Fire Properties of Acrylonitrile Butadiene Styrene Enhanced with Organic Montmorillonite and Exolit Fire Retardant
Abstract
1. Introduction
2. Materials and Methods
- HB: slow burning;
- V-2: burning stops in 30 s on a vertical sample; drops of flaming particles are allowed.
- V-1: burning stops in 30 s on a vertical sample; drops of particles permitted as long as they are not in flame.
- V-0: burning stops in 10 s on a vertical sample; drops of particles acceptable as long as they are not in flame.
- 5VB: burning stops in 60 s; no drops acceptable; samples may develop a hole.
- 5VA: burning stops in 60 s; no drops permitted; samples may not present a hole.
3. Results and Discussion
- TTI is the time taken for the specimen to ignite at 50 kW/m2 flux;
- THR is the total heat released per unit area during the entire test for the tested thickness of product;
- PHRR is the peak heat release rate;
- Residue is measured as percentage;
- Effective heat of combustion is the energy produced per kg of the product. It is averaged over the length of the test;
- FIGRA is the growth rate of the burning intensity, HRR, during a test and is calculated as the maximum value of the function (heat release rate)/(elapsed test time).
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Realinho, V.; Haurie, L.; Formosa, J.; Velasco, J.I. Flame retardancy effect of combined ammonium polyphosphate and aluminium diethyl phosphinate in crylonitrile-butadiene-styrene. Polym. Degrad. Stab. 2018, 155, 208–219. [Google Scholar] [CrossRef]
- Tarantili, P.A. Composites of Engineering Plastics with Layered Silicate Nanofillers: Preparation and Study of Microstructure and Thermomechanical Properties. In Nanocomposites and Polymers with Analytical Methods; Cuppoletti, J., Ed.; IntechOpen: London, UK, 2011; ISBN 978-953-307-352-1. [Google Scholar]
- Wang, S.; Hu, Y.; Lin, Z.; Gui, Z.; Wang, Z.; Chen, Z.; Fan, W. Flammability and thermal stability studies of ABS/Montmorillonite nanocomposite. Polym. Int. 2003, 52, 1045–1049. [Google Scholar] [CrossRef]
- Weng, Z.; Wang, J.; Senthil, T.; Wu, L. Mechanical and thermal properties of ABS/montmorillonite nanocomposites for fused deposition modeling 3D printing. Mater. Des. 2016, 102, 276–283. [Google Scholar] [CrossRef]
- Malas, A.; Pal, P.; Das, C.K. Effect of expanded graphite and modified graphite flakes on the physical and thermo-mechanical properties of styrene butadiene rubber/polybutadiene rubber (SBR/BR) blends. Mater. Des. 2014, 55, 664–673. [Google Scholar] [CrossRef]
- Jian, R.-K.; Chen, L.; Chen, S.; Long, J.-W.; Wang, Y.-Z. A novel flame-retardant acrylonitrile-butadiene-styrene system based on aluminum isobutylphosphinate and red phosphorus: Flame retardance, thermal degradation and pyrolysis behavior. Polym. Degrad. Stab. 2014, 109, 184–193. [Google Scholar] [CrossRef]
- Morgan, A.B.; Bundy, M. Cone calorimeter analysis of UL-94 V-rated plastics. Fire Mater. 2007, 31, 257–283. [Google Scholar] [CrossRef]
- Schartel, B.; Hull, T.R. Development of fire-retarded materials interpretation of cone calorimeter data. Fire Mater. 2007, 31, 327–354. [Google Scholar] [CrossRef]
- Wu, N.; Lang, S. Flame retardancy and toughness modification of flame retardant polycarbonate/acrylonitrile-butadiene-styrene/AHP composites. Polym. Degrad. Stab. 2016, 123, 26–35. [Google Scholar] [CrossRef]
- Cao, X.; Yang, Y.; Luo, H.; Cai, X. High efficiency intumescent flame retardancy between Hexakis (4-nitrophenoxy) cyclotriphosphazene and ammonium polyphosphate on ABS. Polym. Degrad. Stab. 2017, 143, 259–265. [Google Scholar] [CrossRef]
- Singh, P.; Ghosh, A.K. Torsional, tensile and structural properties of acrylonitrile–butadiene–styrene clay nanocomposites. Mater. Des. 2014, 55, 137–145. [Google Scholar] [CrossRef]
- Gilman, J.W. Flammability and Thermal Stability Studies of Polymer Layered-Sili-cate (Clay) Nanocomposites. Appl. Clay Sci. 1999, 15, 31–49. [Google Scholar] [CrossRef]
- Fontaine, P.G.; Gallos, A.; Bourbigot, S. Role of montmorillonite for enhancing fire retardancy of intumescent. In Proceedings of the Eleventh International Symposium on Fire Safety Science, Christchurch, New Zealand, 9–14 February 2014; pp. 808–820. [Google Scholar]
- Pour, R.H.; Soheilmoghaddam, M.; Hassan, A.; Bourbigot, S. Flammability and thermal properties of polycarbonate/acrylonitrilebutadiene-styrene nanocomposites reinforced with multilayer grapheme. Polym. Degrad. Stab. 2015, 120, 88–97. [Google Scholar] [CrossRef]
- Zhong, H.; Wei, P.; Jiang, P.; Wang, G. Thermal degradation behaviors and flame retardancy of PC/ABS with novel silicon-containing flame retardant. Fire Mater. 2007, 31, 411–423. [Google Scholar] [CrossRef]
- Qin, H.; Zhang, S.; Zhao, C.; Hu, G.; Yang, M. Flame retardant mechanism of polymer/clay nanocomposites based on polypropylene. Polymer 2005, 46, 8386–8395. [Google Scholar] [CrossRef]
- Levchik, S.V.; Weil, E.D. Overview of recent developments in the flame retardancy of polycarbonates. Polym. Int. 2005, 54, 981–998. [Google Scholar] [CrossRef]
