Is the Regular Intake of Anticoagulative Agents an Independent Risk Factor for the Severity of Traumatic Brain Injuries in Geriatric Patients? A Retrospective Analysis of 10,559 Patients from the TraumaRegister DGU®
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Population
2.1.1. Inclusion Criteria
2.1.2. Exclusion Criteria
2.2. Definitions
- (1)
- No anticoagulant medication (NM);
- (2)
- Anti-platelet drugs (APD);
- (3)
- Vitamin K antagonists (VKA);
- (4)
- Direct oral anticoagulants (DOACs);
- (5)
- Heparinoids.
2.3. Statistical Analysis
3. Results
4. Discussion
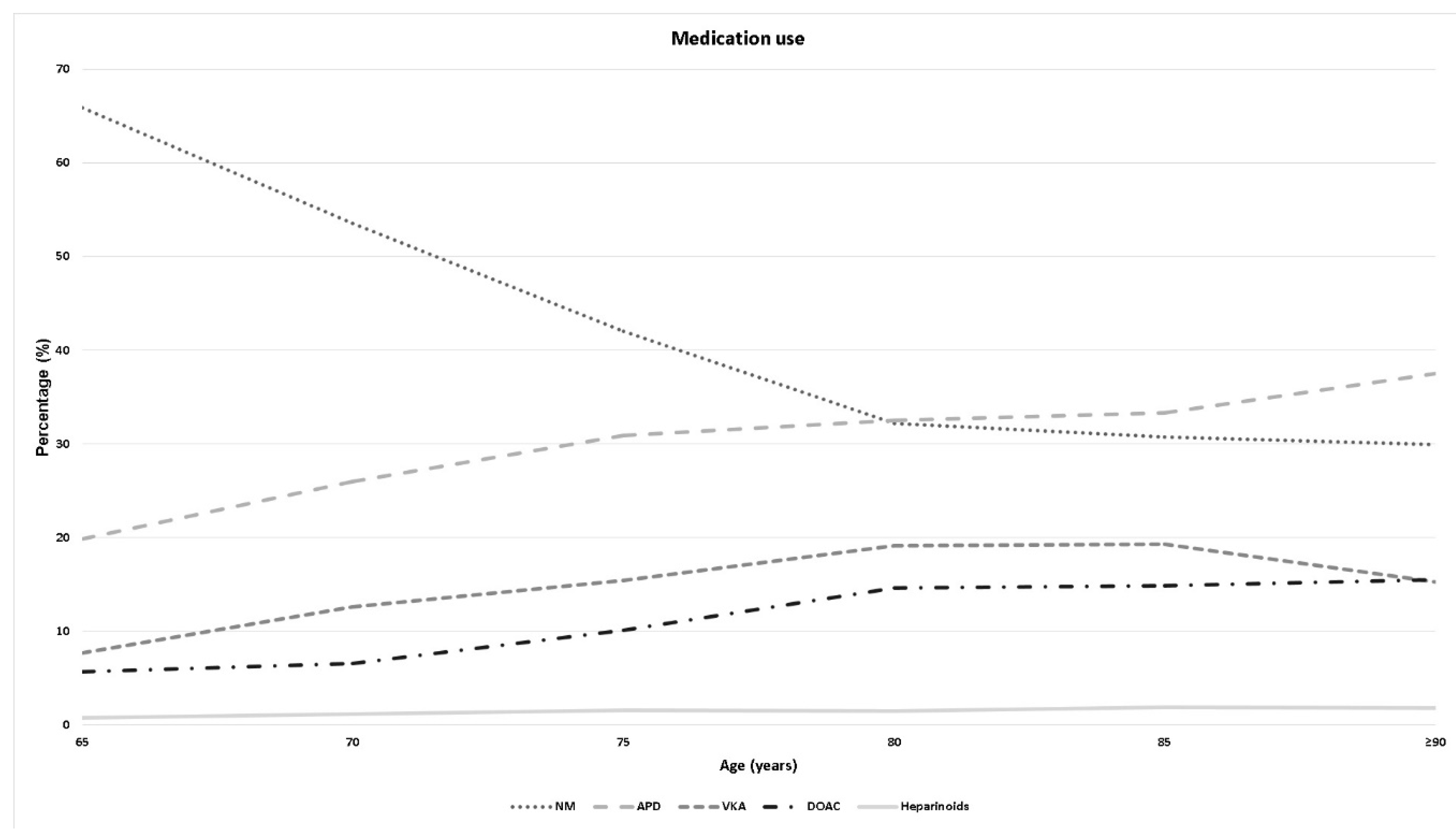
- The rate of patients taking anticoagulative medication increased in the geriatric population with increasing age.
