Potential of Advanced Consolidants for the Application on Sandstone
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of the Nanoparticle-Modified Consolidants
2.3. Methods
2.4. Accelerated Weathering
3. Results and Discussion
3.1. Characterization of the Used Rock Substrate
3.2. Characterization of the Consolidant Xerogels
3.2.1. Effect of Catalyst and Added Nanoparticles on Xerogel Texture Properties
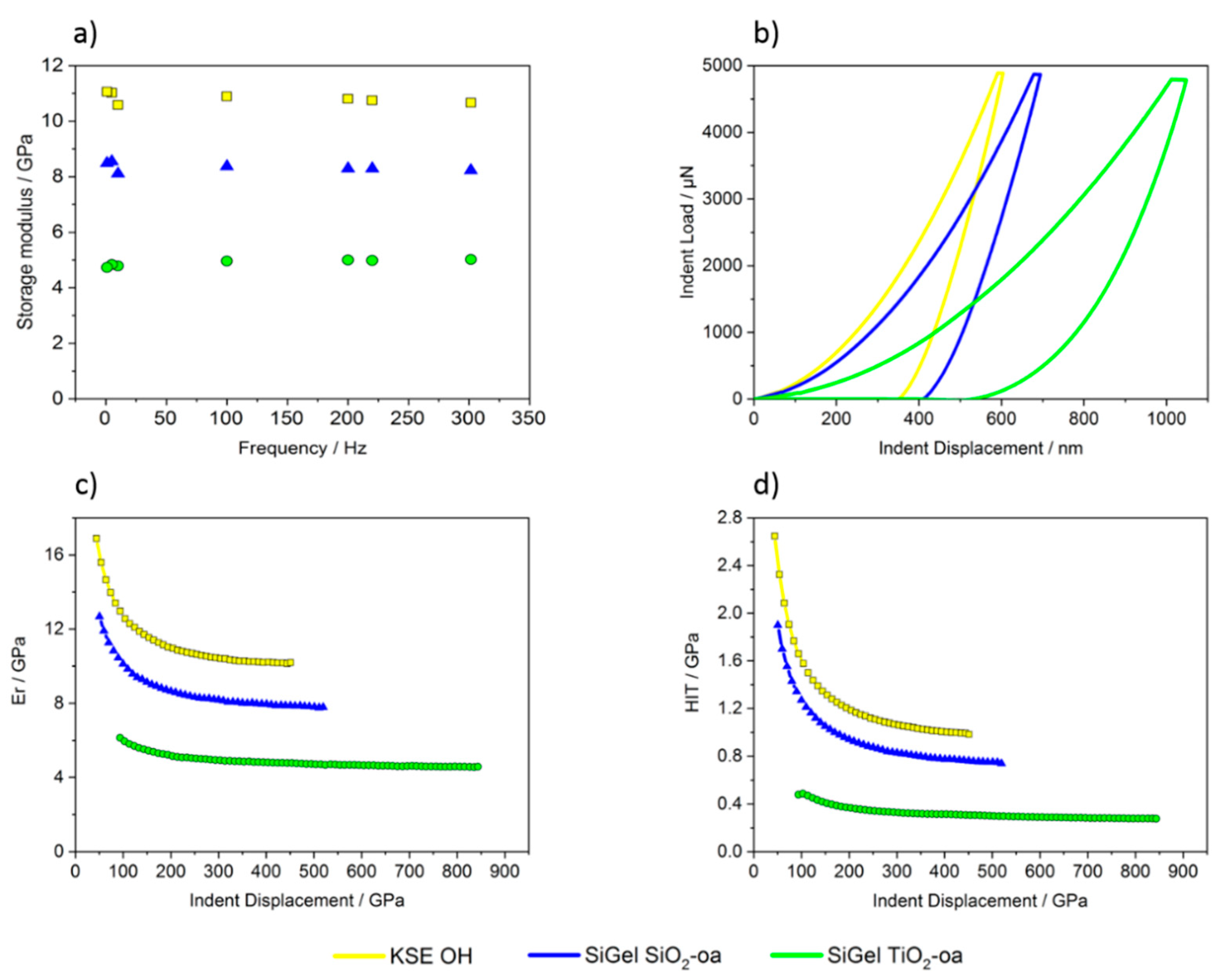
3.2.2. Mechanical Properties of the Xerogels in Microscale
3.2.3. Accelerated Weathering of Xerogels
3.3. Application of the Consolidants on the Sandstone
3.3.1. Uptake of the Consolidants
3.3.2. Mechanical Testing in Microscale
3.3.3. Mechanical Testing in Macroscale
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wheeler, G.; Goins, E.S. Alkoxysilanes and the Consolidation of Stone; The Getty Conservation Institute: Los Angeles, CA, USA, 2005; ISBN 9780892368150. [Google Scholar]
- Brinker, C.J.; Scherer, G.W. Sol-Gel Science: The Physics and Chemistry of Sol-Gel Processing; Academic Press: Boston, MA, USA, 1990; ISBN 0-12-134970-5. [Google Scholar]
- Miliani, C.; Velo-Simpson, M.L.; Scherer, G.W. Particle-modified consolidants: A study on the effect of particles on sol-gel properties and consolidation effectiveness. J. Cult. Herit. 2007, 8, 1–6. [Google Scholar]
- Blanco, E.; Esquivias, L.; Litrán, R.; Piñero, M.; Ramírez-del-Solar, M.; De La Rosa-Fox, N. Sonogels and derived materials. Appl. Organomet. Chem. 1999, 13, 399–418. [Google Scholar] [CrossRef]
- Mosquera, M.J.; De Los Santos, D.M.; Rivas, T. Surfactant-synthesized ormosils with application to stone restoration. Langmuir 2010, 26, 6737–6745. [Google Scholar] [CrossRef] [PubMed]
- Zárraga, R.; Cervantes, J.; Salazar-Hernandez, C.; Wheeler, G. Effect of the addition of hydroxyl-terminated polydimethylsiloxane to TEOS-based stone consolidants. J. Cult. Herit. 2010, 11, 138–144. [Google Scholar] [CrossRef]
- Liu, R.; Han, X.; Huang, X.; Li, W.; Luo, H. Preparation of three-component TEOS-based composites for stone conservation by sol-gel process. J. Sol-Gel Sci. Technol. 2013, 68, 19–30. [Google Scholar] [CrossRef]
- Kim, E.K.; Won, J.; Do, J.Y.; Kim, S.D.; Kang, Y.S. Effects of silica nanoparticle and GPTMS addition on TEOS-based stone consolidants. J. Cult. Herit. 2009, 10, 214–221. [Google Scholar] [CrossRef]
