Apparent False Lateralization of Seizure Onset by Scalp EEG in Temporal Lobe Epilepsy Associated with Cerebral Cavernous Malformation: A Case Report and Overview
Abstract
1. Introduction
| Age (Years)-Sex | Clinical Features and Ictal Semiology | MRI (Structural Findings) | Functional Neuroimaging | Scalp EEG | Intracraneal Ictal EEG | False Lateralization Hypothesis | Ref. |
|---|---|---|---|---|---|---|---|
| 56, female | Focal onset, awareness impairment, non-motor features. | Normal | No area of hypo metabolism | Interictal: sharp waves from both temporal head regions. Ictal: rhythmic 5-6 ictal activity from the left temporal head region. | Ictal seizure onset in the right temporal lobe. 1.5 s after seizure onset on the right: gamma activity in the left mesial temporal and the left lateral temporal electrodes. | Temporal neocortex epilepsy: rapidly involve the contralateral mesolimbic structures; spread through the hippocampal commissures. Initial low voltage fast activity in the right temporal was undetectable on scalp EEG. | [1] |
| 25, female | Focal onset, awareness impairment, non-motor features and motor features (turn her head to the left, chewing, and hyperkinetic movements) | Atrophic process in the left hemisphere | Not reported | Interictal epileptiform in both inferomesial temporal regions, left side predominance. Ictal: right temporal region onset, two seconds later it spread to the contralateral hemisphere, amplitude predominance was right | Seizure onset started in the left temporal. 36 s after the seizure onset, the ictal discharge spread to the right temporal lobe | Cerebral damage reduces the voltage of the ictal discharge on the side of the lesion and higher amplitude in the normal hemisphere. | [2] |
| 21, male | Unknown onset seizure with motor activity. Focal onset, and impairment awareness, motor features (automatism), and non-motor features (epigastralgic sensation) | Left hemisphere atrophy | Not reported | Interictal. sphenoidal EEG: bilateral temporal, independent sharp waves with left sided predominance Ictal: discharge arise from the right frontotemporal regions, predominance over the right temporal region, 12 s after the seizure onset, ictal discharge spread to the contralateral hemisphere. | Onset of the ictal discharge in the left temporal lobe | Cerebral damage reduces the voltage of the ictal discharge on the side of the lesion and higher amplitude in the normal hemisphere. | [2] |
| 30, male | Focal onset, and impairment awareness , non motor features (fear) and motor features (automatism) | Enlarged left lateral ventricle, three small areas of hypo density in the left posterior limb of the internal capsule | Bitemporal independent, interictal sharp waves with right-sided predominance. Ictal EEG: seizure onset in the right temporal lobe. | Seizure discharge started in the left temporal lobe. 2–3 s after seizure onset ictal discharge spread to the right temporal lobe also spread to both frontal regions. | Cerebral damage reduces the voltage of the ictal discharge on the side of the lesion and higher amplitude in the normal hemisphere. | [2] | |
