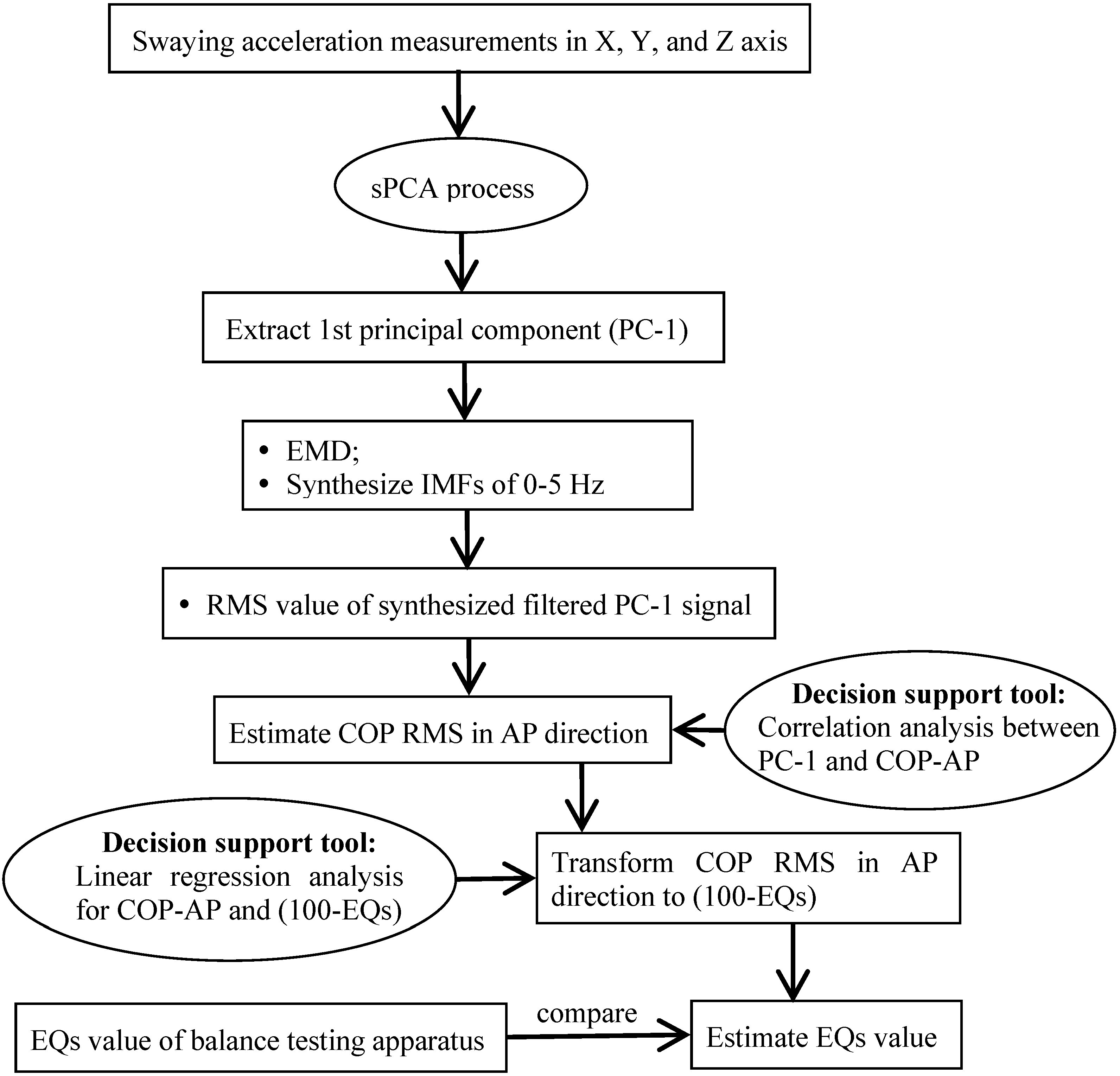
3.1. Signal Separation and Spectrum Analysis
The major difference between the COP and acceleration measurements is the dimension representation. The COP signals, which are recorded through the force plate, consist of the two independent components (in X and Y directions) that are perpendicular to the gravitational direction. According to the SOT, the COP signal in X-direction is defined as the medial-lateral (ML) component and the Y-component is defined as the anterior-posterior (AP) direction. The acceleration signals that are measured by the tri-axial accelerometer consist of the three components in the typical X-Y-Z directions of the Cartesian coordinate system. Since the accelerometer is bound on the human body’s waist and may tilt with human body swaying, the X-Y-Z directions of the tri-axial accelerometer are not fixed in the measurement process.
Through observing the ways that the examinees adjust their posture for balance purposes, both the COP and the acceleration signals have obvious non-stationarity characteristics. The EMD approach that was proposed by Huang et al. [
12] is an adaptive data analysis method and can be utilized to separate the non-stationary signals. The EMD process can be simply expressed as:
where,
x(
t) represents any complicated non-stationary signal,
ck(
t) is the
k-th intrinsic mode function (IMF) of the signal
x(
t) and
rm(
t) is the final residue which can be a constant or the signal mean trend. Based on the concept of EMD, each IMF component satisfies the following conditions [
12]: (1) The number of extrema and the number of zero-crossings must be either equal or differ at most by one in the whole data set, and (2) At any point, the mean value of the envelope defined by the local maxima and the envelope defined by the local minima is zeros.
The measurements of COP and acceleration were first decomposed into the IMFs by the EMD method. With the EMD method, the non-stationary COP and acceleration signals can be separated into the independent signal components of different frequency bands. In the experimental process, on the other hand, the external noises or disturbances that are mixed in the measurements may inevitably interfere with the analysis of the COP and the acceleration signals; therefore, it is crucial to remove the uncorrelated signal components from the measurements of COP and acceleration, and reserve the signal components that contain the information correlated to the balancing ability of the human body.
By taking the Hilbert transform of the signal components (IMFs), the analytical signal of
ck(
t) can be formed as:
where,
represents the Hilbert transform of
ck(
t). The time-dependent amplitude,
Ak(
t), time-dependent phase,
ϕk(
t), and instantaneous frequency,
ωk(
t), of
ck(
t) can be formulated as:
Therefore, the time-frequency-amplitude distributions of the non-stationary COP and acceleration signals can be expressed as:
In order to clearly observe the energy distributions of measurements in the frequency domain, the marginal spectrum of the COP and acceleration signals can be formulated as:
where,
Hk(
ω,
t) represents the time-frequency distribution of the
k-th IMF. The signal compositions and characteristics can then be observed and analyzed within the different frequency scales.
