Evaluating Social Interactions Using the Autism Screening Instrument for Education Planning-3rd Edition (ASIEP-3): Interaction Assessment in Children and Adults with Fragile X Syndrome
Abstract
1. Introduction
1.1. Autism Spectrum Disorder in FXS
1.2. FXS Clinical Trials and Outcome Measures
1.3. Rationale for the Study
1.4. Aims of the Study
1.5. Research Questions
- Can the ASIEP-3 Interaction Assessment be reliably administered and scored in a sample of individuals with fragile X syndrome across a broad range of ages and phenotypes?
- How does performance on the ASIEP-3 Interaction Assessment vary in relation to age, gender, IQ, and behavioral functioning for individuals with FXS?
- How do ASIEP-3 Interaction Assessment results compare between individuals with FXS and a clinical diagnosis of ASD (FX+ASD) and those without ASD (FX−ASD)?
2. Methods
2.1. Study Design
2.1.1. Recruitment
2.1.2. Measures
Stanford Binet Intelligence Scales, Fifth Edition (SB-V)
Vineland Adaptive Behavior Scales 3rd Edition, Comprehensive Interview (Vineland-3)
Aberrant Behavior Checklist-Community Edition (ABC-C)
Social Communication Questionnaire (SCQ)
Social Responsiveness Scale—Second Edition (SRS-2)
Autism Screening Instrument for Educational Planning—Third Edition (ASIEP-3)
Interest Inventory
ASIEP-3 Administration Checklist
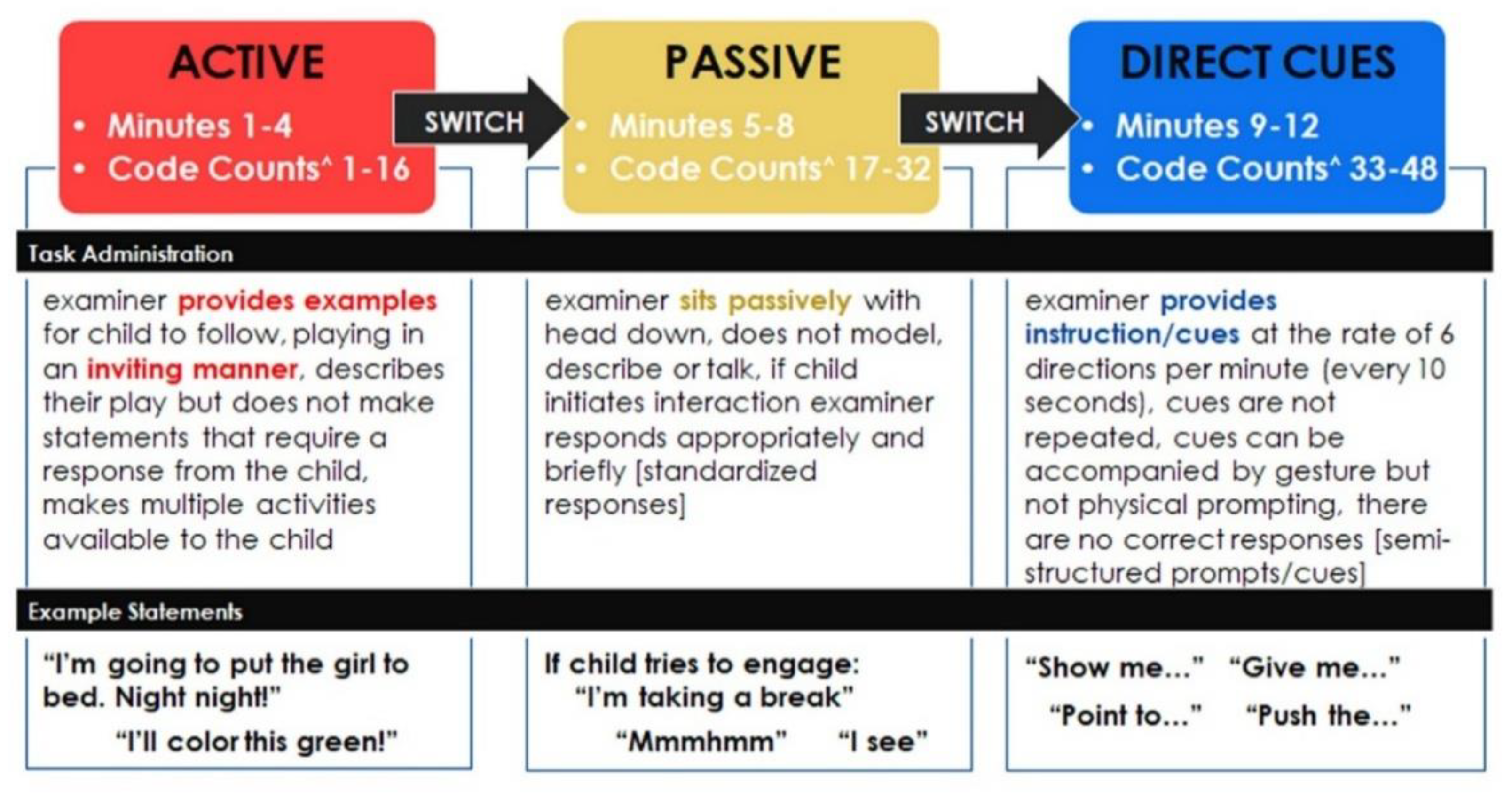
2.2. ASIEP-3 Administration Training
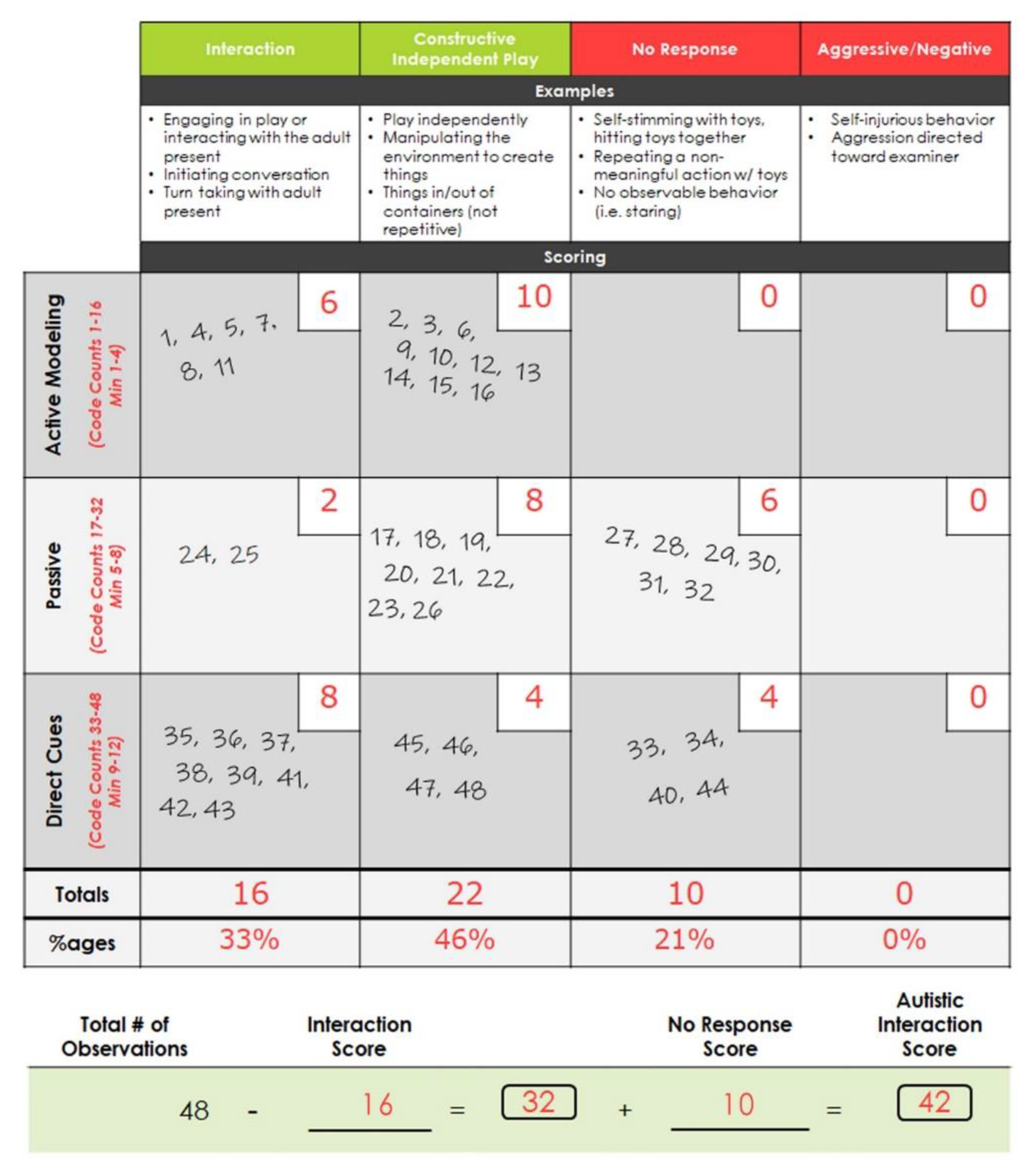
2.3. ASIEP-3 Scoring
2.4. Analysis
- Item 4: “aggressive to other children or adults”
- Item 10 “temper tantrums/outbursts”
- Item 47 “stamps feet or bangs objects or slams doors”
- Item 57 “has temper outbursts or tantrums when he/she does not get own way”
3. Results
3.1. Participant Characteristics
3.2. ASIEP-3
3.2.1. ASIEP-3 Feasibility and Reliability