| 39, female | 1. Focal onset, and impairment awareness , non motor features (epigastric sensation) 2. Focal onset, and impairment awareness and bilateral progression | Atrophy of the left cerebral hemisphere. | Interictal Ictal ECD- SPECT: left hemispheric hypo perfusion and IMZ SPECT revealed slight hypo accumulation in the left hippocampus | Ictal: Right temporal side focus | Interictal subdural EEG: independently in the left hippocampus region and also the right hippocampus region with spread to the lateral temporal regions. | Reduced epileptiform activity in the atrophic hemisphere. Scalp EEG and MEG propagated electricals signals to the right side are easier to detect than signs to the left side. | [3] |
| 23, male | Focal onset, epigastric sensation, impaired awareness, and motor features (automatisms) | Severe left hippocampal sclerosis | Left temporal hypometabolism | Interictal scalp EEG: mild left temporal slow, left midpost, temporal spikes. Ictal scalp: 4 right temporal, 1 left temporal | Left mesial temporal | “Burned-out hippocampus” syndrome with atypical spread of ictal discharges. Not enough neocortical neurons to produce a visible scalp discharge is not accomplished until propagation of the seizure to the contralateral temporal lobe. | [4] |
| 48, male | Focal onset, and motor features (automatisms) | Severe left hippocampal sclerosis | Not reported | Interictal scalp EEG: Bitemporal spikes Ictal scalp: 5 right temporal, 2 bitemporal | Left hippocampus/amygdala and lateral temporal spikes. | “Burned-out hippocampus” with atypical spread of ictal discharges. Rapid bilateral spread. | [4] |
| 38, female | Bilateral tonic-clonic seizures, impaired awareness and non-motor components (panic attacks). Recalled memory, epigastric sensation. | Mild right hippocampal atrophy | PET: Mild right temporal hypo metabolism | Interictal: mild generalized slowing, right anterior temporal spikes, rare left anterior temporal sharps Ictal scalp: theta frequency rhythmic discharge in the left temporal region. | Frequent spikes in the right mesial entorhinal cortex. Right anterior hippocampus and amygdala. | Propagation through the dorsal hippocampal commissure was faster than that to the overlying ipsilateral neocortex, thereby producing falsely discordant ictal scalp EEG recordings. Location and extent of the epileptogenic region: a more posterior hippocampal propagation pattern. | [4] |
| 37, female | Focal onset, impaired awareness and bilateral tonic-clonic seizures. Lightheadedness, staring, fumbling. | Severe right hippocampal atrophy | PET: hypo metabolism: mild right temporal | Interictal infrequent left temporal sharp waves Ictal scalp: left temporal onset | Initial seizures (slowing and spikes and sharp waves) bitemporal, later ones right temporal | “Burned-out hippocampus” with atypical spread of ictal discharges. Interhemispheric propagation. Propagation through the dorsal hippocampal commissure was faster than that to the overlying ipsilateral neocortex, thereby producing falsely discordant ictal scalp EEG recordings | [4] |