- Murashko, E.A.; Levchik, G.F.; Levchik, S.V.; Bright, D.A.; Dashevsky, S. Fire retardant action of resorcinol bis(diphenyl phosphate) in PC-ABS blend. II. Reactions in the condensed phase. J. Appl. Polym. Sci. 1999, 71, 1863–1872. [Google Scholar] [CrossRef]
- Bardziński, P.J. On the impact of intermolecular interactions between the quaternary ammonium ions on interlayer spacing of quat-intercalated montmorillonite: A molecular mechanics and ab-initio study. Appl. Clay Sci. 2014, 95, 323–339. [Google Scholar] [CrossRef]
- Gilman, J.W.; Jackson, C.L.; Morgan, A.B.; Harris, R., Jr.; Manias, E.; Giannelis, E.P.; Wuthenow, M.; Hilton, D. Flammability properties of polymer-layered silicate nanocomposites. Propylene and polystyrene nanocomposites. Chem. Mater. 2000, 12, 1866–1873. [Google Scholar] [CrossRef]
- Meri, R.M.; Zicans, J.; Ivanova, T.; Berzina, R.; Saldabola, R.; Maksimovs, R. The effect of introduction of montmorillonite clay (MMT) on the elastic properties of polycarbonate (PC) composition with acrylonitrile-butadiene styrene (ABS). Compos. Struct. 2015, 134, 950–956. [Google Scholar] [CrossRef]
- Khobragade, P.S.; Hansora, D.P.; Naik, J.B.; Chatterjee, A. Flame retarding performance of elastomeric nanocomposites: A review. Polym. Degrad. Stab. 2016, 130, 194–240. [Google Scholar] [CrossRef]
- Pagacz, J.; Pielichowski, K. Preparation and characterization of PVC/Montmorillonite nanocomposites—A review. J. Vinyl Addit. Technol. 2009, 15, 61–76. [Google Scholar] [CrossRef]
- Kader, M.A.; Kim, K.; Lee, Y.S.; Nah, C. Preparation and properties of nitrile rubber/montmorillonite nanocomposites via latex blending. J. Mater. Sci. 2006, 41, 7341–7352. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhu, W.; Lu, Y.; Gao, Z.; Gu, J. Nano-scale blocking mechanism of MMT and its effects on the properties of polyisocyanate-modified soybean protein adhesive. Ind. Crops Prod. 2014, 57, 35–42. [Google Scholar] [CrossRef]
- Xia, Y.; Jian, X.; Li, J.; Wang, X.; Xu, Y. Synergistic effect of montmorillonite and intumescent flame retardant on flame retardancy enhancement of ABS. Polym. Plast. Technol. Eng. 2007, 46, 227–232. [Google Scholar] [CrossRef]
- Lu, C.; Liu, L.; Chen, N.; Wang, X.; Yang, D.; Huang, X.; Yao, D. Influence of clay dispersion on flame retardancy of ABS/PA6/APP blends. Polym. Degrad. Stab. 2015, 114, 16–29. [Google Scholar] [CrossRef]
- Despinasse, M.C.; Schartel, B. Aryl phosphateearyl phosphate synergy in flame-retarded bisphenol A polycarbonate/acrylonitrile-butadiene-styrene. Thermochim. Acta 2013, 563, 51–61. [Google Scholar] [CrossRef]
- Yoon, P.J.; Hunter, D.L.; Paul, D.R. Polycarbonate Nanocomposites Part 1. Effect of Organoclay Structure on Morphology and Properties. Polymer 2003, 44, 5323–5339. [Google Scholar] [CrossRef]
- Langfeld, K.; Wilke, A.; Sut, A.; Greiser, S.; Ulmer, B.; Andrievici, V.; Limbach, P.; Bastian, M.; Schartel, B. Halogen-free fire retardant styrene–ethylene–butylene–styrene-based thermoplastic elastomers using synergistic aluminum diethylphosphinate-based combinations. J. Fire Sci. 2015, 33, 157–177. [Google Scholar] [CrossRef]
- Ramani, A.; Dahoe, A.E. On flame retardancy in polycaprolactam composites by aluminium diethylphosphinate and melamine polyphosphate in conjunction with organically modified montmorillonite nanoclay. Polym. Degrad. Stab. 2014, 105, 1–11. [Google Scholar] [CrossRef]
- Guo, C.; Zhou, L.; Lv, J. Effects of expandable graphite and modified ammonium polyphosphate on the flame-retardant and mechanical properties of wood flour-polypropylene composites. Polym. Polym. Compos. 2013, 21, 449–456. [Google Scholar] [CrossRef]
- Ma, H.; Tong, L.; Xu, Z.; Fang, Z. Intumescent flame retardant-montmorillonite synergism in ABS nanocomposites. Appl. Clay Sci. 2008, 42, 238–245. [Google Scholar] [CrossRef]
- Yoon, P.J.; Hunter, D.L.; Paul, D.R. Polycarbonate Nanocomposites Part 2. Degradation and Color Formation. Polymer 2003, 44, 5341–5354. [Google Scholar] [CrossRef]
- Nevalainen, K.; Vuorinen, J.; Villman, V. Characterization of Twin-Screw-Extruder-Compounded Polycarbonate Nanoclay Composites. Polym. Eng. Sci. 2009, 49, 631–640. [Google Scholar] [CrossRef]
- Feng, J.; Hao, J.; Du, J.; Yang, R. Effects of Organoclay Modifiers on the Flammability, Thermal and Mechanical Properties of polycarbonate Nanocomposites filled with a Phosphate and Organoclays. Polym. Degrad. Stab. 2012, 97, 108–117. [Google Scholar] [CrossRef]
- Nayak, S.K.; Mohanty, S.; Samal, S.K. Mechanical and Thermal Properties Enhancement of Polycarbonate Nanocomposites Prepared by Melt Compounding. J. Appl. Polym. Sci. 2010, 117, 2101. [Google Scholar] [CrossRef]
- Feyz, E.; Jahani, Y.; Esfandeh, M. Effect of a Nanoclay/Triphenyl Phosphate Hybrid System on the Fire Retardancy of Polycarbonate/Acrylonitrile–Butadiene–Styrene Blend. J. Appl. Polym. Sci. 2011, 120, 3435. [Google Scholar] [CrossRef]
- Table Curve 3D, Systat Software, Inc. Available online: http:// www.systat (accessed on 11 December 2019).
