Feasibility Assessment of a Bioethanol Plant in the Northern Netherlands
Abstract
1. Introduction
2. Materials and Methods
2.1. Feedstocks
2.2. Process Design
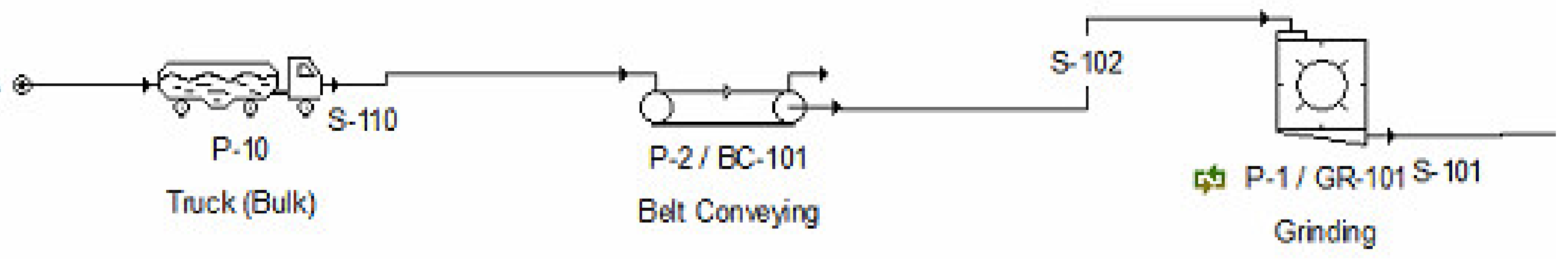
2.2.1. Front End Operations
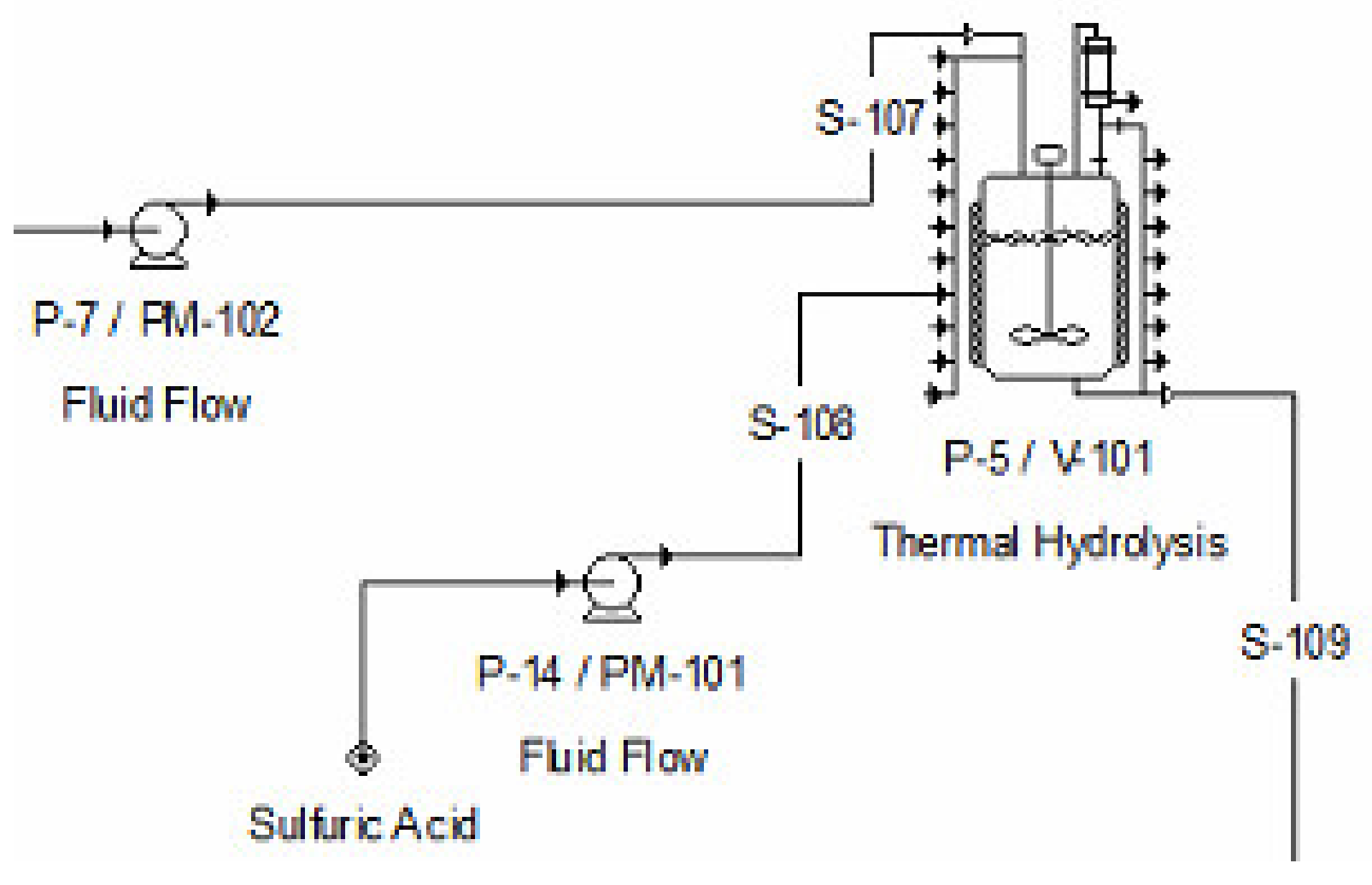
2.2.2. Pretreatment
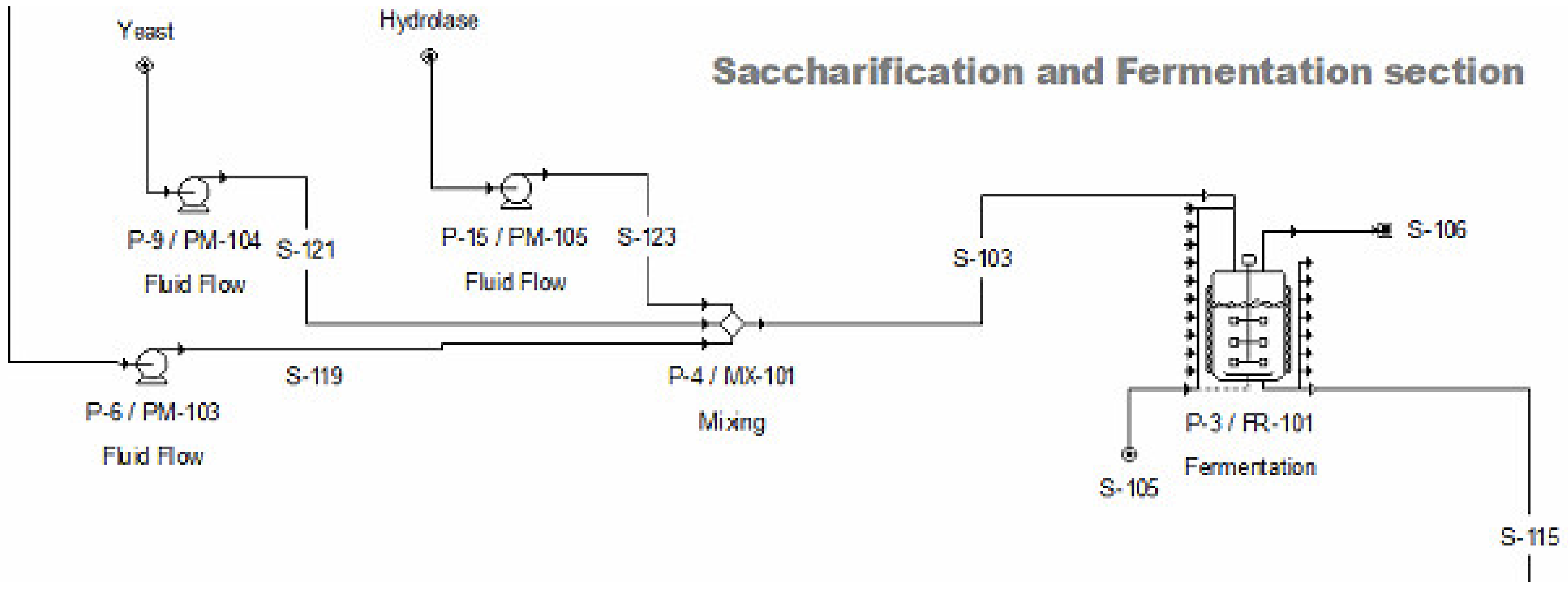
2.2.3. Saccharification and Fermentation
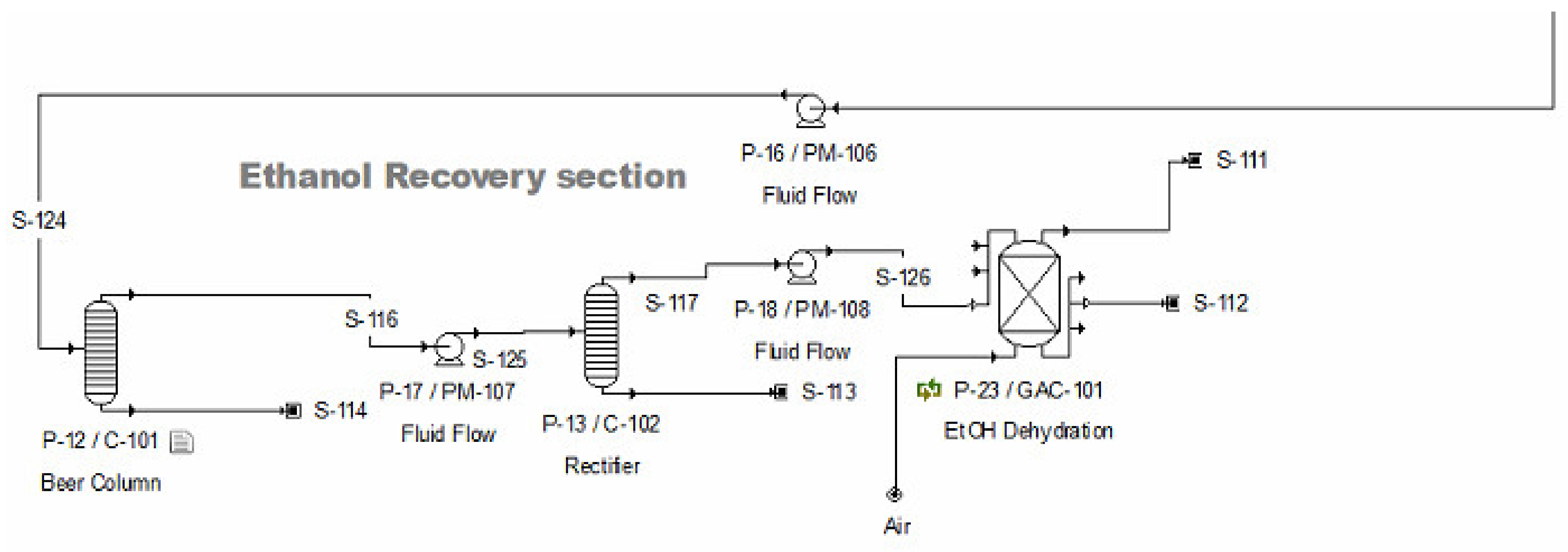
2.2.4. Ethanol Recovery
2.3. Economic Assessment
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Oliveira, A.; Navia, R. Zero waste bio-refineries: The example of Brazilian pulp and paper sector. Waste Manag. Res. 2017, 35, 1193–1194. [Google Scholar] [CrossRef] [PubMed]
- Achinas, S.; Achinas, V.; Euverink, G.J.W. A technological overview of biogas production from biowaste. Engineering 2017, 3, 299–307. [Google Scholar] [CrossRef]
- Chen, J.F. Green chemical engineering for a better life. Engineering 2017, 3, 279. [Google Scholar] [CrossRef]
- Sahajwalla, V. Green processes: Transforming waste into valuable resources. Engineering 2018, 4, 309–310. [Google Scholar] [CrossRef]
- Anjum, M.; Khalid, A.; Qadeer, S.; Miandad, R. Synergistic effect of co-digestion to enhance anaerobic degradation of catering waste and orange peel for biogas production. Waste Manag. Res. 2017, 35, 967–977. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.; Anderson, E.; Addy, M.; Zhang, R.; Cheng, Y.; Peng, P.; Ma, Y.; Fan, L.; Zhang, Y.; Lu, Q.; et al. Breakthrough technologies for the biorefining of organic solid and liquid wastes. Engineering 2018, 4, 574–580. [Google Scholar] [CrossRef]
- Hildebrandt, J.; Bezama, A. Cross-fertilisation of ideas for a more sustainable fertiliser market: The need to incubate business concepts for harnessing organic residues and fertilisers on biotechnological conversion platforms in a circular bioeconomy. Waste Manag. Res. 2018, 36, 1125–1126. [Google Scholar] [CrossRef]