- Elhaddad, F.; Carrascosa, L.A.M.; Mosquera, M.J. Long-term effectiveness, under a coastal environment, of a novel conservation nanomaterial applied on sandstone from a Roman archaeological site. J. Cult. Herit. 2018, 34, 208–217. [Google Scholar] [CrossRef]
- Mosquera, M.J.; Bejarano, M.; De la Rosa-Fox, N.; Esquivias, L. Producing crack-free colloid-polymer hybrid gels by tailoring porosity. Langmuir 2003, 19, 951–957. [Google Scholar] [CrossRef]
- Remzova, M.; Zouzelka, R.; Brzicova, T.; Vrbova, K.; Pinkas, D.; Rőssner, P.; Topinka, J.; Rathousky, J. Toxicity of TiO2, ZnO, and SiO2 Nanoparticles in Human Lung Cells: Safe-by-Design Development of Construction Materials. Nanomaterials 2019, 9, 968. [Google Scholar] [CrossRef]
- Xu, F.; Li, D.; Zhang, Q.; Zhang, H.; Xu, J. Effects of addition of colloidal silica particles on TEOS-based stone protection using n-octylamine as a catalyst. Prog. Org. Coat. 2012, 75, 429–434. [Google Scholar] [CrossRef]
- Remzova, M.; Sasek, P.; Frankeova, D.; Slizkova, Z.; Rathousky, J. Effect of modified ethylsilicate consolidants on the mechanical properties of sandstone. Constr. Build. Mater. 2016, 112, 674–681. [Google Scholar] [CrossRef]
- Remzova, M.; Carrascosa, L.A.M.; Mosquera, M.J.; Rathousky, J. Modified ethylsilicates as efficient innovative consolidants for sedimentary rock. Coatings 2019, 9, 6. [Google Scholar] [CrossRef]
- Ban, M.; Mascha, E.; Weber, J.; Rohatsch, A.; Rodrigues, J.D. Efficiency and compatibility of selected alkoxysilanes on porous carbonate and silicate stones. Materials 2019, 12, 156. [Google Scholar] [CrossRef] [PubMed]
- Verganelaki, A.; Kapridaki, C.; Maravelaki-Kalaitzaki, P. Modified Tetraethoxysilane with Nanocalcium Oxalate in One-Pot Synthesis for Protection of Building Materials. Ind. Eng. Chem. Res. 2015, 54, 7195–7206. [Google Scholar] [CrossRef]
- Verganelaki, A.; Kilikoglou, V.; Karatasios, I.; Maravelaki-Kalaitzaki, P. A biomimetic approach to strengthen and protect construction materials with a novel calcium-oxalate-silica nanocomposite. Constr. Build. Mater. 2014, 62, 8–17. [Google Scholar] [CrossRef]