- Hirayama, D.; Saron, C. Characterisation of recycled acrylonitrile-butadiene-styrene and high-impact polystyrene from waste computer equipment from Brazil. Waste Manag. Res. 2015, 33, 543–549. [Google Scholar] [CrossRef]
- Beigbeder, J.; Perrin, D.; Mascaro, J.F.; Lopez-Cuesta, J.M. Study of the physico-chemical properties of recycled polymers from waste electrical and electronic equipment (WEEE) sorted by high resolution near infrared devices. Conserv. Recycl. 2013, 78, 105–114. [Google Scholar] [CrossRef]
- Du, X.; Yu, H.; Wang, Z.; Tang, T. Effect of anionic organoclay with special aggregate structure on the flame retardancy of acrylonitrile-butadiene-styrene/clay composites. Polym. Degrad. Stab. 2010, 95, 587–592. [Google Scholar] [CrossRef]
- Zhang, J.; Jiang, D.D.; Wang, D.; Wilkie, C.A. Styrenic polimer nanocomposites based on a aligomerically-modified clay with high inorganic content. Polym. Degrad. Stab. 2006, 91, 2665–2674. [Google Scholar] [CrossRef][Green Version]
- Wang, S.; Hu, Y.; Song, L.; Wang, Z.; Chen, Z.; Fan, W. Preparation and thermal properties of ABS/montmorillonite nanocomposite. Polym. Degrad. Stab. 2002, 77, 423–426. [Google Scholar] [CrossRef]