| 27, male | Focal onset, impaired awareness and motor features (bimanual automatisms), epigastric sensation. | Severe left hippocampal atrophy | Not performed | Interictal: left mid temporal spikes Ictal: 3 right midtemporal onset, 1 bitemporal onset | Slowing and frequent spikes in the left hippocampus, some spikes in the right hippocampus. | “Burned-out hippocampus” with atypical spread of ictal discharges. | [4] |
| 12, male | Auditory aura, bimanual and oral automatisms, impaired awareness and non-motor features (déjà vu) | Cavernous malformation in the right superior temporal gyrus | F-FDG-PET focal hypo metabolism on the right, left temporal hypo metabolism. SPECT ictal and interictal: focal hypo perfusion at the location of the cavernoma. Interictal: left temporal hypo perfusion Ictal: hyper perfusion on the left temporal lobe. Abnormal perfusion of the left temporal lobe and along the caudal margin of the right temporal cavernoma. MEG: spikes sources around the cavernoma, but also contralateral in the left miid lateral temporal region. | Left frontal and posterior head quadrant spikes and a single electro clinical seizure originating in the left frontal head region. | Not performed in the presurgical state | Distinct site of epileptogenesis contralateral to the cavernoma (two epileptogenic foci) or rapid contralateral spread of epileptogenic activity. | [10] |
| 34, female | Focal onset, memory deficits, impaired awareness, non motor (“weird feelings”) and motor features (automatisms). | Cavernous angioma in the left medial temporal lobe | Not performed | Interictal: epileptiform patterns (right more than left) Ictal: two seizures with a right temporal origin and two of less certain onset | Right mesial temporal onset (subdural strip electrodes) | Focal neurochemical abnormalities that coincide with ictal onsets. | [11] |
| 33, not reported. | Not seizure description. Left, fronto-temporal dysfunction in the NPT and Wada test. | Left hippocampal sclerosis | Not performed | Interictal: epileptiform discharges in the left temporal region. Ictal: 2 ictal onsets in the left, 4 seizures onset in the right temporal region. | Ictal onset in the left hippocampus. | Seizure spreading to contralateral neocortex through functional commissural connections (i.e., dorsal hippocampal commissure) Rapid contralateral spread by activation of frontal limbic pathways. | [12] |