- Keppert, M.; Žumár, J.; Čáchová, M.; Koňáková, D.; Svora, P.; Pavlík, Z.; Vejmelková, E.; Černý, R. Water Vapor Diffusion and Adsorption of Sandstones: Influence of Rock Texture and Composition. Adv. Mater. Sci. Eng. 2016, 2016, 8039748. [Google Scholar] [CrossRef]
- Al-Naddaf, M.; Wakid, F.; Abu, A.Y. Micro-Drilling Resistance Measurement: A new technique to estimate the porosity of a building stone. Mediterr. Archaeol. Archaeom. 2013, 13, 225–233. [Google Scholar]
- ASTM G 155-05a Standard Practice for Operating Xenon Arc Light Apparatus for Exposure of Non-Metallic Materials; ASTM International: West Conshohocken, PA, USA, 2005.
- Mosquera, M.J.; De Los Santos, D.M.; Montes, A.; Valdez-Castro, L. New nanomaterials for consolidating stone. Langmuir 2008, 24, 2772–2778. [Google Scholar] [CrossRef]
- Sena da Fonseca, B.; Ferreira, M.J.; Taryba, M.G.; Piçarra, S.; Ferreira Pinto, A.P.; de Montemor, M.F. Alkoxysilane-based sols for consolidation of carbonate stones: Impact of the carbonate medium in the sol-gel processes. J. Cult. Herit. 2019, 37, 63–72. [Google Scholar] [CrossRef]
- Danehey, C.; Wheeler, G.S.; Su, S.-C.H. The influence of quartz and calcite on the polymerization of methyltrimethoxysilane. In Proceedings of the 7th International Congress on Deterioration and Conservation of Stone, Lisbon, Portugal, 15–18 June 1992; pp. 1043–1052. [Google Scholar]
- Goins, E.; Wheeler, G.; Wypyski, M. Alkoxysilane film formation on quartz and calcite crystal surfaces. In Proceedings of the 8th International Congress on Deterioration and Conservation of Stone, Berlin, Germany, 30 September–4 November 1996; Volume 30, pp. 1255–1264. [Google Scholar]
- Naidu, S.; Liu, C.; Scherer, G.W. Hydroxyapatite-based consolidant and the acceleration of hydrolysis of silicate-based consolidants. J. Cult. Herit. 2015, 16, 94–101. [Google Scholar] [CrossRef]
- Sassoni, E.; Franzoni, E.; Pigino, B.; Scherer, G.W.; Naidu, S. Consolidation of calcareous and siliceous sandstones by hydroxyapatite: Comparison with a TEOS-based consolidant. J. Cult. Herit. 2013, 14, e103–e108. [Google Scholar] [CrossRef]

| Source | Sample | Consolidating Agent | Solvent | Catalyst | Additive |
|---|---|---|---|---|---|
| IPC 1 | SiGel-d | 50% oligomeric TEOS | 50% IPA 2 | dibutyltin dilaurate | - |
| IPC | SiGel SiO2-d | 50% oligomeric TEOS | 50% IPA | dibutyltin dilaurate | SiO2 |