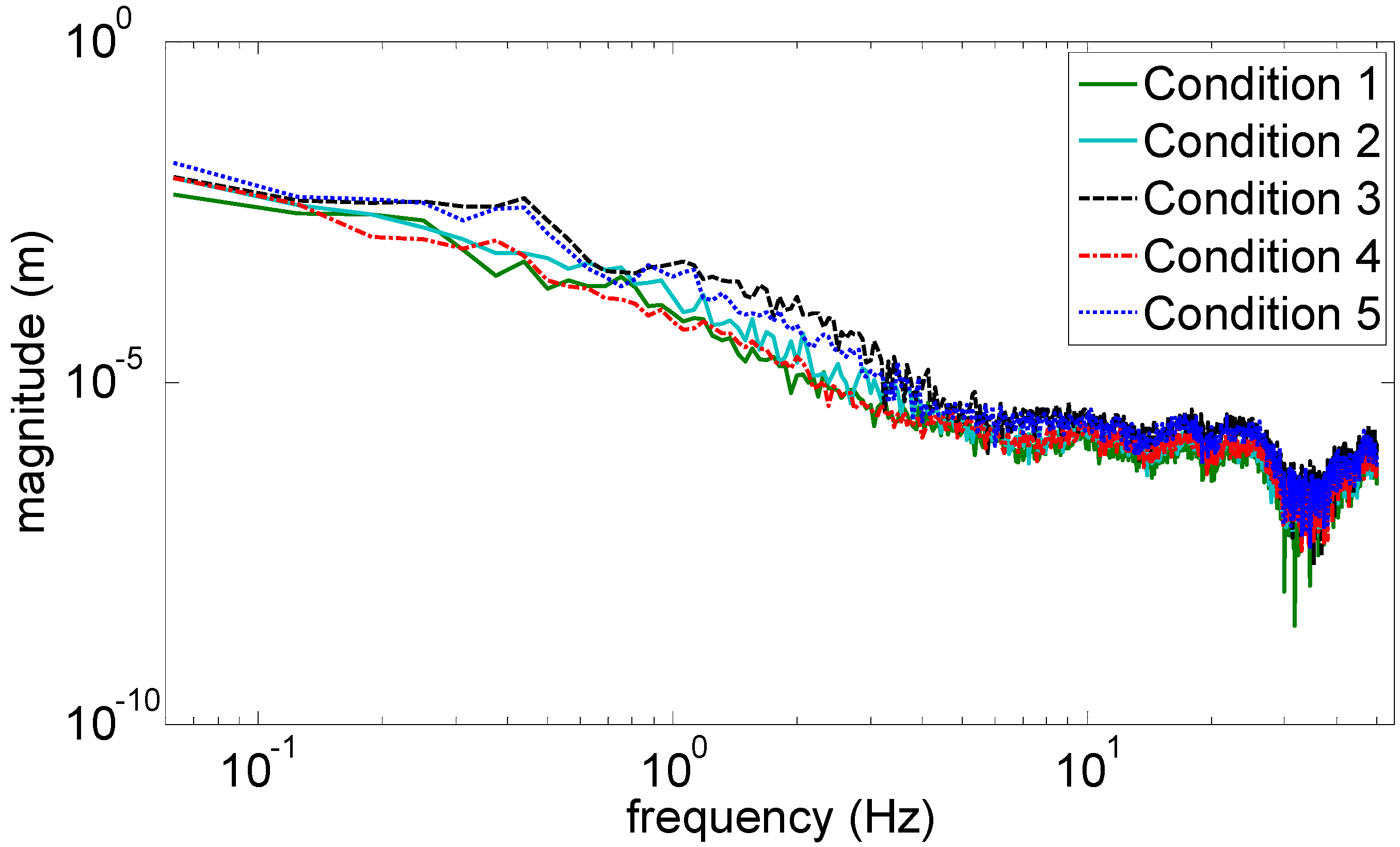
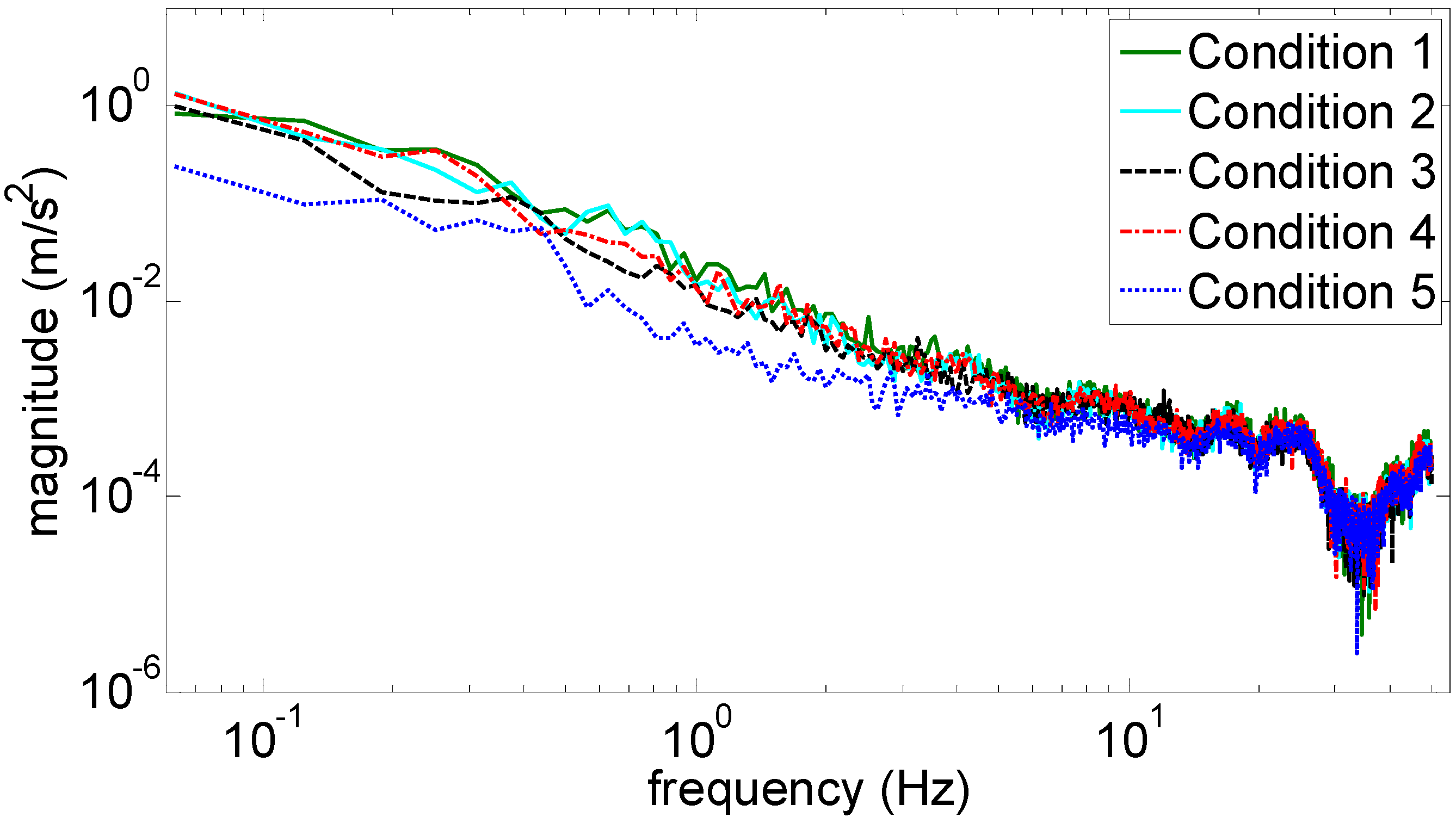
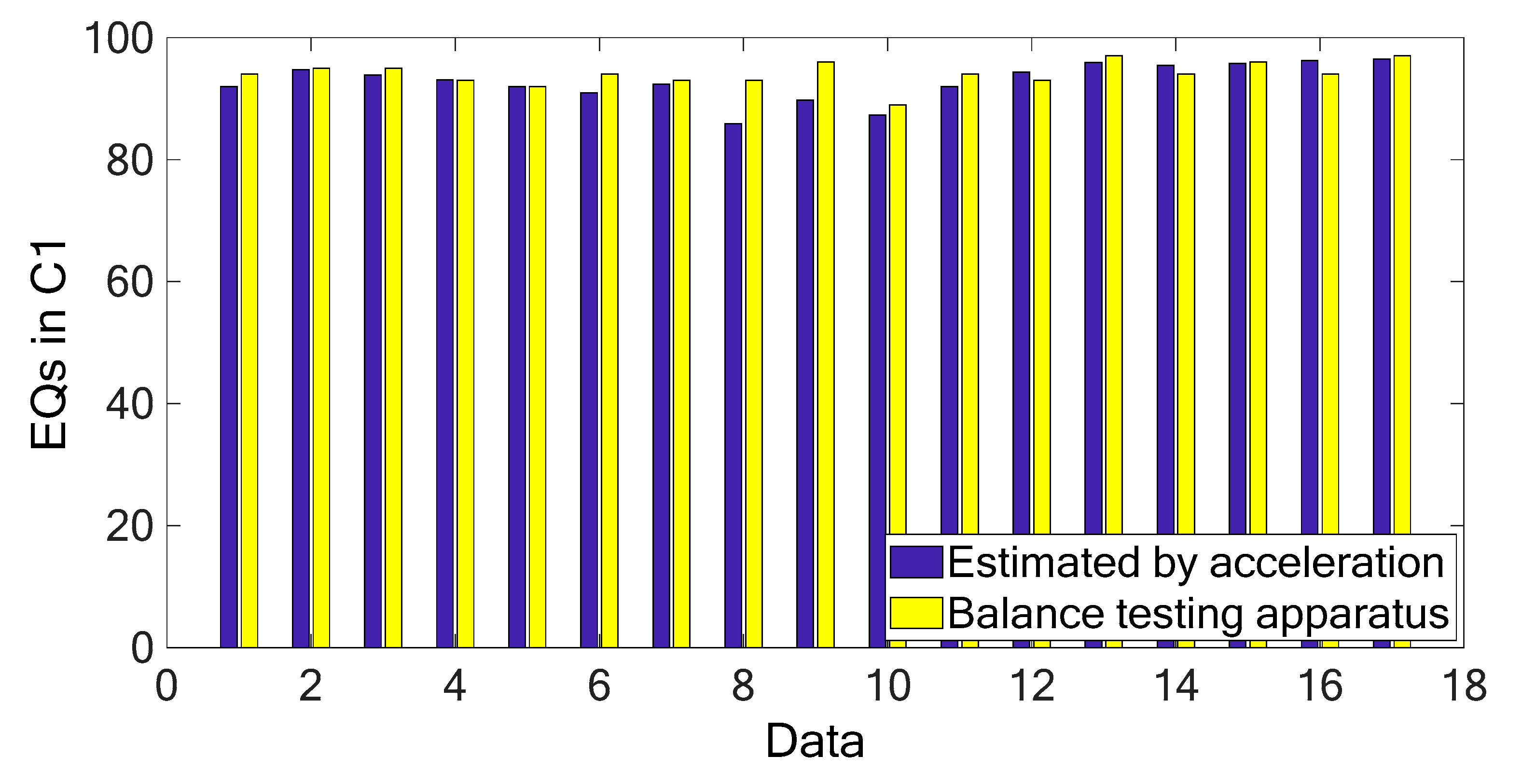
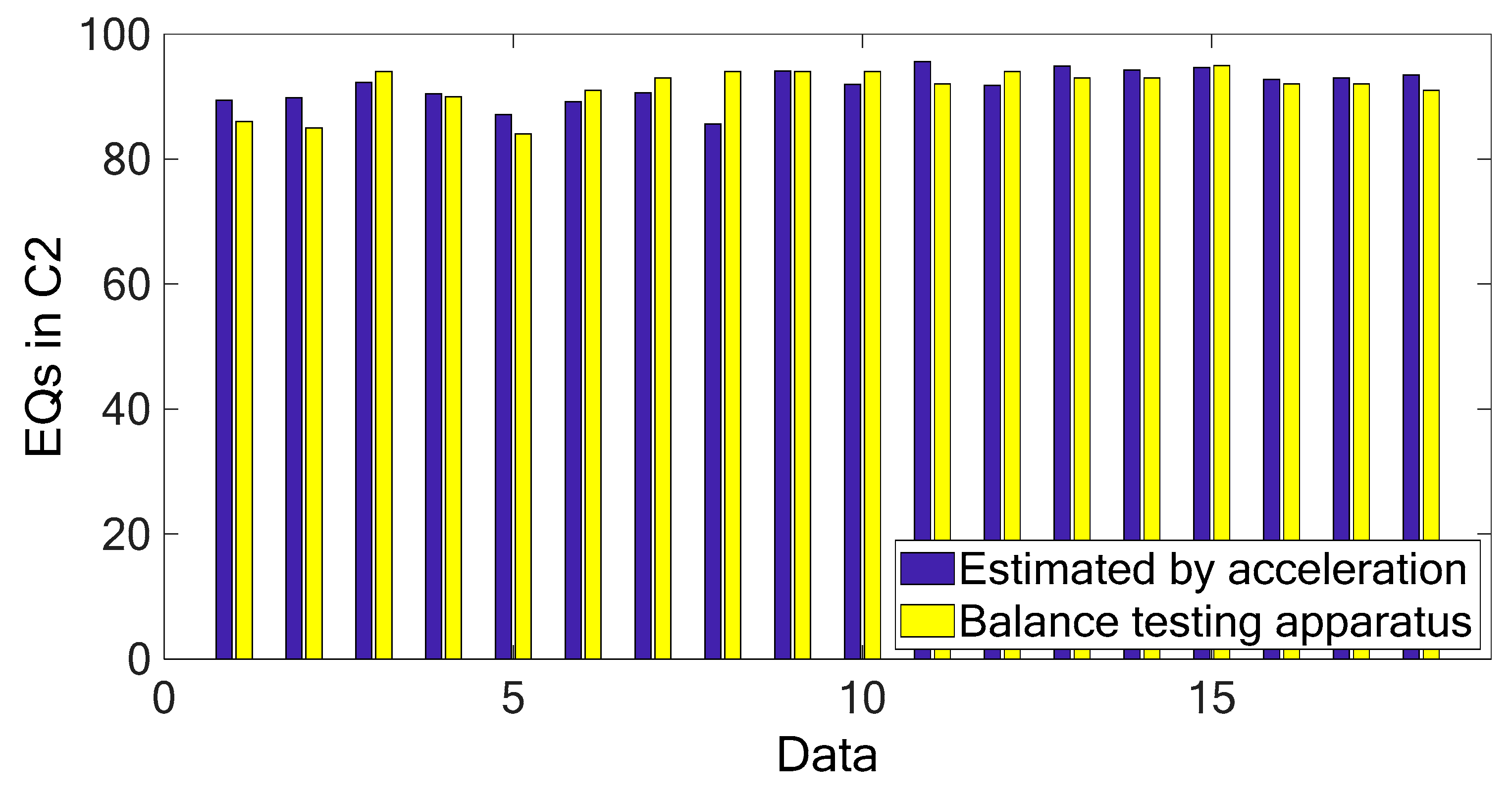
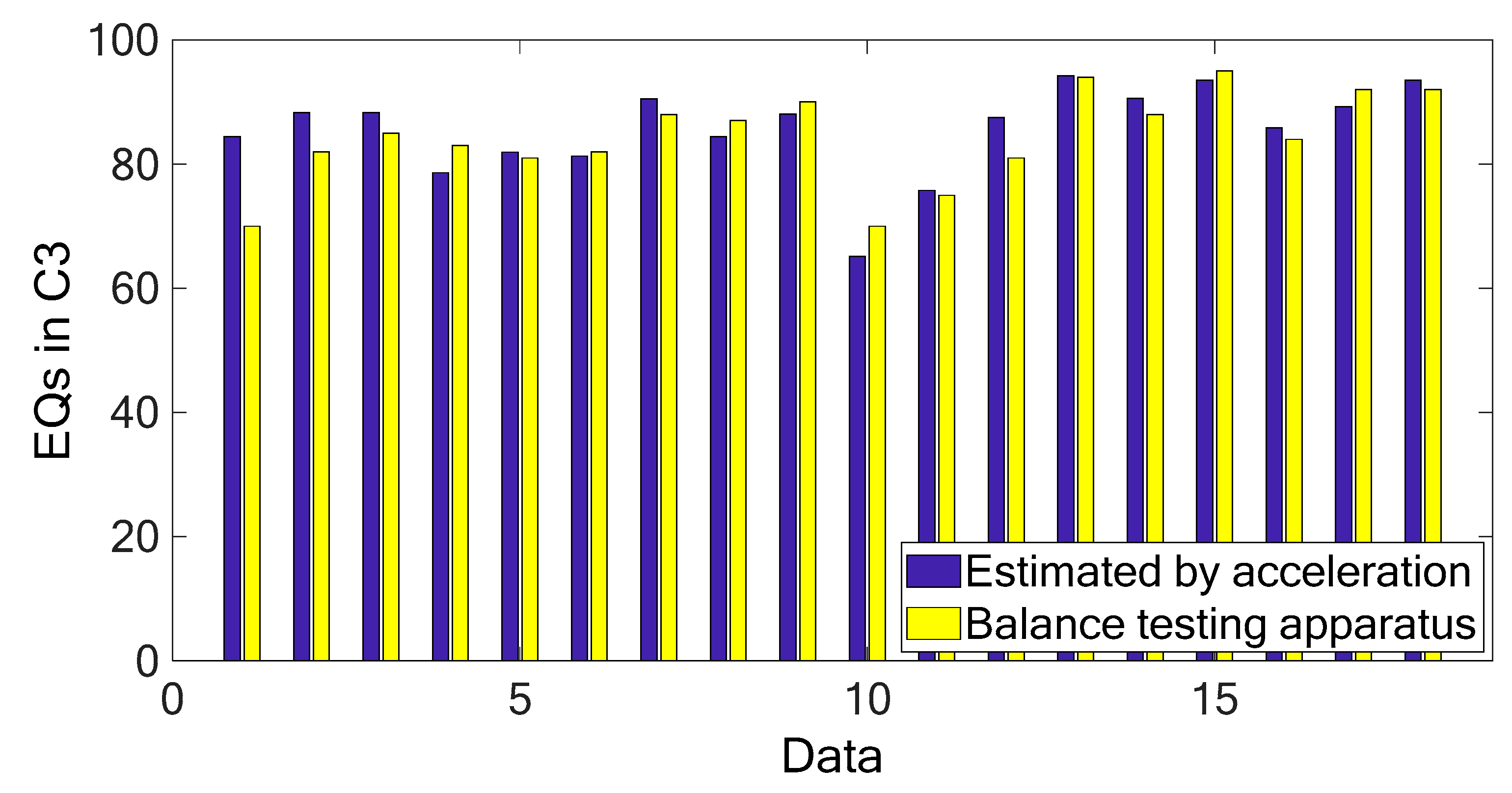
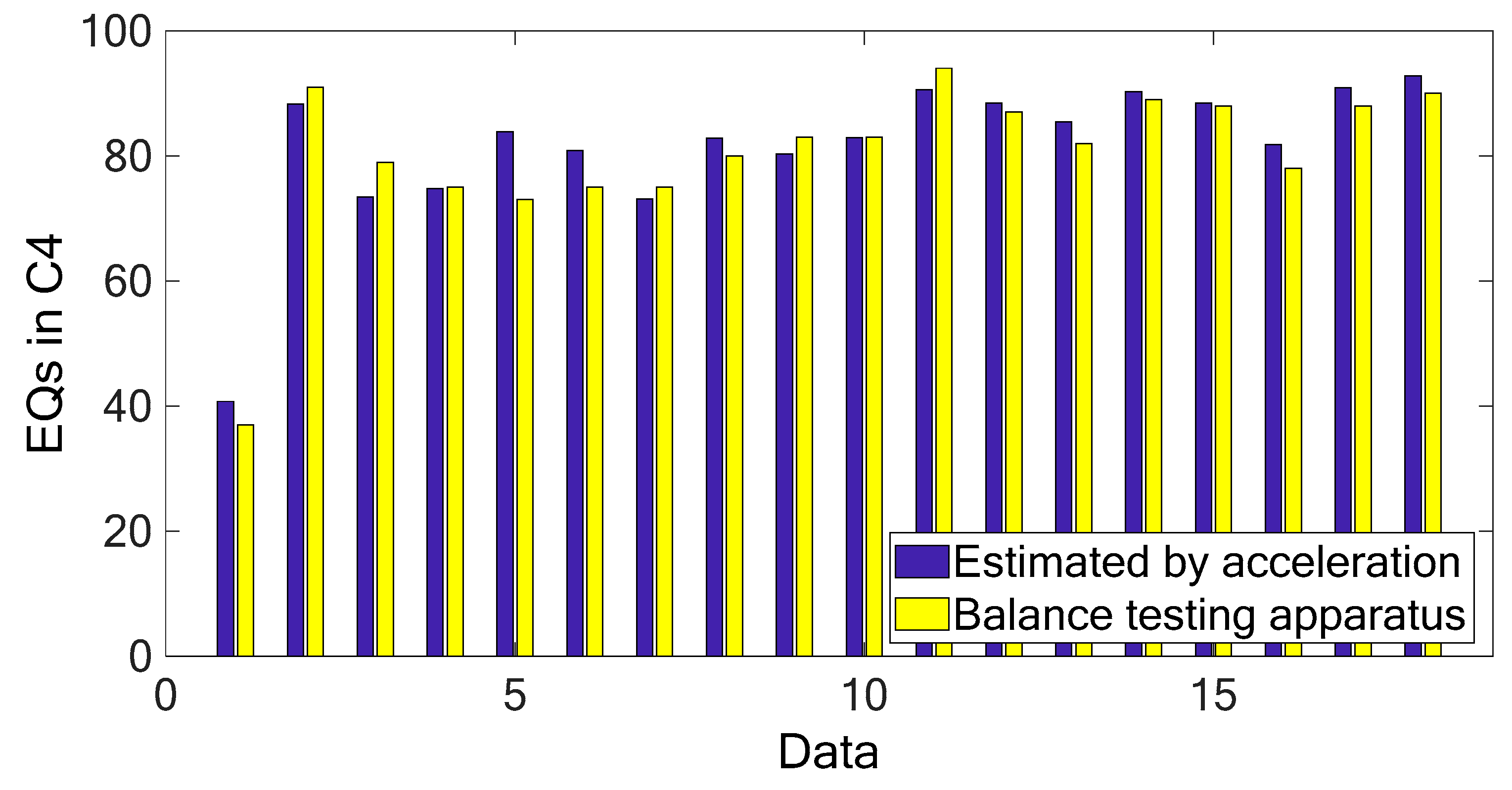
Figure 3 and
Figure 4 show the marginal spectra of the COP and acceleration