- The use of anticoagulant medication increased the severity of TBI in geriatric patients as an independent risk factor.
- The type of anticoagulant medication was associated with the severity of TBI.
- Pre-injury intake of anticoagulant medication increased the mortality rate.
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Dewan, M.C.; Rattani, A.; Gupta, S.; Baticulon, R.E.; Hung, Y.-C.; Punchak, M.; Agrawal, A.; Adeleye, A.O.; Shrime, M.G.; Rubiano, A.M.; et al. Estimating the global incidence of traumatic brain injury. J. Neurosurg. 2018, 1–18. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Czorlich, P.; Mader, M.M.-D.; Emami, P.; Westphal, M.; Lefering, R.; Hoffmann, M. Operative versus non-operative treatment of traumatic brain injuries in patients 80 years of age or older. Neurosurg. Rev. 2019. [Google Scholar] [CrossRef] [PubMed]
- Fu, T.S.; Jing, R.; McFaull, S.R.; Cusimano, M.D. Recent trends in hospitalization and in-hospital mortality associated with traumatic brain injury in Canada: A nationwide, population-based study. J. Trauma Acute Care Surg. 2015, 79, 449–454. [Google Scholar] [CrossRef] [PubMed]
- Spering, C.; Lefering, R.; Bouillon, B.; Lehmann, W.; von Eckardstein, K.; Dresing, K.; Sehmisch, S. It is time for a change in the management of elderly severely injured patients! An analysis of 126,015 patients from the TraumaRegister DGU®. Eur. J. Trauma Emerg. Surg. 2019. [Google Scholar] [CrossRef] [PubMed]
- Giner, J.; Mesa Galán, L.; Yus Teruel, S.; Guallar Espallargas, M.C.; Pérez López, C.; Isla Guerrero, A.; Roda Frade, J. Traumatic brain injury in the new millennium: A new population and new management. Neurologia 2019. [Google Scholar] [CrossRef]
- Filippi, A.; Sessa, E.; Trifirò, G.; Mazzaglia, G.; Pecchioli, S.; Caputi, A.P.; Cricelli, C. Oral anticoagulant therapy in Italy: Prescribing prevalence and clinical reasons. Pharmacol. Res. 2004, 50, 601–603. [Google Scholar] [CrossRef]
- Gardner, R.C.; Dams-O’Connor, K.; Morrissey, M.R.; Manley, G.T. Geriatric Traumatic Brain Injury: Epidemiology, Outcomes, Knowledge Gaps, and Future Directions. J. Neurotrauma 2018, 35, 889–906. [Google Scholar] [CrossRef]
- Gennarelli, T.A.; Wodzin, E. AIS 2005: A contemporary injury scale. Injury 2006, 37, 1083–1091. [Google Scholar] [CrossRef]
