; Piggott, K.; Harocopos, G.J.; Couch, S.M.; Custer, P.; Morgan, J.L.; Kerschensteiner, D. Efficient coding by midget and parasol ganglion cells in the human retina. Neuron 2020, 107, 656–666.e5. [Google Scholar] [CrossRef] [PubMed]
- Dacey, D.M. Circuitry for color coding in the primate retina. Proc. Natl. Acad. Sci. USA 1996, 93, 582–588. [Google Scholar] [CrossRef] [PubMed]
- Calkins, D.J.; Schein, S.J.; Tsukamoto, Y.; Sterling, P. M and L cones in macaque fovea connect to midget ganglion cells by different numbers of excitatory synapses. Nature 1994, 371, 70–72. [Google Scholar] [CrossRef]
- Kolb, H.; Marshak, D. The midget pathways of the primate retina. Doc. Ophthalmol. 2003, 106, 67–81. [Google Scholar] [CrossRef]
- Lee, B.B.; Martin, P.R.; Grünert, U. Retinal connectivity and primate vision. Prog. Retin. Eye Res. 2010, 29, 622–639. [Google Scholar] [CrossRef] [PubMed]
- Provis, J.M.; Dubis, A.M.; Maddess, T.; Carroll, J. Adaptation of the central retina for high acuity vision: Cones, the fovea and the avascular zone. Prog. Retin. Eye Res. 2013, 35, 63–81. [Google Scholar] [CrossRef] [PubMed]
- Peng, Y.R.; Shekhar, K.; Yan, W.; Herrmann, D.; Sappington, A.; Bryman, G.S.; van Zyl, T.; Do, M.T.H.; Regev, A.; Sanes, J.R. Molecular Classification and Comparative Taxonomics of Foveal and Peripheral Cells in Primate Retina. Cell 2019, 176, 1222–1237.e22. [Google Scholar] [CrossRef]
- Sinha, R.; Hoon, M.; Baudin, J.; Okawa, H.; Wong, R.O.L.; Rieke, F. Cellular and Circuit Mechanisms Shaping the Perceptual Properties of the Primate Fovea. Cell 2017, 168, 413–426.e12. [Google Scholar] [CrossRef]
- Jeffries, A.M.; Killian, N.J.; Pezaris, J.S. Mapping the primate lateral geniculate nucleus: A review of experiments and methods. J. Physiol. Paris 2014, 108, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Denison, R.N.; Vu, A.T.; Yacoub, E.; Feinberg, D.A.; Silver, M.A. Functional mapping of the magnocellular and parvocellular subdivisions of human LGN. Neuroimage 2014, 102 Pt 2, 358–369. [Google Scholar] [CrossRef]
- Irvin, G.E.; Norton, T.T.; Sesma, M.A.; Casagrande, V.A. W-like response properties of interlaminar zone cells in the lateral geniculate nucleus of a primate (Galago crassicaudatus). Brain Res. 1986, 362, 254–270. [Google Scholar] [CrossRef]
- Jayakumar, J.; Dreher, B.; Vidyasagar, T.R. Tracking blue cone signals in the primate brain. Clin. Exp. Optom. 2013, 96, 259–266. [Google Scholar] [CrossRef]
- White, A.J.; Solomon, S.G.; Martin, P.R. Spatial properties of koniocellular cells in the lateral geniculate nucleus of the marmoset Callithrix jacchus. J. Physiol. 2001, 533, 519–535. [Google Scholar] [CrossRef]
- Hubel, D.H.; Wiesel, T.N. Laminar and columnar distribution of geniculo-cortical fibers in the macaque monkey. J. Comp. Neurol. 1972, 146, 421–450. [Google Scholar] [CrossRef]
- Blasdel, G.G.; Fitzpatrick, D. Physiological organization of layer 4 in macaque striate cortex. J. Neurosci. 1984, 4, 880–895. [Google Scholar] [CrossRef]
- Merigan, W.H.; Maunsell, J.H. How parallel are the primate visual pathways? Ann. Rev. Neurosci. 1993, 16, 369–402. [Google Scholar] [CrossRef]
- Casagrande, V.A. A third parallel visual pathway to primate area V1. Trends Neurosci. 1994, 17, 305–310. [Google Scholar] [CrossRef]
- Hendry, S.H.C.; Reid, R.C. The koniocellular pathway in primate vision. Annu. Rev. Neurosci. 2000, 23, 127–153. [Google Scholar] [CrossRef] [PubMed]
- Fries, W. The projection from the lateral geniculate nucleus to the prestriate cortex of the macaque monkey. Proc. R. Soc. Lond. B Biol. Sci. 1981, 213, 73–86. [Google Scholar] [PubMed]
- Yukie, M.; Iwai, E. Direct projection from the dorsal lateral geniculate nucleus to the prestriate cortex in macaque monkeys. J. Comp. Neurol. 1981, 201, 81–97. [Google Scholar] [CrossRef] [PubMed]
- Lyon, D.C.; Kaas, J.H. Connectional evidence for dorsal and ventral V3, and other extrastriate areas in the prosimian primate, Galago garnetti. Brain Behav. Evol. 2002, 59, 114–129. [Google Scholar] [CrossRef] [PubMed]
- Ferrera, V.P.; Nealey, T.A.; Maunsell, J.H. Mixed parvocellular and magnocellular geniculate signals in visual area V4. Nature 1992, 358, 756–761. [Google Scholar] [CrossRef] [PubMed]
- Ferrera, V.P.; Nealey, T.A.; Maunsell, J.H. Responses in macaque visual area V4 following inactivation of the parvocellular and magnocellular LGN pathways. J. Neurosci. 1994, 14, 2080–2088. [Google Scholar] [CrossRef]
- Dumoulin, S.O.; Bittar, R.G.; Kabani, N.J.; Baker, C.L., Jr.; Le Goualher, G.; Pike, G.B.; Evans, A.C. A new anatomical landmark for reliable identification of human area V5/MT: A quantitative analysis of sulcal patterning. Cereb. Cortex 2000, 10, 454–463. [Google Scholar] [CrossRef]
- DiCarlo, J.J.; Zoccolan, D.; Rust, N.C. How does the brain solve visual object recognition? Neuron 2012, 73, 415–434. [Google Scholar] [CrossRef] [PubMed]
- Kravitz, D.J.; Saleem, K.S.; Baker, C.I.; Ungerleider, L.G.; Mishkin, M. The ventral visual pathway: An expanded neural framework for the processing of object quality. Trends Cogn. Sci. 2013, 17, 26–49. [Google Scholar] [CrossRef]
- Zeki, S. Area V5-a microcosm of the visual brain. Front. Integr. Neurosci. 2015, 9, 21. [Google Scholar] [CrossRef] [PubMed]
- Maunsell, J.H.; Nealey, T.A.; De Priest, D.D. Magnocellular and parvocellular contributions to responses in the middle temporal visual area (MT) of the macaque monkey. J. Neurosci. 1990, 10, 3323–3334. [Google Scholar] [CrossRef]
- Ffytche, D.H.; Guy, C.N.; Zeki, S. The parallel visual motion inputs into areas V1 and V5 of human cerebral cortex. Brain 1995, 118, 1375–1394. [Google Scholar] [CrossRef] [PubMed]
- Cragg, B.G. The topography of the afferent projections in the circumstriate visual cortex of the monkey studied by the Nauta method. Vis. Res. 1969, 9, 733–747. [Google Scholar] [CrossRef]
- Zeki, S.M. Representation of central visual fields in prestriate cortex of monkey. Brain Res. 1969, 14, 271–291. [Google Scholar] [CrossRef]