resultants with different experimental visual conditions (as tabulated in
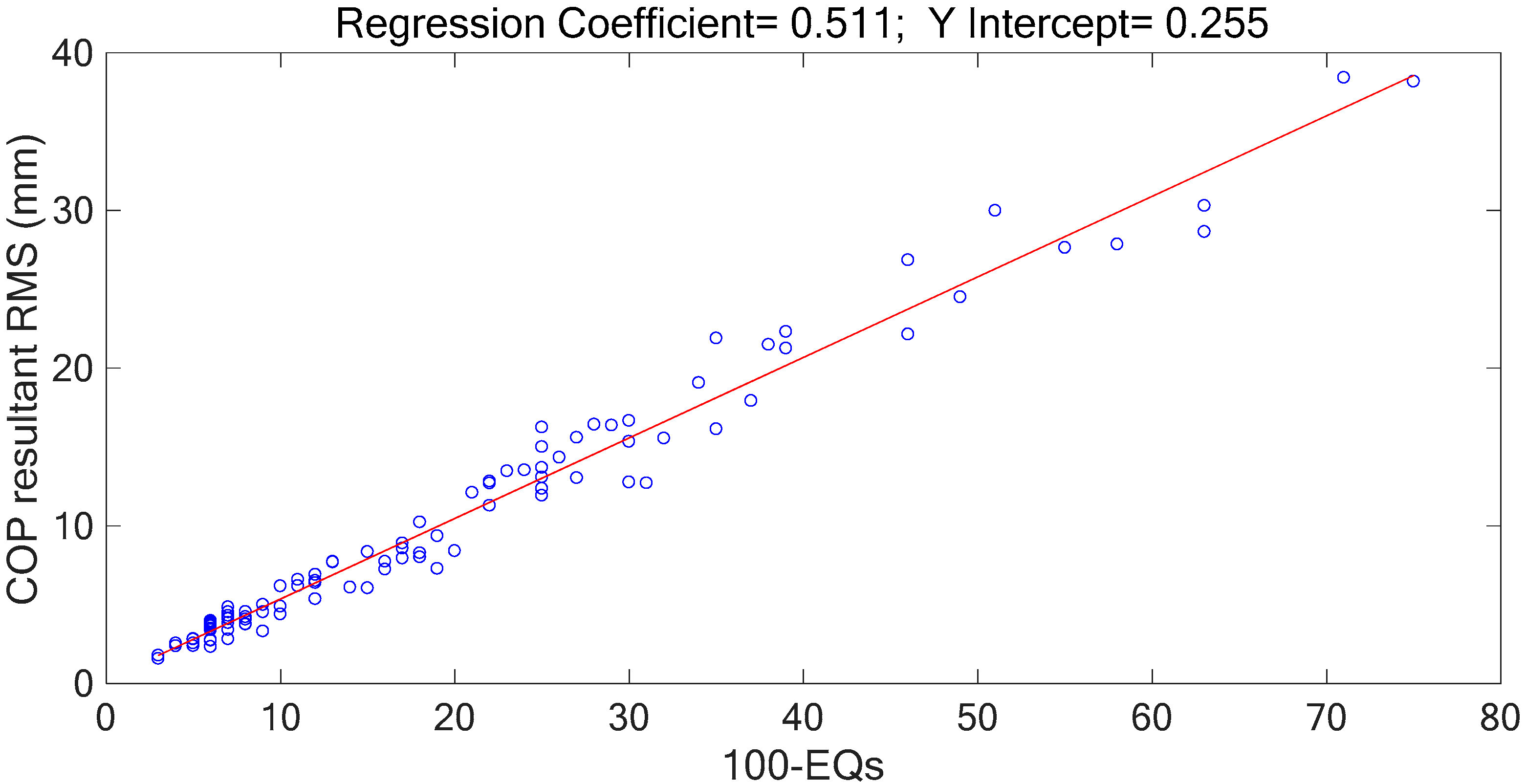
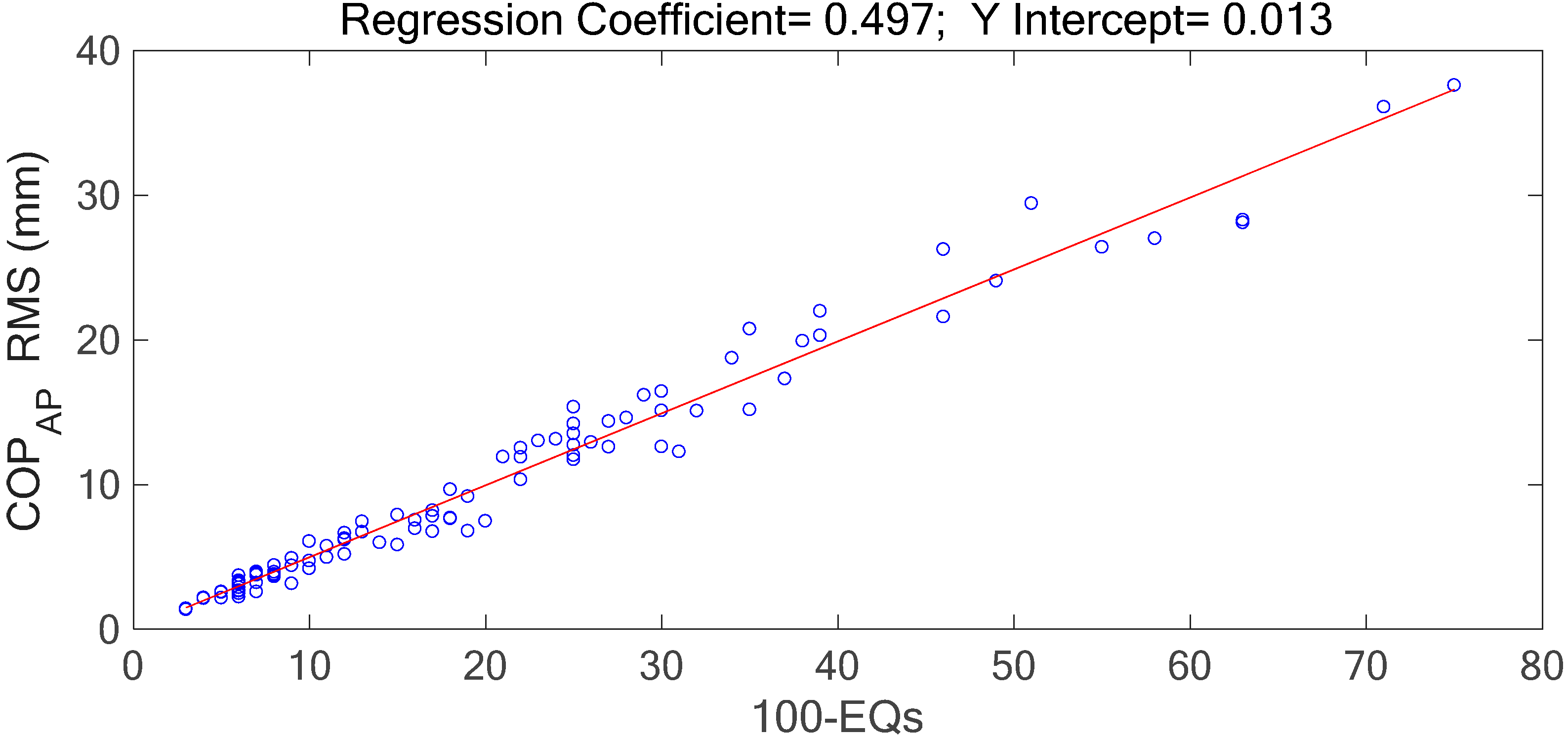
Table 1). The marginal spectra of COP and acceleration resultants apparently show that the signal energy concentrates at the low-frequency range. It is intuitive and coincident with the observation that the signal information correlated to the human body’s swaying for balance is found in the signal components of low frequencies. Based on the inference, the signal analysis of COP and acceleration in this research was focused on the IMFs whose bandwidths are within 0–5 Hz.
