Spatiotemporal Dynamics of Multiple Memory Systems During Category Learning
Abstract
1. Introduction
2. fMRI Pilot Experiment
2.1. Materials and Methods
2.1.1. Participants
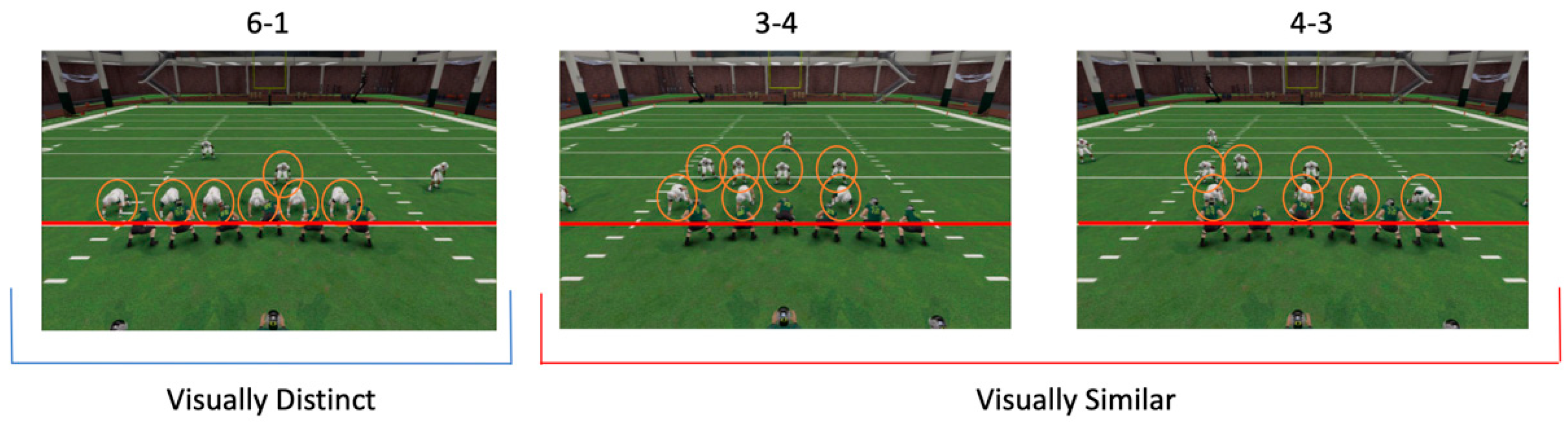
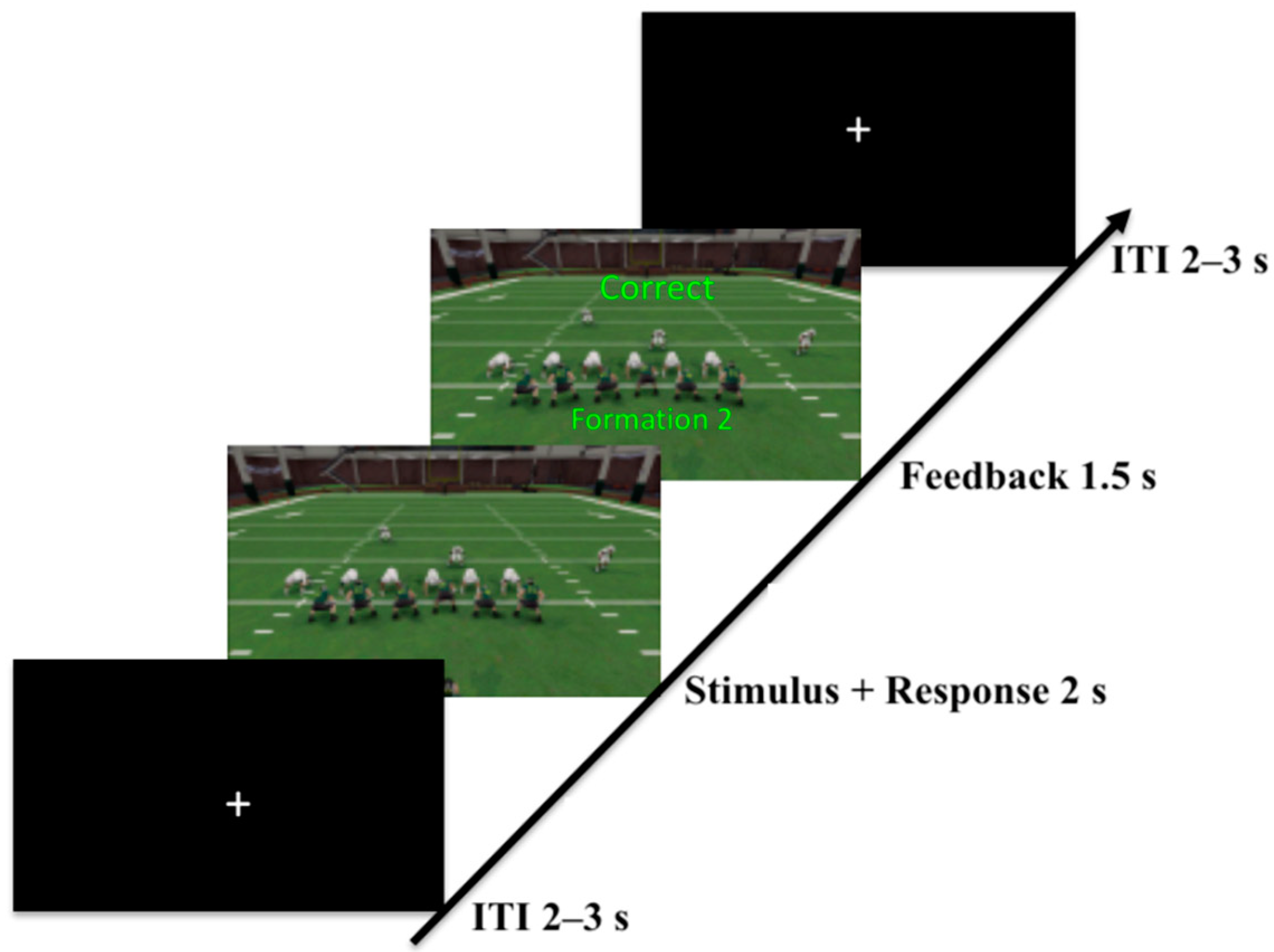
2.1.2. Task
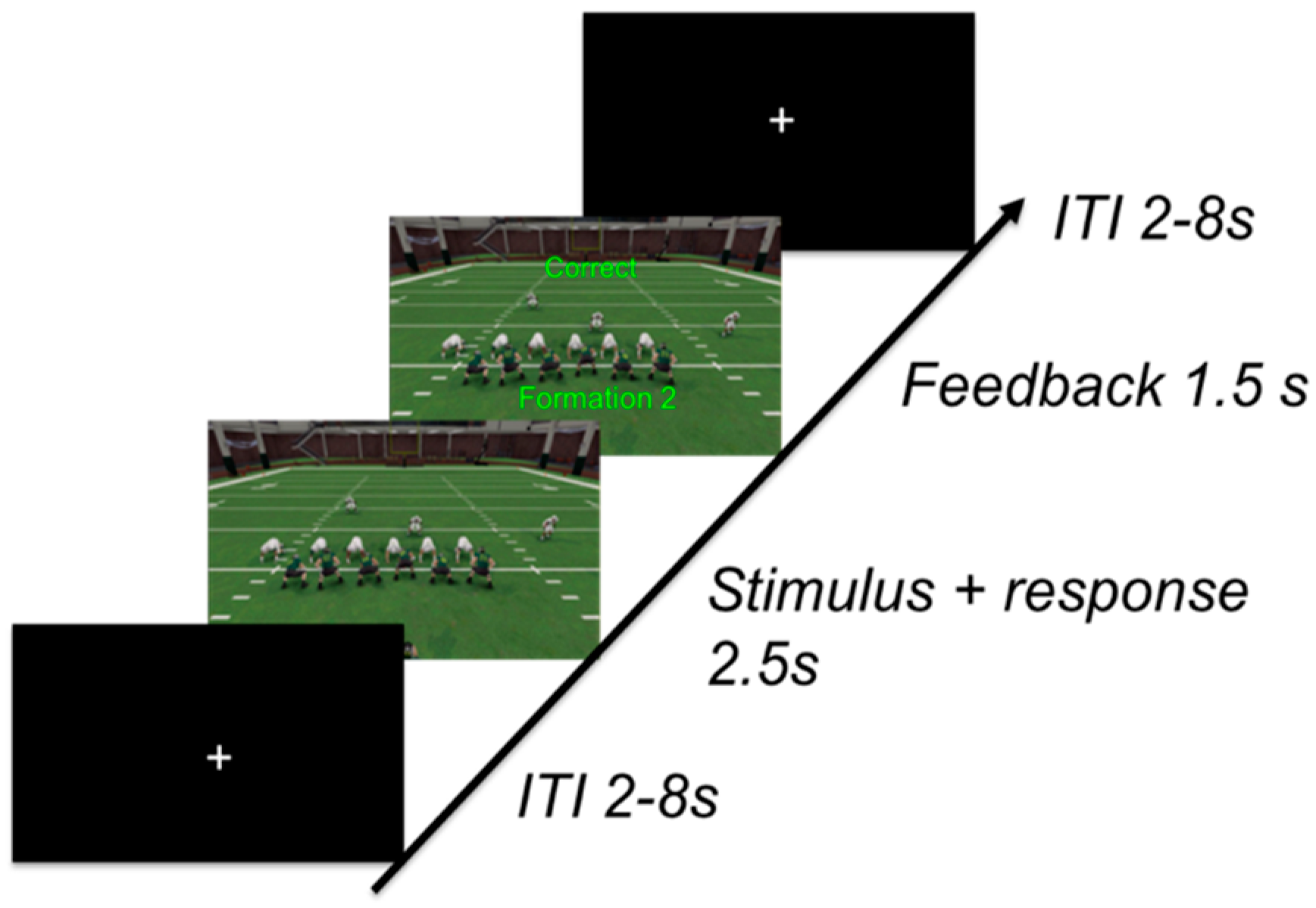
2.1.3. Procedure
2.1.4. fMRI Acquisition and Pre-Processing
2.2. Results
2.2.1. Behavioral Results
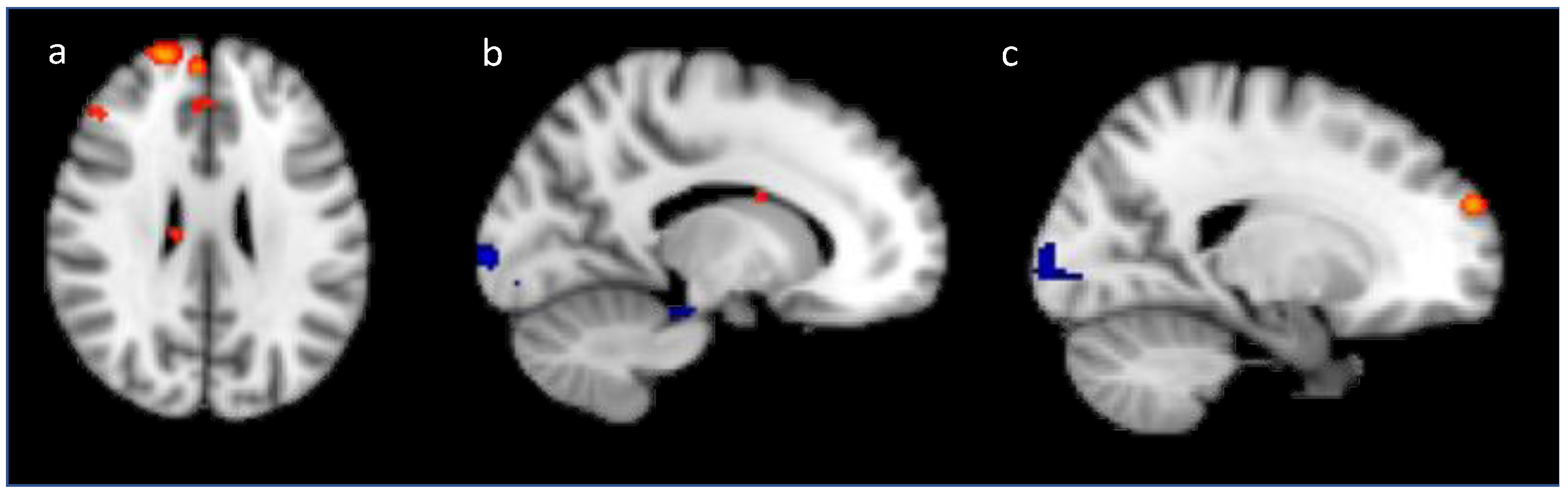
2.2.2. Univariate fMRI Analysis
Training Analysis
Generalization
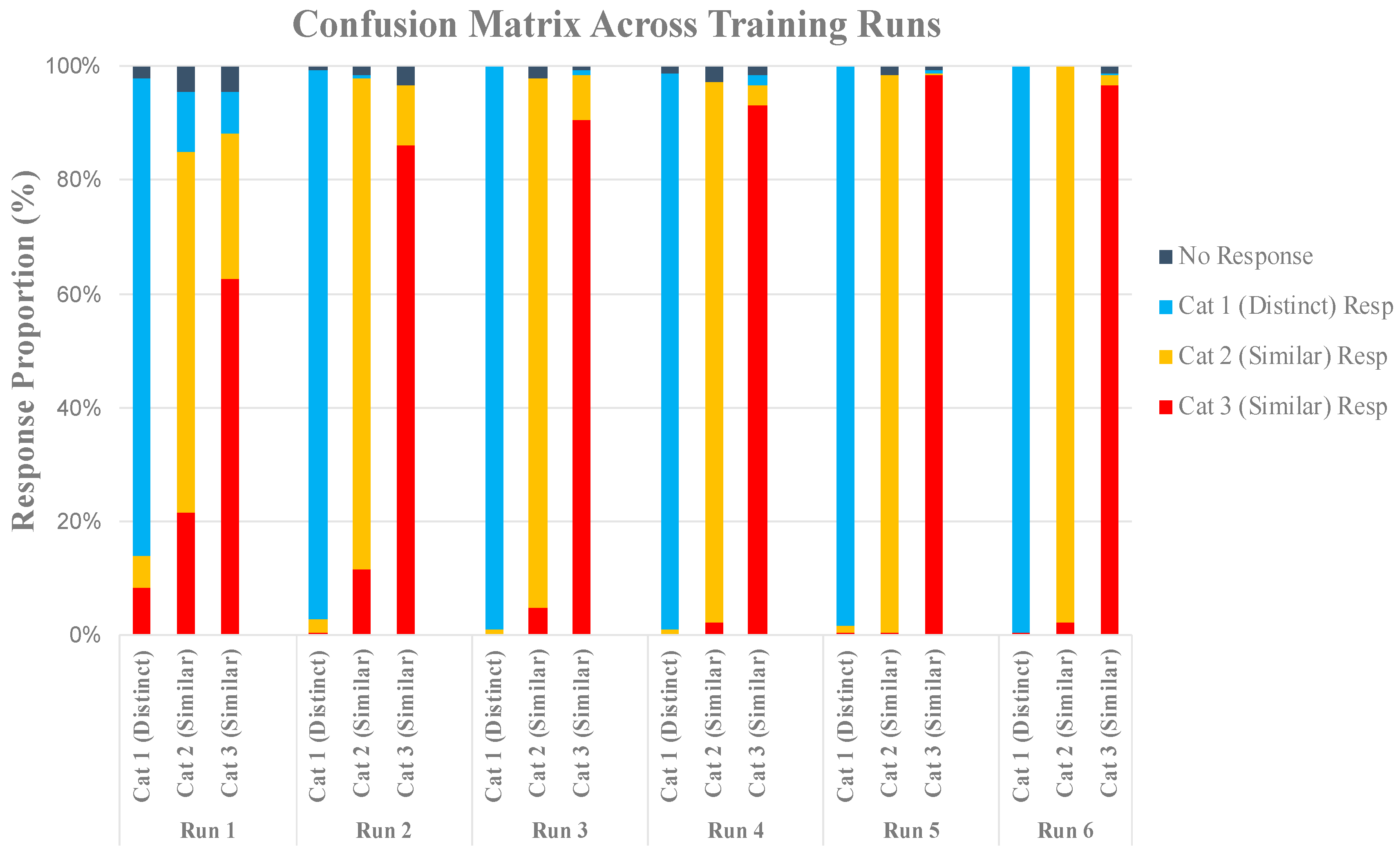
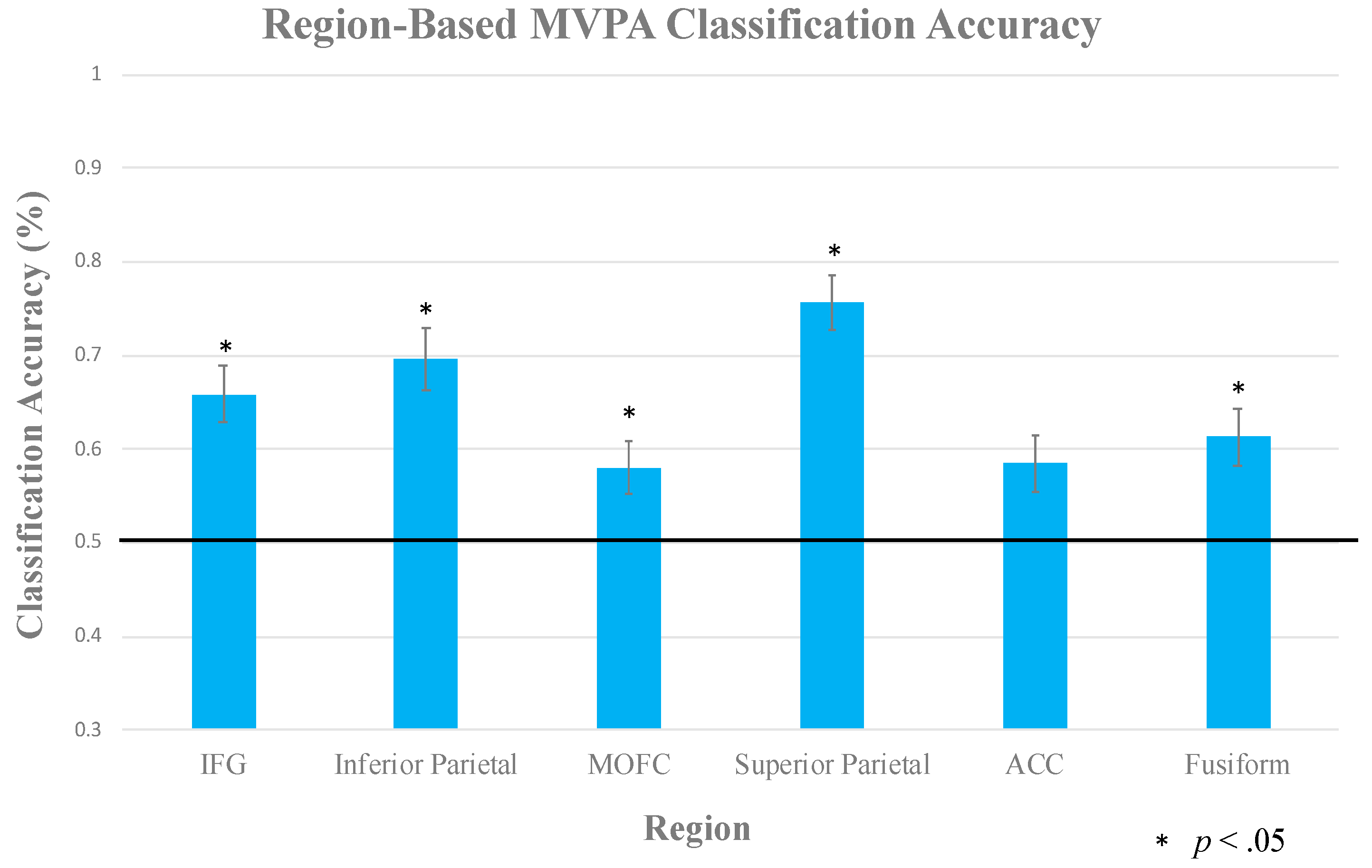
2.2.3. Multi-Voxel Pattern Analysis
3. dEEG Experiment
3.1. Materials and Methods
3.1.1. Participants
3.1.2. Task
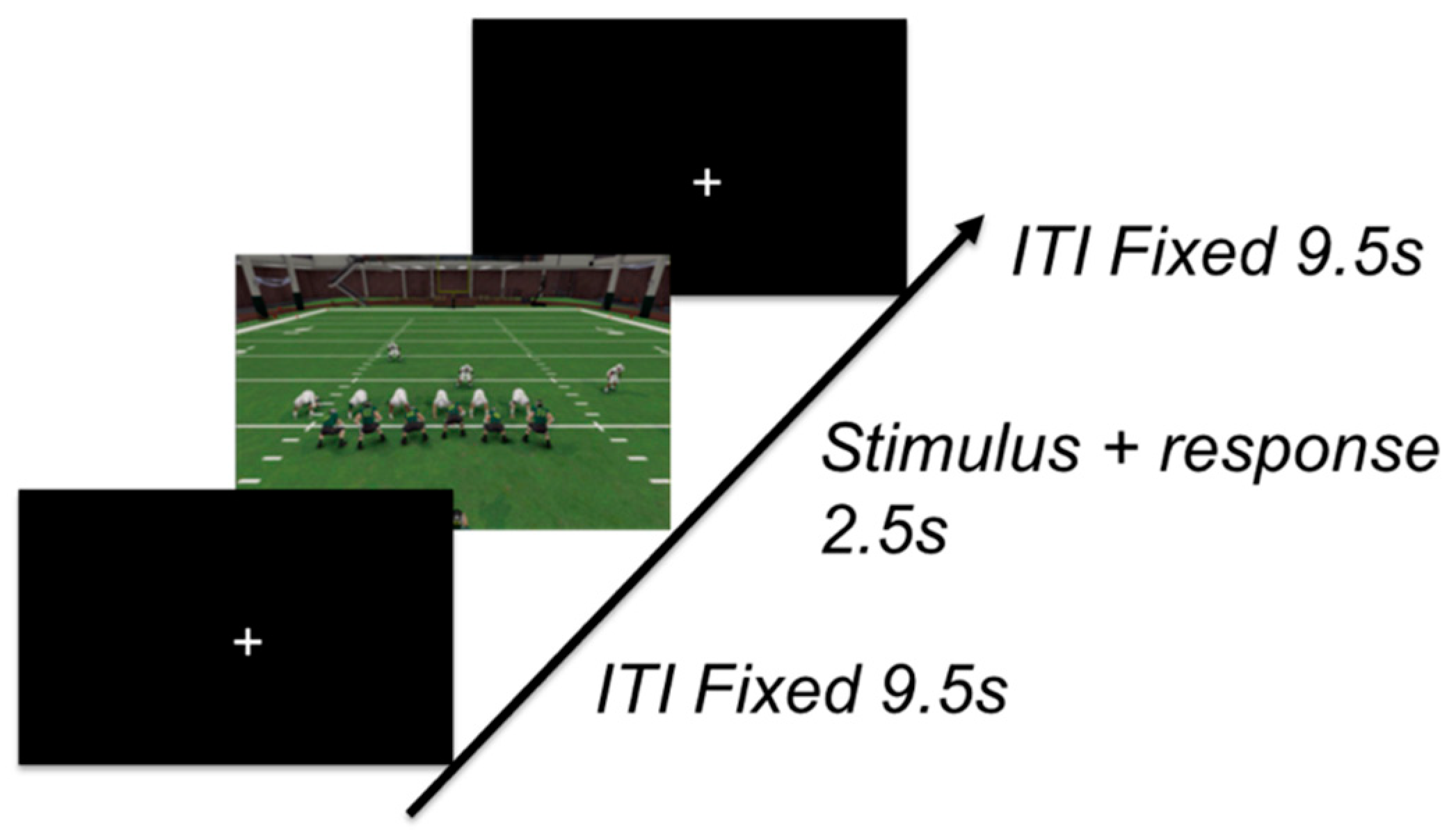
3.1.3. Procedure
3.1.4. Learning Criterion
3.1.5. EEG Recording and Pre-Processing
3.2. Results
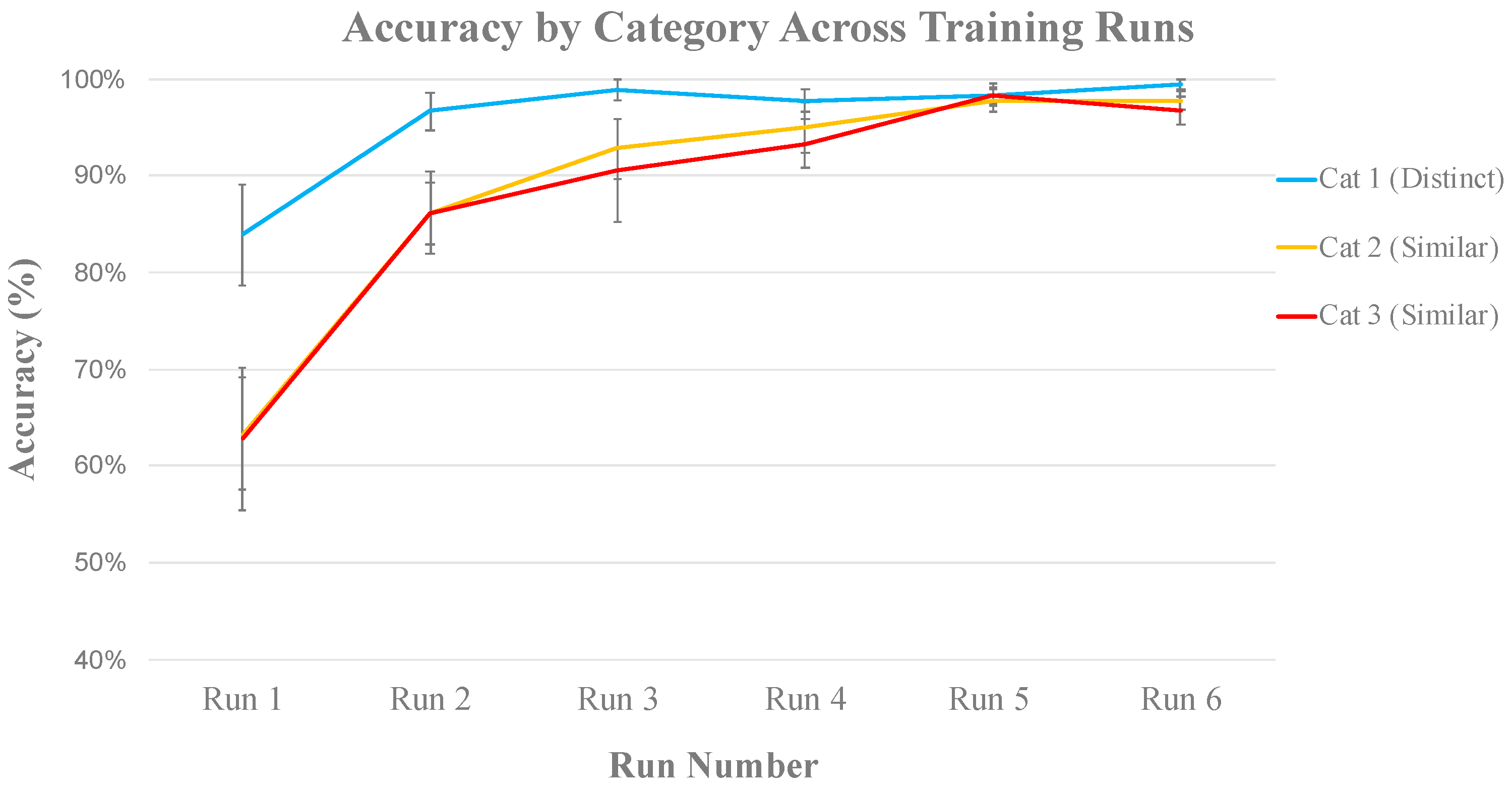
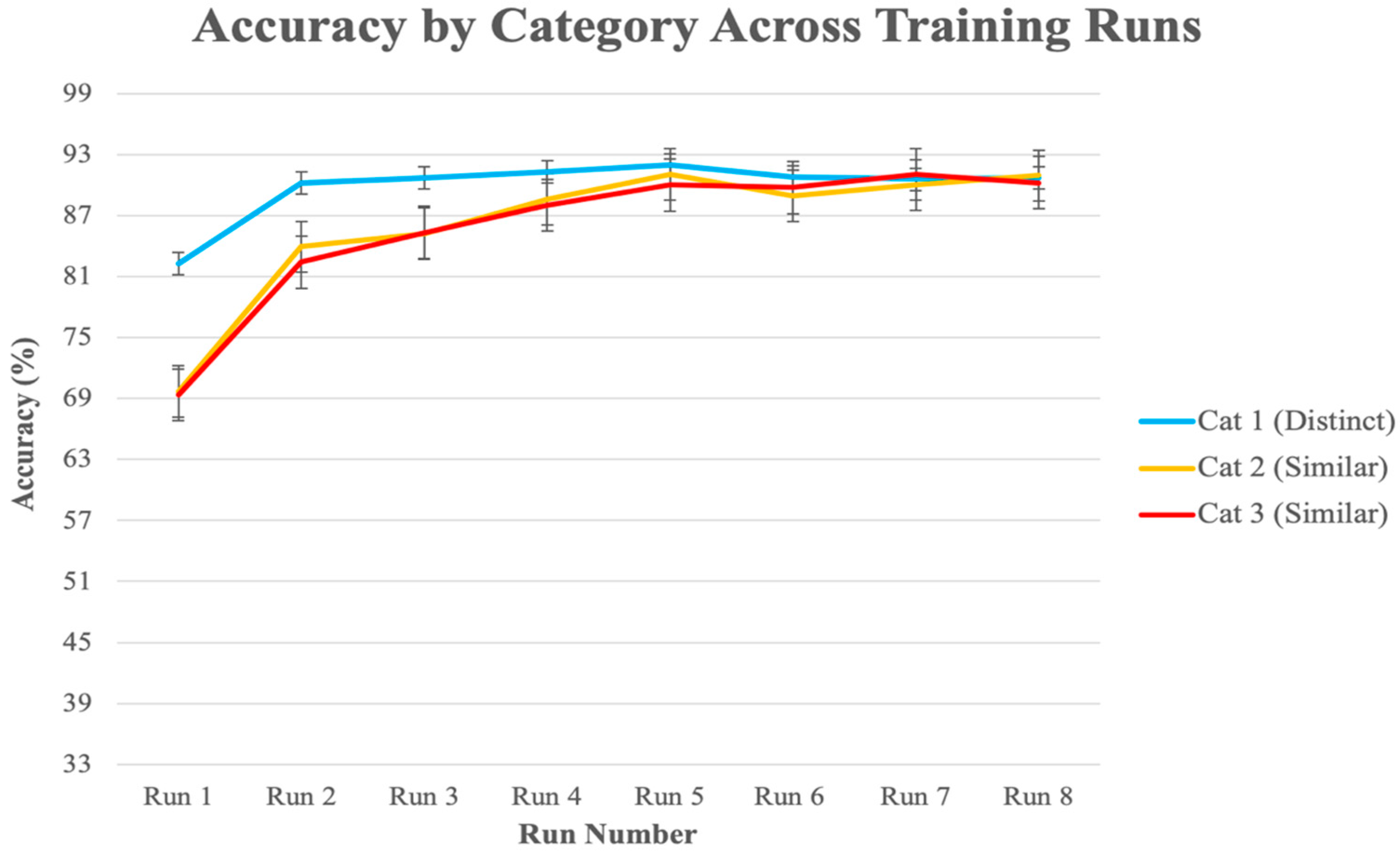
3.2.1. Behavioral Analysis
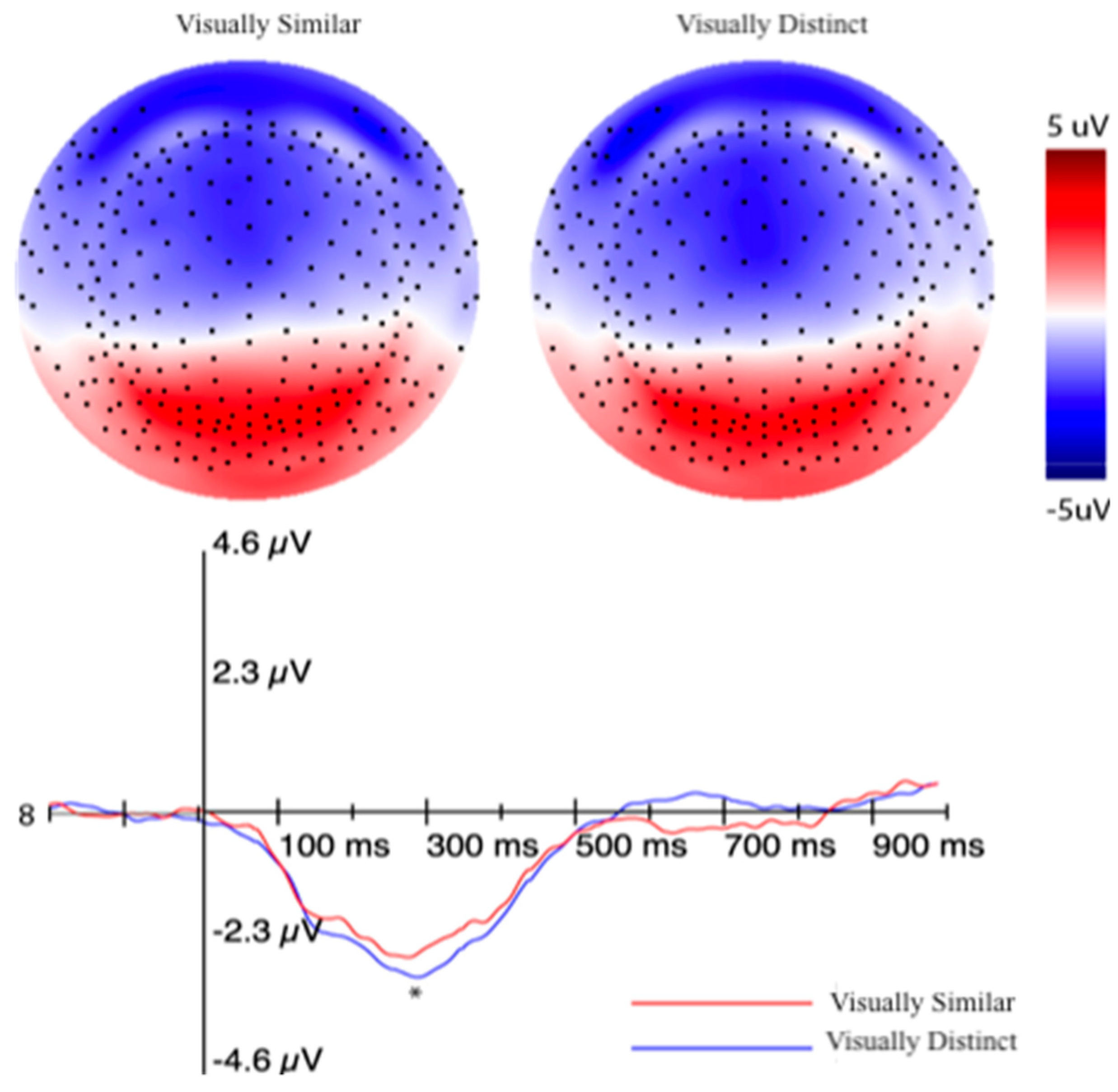
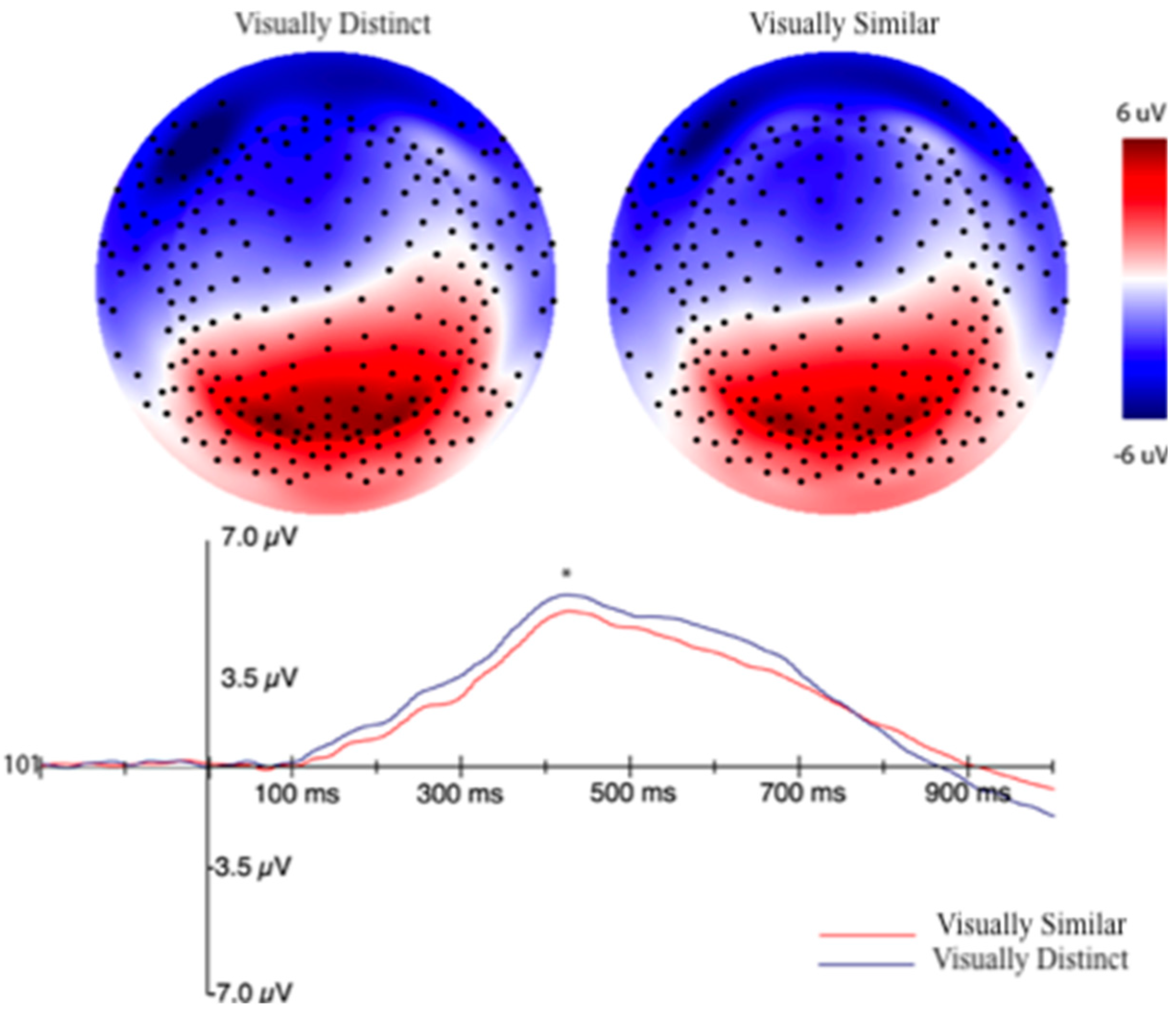
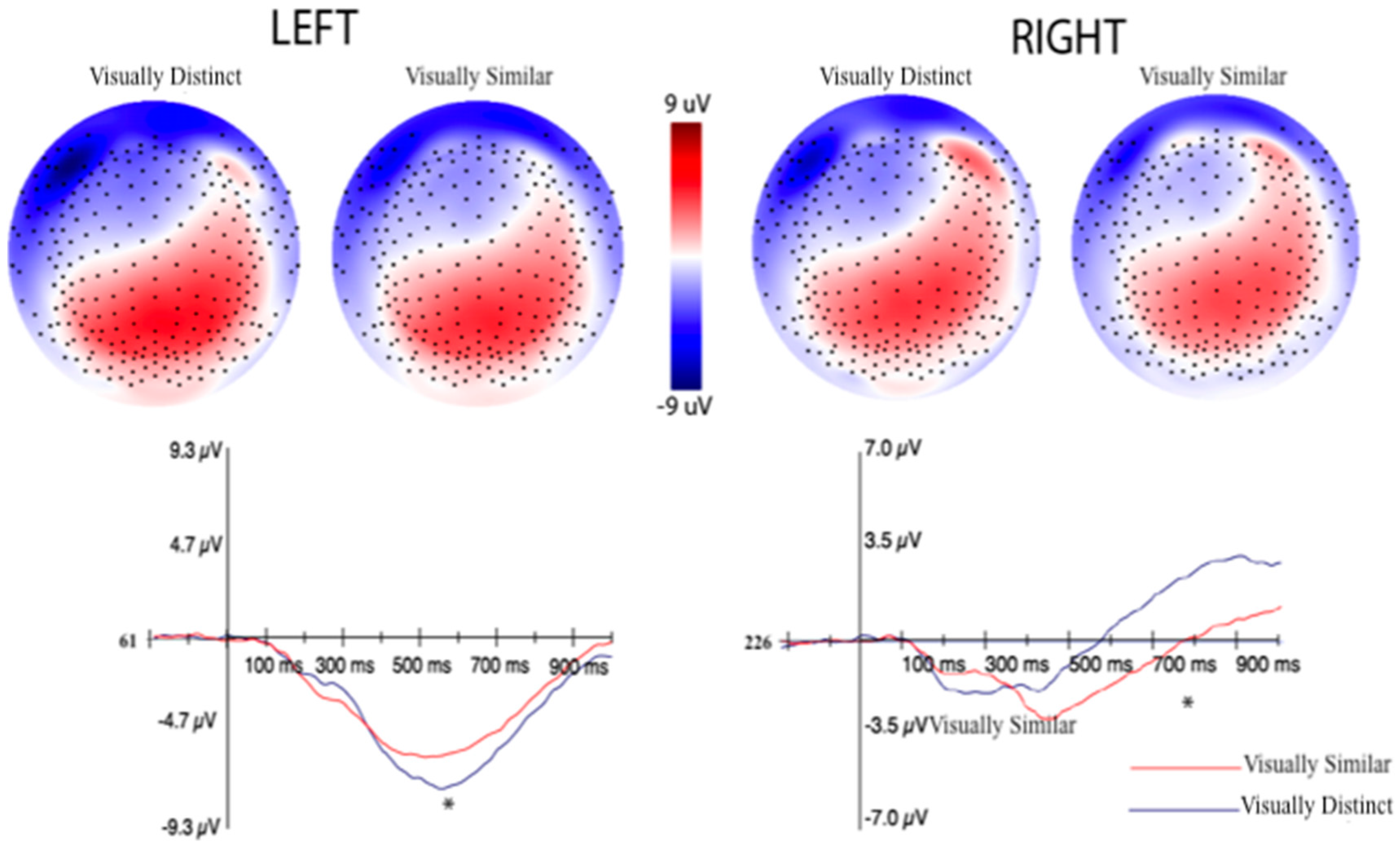
3.2.2. Event-Related Potentials (ERPs) Selection Motivation and Analysis
3.2.3. EEG Machine Learning Analysis
4. Discussion
4.1. fMRI Pilot Experiment
4.1.1. Univariate Analysis
4.1.2. Multi-Voxel Pattern Analysis
4.2. Experiment 2 (dEEG)
4.2.1. ERPs
4.2.2. dEEG Machine Learning
4.3. Category Learning Strategies as a Function of Expertise
4.4. Alternative Interpretations and Limitations
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Bruner, J.; Goodnow, J.J.; Austin, G.A. A Study of Thinking; Science Editions: New York, NY, USA, 1967. [Google Scholar]
- Schneider, W.; Shiffrin, R.M. Controlled and automatic human information processing: I. Detection, search, and attention. Psychol. Rev. 1977, 84, 1–66. [Google Scholar] [CrossRef]
- Zeithamova, D.; Maddox, W.T. Dual task interference in perceptual category learning. Mem. Cogn. 2006, 34, 387–398. [Google Scholar] [CrossRef] [PubMed]
- Knowlton, B.J.; Squire, L.R. The learning of categories: Parallel brain systems for item memory and category knowledge. Science 1993, 262, 1747–1749. [Google Scholar] [CrossRef] [PubMed]
- Gabrieli, J.D.E. Cognitive neuroscience of human memory. Annu. Rev. Psychol. 1998, 49, 87–115. [Google Scholar] [CrossRef]