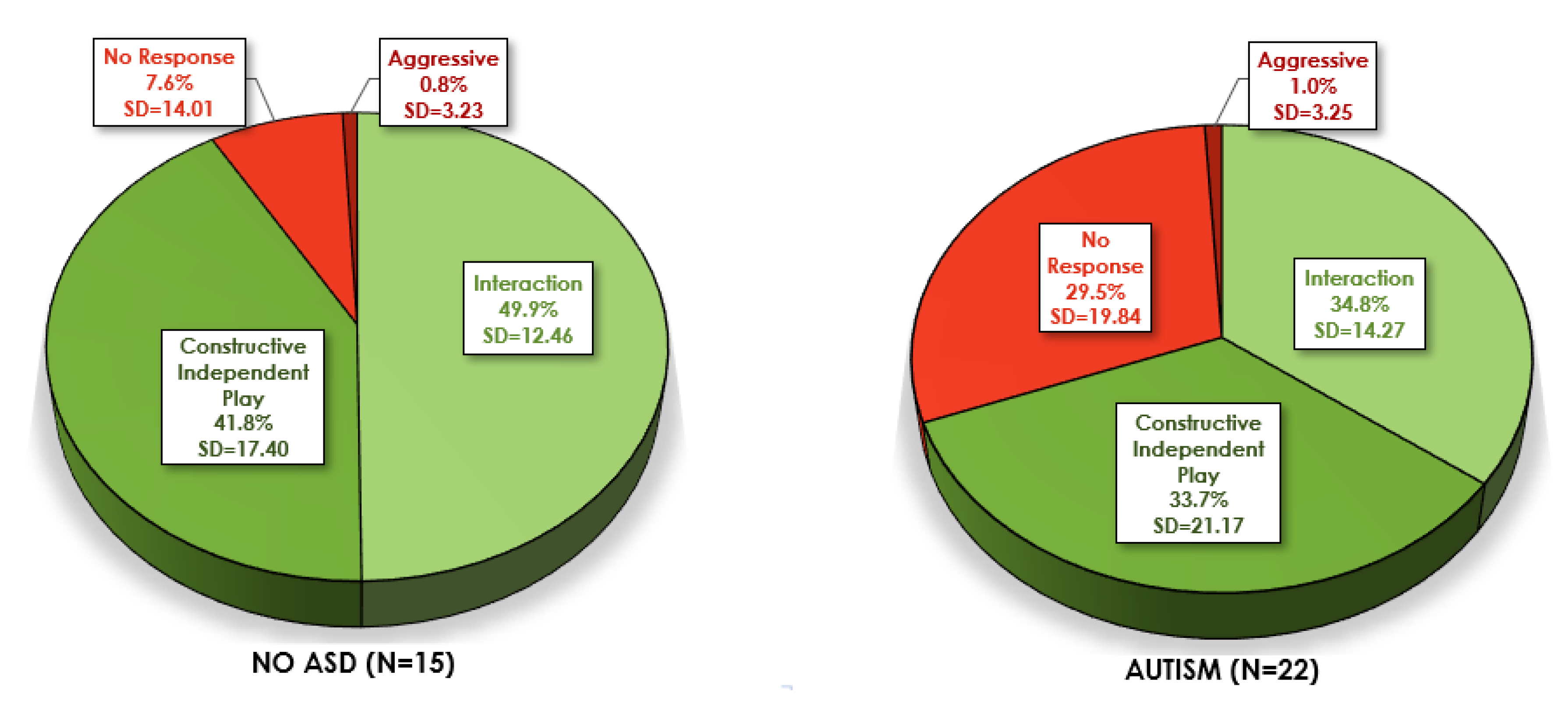
3.2.2. ASIEP-3 Score Profiles
3.2.3. ASIEP Validity with ASD Diagnosis and other Measures
3.2.4. Exploratory and Ad-hoc Analyses
4. Discussion
4.1. Summary of Findings
4.2. Limitations and Next Steps
4.3. Clinical Considerations in the Educational Settings and Beyond
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Crawford, D.C.; Acuña, J.M.; Sherman, S.L.; Acu, J.M. FMR1 and the fragile X syndrome: Human genome epidemiology review. Genet. Med. 2001, 3, 359–371. [Google Scholar] [CrossRef]
- Bassell, G.J.; Warren, S. Fragile X Syndrome: Loss of Local mRNA Regulation Alters Synaptic Development and Function. Neuron 2008, 60, 201–214. [Google Scholar] [CrossRef]
- Loesch, D.Z.; Huggins, R.; Hagerman, R. Phenotypic variation and FMRP levels in fragile X. Ment. Retard. Dev. Disabil. Res. Rev. 2004, 10, 31–41. [Google Scholar] [CrossRef] [PubMed]
- Scerif, G.; Cornish, K.; Wilding, J.; Driver, J.; Karmiloff-Smith, A. Delineation of early attentional control difficulties in fragile X syndrome: Focus on neurocomputational changes. Neuropsychologia 2007, 45, 1889–1898. [Google Scholar] [CrossRef] [PubMed]
- Hagerman, R.J.; Hull, C.E.; Safanda, J.F.; Carpenter, I.; Staley, L.W.; O’Connor, R.A.; Seydel, C.; Mazzocco, M.M.M.; Snow, K.; Thibodeau, S.N.; et al. High functioning fragile X males: Demonstration of an unmethylated fully expanded FMR-1 mutation associated with protein expression. Am. J. Med Genet. 1994, 51, 298–308. [Google Scholar] [CrossRef] [PubMed]
- Murphy, M.M. A review of mathematical learning disabilities in children with fragile X syndrome. Dev. Disabil. Res. Rev. 2009, 15, 21–27. [Google Scholar] [CrossRef]
- Lachiewicz, A.M.; Dawson, D.V.; Spiridigliozzi, G.A.; McConkie-Rosell, A. Arithmetic difficulties in females with the fragile X premutation. Am. J. Med. Genet. Part A 2006, 140, 665–672. [Google Scholar] [CrossRef] [PubMed]