- Lefering, R.; Paffrath, T.; Linker, R.; Bouillon, B.; Neugebauer, E.A.M. Deutsche Gesellschaft für Unfallchirurgie/German Society for Trauma Surgery Head injury and outcome--what influence do concomitant injuries have? J. Trauma 2008, 65, 1036–1044. [Google Scholar] [CrossRef]
- Fröhlich, M.; Caspers, M.; Lefering, R.; Driessen, A.; Bouillon, B.; Maegele, M.; Wafaisade, A. TraumaRegister DGU Do elderly trauma patients receive the required treatment? Epidemiology and outcome of geriatric trauma patients treated at different levels of trauma care. Eur. J. Trauma Emerg. Surg. 2019. [Google Scholar] [CrossRef]
- Peterer, L.; Ossendorf, C.; Jensen, K.O.; Osterhoff, G.; Mica, L.; Seifert, B.; Werner, C.M.L.; Simmen, H.-P.; Pape, H.-C.; Sprengel, K. Implementation of new standard operating procedures for geriatric trauma patients with multiple injuries: A single level I trauma centre study. BMC Geriatr. 2019, 19, 359. [Google Scholar] [CrossRef] [PubMed]
- Tykocki, T.; Guzek, K. Anticoagulation Therapy in Traumatic Brain Injury. World Neurosurg. 2016, 89, 497–504. [Google Scholar] [CrossRef] [PubMed]
- Nishijima, D.K.; Shahlaie, K.; Sarkar, K.; Rudisill, N.; Holmes, J.F. Risk of unfavorable long-term outcome in older adults with traumatic intracranial hemorrhage and anticoagulant or antiplatelet use. Am. J. Emerg. Med. 2013, 31, 1244–1247. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Scotti, P.; Séguin, C.; Lo, B.W.Y.; de Guise, E.; Troquet, J.-M.; Marcoux, J. Antithrombotic agents and traumatic brain injury in the elderly population: Hemorrhage patterns and outcomes. J. Neurosurg. 2019, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Batchelor, J.S.; Grayson, A. A meta-analysis to determine the effect of anticoagulation on mortality in patients with blunt head trauma. Br. J. Neurosurg. 2012, 26, 525–530. [Google Scholar] [CrossRef]
- Batchelor, J.S.; Grayson, A. A meta-analysis to determine the effect of preinjury antiplatelet agents on mortality in patients with blunt head trauma. Br. J. Neurosurg. 2013, 27, 12–18. [Google Scholar] [CrossRef]
- Bonville, D.J.; Ata, A.; Jahraus, C.B.; Arnold-Lloyd, T.; Salem, L.; Rosati, C.; Stain, S.C. Impact of preinjury warfarin and antiplatelet agents on outcomes of trauma patients. Surgery 2011, 150, 861–868. [Google Scholar] [CrossRef]
