The Effects of Spinal Manipulation on Motor Unit Behavior
Abstract
1. Introduction
2. Materials and Methods
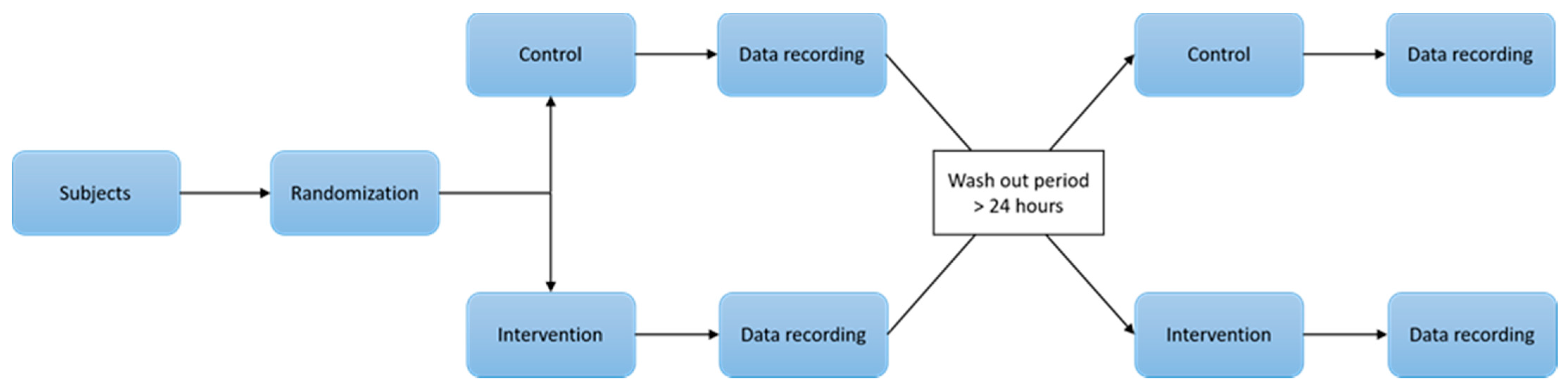
2.1. Experimental Design and Setting
2.2. Participants
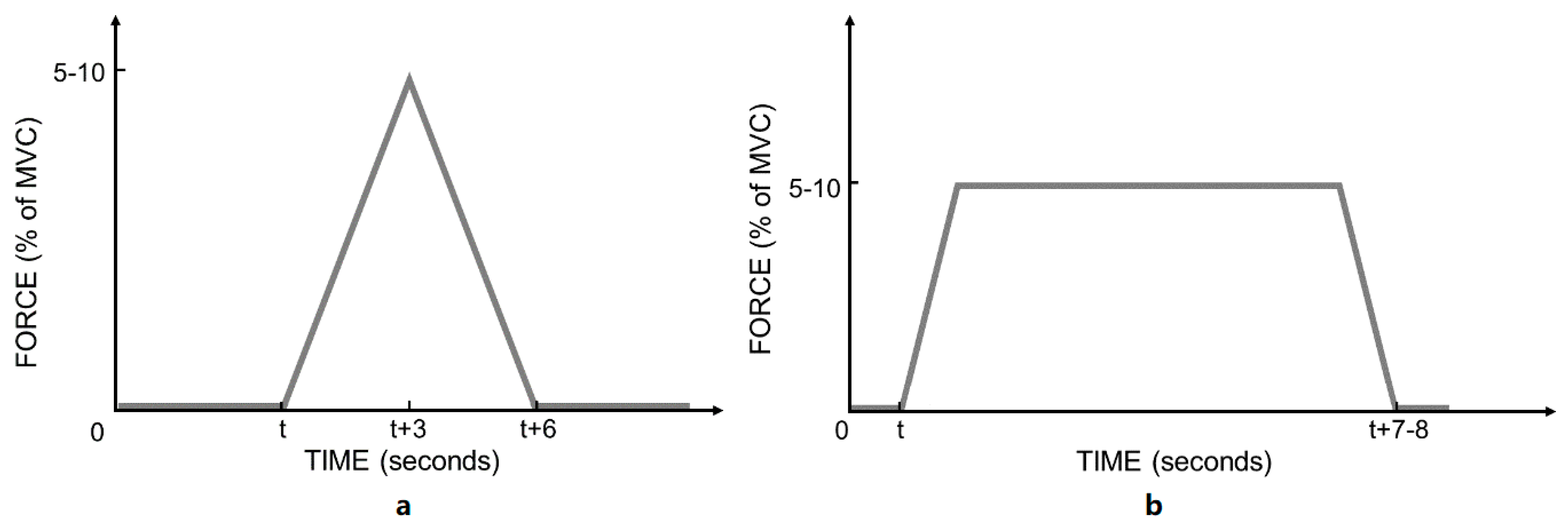
2.3. Spinal Manipulation and Sham Intervention
2.4. Procedure
2.5. Equipment
2.5.1. Ankle Ergometer
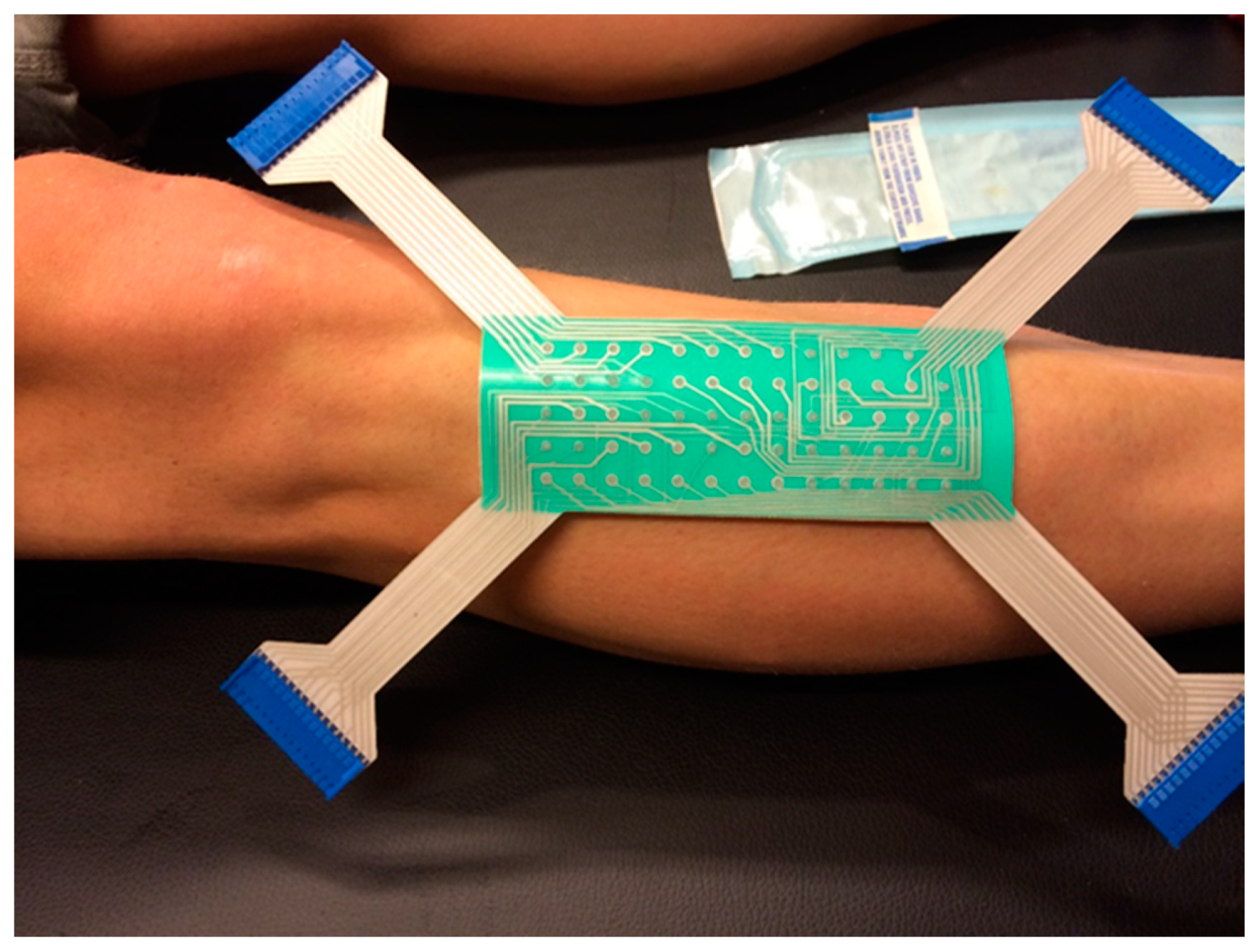
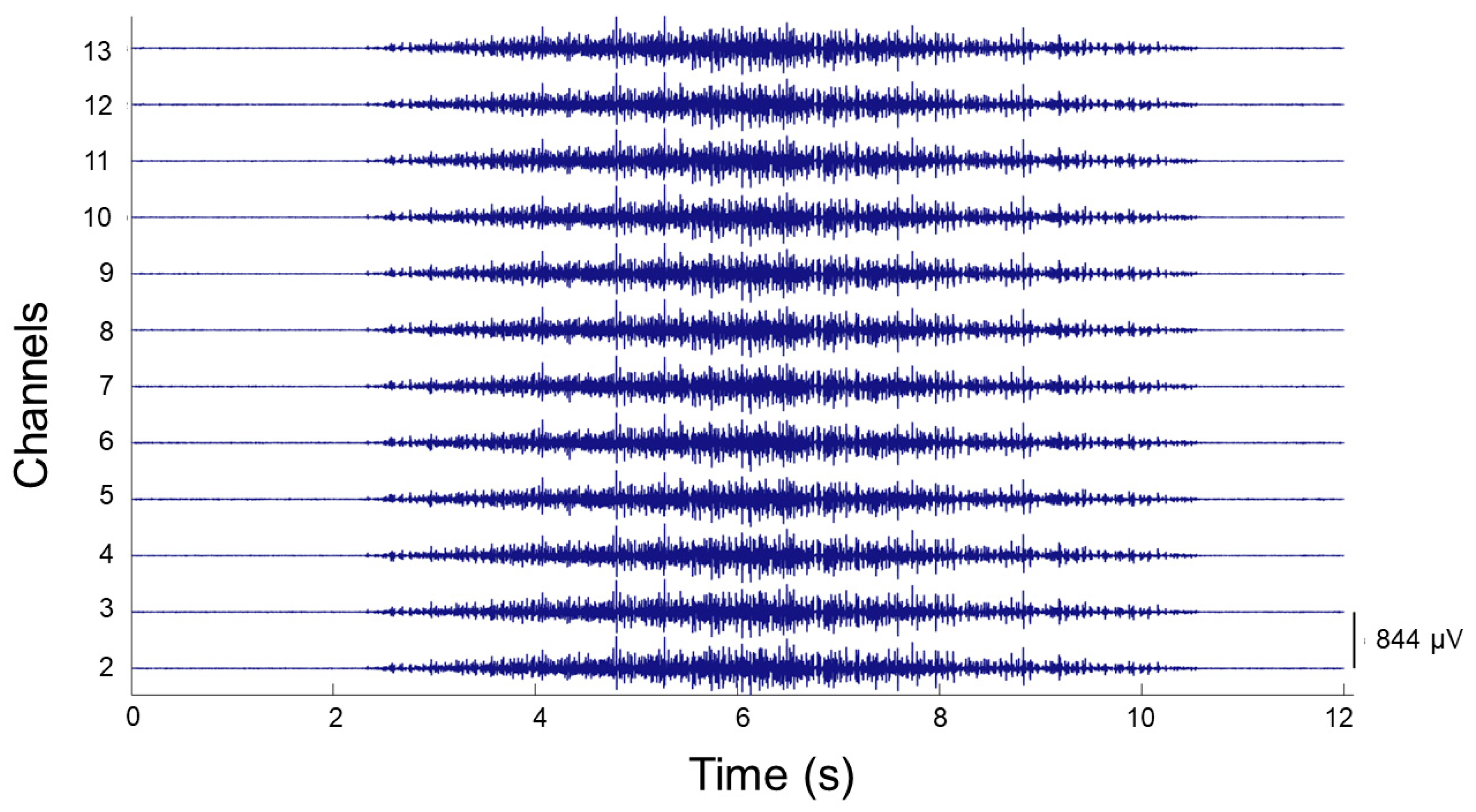
2.5.2. HD EMG
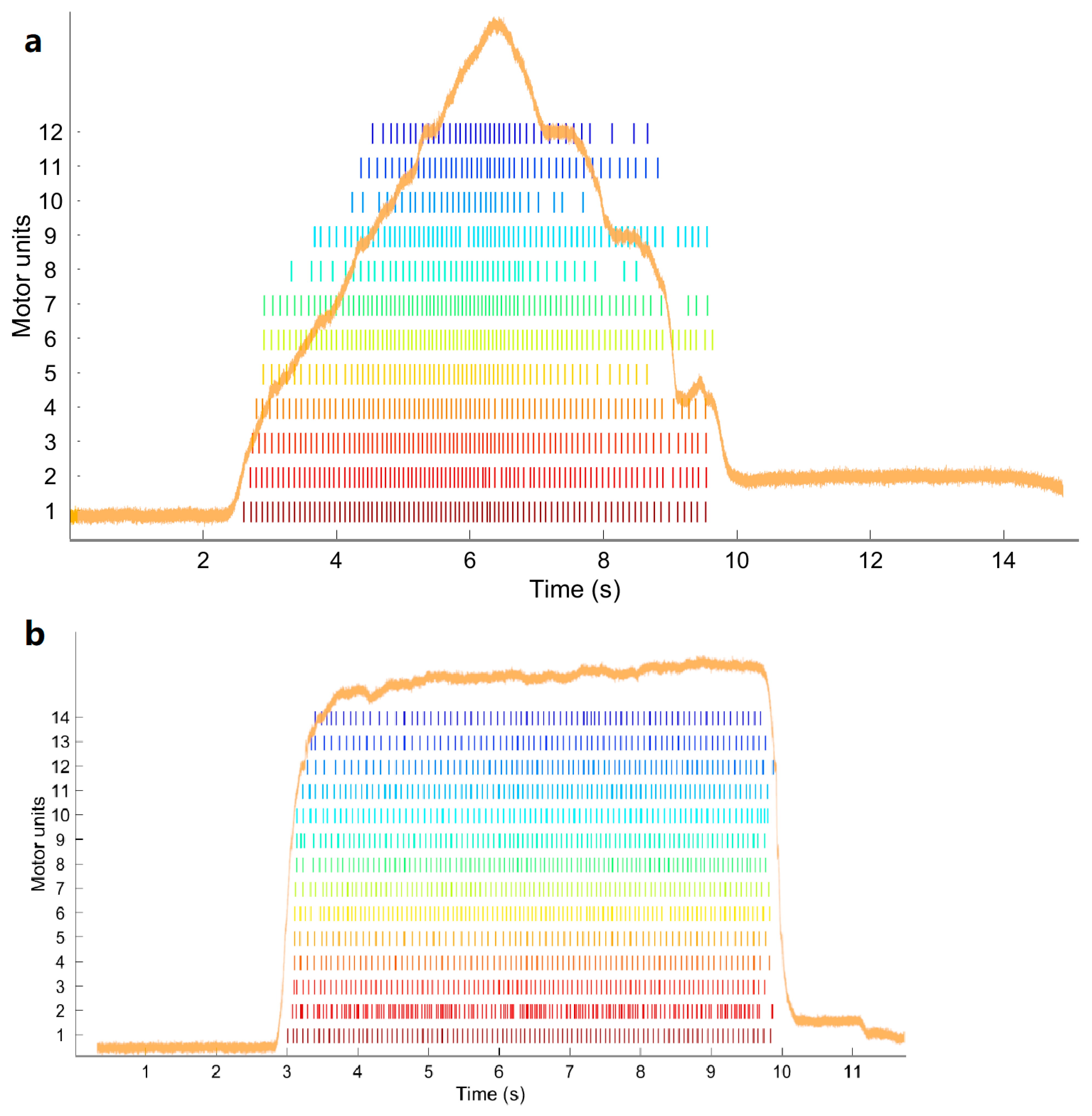
2.6. Data Processing
- Conduction velocity (CV): The speed which an action potential travels along the membrane of a skeletal muscle fiber [68]. The estimation of the conduction velocity was calculated using the techniques described by Farina et al. [69,70], which use a maximum likelihood multiple-channel method, using the Newton method for efficient optimization. Coefficient of correlation (CC) was used to measure the goodness of match of MUAPs on neighboring HDsEMG channels and, thus, the accuracy of CV estimation. Only the CV values with corresponding CC ≥0.7 were used for the statistical analysis.