- Smith, E.E.; Patalano, A.L.; Jonides, J. Alternative strategies of categorization. Cognition 1998, 65, 167–196. [Google Scholar] [CrossRef]
- Casale, M.B.; Ashby, F.G. A role for the perceptual representation memory system in category learning. Percept. Psychophys. 2008, 70, 983–999. [Google Scholar] [CrossRef]
- Chein, J.M.; Schneider, W. Neuroimaging studies of practice-related change: fMRI and meta-analytic evidence of a domain-general control network for learning. Cogn. Brain Res. 2005, 25, 607–623. [Google Scholar] [CrossRef]
- Gabriel, M.; Burhans, L.; Talk, A.; Scalf, P. Cingulate cortex. In Encyclopedia of the Human Brain; Ramachandran, V.S., Ed.; Elsevier Science: Amsterdam, The Netherlands, 2002; pp. 775–791. [Google Scholar]
- Schneider, G.E. Two visual systems. Science 1969, 163, 895–902. [Google Scholar] [CrossRef]
- Ungerleider, L.G.; Mishkin, M. Two cortical visual systems. In Analysis of Visual Behavior; Ingle, D.J., Goodale, M.A., Mansfield, R.J.W., Eds.; MIT Press: Cambridge, MA, USA, 1982; pp. 549–586. [Google Scholar]
- Keele, S.W.; Ivry, R.; Mayr, U.; Hazeltine, E.; Heuer, H. The cognitive and neural architecture of sequence representation. Psychol. Rev. 2003, 110, 316–339. [Google Scholar] [CrossRef]
- Luu, P.; Jiang, Z.; Poulsen, C.; Mattson, C.; Smith, A.; Tucker, D.M. Learning and the development of contexts for action. Front. Hum. Neurosci. 2011, 5, 159. [Google Scholar] [CrossRef]
- Kemler-Nelson, D.G. The effect of intention on what concepts are acquired. J. Verbal Learn. Verbal Behav. 1984, 23, 734–759. [Google Scholar] [CrossRef]
- Ashby, F.G.; Alfonso-Reese, L.A.; Turken, A.U.; Waldron, E.M. A neuropsychological theory of multiple systems in category learning. Psychol. Rev. 1998, 105, 442–481. [Google Scholar] [CrossRef] [PubMed]
- Rosch, E. Cognitive representations of semantic categories. J. Exp. Psychol. Gen. 1975, 104, 192–233. [Google Scholar] [CrossRef]
- Rosch, E. Principles of Categorization; Lawrence Erlbaum Associates: Hillsdale, NJ, USA, 1978. [Google Scholar]
- Aizenstein, H.J.; MacDonald, A.W.; Stenger, V.A.; Nebes, R.D.; Larson, J.K.; Ursu, S.; Carter, C.S. Complementary category learning systems identified using event-related functional mri. J. Cogn. Neurosci. 2000, 12, 977–987. [Google Scholar] [CrossRef] [PubMed]