- Sudhalter, V.; Cohen, I.L.; Silverman, W.; Wolf-Schein, E.G. Conversational analyses of males with fragile X, Down syndrome, and autism: Comparison of the emergence of deviant language. Am. J. Ment. Retard. 1990, 94, 431–441. [Google Scholar]
- Reiss, A.L.; Freund, L. Behavioral phenotype of fragile X syndrome: DSM-III-R autistic behavior in male children. Am. J. Med. Genet. 1992, 43, 35–46. [Google Scholar] [CrossRef] [PubMed]
- Cohen, I.L.; Vietze, P.M.; Sudhalter, V.; Jenkins, E.C.; Brown, W.T. Effects of age and communication level on eye contact in fragile X males and non-fragile X autistic males. Am. J. Med. Genet. 1991, 38, 498–502. [Google Scholar] [CrossRef]
- Hessl, D.; Dyer-Friedman, J.; Glaser, B.; Wisbeck, J.; Barajas-Gonzalez, R.G.; Taylor, A.; Reiss, A.L. The influence of environmental and genetic factors on behavior problems and autistic symptoms in boys and girls with fragile X syndrome. Pediatrics 2001, 108, e88. [Google Scholar] [CrossRef] [PubMed]
- Cordeiro, L.; Ballinger, E.; Hagerman, R.; Hessl, D. Clinical assessment of DSM-IV anxiety disorders in fragile X syndrome: Prevalence and characterization. J. Neurodev. Disord. 2010, 3, 57–67. [Google Scholar] [CrossRef] [PubMed]
- Lachiewicz, A.M. Abnormal behaviors of young girls with fragile X syndrome. Am. J. Med. Genet. 1992, 43, 72–77. [Google Scholar] [CrossRef]
- Lachiewicz, A.M.; Dawson, D.V. Behavior problems of young girls with fragile X syndrome: Factor scores on the Conners’ parent’s questionnaire. Am. J. Med. Genet. 1994, 51, 364–369. [Google Scholar] [CrossRef] [PubMed]
- Braden, M. Braden on Behavior: The Uniqueness of Females with FXS. Available online: https://fragilex.org/fxs/uniqueness-females-fragile-x-syndrome/ (accessed on 15 April 2020).