| 27, not reported | Not seizure description. Left, temporal dysfunction in the NPT and Wada test. | Left hippocampal sclerosis | Not performed | Interictal: epileptiform discharges in the left side. Ictal: 2 ictal seizure onset in the right temporal region | Ictal onset in the left hippocampus. | Seizure spreading to contralateral neocortex through functional commissural connections (i.e., dorsal hippocampal commissure) Rapid contralateral spread by activation of frontal limbic pathways. | [12] |
| 25, not reported | Not clinical information | Left hippocampal sclerosis | Not performed | Interictal: epileptiform discharges in the left side. Ictal: 3 ictal seizure onset in the right side | Ictal onset in the left anterior inferior–mesial temporal region. | Seizure spreading to contralateral neocortex through functional commissural connections (i.e., dorsal hippocampal commissure) Rapid contralateral spread by activation of frontal limbic pathways. | [12] |
| 33, not reported | Not seizure description. Right, temporal dysfunction in the NPT and Wada test. | Right hippocampal sclerosis | Not information | Interictal: epileptiform discharges in the right side. Ictal: 4 ictal seizure onset in the left temporal region | Ictal onset: right hippocampus | Seizure spreading to contralateral neocortex through functional commissural connections (i.e., dorsal hippocampal commissure) Rapid contralateral spread by activation of frontal limbic pathways. | [12] |
| 45, male | Focal onset, impaired awareness, non-motor features (déjà vu and periodic nausea). Impairment of both verbal and visual memory in the NPT. | Several bilateral scattered white matter hyper intensities, most prominent in the left frontal lobe. | Decreased glucose uptake in the left temporal lobe as compared with the right temporal lobe in both lateral and mesial cortical regions. | Onset in the right amygdala, although one subclinical seizure began in the left temporal lobe. | EEG from the right amygdala showed continuous low-amplitude fast activity (>13 Hz) for extended times; at other times, this pattern was entirely absent. | The fast activity was believed to be epileptic, although it was asymptomatic. Several discrete subclinical seizures with a different electrographic pattern originated in the right amygdala, later spreading to the right hippocampus. Abnormal increase in temporal lobe metabolism. | [13] |
| 27, female | Focal onset, aware, non motor features (unpleasant smell and taste, salivation, noted strange feelings in her face or body) | Dilated right temporal ventricular horn with questionable decreased hippocampal volume | Less FDG uptake in the left temporal lobe than in the right temporal lobe, mainly in lateral temporal cortex. | Right sphenoidal spikes and intermittent arrhythmic right sphenoidal theta in wakefulness and sleep. | All arose from the right amygdala. The EEG also showed frequent (several per second) sharp waves and occasional subclinical electrographic seizures in the right amygdala. | Sharp waves amplify neuronal activity in temporal lobe neocortex through amygdalocortical connections, possibly increasing lateral temporal cortical metabolism. | [13] |