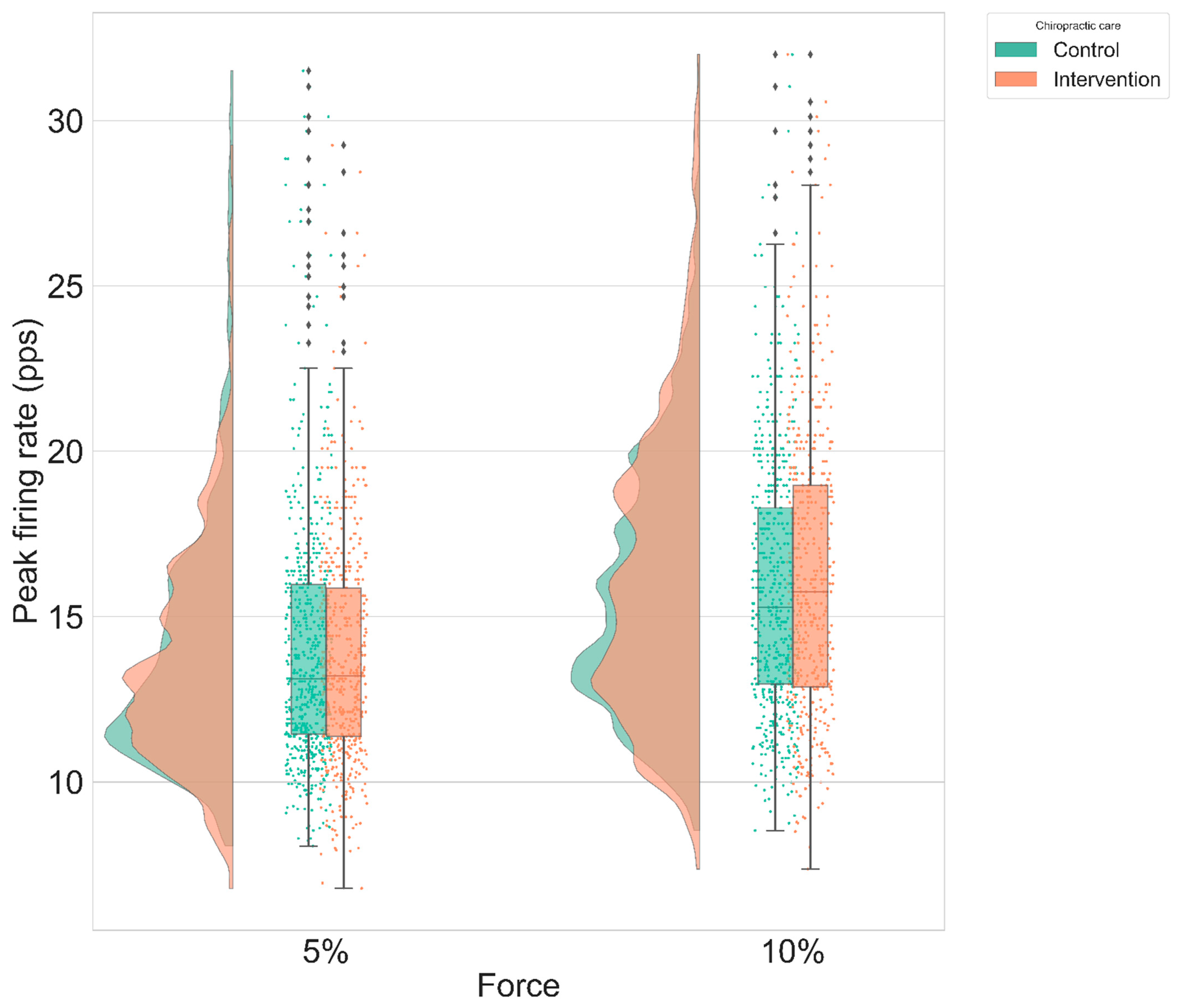
- Peak firing rate (PFR): The PFR of a MU was defined as the maximum instantaneous firing rate value for the MU. The instantaneous firing rate (IFR) of each MU was calculated:with in pps, and and in seconds.