- Clifford, S.; Dissanayake, C.; Bui, M.; Huggins, R.; Taylor, A.K.; Loesch, D.Z. Autism Spectrum Phenotype in Males and Females with Fragile X Full Mutation and Premutation. J. Autism Dev. Disord. 2006, 37, 738–747. [Google Scholar] [CrossRef] [PubMed]
- Hall, S.; Lightbody, A.A.; Reiss, A.L. Compulsive, self-injurious, and autistic behavior in children and adolescents with fragile X syndrome. Am. J. Ment. Retard. 2008, 113, 44–53. [Google Scholar] [CrossRef]
- Harris, S.W.; Hessl, D.; Goodlin-Jones, B.; Ferranti, J.; Bacalman, S.; Barbato, I.; Tassone, F.; Hagerman, P.J.; Herman, H.; Hagerman, R.J. Autism profiles of males with fragile X syndrome. Am. J. Ment. Retard. 2008, 113, 427–438. [Google Scholar] [CrossRef] [PubMed]
- Kaufmann, W.E.; Cortell, R.; Kau, A.S.; Bukelis, I.; Tierney, E.; Gray, R.M.; Cox, C.; Capone, G.T.; Stanard, P. Autism spectrum disorder in fragile X syndrome: Communication, social interaction, and specific behaviors. Am. J. Med. Genet. 2004, 129, 225–234. [Google Scholar] [CrossRef]
- Bailey, D.B.; Raspa, M.; Olmsted, M.; Holiday, D.B. Co-occurring conditions associated withFMR1gene variations: Findings from a national parent survey. Am. J. Med. Genet. Part A 2008, 146, 2060–2069. [Google Scholar] [CrossRef]
- Budimirovic, D.; Bukelis, I.; Cox, C.; Gray, R.M.; Tierney, E.; Kaufmann, W.E. Autism spectrum disorder in Fragile X syndrome: Differential contribution of adaptive socialization and social withdrawal. Am. J. Med. Genet. Part A 2006, 140, 1814–1826. [Google Scholar] [CrossRef]
- Hernandez, R.N.; Feinberg, R.L.; Vaurio, R.; Passanante, N.M.; Thompson, R.E.; Kaufmann, W.E. Autism spectrum disorder in fragile X syndrome: A longitudinal evaluation. Am. J. Med. Genet. Part A 2009, 149, 1125–1137. [Google Scholar] [CrossRef] [PubMed]
- Kaufmann, W.E.; Kidd, S.A.; Andrews, H.F.; Budimirovic, D.B.; Esler, A.; Haas-Givler, B.; Stackhouse, T.; Riley, C.; Peacock, G.; Sherman, S.L.; et al. Autism Spectrum Disorder in Fragile X Syndrome: Cooccurring Conditions and Current Treatment. Pediatrics 2017, 139, S194–S206. [Google Scholar] [CrossRef]
- Lewis, P.; Abbeduto, L.; Murphy, M.; Richmond, E.; Giles, N.; Bruno, L.; Schroeder, S. Cognitive, language and social-cognitive skills of individuals with fragile X syndrome with and without autism. J. Intellect. Disabil. Res. 2006, 50, 532–545. [Google Scholar] [CrossRef] [PubMed]
- Niu, M.; Han, Y.; Dy, A.B.; Du, J.; Jin, H.; Qin, J.; Zhang, J.; Li, Q.; Hagerman, R.J. Autism Symptoms in Fragile X Syndrome. J. Child Neurol. 2017, 32, 903–909. [Google Scholar] [CrossRef]
- Hogan, A.L.; Caravella, K.E.; Ezell, J.; Rague, L.; Hills, K.; Roberts, J.E. Autism Spectrum Disorder Symptoms in Infants with Fragile X Syndrome: A Prospective Case Series. J. Autism Dev. Disord. 2017, 47, 1628–1644. [Google Scholar] [CrossRef] [PubMed]
- Boyle, L.; Kaufmann, W.E. The behavioral phenotype of FMR1 mutations. Am. J. Med. Genet. Part C Semin. Med Genet. 2010, 154, 469–476. [Google Scholar] [CrossRef]
- Talisa, V.B.; Boyle, L.; Crafa, D.; Kaufmann, W.E. Autism and anxiety in males with fragile X syndrome: An exploratory analysis of neurobehavioral profiles from a parent survey. Am. J. Med Genet. Part A 2014, 164, 1198–1203. [Google Scholar] [CrossRef]
- Berry-Kravis, E. Mechanism-Based Treatments in Neurodevelopmental Disorders: Fragile X Syndrome. Pediatr. Neurol. 2014, 50, 297–302. [Google Scholar] [CrossRef]
- Berry-Kravis, E.; Knox, A.; Hervey, C. Targeted treatments for fragile X syndrome. J. Neurodev. Disord. 2011, 3, 193–210. [Google Scholar] [CrossRef]
- Berry-Kravis, E.; Hessl, D.; Rathmell, B.; Zarevics, P.; Cherubini, M.; Walton-Bowen, K.; Mu, Y.; Nguyen, D.V.; Gonzalez-Heydrich, J.; Wang, P.P.; et al. Effects of STX209 (Arbaclofen) on Neurobehavioral Function in Children and Adults with Fragile X Syndrome: A Randomized, Controlled, Phase 2 Trial. Sci. Transl. Med. 2012, 4, 152ra127. [Google Scholar] [CrossRef]