| 47, male | Focal onset, awareness impairment, non-motor features (déjà vu) | Left hippocampal atrophy | FDG PET: decreased uptake in the left temporal region | Ictal EEG: Left temporal (1 min after the clinical onset of the seizure) | SDE: seizure onset in the right lateral temporal region before spreading to the let lateral temporal SDE in 4 to 10 s. SEEG: Regional onset seizure arising from the left SEEG electrodes suggesting a left temporal onset | Rapid contralateral spread through the hippocampal commissure or frontal limbic pathways | [14] |
| 45, female | 1. Unknown onset seizure with motor activity 2. Focal onset, awareness impairment, non motor features 3. Focal onset, awareness impairment, and progression to bilateral tonic–clonic seizures | Normal | FDG PET: normal. | Interictal: Left temporal epileptiform discharges. Ictal recordings: no localizing and no lateralizing | SEEG electrodes: seizures arising from the left temporal electrode, then spreading to the contralateral electrode first, followed 5 to 20 s later by spread to the contralateral temporal SDE; the ipsilateral SDE recordings showed no change | Rapid contralateral spread through the hippocampal commissure or frontal limbic pathways | [14] |
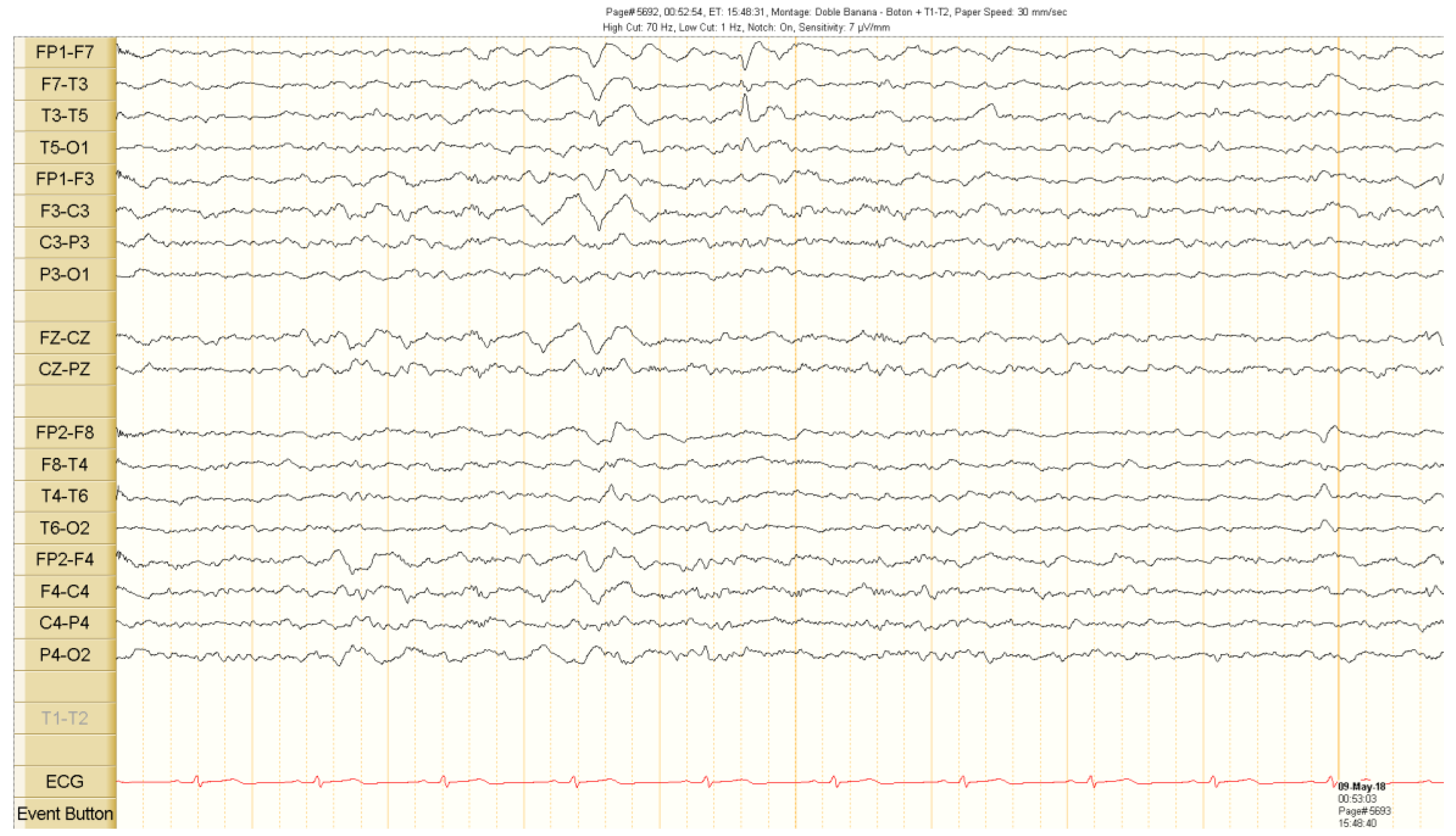
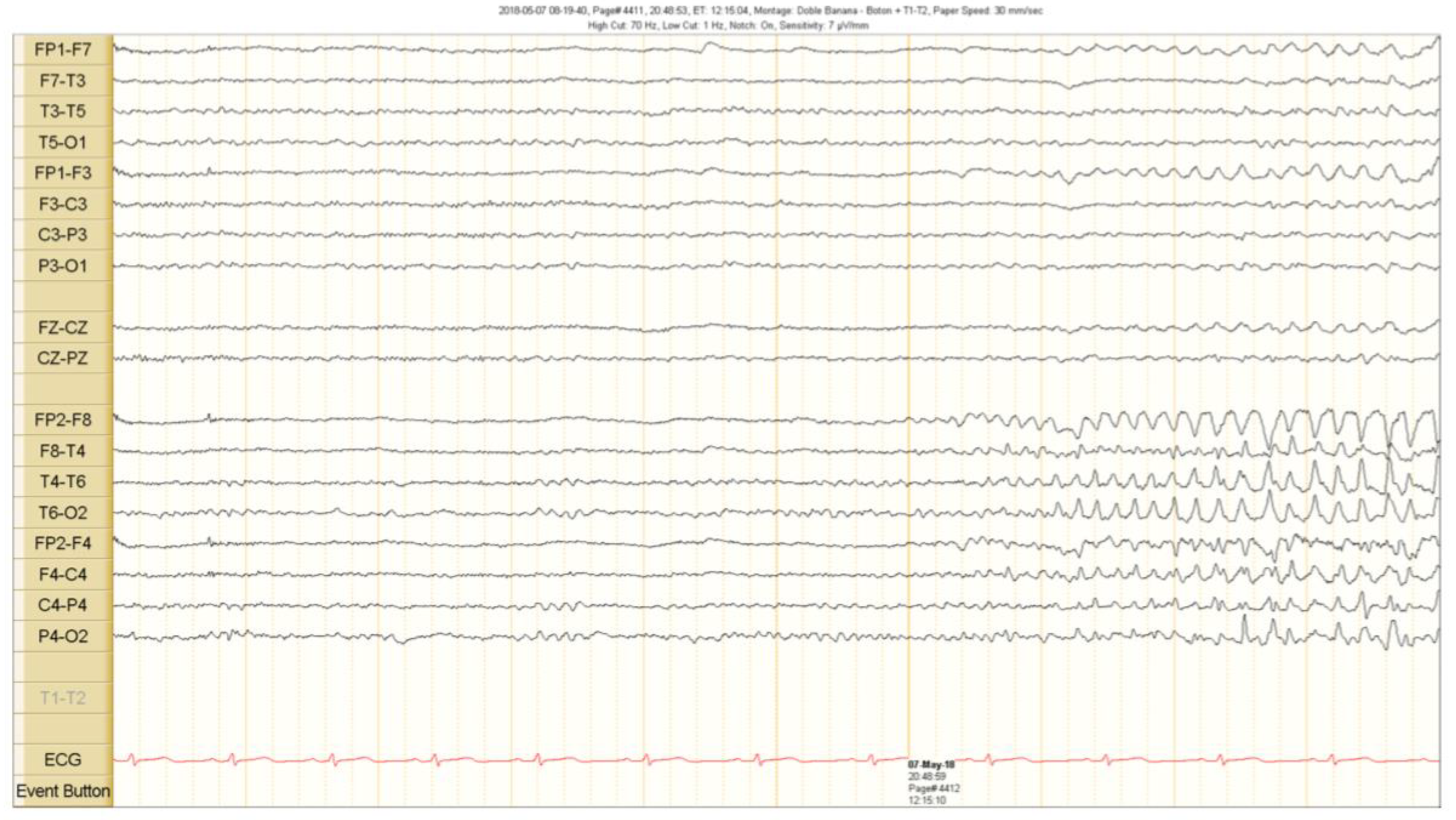
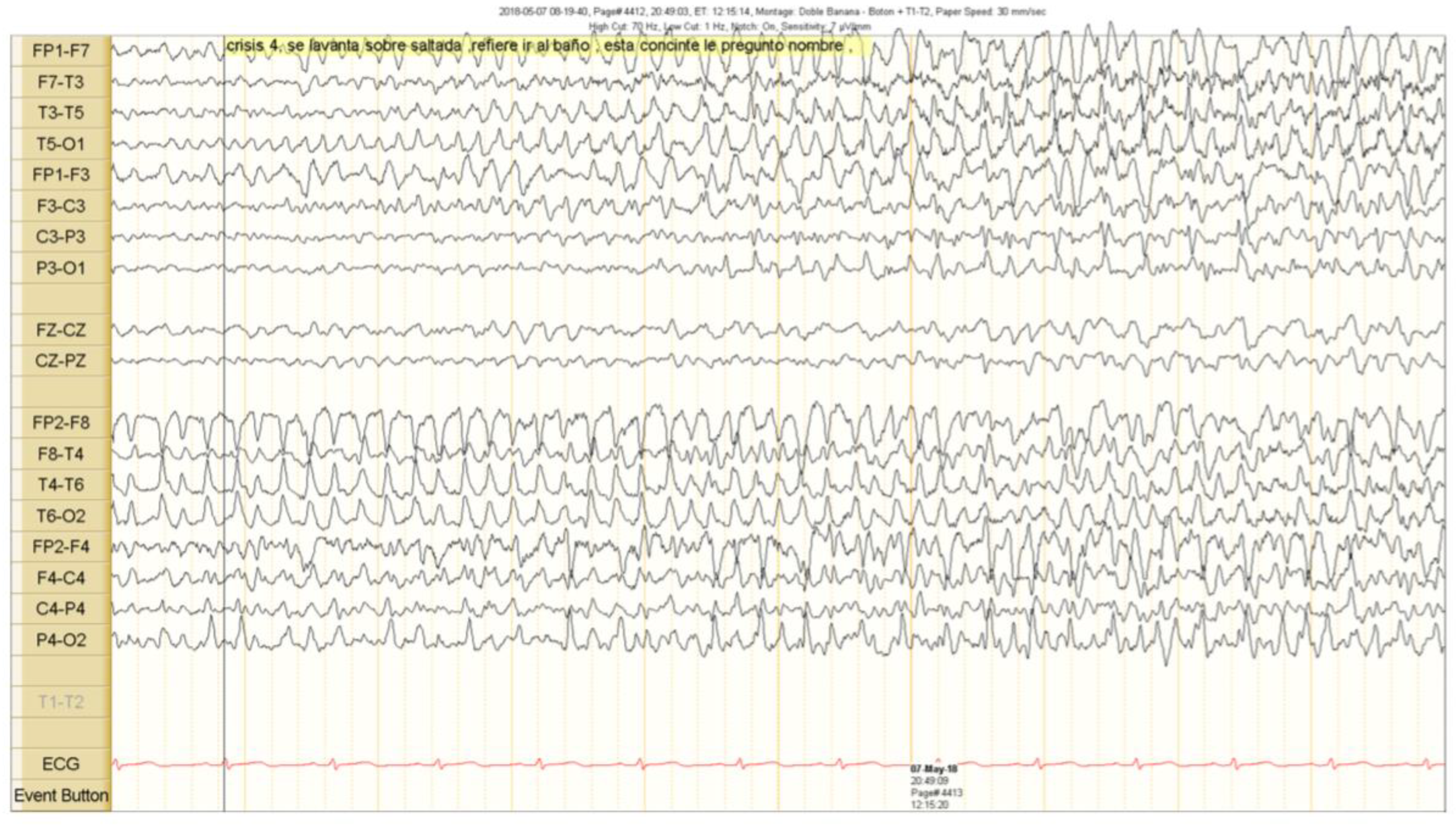
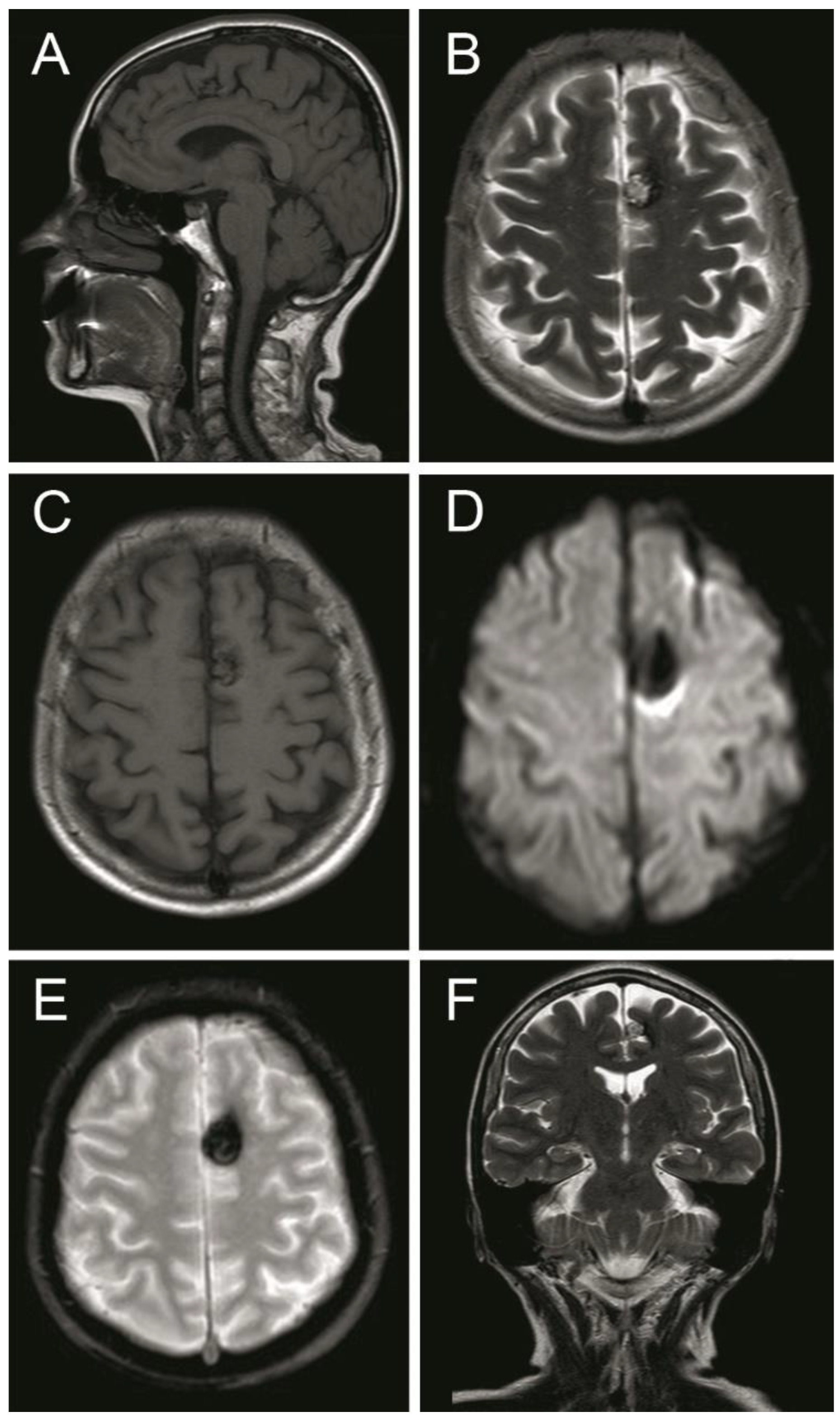
2. Case Presentation
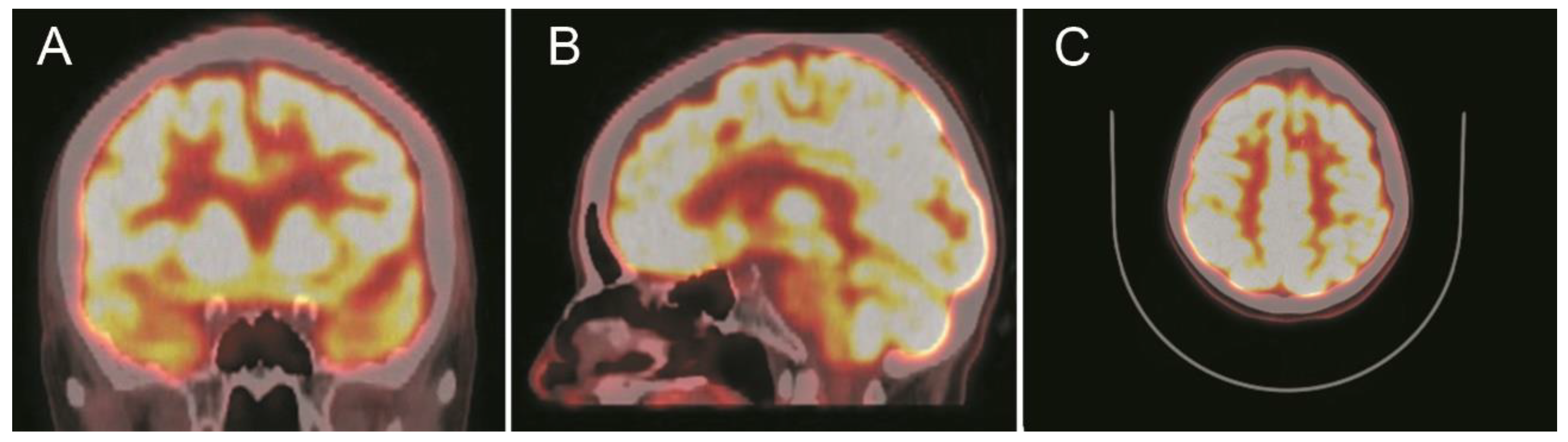
Outcome and Follow Up
3. Discussion
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Adamolekun, B.; Afra, P.; Boop, F.A. False lateralization of seizure onset by scalp EEG in neocortical temporal lobe epilepsy. Seizure 2011, 20, 494–499. [Google Scholar] [CrossRef] [PubMed]
- Sammaritano, M.; de Lotbinière, A.; Andermann, F.; Olivier, A.; Gloor, P.; Quesney, L.F. False lateralization by surface EEG of seizure onset in patients with temporal lobe epilepsy and gross focal cerebral lesions: False Lateralization by Surface EEG. Ann. Neurol. 1987, 21, 361–369. [Google Scholar] [CrossRef] [PubMed]
- Fujimoto, A.; Masuda, H.; Homma, J.; Sasagawa, M.; Kameyama, S. False Lateralization of Mesial Temporal Lobe Epilepsy by Noninvasive Neurophysiological Examinations-Case Report. Neurol. Med. Chir. 2006, 46, 518–521. [Google Scholar] [CrossRef] [PubMed]
- Mintzer, S.; Cendes, F.; Soss, J.; Andermann, F.; Engel, J.; Dubeau, F.; Olivier, A.; Fried, I. Unilateral Hippocampal Sclerosis with Contralateral Temporal Scalp Ictal Onset. Epilepsia 2004, 45, 792–802. [Google Scholar] [CrossRef] [PubMed]
- Haasdijk, R.A.; Cheng, C.; Maat-Kievit, A.J.; Duckers, H.J. Cerebral cavernous malformations: From molecular pathogenesis to genetic counselling and clinical management. Eur. J. Hum. Genet. 2012, 20, 134–140. [Google Scholar] [CrossRef]
