Epigallocatechin-3-Gallate (EGCG), An Active Constituent of Green Tea: Implications in the Prevention of Liver Injury Induced by Diethylnitrosamine (DEN) in Rats
Abstract
Featured Application
Abstract
1. Introduction
2. Materials and Methods
2.1. Animal Model and Sample Collection
2.2. Determination of Liver Function (ALT, ALP, and AST) Enzymes, SOD, CAT, and GPx Antioxidant Enzymes and Total Antioxidant Capacity
2.3. Determination of C-Reactive Protein, Interleukin-6 (IL-6), and Tumor necrosis factor-α (TNF-α)
2.4. Histopathological Analysis
2.5. Immunohistochemical Analysis
Scoring Method
2.6. TUNEL Assay
2.7. Transmission Electron Microscopy (TEM)
2.8. Cell Cycle Analysis
2.9. Statistical Analysis
3. Results
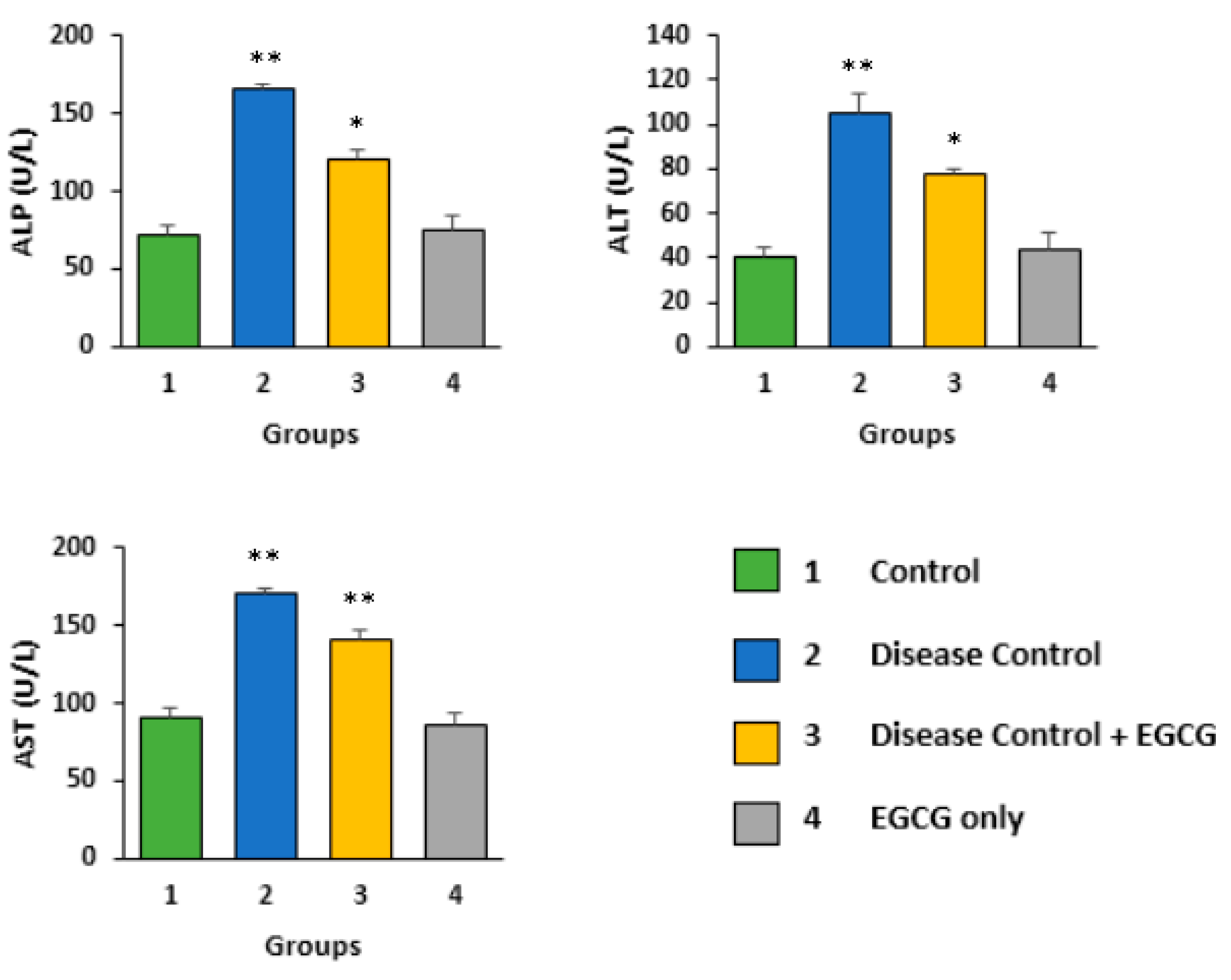
3.1. EGCG Reduces the Serum Level of Biochemical Enzymes
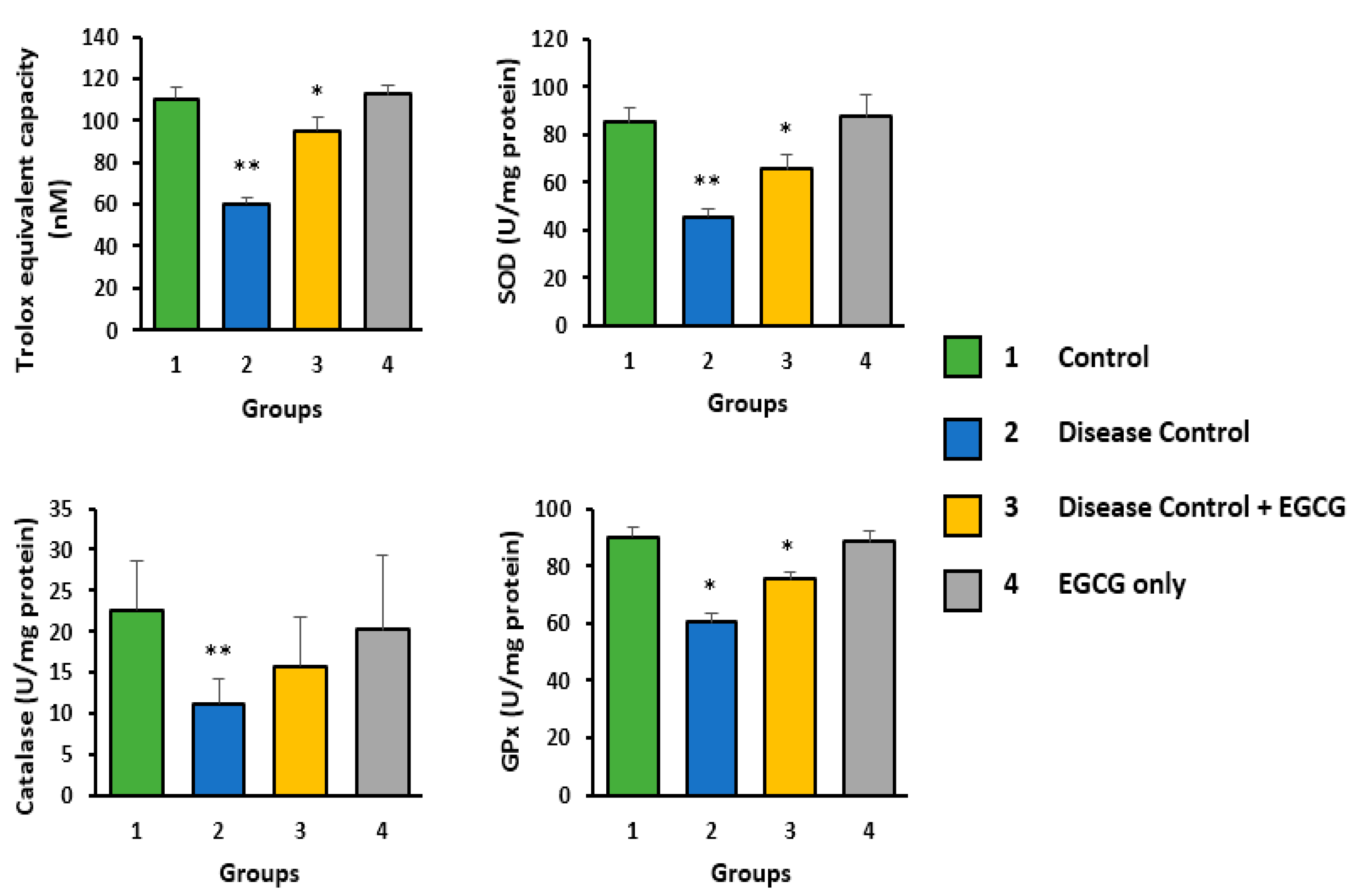
3.2. Antioxidant Activity of EGCG
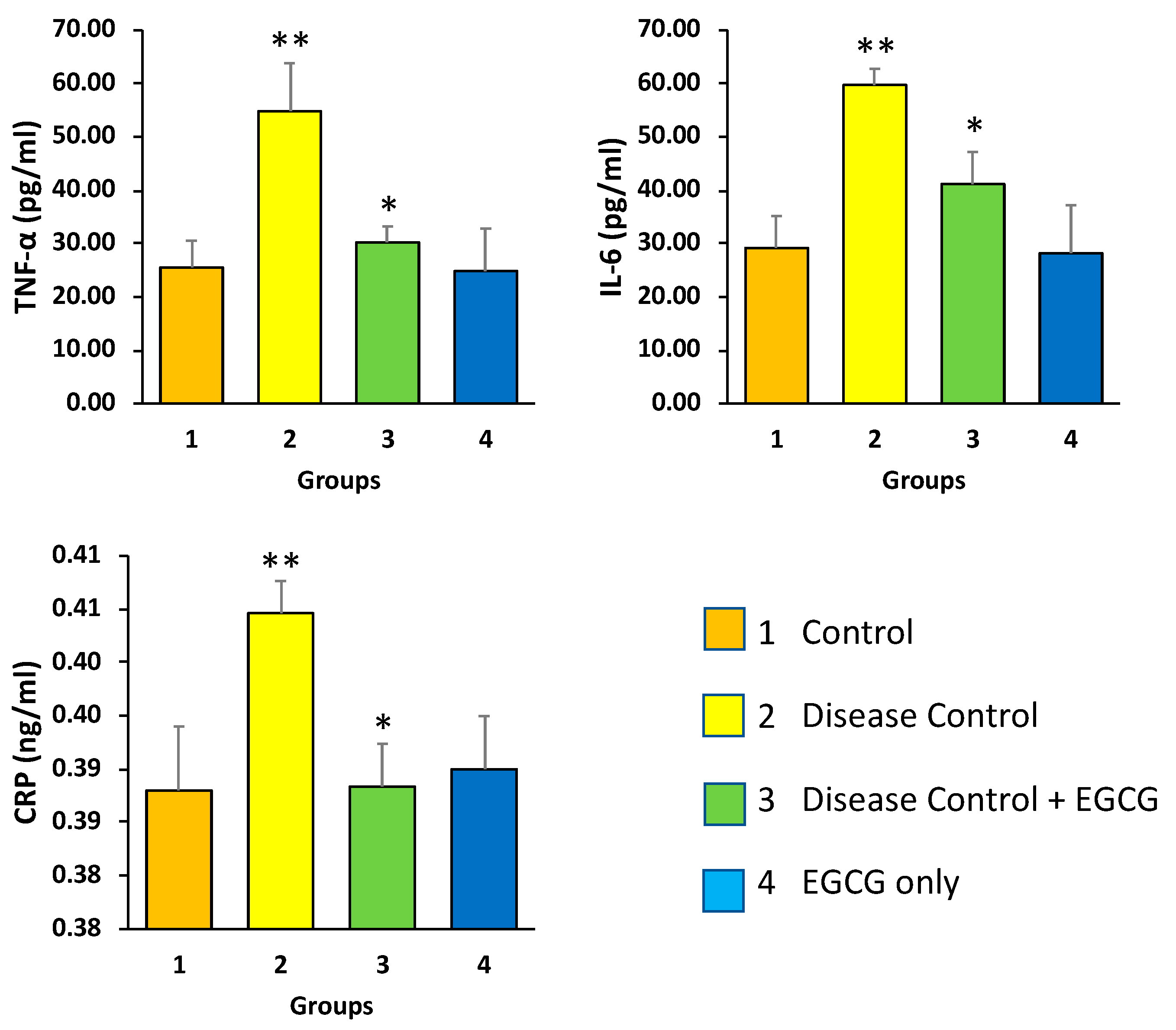
3.3. EGCG Reduces the Serum Levels of CRP and Pro-Inflammatory Mediators—TNF-α and IL-6
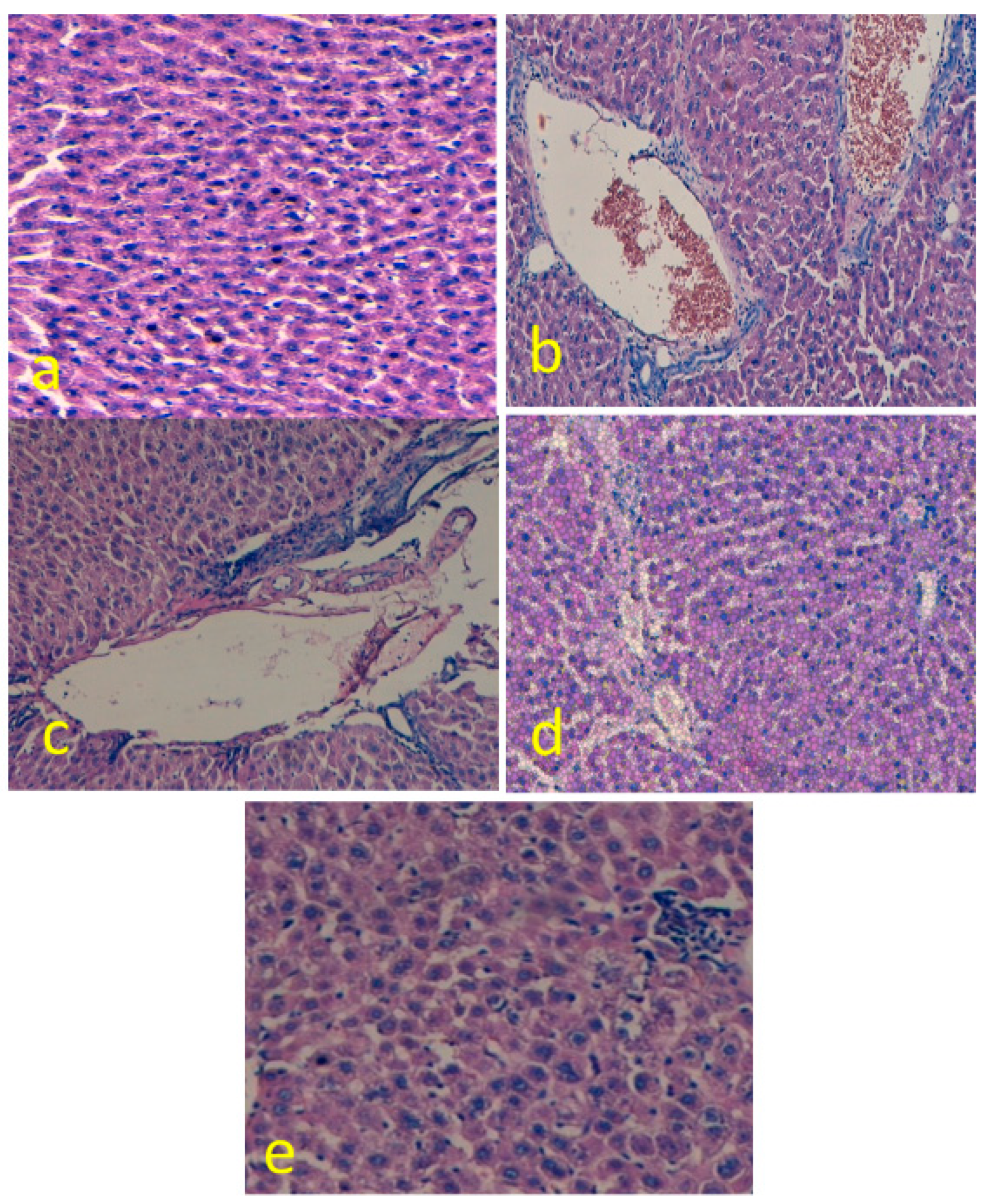
3.4. EGCG Reduces Hepatic Histological Alterations
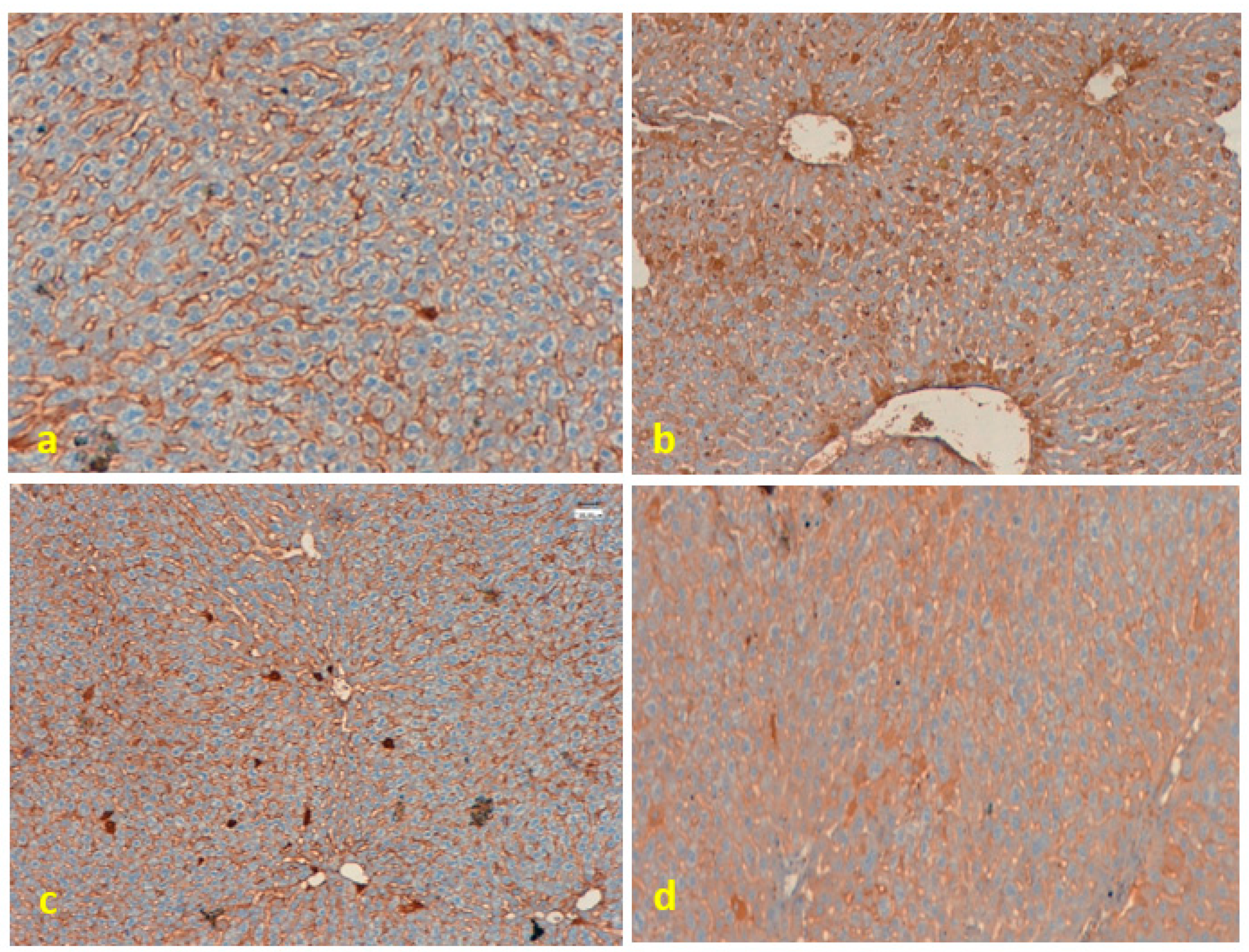
3.5. Immunohistochemical Analysis
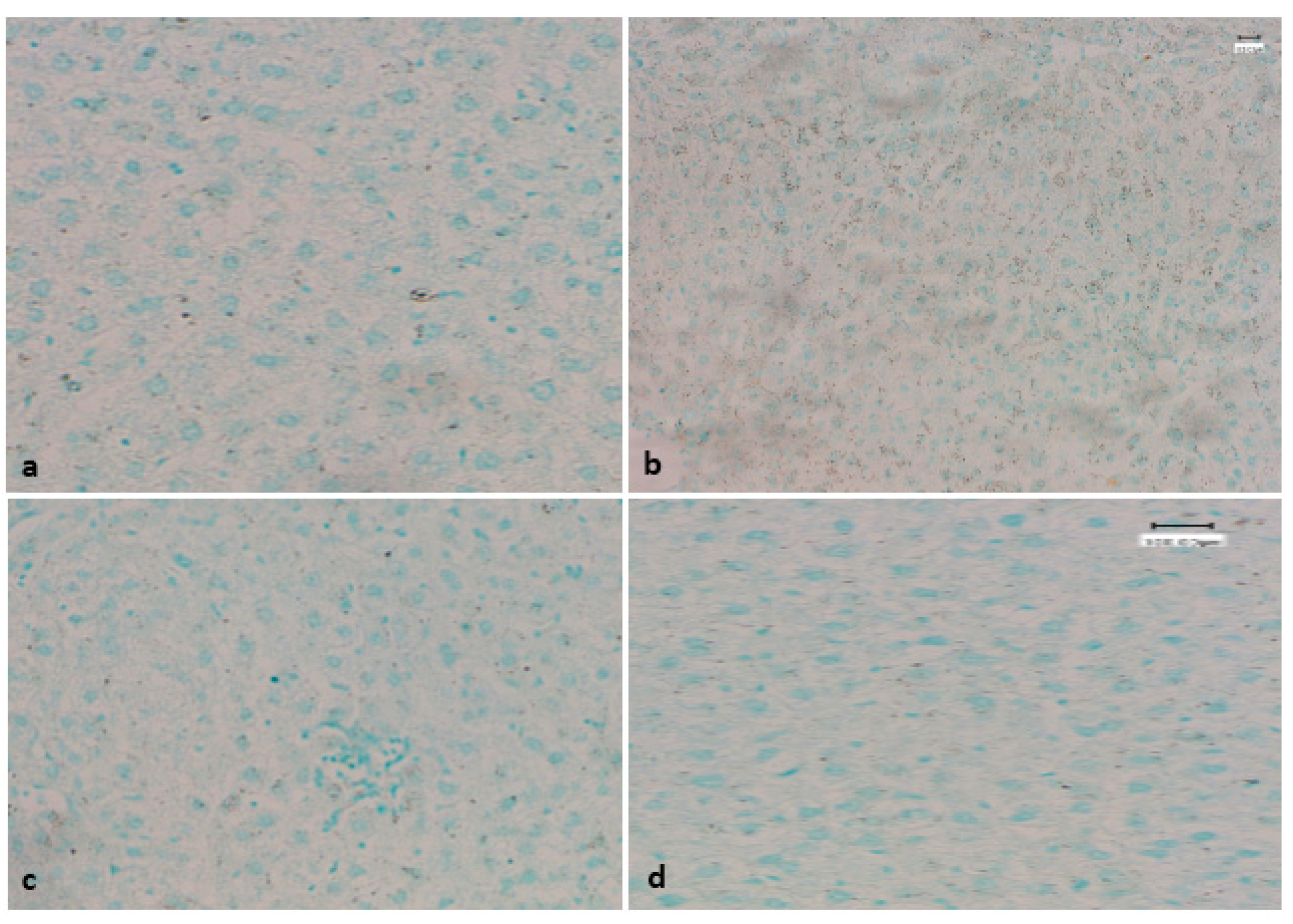
3.6. Evaluation of Apoptotic Bodies Via TUNEL Assay
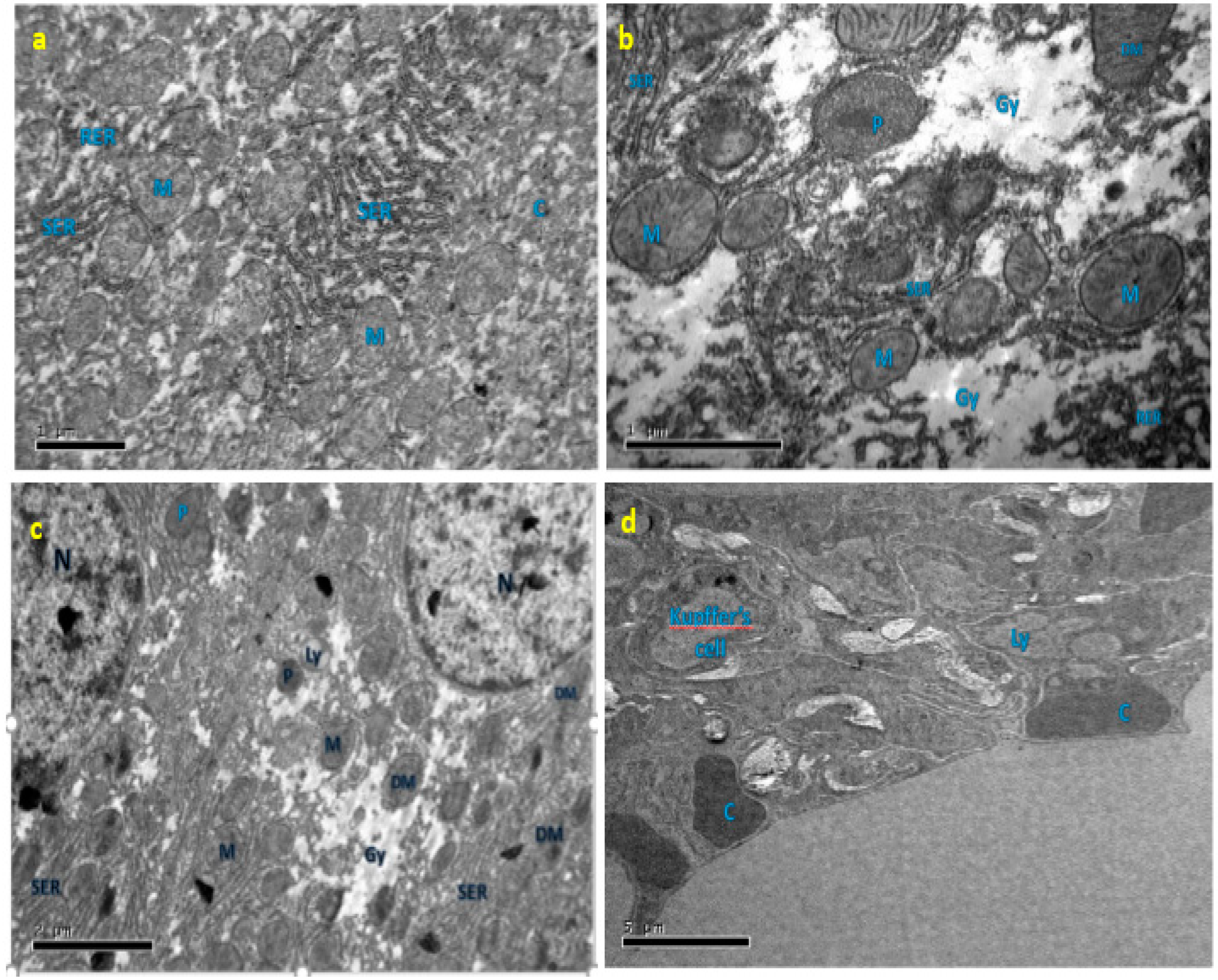
3.7. Effect of EGCG on Ultrastructure Changes of Liver
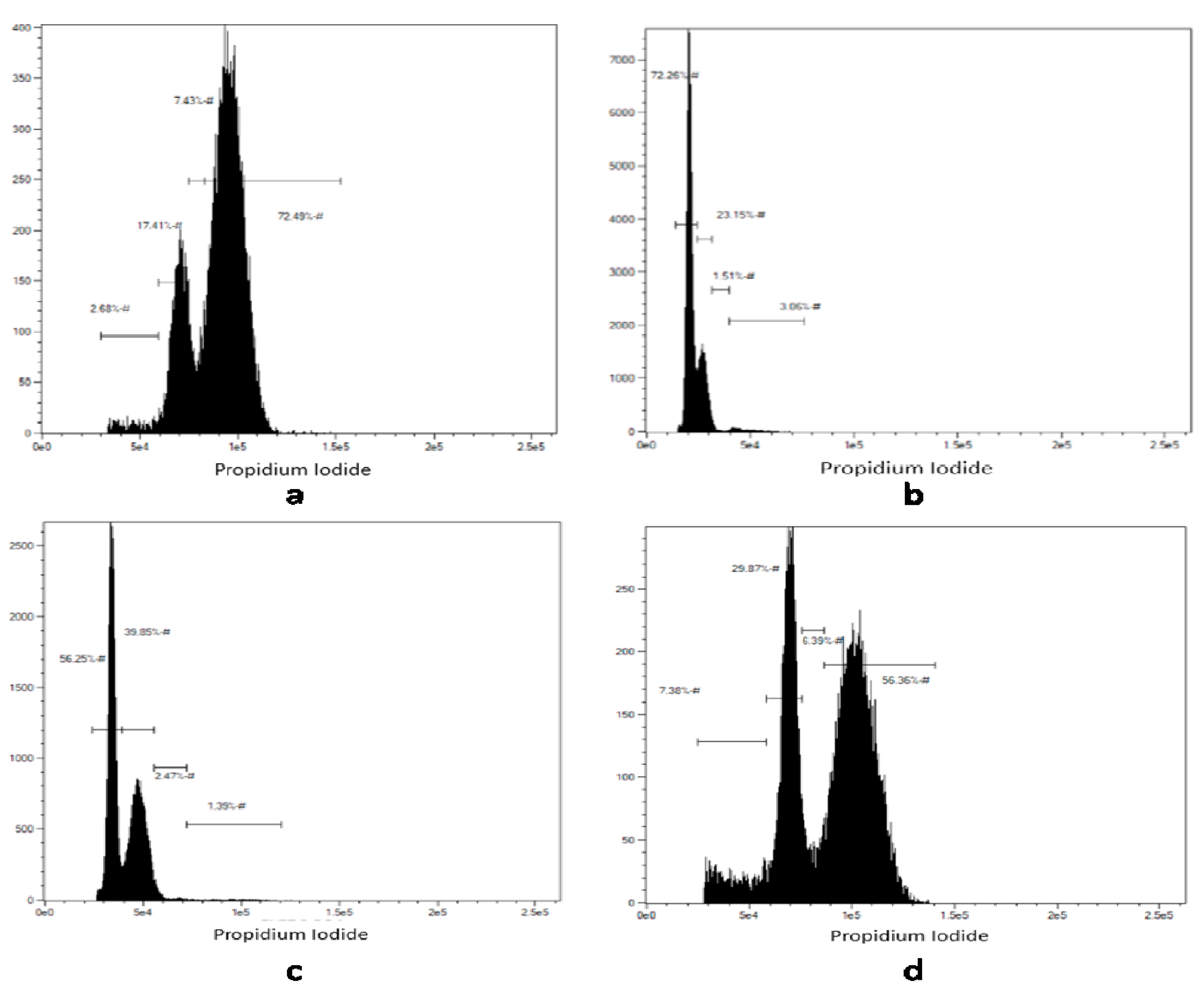
3.8. Effect of EGCG on the Cell Cycle
4. Discussion
Author Contributions
Funding
Conflicts of Interest
Data Availability
References
- Samuel, A.J.S.J.; Mohan, S.; Chellappan, D.K.; Kalusalingam, A.; Ariamuthu, S. Hibiscus vitifolius (Linn.) root extracts shows potent protective action against anti-tubercular drug induced hepatotoxicity. J. Ethnopharmacol. 2012, 141, 396–402. [Google Scholar] [CrossRef] [PubMed]