- Erickson, C.A.; Veenstra-VanderWeele, J.; Melmed, R.D.; McCracken, J.T.; Ginsberg, L.D.; Sikich, L.; Scahill, L.; Cherubini, M.; Zarevics, P.; Walton-Bowen, K.; et al. STX209 (Arbaclofen) for Autism Spectrum Disorders: An 8-Week Open-Label Study. J. Autism Dev. Disord. 2013, 44, 958–964. [Google Scholar] [CrossRef] [PubMed]
- Jacquemont, S.; Curie, A.; Portes, V.D.; Torrioli, M.G.; Berry-Kravis, E.; Hagerman, R.; Ramos, F.J.; Cornish, K.; He, Y.; Paulding, C.; et al. Epigenetic Modification of the FMR1 Gene in Fragile X Syndrome Is Associated with Differential Response to the mGluR5 Antagonist AFQ056. Sci. Transl. Med. 2011, 3, 64ra1. [Google Scholar] [CrossRef] [PubMed]
- Berry-Kravis, E.; Hagerman, R.; Visootsak, J.; Budimirovic, D.; Kaufmann, W.E.; Cherubini, M.; Zarevics, P.; Walton-Bowen, K.; Wang, P.; Bear, M.F.; et al. Arbaclofen in fragile X syndrome: Results of phase 3 trials. J. Neurodev. Disord. 2017, 9, 3–18. [Google Scholar] [CrossRef]
- De Sonia, A.V.J.; Smith, M. FXCRC Analysis of Arbaclofen Responses in Fragile X Syndrome; Springer: Orange County, CA, USA, 2014. [Google Scholar]
- Lee, A.W.; Ventola, P.E.; Budimirovic, D.; Berry-Kravis, E.; Visootsak, J. Clinical Development of Targeted Fragile X Syndrome Treatments: An Industry Perspective. Brain Sci. 2018, 8, 214. [Google Scholar] [CrossRef]
- Berry-Kravis, E.; Hessl, D.; Abbeduto, L.; Reiss, A.L.; Beckel-Mitchener, A.; Urv, T.K. Outcome Measures for Clinical Trials in Fragile X Syndrome. J. Dev. Behav. Pediatr. 2013, 34, 508–522. [Google Scholar] [CrossRef] [PubMed]
- Budimirovic, D.B.; Berry-Kravis, E.; Erickson, C.A.; Hall, S.; Hessl, D.; Reiss, A.L.; King, M.K.; Abbeduto, L.; Kaufmann, W.E. Updated report on tools to measure outcomes of clinical trials in fragile X syndrome. J. Neurodev. Disord. 2017, 9, 14. [Google Scholar] [CrossRef]
- Erickson, C.A.; Kaufmann, W.E.; Budimirovic, D.; Lachiewicz, A.; Haas-Givler, B.; Miller, R.M.; Weber, J.D.; Abbeduto, L.; Hessl, D.; Hagerman, R.; et al. Best Practices in Fragile X Syndrome Treatment Development. Brain Sci. 2018, 8, 224. [Google Scholar] [CrossRef]
- Knox, A.; Schneider, A.; Abucayan, F.; Hervey, C.; Tran, C.; Hessl, D.; Berry-Kravis, E. Feasibility, reliability, and clinical validity of the Test of Attentional Performance for Children (KiTAP) in Fragile X syndrome (FXS). J. Neurodev. Disord. 2012, 4, 2. [Google Scholar] [CrossRef]
- Berry-Kravis, E.; Doll, E.; Sterling, A.; Kover, S.T.; Schroeder, S.M.; Mathur, S.; Abbeduto, L. Development of an Expressive Language Sampling Procedure in Fragile X Syndrome: A Pilot Study. J. Dev. Behav. Pediatr. 2013, 34, 245–251. [Google Scholar] [CrossRef]
- Abbeduto, L.; Berry-Kravis, E.; Sterling, A.; Sherman, S.; Edgin, J.O.; McDuffie, A.; Hoffmann, A.; Hamilton, D.; Nelson, M.; Aschkenasy, J.; et al. Expressive language sampling as a source of outcome measures for treatment studies in fragile X syndrome: Feasibility, practice effects, test-retest reliability, and construct validity. J. Neurodev. Disord. 2020, 12, 10. [Google Scholar] [CrossRef]
- Hessl, D.; Sansone, S.M.; Berry-Kravis, E.; Riley, K.; Widaman, K.F.; Abbeduto, L.; Schneider, A.; Coleman, J.; Oaklander, D.; Rhodes, K.C.; et al. The NIH Toolbox Cognitive Battery for intellectual disabilities: Three preliminary studies and future directions. J. Neurodev. Disord. 2016, 8, 35. [Google Scholar] [CrossRef] [PubMed]
- Shields, R.H.; Kaat, A.J.; McKenzie, F.J.; Drayton, A.; Sansone, S.M.; Coleman, J.; Michalak, C.; Riley, K.; Berry-Kravis, E.; Gershon, R.C.; et al. Validation of the NIH Toolbox Cognitive Battery in intellectual disability. Neurology 2020, 94, e1229–e1240. [Google Scholar] [CrossRef] [PubMed]
- Benjamin, D.P.; Mastergeorge, A.M.; McDuffie, A.; Kover, S.T.; Hagerman, R.; Abbeduto, L. Effects of labeling and pointing on object gaze in boys with fragile X syndrome: An eye-tracking study. Res. Dev. Disabil. 2014, 35, 2658–2672. [Google Scholar] [CrossRef]