- Al-Shahi Salman, R. The outlook for adults with epileptic seizure(s) associated with cerebral cavernous malformations or arteriovenous malformations: Epilepsy Caused by Cavernomas and Arteriovenous Malformations. Epilepsia 2012, 53, 34–42. [Google Scholar] [CrossRef]
- Josephson, C.; Rosenow, F.; Al-Shahi Salman, R. Intracranial Vascular Malformations and Epilepsy. Semin. Neurol. 2015, 35, 223–234. [Google Scholar] [CrossRef]
- Hauser, W.A.; Mohr, J.P. Seizures, epilepsy, and vascular malformations. Neurology 2011, 76, 1540–1541. [Google Scholar] [CrossRef]
- Rosenow, F.; Alonso-Vanegas, M.A.; Baumgartner, C.; Blümcke, I.; Carreño, M.; Gizewski, E.R.; Hamer, H.M.; Knake, S.; Kahane, P.; Lüders, H.O.; et al. Cavernoma-related epilepsy: Review and recommendations for management-Report of the Surgical Task Force of the ILAE Commission on Therapeutic Strategies. Epilepsia 2013, 54, 2025–2035. [Google Scholar] [CrossRef]
- Rammo, R.; Greiner, H.; Trout, A.T.; Leach, J.; Rozhkov, L.; Fujiwara, H.; Rose, D.F.; Mangano, F. False lateralization of pre-surgical work-up in a child with a cortical cavernous malformation and intractable epilepsy. J. Neurosurg. Sci. 2018, 62, 91–94. [Google Scholar] [CrossRef]
- Holmes, M.D.; Wilensky, A.J.; Ojemann, G.A.; Ojemann, L.M. Hippocampal or neocortical lesions on magnetic resonance imaging do not necessarily indicate site of ictal onsets in partial epilepsy. Ann. Neurol. 1999, 45, 461–465. [Google Scholar] [CrossRef]
- Tezer, F.I.; Dericioglu, N.; Bozkurt, G.; Bilginer, B.; Akalan, N.; Saygi, S. Epilepsy surgery in patients with unilateral mesial temporal sclerosis and contralateral scalp ictal onset. Turk. Neurosurg. 2011. [Google Scholar] [CrossRef] [PubMed]
- Sperling, M.R.; Alavi, A.; Reivich, M.; French, J.A.; O’Connor, M.J. False lateralization of temporal lobe epilepsy with FDG positron emission tomography. Epilepsia 1995, 36, 722–727. [Google Scholar] [CrossRef] [PubMed]
- Alsaadi, T.M.; Laxer, K.; Barbaro, N.; Marks, W.; Garcia, P. False lateralization by subdural electrodes in two patients with temporal lobe epilepsy. Neurology 2001, 57, 532–534. [Google Scholar] [CrossRef] [PubMed]
- Niranjan, A.; Lunsford, L.D. Stereotactic radiosurgery guidelines for the management of patients with intracranial cavernous malformations. Prog. Neurol. Surg. 2013, 27, 166–175. [Google Scholar] [CrossRef] [PubMed]
- Foldvary-Schaefer, N.; Unnwongse, K. Localizing and lateralizing features of auras and seizures. Epilepsy Behav. 2011, 20, 160–166. [Google Scholar] [CrossRef]
- Loddenkemper, T.; Kotagal, P. Lateralizing signs during seizures in focal epilepsy. Epilepsy Behav. 2005, 7, 1–17. [Google Scholar] [CrossRef]
- Spencer, S.S.; Williamson, P.D.; Spencer, D.D.; Mattson, R.H. Human Hippocampal Seizure Spread Studied by Depth and Subdural Recording: The Hippocampal Commissure. Epilepsia 1987, 28, 479–489. [Google Scholar] [CrossRef]
- Lieb, J.; Dasheiff, R.M.; Engel, J. Role of the frontal lobes in the propagation of mesial temporal lobe seizures. Epilepsia 1991, 32, 822–837. [Google Scholar] [CrossRef]