- Marzo, C.; Díaz, A.B.; Caro, I.; Blandino, A. Valorization of agro-industrial wastes to produce hydrolytic enzymes by fungal solid-state fermentation. Waste Manag. Res. 2019, 37, 149–156. [Google Scholar] [CrossRef]
- Zhou, Z.; Tang, Y.; Chi, Y.; Ni, M.; Buekens, A. Waste-to-energy: A review of life cycle assessment and its extension methods. Waste Manag. Res. 2018, 36, 3–16. [Google Scholar] [CrossRef]
- Philippidis, G.; Bartelings, H.; Helming, J.; M’barek, R.; Smeets, E.; Van Meijl, H. The good, the bad and the uncertain: Bioenergy use in the European Union. Energies 2018, 11, 2703. [Google Scholar] [CrossRef]
- Aragie, E.; Balié, J.; MoralesOpazo, C. Does reducing food losses and wastes in sub-Saharan Africa make economic sense? Waste Manag. Res. 2018, 36, 483–494. [Google Scholar] [CrossRef] [PubMed]
- Park, J.H.; Jin, M.H.; Lee, Y.J.; Song, G.S.; Choi, J.W.; Lee, D.W.; Kim, J.G. Two-in-one fuel synthetic bioethanol-lignin from lignocellulose with sewage sludge and its air pollutants reduction effects. Energies 2019, 12, 3072. [Google Scholar] [CrossRef]
- Chang, C.-L.; Li, Y.; McAleer, M. Volatility spillovers between energy and agricultural markets: A critical appraisal of theory and practice. Energies 2018, 11, 1595. [Google Scholar] [CrossRef]
- Bilandžija, N.; Krička, T.; Matin, A.; Leto, J.; Grubor, M. Effect of harvest season on the fuel properties of Sida hermaphrodita (L.) Rusby biomass as solid biofuel. Energies 2018, 11, 3398. [Google Scholar] [CrossRef]
- Uçar, S.; Karagöz, S. Co-processing of olive bagasse with crude rapeseed oil via pyrolysis. Waste Manag. Res. 2017, 35, 480–490. [Google Scholar] [CrossRef] [PubMed]