- Mishra, B.B.; Tiwari, V.K. Natural products: An evolving role in future drug discovery. Eur. J. Med. Chem. 2011, 46, 4769–4807. [Google Scholar] [CrossRef] [PubMed]
- Gao, M.; Nettles, R.E.; Belema, M.; Snyder, L.B.; Nguyen, V.N.; Fridell, R.A.; Serrano-Wu, M.H.; Langley, D.R.; Sun, J.H.; O’Boyle, D.R., II; et al. Chemical genetics strategy identifies an HCV NS5A inhibitor with a potent clinical effect. Nature 2010, 465, 96. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, N.; Ghosh, R.; Mandal, V.; Mandal, S.C. Recent advances in herbal medicine for treatment of liver diseases. Pharm. Biol. 2011, 49, 970–988. [Google Scholar] [CrossRef] [PubMed]
- Tipoe, G.L.; Leung, T.M.; Liong, E.C.; Lau, T.Y.H.; Fung, M.L.; Nanji, A.A. Epigallocatechin-3-gallate (EGCG) reduces liver inflammation, oxidative stress and fibrosis in carbon tetrachloride (CCl4)-induced liver injury in mice. Toxicology 2010, 273, 45–52. [Google Scholar] [CrossRef] [PubMed]
- Louvet, A.; Mathurin, P. Alcoholic liver disease: Mechanisms of injury and targeted treatment. Nat. Rev. Gastroenterol. Hepatol. 2015, 12, 231. [Google Scholar] [CrossRef] [PubMed]
- Björnsson, E. Drug-induced liver injury in clinical practice. Aliment. Pharmacol. Ther. 2010, 32, 3–13. [Google Scholar] [CrossRef] [PubMed]
- Park, D.H.; Shin, J.W.; Park, S.K.; Seo, J.N.; Li, L.; Jang, J.J.; Lee, M.J. Diethylnitrosamine (DEN) induces irreversible hepatocellular carcinogenesis through overexpression of G1/S-phase regulatory proteins in rat. Toxicol. Lett. 2009, 191, 321–326. [Google Scholar] [CrossRef] [PubMed]
- Magee, P.N.; Barnes, J. The production of malignant primary hepatic tumours in the rat by feeding dimethylnitrosamine. Br. J. Cancer 1956, 10, 114. [Google Scholar] [CrossRef] [PubMed]