3.2. Segmentalized Principal Component Analysis (sPCA) for Acceleration Signal
As mentioned before, the frequency characteristics of the human body’s sway mainly focuses on the very low-frequency range, the capacitive accelerometer has satisfactory measurement performance within the bandwidth of very low frequencies, and thus, was utilized to detect the acceleration of the examinee’s body sway in the experiment. However, the bind-in accelerometer inevitably tilted along with the examinee’s body sway and hence, the X-Y-Z directions of the acceleration measurements were time-varying with respect to the fixed reference frame. Furthermore, the measurement of the capacitive accelerometer also includes the constant gravitation (g = 9.81 m/s2) in the vertical direction and thus, the acceleration measurements in the three directions may contain the gravitation projection components while the accelerometer was tilted along with the examinee’s body sway. To the contrary, the COP signal components in ML and AP directions were obtained in the horizontal plane and were independent of the gravitation in the vertical direction. Therefore, it is definitely beneficial for deriving the correlation between the COP and acceleration measurements if the gravitation projection onto the three directions of acceleration signals can be removed.
The concept of PCA that was proposed by Pearson [
13] is to convert the measurements in which the variables may be correlated with each other into another sets of variables which are uncorrelated. The sets of variables in the other space are called principal components. The mathematical procedure of PCA is to transform the data sets of measurement into another coordinate system, where the principal components are located through an orthogonal matrix. In this coordinate system, the first principal component has the highest variance, and the subsequent components in turn have as high variance as possible. Therefore, the principal components in the new space are uncorrelated with each other or even orthogonal to each other.
In order to resolve the stated problem of the axial directions variation as well as the gravitation projection issue, and to thereafter derive the relationship between the COP measurement and the acceleration signal, the PCA was employed to process the acceleration signals in this research. The acceleration measurements in X-, Y- and Z-directions were first transformed into another coordinate system where the principal components were located. The principal components in the new coordinate system are theoretically orthogonal to each other, and thus the operation of PCA can reflect the transformed signals in the way of most possible variability in the data [
14,
15]. In this research, therefore, the variability of acceleration measurements along the three directions was first analyzed.
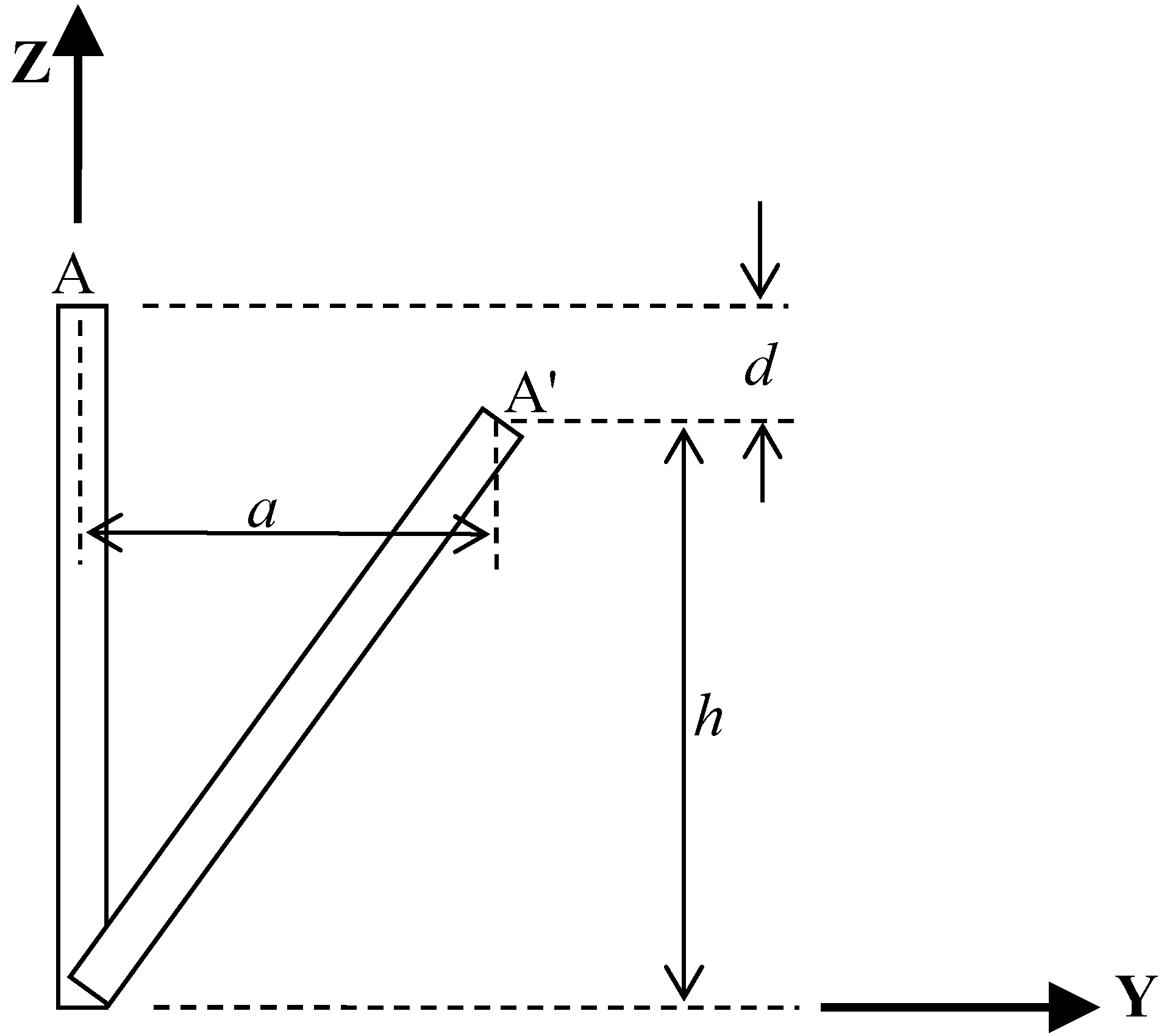
Simply consider that the accelerometer is bound at the point A of the examinee’s waist and sways in the AP direction, as shown in
Figure 5. As the examinee sways to maintain the body’s balance status, the accelerometer moves from A to A’, as shown in
Figure 5. The acceleration measurements consist of the components perpendicular to the Z-direction as well as the component parallel to the Z-direction. Since the two acceleration components in Y- and Z-direction are proportional to the distance
a and
d respectively, it can be briefly proven that the acceleration has larger variance in Y-direction than in Z-direction if
a is always greater than
d. First, assume
d > a contradictorily. The distance
a in Y-direction can be determined from the geometric relationship,
, and hence,
. It is easy to derive the contradiction that the condition for
d >
a implies
d >
h. Similarly, the same inference can be applied in the X-direction instead of the Y-direction. Therefore, it is reasonable to infer that the acceleration variance in Z-direction is smaller than those in X- and Y-directions.
