Obesity is Associated with Reduced Plasticity of the Human Motor Cortex
Abstract
1. Introduction
2. Methods
2.1. Study Design and Participants
2.2. Experimental Procedures
2.3. Participant Characteristics
2.4. Electromyography
2.5. Transcranial Magnetic Stimulation
2.6. Theta Burst Stimulation
2.7. Data Analysis and Statistics
3. Results
3.1. Participant Characteristics
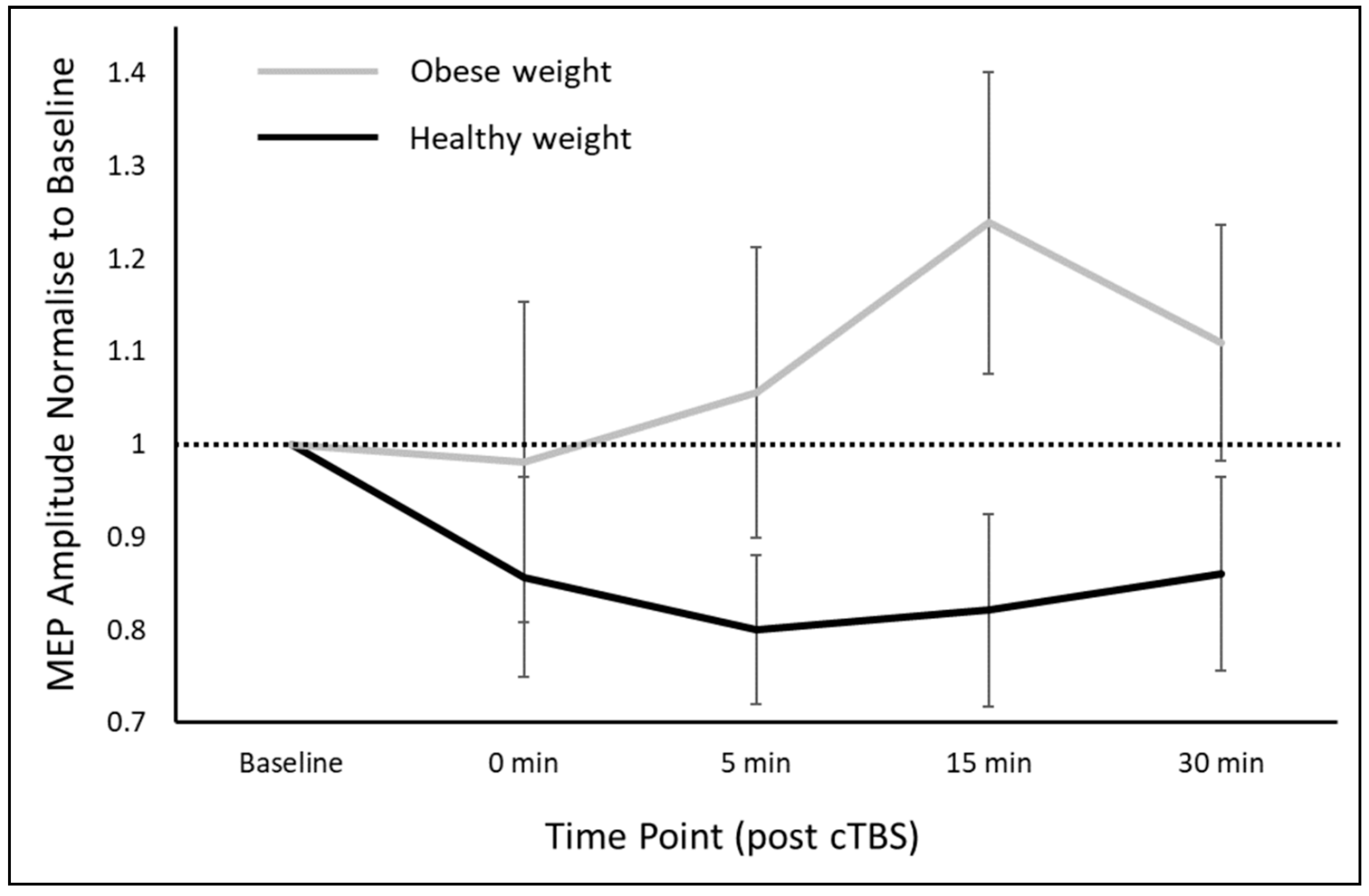
3.2. cTBS Responses
4. Discussion
Limitations
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Blüher, M. Obesity: Global epidemiology and pathogenesis. Nat. Rev. Endocrinol. 2019, 15, 288–298. [Google Scholar] [CrossRef] [PubMed]
- Manson, J.E.; Skerrett, P.J.; Greenland, P.; Van Itallie, T.B. The escalating pandemics of obesity and sedentary lifestyle. A call to action for clinicians. Arch. Intern. Med. 2004, 164, 249–258. [Google Scholar] [CrossRef] [PubMed]
- Van Gaal, L.F.; Mertens, I.L.; De Block, C.E. Mechanisms linking obesity with cardiovascular disease. Nature 2006, 444, 875–880. [Google Scholar] [CrossRef]
- Livingston, G.; Huntley, J.; Sommerlad, A.; Ames, D.; Ballard, C.; Banerjee, S.; Brayne, C.; Burns, A.; Cohen-Mansfield, J.; Cooper, C.; et al. Dementia prevention, intervention, and care: 2020 report of the Lancet Commission. Lancet 2020, 396, 413–446. [Google Scholar] [CrossRef]
- Lindqvist, A.; Mohapel, P.; Bouter, B.; Frielingsdorf, H.; Pizzo, D.; Brundin, P.; Erlanson-Albertsson, C. High-fat diet impairs hippocampal neurogenesis in male rats. Eur. J. Neurol. 2006, 13, 1385–1388. [Google Scholar] [CrossRef]
- Grillo, C.A.; Piroli, G.G.; Junor, L.; Wilson, S.P.; Mott, D.D.; Wilson, M.A.; Reagan, L.P. Obesity/hyperleptinemic phenotype impairs structural and functional plasticity in the rat hippocampus. Physiol. Behav. 2011, 105, 138–144. [Google Scholar] [CrossRef]
- Hsuchou, H.; He, Y.; Kastin, A.J.; Tu, H.; Markadakis, E.N.; Rogers, R.C.; Fossier, P.B.; Pan, W. Obesity induces functional astrocytic leptin receptors in hypothalamus. Brain J. Neurol. 2009, 132 Pt 4, 889–902. [Google Scholar] [CrossRef]