- Zeki, S.M. The functional organization of projections from striate to prestriate visual cortex in the rhesus monkey. Cold Spring Harb. Symp. Quant. Biol. 1975, 40, 591–600. [Google Scholar] [CrossRef]
- Zeki, S.M. The projections to the superior temporal sulcus from areas 17 and 18 in the rhesus monkey. Proc. R. Soc. B 1976, 193, 199–207. [Google Scholar]
- Zeki, S.M. Simultaneous anatomical demonstration of the representation of the vertical and horizontal meridians in areas V2 and V3 of rhesus monkey visual cortex. Proc. R. Soc. B 1977, 195, 517–523. [Google Scholar]
- Zeki, S.M. Colour coding in the superior temporal sulcus of rhesus monkey visual cortex. Proc. R. Soc. B 1977, 197, 195–223. [Google Scholar]
- Zeki, S.M. Colour coding in rhesus monkey prestriate cortex. Brain Res. 1978, 53, 422–427. [Google Scholar] [CrossRef]
- Zeki, S.M. The cortical projections of foveal striate cortex in the rhesus monkey. J. Physiol. 1978, 277, 227–244. [Google Scholar] [CrossRef] [PubMed]
- Weiner, K.S. Area TEO and “Area?”: Cytoarchitectonic confusion corrected by connectivity and cortical ablation. Brain Struct. Funct. 2018, 223, 3515–3529. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, H.; Gattass, R.; Desimone, R.; Ungerleider, L.G. The modular organization of projections from areas V1 and V2 to areas V4 and TEO in macaques. J. Neurosci. 1993, 13, 3681–3691. [Google Scholar] [CrossRef]
- Distler, C.; Boussaoud, D.; Desimone, R.; Ungerleider, L.G. Cortical connections of inferior temporal area TEO in macaque monkeys. J. Comp. Neurol. 1993, 334, 125–150. [Google Scholar] [CrossRef]
- Dubner, R.; Zeki, S.M. Response properties and receptive fields of cells in an anatomically defined region of the superior temporal sulcus in the monkey. Brain Res. 1971, 35, 528–532. [Google Scholar] [CrossRef]
- Albright, T.D. Form-cue invariant motion processing in primate visual cortex. Science 1992, 255, 1141–1143. [Google Scholar] [CrossRef]
- Bisley, J.W.; Pasternak, T. The multiple roles of visual cortical areas MT/MST in remembering the direction of visual motion. Cereb. Cortex 2000, 10, 1053–1065. [Google Scholar] [CrossRef]
- Tootell, R.B.; Mendola, J.D.; Hadjikhani, N.K.; Ledden, P.J.; Liu, A.K.; Reppas, J.B.; Sereno, M.I.; Dale, A.M. Functional analysis of V3A and related areas in human visual cortex. J. Neurosci. 1997, 17, 7060–7078. [Google Scholar] [CrossRef]
- Bar, M.; Tootell, R.B.; Schacter, D.L.; Greve, D.N.; Fischl, B.; Mendola, J.D.; Rosen, B.R.; Dale, A.M. Cortical mechanisms specific to explicit visual object recognition. Neuron 2001, 29, 529–535. [Google Scholar] [CrossRef]
- Felleman, D.J.; Van Essen, D.C. Distributed hierarchical processing in the primate cerebral cortex. Cereb. Cortex 1991, 1, 1–47. [Google Scholar] [CrossRef]
- Schneider, G.E. Two visual systems. Science 1969, 163, 895–902. [Google Scholar] [CrossRef] [PubMed]
- Poeppel, E.; Held, R.; Frost, D. Residual visual function in patients with lesions oft he central visual pathways. Nature 1973, 256, 489–490. [Google Scholar] [CrossRef] [PubMed]
- Perenin, M.T.; Jeannerod, M. Visual function within the hemianopic field following early cerebral hemidecortication in man. Neuropsychologia 1978, 16, 1–13. [Google Scholar] [CrossRef]
- Zihl, J.; Werth, R. Contributions tot he study of “blindsight”-II. The role of specific practice for saccadic localization in patients with postgeniculate visual field defects. Neiropsychologia 1984, 22, 13–22. [Google Scholar] [CrossRef]
- Werth, R. Residual visual functions after loss of both cerebral hemispheres in infancy. Invest. Ophthalmol. Vis. Sci. 2007, 48, 3098–3106. [Google Scholar] [CrossRef] [PubMed]
- Ungerleider, L.; Mishkin, M. Two cortical visual systems. In Analysis of Motor Behavior; Ingle, D.J., Goodale, M.A., Mansfield, R.J.W., Eds.; MIT Press: Cambridge, MA, USA, 1982; pp. 549–586. [Google Scholar]
- Mishkin, M.; Ungerleider, L.G.; Macko, K.A. Object vision and spatial vision: Two cortical pathways. Trends Neurosci. 1983, 6, 414–417. [Google Scholar] [CrossRef]
- Goodale, M.A.; Milner, A.D. Separate visual pathways for perception and action. Trends Neurosci. 1992, 15, 20–25. [Google Scholar] [CrossRef] [PubMed]
- Goodale, M.A.; Milner, A.D.; Jakobson, L.S.; Carey, D.P. A neurological dissociation between perceiving objects and grasping them. Nature 1991, 349, 154–156. [Google Scholar] [CrossRef] [PubMed]
- James, T.W.; Culham, J.; Humphrey, G.K.; Milner, A.D.; Goodale, M.A. Ventral occipital lesions impair object recognition but not object-directed grasping: An fMRI study. Brain 2003, 126, 2463–2475. [Google Scholar] [CrossRef]
- Milner, A.D. How do the two visual streams interact with each other? Exp. Brain Res. 2017, 235, 1297–1308. [Google Scholar] [CrossRef] [PubMed]
- Fridriksson, J.; Yourganov, G.; Bonilha, L.; Basilakos, A.; Den Ouden, D.B.; Rorden, C. Revealing the dual streams of speech processing. Proc. Natl. Acad. Sci. USA 2016, 113, 15108–15113. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.H.; Jeong, G.; Kim, Y.B.; Cho, Z.H. Proposal for human visual pathway in the extrastriate cortex by fiber tracking method using diffusion-weighted MRI. Neuroimage 2020, 220, 117145. [Google Scholar] [CrossRef]
- Jitsuishi, T.; Yamaguchi, A. Identification of a distinct association fiber tract “IPS-FG” to connect the intraparietal sulcus areas and fusiform gyrus by white matter dissection and tractography. Sci. Rep. 2020, 10, 15475. [Google Scholar] [CrossRef]
- Uddin, L.Q.; Supekar, K.; Amin, H.; Rykhlevskaia, E.; Nguyen, D.A.; Greicius, M.D.; Menon, V. Dissociable connectivity within human angular gyrus and intraparietal sulcus: Evidence from functional and structural connectivity. Cereb. Cortex 2010, 20, 2636–2646. [Google Scholar] [CrossRef]