- Verna, L.; Whysner, J.; Williams, G.M. N-nitrosodiethylamine mechanistic data and risk assessment: Bioactivation, DNA-adduct formation, mutagenicity, and tumor initiation. Pharmacol. Ther. 1996, 71, 57–81. [Google Scholar] [CrossRef]
- Paterson, I.; Anderson, E.A. The renaissance of natural products as drug candidates. Science 2005, 310, 451–453. [Google Scholar] [CrossRef] [PubMed]
- Teuten, E.L.; Xu, L.; Reddy, C.M. Two abundant bioaccumulated halogenated compounds are natural products. Science 2005, 307, 917–920. [Google Scholar] [CrossRef] [PubMed]
- Rollinger, J.; Langer, T.; Stuppner, H. Strategies for efficient lead structure discovery from natural products. Curr. Med. Chem. 2006, 13, 1491–1507. [Google Scholar] [CrossRef] [PubMed]
- Majeed, R.; Reddy, M.V.; Chinthakindi, P.K.; Sangwan, P.L.; Hamid, A.; Chashoo, G.; Saxena, A.K.; Koul, S. Bakuchiol derivatives as novel and potent cytotoxic agents: A report. Eur. J. Med. Chem. 2012, 49, 55–67. [Google Scholar] [CrossRef] [PubMed]
- Beecher, G.R.; Warden, B.A.; Merken, H. Analysis of tea polyphenols. Proc. Soc. Exp. Biol. Med. 1999, 220, 267–270. [Google Scholar] [PubMed]
- Salah, N.; Miller, N.J.; Paganga, G.; Tijburg, L.; Bolwell, G.P.; Riceevans, C. Polyphenolic flavanols as scavengers of aqueous phase radicals and as chain-breaking antioxidants. Arch. Biochem. Biophys. 1995, 322, 339–346. [Google Scholar] [CrossRef] [PubMed]
- Isbrucker, R.A.; Bausch, J.; Edwards, J.A.; Wolz, E. Safety studies on epigallocatechin gallate (EGCG) preparations. Part 1: Genotoxicity. Food Chem. Toxicol. 2006, 44, 626–635. [Google Scholar] [CrossRef] [PubMed]
- Lambert, J.D.; Yang, C.S. Mechanisms of cancer prevention by tea constituents. J. Nutr. 2003, 133, 3262S–3267S. [Google Scholar] [CrossRef] [PubMed]