- Semple, B.D.; Blomgren, K.; Gimlin, K.; Ferriero, D.M.; Noble-Haeusslein, L.J. Brain development in rodents and humans: Identifying benchmarks of maturation and vulnerability to injury across species. Prog. Neurobiol. 2013, 106, 1–16. [Google Scholar] [CrossRef]
- Erion, J.R.; Wosiski-Kuhn, M.; Dey, A.; Hao, S.; Davis, C.L.; Pollock, N.K.; Stranahan, A.M. Obesity elicits interleukin 1-mediated deficits in hippocampal synaptic plasticity. J. Neurosci. 2014, 34, 2618–2631. [Google Scholar] [CrossRef]
- Gazdzinski, S.; Kornak, J.; Weiner, M.W.; Meyerhoff, D.J. Body mass index and magnetic resonance markers of brain integrity in adults. Ann. Neurol. 2008, 63, 652–657. [Google Scholar] [CrossRef]
- Lee, E.B.; Mattson, M.P. The neuropathology of obesity: Insights from human disease. Acta Neuropathol. 2014, 127, 3–28. [Google Scholar] [CrossRef] [PubMed]
- Park, H.; Poo, M.M. Neurotrophin regulation of neural circuit development and function. Nat. Rev. Neurosci. 2013, 14, 7–23. [Google Scholar] [CrossRef]
- Boyd, L.A.; Linsdell, M.A. Excitatory repetitive transcranial magnetic stimulation to left dorsal premotor cortex enhances motor consolidation of new skills. BMC Neurosci. 2009, 10, 72. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.Z.; Chen, R.S.; Rothwell, J.C.; Wen, H.Y. The after-effect of human theta burst stimulation is NMDA receptor dependent. Clin. Neurophysiol. 2007, 118, 1028–1032. [Google Scholar] [CrossRef] [PubMed]
- Rossi, S.; Hallett, M.; Rossini, P.M.; Pascual-Leone, A. Screening questionnaire before TMS: An update. Clin. Neurophysiol. 2011, 122, 1686. [Google Scholar] [CrossRef]
- Sale, M.V.; Ridding, M.C.; Nordstrom, M.A. Cortisol inhibits neuroplasticity induction in human motor cortex. J. Neurosci. 2008, 28, 8285–8293. [Google Scholar] [CrossRef]
- WHO. Global Database on Bady Mass Index WHO Website. 2015. Available online: http://appswhoint/bmi/indexjsp (accessed on 21 August 2020).