| IPC | SiGel SiO2-octyl-d | 50% oligomeric TEOS | 50% IPA | dibutyltin dilaurate | SiO2-octyl |
| IPC | SiGel-oa | 50% oligomeric TEOS | 50% IPA | n-octylamine | - |
| IPC | SiGel SiO2-oa | 50% oligomeric TEOS | 50% IPA | n-octylamine | SiO2 |
| IPC | SiGel SiO2-octyl-oa | 50% oligomeric TEOS | 50% IPA | n-octylamine | SiO2-octyl |
| IPC | SiGel SiO2-methyl-oa | 50% oligomeric TEOS | 50% IPA | n-octylamine | SiO2-methyl |
| IPC | SiGel TiO2-oa | 50% oligomeric TEOS | 50% IPA | n-octylamine | TiO2 |
| IPC | SiGel ZnO-oa/d | 50% oligomeric TEOS | 50% IPA | n-octylamine + dibutyltin dilaurate | ZnO |
| IPC | HAP | 23% DAP | 77% H2O | - | Ca(OH)2 |
| Remmers | KSE OH | 75% oligomeric TEOS | 25% MEK | dibutyltin dilaurate | - |
| NanoPhos SA | SP FX | 20% TEOS | 80% IPA | n-octylamine | CaC2O4 |
| Steps | Exposure Period/ min | Irradiance 1/ W m−2 | Black Standard Temperature/ °C | Chamber Temperature/ °C | Relative Humidity/ % |
|---|---|---|---|---|---|
| 1. light | 40 | 0.55 | 70 ± 2 | 47 ± 2 | 50 ± 5 |
| 2. light and spray | 20 | 0.55 | 70 ± 2 | 47 ± 2 | 50 ± 5 |
| 3. light | 60 | 0.55 | 70 ± 2 | 47 ± 2 | 50 ± 5 |
| 4. dark and spray | 60 | 0.00 | 38 ± 2 | 38 ± 2 | 95 ± 5 |
| Xerogel | Micropore Volume/ cm3 g−1 | Mesopore Volume/ cm3 g−1 | BET Surface Area 1/ m2 g−1 | Pore Width/ nm | Total Porosity 2/ % |
|---|---|---|---|---|---|
| SiGel-d | 0.15 | 0.00 | * | 0.8 | 25 |
| SiGel-oa | 0.00 | 0.23 | 198 | 5.0 | 34 |
| SiGel-SiO2-oa | 0.00 | 0.24 | 187 | 6.6 | 35 |
| SiGel-SiO2-methyl-oa | 0.00 | 0.26 | 223 | 5.6 | 39 |
| SiGel-SiO2-octyl-oa | 0.00 | 0.24 | 209 | 5.4 | 35 |
| SiGel-TiO2-oa | 0.00 | 0.22 | 207 | 5.0 | 33 |
| SiGel-ZnO-oa/d | 0.14 | 0.17 | 55 | 1.9; 4–5 | 41 |
| KSE OH | 0.06 | 0.27 | 379 | 1.9; 4–5 | 38 |
| SP FX | 0.00 | 0.01 | 12 | 2–4 | 2 |
| Xerogel | Young’s Modulus/GPa | Hardness/ GPa | Contact Depth/nm |
|---|---|---|---|
| SiGel-d | 7.74 ± 0.52 | 1.04 ± 0.09 | 182.21 ± 9.35 |
| SiGel-oa | 4.49 ± 0.14 | 0.41 ± 0.02 | 297.88 ± 6.50 |
| SiGel-SiO2-methyl-oa | 3.83 ± 0.06 | 0.45 ± 0.01 | 285.80 ± 4.43 |
| SiGel-TiO2-oa | 4.72 ± 0.03 | 0.33 ± 0.00 | 349.96 ± 2.10 |
| KSE OH | 11.44 ± 0.25 | 1.32 ± 0.05 | 175.75 ± 4.34 |
| Sandstone Treatment | Consolidant Uptake/ wt.% | Dry Matter Yield/ wt.% | Water Uptake/ wt.% |
|---|---|---|---|