- Wolfram, S.; Wang, Y.; Thielecke, F. Anti-obesity effects of green tea: From bedside to bench. Mol. Nutr. Food Res. 2006, 50, 176–187. [Google Scholar] [CrossRef] [PubMed]
- Frei, B.; Higdon, J.V. Antioxidant activity of tea polyphenols in vivo: Evidence from animal studies. J. Nutr. 2003, 133, 3275S–3284S. [Google Scholar] [CrossRef] [PubMed]
- Kundu, J.K.; Na, H.K.; Chun, K.S.; Kim, Y.K.; Lee, S.J.; Lee, S.S.; Lee, O.S.; Sim, Y.C.; Surh, Y.J. Inhibition of Phorbol Ester–Induced COX-2 Expression by Epigallocatechin Gallate in Mouse Skin and Cultured Human Mammary Epithelial Cells. J. Nutr. 2003, 133, 3805S–3810S. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.; Oz, H.S.; Barve, S.; De Villiers, W.J.; McClain, C.J.; Varilek, G.W. The green tea polyphenol (−)-epigallocatechin-3-gallate blocks nuclear factor-κB activation by inhibiting IκB kinase activity in the intestinal epithelial cell line IEC-6. Mol. Pharmacol. 2001, 60, 528–533. [Google Scholar] [PubMed]
- Bancroft, J.D.; Gamble, M. Theory and Practice of Histological Techniques, 6th ed.; Churchill Livingstone, Elsevier: London, UK, 2008. [Google Scholar]
- Rahmani, A.; Alzohairy, M.; Khadri, H.; Mandal, A.K.; Rizvi, M.A. Expressional evaluation of vascular endothelial growth factor (VEGF) protein in urinary bladder carcinoma patients exposed to cigarette smoke. Int. J. Clin. Exp. Pathol. 2012, 5, 195–200. [Google Scholar] [PubMed]
- Rahmani, A.H.; Babiker, A.Y.; AlWanian, W.M.; Elsiddig, S.A.; Faragalla, H.E.; Aly, S.M. Association of cytokeratin and vimentin protein in the genesis of transitional cell carcinoma of urinary bladder patients. Dis. Mark. 2015, 2015, 204759. [Google Scholar] [CrossRef] [PubMed]