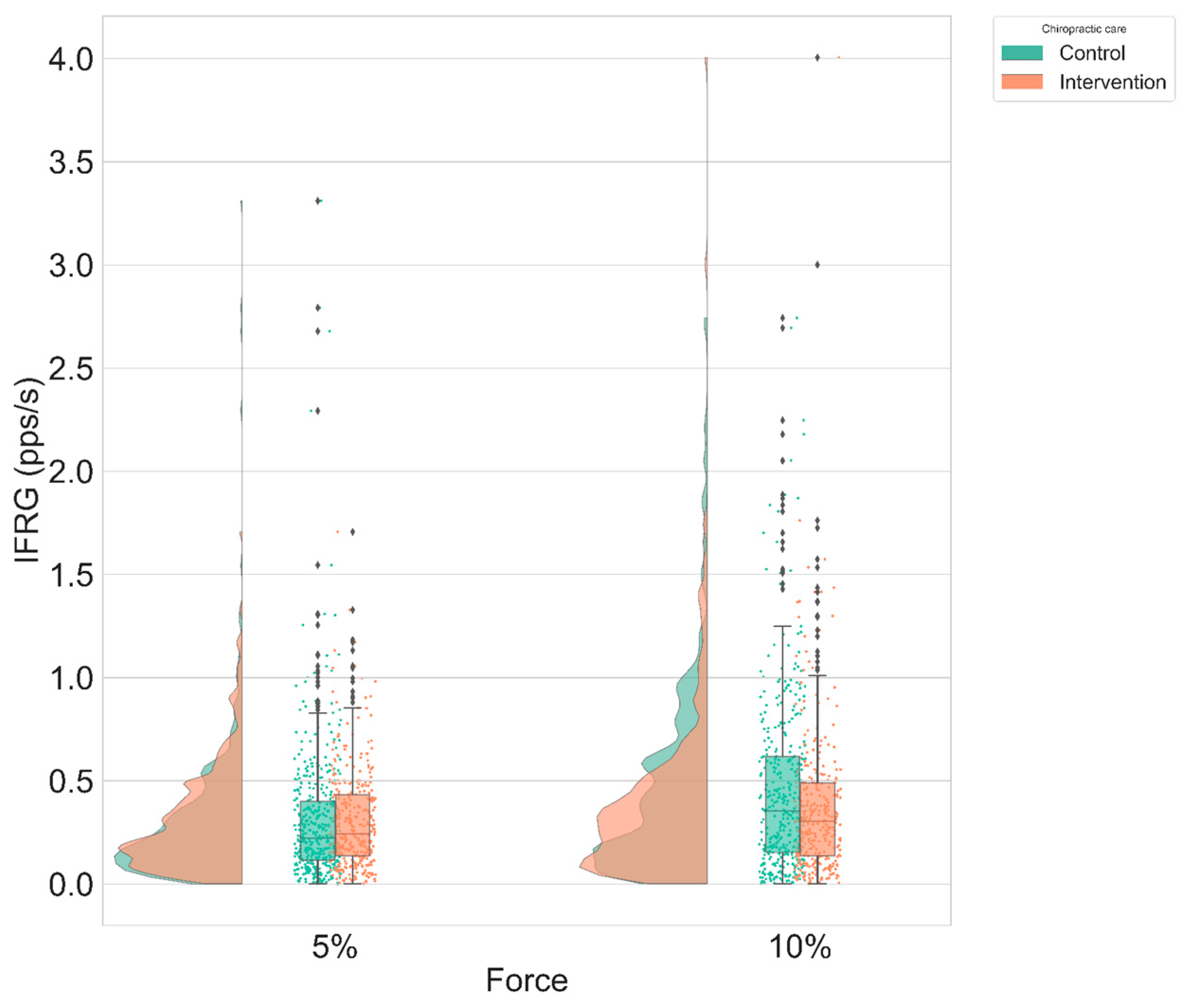
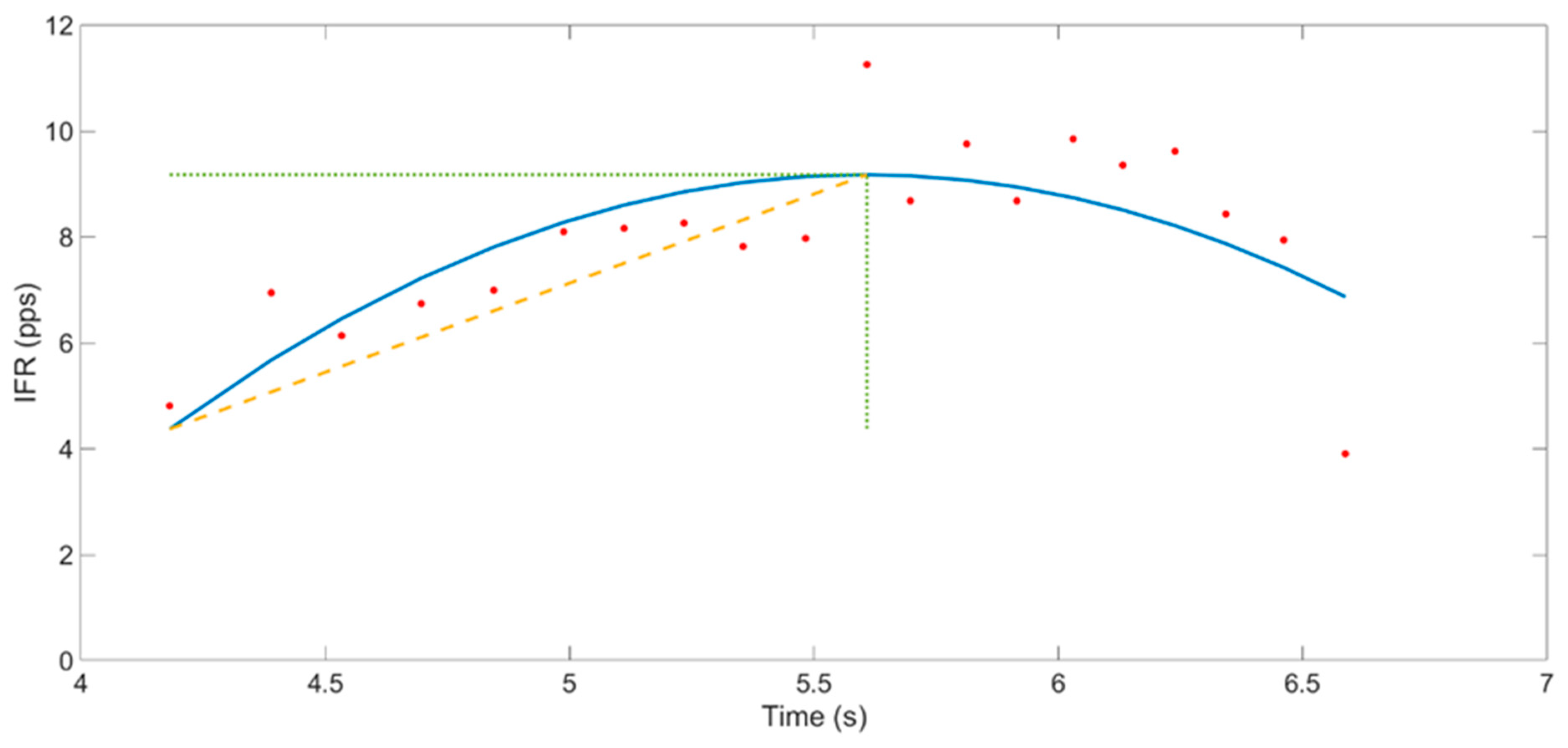
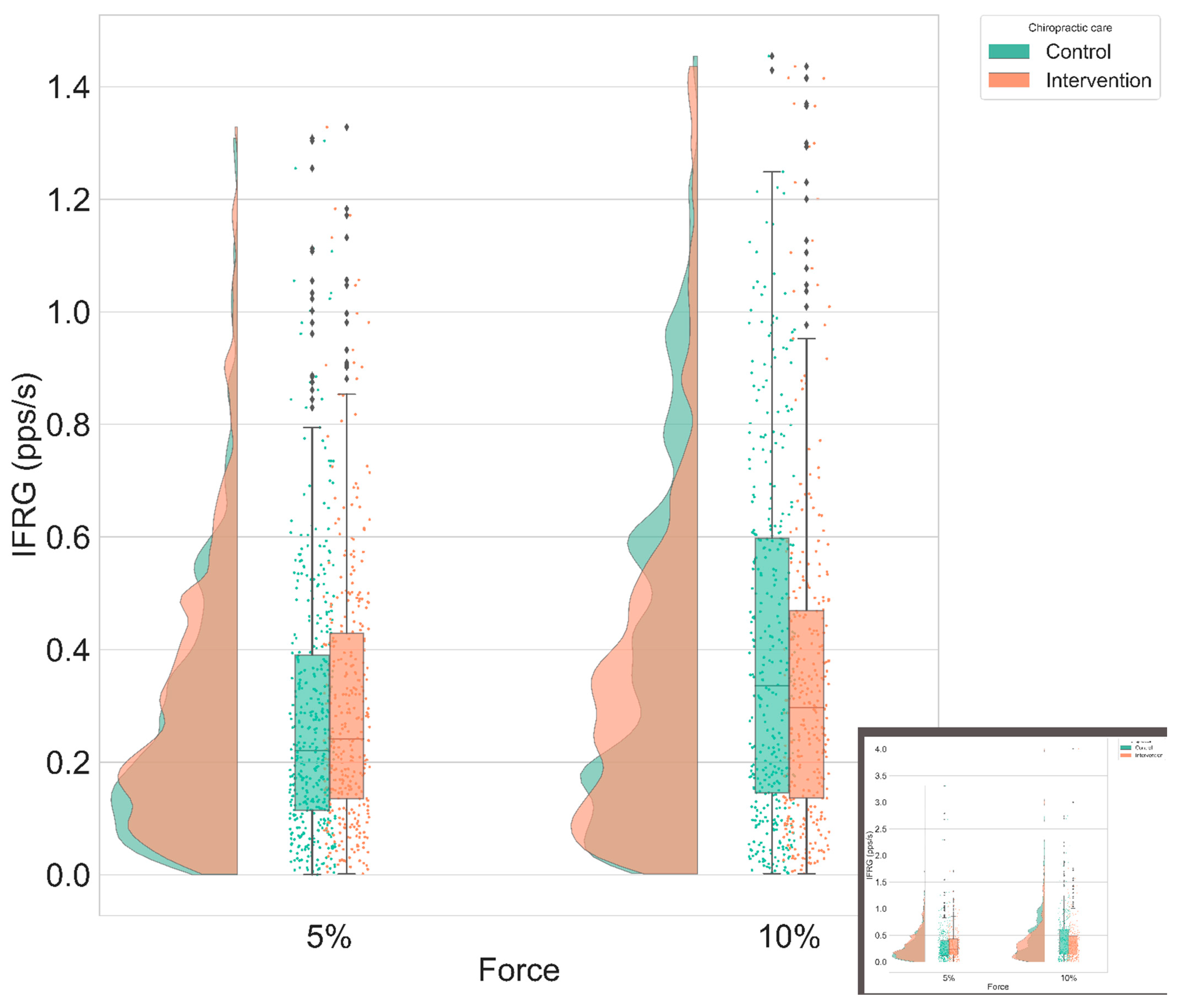
- Instantaneous MU firing rate gradient (IFRG): The IFRG is the slope of linear approximation of MU firing rate increase from recruitment to its peak IFR value (Figure 6). The following technique was used to compute this value: a polynomial of the second order was fitted on the instantaneous firing rate curve of the MU, using the polyfit() function from MATLAB 2018a (MathWorks, Natick, MA, United States). Then, a linear function was fitted on the first value and the peak value of this polynomial. The slope of the linear expression defined the IFRG. In each participant, IFRG values were averaged across contraction repetitions and across condition and intensity before being statistically analyzed.