- Craig, C.L.; Marshall, A.L.; Sjöström, M.; Bauman, A.E.; Booth, M.L.; Ainsworth, B.E.; Pratt, M.; Ekelund, U.L.F.; Yngve, A.; Sallis, J.F.; et al. International physical activity questionnaire: 12-country reliability and validity. Med. Sci. Sports Exerc. 2003, 35, 1381–1395. [Google Scholar] [CrossRef]
- Buysse, D.J.; Reynolds, C.F., 3rd; Monk, T.H.; Berman, S.R.; Kupfer, D.J. The Pittsburgh Sleep Quality Index: A new instrument for psychiatric practice and research. Psychiatry Res. 1989, 28, 193–213. [Google Scholar] [CrossRef]
- Shiri, R.; Pourmemari, M.H.; Falah-Hassani, K.; Viikari-Juntura, E. The effect of excess body mass on the risk of carpal tunnel syndrome: A meta-analysis of 58 studies. Obes. Rev. 2015, 16, 1094–1104. [Google Scholar] [CrossRef]
- Hordacre, B.; Goldsworthy, M.R.; Vallence, A.M.; Darvishi, S.; Moezzi, B.; Hamada, M.; Rothwell, J.C.; Ridding, M.C. Variability in neural excitability and plasticity induction in the human cortex: A brain stimulation study. Brain Stimul. 2017, 10, 588–595. [Google Scholar] [CrossRef]
- Goldsworthy, M.R.; Pitcher, J.B.; Ridding, M.C. The application of spaced theta burst protocols induces long-lasting neuroplastic changes in the human motor cortex. Eur. J. Neurosci. 2012, 35, 125–134. [Google Scholar] [CrossRef] [PubMed]
- Goldsworthy, M.R.; Pitcher, J.B.; Ridding, M.C. Neuroplastic modulation of inhibitory motor cortical networks by spaced theta burst stimulation protocols. Brain Stimul. 2013, 6, 340–345. [Google Scholar] [CrossRef] [PubMed]
- Goldsworthy, M.R.; Müller-Dahlhaus, F.; Ridding, M.C.; Ziemann, U. Resistant against De-depression: LTD-Like Plasticity in the Human Motor Cortex Induced by Spaced cTBS. Cereb. Cortex. 2015, 25, 1724–1734. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.Z.; Edwards, M.J.; Rounis, E.; Bhatia, K.P.; Rothwell, J.C. Theta burst stimulation of the human motor cortex. Neuron 2005, 45, 201–206. [Google Scholar] [CrossRef]
- Hordacre, B.; Moezzi, B.; Goldsworthy, M.R.; Rogasch, N.C.; Graetz, L.J.; Ridding, M.C. Resting state functional connectivity measures correlate with the response to anodal transcranial direct current stimulation. Eur. J. Neurosci. 2017, 45, 837–845. [Google Scholar] [CrossRef]
- Hordacre, B.; Moezzi, B.; Ridding, M.C. Neuroplasticity and network connectivity of the motor cortex following stroke: A transcranial direct current stimulation study. Hum. Brain Mapp. 2018, 39, 3326–3339. [Google Scholar] [CrossRef]
- Ridding, M.C.; Ziemann, U. Determinants of the induction of cortical plasticity by non-invasive brain stimulation in healthy subjects. J. Physiol. 2010, 588 Pt 13, 2291–2304. [Google Scholar] [CrossRef]
- Hamada, M.; Murase, N.; Hasan, A.; Balaratnam, M.; Rothwell, J.C. The role of interneuron networks in driving human motor cortical plasticity. Cereb. Cortex. 2013, 23, 1593–1605. [Google Scholar] [CrossRef]
- Di Lazzaro, V.; Pilato, F.; Saturno, E.; Oliviero, A.; Dileone, M.; Mazzone, P.; Insola, A.; Tonali, P.A.; Ranieri, F.; Huang, Y.Z.; et al. Theta-burst repetitive transcranial magnetic stimulation suppresses specific excitatory circuits in the human motor cortex. J. Physiol. 2005, 565 Pt 3, 945–950. [Google Scholar] [CrossRef]