- Solarte-Toro, J.C.; Chacón-Pérez, Y.; Cardona-Alzate, C.A. Evaluation of biogas and syngas as energy vectors for heat and power generation using lignocellulosic biomass as raw material. Electron. J. Biotechnol. 2018, 33, 52–62. [Google Scholar] [CrossRef]
- Ahorsu, R.; Medina, F.; Constantí, M. Significance and challenges of biomass as a suitable feedstock for bioenergy and biochemical production: A review. Energies 2018, 11, 3366. [Google Scholar] [CrossRef]
- Park, J.H.; Jin, M.H.; Lee, Y.J.; Song, G.S.; Choi, J.W.; Lee, D.W.; Choi, Y.C.; Park, S.J.; Song, K.H.; Kim, J.G.; et al. A Solid-state fermentation and composting as alternatives to treat hair waste: A life-cycle assessment comparative approach. Waste Manag. Res. 2017, 35, 786–790. [Google Scholar]
- Smuga-Kogut, M.; Piskier, T.; Walendzik, B.; Szymanowska-Powałowska, D. Assessment of wasteland derived biomass for bioethanol production. Electron. J. Biotechnol. 2019, 41, 1–8. [Google Scholar] [CrossRef]
- Sharifyazd, S.; Karimi, K. Effects of fermentation conditions on valuable products of ethanolic fungus Mucor indicus. Electron. J. Biotechnol. 2017, 30, 77–82. [Google Scholar] [CrossRef]
- Tremouli, A.; Papadopoulou, K.; Kanellos, G.; Lampropoulos, I.; Lyberatos, G. Bioelectricity production from fermentable household waste in a dual-chamber microbial fuel cell. Waste Manag. Res. 2018, 36, 1037–1042. [Google Scholar]
- RedCorn, R.; Fatemi, S.; Engelberth, A.S. Comparing end-use potential for industrial food-waste sources. Engineering 2018, 4, 371–380. [Google Scholar] [CrossRef]
- Wang, J.; Wang, H.; Fan, Y. Techno-economic challenges of fuel cell commercialization. Engineering 2018, 4, 352–360. [Google Scholar] [CrossRef]