- Zhang, Y.; Chen, X.; Fang, Z. Flame retardant ABS with a novel polyphosphate derived from biomass. Adv. Mater. Res. 2011, 284–286, 187–192. [Google Scholar] [CrossRef]
- Wu, N.; Li, X. Flame retardancy and synergistic flame retardant mechanisms of acrylonitrile-butadiene-styrene composites based on aluminum hypophosphite. Polym. Degrad. Stab. 2014, 105, 265–276. [Google Scholar] [CrossRef]
- Marcilla, A.; Garcia Quesada, J.C.; Gomez-Siurana, A.; Berenguer, D. Characterization of styrene–butadiene copolymers by catalytic pyrolysis over Al-MCM-41. J. Anal. Appl. Pyrol. 2009, 85, 327–333. [Google Scholar] [CrossRef]
- Mattausch, H. Properties and applications of nanoclay composites. In Polymer Nanoclay Composites; Elsevier: Amsterdam, The Netherlands, 2015; pp. 127–155. [Google Scholar]
- Simionescu, T.M.; Minea, A.A. The effect of montmorillonite clay and fire retardants on the heat of combustion of recycled acrylonitrile-butadiene styrene. Environ. Eng. Manag. J. 2019, 18, 317–326. [Google Scholar]
- Zhuge, J.; Tang, Y.; Chen, R.-H.; Ibeh, C.; Hu, Y. Flammability of carbon nanofiber-clay nano paper based polymer composites. Polym. Adv. Technol. 2011, 22, 1403–1413. [Google Scholar] [CrossRef]