- Reber, P.J.; Stark, C.E.L.; Squire, L.R. Contrasting cortical activity associated with category memory and recognition memory. Learn. Mem. 1998, 5, 420–428. [Google Scholar] [PubMed]
- Reber, P.J.; Squire, L.R. Intact learning of artificial grammars and intact category learning by patients with Parkinson’s disease. Behav. Neurosci. 1999, 113, 235–242. [Google Scholar] [CrossRef] [PubMed]
- Nosofsky, R.M. Attention, similarity, and the identification-categorization relationship. J. Exp. Psychol. Gen. 1986, 115, 39–57. [Google Scholar] [CrossRef]
- Ashby, F.G.; Ell, S.W. The neurobiology of human category learning. Trends Cogn. Sci. 2001, 5, 204–210. [Google Scholar] [CrossRef]
- Waldron, E.M.; Ashby, F.G. The effects of concurrent task interference on category learning: Evidence from multiple category learning systems. Psychon. Bull. Rev. 2001, 8, 168–176. [Google Scholar] [CrossRef]
- Minda, J.P.; Miles, S.J. The influence of verbal and nonverbal processing on category learning. Psychol. Learn. Motiv. 2010, 52, 117–162. [Google Scholar]
- Dale, A.M. Optimal experimental design for event-related mri. Hum. Brain Mapp. 1999, 8, 109–114. [Google Scholar] [CrossRef]
- Jenkinson, M.; Beckmann, C.F.; Behrens, T.E.; Woolrich, M.W.; Smith, S.M. FSL. NeuroImage 2012, 62, 782–790. [Google Scholar] [CrossRef] [PubMed]
- Worsley, K.J. Statistical analysis of activation images. In Functional Mri: An Introduction to Methods; Jezzard, P., Matthews, P.M., Smith, S.M., Eds.; Oxford University Press: New York, NY, USA, 2001; pp. 251–270. [Google Scholar]
- Nomura, E.M.; Maddox, W.T.; Filoteo, J.V.; Ing, A.D.; Gitelman, D.R.; Parrish, T.B.; Mesulam, M.M.; Reber, P.J. Neural correlates of rule-based and information-integration visual category learning. Cereb. Cortex 2006, 17, 37–43. [Google Scholar] [CrossRef] [PubMed]
- Norman, K.A.; Polyn, S.M.; Deltre, G.J.; Haxby, J.V. Beyond mind-reading: Multi-voxel pattern analysis of fMRI data. Trends Cogn. Sci. 2006, 10, 424–430. [Google Scholar] [CrossRef]
- Dale, A.M.; Fischl, B.; Sereno, M.I. Cortical surface-based analysis. I. Segmentation and surface reconstruction. NeuroImage 1999, 9, 179–194. [Google Scholar] [CrossRef]
- Fischl, B.; Salat, D.H.; Busa, E.; Albert, M.; Dieterich, M.; Haselgrove, C.; van der Kouwe, A.; Killiany, R.; Kennedy, D.; Klaveness, S.; et al. Whole brain segmentation: Automated labeling of neuroanatomical structures in the human brain. Neuron 2002, 33, 341–355. [Google Scholar] [CrossRef]