- Fortuna, G.R.; Mueller, E.W.; James, L.E.; Shutter, L.A.; Butler, K.L. The impact of preinjury antiplatelet and anticoagulant pharmacotherapy on outcomes in elderly patients with hemorrhagic brain injury. Surgery 2008, 144, 598–603; discussion 603–605. [Google Scholar] [CrossRef]
- Ivascu, F.A.; Howells, G.A.; Junn, F.S.; Bair, H.A.; Bendick, P.J.; Janczyk, R.J. Predictors of mortality in trauma patients with intracranial hemorrhage on preinjury aspirin or clopidogrel. J. Trauma 2008, 65, 785–788. [Google Scholar] [CrossRef]
- Franko, J.; Kish, K.J.; O’Connell, B.G.; Subramanian, S.; Yuschak, J.V. Advanced age and preinjury warfarin anticoagulation increase the risk of mortality after head trauma. J. Trauma 2006, 61, 107–110. [Google Scholar] [CrossRef]
- Mak, C.H.K.; Wong, S.K.H.; Wong, G.K.; Ng, S.; Wang, K.K.W.; Lam, P.K.; Poon, W.S. Traumatic Brain Injury in the Elderly: Is it as Bad as we Think? Curr. Transl. Geriatr. Exp. Gerontol. Rep. 2012, 1, 171–178. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Julien, J.; Alsideiri, G.; Marcoux, J.; Hasen, M.; Correa, J.A.; Feyz, M.; Maleki, M.; de Guise, E. Antithrombotic agents intake prior to injury does not affect outcome after a traumatic brain injury in hospitalized elderly patients. J. Clin. Neurosci. 2017, 38, 122–125. [Google Scholar] [CrossRef] [PubMed]
- McIntyre, A.; Mehta, S.; Aubut, J.; Dijkers, M.; Teasell, R.W. Mortality among older adults after a traumatic brain injury: A meta-analysis. Brain Inj. 2013, 27, 31–40. [Google Scholar] [CrossRef] [PubMed]
- Panczykowski, D.M.; Okonkwo, D.O. Premorbid oral antithrombotic therapy and risk for reaccumulation, reoperation, and mortality in acute subdural hematomas. J. Neurosurg. 2011, 114, 47–52. [Google Scholar] [CrossRef] [PubMed]
- Major, J.; Reed, M.J. A retrospective review of patients with head injury with coexistent anticoagulant and antiplatelet use admitted from a UK emergency department. Emerg. Med. J. 2009, 26, 871–876. [Google Scholar] [CrossRef]
- Flibotte, J.J.; Hagan, N.; O’Donnell, J.; Greenberg, S.M.; Rosand, J. Warfarin, hematoma expansion, and outcome of intracerebral hemorrhage. Neurology 2004, 63, 1059–1064. [Google Scholar] [CrossRef]
- Karni, A.; Holtzman, R.; Bass, T.; Zorman, G.; Carter, L.; Rodriguez, L.; Bennett-Shipman, V.J.; Lottenberg, L. Traumatic head injury in the anticoagulated elderly patient: A lethal combination. Am. Surg. 2001, 67, 1098–1100. [Google Scholar]