- Young, M.; Sazgar, M.; Sen-Gupta, I.; Losey, T.; Bannout, F.; Hwang, J.; Pietzsch, E.; Vadera, S.; Hsu, F.; Lin, J.; et al. The Rate of False Lateralization from Scalp EEG in Epilepsy Patients with Bilaterally Implanted Intracranial EEG (P4.195). Neurology 2016, 86 (Suppl. 16). [Google Scholar]
- Giulioni, M.; Zucchelli, M.; Riguzzi, P.; Marucci, G.; Tassinari, C.A.; Calbucci, F. Co-existence of cavernoma and cortical dysplasia in temporal lobe epilepsy. J. Clin. Neurosci. 2007, 14, 1122–1124. [Google Scholar] [CrossRef]
- Chen, D.J.; Severson, E.; Prayson, R.A. Cavernous angiomas in chronic epilepsy associated with focal cortical dysplasia. Clin. Neuropathol. 2013, 32, 31. [Google Scholar] [CrossRef] [PubMed]
- Niehusmann, P.; Becker, A.J.; Malter, M.P.; Raabe, A.; Boström, A.; von der Brelie, C. Focal cortical dysplasia type IIIc associates with multiple cerebral cavernomas. Epilepsy Res. 2013, 107, 190–194. [Google Scholar] [CrossRef] [PubMed]
- Ferrier, C.H.; Aronica, E.; Leijten, F.S.S.; Spliet, W.G.M.; Boer, K.; Van Rijen, P.C.; Van Huffelen, A.C. Electrocorticography discharge patterns in patients with a cavernous hemangioma and pharmacoresistent epilepsy. J. Neurosurg. 2007, 107, 495–503. [Google Scholar] [CrossRef] [PubMed]
- Maciunas, J.A.; Syed, T.U.; Cohen, M.L.; Werz, M.A.; Maciunas, R.J.; Koubeissi, M.Z. Triple pathology in epilepsy: Coexistence of cavernous angiomas and cortical dysplasias with other lesions. Epilepsy Res. 2010, 91, 106–110. [Google Scholar] [CrossRef] [PubMed]
- Friedman, E. Epilepsy Imaging in Adults: Getting It Right. Am. J. Roentgenol. 2014, 203, 1093–1103. [Google Scholar] [CrossRef]
- Sarikaya, I. PET studies in epilepsy. Am. J. Nucl. Med. Mol. Imaging 2015, 5, 416–430. [Google Scholar]
- Chan, A.Y.; Kharrat, S.; Lundeen, K.; Mnatsakanyan, L.; Sazgar, M.; Sen-Gupta, I.; Lin, J.J.; Hsu, F.P.K.; Vadera, S. Length of stay for patients undergoing invasive electrode monitoring with stereoelectroencephalography and subdural grids correlates positively with increased institutional profitability. Epilepsia 2017, 58, 1023–1026. [Google Scholar] [CrossRef]
- Cendes, F.; Theodore, W.H.; Brinkmann, B.H.; Sulc, V.; Cascino, G.D. Neuroimaging of epilepsy. In Handbook of Clinical Neurology; Elsevier: Amsterdam, The Netherlands, 2016; Volume 136, pp. 985–1014. ISBN 978-0-444-53486-6. [Google Scholar]
- Batra, S.; Lin, D.; Recinos, P.F.; Zhang, J.; Rigamonti, D. Cavernous malformations: Natural history, diagnosis and treatment. Nat. Rev. Neurol. 2009, 5, 659–670. [Google Scholar] [CrossRef]
- Josephson, C.B.; Leach, J.-P.; Duncan, R.; Roberts, R.C.; Counsell, C.E.; Al-Shahi Salman, R. Scottish Audit of Intracranial Vascular Malformations (SAIVMs) steering committee and collaborators Seizure risk from cavernous or arteriovenous malformations: Prospective population-based study. Neurology 2011, 76, 1548–1554. [Google Scholar] [CrossRef]