- Singh, B.N.; Shankar, S.; Srivastava, R.K. Green tea catechin, epigallocatechin-3-gallate (EGCG): Mechanisms, perspectives and clinical applications. Biochem. Pharmacol. 2011, 82, 1807–1821. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.S.; Wang, X. Green tea and cancer prevention. Nutr. Cancer 2010, 62, 931–937. [Google Scholar] [CrossRef] [PubMed]
- Thangapandiyan, S.; Miltonprabu, S. Epigallocatechin gallate effectively ameliorates fluoride-induced oxidative stress and DNA damage in the liver of rats. Can. J. Physiol. Pharmacol. 2013, 91, 528–537. [Google Scholar] [CrossRef] [PubMed]
- Moravcová, A.; Cervinkova, Z.; Kucera, O.; Mezera, V.; Lotková, H. Antioxidative effect of epigallocatechin gallate against D-galactosamine-induced injury in primary culture of rat hepatocytes. Acta Med. 2014, 57, 3–8. [Google Scholar]
- Yao, H.T.; Yang, Y.C.; Chang, C.H.; Yang, H.T.; Yin, M.C. Protective effects of (-)-epigallocatechin-3-gallate against acetaminophen-induced liver injury in rats. Biomedicine 2015, 5, 15. [Google Scholar] [CrossRef] [PubMed]
- Tak, E.; Park, G.C.; Kim, S.H.; Jun, D.Y.; Lee, J.; Hwang, S.; Song, G.W.; Lee, S.G. Epigallocatechin-3-gallate protects against hepatic ischaemia-reperfusion injury by reducing oxidative stress and apoptotic cell death. J. Int. Med. Res. 2016, 44, 1248–1262. [Google Scholar] [CrossRef] [PubMed]
- Karatayli, E.; Hall, R.A.; Weber, S.N.; Dooley, S.; Lammert, F. Effect of alcohol on the interleukin 6-mediated inflammatory response in a new mouse model of acute-on-chronic liver injury. Biochim. Biophys. Acta Mol. Basis Dis. 2019, 1865, 298–307. [Google Scholar] [CrossRef] [PubMed]
- Farouk, S.; Sabet, S.; Zahra, F.A.A.; El-Ghor, A.A. Bone marrow derived-mesenchymal stem cells downregulate IL17A dependent IL6/STAT3 signaling pathway in CCl4-induced rat liver fibrosis. PLoS ONE 2018, 13, e0206130. [Google Scholar] [CrossRef] [PubMed]