2.7. Statistical Analysis
3. Results
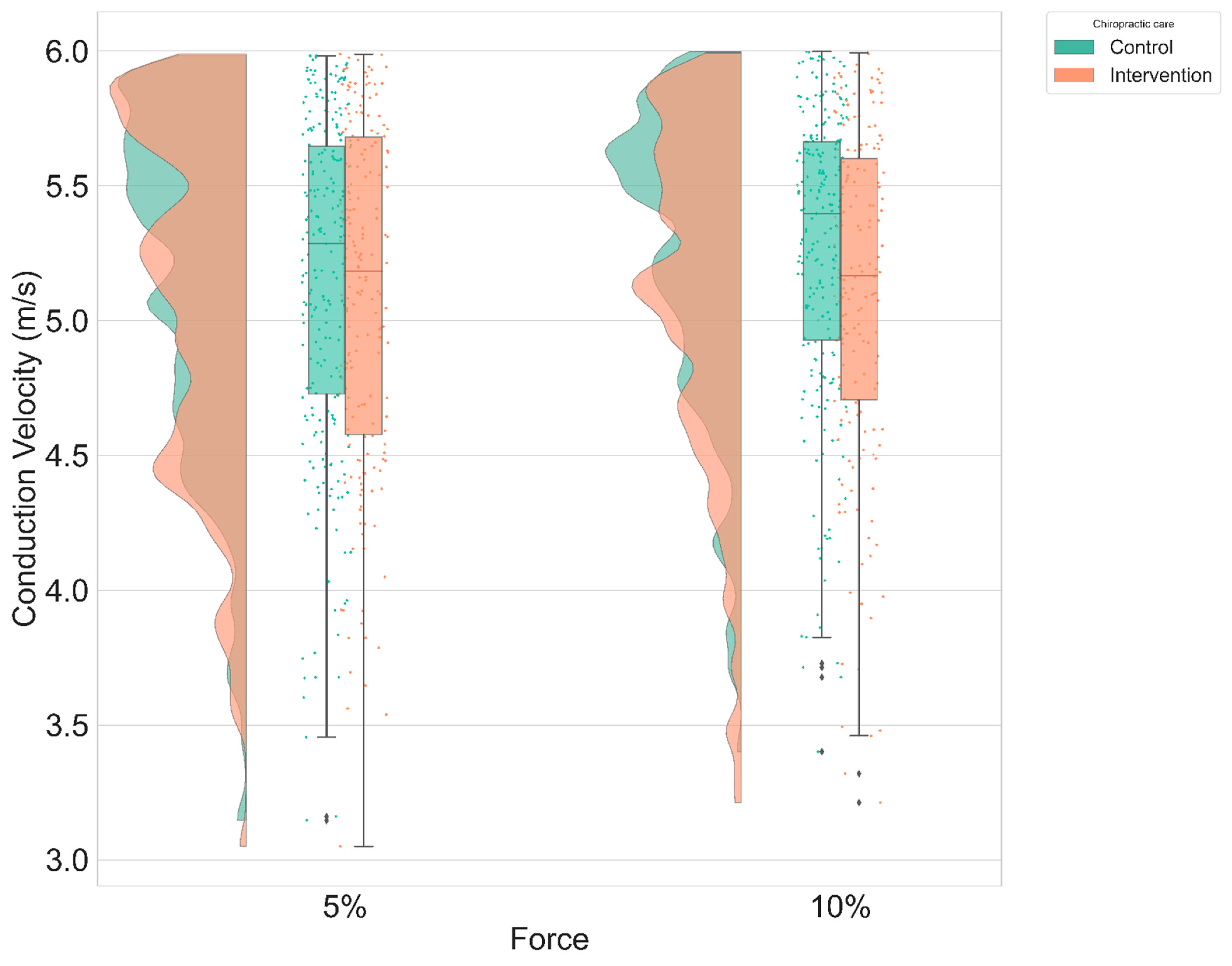
3.1. Conduction Velocity
3.2. Instantaneous MU Firing Rate Gradient & Peak Firing Rate
4. Discussion
5. Limitation
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
References
- Jenkins, H.J.; Downie, A.S.; Moore, C.S.; French, S.D. Current evidence for spinal X-ray use in the chiropractic profession: A narrative review. Chiropr. Man. Therap. 2018, 26, 48. [Google Scholar] [CrossRef] [PubMed]
- McGill, S.M. Stability: From biomechanical concept to chiropractic practice. J. Can. Chiropr. Assoc. 1999, 43, 75–88. [Google Scholar]
- Christiansen, T.L.; Niazi, I.K.; Holt, K.; Nedergaard, R.W.; Duehr, J.; Allen, K.; Marshall, P.; Türker, K.S.; Hartvigsen, J.; Haavik, H. The effects of a single session of spinal manipulation on strength and cortical drive in athletes. Eur. J. Appl. Physiol. 2018, 118, 737–749. [Google Scholar] [CrossRef] [PubMed]
- Haavik, H.; Murphy, B. The role of spinal manipulation in addressing disordered sensorimotor integration and altered motor control. J. Electromyogr. Kinesiol. 2012, 22, 768–776. [Google Scholar] [CrossRef]
- Haavik, H.; Murphy, B. Subclinical Neck Pain and the Effects of Cervical Manipulation on Elbow Joint Position Sense. J. Manip. Physiol. Ther. 2011, 34, 88–97. [Google Scholar] [CrossRef]
- Holt, K.R.; Haavik, H.; Lee, A.C.L.; Murphy, B.; Elley, C.R. Effectiveness of Chiropractic Care to Improve Sensorimotor Function Associated With Falls Risk in Older People: A Randomized Controlled Trial. J. Manip. Physiol. Ther. 2016, 39, 267–278. [Google Scholar] [CrossRef] [PubMed]
- Holt, K.; Niazi, I.K.; Nedergaard, R.W.; Duehr, J.; Amjad, I.; Shafique, M.; Anwar, M.N.; Ndetan, H.; Turker, K.S.; Haavik, H. The effects of a single session of chiropractic care on strength, cortical drive, and spinal excitability in stroke patients. Sci. Rep. 2019, 9, 2673. [Google Scholar] [CrossRef]
- Daligadu, J.; Haavik, H.; Yielder, P.C.; Baarbe, J.; Murphy, B. Alterations in Cortical and Cerebellar Motor Processing in Subclinical Neck Pain Patients Following Spinal Manipulation. J. Manip. Physiol. Ther. 2013, 36, 527–537. [Google Scholar] [CrossRef]
- Haavik, H.; Murphy, B. Transient modulation of intracortical inhibition following spinal manipulation. Chiropr. J. Aust. 2007, 37, 106–116. [Google Scholar]
- Haavik, H.; Niazi, I.; Jochumsen, M.; Sherwin, D.; Flavel, S.; Türker, K. Impact of Spinal Manipulation on Cortical Drive to Upper and Lower Limb Muscles. Brain Sci. 2016, 7, 2. [Google Scholar] [CrossRef]
- Taylor, H.H.; Murphy, B. The Effects of Spinal Manipulation on Central Integration of Dual Somatosensory Input Observed After Motor Training: A Crossover Study. J. Manip. Physiol. Ther. 2010, 33, 261–272. [Google Scholar] [CrossRef] [PubMed]
- Mauguiere, F. Somatosensory evoked potentials: Normal responses, abnormal waveforms, and clinical applications in neurological disease. In Electroencephalography. Basic Principles, Clinical Applications, and Related Fields; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2005; pp. 1067–1119. [Google Scholar]
- Taylor, H.H.; Murphy, B. Altered Central Integration of Dual Somatosensory Input After Cervical Spine Manipulation. J. Manip. Physiol. Ther. 2010, 33, 178–188. [Google Scholar] [CrossRef] [PubMed]
- Taylor, H.H.; Murphy, B. Altered Sensorimotor Integration with Cervical Spine Manipulation. J. Manip. Physiol. Ther. 2008, 31, 115–126. [Google Scholar] [CrossRef]
- Mendonca, G.V.; Teodósio, C.; Mouro, M.; Freitas, S.; Mil-Homens, P.; Pezarat-Correia, P.; Vila-Chã, C. Improving the Reliability of V-Wave Responses in the Soleus Muscle. J. Clin. Neurophysiol. 2019, 36, 97–103. [Google Scholar] [CrossRef] [PubMed]
- Mills, K. Oxford Textbook of Clinical Neurophysiology; Oxford University Press: Oxford, UK, 2017. [Google Scholar]
- Triccas, L.T.; Meyer, S.; Mantini, D.; Camilleri, K.; Falzon, O.; Camilleri, T.; Verheyden, G. A systematic review investigating the relationship of electroencephalography and magnetoencephalography measurements with sensorimotor upper limb impairments after stroke. J. Neurosci. Methods 2019, 311, 318–330. [Google Scholar] [CrossRef] [PubMed]