- Wang, C.; You, C.; Han, G.-Q.; Wang, J.; Xiong, Y.-B.; Liu, C.-X. Electrocorticography-guided surgical treatment of solitary supratentorial cavernous malformations with secondary epilepsy. Chin. Med. Sci. J. 2014, 29, 112–116. [Google Scholar] [CrossRef]
- Jin, K.; Nakasato, N.; Shamoto, H.; Kanno, A.; Itoyama, Y.; Tominaga, T. Neuromagnetic localization of spike sources in perilesional, contralateral mirror, and ipsilateral remote areas in patients with cavernoma. Epilepsia 2007, 48, 2160–2166. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; Lin, F.; Kang, D.; Jiao, Y.; Cao, Y.; Wang, S. Supratentorial cavernous malformations adjacent to the corticospinal tract: Surgical outcomes and predictive value of diffusion tensor imaging findings. J. Neurosurg. 2018, 128, 541–552. [Google Scholar] [CrossRef] [PubMed]
- Duffau, H.; Fontaine, D. Successful resection of a left insular cavernous angioma using neuronavigation and intraoperative language mapping. Acta Neurochir. (Wien) 2005, 147, 205–208. [Google Scholar] [CrossRef]
- Cavalcanti, D.D.; Preul, M.C.; Kalani, M.Y.S.; Spetzler, R.F. Microsurgical anatomy of safe entry zones to the brainstem. J. Neurosurg. 2016, 124, 1359–1376. [Google Scholar] [CrossRef]
- Chohan, M.O.; Marchiò, S.; Morrison, L.A.; Sidman, R.L.; Cavenee, W.K.; Dejana, E.; Yonas, H.; Pasqualini, R.; Arap, W. Emerging Pharmacologic Targets in Cerebral Cavernous Malformation and Potential Strategies to Alter the Natural History of a Difficult Disease: A Review. JAMA Neurol. 2019, 76, 492. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gaviria Carrillo, M.; López, J.; Rodríguez Q., J.H.; Gaona, I.; Ortiz-Guerrero, G.; Nava-Mesa, M.O. Apparent False Lateralization of Seizure Onset by Scalp EEG in Temporal Lobe Epilepsy Associated with Cerebral Cavernous Malformation: A Case Report and Overview. Brain Sci. 2020, 10, 584. https://doi.org/10.3390/brainsci10090584
Gaviria Carrillo M, López J, Rodríguez Q. JH, Gaona I, Ortiz-Guerrero G, Nava-Mesa MO. Apparent False Lateralization of Seizure Onset by Scalp EEG in Temporal Lobe Epilepsy Associated with Cerebral Cavernous Malformation: A Case Report and Overview. Brain Sciences. 2020; 10(9):584. https://doi.org/10.3390/brainsci10090584
Chicago/Turabian StyleGaviria Carrillo, Mariana, Jonathan López, Jesús H. Rodríguez Q., Ivan Gaona, Gloria Ortiz-Guerrero, and Mauricio O. Nava-Mesa. 2020. "Apparent False Lateralization of Seizure Onset by Scalp EEG in Temporal Lobe Epilepsy Associated with Cerebral Cavernous Malformation: A Case Report and Overview" Brain Sciences 10, no. 9: 584. https://doi.org/10.3390/brainsci10090584
APA StyleGaviria Carrillo, M., López, J., Rodríguez Q., J. H., Gaona, I., Ortiz-Guerrero, G., & Nava-Mesa, M. O. (2020). Apparent False Lateralization of Seizure Onset by Scalp EEG in Temporal Lobe Epilepsy Associated with Cerebral Cavernous Malformation: A Case Report and Overview. Brain Sciences, 10(9), 584. https://doi.org/10.3390/brainsci10090584