- Takemura, H.; Rokem, A.; Winawer, J.; Yeatman, J.D.; Wandell, B.A.; Pestilli, F. A Major Human White Matter Pathway Between Dorsal and Ventral Visual Cortex. Cereb. Cortex 2016, 26, 2205–2214. [Google Scholar] [CrossRef] [PubMed]
- Tootell, R.B.; Mendola, J.D.; Hadjikhani, N.K.; Liu, A.K.; Dale, A.M. The representation of the ipsilateral visual field in human cerebral cortex. Proc. Natl. Acad. Sci. USA 1998, 95, 818–824. [Google Scholar] [CrossRef] [PubMed]
- Lorenz, S.; Weiner, K.S.; Caspers, J.; Mohlberg, H.; Schleicher, A.; Bludau, S.; Eickhoff, S.B.; Grill-Spector, K.; Zilles, K.; Amunts, K. Two New Cytoarchitectonic Areas on the Human Mid-Fusiform Gyrus. Cereb. Cortex 2017, 27, 373–385. [Google Scholar] [CrossRef]
- Wu, Y.; Sun, D.; Wang, Y.; Wang, Y.; Wang, Y. Tracing short connections of the temporo-parieto-occipital region in the human brain using diffusion spectrum imaging and fiber dissection. Brain Res. 2016, 1646, 152–159. [Google Scholar] [CrossRef]
- Takemura, H.; Pestilli, F.; Weiner, K.S.; Keliris, G.A.; Landi, S.M.; Sliwa, J.; Ye, F.Q.; Barnett, M.A.; Leopold, D.A.; Freiwald, W.A.; et al. Occipital White Matter Tracts in Human and Macaque. Cereb. Cortex 2017, 27, 3346–3359. [Google Scholar] [CrossRef] [PubMed]
- Yeatman, J.D.; Weiner, K.S.; Pestilli, F.; Rokem, A.; Mezer, A.; Wandell, B.A. The vertical occipital fasciculus: A century of controversy resolved by in vivo measurements. Proc. Natl. Acad. Sci. USA 2014, 111, E5214–E5223. [Google Scholar] [CrossRef]
- Jitsuishi, T.; Hirono, S.; Yamamoto, T.; Kitajo, K.; Iwadate, Y.; Yamaguchi, A. White matter dissection and structural connectivity of the human vertical occipital fasciculus to link vision-associated brain cortex. Sci. Rep. 2020, 10, 820. [Google Scholar] [CrossRef]
- Tootell, R.B.; Reppas, J.B.; Dale, A.M.; Look, R.B.; Sereno, M.I.; Malach, R.; Brady, T.J.; Rosen, B.R. Visual motion aftereffect in human cortical area MT revealed by functional magnetic resonance imaging. Nature 1995, 375, 139–141. [Google Scholar] [CrossRef]
- Avidan, G.; Harel, M.; Hendler, T.; Ben-Bashat, D.; Zohary, E.; Malach, R. Contrast sensitivity in human visual areas and its relationship to object recognition. J. Neurophysiol. 2002, 87, 3102–3116. [Google Scholar] [CrossRef]
- Nasr, S.; Polimeni, J.R.; Tootell, R.B. Interdigitated Color- and Disparity-Selective Columns within Human Visual Cortical Areas V2 and V3. J. Neurosci. 2016, 36, 1841–1857. [Google Scholar] [CrossRef] [PubMed]
- Tootell, R.B.H.; Nasr, S. Columnar Segregation of Magnocellular and Parvocellular Streams in Human Extrastriate Cortex. J. Neurosci. 2017, 37, 8014–8032. [Google Scholar] [CrossRef]
- Winawer, J.; Witthoft, N. Human V4 and ventral occipital retinotopic maps. Vis. Neurosci. 2015, 32, E020. [Google Scholar] [CrossRef] [PubMed]
- Cohen, D.; Goddard, E.; Mullen, K.T. Reevaluating hMT+ and hV4 functional specialization for motion and static contrast using fMRI-guided repetitive transcranial magnetic stimulation. J. Vis. 2019, 19, 11. [Google Scholar] [CrossRef]
- Himmelberg, M.M.; Wade, A.R. Eccentricity-dependent temporal contrast tuning in human visual cortex measured with fMRI. Neuroimage 2019, 184, 462–474. [Google Scholar] [CrossRef]
- May, J.G.; Lovegrove, W.J.; Martin, F.; Nelson, P. Pattern-elicited visual evoked potentials in good and poor readers. Clin. Vis. Sci. 1991, 6, 131–136. [Google Scholar]
- Lehmkuhle, S.; Garzia, R.P.; Turner, L.; Hash, T.; Baro, J.A. A Defective Visual Pathway in Children with Reading Disability. N. Engl. J. Med. 1993, 328, 989–996. [Google Scholar] [CrossRef]
- Kubová, Z.; Kuba, M.; Kremláček, J.; Langrová, J.; Szanyi, J.; Vít, F.; Chutná, M. Comparison of visual information processing in school-age dyslexics and normal readers via motion-onset visual evoked potentials. Vis. Res. 2015, 111, 97–104. [Google Scholar] [CrossRef]
- Franceschini, S.; Gori, S.; Ruffino, M.; Viola, S.; Molteni, M.; Facoetti, A. Action video games make dyslexic children read better. Curr. Biol. 2013, 23, 462–466. [Google Scholar] [CrossRef] [PubMed]
- Brown, A.C.; Peters, J.L.; Parsons, C.; Crewther, D.P.; Crewther, S.G. Efficiency in Magnocellular Processing: A Common Deficit in Neurodevelopmental Disorders. Front. Hum. Neurosci. 2020, 14, 49. [Google Scholar] [CrossRef] [PubMed]
- Rao, S.C.; Rainer, G.; Miller, E.K. Integration of what and where in the primate prefrontal cortex. Science 1997, 276, 821–824. [Google Scholar] [CrossRef] [PubMed]
- Skoyles, J.; Skottun, B.C. On the prevalence of magnocellular deficits in the visual system of non-dyslexic individuals. Brain Lang. 2004, 88, 79–82. [Google Scholar] [CrossRef]
- Heim, S.; Grande, M.; Pape-Neumann, J.; Van Ermingen, M.; Meffert, E.; Grabowska, A.; Huber, W.; Amunts, K. Interaction of phonological awareness and ‘magnocellular’ processing during normal and dyslexic reading: Behavioural and fMRI investigations. Dyslexia 2010, 16, 258–282. [Google Scholar] [CrossRef]
- Blythe, H.I.; Kirkby, J.A.; Liversedge, S.P. Comments on: “What is developmental dyslexia?” Brain Sci. 2018, 8, 26. The relationship between eye movements and reading difficulties. Brain Sci. 2018, 8, 100. [Google Scholar] [CrossRef]
- Contemori, G.; Battaglini, L.; Barollo, M.; Ciavarelli, A.; Casco, C. Developmental dyslexia: A deficit in magnocellular-parvocellular co-activation, not simply in pure magnocellular activation. Vis. Res. 2019, 159, 61–67. [Google Scholar] [CrossRef] [PubMed]
- Hutzler, F.; Kronbichler, M.; Jacobs, A.M.; Wimmer, H. Perhaps correlational but not causal: No effect of dyslexic readers’ magnocellular system on their eye movements during reading. Neuropsychologia 2006, 44, 637–648. [Google Scholar] [CrossRef] [PubMed]
- Harms, H. Die Technik der statischen Perimetrie. Ophthalmologica 1969, 158, 387–405. [Google Scholar] [CrossRef]
- Krakau, C.E. Temporal summation and perimetry. Ophthalmic Res. 1989, 21, 49–55. [Google Scholar] [CrossRef]