- Woolgar, A.; Thompson, R.; Bor, D.; Duncan, J. Multi-voxel coding of stimuli, rules, and responses in human frontoparietal cortex. NeuroImage 2011, 56, 744–752. [Google Scholar] [CrossRef]
- Reverberi, C.; Görgen, K.; Haynes, J.D. Compositionality of rule representations in human prefrontal cortex. Cereb. Cortex 2012, 22, 1237–1246. [Google Scholar] [CrossRef]
- Nelissen, N.; Strokes, M.; Nobre, A.C.; Rushworth, M.F. Frontal and parietal cortical interactions with distributed visual representations during selection attention and action selection. J. Neurosci. 2013, 33, 16443–16458. [Google Scholar] [CrossRef]
- Rissman, J.; Gazzaley, A.; D’Esposito, M. Measuring functional connectivity during distinct stages of a cognitive task. NeuroImage 2004, 23, 752–763. [Google Scholar] [CrossRef]
- Avants, B.B.; Tustison, N.J.; Song, G.; Cook, P.A.; Klein, A.; Gee, J.C. A reproducible evaluation of ANTs similarity metric performance in brain image registration. NeuroImage 2011, 54, 2033–2044. [Google Scholar] [CrossRef] [PubMed]
- Mur, M.; Bandettini, P.A.; Kriegeskorte, N. Revealing representational content with pattern-information fMRI—An introductory guide. Soc. Cogn. Affect. Neurosci. 2009, 4, 101–109. [Google Scholar] [CrossRef] [PubMed]
- Esterman, M.; Tamber-Rosenau, B.J.; Chiu, Y.; Yantis, S. Avoiding non-independence in fMRI data analysis: Leave one subject out. NeuroImage 2010, 50, 572–576. [Google Scholar] [CrossRef] [PubMed]
- Luu, P.; Tucker, D.M.; Stripling, R. Neural mechanisms for learning action in context. Brain Res. 2007, 1179, 89–105. [Google Scholar] [CrossRef]
- Morgan, K.K.; Luu, P.; Tucker, D.M. Changes in p3b latency and amplitude reflect expertise acquisition in a football visuomotor learning task. PLoS ONE 2016, 11, e0154021. [Google Scholar] [CrossRef] [PubMed]
- Bush, G.; Vogt, B.A.; Holmes, J.; Dale, A.M.; Greve, D.; Jenike, M.A.; Rosen, B.R. Dorsal anterior cingulate cortex: A role in reward-based decision making. Proc. Natl. Acad. Sci. USA 2002, 99, 523–528. [Google Scholar] [CrossRef] [PubMed]
- Halgren, E.; Baudena, P.; Heit, G.; Clarke, J.M.; Marinkovic, K.; Chauvel, P. Spatio-temporal stages in face and word processing. 2. Depth-recorded potentials in the human frontal and Rolandic cortices. J. Physiol. 1994, 88, 51–80. [Google Scholar] [CrossRef]