- Banerjee, M.; Bouillon, B.; Shafizadeh, S.; Paffrath, T.; Lefering, R.; Wafaisade, A. German Trauma Registry Group Epidemiology of extremity injuries in multiple trauma patients. Injury 2013, 44, 1015–1021. [Google Scholar] [CrossRef]
- Bai, B.; Jazrawi, L.M.; Kummer, F.J.; Spivak, J.M. The use of an injectable, biodegradable calcium phosphate bone substitute for the prophylactic augmentation of osteoporotic vertebrae and the management of vertebral compression fractures. Spine 1999, 24, 1521–1526. [Google Scholar] [CrossRef]
- Rimaitis, M.; Bilskienė, D.; Tamošuitis, T.; Vilcinis, R.; Rimaitis, K.; Macas, A. Implementation of Thromboelastometry for Coagulation Management in Isolated Traumatic Brain Injury Patients Undergoing Craniotomy. Med. Sci. Monit. 2020, 26, e922879-1–e922879-11. [Google Scholar] [CrossRef]
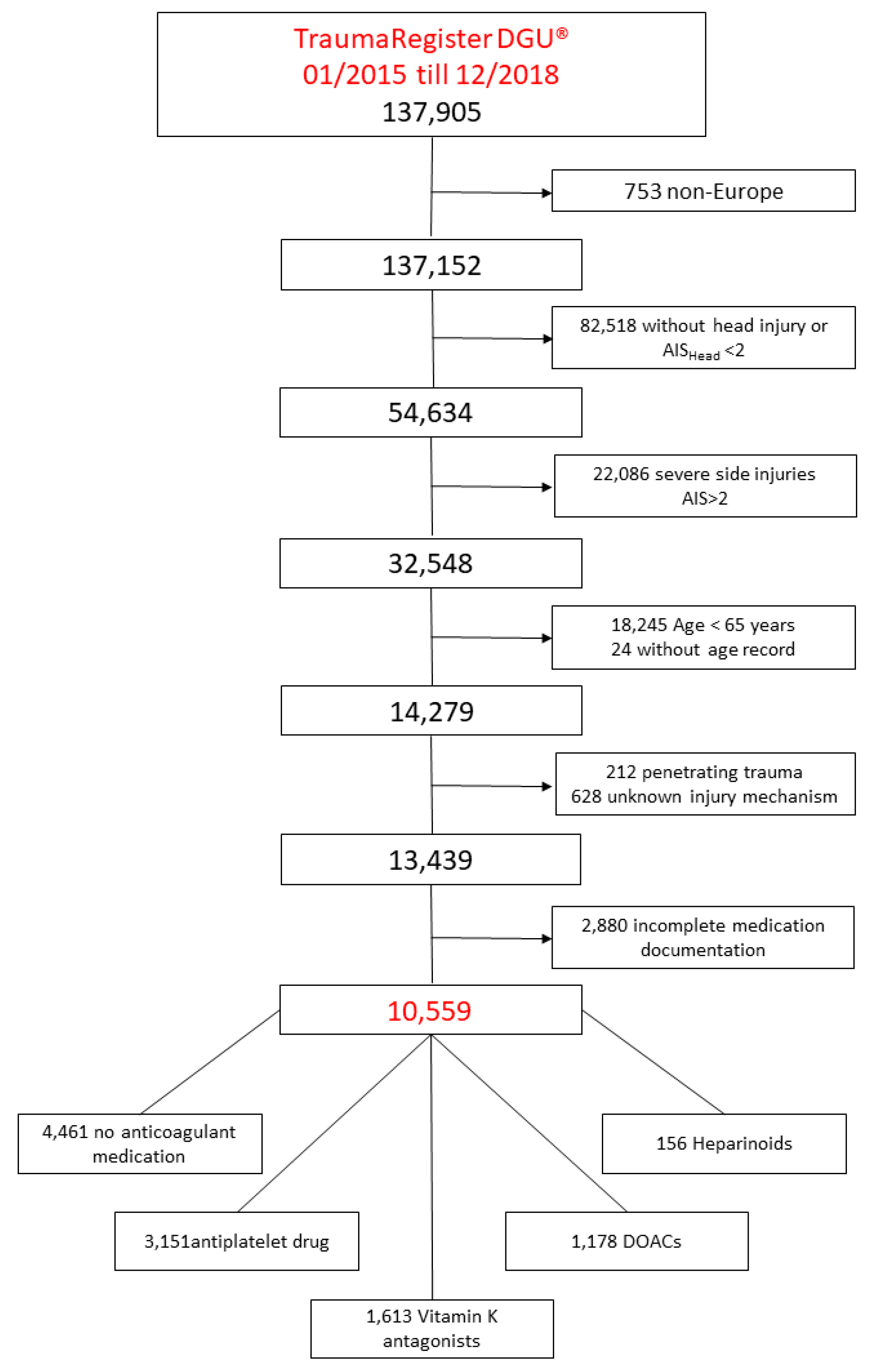
| Inclusion Criteria |
|---|
| Age ≥ 65 years |
| AISHead ≥ 2 |
| AISother then head ≤ 2 |
| Blunt trauma mechanism |
| Complete anticoagulant medication documentation |
| Admitted to a European trauma center |
| Exclusion Criteria |
| Unknown age or <65 years |
| AISHead < 2 |
| AISother then head ≥ 3 |
| Penetrating or unknown trauma mechanism |
| Incomplete anticoagulant medication documentation |
| Admitted to a non-European Trauma centre |
| NM | APD | VKA | DOACs | Heparinoids | Total | |
|---|---|---|---|---|---|---|
| Number | 4461 | 3151 | 1613 | 1178 | 156 | 10,559 |
| Proportion (%) | 42.2 | 29.8 | 15.3 | 11.2 | 1.5 | 100 |
| Age (Mean ± SD) | 76.6 (±7.7) | 80.1 (±7.3) | 80.4 (±6.8) | 81.2 (±7.0) | 80.8 (±7.4) | 78.8 (±7.6) |
| ISS (Mean ± SD) | 16.0 (±8.0) | 16.7 (±8.2) | 18.6 (±8.6) | 17.9 (±8.5) | 18.0 (±8.5) | 16.9 (±8.3) |
| AISHead | 3.5 | 3.7 | 3.9 | 3.8 | 3.9 | 3.7 |
| ASA 3/4 (%) | 50.1 | |||||
| Male (%) | 54.9 |
| Moderate TBI | 95% Confidence Interval | Severe TBI | 95% Confidence Interval | Total | 95% Confidence Interval | |
|---|---|---|---|---|---|---|
| NM | 5.3 | 27.9 | 16.9 | |||
| APD | 8.2 | 6.8–9.5 | 34.8 | 32.6–37.0 | 23.2 | 21.7–24.7 |
| VKA | 10.7 | 8.1–12.6 | 43.9 | 40.9–46.9 | 32.7 | 30.4–35.0 |
| DOAC | 10.1 | 7.3–12.3 | 41.7 | 38.2–45.2 | 30.1 | 27.5–32.7 |