- Sandberg, K.; Bibby, B.M.; Timmermans, B.; Cleeremans, A.; Overgaard, M. Measuring consciousness: Task accuracy and awareness as sigmoid functions of stimulus duration. Conscious Cogn. 2011, 20, 1659–1675. [Google Scholar] [CrossRef]
- Windey, B.; Vermeiren, A.; Atas, A.; Cleeremans, A. The graded and dichotomous nature of visual awareness. Philos. Trans. R. Soc. B Biol. Sci. 2014, 369, 20130282. [Google Scholar] [CrossRef][Green Version]
- Mulholland, P.J.; Redmond, T.; Garway-Heath, D.F.; Zlatkova, M.B.; Anderson, R.S. The Effect of Age on the Temporal Summation of Achromatic Perimetric Stimuli. Investig. Ophthalmol. Vis. Sci. 2015, 56, 6467–6472. [Google Scholar] [CrossRef][Green Version]
- Beauny, A.; de Heering, A.; Muñoz Moldes, S.; Martin, J.-R.; de Beir, A.; Cleeremans, A. Unconscious categorization of sub-millisecond complex images. PLoS ONE 2020, 15, e0236467. [Google Scholar] [CrossRef] [PubMed]
- Holmes, R.; Victora, M.; Wang, R.F.; Kwiat, P.G. Measuring temporal summation in visual detection with a single-photon source. Vis. Res. 2017, 140, 33–43. [Google Scholar] [CrossRef]
- Stigliani, A.; Jeska, B.; Grill-Spector, K. Encoding model of temporal processing in human visual cortex. Proc. Natl. Acad. Sci. USA 2017, 114, E11047–E11056. [Google Scholar] [CrossRef]
- Graham, C.H.; Cook, C. Visual acuity as a function of intensity and exposure-time. Am. J. Psychol. 1937, 49, 654–661. [Google Scholar] [CrossRef]
- Schwarz, F. Der Einfluß der Darbietungszeit auf die Erkennbarkeit von Sehproben. Pfluegers Arch. 1947, 249, 354–360. [Google Scholar] [CrossRef]
- Barlow, H.B. Temporal and spatial summation in human vision at different background intensities. J. Physiol. Lond. 1958, 141, 337–350. [Google Scholar] [CrossRef] [PubMed]
- Niwa, K.; Tokoro, T. Measurement of temporal summation of visual acuity with use of modified tachistoscope. Jpn. J. Ophthalmol. 1997, 41, 403–408. [Google Scholar] [CrossRef] [PubMed]
- McAnany, J.J. The effect of exposure duration on visual acuity for letter optotypes and gratings. Vis. Res. 2014, 105, 86–91. [Google Scholar] [CrossRef]
- Heinrich, S.P.; Blechenberg, T.; Reichel, C.; Bach, M. The “speed” of acuity in scotopic vs. photopic vision. Graefes Arch. Clin. Exp. Ophthalmol. 2020, 258, 2791–2798. [Google Scholar] [CrossRef] [PubMed]
- Petersen, S.E.; Fox, P.T.; Posner, M.I.; Mintun, M.; Raichle, M.E. Positron emission tomography studies of the cortical anatomy of single-word processing. Nature 1988, 331, 585–588. [Google Scholar] [CrossRef]
- Petersen, S.E.; Fox, P.T.; Synder, A.Z.; Raichle, M.E. Activation of extrastriate and frontal cortices areas by words and word-like stimuli. Science 1990, 249, 1041–1044. [Google Scholar] [CrossRef] [PubMed]
- Price, C.J.; Moore, C.J.; Frackowiak, R.S.J. The effect of varying stimulus rate and duration on brain activity during reading. NeuroImage 1996, 3, 40–52. [Google Scholar] [CrossRef] [PubMed]
- Price, C.J.; Wise, R.J.S.; Frackowiak, R.S.J. Demonstrating the implicit processing of visually presented words and pseudowords. Cereb. Cortex 1996, 6, 62–70. [Google Scholar] [CrossRef]
- Jernigan, T.L.; Ostergaard, A.L.; Law, I.; Svarer, C.; Gerlach, C.; Paulson, O. Brain activation during word identification and word recognition. NeuroImage 1998, 8, 93–105. [Google Scholar] [CrossRef][Green Version]
- Yeatman, J.D.; Rauschecker, A.M.; Wandell, B.A. Anatomy of the visual word form area: Adjacent cortical circuits and long-range white matter connections. Brain Lang. 2013, 125, 146–155. [Google Scholar] [CrossRef]
- Weiner, K.S.; Sayres, R.; Vinberg, J.; Grill-Spector, K. fMRI-adaptation and category selectivity in human ventral temporal cortex: Regional differences across time scales. J. Neurophysiol. 2010, 103, 3349–3365. [Google Scholar] [CrossRef]
- Zhou, J.; Benson, N.C.; Kendrick, N.; Kay, K.N.; Winawer, J. Compressive temporal summation in human visual cortex. J. Neurosci. 2018, 38, 691–709. [Google Scholar] [CrossRef]
- Stigliani, A.; Jeska, B.; Grill-Spector, K. Differential sustained and transient temporal processing across visual streams. PLoS Comput. Biol. 2019, 15, e1007011. [Google Scholar] [CrossRef]
- Mechelli, A.; Humphreys, G.W.; Mayall, K.; Olson, A.; Price, C.J. Differential effects of word length and visual contrast in the fusiform and lingual gyri during reading. Proc. R. Soc. Lond. Ser. B 2000, 267, 1909–1913. [Google Scholar] [CrossRef] [PubMed]
- Schurz, M.; Sturm, D.; Richlan, F.; Kronbichler, M.; Ladurner, G.; Wimmer, H. A dual-route perspective on brain activation in response to visual words: Evidence for a length by lexicality interaction in the visual word form area (VWFA). Neuroimage 2010, 49, 2649–2661. [Google Scholar] [CrossRef] [PubMed]
- Martens, V.E.; de Jong, P.F. The effect of word length on lexical decision in dyslexic and normal reading children. Brain Lang. 2006, 98, 140–149. [Google Scholar] [CrossRef]
- Warrington, E.K.; Shallice, T. Word-form dyslexia. Brain 1980, 103, 99–112. [Google Scholar] [CrossRef] [PubMed]
- Cohen, L.; Dehaene, S.; Naccache, L.; Lehéricy, S.; Dehaene-Lambertz, G.; Hénaff, M.A.; Michel, F. The visual word form area: Spatial and temporal characterization of an initial stage of reading in normal subjects and posterior split-brain patients. Brain 2000, 123, 291–307. [Google Scholar] [CrossRef]
- Cohen, L.; Lehericy, S.; Chochon, F.; Lemer, C.; Rivaud, S.; Dehaene, S. Language-specific tuning of visual cortex? Functional properties of the visual word form area. Brain 2002, 125, 1054–1069. [Google Scholar] [PubMed]
- Cohen, L.; Martinaud, O.; Lemer, C.; Lehéricy, S.; Samson, Y.; Obadia, M.; Slachevsky, A.; Dehaene, S. Visual word recognition in the left and right hemispheres: Anatomical and functional correlates of peripheral alexias. Cereb. Cortex 2003, 13, 1313–1333. [Google Scholar] [CrossRef] [PubMed]
- Nasr, S.; Liu, N.; Devaney, K.J.; Yue, X.; Rajimehr, R.; Ungerleider, L.G.; Tootell, R.B. Scene-selective cortical regions in human and nonhuman primates. J. Neurosci. 2011, 31, 13771–13785. [Google Scholar] [CrossRef] [PubMed]