- Ahmed, L.; Zhang, B.; Hawkins, S.; Mannan, M.S.; Cheng, Z. Study of thermal and mechanical behaviors of flame retardant polystyrene-based nanocomposites prepared via in-situ polymerization method. J. Loss Prev. Process 2017, 49, 228–239. [Google Scholar] [CrossRef]
- Wang, L.; He, X.; Wilkie, C.A. The utility of Nanocomposites in fire retardancy. Materials 2010, 3, 4580–4606. [Google Scholar] [CrossRef]
| Exolit OP 1230 | Exolit AP 422 | |
|---|---|---|
| Chemical formula | [(C2H5)2PO2]3Al | (NH4PO3)n |
| Phosphor content, % | 23.3%–24% | 31%–32% |
| Humidity, % | max 0.2% | max 0.25% |
| Density, g/cm3 | 1.35 | 1.9 |
| Decomposing temperature, °C | >300 | >275 |
| Particle dimension | 20–40 µm | 17 µm |
| Specimen | Recycled Acrylonitrile-Butadiene-Styrene, reABS (wt%) | Organic Montmorillonite, OMT (wt%) | Exolit OP 1230 (wt%) | Exolit AP 422 (wt%) |
|---|---|---|---|---|
| reABS | 100.0 | - | - | - |
| reABS 1% OMT | 99.0 | 1.0 | - | - |
| reABS 15% FR | 85.0 | - | 7.5 | 7,.5 |
| reABS 1% OMT 15% FR | 84.0 | 1.0 | 7.5 | 7.5 |
| reABS 1% OMT 18% FR | 81.0 | 1.0 | 9.0 | 9.0 |
| reABS 1% OMT 20% FR | 79.0 | 1.0 | 10.0 | 10.0 |
| reABS 2% OMT | 98.0 | 2.0 | - | - |