- Halgren, E.; Baudena, P.; Clarke, J.M.; Heit, G.; Liegeois, C.; Chauvel, P.; Musolino, A. Intracerebral potential to rare target and distractor auditory and visual stimuli. I. Superior temporal plane and parietal lobe. Electroencephalogr. Clin. Neurophysiol. 1995, 94, 191–220. [Google Scholar] [CrossRef]
- Halgren, E.; Baudena, P.; Clarke, J.M.; Heit, G.; Marinkovic, K.; Devaux, B.; Vignal, J.P.; Biraben, A. Intracerebral potential to rare target and distractor auditory and visual stimuli. II. Medial, lateral, and posterior temporal lobe. Electroencephalogr. Clin. Neurophysiol. 1995, 94, 229–250. [Google Scholar] [CrossRef]
- Baudena, P.; Halgren, E.; Heit, G.; Clarke, J.M. Intracerebral potentials to rare target and distractor auditory and visual stimuli. III. Frontal cortex. Electroencephalogr. Clin. Neurophysiol. 1995, 94, 251–264. [Google Scholar] [CrossRef]
- Smith, M.E.; Halgren, E.; Sokolik, M.; Baudena, P.; Musolino, A.; Liegeois-Chauvel, C.; Chauvel, P. The intracranial topography of the p3 event-related potential elicited during auditory oddball. Electroencephalogr. Clin. Neurophysiol. 1990, 76, 235–248. [Google Scholar] [CrossRef]
- Brankack, J.; Seidenbecher, T.; Muller-Gartner, H.W. Task-relevant late positive component in rats: Is it related to hippocampal theta rhythm? Hippocampus 1996, 6, 475–482. [Google Scholar] [CrossRef]
- Shin, J. The interrelationship between movement and cognition: Theta rhythm and the p330 event-related potential. Hippocampus 2011, 21, 744–752. [Google Scholar] [CrossRef] [PubMed]
- Kahana, M.J.; Seelig, D.; Madsen, J.R. Theta returns. Curr. Opin. Neurobiol. 2001, 11, 739–744. [Google Scholar] [CrossRef]
- Miles, S.J.; Matsuki, K.; Minda, J.P. Continuous executive function disruption interferes with application of an information integration categorization strategy. Atten. Percept. Psychophys. 2014, 76, 1318–1334. [Google Scholar] [CrossRef]
- Lombardi, W.J.; Andreason, P.J.; Sirocco, K.Y.; Rio, D.E.; Gross, R.E.; Umhau, J.C.; Hommer, D.W. Wisconsin card sorting test performance following head-injury: Dorsolateral fronto-striatal circuit activity predicts perseveration. J. Exp. Neuropsychol. 1999, 21, 2–16. [Google Scholar] [CrossRef]