- Klusek, J.; Moser, C.; Schmidt, J.; Abbeduto, L.; Roberts, J.E. A novel eye-tracking paradigm for indexing social avoidance-related behavior in fragile X syndrome. Am. J. Med. Genet. Part B Neuropsychiatr. Genet. 2019, 183, 5–16. [Google Scholar] [CrossRef] [PubMed]
- Hall, S.S.; Frank, M.C.; Pusiol, G.T.; Farzin, F.; Lightbody, A.A.; Reiss, A.L. Quantifying naturalistic social gaze in fragile X syndrome using a novel eye tracking paradigm. Am. J. Med. Genet. Part B Neuropsychiatr. Genet. 2015, 168, 564–572. [Google Scholar] [CrossRef]
- Kidd, S.A.; Berry-Kravis, E.; Choo, T.H.; Chen, C.; Esler, A.; Hoffmann, A.; Andrews, H.F.; Kaufmann, W.E. Improving the Diagnosis of Autism Spectrum Disorder in Fragile X Syndrome by Adapting the Social Communication Questionnaire and the Social Responsiveness Scale-2. J. Autism Dev. Disord. 2019, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Lord, C.; Rutter, M. Autism Diagnostic Observation Scales-2nd Edition (ADOS-2); Western Psychological Services: Torrance, CA, USA, 2012. [Google Scholar]
- Levitas, A.; Hagerman, R.J.; Braden, M.; Rimland, B.; McBOGG, P.; Matus, I. Autism and the Fragile X Syndrome. J. Dev. Behav. Pediatr. 1983, 4, 151–158. [Google Scholar] [CrossRef]
- Krug, D.; Arick, J.; Almond, P. Autism Screening Instrument for Educational Planning; ProEd: Austin, TX, USA, 1978. [Google Scholar]
- Krug, D.; Almond P, A.J. ASIEP-3: Autism Screening Instrument for Educational Planning, 3rd ed.; PRO-ED Inc.: Austin, TX, USA, 2008. [Google Scholar]
- Teal, M.B.; Wiebe, M.J. A validity analysis of selected instruments used to assess autism. J. Autism Dev. Disord. 1986, 16, 485–494. [Google Scholar] [CrossRef]
- Sherman, S.L.; Kidd, S.A.; Riley, C.; Berry-Kravis, E.; Andrews, H.F.; Miller, R.M.; Lincoln, S.; Swanson, M.; Kaufmann, W.E.; Brown, W.T. FORWARD: A Registry and Longitudinal Clinical Database to Study Fragile X Syndrome. Pediatrics 2017, 139, S183–S193. [Google Scholar] [CrossRef]
- Roid, H.G.; Pomplun, M. The Stanford-Binet Intelligence Scales; The Guilford Press: New York, NY, USA, 2012. [Google Scholar]
- Sansone, S.M.; Schneider, A.; Bickel, E.; Berry-Kravis, E.; Prescott, C.; Hessl, D. Improving IQ measurement in intellectual disabilities using true deviation from population norms. J. Neurodev. Disord. 2014, 6, 16. [Google Scholar] [CrossRef]
- Sparrow, S.; Balla, D.; Cicchetti, D. Vineland Adaptive Behavior Scales; American Guidance Service: Circle Pines, MN, USA, 1984. [Google Scholar]
- Sparrow, S.; Cicchetti, D.; Saulnier, C. Vineland Adaptive Behavior Scales–Third Edition (Vineland-3); Pearson: San Antonio, TX, USA, 2016. [Google Scholar]
- Pepperdine, C.R.; McCrimmon, A. Test Review: Vineland Adaptive Behavior Scales, Third Edition (Vineland-3) by Sparrow, S. S., Cicchetti, D. V., & Saulnier, C. A. Can. J. Sch. Psychol. 2017, 33, 157–163. [Google Scholar] [CrossRef]
- Aman, G.M.; Singh, N. Aberrant Behavior Checklist; Community (ABC-C); Slosson Educational Publications: East Aurora, NY, USA, 1994. [Google Scholar]
- Aman, M.G.; Burrow, W.H.; Wolford, P.L. The Aberrant Behavior Checklist-Community: Factor validity and effect of subject variables for adults in group homes. Am. J. Ment. Retard. 1995, 100, 283–292. [Google Scholar] [PubMed]
- Brown, E.C.; Aman, M.G.; Havercamp, S.M. Factor analysis and norms for parent ratings on the Aberrant Behavior Checklist-Community for young people in special education. Res. Dev. Disabil. 2002, 23, 45–60. [Google Scholar] [CrossRef]
- Brinkley, J.; Nations, L.; Abramson, R.K.; Hall, A.; Wright, H.H.; Gabriels, R.; Gilbert, J.R.; Pericak-Vance, M.A.O.; Cuccaro, M.L. Factor Analysis of the Aberrant Behavior Checklist in Individuals with Autism Spectrum Disorders. J. Autism Dev. Disord. 2006, 37, 1949–1959. [Google Scholar] [CrossRef]
- Berry-Kravis, E.; Krause, S.E.; Block, S.S.; Guter, S.; Wuu, J.; Leurgans, S.; Decle, P.; Potanos, K.; Cook, E.; Salt, J.; et al. Effect of CX516, an AMPA-Modulating Compound, on Cognition and Behavior in Fragile X Syndrome: AControlled Trial. J. Child Adolesc. Psychopharmacol. 2006, 16, 525–540. [Google Scholar] [CrossRef]