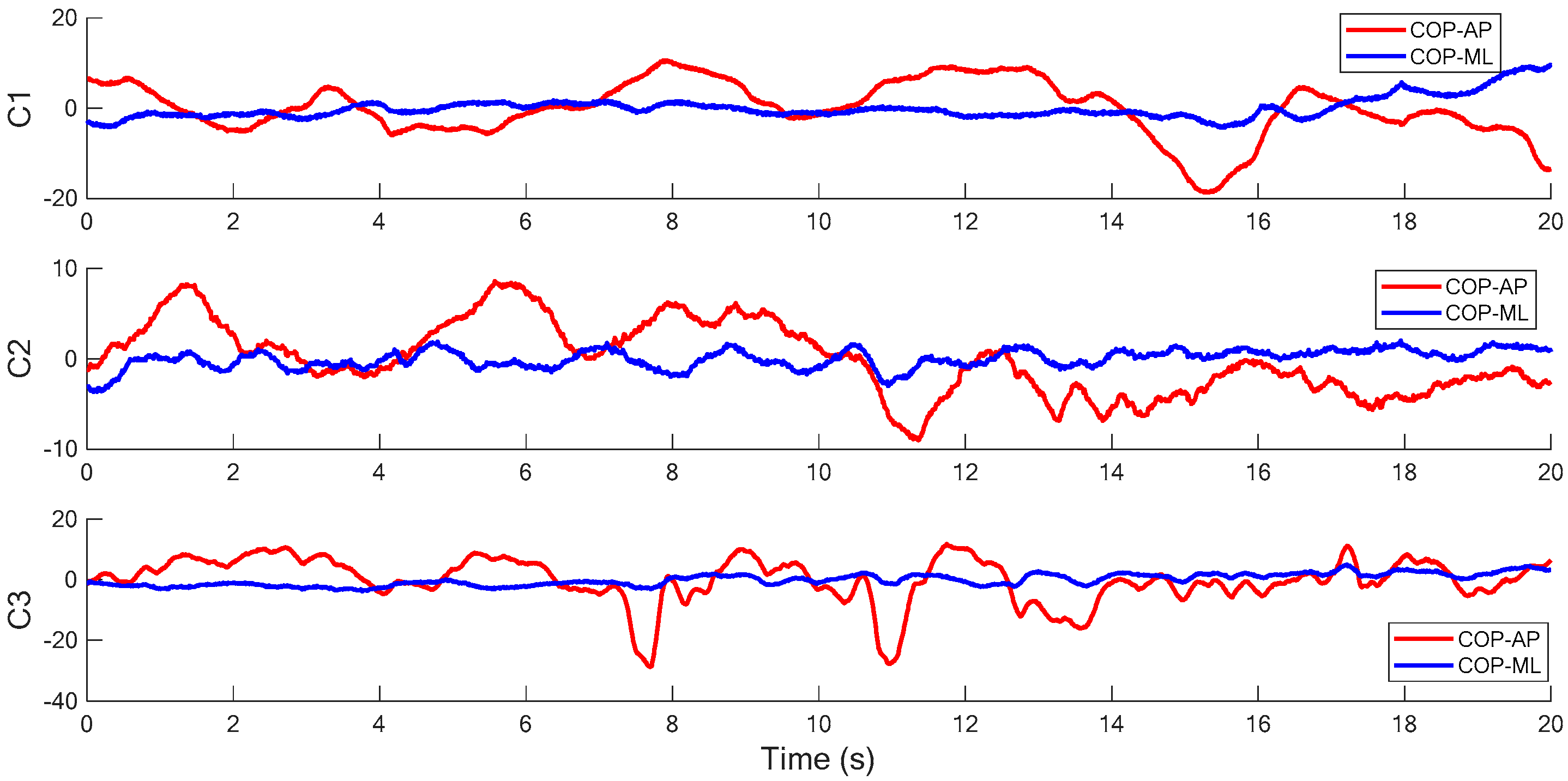
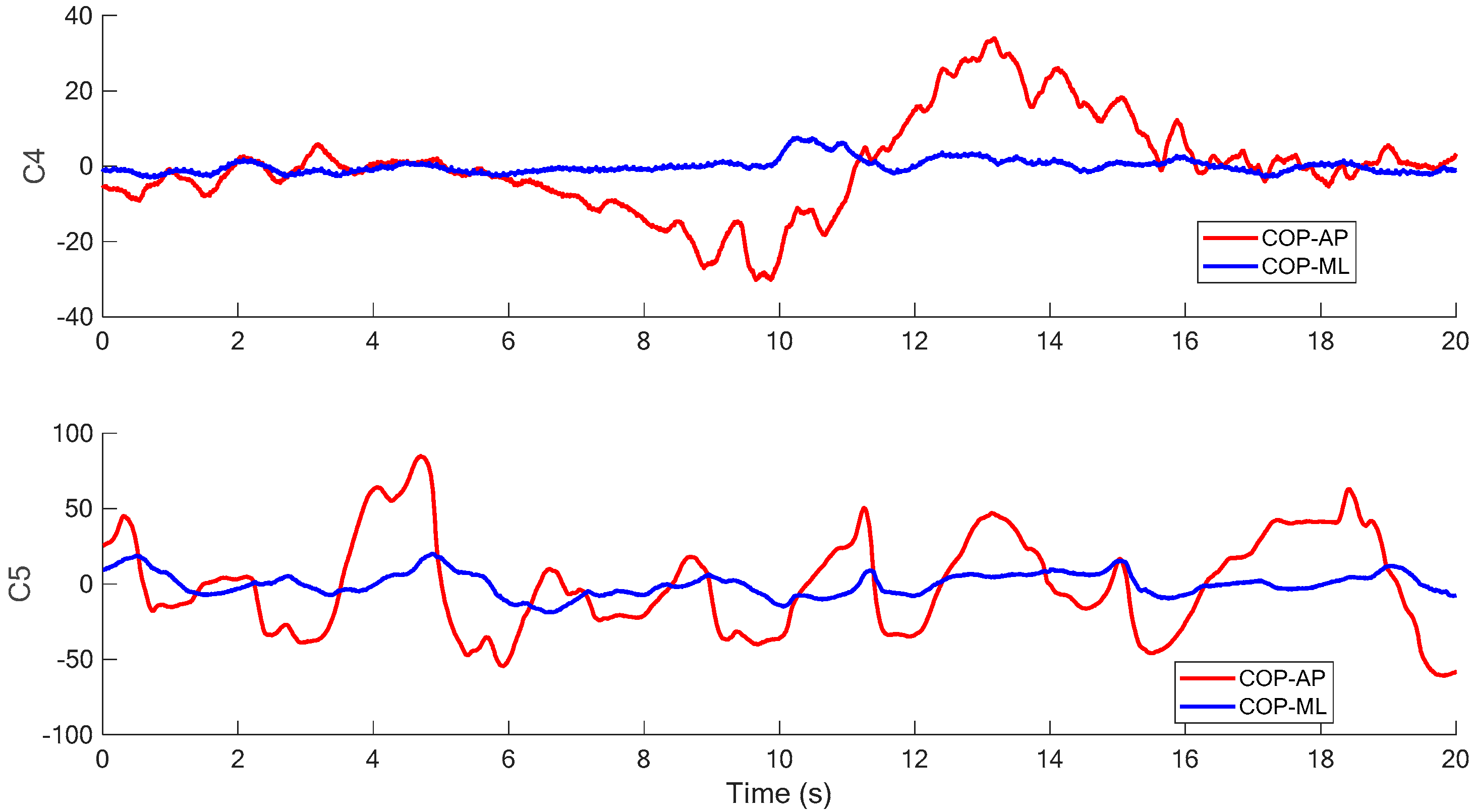
Figure 6 and
Figure 7 illustrate the COP measurements of the five visual conditions (Conditions 1 to 5, as shown in
Table 1) in the SOT of this research. It is found in the figures that the examinee generally has more sway variance in the AP direction than in the ML direction. A similar phenomenon can also be observed in most of the other COP measurements. The inference is reasonable because the structure of legs and ankles has more capability against the perturbation in the ML direction than in the AP direction while the examinee stands in the normal posture. Therefore, it is assumed in this study that the acceleration measurement in AP direction dominates the balancing characteristics as the examinee sways for most tests.
Based on the concept of PCA, the first principal component corresponds to the COP signal component in the AP direction. The second principal component corresponds to the COP signal component in the ML direction. The third principal component is attributed to the gravitation of the vertical component. This inference was drawn from the fact that the acceleration measurement had less variance in the direction parallel to the gravitation than the directions perpendicular to the gravitation.
Although the PCA is capable of transforming the acceleration signals into the three principal components corresponding to the AP, ML and vertical directions conceptually, it is, however, apparently unable to reflect the instantaneous variation in the projection of gravitation onto the three measured components in case of the time-varying tilting accelerometer if the PCA is employed to process the signal throughout the whole data length (20 s). In order to address this concern and to deal with the time-varying problem of directivity variation on the oblique accelerometer, the segmentalized PCA (sPCA) was proposed in this research to accurately extract the time-varying components corresponding to the acceleration components that are perpendicular to the gravity at the different instants during the human body’s sway.