| Untreated | - | - | 11.60 ± 0.5 |
| SiGel-oa | 10.1 ± 0.5 | 4.0 ± 0.2 | 0.09 ± 0.01 |
| SiGel-SiO2-oa | 8.0 ± 0.4 | 4.3 ± 0.2 | 0.06 ± 0.01 |
| SiGel-SiO2-methyl-oa | 10.3 ± 0.5 | 4.2 ± 0.3 | 0.12 ± 0.02 |
| SiGel-SiO2-octyl-oa | 9.2 ± 0.4 | 4.8 ± 0.3 | 0.12 ± 0.01 |
| SiGel-TiO2-oa | 10.1 ± 0.6 | 4.8 ± 0.4 | 0.11 ± 0.01 |
| SiGel-ZnO-oa/d | 10.2 ± 0.6 | 3.7 ± 0.2 | 0.32 ± 0.02 |
| HAP | 13.9 ± 0.7 | 0.24 ± 0.03 | 9.8 ± 0.5 |
| HAP+Ca(OH)2 | 10.8 ± 0.5 | 0.31 ± 0.03 | 9.8 ± 0.6 |
| KSE OH | 10.6 ± 0.4 | 2.7 ± 0.11 | 2.3 ± 0.1 |
| SP FX | 9.6 ± 0.4 | 1.1 ± 0 05 | 9.5 ± 0.5 |
| Sandstone Treatment | BET Area Fresh 1/ m2 g−1 | BET Area Aged 2/ m2 g−1 | DRMS Fresh 3/ N | DRMS Aged 4/ N | ∆ DRMS 5/ % |
|---|---|---|---|---|---|
| Untreated | 0.56 | - | 9.7 ± 0.5 | 3.2 ± 0.2 | −67 |
| SiGel-oa | 0.05 | 5.90 | 11.6 ± 0.8 | 9.2 ± 1.2 | −21 |
| SiGel SiO2-oa | 0.05 | 16.77 | 15.5 ± 0.8 | 12.1 ± 0.8 | −22 |
| SiGel SiO2-octyl-oa | 0.15 | 8.41 | 10.5 ± 0.7 | 4.0 ± 1.0 | −62 |
| SiGel SiO2-methyl-oa | 0.06 | 7.42 | 17.3 ± 1.9 | 12.7 ± 1.0 | −27 |
| SiGel TiO2-oa | 0.02 | 0.17 | 23.3 ± 2.5 | 18.1 ± 1.4 | −22 |
| SiGel ZnO-oa/d | 5.10 * | 10.60 * | 54.9 ± 1.7 | 60.9 ± 2.2 | +11 |
| KSE OH | 0.46 | 2.54 | 25.2 ± 1.4 | 18.5 ± 1.5 | −27 |
| SP FX | 2.10 * | 3.00 | 22.7 ± 1.6 | 16.4 ± 1.4 | −28 |
| HAP | 0.23 | 0.42 | 18.5 ± 1.3 | 18.1 ± 1.6 | −2 |
| HAP+Ca(OH)2 | 0.23 | 0.73 | 17.8 ± 1.4 | 17.6 ± 1.5 | −1 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Remzova, M.; Zouzelka, R.; Lukes, J.; Rathousky, J. Potential of Advanced Consolidants for the Application on Sandstone. Appl. Sci. 2019, 9, 5252. https://doi.org/10.3390/app9235252
Remzova M, Zouzelka R, Lukes J, Rathousky J. Potential of Advanced Consolidants for the Application on Sandstone. Applied Sciences. 2019; 9(23):5252. https://doi.org/10.3390/app9235252
Chicago/Turabian StyleRemzova, Monika, Radek Zouzelka, Jaroslav Lukes, and Jiri Rathousky. 2019. "Potential of Advanced Consolidants for the Application on Sandstone" Applied Sciences 9, no. 23: 5252. https://doi.org/10.3390/app9235252
APA StyleRemzova, M., Zouzelka, R., Lukes, J., & Rathousky, J. (2019). Potential of Advanced Consolidants for the Application on Sandstone. Applied Sciences, 9(23), 5252. https://doi.org/10.3390/app9235252