- Sansone, S.M.; Widaman, K.F.; Hall, S.S.; Reiss, A.L.; Lightbody, A.; Kaufmann, W.E.; Berry-Kravis, E.; Lachiewicz, A.; Brown, E.C.; Hessl, D. Psychometric Study of the Aberrant Behavior Checklist in Fragile X Syndrome and Implications for Targeted Treatment. J. Autism Dev. Disord. 2011, 42, 1377–1392. [Google Scholar] [CrossRef] [PubMed]
- Wheeler, A.; Raspa, M.; Bann, C.; Bishop, E.; Hessl, D.; Sacco, P.; Bailey, D.B. Anxiety, attention problems, hyperactivity, and the Aberrant Behavior Checklist in fragile X syndrome. Am. J. Med. Genet. Part A 2013, 164, 141–155. [Google Scholar] [CrossRef]
- Kau, A.S.; Tierney, E.; Bukelis, I.; Stump, M.H.; Kates, W.; Trescher, W.H.; Kaufmann, W.E. Social behavior profile in young males with fragile X syndrome: Characteristics and specificity. Am. J. Med. Genet. 2004, 126, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Rutter, M.; Rutter, M.; Bailey, A.; Lord, C. Social Communication Questionnaire (SCQ); Western Psychological Services: Los Angeles, CA, USA, 2003. [Google Scholar]
- Ozonoff, S.; Goodlin-Jones, B.L.; Solomon, M. Evidence-Based Assessment of Autism Spectrum Disorders in Children and Adolescents. J. Clin. Child Adolesc. Psychol. 2005, 34, 523–540. [Google Scholar] [CrossRef] [PubMed]
- Constantino, J.N. Social Responsiveness Scale-Second Edition (SRS-2); Western Psychological Services: Los Angeles, CA, USA, 2012. [Google Scholar]
- Shaffer, R.; Schmitt, L.M.; Thurman, A.J.; Abbeduto, L.; Hong, M.; Pedapati, E.V.; Dominick, K.; Sweeney, J.; Erickson, C.A. The Relationship between Expressive Language Sampling and Clinical Measures in Fragile X Syndrome and Typical Development. Brain Sci. 2020, 10, 66. [Google Scholar] [CrossRef]
- Hessl, D.; Tassone, F.; Cordeiro, L.; Koldewyn, K.; McCormick, C.; Green, C.; Wegelin, J.; Yuhas, J.; Hagerman, R.J. Brief Report: Aggression and Stereotypic Behavior in Males with Fragile X Syndrome—Moderating Secondary Genes in a “Single Gene” Disorder. J. Autism Dev. Disord. 2007, 38, 184–189. [Google Scholar] [CrossRef] [PubMed]
- Wheeler, A.C.; Raspa, M.; Bishop, E.; Bailey, D.B. Aggression in fragile X syndrome. J. Intellect. Disabil. Res. 2015, 60, 113–125. [Google Scholar] [CrossRef] [PubMed]
- Mirrett, P.L.; Roberts, J.E.; Price, J. Early Intervention Practices and Communication Intervention Strategies for Young Males With Fragile X Syndrome. Lang. Speech. Hear. Serv. Sch. 2003, 34, 320–331. [Google Scholar] [CrossRef]
- Riley, K.; Braden M, S.V. Braden on Behavior: Assessment Considerations for Children with Fragile X Syndrome. Available online: https://marciabraden.com/images/ebooks/Reflecting-on-FXS.pdf (accessed on 15 April 2020).
- Thompson, T.; Coleman, J.M.; Riley, K.; Snider, L.A.; Howard, L.J.; Sansone, S.M.; Hessl, D. Standardized Assessment Accommodations for Individuals with Intellectual Disability. Contemp. Sch. Psychol. 2018, 22, 443–457. [Google Scholar] [CrossRef] [PubMed]
- Symons, F.J.; Clark, R.D.; Roberts, J.P.; Bailey, N.B. Classroom Behavior of Elementary School-Age Boys with Fragile X Syndrome. J. Spéc. Educ. 2001, 34, 194–202. [Google Scholar] [CrossRef]
| Males | Females | Total | Statistical Results (t–Tests and Fisher’s Exact Tests) | |
|---|---|---|---|---|
| Age N | 26 | 13 | 39 | |
| M (SD) | 13.1 (8.1) | 17.1 (13.4) | 14.4 (10.2) | t(37) = −1.177, p = 0.247 |
| Range | 3.2–29.9 | 1.6–46.5 | 1.6–46.5 | |
| Ethnicity: Hispanic N (%) | 5 (19.2%) | 1 (7.7%) | 6 (15.8%) | p = 0.643 |
| Race: Caucasian N (%) | 5 (19.2%) | 1 (7.7%) | 31 (81.6%) | p = 0.189 |
| Stanford-Binet-V^ N | 25 | 11 | 36 | |
| ABIQ-M (SD) | 54.0 (12.9) | 70.1 (21.3) | 58.9 (17.3) | t(34) = −2.813, p = 0.036 * |
| ABIQ-Range | 47–100 | 47–109 | 47–109 | |
| FSIQ-M (SD) | 46.5 (8.4) | 66.7 (17.2) | 53.7 (15.5) | t(29) = −4.423, p = 0.003 ** |