- Longjan, G.G.; Dehouche, Z. Nutrient characterisation and bioenergy potential of common Nigerian food wastes. Waste Manag. Res. 2018, 36, 426–435. [Google Scholar] [CrossRef]
- Reißmann, D.; Thrän, D.; Bezama, A. How to identify suitable ways for the hydrothermal treatment of wet bio-waste? A critical review and methods proposal. Waste Manag. Res. 2018, 36, 912–923. [Google Scholar] [CrossRef]
- Sepúlveda-Cervantes, C.V.; Soto-Regalado, E.; Rivas-García, P.; Loredo-Cancino, M.; Cerino-Córdova, F.dJ.; García Reyes, R.B. Technical-environmental optimisation of the activated carbon production of an agroindustrial waste by means response surface and life cycle assessment. Waste Manag. Res. 2018, 36, 121–130. [Google Scholar] [CrossRef]
- Chen, J.F. Green chemical engineering. Engineering 2017, 3, 283–284. [Google Scholar] [CrossRef]
- Nelson, M.J.; Nakhla, G.; Zhu, J. Fluidized-bed bioreactor applications for biological wastewater treatment: A review of research and developments. Engineering 2017, 3, 330–342. [Google Scholar] [CrossRef]
- Kupczyk, A.; Mączyńska, J.; Redlarski, G.; Tucki, K.; Bączyk, A.; Rutkowski, D. Selected aspects of biofuels market and the electromobility development in Poland: Current trends and forecasting changes. Appl. Sci. 2019, 9, 254. [Google Scholar] [CrossRef]
- Demirbas, A. Bioethanol from cellulosic materials: A renewable motor fuel from biomass. Energy Sources 2005, 27, 327–337. [Google Scholar] [CrossRef]
- Matsakas, L.; Gao, Q.; Jansson, S.; Rova, U.; Christakopoulos, P. Green conversion of municipal solid wastes into fuels and chemicals. Electron. J. Biotechnol. 2017, 26, 69–83. [Google Scholar] [CrossRef]
- Koçer, A.T.; Özçimen, D. Investigation of the biogas production potential from algal wastes. Waste Manag. Res. 2018, 36, 1100–1105. [Google Scholar] [CrossRef] [PubMed]