- James, K.H.; James, T.W.; Jobard, G.; Wong, A.C.; Gauthier, I. Letter processing in the visual system: Different activation patterns for single letters and strings. Cogn. Affect. Behav. Neurosci. 2005, 5, 452–466. [Google Scholar] [CrossRef]
- Rosenke, M.; Weiner, K.S.; Barnett, M.A.; Zilles, K.; Amunts, K.; Goebel, R.; Grill-Spector, K. A cross-validated cytoarchitectonic atlas of the A cross-validated cytoarchitectonic atlas of the human ventral visual stream. Neuroimage 2018, 170, 257–270. [Google Scholar] [CrossRef]
- Weiner, K.S.; Barnett, M.A.; Lorenz, S.; Caspers, J.; Stigliani, A.; Amunts, K.; Zilles, K.; Fischl, B.; Grill-Spector, K. The Cytoarchitecture of Domain-specific Regions in Human High-level Visual Cortex. Cereb. Cortex 2017, 27, 146–161. [Google Scholar] [CrossRef]
- Epelbaum, S.; Pinel, P.; Gaillard, R.; Delmaire, C.; Perrin, M.; Dupont, S.; Dehaene, S.; Cohen, L. Pure alexia as a disconnection syndrome: New diffusion imaging evidence for an old concept. Cortex 2008, 44, 962–974. [Google Scholar] [CrossRef]
- Polk, T.A.; Stallcup, M.; Aguirre, G.K.; Alsop, D.C.; D’Esposito, M.; Detre, J.A.; Farah, M.J. Neural specialization for letter recognition. J. Cogn. Neurosci. 2002, 14, 145–159. [Google Scholar] [CrossRef]
- McCandliss, B.D.; Cohen, L.; Dehaene, S. The visual word form area: Expertise for reading in the fusiform gyrus. Trends Cogn. Sci. 2003, 7, 293–299. [Google Scholar] [CrossRef]
- Baker, C.I.; Liu, J.; Wald, L.L.; Kwong, K.K.; Benner, T.; Kanwisher, N. Visual word processing and experiential origins of functional selectivity in human extrastriate cortex. Proc. Natl. Acad. Sci. USA 2007, 104, 9087–9092. [Google Scholar] [CrossRef]
- Stevens, W.D.; Kravitz, D.J.; Peng, C.S.; Tessler, M.H.; Martin, A. Privileged Functional Connectivity between the Visual Word Form Area and the Language System. J. Neurosci. 2017, 37, 5288–5297. [Google Scholar] [CrossRef]
- Ben-Shachar, M.; Dougherty, R.F.; Deutsch, G.K.; Wandell, B.A. The development of cortical sensitivity to visual word forms. J. Cogn. Neurosci. 2011, 23, 2387–2399. [Google Scholar] [CrossRef] [PubMed]
- Stigliani, A.; Weiner, K.S.; Grill-Spector, K. Temporal Processing Capacity in High-Level Visual Cortex Is Domain Specific. J. Neurosci. 2015, 35, 12412–12424. [Google Scholar] [CrossRef]
- Binder, J.R.; Medler, D.A.; Westbury, C.F.; Liebenthal, E.; Buchanan, L. Tuning of the human left fusiform gyrus to sublexical orthographic structure. Neuroimage 2006, 33, 739–748. [Google Scholar] [CrossRef] [PubMed]
- Graves, W.W.; Desai, R.; Humphries, C.; Seidenberg, M.S.; Binder, J.R. Neural systems for reading aloud: A multiparametric approach. Cereb. Cortex 2010, 20, 1799–1815. [Google Scholar] [CrossRef]
- Dębska, A.; Chyl, K.; Dzięgiel, G.; Kacprzak, A.; Łuniewska, M.; Plewko, J.; Marchewka, A.; Grabowska, A.; Jednoróg, K. Reading and spelling skills are differentially related to phonological processing: Behavioral and fMRI study. Dev. Cogn. Neurosci. 2019, 39, 100683. [Google Scholar] [CrossRef] [PubMed]
- Reich, L.; Szwed, M.; Cohen, L.; Amedi, A. A ventral visual stream reading center independent of visual experience. Curr. Biol. 2011, 21, 363–368. [Google Scholar] [CrossRef]
- Taylor, J.S.; Rastle, K.; Davis, M.H. Can cognitive models explain brain activation during word and pseudoword reading? A meta-analysis of 36 neuroimaging studies. Psychol. Bull. 2013, 139, 766–791. [Google Scholar] [CrossRef]
- Fischer-Baum, S.; Kook, J.H.; Lee, Y.; Ramos-Nuñez, A.; Vannucci, M. Individual Differences in the Neural and Cognitive Mechanisms of Single Word Reading. Front. Hum. Neurosci. 2018, 12, 271. [Google Scholar] [CrossRef]
- Woolnough, O.; Donos, C.; Rollo, P.S.; Forseth, K.J.; Lakretz, Y.; Crone, N.E.; Fischer-Baum, S.; Dehaene, S.; Tandon, N. Spatiotemporal dynamics of orthographic and lexical processing in the ventral visual pathway. Nat. Hum. Behav. 2021, 5, 389–398. [Google Scholar] [CrossRef] [PubMed]
- Jobard, G.; Crivello, F.; Tzourio-Mazoyer, N. Evaluation of the dual route theory of reading: A metanalysis of 35 neuroimaging studies. Neuroimage 2003, 20, 693–712. [Google Scholar] [CrossRef]
- Dehaene, S.; Le Clec’H, G.; Poline, J.B.; Le Bihan, D.; Cohen, L. The visual word-form area: A prelexical representation of visualwords in the fusiform gyrus. Neuroreport 2002, 13, 321–325. [Google Scholar] [CrossRef] [PubMed]
- Wydell, T.N.; Vuorinen, T.; Helenius, P.; Salmelin, R. Neural correlates of letter-string length and lexicality during reading in a regular orthography. J. Cogn. Neurosci. 2003, 15, 1052–1062. [Google Scholar] [CrossRef] [PubMed]
- Lochy, A.; Jacques, C.; Maillard, L.; Colnat-Coulbois, S.; Rossion, B.; Jonas, J. Selective visual representation of letters and words in the left ventral occipito-temporal cortex with intracerebral recordings. Proc. Natl. Acad. Sci. USA 2018, 115, E7595–E7604. [Google Scholar] [CrossRef]
- Fiez, J.A.; Balota, D.A.; Raichle, M.E.; Petersen, S.E. Effects of lexicality, frequency, and spelling-to-sound consistencyon the functional anatomy of reading. Neuron 1999, 24, 205–218. [Google Scholar] [CrossRef]
- Xu, B.; Grafman, J.; Gaillard, W.D.; Ishii, K.; Vega-Bermudez, F.; Pietrini, P.; Reeves-Tyer, P.; DiCamillo, P.; Theodore, W. Conjoint and extended neural networks for the computation of speechcodes: The neural basis of selective impairment in reading words and pseudowords. Cereb. Cortex 2001, 11, 267–277. [Google Scholar] [CrossRef]
- Binder, J.R.; Medler, D.A.; Desai, R.; Conant, L.L.; Liebenthal, E. Some neurophysiological constraints on models of word naming. Neuroimage 2005, 27, 677–693. [Google Scholar] [CrossRef]
- Kronbichler, M.; Bergmann, J.; Hutzler, F.; Staffen, W.; Mair, A.; Ladurner, G.; Wimmer, H. Taxi vs. Taksi: On orthographic word recognition in the left ventral occipitotemporal cortex. J. Cogn. Neurosci. 2007, 19, 1584–1594. [Google Scholar] [CrossRef]