| reABS 2% OMT 15% FR | 83.0 | 2.0 | 7.5 | 7.5 |
| reABS 2% OMT 18% FR | 80.0 | 2.0 | 9.0 | 9.0 |
| reABS 2% OMT 20% FR | 78.0 | 2.0 | 10.0 | 10.0 |
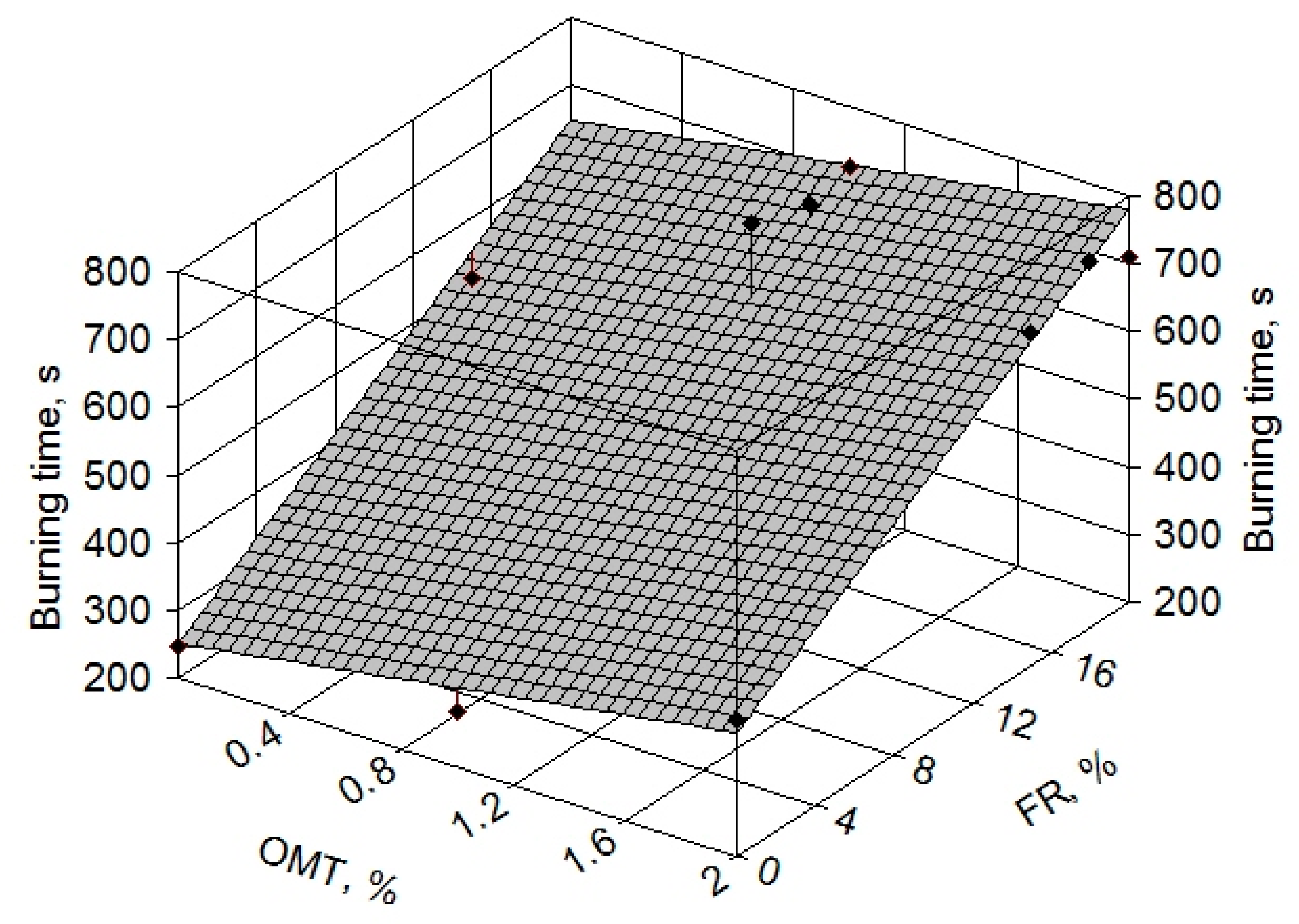
| Specimen | LOI [%] | Behavior at Burning after First Ignition | Drop Type | UL 94 |
|---|---|---|---|---|
| reABS | 18.5 | burnt completely | burning drops | HB |
| reABS 15% FR | 25.5 | burned 10 s | burning drops | V-2 |
| reABS 1% OMT | 20.8 | burnt completely | burning drops | HB |
| reABS 1% OMT 15% FR | 25.8 | burned 9 s | no burning drops | V-1 |
| reABS 1% OMT 18% FR | 27.7 | burned 2 s | no drops | V-0 |
| reABS 1% OMT 20% FR | 28 | burned 0 s | no drops | V-0 |
| reABS 2% OMT | 22 | burnt completely | burning drops | HB |
| reABS 2% OMT 15% FR | 27 | burned 1 s | no drops | V-0 |
| reABS 2% OMT 18% FR | 28.3 | burned 0 s | no drops | V-0 |
| reABS 2% OMT 20% FR | 28.7 | burned 0 s | no drops | V-0 |
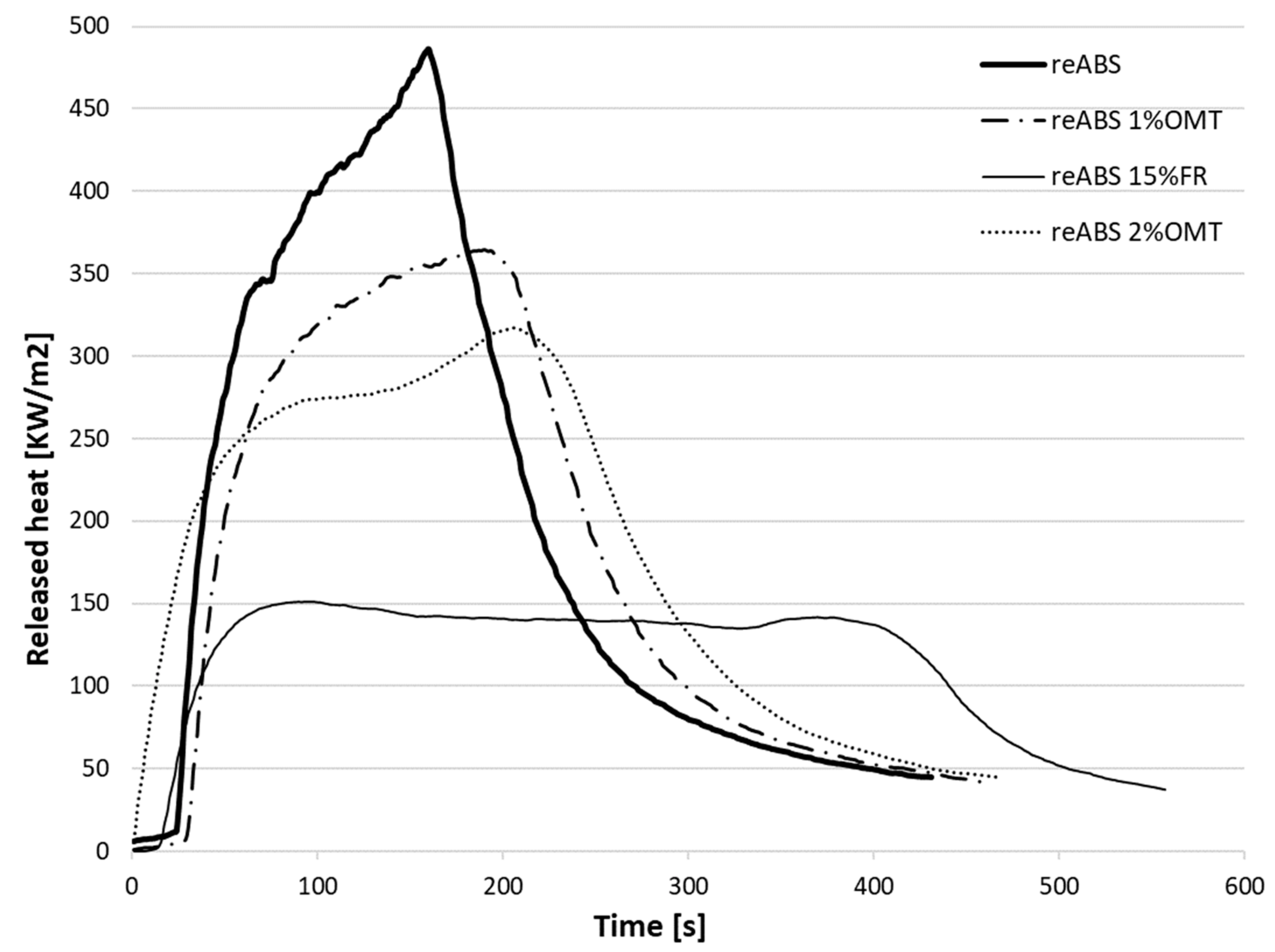
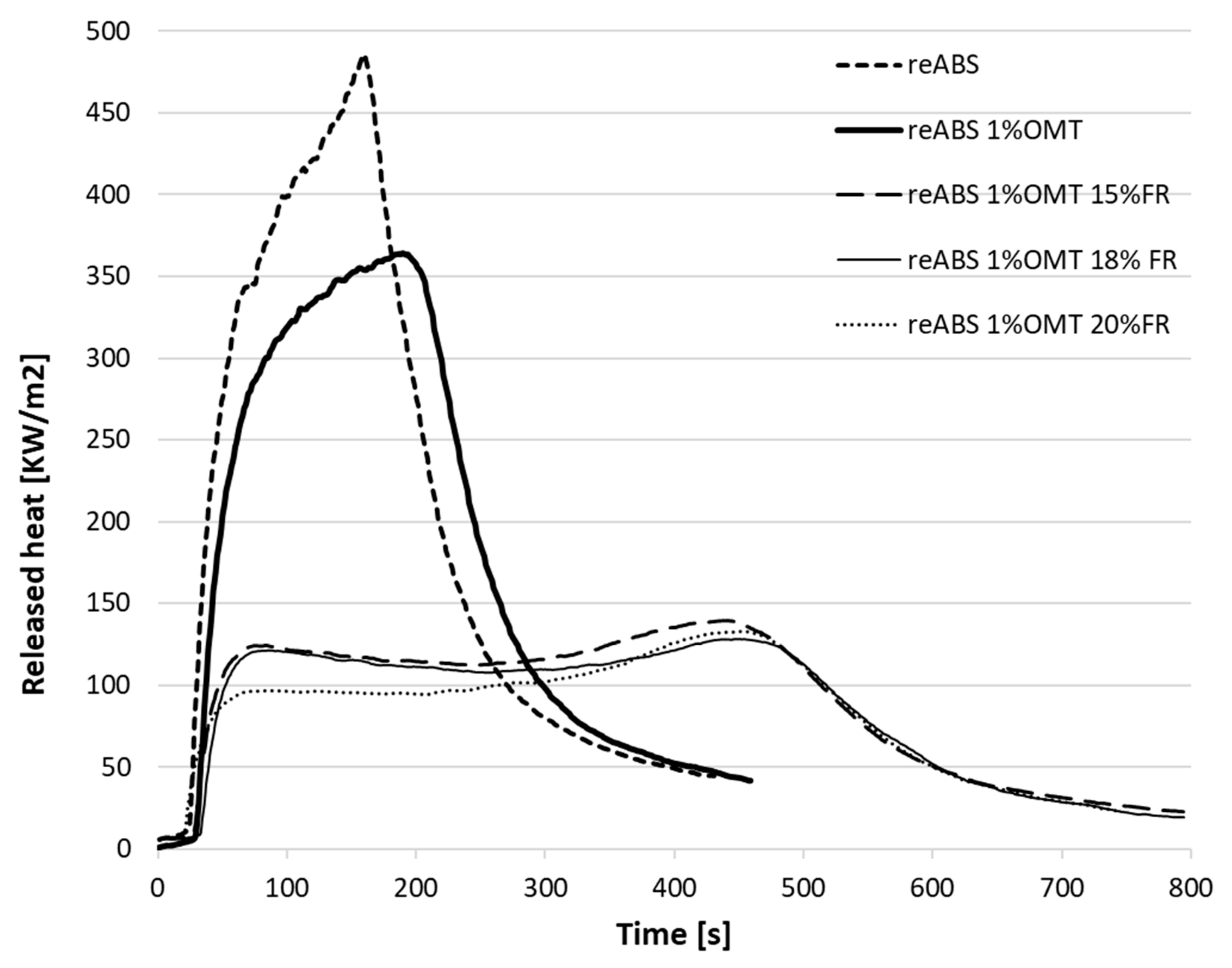
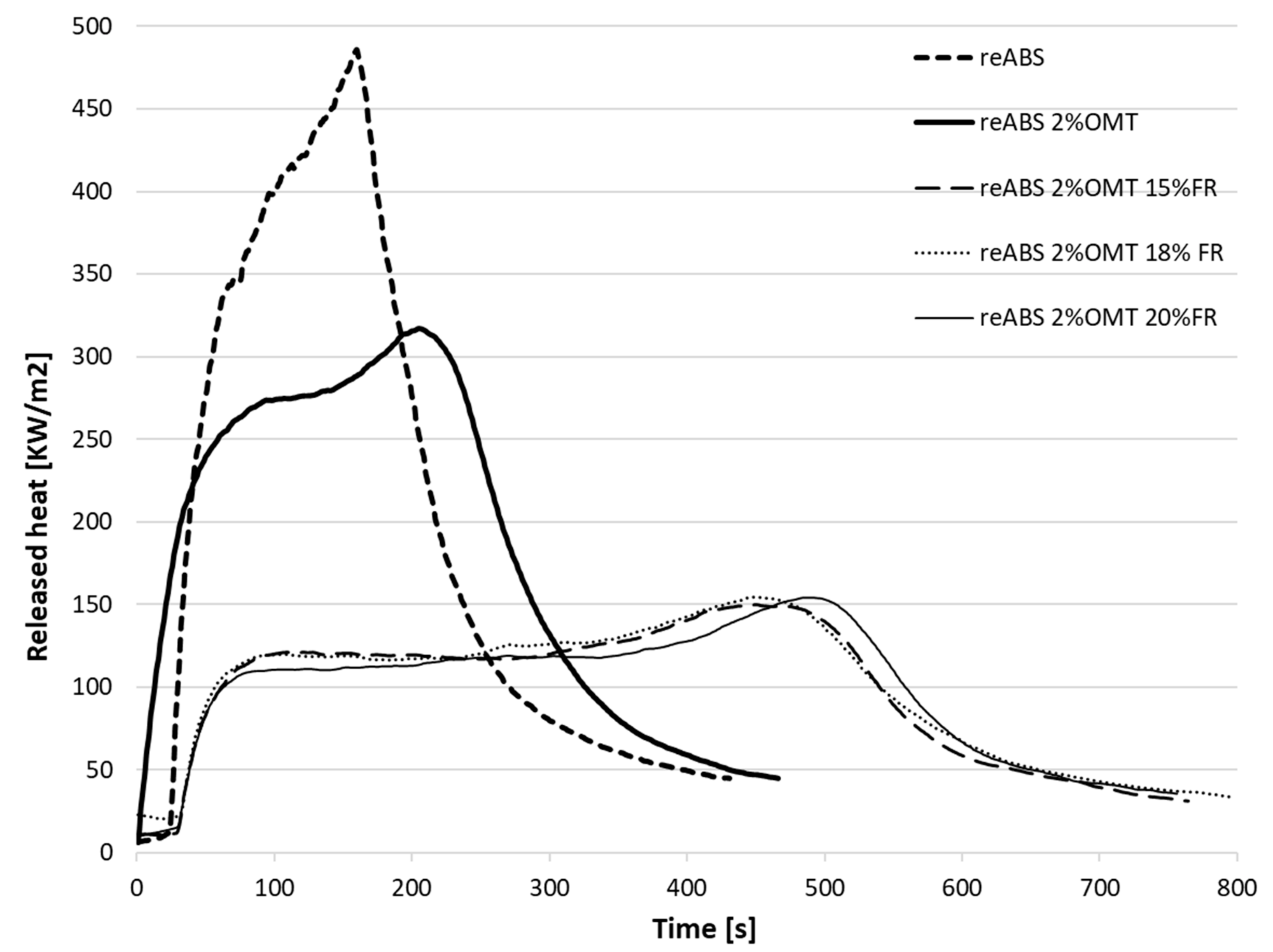
| Specimen | Total Heat Released Per Unit Area, THR (MJ/m2) | Peak Heat Release Rate, PHRR (kW/m2) | Time to Ignition, TTI (s) | Residue (wt%) | Time to PHRR (s) | Growth Rate of the Burning Intensity, FIGRA(W/s) |