- Marqués-Iturria, I.; Pueyo, R.; Garolera, M.; Segura, B.; Junqué, C.; García-García, I.; Sender-Palacios, M.J.; Vernet-Vernet, M.; Narberhaus, A.; Ariza, M.; et al. Frontal cortical thinning and subcortical volume reductions in early adulthood obesity. Psychiatry Res. 2013, 214, 109–115. [Google Scholar] [CrossRef]
- Prickett, C.; Brennan, L.; Stolwyk, R. Examining the relationship between obesity and cognitive function: A systematic literature review. Obes. Res. Clin. Pract. 2015, 9, 93–113. [Google Scholar] [CrossRef] [PubMed]
- Gunstad, J.; Lhotsky, A.; Wendell, C.R.; Ferrucci, L.; Zonderman, A.B. Longitudinal examination of obesity and cognitive function: Results from the Baltimore longitudinal study of aging. Neuroepidemiology 2010, 34, 222–229. [Google Scholar] [CrossRef] [PubMed]
- Loprinzi, P.D.; Frith, E. Obesity and episodic memory function. J. Physiol. Sci. 2018, 68, 321–331. [Google Scholar] [CrossRef] [PubMed]
- Coppin, G.; Nolan-Poupart, S.; Jones-Gotman, M.; Small, D.M. Working memory and reward association learning impairments in obesity. Neuropsychologia 2014, 65, 146–155. [Google Scholar] [CrossRef]
- Bramham, C.R.; Messaoudi, E. BDNF function in adult synaptic plasticity: The synaptic consolidation hypothesis. Prog. Neurobiol. 2005, 76, 99–125. [Google Scholar] [CrossRef]
- Lu, B. BDNF and activity-dependent synaptic modulation. Learn Mem. 2003, 10, 86–98. [Google Scholar] [CrossRef]
- Minichiello, L. TrkB signalling pathways in LTP and learning. Nat. Rev. Neurosci. 2009, 10, 850–860. [Google Scholar] [CrossRef]
- Waterhouse, E.G.; Xu, B. New insights into the role of brain-derived neurotrophic factor in synaptic plasticity. Mol. Cell Neurosci. 2009, 42, 81–89. [Google Scholar] [CrossRef]
- Takei, N.; Numakawa, T.; Kozaki, S.; Sakai, N.; Endo, Y.; Takahashi, M.; Hatanaka, H. Brain-derived neurotrophic factor induces rapid and transient release of glutamate through the non-exocytotic pathway from cortical neurons. J. Biol. Chem. 1998, 273, 27620–27624. [Google Scholar] [CrossRef]
- Levine, E.S.; Kolb, J.E. Brain-derived neurotrophic factor increases activity of NR2B-containing N-methyl-D-aspartate receptors in excised patches from hippocampal neurons. J. Neurosci. Res. 2000, 62, 357–362. [Google Scholar] [CrossRef]
- Suen, P.C.; Wu, K.; Levine, E.S.; Mount, H.T.; Xu, J.L.; Lin, S.Y.; Black, I.B. Brain-derived neurotrophic factor rapidly enhances phosphorylation of the postsynaptic N-methyl-D-aspartate receptor subunit 1. Proc. Natl. Acad. Sci. USA 1997, 94, 8191–8195. [Google Scholar] [CrossRef] [PubMed]
- Lessmann, V.; Heumann, R. Modulation of unitary glutamatergic synapses by neurotrophin-4/5 or brain-derived neurotrophic factor in hippocampal microcultures: Presynaptic enhancement depends on pre-established paired-pulse facilitation. Neuroscience 1998, 86, 399–413. [Google Scholar] [CrossRef]
- Marty, S.; Berninger, B.; Carroll, P.; Thoenen, H. GABAergic stimulation regulates the phenotype of hippocampal interneurons through the regulation of brain-derived neurotrophic factor. Neuron 1996, 16, 565–570. [Google Scholar] [CrossRef]