- Church, J.A.; Balota, D.A.; Petersen, S.E.; Schlaggar, B.L. Manipulation of length and lexicality localizes the functional neuroanatomy of phonological processing in adult readers. J. Cogn. Neurosci. 2011, 23, 1475–1493. [Google Scholar] [CrossRef]
- Bruno, J.L.; Zumberge, A.; Manis, F.R.; Lu, Z.L.; Goldman, J.G. Sensitivity to orthographic familiarity in the occipito-temporal region. Neuroimage 2008, 39, 1988–2001. [Google Scholar] [CrossRef]
- Spinelli, D.; De Luca, M.; Di Filippo, G.; Mancini, M.; Martelli, M.; Zoccolotti, P. Length effect in word naming in reading: Role of reading experience and reading deficit in italian readers. Dev. Neuropsychol. 2005, 27, 217–235. [Google Scholar] [CrossRef]
- Price, C.J.; Devlin, J.T. The myth of the visual word form area. Neuroimage 2003, 19, 473–481. [Google Scholar] [CrossRef]
- Vogel, A.C.; Petersen, S.E.; Schlaggar, B.L. The left occipitotemporal cortex does not show preferential activity for words. Cereb. Cortex 2012, 22, 2715–2732. [Google Scholar] [CrossRef]
- Vogel, A.C.; Petersen, S.E.; Schlaggar, B.L. The VWFA: It’s not just for words anymore. Front. Hum. Neurosci. 2014, 8, 88. [Google Scholar] [CrossRef] [PubMed]
- Tagamets, M.A.; Novick, J.M.; Chalmers, M.L.; Friedman, R.B. A parametric approach to orthographic processing in the brain: An fMRI study. J. Cogn. Neurosci. 2000, 12, 281–297. [Google Scholar] [CrossRef] [PubMed]
- Wright, N.D.; Mechelli, A.; Noppeney, U.; Veltman, D.J.; Rombouts, S.A.; Glensman, J.; Haynes, J.D.; Price, C.J. Selective activation around the left occipito-temporal sulcus for words relative to pictures: Individual variability or false positives? Hum. Brain Mapp. 2008, 29, 986–1000. [Google Scholar] [CrossRef]
- Kay, K.N.; Yeatman, J.D. Bottom-up and top-down computations in word- and face-selective cortex. Elife 2017, 6, e22341. [Google Scholar] [CrossRef]
- Kuo, W.J.; Yeh, T.C.; Lee, J.R.; Chen, L.F.; Lee, P.L.; Chen, S.S.; Ho, L.T.; Hung, D.L.; Tzeng, O.J.; Hsieh, J.C. Orthographic and phonological processing of Chinese characters: An fMRI study. Neuroimage 2004, 21, 1721–1731. [Google Scholar] [CrossRef]
- Callan, A.M.; Callan, D.E.; Masaki, S. When meaningless symbols become letters: Neural activity change in learning new phonograms. Neuroimage 2005, 28, 553–562. [Google Scholar] [CrossRef] [PubMed]
- Xue, G.; Chen, C.; Jin, Z.; Dong, Q. Language experience shapes fusiform activation when processing a logographic artificial language: An fMRI training study. Neuroimage 2006, 31, 1315–1326. [Google Scholar] [CrossRef]
- Xue, G.; Poldrack, R.A. The neural substrates of visual perceptual learning of words: Implications for the visual word form area hypothesis. J. Cogn. Neurosci. 2007, 19, 1643–1655. [Google Scholar] [CrossRef]
- Moore, C.J.; Price, C.J. Three distinct ventral occipitotemporal regions for reading and object naming. Neuroimage 1999, 10, 181–192. [Google Scholar] [CrossRef]
- Ben-Shachar, M.; Dougherty, R.F.; Deutsch, G.K.; Wandell, B.A. Differential sensitivity to words and shapes in ventral occipito-temporal cortex. Cereb. Cortex 2007, 17, 1604–1611. [Google Scholar] [CrossRef]
- Ploran, E.J.; Nelson, S.M.; Velanova, K.; Donaldson, D.I.; Petersen, S.E.; Wheeler, M.E. Evidence accumulation and the moment of recognition: Dissociating perceptual recognition processes using fMRI. J. Neurosci. 2007, 27, 11912–11924. [Google Scholar] [CrossRef]
- Starrfelt, R.; Gerlach, C. The visual what for area: Words and pictures in the left fusiform gyrus. Neuroimage 2007, 35, 334–342. [Google Scholar] [CrossRef] [PubMed]
- Kherif, F.; Josse, G.; Price, C.J. Automatic top-down processing explains common left occipito-temporal responses to visual words and objects. Cereb. Cortex 2011, 21, 103–114. [Google Scholar] [CrossRef]
- Van Doren, L.; Dupont, P.; De Grauwe, S.; Peeters, R.; Vandenberghe, R. The amodal system for conscious word and picture identification in the absence of a semantic task. Neuroimage 2010, 49, 3295–3307. [Google Scholar] [CrossRef]
- Chen, L.; Wassermann, D.; Abrams, D.A.; Kochalka, J.; Gallardo-Diez, G.; Menon, V. The visual word form area (VWFA) is part of both language and attention circuitry. Nat. Commun. 2019, 10, 5601. [Google Scholar] [CrossRef] [PubMed]
- Tsapkini, K.; Rapp, B. The orthography-specific functions of the left fusiform gyrus: Evidence of modality and category specificity. Cortex 2010, 46, 185–205. [Google Scholar] [CrossRef] [PubMed]
- Vinckier, F.; Dehaene, S.; Jobert, A.; Dubus, J.P.; Sigman, M.; Cohen, L. Hierarchical coding of letter strings in the ventral stream: Dissecting the inner organization of the visual word-form system. Neuron 2007, 55, 143–156. [Google Scholar] [CrossRef] [PubMed]
- Olulade, O.A.; Flowers, D.L.; Napoliello, E.M.; Eden, G.F. Dyslexic children lack word selectivity gradients in occipito-temporal and inferior frontal cortex. Neuroimage Clin. 2015, 7, 742–754. [Google Scholar] [CrossRef] [PubMed]
- Lerma-Usabiaga, G.; Carreiras, M.; Paz-Alonso, P.M. Converging evidence for functional and structural segregation within the left ventral occipitotemporal cortex in reading. Proc. Natl. Acad. Sci USA 2018, 115, E9981–E9990. [Google Scholar] [CrossRef]
- Dehaene, S.; Cohen, L. The unique role of the visual word form area in reading. Trends Cogn. Sci. 2011, 15, 254–262. [Google Scholar] [CrossRef] [PubMed]
- Van der Mark, S.; Klaver, P.; Bucher, K.; Maurer, U.; Schulz, E.; Brem, S.; Martin, E.; Brandeis, D. The left occipitotemporal system in reading: Disruption of focal fMRI connectivity to left inferior frontal and inferior parietal language areas in children with dyslexia. Neuroimage 2011, 54, 2426–2436. [Google Scholar] [CrossRef]
- Martin, A.; Kronbichler, M.; Richlan, F. Dyslexic brain activation abnormalities in deep and shallow orthographies: A meta-analysis of 28 functional neuroimaging studies. Hum. Brain Mapp. 2016, 37, 2676–2699. [Google Scholar] [CrossRef]