- Navid, M.S.; Niazi, I.K.; Lelic, D.; Nedergaard, R.B.; Holt, K.; Amjad, I.; Drewes, A.M.; Haavik, H. Investigating the Effects of Chiropractic Spinal Manipulation on EEG in Stroke Patients. Brain Sci. 2020, 10, 253. [Google Scholar] [CrossRef] [PubMed]
- Niazi, I.K.; Türker, K.S.; Flavel, S.; Kinget, M.; Duehr, J.; Haavik, H. Changes in H-reflex and V-waves following spinal manipulation. Exp. Brain Res. 2015, 233, 1165–1173. [Google Scholar] [CrossRef]
- Holobar, A.; Glaser, V.; Gallego, J.A.; Dideriksen, J.L.; Farina, D. Non-invasive characterization of motor unit behaviour in pathological tremor. J. Neural Eng. 2012, 9, 056011. [Google Scholar] [CrossRef]
- Radeke, J.; van Dijk, J.P.; Holobar, A.; Lapatki, B.G. Electrophysiological method to examine muscle fiber architecture in the upper lip in cleft-lip patients. J. Orofac. Orthop. 2014, 75, 51–61. [Google Scholar] [CrossRef]
- Perry, J.; Easterday, C.S.; Antonelli, D.J. Surface Versus Intramuscular Electrodes for Electromyography of Superficial and Deep Muscles. Phys. Ther. 1981, 61, 7–15. [Google Scholar] [CrossRef]
- Farina, D.; Holobar, A.; Merletti, R.; Enoka, R.M. Decoding the neural drive to muscles from the surface electromyogram. Clin. Neurophysiol. 2010, 121, 1616–1623. [Google Scholar] [CrossRef] [PubMed]
- Gazzoni, M.; Farina, D.; Merletti, R. A new method for the extraction and classification of single motor unit action potentials from surface EMG signals. J. Neurosci. Methods 2004, 136, 165–177. [Google Scholar] [CrossRef]
- Hogrel, J.-Y. Use of surface EMG for studying motor unit recruitment during isometric linear force ramp. J. Electromyogr. Kinesiol. 2003, 13, 417–423. [Google Scholar] [CrossRef]
- Holobar, A.; Zazula, D. Multichannel Blind Source Separation Using Convolution Kernel Compensation. IEEE Trans. Signal. Process. 2007, 55, 4487–4496. [Google Scholar] [CrossRef]
- Merletti, R. Surface Electromyography, 1st ed.; Wiley-IEEE Press: Piscataway, NJ, USA, 2016. [Google Scholar]
- Kleine, B.U.; van Dijk, J.P.; Lapatki, B.G.; Zwarts, M.J.; Stegeman, D.F. Using two-dimensional spatial information in decomposition of surface EMG signals. J. Electromyogr. Kinesiol. 2007, 17, 535–548. [Google Scholar] [CrossRef]
- Nawab, S.H.; Chang, S.-S.; De Luca, C.J. High-yield decomposition of surface EMG signals. Clin. Neurophysiol. 2010, 121, 1602–1615. [Google Scholar] [CrossRef] [PubMed]
- Vila-Chã, C.; Falla, D.; Correia, M.V.; Farina, D. Adjustments in Motor Unit Properties during Fatiguing Contractions after Training. Med. Sci. Sports Exerc. 2012, 44, 616–624. [Google Scholar] [CrossRef]
- Duchateau, J.; Semmler, J.; Enoka, R. Training adaptations in the behavior of human motor units. J. Appl. Physiol. 2006, 101, 1766–1775. [Google Scholar] [CrossRef]
- Vila-Chã, C.; Falla, D.; Farina, D. Motor unit behavior during submaximal contractions following six weeks of either endurance or strength training. J. Appl. Physiol. 2010, 109, 1455–1466. [Google Scholar] [CrossRef]
- Deschenes, M. Short review: Rate coding and motor unit recruitment patterns. J. Appl. Sport Sci. Res. 1989, 3, 33–39. [Google Scholar] [CrossRef]
- Merletti, R.; Holobar, A.; Farina, D. Analysis of motor units with high-density surface electromyography. J. Electromyogr. Kinesiol. 2008, 18, 879–890. [Google Scholar] [CrossRef] [PubMed]
- Andreassen, S.; Arendt-Nielsen, L. Muscle fibre conduction velocity in motor units of the human anterior tibial muscle: A new size principle parameter. J. Physiol. 1987, 391, 561–571. [Google Scholar] [CrossRef] [PubMed]
- Andrew, D.; Yielder, P.; Haavik, H.; Murphy, B. The effects of subclinical neck pain on sensorimotor integration following a complex motor pursuit task. Exp. Brain Res. 2018, 236, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Baarbé, J.; Yielder, P.; Haavik, H.; Debison-Larabie, C.; Holmes, M.; Murphy, B. Enhanced cerebellar disinhibition when cervical manipulation precedes motor learning in patients with subclinical neck pain. J. Chiropr. Med. 2015, 14, 115–116. [Google Scholar]
- Baarbé, J.K.; Holmes, M.W.R.; Murphy, H.E.; Haavik, H.; Murphy, B.A. Influence of Subclinical Neck Pain on the Ability to Perform a Mental Rotation Task: A 4-Week Longitudinal Study with a Healthy Control Group Comparison. J. Manip. Physiol. Ther. 2016, 39, 23–30. [Google Scholar] [CrossRef]