- Jamal, M.H.; Ali, H.; Dashti, A.; Al-Abbad, J.; Dashti, H.; Mathew, C.; Al-Ali, W.; Asfar, S. Effect of epigallocatechin gallate on uncoupling protein 2 in acute liver injury. Int. J. Clin. Exp. Pathol. 2015, 8, 649–654. [Google Scholar] [PubMed]
- Oliva, J.; Bardag-Gorce, F.; Tillman, B.; French, S.W. Protective effect of quercetin, EGCG, catechin and betaine against oxidative stress induced by ethanol in vitro. Exp. Mol. Pathol. 2011, 90, 295–299. [Google Scholar] [CrossRef] [PubMed]
- Milella, M.; Falcone, I.; Conciatori, F.; Cesta Incani, U.; Del Curatolo, A.; Inzerilli, N.; Nuzzo, C.; Vaccaro, V.; Vari, S.; Cognetti, F.; et al. PTEN: Multiple Functions in Human Malignant Tumors. Front. Oncol. 2015, 5, 24. [Google Scholar] [CrossRef] [PubMed]
- Rahmani, A.; Alzohairy, M.; Babiker, A.Y.; Rizvi, M.A.; Elkarimahmad, H.G. Clinicopathological significance of PTEN and bcl2 expressions in oral squamous cell carcinoma. Int. J. Clin. Exp. Pathol. 2012, 5, 965–971. [Google Scholar] [PubMed]
- Su, S.; Luo, D.; Liu, X.; Liu, J.; Peng, F.; Fang, C.; Li, B. miR-494 up-regulates the PI3K/Akt pathway via targetting PTEN and attenuates hepatic ischemia/reperfusion injury in a rat model. Biosci. Rep. 2017, 37. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.; Tian, Y.; Xia, J.; Wu, X.; Yang, Y.; Li, X.; Huang, C.; Meng, X.; Ma, T.; Li, J. The role of PTEN in regulation of hepatic macrophages activation and function in progression and reversal of liver fibrosis. Toxicol. Appl. Pharmacol. 2017, 317, 51–62. [Google Scholar] [CrossRef] [PubMed]
- Chang, W.; He, W.; Li, P.P.; Song, S.S.; Yuan, P.F.; Lu, J.T.; Wei, W. Protective effects of Celastrol on diethylnitrosamine-induced hepatocellular carcinoma in rats and its mechanisms. Eur. J. Pharmacol. 2016, 784, 173–180. [Google Scholar] [CrossRef] [PubMed]