- Rao, S.M.; Bobholz, J.A.; Hammeke, T.A.; Tosen, A.C.; Woodley, S.J.; Cunningham, J.M.; Cox, R.W.; Stein, E.A.; Binder, J.R. Functional mri evidence for subcortical participation in conceptual reasoning skills. Neuroreport 1997, 8, 1987–1993. [Google Scholar] [CrossRef]
- Rogers, R.D.; Andrews, T.C.; Grasby, P.M.; Brooks, D.J.; Robbins, T.W. Contrasting cortical and subcortical activations produced by attentional-set shifting and reversal learning in humans. J. Cogn. Neurosci. 2000, 12, 142–162. [Google Scholar] [CrossRef]
- Ashby, F.G.; Paul, E.J.; Maddox, W.T. COVIS. In Formal Approaches in Categorization; Pothos, E.M., Wills, A.J., Eds.; Cambridge University Press: New York, NY, USA, 2011; pp. 65–87. [Google Scholar]
- Eichenbaum, H. A cortical-hippocampal system for declarative memory. Nat. Rev. Neurosci. 2000, 1, 41–50. [Google Scholar] [CrossRef]
- Haynes, J.D.; Rees, G. Decoding mental states from brain activity in humans. Nat. Rev. Neurosci. 2006, 7, 523–534. [Google Scholar] [CrossRef]
- Desimone, R.; Duncan, J. Neural mechanisms of selective visual attention. Annu. Rev. Neurosci. 1995, 18, 193–222. [Google Scholar] [CrossRef] [PubMed]
- Miller, E.K.; Cohen, J.D. An integrative theory of prefrontal cortex function. Annu. Rev. Neurosci. 2001, 24, 167–202. [Google Scholar] [CrossRef] [PubMed]
- Asaad, W.F.; Rainer, G.; Miller, E.K. Neural activity in the primate prefrontal cortex during associative learning. Neuron 1998, 21, 1399–1407. [Google Scholar] [CrossRef]
- Freedman, D.J.; Assad, J.A. Experience-dependent representation of visual categories in parietal cortex. Nature 2006, 443, 85–88. [Google Scholar] [CrossRef] [PubMed]
- White, I.M.; Wise, S.P. Rule-dependent neuronal activity in the prefrontal cortex. Exp. Brain Res. 1999, 126, 315–335. [Google Scholar] [CrossRef] [PubMed]
- Bode, S.; Haynes, J.D. Decoding sequential stages of task preparation in the human brain. NeuroImage 2009, 45, 606–613. [Google Scholar] [CrossRef] [PubMed]
- Haynes, J.D.; Sakai, K.; Rees, G.; Gilbert, S.; Frith, C.; Passingham, R.E. Reading hidden intentions in the human brain. Curr. Biol. 2007, 17, 323–328. [Google Scholar] [CrossRef]
- Toni, I.; Rammani, N.; Josephs, O.; Ashburner, J.; Passingham, R.E. Learning arbitrary visuomotor associations: Temporal dynamics of brain activity. NeuroImage 2001, 14, 1048–1057. [Google Scholar] [CrossRef]