- Baarbé, J.K.; Yielder, P.; Haavik, H.; Holmes, M.W.R.; Murphy, B.A. Subclinical recurrent neck pain and its treatment impacts motor training-induced plasticity of the cerebellum and motor cortex. PLoS ONE 2018, 13, e0193413. [Google Scholar] [CrossRef]
- Farid, B.; Yielder, P.; Holmes, M.; Haavik, H.; Murphy, B.A. Association of Subclinical Neck Pain with Altered Multisensory Integration at Baseline and 4-Week Follow-up Relative to Asymptomatic Controls. J. Manip. Physiol. Ther. 2018, 41, 81–91. [Google Scholar] [CrossRef]
- Lee, H.; Nicholson, L.L.; Adams, R.D.; Bae, S.-S. Proprioception and Rotation Range Sensitization Associated with Subclinical Neck Pain. Spine 2005, 30, E60–E67. [Google Scholar] [CrossRef]
- Lee, H.-Y.; Wang, J.-D.; Yao, G.; Wang, S.-F. Association between cervicocephalic kinesthetic sensibility and frequency of subclinical neck pain. Man. Ther. 2008, 13, 419–425. [Google Scholar] [CrossRef]
- Paulus, I.; Brumagne, S. Altered interpretation of neck proprioceptive signals in persons with subclinical recurrent neck pain. J. Rehabil. Med. 2008, 40, 426–432. [Google Scholar] [CrossRef]
- Rossi, S.; della Volpe, R.; Ginanneschi, F.; Ulivelli, M.; Bartalini, S.; Spidalieri, R.; Rossi, A. Early somatosensory processing during tonic muscle pain in humans: Relation to loss of proprioception and motor ‘defensive’ strategies. Clin. Neurophysiol. 2003, 114, 1351–1358. [Google Scholar] [CrossRef]
- Hawker, G.A.; Mian, S.; Kendzerska, T.; French, M. Measures of adult pain: Visual Analog Scale for Pain (VAS Pain), Numeric Rating Scale for Pain (NRS Pain), McGill Pain Questionnaire (MPQ), Short-Form McGill Pain Questionnaire (SF-MPQ), Chronic Pain Grade Scale (CPGS), Short Form-36 Bodily Pain Scale (SF). Arthritis Care Res. 2011, 63, S240–S252. [Google Scholar] [CrossRef]
- Triano, J.J.; Budgell, B.; Bagnulo, A.; Roffey, B.; Bergmann, T.; Cooperstein, R.; Gleberzon, B.; Good, C.; Perron, J.; Tepe, R. Review of methods used by chiropractors to determine the site for applying manipulation. Chiropr. Man. Therap. 2013, 21, 36. [Google Scholar] [CrossRef] [PubMed]
- WHO. WHO Guidelines on Basic Training and Safety in Chiropractic; WHO: Geneva, Switzerland, 2005. [Google Scholar]
- The Rubicon Group Definition and Position Statement on the Chiropractic Subluxation. 2017. Available online: http://www.therubicongroup.org/#/policies/ (accessed on 10 December 2020).
- Holt, K.; Russell, D.; Cooperstein, R.; Young, M.; Sherson, M.; Haavik, H. Interexaminer reliability of a multidimensional battery of tests used to assess for vertebral subluxations. Chiropr. J. Aust. 2018, 46, 100–117. [Google Scholar]
- Cooperstein, R.; Gleberzon, B. Technique Systems in Chiropractic; Churchill-Livingstone: New York, NY, USA, 2004. [Google Scholar]
- Hessell, B.; Herzog, W.; Conway, P.; McEwen, M. Experimental measurement of the force exerted during spinal manipulation using the Thompson technique. J. Manip. Physiol. Ther. 1990, 13, 448–453. [Google Scholar]
- Herzog, W.; Conway, P.; Zhang, Y.; Gál, J.; Guimaraes, A. Reflex responses associated with manipulative treatments on the thoracic spine: A pilot study. J. Manip. Physiol. Ther. 1995, 18, 233–236. [Google Scholar]
- Herzog, W. Mechanical, physiologic, and neuromuscular considerations of chiropractic treatments. Adv. Chiropr. 1996, 3, 69–285. [Google Scholar]
- Pickar, J.; Wheeler, J. Response of muscle proprioceptors to spinal manipulative-like loads in the anesthetized cat. J. Manip. Physiol. Ther. 2001, 24, 2–11. [Google Scholar] [CrossRef]
- Masakado, Y.; Nielsen, J.B. Task-and Phase-related Changes in Cortico-muscular Coherence. Keio J. Med. 2008, 57, 50–56. [Google Scholar] [CrossRef]
- Duchateau, J.; Enoka, R.M. Human motor unit recordings: Origins and insight into the integrated motor system. Brain Res. 2011, 1409, 42–61. [Google Scholar] [CrossRef]
- Conway, B.A.; Reid, C.; Halliday, D.M. Low Frequency Cortico-Muscular Coherence During Voluntary Rapid Movements of the Wrist Joint. Brain Topogr. 2003, 16, 221–224. [Google Scholar] [CrossRef] [PubMed]
- Farina, D.; Holobar, A. Characterization of Human Motor Units from Surface EMG Decomposition. Proc. IEEE. 2016, 104, 353–373. [Google Scholar] [CrossRef]
- OTBioelettronica. OT BioLab+; OTBioelettronica: Turin, Italy, 2017. [Google Scholar]
- Holobar, A. DEMUSE® Tool. 2020. Available online: https://demuse.feri.um.si/ (accessed on 10 March 2019).