| FSIQ-Range | 40–67 | 40–88 | 40–88 | |
| FSIQz deviation score- M (SD) | 47.2 (17.6) | 73.6 (18.2) | 57.0 (21.74) | t(28) = −3.909, p = 0.001 ** |
| FSIQz deviation score- Range | 10.5–79.5 | 35.1–98.0 | 10.5–98.0 | |
| Vineland Adaptive Behavior Scales-3 | ||||
| Adaptive Composite-M (SD) | 51.2 (16.8) | 65.6 (30.2) | 55.4 (22.1) | t(36) = −1.418, p = 0.183 |
| Adaptive Composite-Range | 20–80 | 20–98 | 20–98 | |
| Socialization-M (SD) | 55.1 (18.0) | 70.2 (27.7) | 59.6 (22.1) | t(36) = −1.978, p = 0.056 |
| Socialization-Range | 20–92 | 20–105 | 20–105 | |
| Communication-M (SD) | 49.2 (23.0) | 57.9 (28.4) | 52.0 (24.7) | t(36) = −1.007, p = 0.321 |
| Communication-Range | 20–95 | 20–91 | 20–95 | |
| Clinical Autism Diagnosis | ||||
| No ASD- N (%) | 4 (16.7%) | 11 (84.6%) | 15 (40.5%) | p < 0.001 |
| ASD- N (%) | 20 (83.3%) | 2 (15.4%) | 22 (56.4%) | |
| Aberrant Behavior Checklist | ||||
| Total Score-M (SD) | 43.1 (28.1) | 25.4 (22.5) | 37.0 (27.3) | t(30) = 1.806, p = 0.081 |
| Total Score-Range | 9–98 | 0–77 | 0–98 | |
| Social Avoidance Score-M (SD) | 2.10 (2.68) | 2.18 (3.66) | 2.13 (2.99) | t(30) = −0.076, p = 0.940 |
| Social Avoidance Score-Range | 0–8 | 0–12 | 0–12 | |
| Social Communication Questionnaire | ||||
| Total Score-M (SD) | 15.4 (6.1) | 9.1 (7.2) | 13.6 (6.9) | t(26) = 2.334, p = 0.028 * |
| Total T-score Range | 5–26 | 3–25 | 3–26 | |
| Social Responsiveness Scale-2 | ||||
| Total T-score-M (SD) | 69.8 (10.7) | 62.4 (14.8) | 67.7 (12.2) | t(22) = 1.3807, p = 0.181 |
| Total T-score Range | 48–87 | 47–88 | 47–88 |
| Males (N = 26) | Females (N = 13) | Total (N = 39) | Statistical Results (t-Tests) | |
|---|---|---|---|---|
| ASIEP-3 Scores | ||||
| Interaction Percentage-M (SD) | 36.5% (14.5) | 48.7% (14.3) | 40.6% (15.4) | t(37) = −2.491, p = 0.017 * |
| Interaction-Range | 8.3%–56.3% | 16.7%–68.8% | 8.3%–68.8% | |
| CIP Percentage-M (SD) | 33.8% (21.3) | 42.8% (16.2) | 36.8% (20.0) | t(37) = −1.336, p = 0.190 |
| CIP Range | 4.2%–75.0% | 6.3%–70.8% | 4.2%–75% | |
| No Response Percentage-M (SD) | 27.4% (21.2) | 8.5% (12.5) | 21.1% (20.7) | t(37) = 3.491, p = 0.001 *** |
| No Response-Range | 0%–75.0% | 0%–35.4% | 0%–75% | |
| Aggressive Percentage-M (SD) | 1.4% (3.9) | 0% (0) | 9.1% (3.2) | t(37) = 1.799, p = 0.084 |
| Aggressive-Range | 0%–12.5% | 0%–0% | 0%–12.5% | |
| Autistic Interaction Score-M (SD) | 43.6% (14.0) | 28.7% (10.2) | 38.6% (14.6) | t(37) = 3.397, p = 0.002 ** |
| Autistic Interaction Score-Range | 21%–80% | 15%–53% | 15%–80% |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cordeiro, L.; Braden, M.; Coan, E.; Welnick, N.; Tanda, T.; Tartaglia, N. Evaluating Social Interactions Using the Autism Screening Instrument for Education Planning-3rd Edition (ASIEP-3): Interaction Assessment in Children and Adults with Fragile X Syndrome. Brain Sci. 2020, 10, 248. https://doi.org/10.3390/brainsci10040248
Cordeiro L, Braden M, Coan E, Welnick N, Tanda T, Tartaglia N. Evaluating Social Interactions Using the Autism Screening Instrument for Education Planning-3rd Edition (ASIEP-3): Interaction Assessment in Children and Adults with Fragile X Syndrome. Brain Sciences. 2020; 10(4):248. https://doi.org/10.3390/brainsci10040248
Chicago/Turabian StyleCordeiro, Lisa, Marcia Braden, Elizabeth Coan, Nanastasia Welnick, Tanea Tanda, and Nicole Tartaglia. 2020. "Evaluating Social Interactions Using the Autism Screening Instrument for Education Planning-3rd Edition (ASIEP-3): Interaction Assessment in Children and Adults with Fragile X Syndrome" Brain Sciences 10, no. 4: 248. https://doi.org/10.3390/brainsci10040248
APA StyleCordeiro, L., Braden, M., Coan, E., Welnick, N., Tanda, T., & Tartaglia, N. (2020). Evaluating Social Interactions Using the Autism Screening Instrument for Education Planning-3rd Edition (ASIEP-3): Interaction Assessment in Children and Adults with Fragile X Syndrome. Brain Sciences, 10(4), 248. https://doi.org/10.3390/brainsci10040248