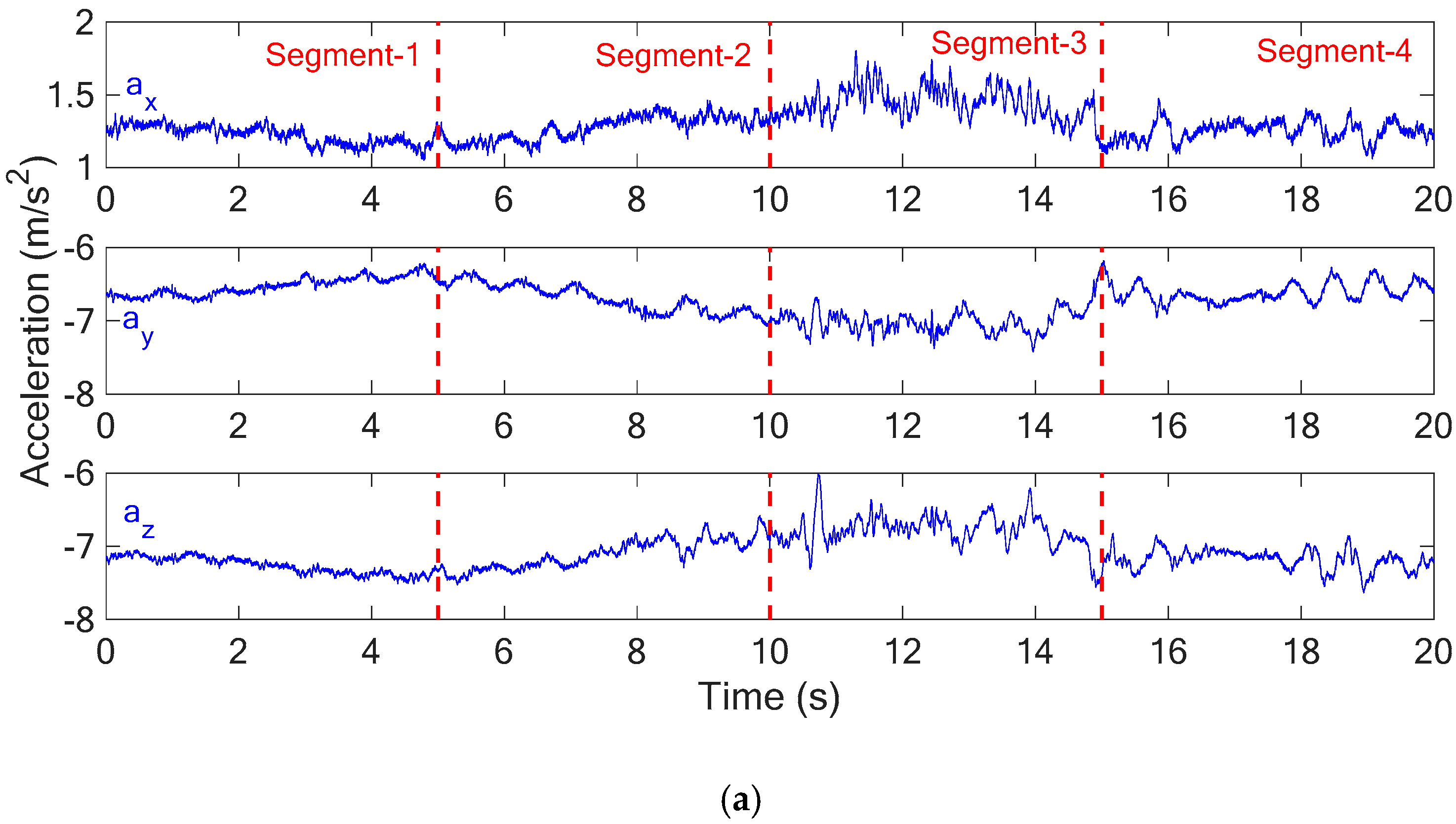
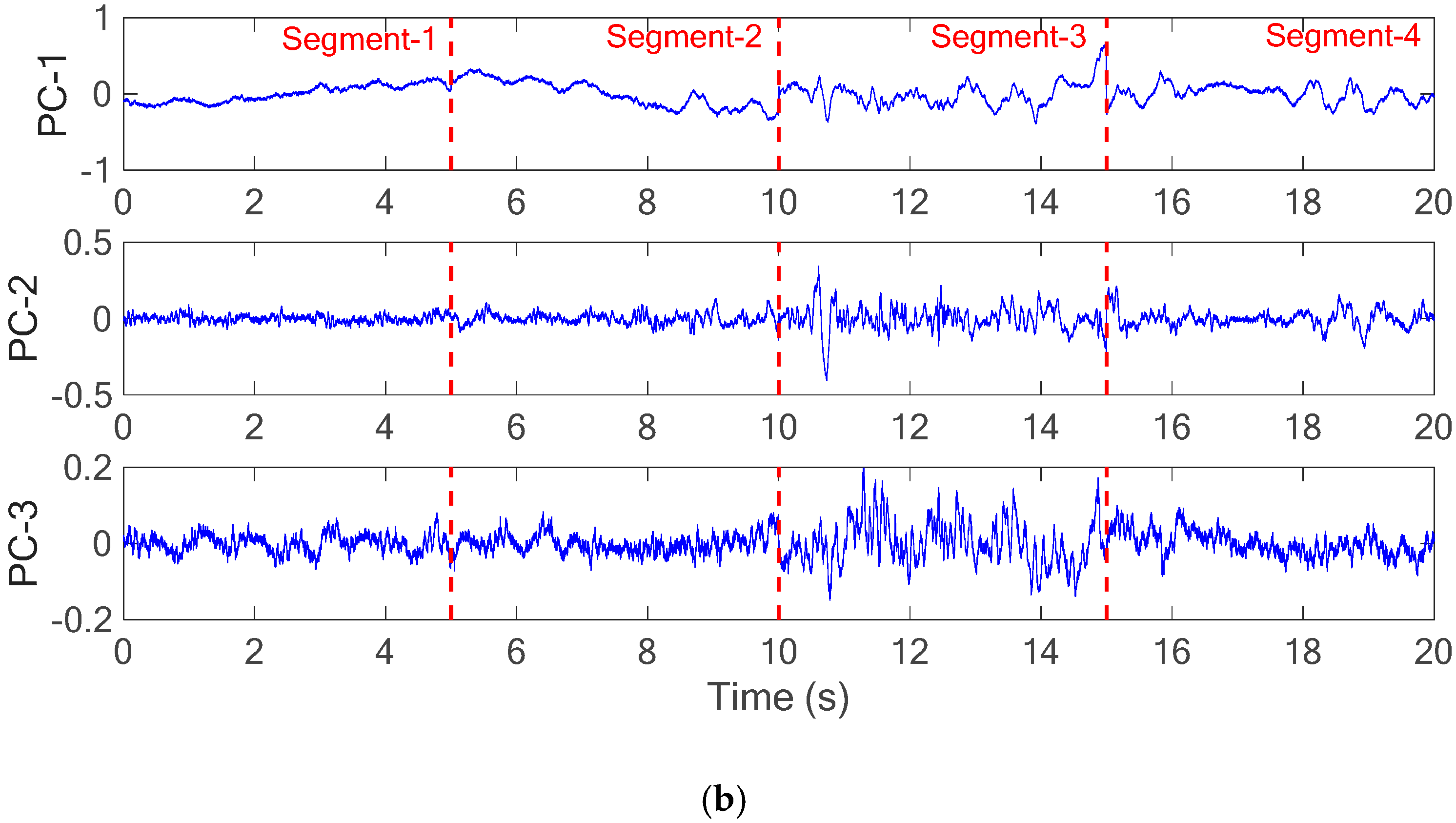
As illustrated in
Figure 8a, the acceleration measurements in X-, Y- and Z-directions were divided into four segments and then the PCA was applied for each segment to determine the three principal components (PC-1, PC-2 and PC-3), as shown in
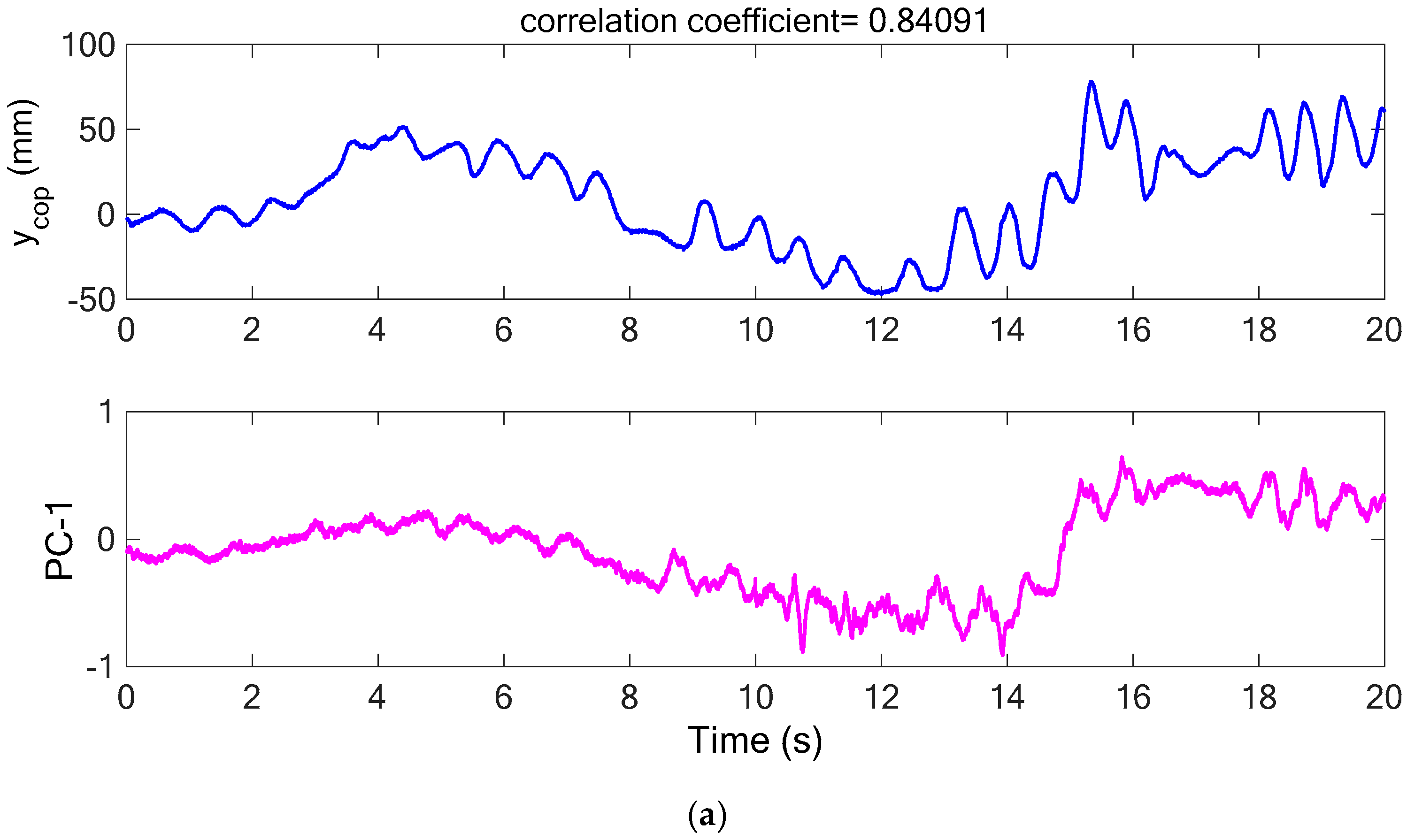
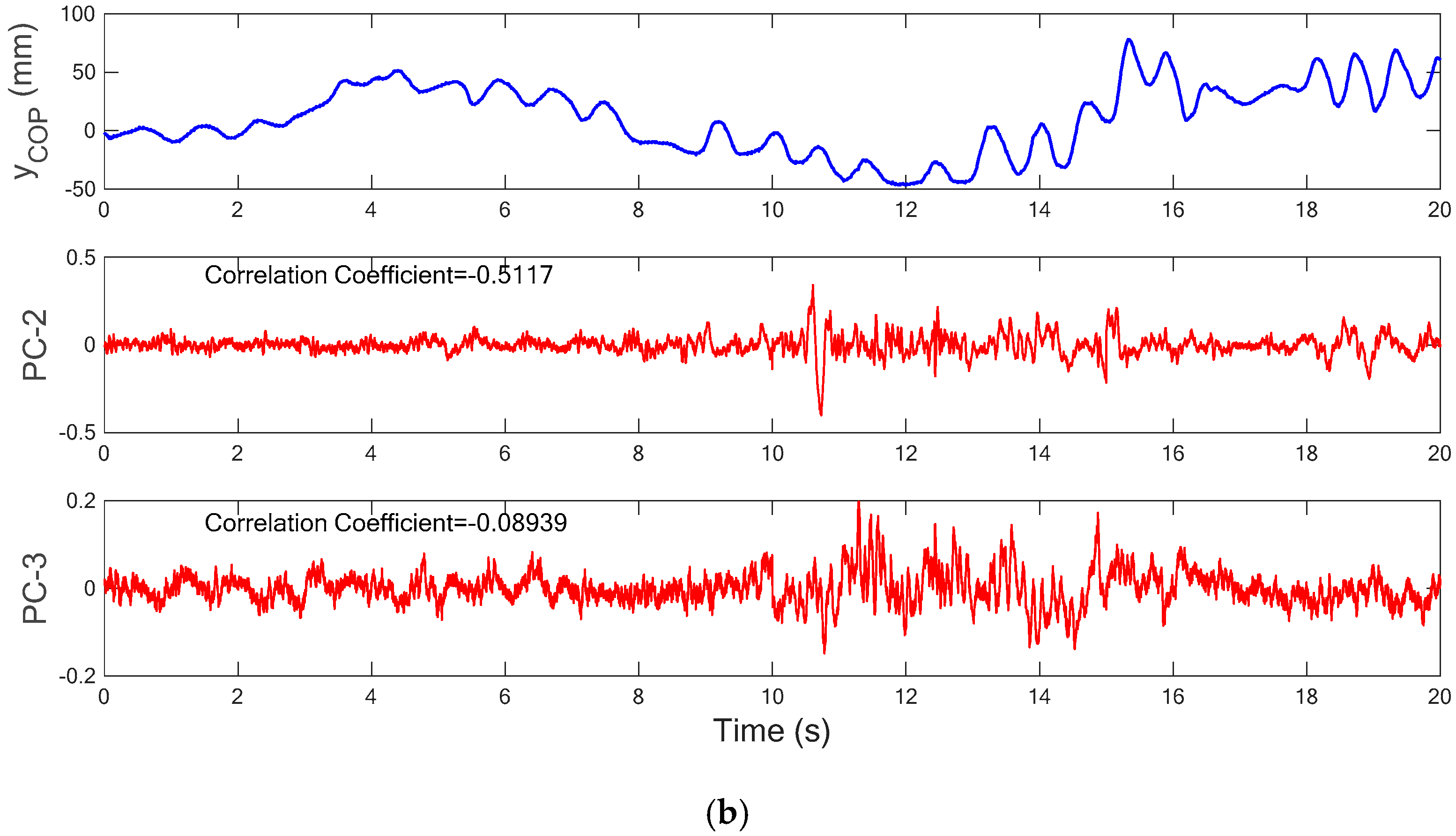
Figure 8b. With the connected principal components of each segment,
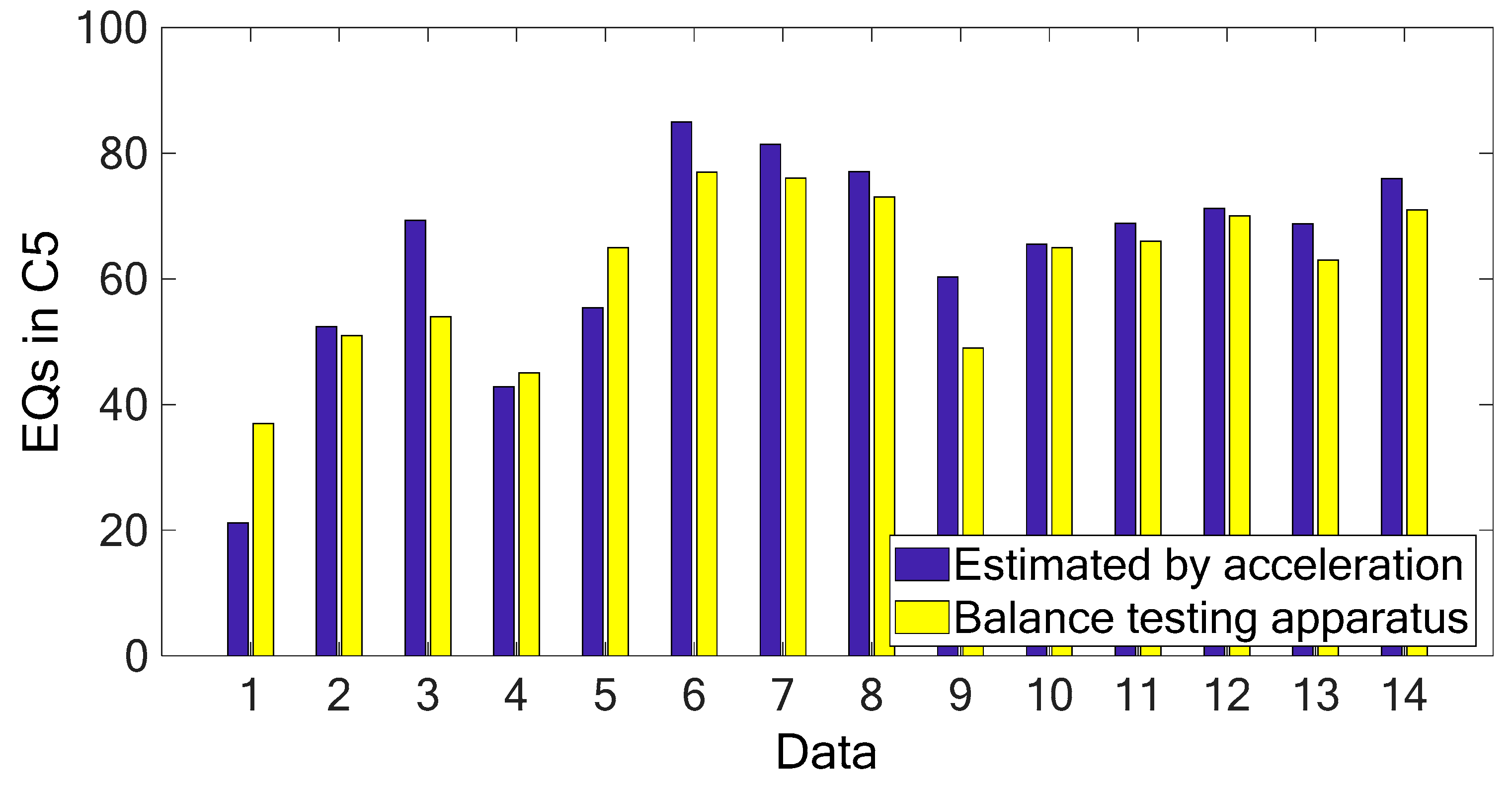
Figure 9a shows the comparison between the COP measurement in the Y-axis (AP) and the first principal component (PC-1) of acceleration signals. It is noted that the PC-1 of the acceleration measurements has similar variation trend with the COP measurement in the AP direction and the correlation coefficient between the two sets of series was calculated as 0.841. As compared with the PC-2 and PC-3 of the acceleration signals, their variation trends are very different from the COP measurement in the AP direction (as shown in
Figure 9b), and thus, they have correlation coefficients of −0.512 and −0.089, respectively. The high correlation coefficient between the COP measurement in the AP direction and the PC-1 of the acceleration measurements demonstrates that it is sufficient to accurately predict the COP measurement in the AP direction. Therefore, it is feasible to utilize the processed acceleration measurement to represent the COP signal for evaluating the human body’s balancing ability.