- Boros, M.; Anton, J.-L.; Pech, C.; Grainger, J.; Szwed, M.; Ziegler, J.C. Orthographic processing deficits in developmental dyslexia: Beyond the ventral visual stream. Neuroimage 2016, 128, 316–327. [Google Scholar] [CrossRef]
- Salmelin, R.; Service, E.; Kiesilä, P.; Uutela, K.; Salonen, O. Impaired visual word processing in dyslexia revealed with magnetoencephalography. Ann. Neurol. 1996, 40, 157–162. [Google Scholar] [CrossRef]
- Rumsey, J.M.; Horwitz, B.; Donohue, B.C.; Nace, K.; Maisog, J.M.; Andreason, P. Phonological and orthographic components of word recognition. A PET-rCBF study. Brain 1997, 120, 739–759. [Google Scholar] [CrossRef]
- Brunswick, N. Explicit and implicit processing of words and pseudowords by adult developmental dyslexics: A search for Wernicke’s Wortschatz? Brain 1999, 122, 1901–1917. [Google Scholar] [CrossRef]
- Paulesu, E.; Démonet, J.F.; Fazio, F.; McCrory, E.; Chanoine, V.; Brunswick, N.; Cappa, S.F.; Cossu, G.; Habib, M.; Frith, C.D.; et al. Dyslexia: Cultural diversity and biological unity. Science 2001, 291, 2165–2167. [Google Scholar] [CrossRef]
- Shaywitz, B.A.; Shaywitz, S.E.; Pugh, K.R.; Mencl, W.E.; Fulbright, R.K.; Skudlarski, P.; Constable, R.T.; Marchione, K.E.; Fletcher, J.M.; Lyon, G.R.; et al. Disruption of posterior brain systems for reading in children with developmental dyslexia. Biol. Psychiatry 2002, 52, 101–110. [Google Scholar] [CrossRef]
- Cao, F.; Bitan, T.; Chou, T.; Burman, D.D.; Booth, J.R. Deficient orthographic and phonological representations in children with dyslexia revealed by brain activation patterns. J. Child. Psychol. Psychiatry 2006, 47, 1041–1050. [Google Scholar] [CrossRef]
- Maurer, U.; Brem, S.; Bucher, K.; Kranz, F.; Benz, R.; Steinhausen, H.C.; Brandeis, D. Impaired tuning of a fast occipito-temporal response for print in dyslexic children learning to read. Brain 2007, 130, 3200–3210. [Google Scholar] [CrossRef] [PubMed]
- Kronbichler, L.; Kronbichler, M. The importance of the left occipitotemporal cortex in developmental dyslexia. Curr. Dev. Disord. Rep. 2018, 5, 1–8. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Rodrigues, A.P.; Rebola, J.; Pereira, M.; van Asselen, M.; Castelo-Branco, M. Neural Responses of the AnteriorVentral Occipitotemporal Cortex in Developmental Dyslexia: Beyond the Visual Word Form Area. IOVS Vis. Neurosci. 2019, 60, 1063–1068. [Google Scholar]
- Tarkiainen, A.; Helenius, P.; Hansen, P.C.; Cornelissen, P.L.; Salmelin, R. Dynamics of letter string perception in the human occipitotemporal cortex. Brain 1999, 122, 2119–2131. [Google Scholar] [CrossRef] [PubMed]
- Polk, T.A.; Farah, M.J. Functional MRI evidence for an abstract, not perceptual, word-form area. J. Exp. Psychol. Gen. 2002, 131, 65–72. [Google Scholar] [CrossRef]
- Brem, S.; Maurer, U.; Kronbichler, M.; Schurz, M.; Richlan, F.; Blau, V.; Reithler, J.; van der Mark, S.; Schulz, E.; Bucher, K.; et al. Visual word form processing deficits driven by severity of reading impairments in children with developmental dyslexia. Sci. Rep. 2020, 10, 18728. [Google Scholar] [CrossRef]
- Cohen, L.; Dehaene, S. Specialization within the ventral stream: The case for the visual word form area. Neuroimage 2004, 22, 466–476. [Google Scholar] [CrossRef]
- Glezer, L.S.; Jiang, X.; Riesenhuber, M. Evidence for highly selective neuronal tuning to whole words in the “visual word form area”. Neuron 2009, 62, 199–204. [Google Scholar] [CrossRef]
- Saygin, Z.M.; Osher, D.E.; Norton, E.S.; Youssoufian, D.A.; Beach, S.D.; Feather, J.; Gaab, N.; Gabrieli, J.D.; Kanwisher, N. Connectivity precedes function in the development of the visual word form area. Nat. Neurosci. 2016, 19, 1250–1255. [Google Scholar] [CrossRef]
- Beauvois, M.F.; Dérouesné, J. Phonological alexia: Three dissociations. J. Neurol. Neurosurg. Psychiatry 1979, 42, 1115–1124. [Google Scholar] [CrossRef] [PubMed]
- Behrmann, M.; Nelson, J.; Sekuler, E.B. Visual complexity in letter-by-letter reading: “Pure” alexia is not pure. Neuropsychologia 1998, 36, 1115–1132. [Google Scholar] [CrossRef]
- Hillis, A.E.; Newhart, M.; Heidler, J.; Barker, P.; Herskovits, E.; Degaonkar, M. The roles of the “visual word form area” in reading. Neuroimage 2005, 24, 548–559. [Google Scholar] [CrossRef]
- Gaillard, R.; Naccache, L.; Pinel, P.; Clémenceau, S.; Volle, E.; Hasboun, D.; Dupont, S.; Baulac, M.; Dehaene, S.; Adam, C.; et al. Direct intracranial, FMRI, and lesion evidence for the causal role of left inferotemporal cortex in reading. Neuron 2006, 50, 191–204. [Google Scholar] [CrossRef]
- Henry, C.; Gaillard, R.; Volle, E.; Chiras, J.; Ferrieux, S.; Dehaene, S.; Cohen, L. Brain activations during letter-by-letter reading: A follow-up study. Neuropsychologia 2005, 43, 1983–1989. [Google Scholar] [CrossRef] [PubMed]
- Richlan, F.; Sturm, D.; Schurz, M.; Kronbichler, M.; Ladurner, G.; Wimmer, H. A common left occipito-temporal dysfunction in developmental dyslexia and acquired letter-by-letter reading? PLoS ONE 2010, 5, e12073. [Google Scholar] [CrossRef]
- Seghier, M.L.; Neufeld, N.H.; Zeidman, P.; Leff, A.P.; Mechelli, A.; Nagendran, A.; Riddoch, J.M.; Humphreys, G.W.; Price, C.J. Reading without the left ventral occipito-temporal cortex. Neuropsychologia 2012, 50, 3621–3635. [Google Scholar] [CrossRef]
- Horwitz, B.; Rumsey, J.M.; Donohue, B.C. Functional connectivity of the angular gyrus in normal reading and dyslexia. Proc. Natl. Acad. Sci. USA 1998, 95, 8939–8944. [Google Scholar] [CrossRef]
- Cao, F.; Bitan, T.; Booth, J.R. Effective brain connectivity in children with reading difficulties during phonological processing. Brain Lang. 2008, 107, 91–101. [Google Scholar] [CrossRef] [PubMed]
- Blau, V.; van Atteveldt, N.; Ekkebus, M.; Goebel, R.; Blomert, L. Reduced neural integration of letters and speech sounds links phonological and reading deficits in adult dyslexia. Curr. Biol. 2009, 19, 503–508. [Google Scholar] [CrossRef]