|---|---|---|---|---|---|---|
| reABS | 85.52 | 486.05 | 24 | 1.6 | 152 | 3.45 |
| reABS 15% FR | 64.05 | 150.92 | 21 | 10.7 | 85 | 1.74 |
| reABS 1% OMT | 82.73 | 364.38 | 23 | 4.1 | 182 | 2.23 |
| reABS 1% OMT 15% FR | 71.06 | 139.75 | 28 | 13.5 | 429 | 0.38 |
| reABS 1% OMT 18% FR | 67.37 | 128.14 | 33 | 13.1 | 430 | 0.34 |
| reABS 1% OMT 20% FR | 62.54 | 132.76 | 42 | 17.7 | 464 | 0.33 |
| reABS 2% OMT | 84.99 | 317.29 | 20 | 6.4 | 215 | 1.66 |
| reABS 2% OMT 15% FR | 74.57 | 149.69 | 29 | 15.8 | 434 | 0.39 |
| reABS 2% OMT 18% FR | 77.85 | 154.26 | 30 | 17.3 | 429 | 0.41 |
| reABS 2% OMT 20% FR | 74.15 | 154.15 | 29 | 18.9 | 475 | 0.38 |
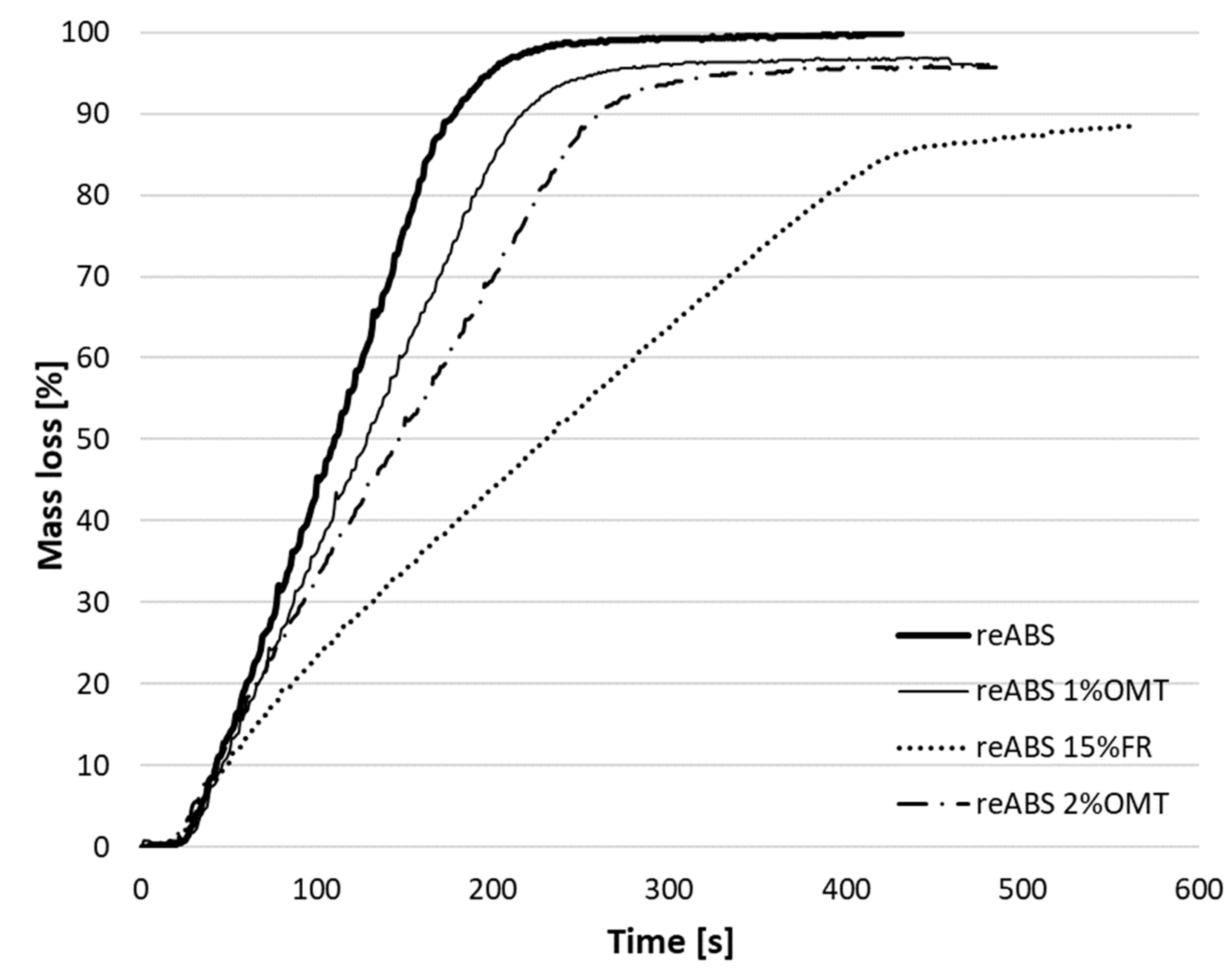
| Specimen | Mass after Burning [%] | ||
|---|---|---|---|
| 100 s | 200 s | 300 s | |
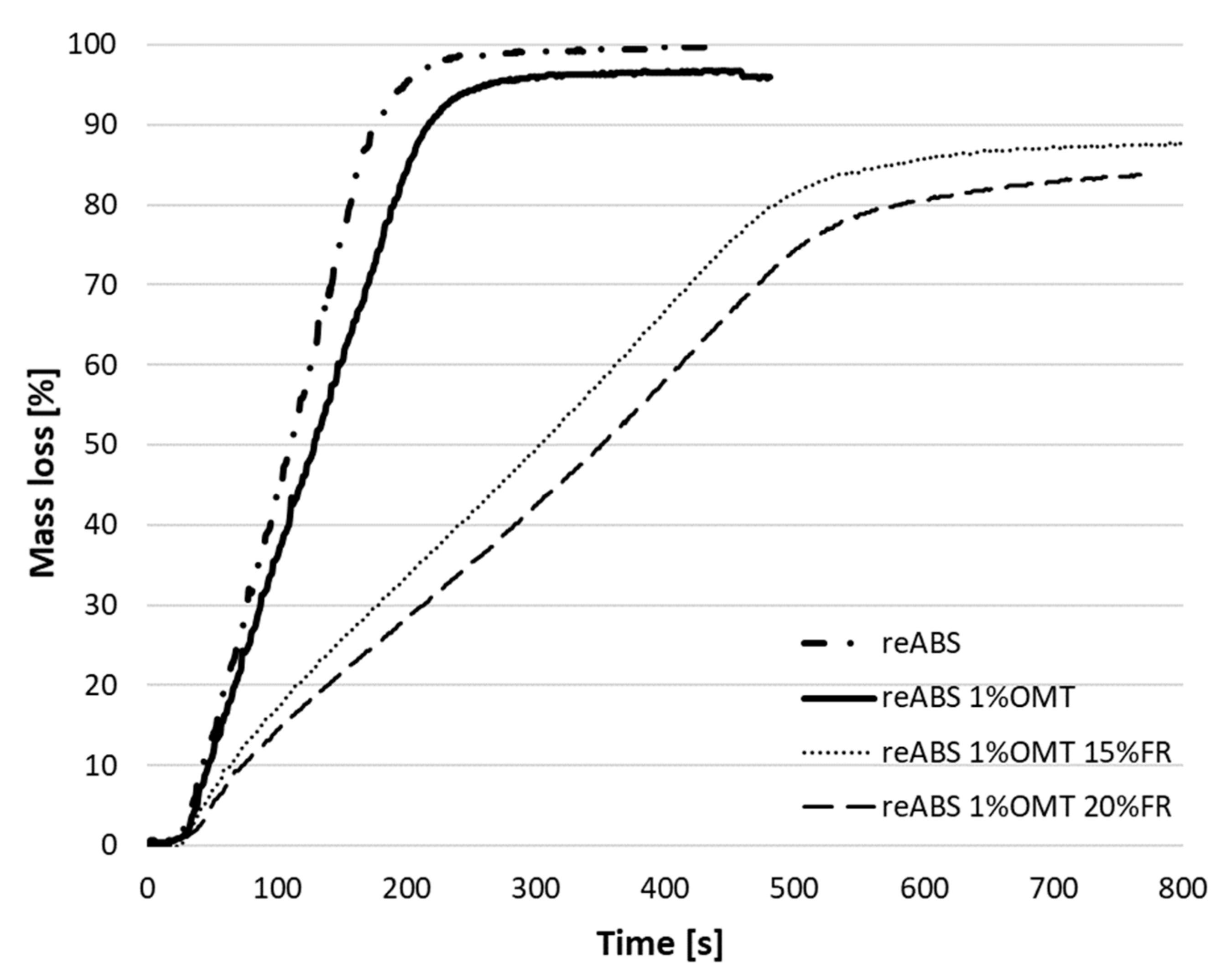
| reABS | 55.03 | 4.35 | 0.74 |
| reABS 15% FR | 76.34 | 55.79 | 35.99 |
| reABS 1% OMT | 63.45 | 15.28 | 4.16 |
| reABS 1% OMT 15% FR | 82.39 | 66.29 | 50.45 |
| reABS 1% OMT 18% FR | 84.29 | 69.23 | 55.47 |
| reABS 1% OMT 20% FR | 85.49 | 71.44 | 57.35 |
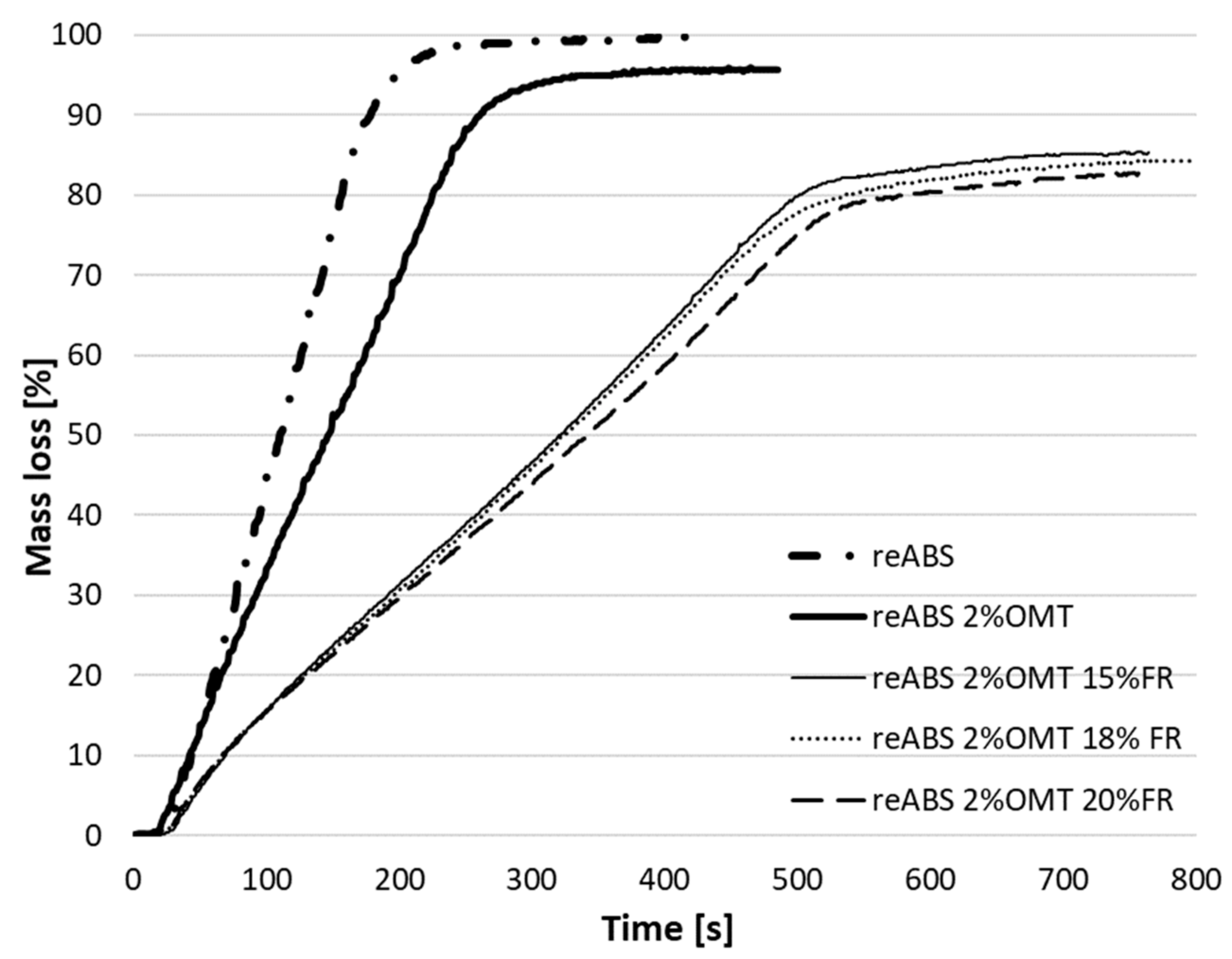
| reABS 2% OMT | 66.41 | 29.71 | 6.3 |