| Heparinoids | 23.0 | 12.4–31.5 | 45.2 | 35.2–55.2 | 36.5 | 28.9–44.1 |
| Total | 7.5 | 34.8 | 22.9 |
| OR for Severe TBI | 95% Confidence Interval | |
|---|---|---|
| Unadjusted Odds Ratios | ||
| APD | 1.23 | 1.12–1.34 |
| VKA | 1.89 | 1.67–2.21 |
| DOAC | 1.63 | 1.43–1.86 |
| Heparinoids | 1.48 | 1.07–2.05 |
| Adjusted Odds Ratios | ||
| APD | 1.03 | 0.93–1.15 |
| VKA | 1.43 | 1.25–1.63 |
| DOAC | 1.24 | 1.07–1.43 |
| Heparinoids | 1.11 | 0.78–1.57 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Eibinger, N.; Halvachizadeh, S.; Hallmann, B.; Seibert, F.J.; Puchwein, P.; Berk, T.; Lefering, R.; Sprengel, K.; Pape, H.C.; Jensen, K.O.; et al. Is the Regular Intake of Anticoagulative Agents an Independent Risk Factor for the Severity of Traumatic Brain Injuries in Geriatric Patients? A Retrospective Analysis of 10,559 Patients from the TraumaRegister DGU®. Brain Sci. 2020, 10, 842. https://doi.org/10.3390/brainsci10110842
Eibinger N, Halvachizadeh S, Hallmann B, Seibert FJ, Puchwein P, Berk T, Lefering R, Sprengel K, Pape HC, Jensen KO, et al. Is the Regular Intake of Anticoagulative Agents an Independent Risk Factor for the Severity of Traumatic Brain Injuries in Geriatric Patients? A Retrospective Analysis of 10,559 Patients from the TraumaRegister DGU®. Brain Sciences. 2020; 10(11):842. https://doi.org/10.3390/brainsci10110842
Chicago/Turabian StyleEibinger, Nicolas, Sascha Halvachizadeh, Barbara Hallmann, Franz Josef Seibert, Paul Puchwein, Till Berk, Rolf Lefering, Kai Sprengel, Hans Christoph Pape, Kai Oliver Jensen, and et al. 2020. "Is the Regular Intake of Anticoagulative Agents an Independent Risk Factor for the Severity of Traumatic Brain Injuries in Geriatric Patients? A Retrospective Analysis of 10,559 Patients from the TraumaRegister DGU®" Brain Sciences 10, no. 11: 842. https://doi.org/10.3390/brainsci10110842
APA StyleEibinger, N., Halvachizadeh, S., Hallmann, B., Seibert, F. J., Puchwein, P., Berk, T., Lefering, R., Sprengel, K., Pape, H. C., Jensen, K. O., & The TraumaRegister DGU. (2020). Is the Regular Intake of Anticoagulative Agents an Independent Risk Factor for the Severity of Traumatic Brain Injuries in Geriatric Patients? A Retrospective Analysis of 10,559 Patients from the TraumaRegister DGU®. Brain Sciences, 10(11), 842. https://doi.org/10.3390/brainsci10110842