- Glezer, L.S.; Eden, G.; Jiang, X.; Luetje, M.; Napoliello, E.; Kim, J.; Riesenhuber, M. Uncovering phonological and orthographic selectivity across the reading network using fMRI-RA. Neuroimage 2016, 138, 248–256. [Google Scholar] [CrossRef]
- Fiez, J.A.; Petersen, S.E. Neuroimaging studies of word reading. Proc. Natl. Acad. Sci. USA 1998, 95, 914–921. [Google Scholar] [CrossRef] [PubMed]
- Price, C.J. The anatomy of language: Contributions from functional neuroimaging. J. Anat. 2000, 197, 335–359. [Google Scholar] [CrossRef]
- Paulesu, E.; Danelli, L.; Berlingeri, M. Reading the dyslexic brain: Multiple dysfunctional routes revealed by a new meta-analysis of PET and fMRI activation studies. Front. Hum. Neurosci. 2014, 8, 830. [Google Scholar] [CrossRef] [PubMed]
- Dickens, J.V.; Fama, M.E.; DeMarco, A.T.; Lacey, E.H.; Friedman, R.B.; Turkeltaub, P.E. Localization of Phonological and Semantic Contributions to Reading. J. Neurosci. 2019, 39, 5361–5368. [Google Scholar] [CrossRef] [PubMed]
- Mechelli, A.; Crinion, J.T.; Long, S.; Friston, K.J.; Ralph, M.A.L.; Patterson, K.; McClelland, J.L.; Price, C.J. Dissociating reading processes on the basis of neuronal interactions. J. Cogn. Neurosci. 2005, 17, 1753–1765. [Google Scholar] [CrossRef]
- Maisog, J.M.; Einbinder, E.R.; Flowers, D.L.; Turkeltaub, P.E.; Eden, G.F. A meta-analysis of functional neuroimaging studies of dyslexia. Ann. N. Y. Acad. Sci. 2008, 1145, 237–259. [Google Scholar] [CrossRef]
- Werth, R. Visual functions without the occipital lobe or after cerebral hemispherectomy in infancy. Eur. J. Neurosci. 2006, 24, 2932–2944. [Google Scholar] [CrossRef]
- Werth, R. Cerebral blindness and plasticity of the visual system in children. A review of visual capacities in patients with occipital lesions, hemispherectomy or hydranencephaly. Restor. Neurol. Neurosci. 2008, 26, 377–389. [Google Scholar] [PubMed]
- Starrfelt, R.; Habekost, T.; Leff, A.P. Too little, too late: Reduced visual span and speed characterize pure alexia. Cereb. Cortex 2009, 19, 2880–2890. [Google Scholar] [CrossRef]
- Mauthner, L. Gehirn und Auge; J.F. Bergmann: Wiesbaden, Germany, 1881. [Google Scholar]
- Wilbrand, H. Ueber die makulär-hemianopische Lesestörung und die von Monakow’sche Projektion der Makula auf die Sehsphäre. Klin. Monatsbl. Augenheilkd. 1907, 45, 1–39. [Google Scholar]
- Mackensen, G. Die Untersuchung der Lesefähigkeit als klinische Funktionsprüfung. Fortschr. Augenheilkd. 1962, 12, 344–379. [Google Scholar]
- Zihl, J.; Krischer, C.; Meißen, R. Die hemianopische Lesestörung und ihre Behandlung. Nervenarzt 1984, 55, 317–323. [Google Scholar]
- Bálint, D. Seelenlähmung des “Schauens”, optische Ataxie, räumliche Störung der Aufmerksamkeit. Mschr. Psychiat. Neurol. 1909, 25, 51–81. [Google Scholar] [CrossRef]
- Poppelreuter, W. Die Psychischen Schädigungen durch Kopfschuß im Kriege 1914–1918. Bd. I. Die Störungen der Niederen und Höheren Sehleistungen durch Verletzungen des Okzipitalhirns; L. Voss: Leipzig, Germany, 1917. [Google Scholar]
- Luria, A.R. Disorders of “simultaneous perception” in a case of bilateral occipito-parietal brain injury. Brain 1959, 82, 437–449. [Google Scholar] [CrossRef] [PubMed]
- Baylis, G.C.; Driver, J.; Baylis, L.L.; Rafal, R.D. Reading of letters and words in a patient with Balint’s syndrome. Neuropsychologia 1994, 32, 1273–1286. [Google Scholar] [CrossRef]
- Rizzo, M.; Vecera, S.P. Psychoanatomical substrates of Bálint’s syndrome. J. Neurol. Neurosurg. Psychiatry 2002, 72, 162–178. [Google Scholar] [CrossRef]
- Moreaud, O. Balint syndrome. Arch. Neurol. 2003, 60, 1329–1331. [Google Scholar] [CrossRef] [PubMed]
- Michel, F.; Henaff, M.A. Seeing without the occipito-parietal cortex: Simultagnosia as a shrinkage of the attentional visual field. Behav. Neurol. 2004, 15, 3–13. [Google Scholar] [CrossRef] [PubMed]
- Farah, M.J. Visual Agnosia, 2nd ed.; MIT Press: Cambridge, MA, USA; London, UK, 2004; pp. 43–58. [Google Scholar]
- Chechlacz, M.; Rotshtein, P.; Hansen, P.C.; Riddoch, J.M.; Deb, S.; Humphreys, G.W. The neural underpinings of simultanagnosia: Disconnecting the visuospatial attention network. J. Cogn. Neurosci. 2012, 247, 18–35. [Google Scholar] [CrossRef] [PubMed]
- Chechlacz, M.; Humphreys, G.W. The enigma of Balint’s syndrome: Neural substrates and cognitive deficits. Front. Hum. Neurosci. 2014, 8, 123. [Google Scholar] [CrossRef] [PubMed]
- Chechlacz, M. Bilateral parietal dysfunctions and disconnections in simultanagnosia and Bálint syndrome. Handb. Clin. Neurol. 2018, 151, 249–267. [Google Scholar] [CrossRef] [PubMed]
- Faust, C. Über Gestaltzerfall als Symptom des parieto-okzipitalen Übergangsbereichs bei doppelseitiger Verletzung nach Hirnschuss. Nervenarzt 1947, 18, 103–114. [Google Scholar] [PubMed]

| Fixation Time Milliseconds | Number of Letters Recognized | |||
|---|---|---|---|---|
| 3 Letters | 4 Letters | 5 Letters | 6 Letters | |
| Number of Subjects Who Recognized ≥ 95% of the Pseudowords Correctly | ||||
| 250 ms | 24 | 30 | 28 | 11 |
| 300 ms | 7 | 4 | 5 | 0 |
| 350 ms | 4 | 9 | 11 | 0 |
| 400 ms | 9 | 20 | 3 | 6 |
| 450 ms | 5 | 1 | 1 | 0 |
| 500 ms | 4 | 4 | 3 | 0 |
| ∑Subjects | 52 | 80 | 51 | 17 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Werth, R. Is Developmental Dyslexia Due to a Visual and Not a Phonological Impairment? Brain Sci. 2021, 11, 1313. https://doi.org/10.3390/brainsci11101313
Werth R. Is Developmental Dyslexia Due to a Visual and Not a Phonological Impairment? Brain Sciences. 2021; 11(10):1313. https://doi.org/10.3390/brainsci11101313
Chicago/Turabian StyleWerth, Reinhard. 2021. "Is Developmental Dyslexia Due to a Visual and Not a Phonological Impairment?" Brain Sciences 11, no. 10: 1313. https://doi.org/10.3390/brainsci11101313
APA StyleWerth, R. (2021). Is Developmental Dyslexia Due to a Visual and Not a Phonological Impairment? Brain Sciences, 11(10), 1313. https://doi.org/10.3390/brainsci11101313