- Sun, Q.; Long, Z.; Wu, H.; Liu, Y.; Wang, L.; Zhang, X.; Wang, X.; Hai, C. Effect of alcohol on diethylnitrosamine-induced hepatic toxicity: Critical role of ROS, lipid accumulation, and mitochondrial dysfunction. Exp. Toxicol. Pathol. 2015, 67, 491–498. [Google Scholar] [CrossRef] [PubMed]
- James, K.D.; Kennett, M.J.; Lambert, J.D. Potential role of the mitochondria as a target for the hepatotoxic effects of (-)-epigallocatechin-3-gallate in mice. Food Chem. Toxicol. 2018, 111, 302–309. [Google Scholar] [CrossRef] [PubMed]
- Shen, K.; Feng, X.; Su, R.; Xie, H.; Zhou, L.; Zheng, S. Epigallocatechin 3-gallate ameliorates bile duct ligation induced liver injury in mice by modulation of mitochondrial oxidative stress and inflammation. PLoS ONE 2015, 10, e0126278. [Google Scholar] [CrossRef] [PubMed]
- Mezera, V.; Endlicher, R.; Kucera, O.; Sobotka, O.; Drahota, Z.; Cervinkova, Z. Effects of Epigallocatechin Gallate on Tert-Butyl Hydroperoxide-Induced Mitochondrial Dysfunction in Rat Liver Mitochondria and Hepatocytes. Oxid. Med. Cell. Longev. 2016, 2016, 7573131. [Google Scholar] [CrossRef] [PubMed]
- Luo, X.; Kraus, W.L. On PAR with PARP: Cellular stress signaling through poly (ADP-ribose) and PARP-1. Genes Dev. 2012, 26, 417–432. [Google Scholar] [CrossRef] [PubMed]
- Jiang, F.; Jiang, R.; Zhu, X.; Zhang, X.; Zhan, Z. Genipin inhibits TNF-alpha-induced vascular smooth muscle cell proliferation and migration via induction of HO-1. PLoS ONE 2013, 8, e74826. [Google Scholar]
- Liu, P.L.; Liu, J.T.; Kuo, H.F.; Chong, I.W.; Hsieh, C.C. Epigallocatechin gallate attenuates proliferation and oxidative stress in human vascular smooth muscle cells induced by interleukin-1beta via heme oxygenase-1. Mediat. Inflamm. 2014, 2014, 523684. [Google Scholar] [CrossRef] [PubMed]