- Rutherford, L.C.; DeWan, A.; Lauer, H.M.; Turrigiano, G.G. Brain-derived neurotrophic factor mediates the activity-dependent regulation of inhibition in neocortical cultures. J. Neurosci. 1997, 17, 4527–4535. [Google Scholar] [CrossRef]
- Mainardi, M.; Fusco, S.; Grassi, C. Modulation of hippocampal neural plasticity by glucose-related signaling. Neural. Plast. 2015, 2015, 657928. [Google Scholar] [CrossRef] [PubMed]
- Fung, P.K.; Robinson, P.A. Neural field theory of synaptic metaplasticity with applications to theta burst stimulation. J. Theor. Biol. 2014, 340, 164–176. [Google Scholar] [CrossRef]
- Cosentino, G.; Fierro, B.; Vigneri, S.; Talamanca, S.; Paladino, P.; Baschi, R.; Indovino, S.; Maccora, S.; Valentino, F.; Fileccia, E.; et al. Cyclical changes of cortical excitability and metaplasticity in migraine: Evidence from a repetitive transcranial magnetic stimulation study. Pain 2014, 155, 1070–1078. [Google Scholar] [CrossRef]
- Brighina, F.; Cosentino, G.; Vigneri, S.; Fierro, B. Mechanisms of cortical plasticity in migraine patients. Pain 2014, 155, 643–644. [Google Scholar] [CrossRef]
| Healthy | Obese | Statistics | |
|---|---|---|---|
| Demographic data | n = 16 | n = 14 | |
| Handedness (n, right/left) | (14:2) | (11:3) | p = 0.642 |
| Gender (n, male/female) | (7:9) | (9:5) | p = 0.299 |
| Age (years, mean (SD)) | 26.3 (8.5) | 35.4 (14.3) | U = 68.5, p = 0.070 |
| IPAQ total (MET mins, mean (SD)) | 4487.6 (5339.1) | 5381.7 (9229.5) | U = 106.0, p = 0.822 |
| PSQI (mean (SD)) | 4.25 (3.17) | 6.14 (3.72) | U = 73.5, p = 0.110 |
| BMI (kg/m2, range) | 22.0 (20.0–24.9) | 43.7 (30.0–68.0) | U = 0.0, p < 0.001 |
| Percentage of body fat (%, mean (SD)) | 19.7 (6.4) | 36.1 (15.3) | U = 32.0, p = 0.010 |
| Waist–hip ratio mean (SD) | 0.81 (0.06) | 0.95 (0.06) | t(28) = 6.16, p < 0.001 |
| Baseline excitability data | |||
| RMT (% MSO, mean (SD) | 41.4 (7.0) | 43.9 (5.0) | t(28) = 1.11, p = 0.279 |
| SI1mV (% MSO, mean (SD)) | 57.4 (14.6) | 62.6 (9.3) | t(28) = 1.14, p = 0.263 |
| Baseline MEP amplitude (mV, mean (SD)) | 1.01 (0.21) | 0.98 (0.32) | t(28) = 0.32, p = 0.750 |
| MEP onset latency (ms, mean (SD)) | 24.16 (2.52) | 24.25 (1.77) | t(28) = 0.11, p = 0.916 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sui, S.X.; Ridding, M.C.; Hordacre, B. Obesity is Associated with Reduced Plasticity of the Human Motor Cortex. Brain Sci. 2020, 10, 579. https://doi.org/10.3390/brainsci10090579
Sui SX, Ridding MC, Hordacre B. Obesity is Associated with Reduced Plasticity of the Human Motor Cortex. Brain Sciences. 2020; 10(9):579. https://doi.org/10.3390/brainsci10090579
Chicago/Turabian StyleSui, Sophia X., Michael C. Ridding, and Brenton Hordacre. 2020. "Obesity is Associated with Reduced Plasticity of the Human Motor Cortex" Brain Sciences 10, no. 9: 579. https://doi.org/10.3390/brainsci10090579
APA StyleSui, S. X., Ridding, M. C., & Hordacre, B. (2020). Obesity is Associated with Reduced Plasticity of the Human Motor Cortex. Brain Sciences, 10(9), 579. https://doi.org/10.3390/brainsci10090579