- Groll, M.J.; de Lange, F.P.; Verstraten, F.A.J.; Passingham, R.E.; Toni, I. Cerebral changes during performance of overlearned arbitrary visuomotor associations. J. Neurosci. 2006, 26, 117–125. [Google Scholar] [CrossRef]
- Donchin, E.; Coles, M.G.H. Is the p300 component a manifestation of context updating? Behav. Brain Sci. 1988, 11, 357–374. [Google Scholar] [CrossRef]
- Palmeri, T.J. Exemplar similarity and the development of automaticity. J. Exp. Psychol. Learn. Mem. Cogn. 1997, 23, 324–354. [Google Scholar] [CrossRef] [PubMed]
- Nosofsky, R.M.; Palmeri, T.J.; McKinley, S.C. Rule-plus-exception model of classification learning. Psychol. Rev. 1994, 101, 53–79. [Google Scholar] [CrossRef] [PubMed]
- Palmeri, T.J.; Nosofsky, R.M. Recognition memory for exceptions to the category rule. J. Exp. Psychol. Learn. Mem. Cogn. 1995, 21, 548–568. [Google Scholar] [CrossRef] [PubMed]
- Zeithamova, D.; Maddox, W.T.; Schnyer, D.M. Dissociable prototype learning systems: Evidence from brain imaging and behavior. J. Neurosci. 2008, 28, 13194–13201. [Google Scholar] [CrossRef] [PubMed]




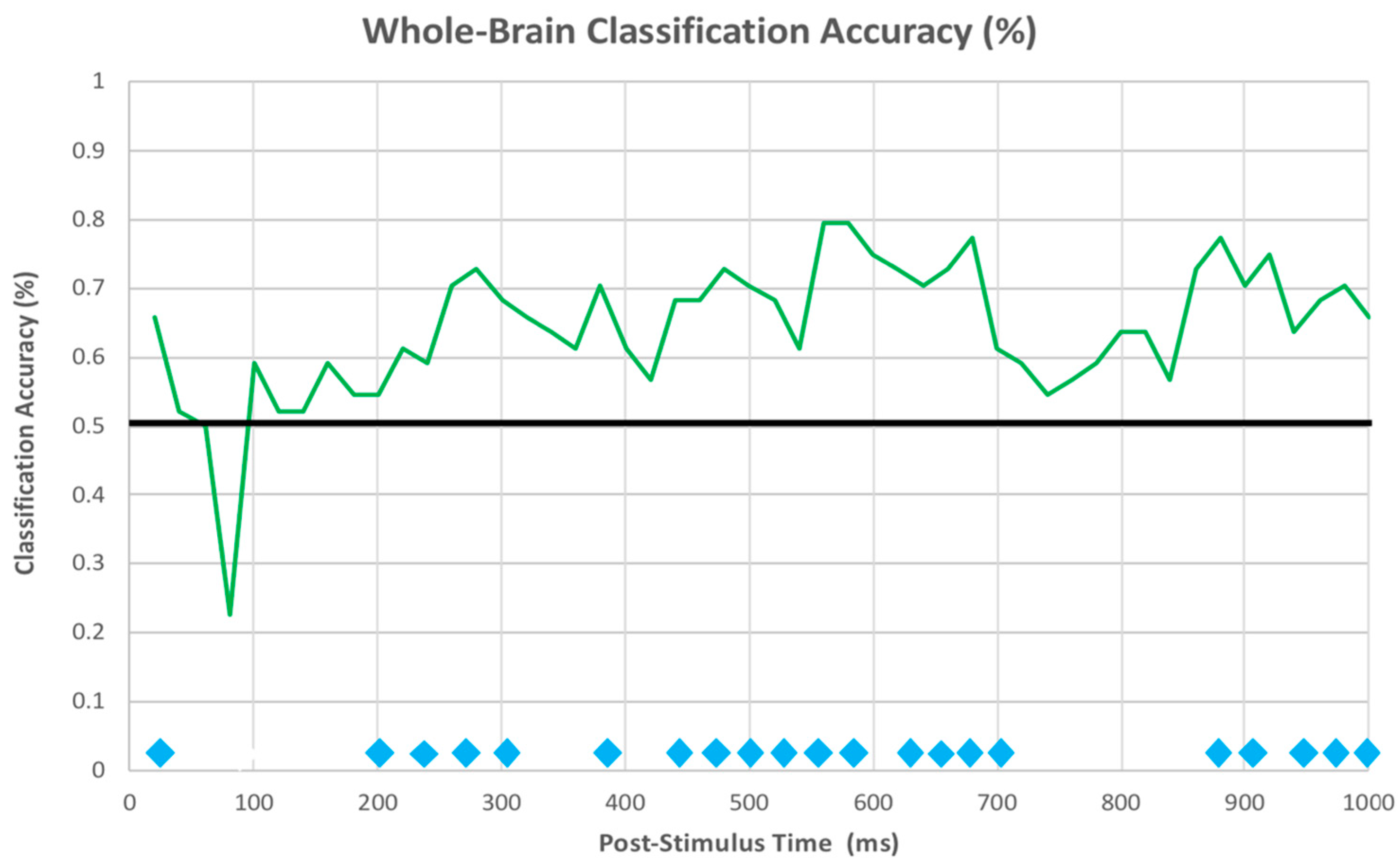
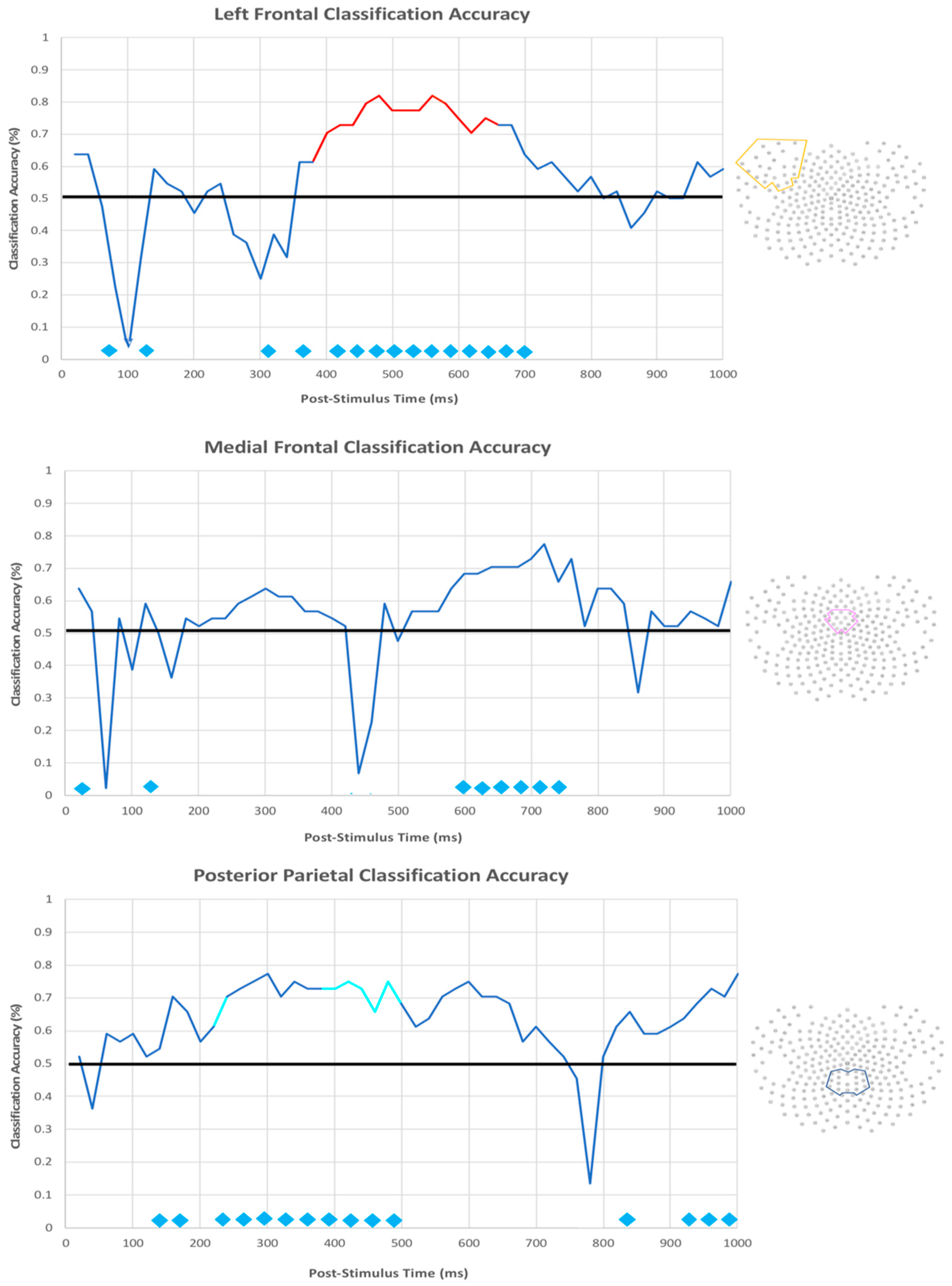
| Location | Cluster Size | Z-Value | X | Y | Z |
|---|---|---|---|---|---|
| L Sup. Fr. Gyrus | 58 | 2.79 | −54 | 44 | −10 |
| L. IFG | 50 | 2.95 | −50 | 30 | 14 |
| L. Sup. Fr. Gyrus | 38 | 2.72 | −12 | 40 | 56 |
| L. Sup. Fr. Gyrus | 34 | 2.47 | −16 | 56 | 38 |
| R. Hippocampus | 26 | 2.88 | 22 | −34 | −10 |
| L. Sup. Temp. Gyrus | 25 | 2.67 | −50 | 10 | −16 |
| R. Fusiform Gyrus | 25 | 3.04 | 40 | −44 | −20 |
| R. Lateral Occipital Cortex | 24 | 2.72 | −10 | −12 | 56 |
| L. Suppl. Motor Cortex | 22 | 2.42 | 58 | −64 | 24 |
| Brain Stem | 22 | 2.63 | 6 | −22 | −28 |
| R. Mid. Temp. Gyrus | 20 | 2.56 | 40 | −58 | 2 |
| Location | Cluster Size | Z-Value | X | Y | Z |
|---|---|---|---|---|---|
| R. Lateral Occipital Cortex | 519 | 3.16 | 6 | −74 | 36 |
| R. Lateral Occipital Cortex | 154 | 2.87 | 34 | −62 | 62 |
| L. Fusiform Gyrus | 106 | 3.17 | −20 | −66 | −18 |
| L. Lateral Occipital Cortex | 98 | 2.83 | −36 | −56 | 38 |
| R. IFG | 89 | 3.25 | 20 | 56 | −6 |
| L. Post. Cingulate Gyrus | 70 | 2.88 | −8 | −40 | 48 |
| R. Lateral Occipital Cortex | 55 | 2.47 | 20 | −88 | 38 |
| R. Fusiform Gyrus | 52 | 2.62 | 20 | −54 | −16 |