| Experimental Group | Group Number | Treatment | Number of Animals Per Group (n) |
|---|---|---|---|
| Negative control | 1 | Normal rats administered with vehicle solution | 08 |
| Disease control | 2 | DEN administered via oral gavage | 08 |
| Treatment | 3 | DEN and EGCG | 08 |
| Treatment | 4 | EGCG with vehicle solution | 08 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
A. Almatroodi, S.; A. Alsahli, M.; Alharbi, H.M.; Khan, A.A.; Husain Rahmani, A. Epigallocatechin-3-Gallate (EGCG), An Active Constituent of Green Tea: Implications in the Prevention of Liver Injury Induced by Diethylnitrosamine (DEN) in Rats. Appl. Sci. 2019, 9, 4821. https://doi.org/10.3390/app9224821
A. Almatroodi S, A. Alsahli M, Alharbi HM, Khan AA, Husain Rahmani A. Epigallocatechin-3-Gallate (EGCG), An Active Constituent of Green Tea: Implications in the Prevention of Liver Injury Induced by Diethylnitrosamine (DEN) in Rats. Applied Sciences. 2019; 9(22):4821. https://doi.org/10.3390/app9224821
Chicago/Turabian StyleA. Almatroodi, Saleh, Mohammed A. Alsahli, Hanan Marzoq Alharbi, Amjad Ali Khan, and Arshad Husain Rahmani. 2019. "Epigallocatechin-3-Gallate (EGCG), An Active Constituent of Green Tea: Implications in the Prevention of Liver Injury Induced by Diethylnitrosamine (DEN) in Rats" Applied Sciences 9, no. 22: 4821. https://doi.org/10.3390/app9224821
APA StyleA. Almatroodi, S., A. Alsahli, M., Alharbi, H. M., Khan, A. A., & Husain Rahmani, A. (2019). Epigallocatechin-3-Gallate (EGCG), An Active Constituent of Green Tea: Implications in the Prevention of Liver Injury Induced by Diethylnitrosamine (DEN) in Rats. Applied Sciences, 9(22), 4821. https://doi.org/10.3390/app9224821