- Holobar, A.; Zazula, D. Correlation-based decomposition of surface electromyograms at low contraction forces. Med. Biol. Eng. Comput. 2004, 42, 487–495. [Google Scholar] [CrossRef] [PubMed]
- Holobar, A.; Zazula, D. On the selection of the cost function for gradient-based decomposition of surface electromyograms. In Proceedings of the 2008 30th Annual International Conference of the IEEE Engineering in Medicine and Biology Society, Vancouver, BC, Canada, 20–25 August 2008; pp. 4668–4671. [Google Scholar] [CrossRef]
- Holobar, A.; Farina, D.; Gazzoni, M.; Merletti, R.; Zazula, D. Estimating motor unit discharge patterns from high-density surface electromyogram. Clin. Neurophysiol. 2009, 120, 551–562. [Google Scholar] [CrossRef] [PubMed]
- Minetto, M.A.; Holobar, A.; Botter, A.; Farina, D. Discharge Properties of Motor Units of the Abductor Hallucis Muscle during Cramp Contractions. J. Neurophysiol. 2009, 102, 1890–1901. [Google Scholar] [CrossRef]
- Grassi, B.; Quaresima, V. Near-infrared spectroscopy and skeletal muscle oxidative function in vivo in health and disease: A review from an exercise physiology perspective. J. Biomed. Opt. 2016, 21, 091313. [Google Scholar] [CrossRef] [PubMed]
- Gross, A.; Langevin, P.; Burnie, S.J.; Bédard-Brochu, M.S.; Empey, B.; Dugas, E.; Faber-Dobrescu, M.; Andres, C.; Graham, N.; Goldsmith, C.H.; et al. Manipulation and mobilisation for neck pain contrasted against an inactive control or another active treatment. In Cochrane Database of Systematic Reviews; Cochrane Back and Neck Group, Ed.; John Wiley & Sons: Hoboken, NJ, USA, 2015. [Google Scholar] [CrossRef]
- Holobar, A.; Minetto, M.A.; Farina, D. Accurate identification of motor unit discharge patterns from high-density surface EMG and validation with a novel signal-based performance metric. J. Neural Eng. 2014, 11, 016008. [Google Scholar] [CrossRef]
- Beck, R. Muscle fiber conduction velocity. In Wiley Encyclopedia of Biomedical Engineering; John Wiley & Sons: Hoboken, NJ, USA, 2006. [Google Scholar]
- Farina, D.; Fortunato, E.; Merletti, R. Noninvasive estimation of motor unit conduction velocity distribution using linear electrode arrays. IEEE Trans. Biomed. Eng. 2000, 47, 380–388. [Google Scholar] [CrossRef]
- Farina, D.; Muhammad, W.; Fortunato, E.; Meste, O.; Merletti, R.; Rix, H. Estimation of single motor unit conduction velocity from surface electromyogram signals detected with linear electrode arrays. Med. Biol. Eng. Comput. 2001, 39, 225–236. [Google Scholar] [CrossRef]
- Bates, D.; Mächler, M.; Bolker, B.; Walker, S. Fitting Linear Mixed-Effects Models using lme4. arXiv 2014, arXiv:14065823. Available online: http://arxiv.org/abs/1406.5823 (accessed on 5 March 2020).
- Jochumsen, M.; Cremoux, S.; Robinault, L.; Lauber, J.; Arceo, J.C.; Navid, M.S.; Nedergaard, R.W.; Rashid, U.; Haavik, H.; Niazi, I.K. Investigation of Optimal Afferent Feedback Modality for Inducing Neural Plasticity with A Self-Paced Brain-Computer Interface. Sensors 2018, 18, 3761. [Google Scholar] [CrossRef] [PubMed]
- Lenth, R.; Singmann, H.; Love, J. Emmeans: Estimated marginal means, aka least-squares means. R Package Version 2018, 1, 3. [Google Scholar]
- Niazi, I.K.; Kamavuako, E.N.; Holt, K.; Janjua, T.A.M.; Kumari, N.; Amjad, I.; Haavik, H. The Effect of Spinal Manipulation on the Electrophysiological and Metabolic Properties of the Tibialis Anterior Muscle. Healthcare 2020, 8, 548. [Google Scholar] [CrossRef] [PubMed]
- Farina, D.; Pozzo, M.; Merlo, E.; Bottin, A.; Merletti, R. Assessment of average muscle fiber conduction velocity from surface EMG signals during fatiguing dynamic contractions. IEEE Trans. Biomed. Eng. 2004, 51, 1383–1393. [Google Scholar] [CrossRef] [PubMed]
- Kernell, D. The Limits of Firing Frequency in Cat Lumbosacral Motoneurones Possessing Different Time Course of Afterhyperpolarization. Acta Physiol. Scand. 1965, 65, 87–100. [Google Scholar] [CrossRef]
- Kernell, D. Principles of Force Gradation in Skeletal Muscles. Neural Plast. 2003, 10, 69–76. [Google Scholar] [CrossRef]
- De Luca, C.J.; Contessa, P. Biomechanical benefits of the onion-skin motor unit control scheme. J. Biomech. 2015, 48, 195–203. [Google Scholar] [CrossRef]
- Gawel, M.; Kostera-Pruszczyk, A. Effect of Age and Gender on the Number of Motor Units in Healthy Subjects Estimated by the Multipoint Incremental MUNE Method. J. Clin. Neurophysiol. 2014, 31, 272–278. [Google Scholar] [CrossRef]
| Control Group (N = 14) | Intervention Group (N = 12) | |
|---|---|---|
| Conduction velocity (m.s−1) | 5.11 ± 0.62 | 5.12 ± 0.61 |
| Peak firing rate (pps) | 15.79 ± 4.04 | 15.45 ± 3.9 |
| Instantaneous MU firing rate gradient (pps.s−1) | 1.33 ± 1.48 | 1.37 ± 1.51 |
| Condition, Intensity | Control—Intervention Difference (Mean ± SE) | df | T Ratio | p Value |
|---|---|---|---|---|
| Ramp and maintain, 5% | 0.3 ± 0.11 m.s−1 | 53.7 | 2.64 | 0.01 |
| Ramp and maintain, 10% | 0.2 ± 0.12 m.s−1 | 53.8 | 1.65 | 0.11 |
| Ramp, 5% | 0.07 ± 0.11 m.s−1 | 53.8 | 0.66 | 0.51 |
| Ramp, 10% | 0.03 ± 0.12 m.s−1 | 54.4 | 0.28 | 0.78 |
| Condition, Intensity | Ratio Control/Intervention Difference (Mean ± SE) | z-Value | p-Value |
|---|---|---|---|
| Ramp and maintain, 5% | 0.99 ± 0.15 | −0.05 | 0.96 |
| Ramp and maintain, 10% | 0.95 ± 0.15 | −0.32 | 0.75 |
| Ramp, 5% | 1.27 ± 0.2 | 1.52 | 0.13 |
| Ramp, 10% | 1.174 ± 0.19 | 0.98 | 0.33 |
| Condition, Intensity | Control—Intervention Difference (Mean ± SE) | z-Value | p-Value |
| Ramp and maintain, 5% | 0.286 ± 0.4 pps | 0.715 | 0.47 |
| Ramp and maintain, 10% | −0.24 ± 0.47 pps | −0.508 | 0.61 |
| Ramp, 5% | 0.14 ± 0.42 pps | 0.33 | 0.74 |
| Ramp, 10% | 0.1 ± 0.52 pps | 0.2 | 0.84 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Robinault, L.; Holobar, A.; Crémoux, S.; Rashid, U.; Niazi, I.K.; Holt, K.; Lauber, J.; Haavik, H. The Effects of Spinal Manipulation on Motor Unit Behavior. Brain Sci. 2021, 11, 105. https://doi.org/10.3390/brainsci11010105
Robinault L, Holobar A, Crémoux S, Rashid U, Niazi IK, Holt K, Lauber J, Haavik H. The Effects of Spinal Manipulation on Motor Unit Behavior. Brain Sciences. 2021; 11(1):105. https://doi.org/10.3390/brainsci11010105
Chicago/Turabian StyleRobinault, Lucien, Aleš Holobar, Sylvain Crémoux, Usman Rashid, Imran Khan Niazi, Kelly Holt, Jimmy Lauber, and Heidi Haavik. 2021. "The Effects of Spinal Manipulation on Motor Unit Behavior" Brain Sciences 11, no. 1: 105. https://doi.org/10.3390/brainsci11010105
APA StyleRobinault, L., Holobar, A., Crémoux, S., Rashid, U., Niazi, I. K., Holt, K., Lauber, J., & Haavik, H. (2021). The Effects of Spinal Manipulation on Motor Unit Behavior. Brain Sciences, 11(1), 105. https://doi.org/10.3390/brainsci11010105