| L. Middle Frontal Gyrus | 49 | 2.77 | −38 | 45 | 18 |
| R. Occipital Pole | 41 | 2.34 | 20 | −104 | −10 |
| Brain Stem | 40 | 3.05 | 22 | −32 | −42 |
| Location | Cluster Size | Z-Value | X | Y | Z |
|---|---|---|---|---|---|
| L. Caudate Nucleus | 290 | 3.5 | −8 | −10 | 24 |
| Cerebellum | 129 | 3.22 | 16 | −72 | −28 |
| Cerebellum | 125 | 3.43 | 32 | −80 | −22 |
| Cerebellum | 90 | 3.18 | 4 | −50 | −10 |
| L. Sup. Frontal Gyrus | 88 | 3.21 | −28 | 6 | 64 |
| L. Lateral Occipital Cortex | 73 | 3.17 | −26 | −78 | 50 |
| R. Lateral Occipital Cortex | 71 | 3.08 | 40 | −74 | 42 |
| L. Inf. Frontal Gyrus | 67 | 3.22 | −42 | 22 | 4 |
| Cerebellum | 63 | 3.3 | −26 | −90 | −26 |
| L. Sup. Frontal Gyrus | 59 | 3.17 | −42 | 46 | 20 |
| Brain Stem | 58 | 2.92 | 14 | −16 | −38 |
| Location | Cluster Size | Z-Value | X | Y | Z |
|---|---|---|---|---|---|
| R. Lateral Occipital Cortex | 1922 | 4 | 18 | −100 | 6 |
| R. Fusiform Gyrus | 335 | 3.41 | 12 | −72 | −2 |
| R. Inf. Frontal Gyrus | 213 | 3.11 | 62 | 6 | 12 |
| Postcentral Gyrus | 144 | 3.35 | −40 | −26 | 54 |
| L. Sup. Temporal Gyrus | 143 | 3.49 | 68 | −24 | 28 |
| Cerebellum | 113 | 3.09 | −20 | −72 | −52 |
| L. Fusiform Gyrus | 100 | 2.88 | 38 | −54 | −24 |
| R. Mid. Temporal Gyrus | 99 | 3.13 | 66 | −40 | 2 |
| R. Mid. Frontal Gyrus | 82 | 3.25 | 32 | 18 | 30 |
| R. Mid Temporal Gyrus | 79 | 3.37 | 54 | −6 | −28 |
| R. Angular Gyrus | 70 | 3.7 | 56 | −46 | 30 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
K. Morgan, K.; Zeithamova, D.; Luu, P.; Tucker, D. Spatiotemporal Dynamics of Multiple Memory Systems During Category Learning. Brain Sci. 2020, 10, 224. https://doi.org/10.3390/brainsci10040224
K. Morgan K, Zeithamova D, Luu P, Tucker D. Spatiotemporal Dynamics of Multiple Memory Systems During Category Learning. Brain Sciences. 2020; 10(4):224. https://doi.org/10.3390/brainsci10040224
Chicago/Turabian StyleK. Morgan, Kyle, Dagmar Zeithamova, Phan Luu, and Don Tucker. 2020. "Spatiotemporal Dynamics of Multiple Memory Systems During Category Learning" Brain Sciences 10, no. 4: 224. https://doi.org/10.3390/brainsci10040224
APA StyleK. Morgan, K., Zeithamova, D., Luu, P., & Tucker, D. (2020). Spatiotemporal Dynamics of Multiple Memory Systems During Category Learning. Brain Sciences, 10(4), 224. https://doi.org/10.3390/brainsci10040224