| reABS 2% OMT 15% FR | 84.17 | 68.37 | 53.2 |
| reABS 2% OMT 18% FR | 84.03 | 69.2 | 54.11 |
| reABS 2% OMT 20% FR | 84.33 | 70.13 | 55.95 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Simionescu, T.M.; Minea, A.A.; Reis, P.N.B.d. Fire Properties of Acrylonitrile Butadiene Styrene Enhanced with Organic Montmorillonite and Exolit Fire Retardant. Appl. Sci. 2019, 9, 5433. https://doi.org/10.3390/app9245433
Simionescu TM, Minea AA, Reis PNBd. Fire Properties of Acrylonitrile Butadiene Styrene Enhanced with Organic Montmorillonite and Exolit Fire Retardant. Applied Sciences. 2019; 9(24):5433. https://doi.org/10.3390/app9245433
Chicago/Turabian StyleSimionescu, Tudor Mihai, Alina Adriana Minea, and Paulo Nobre Balbis dos Reis. 2019. "Fire Properties of Acrylonitrile Butadiene Styrene Enhanced with Organic Montmorillonite and Exolit Fire Retardant" Applied Sciences 9, no. 24: 5433. https://doi.org/10.3390/app9245433
APA StyleSimionescu, T. M., Minea, A. A., & Reis, P. N. B. d. (2019). Fire Properties of Acrylonitrile Butadiene Styrene Enhanced with Organic Montmorillonite and Exolit Fire Retardant. Applied Sciences, 9(24), 5433. https://doi.org/10.3390/app9245433