- Sotelo-Navarro, P.; Poggi-Varaldo, H.; Turpin-Marion, S.; Vázquez-Morillas, A.; Beltrán-Villavicencio, M.; Espinosa-Valdemar, R. Biohydrogen production from used diapers: Evaluation of effect of temperature and substrate conditioning. Waste Manag. Res. 2017, 35, 267–275. [Google Scholar] [CrossRef] [PubMed]
- EurObserv′er. The State of Renewable Energies in Europe, 17th ed.; Observ’ER: Pairs, France, 2017. [Google Scholar]
- Phukoetphim, N.; Salakkam, A.; Laopaiboon, P.; Laopaiboon, L. Improvement of ethanol production from sweet sorghum juice under batch and fed-batch fermentations: Effect of sugar levels, nitrogen supplementation, and feeding regimes. Electron. J. Biotechnol. 2017, 26, 84–92. [Google Scholar] [CrossRef]
- Pradyawong, S.; Juneja, A.; Sadiq, M.B.; Noomhorm, A.; Singh, V. Comparison of cassava starch with corn as a feedstock for bioethanol production. Energies 2018, 11, 3476. [Google Scholar] [CrossRef]
- Taški-Ajduković, K.; Nagl, N.; Ćurčić, Z.; Zorić, M. Estimation of genetic diversity and relationship in sugar beet pollinators based on SSR markers. Electron. J. Biotechnol. 2017, 27, 1–7. [Google Scholar] [CrossRef]
- Taiwo, A.E.; Madzimbamuto, T.N.; Ojumu, T.V. Optimization of corn steep liquor dosage and other fermentation parameters for ethanol production by Saccharomyces cerevisiae Type 1 and Anchor Instant Yeast. Energies 2018, 11, 1740. [Google Scholar] [CrossRef]
- Gronchi, N.; Favaro, L.; Cagnin, L.; Brojanigo, S.; Pizzocchero, V.; Basaglia, M.; Casella, S. Novel yeast strains for the efficient saccharification and fermentation of starchy by-products to bioethanol. Energies 2019, 12, 714. [Google Scholar] [CrossRef]
- Domínguez, J.; Gómez-Brandón, M.; Martínez-Cordeiro, H.; Lores, M. Bioconversion of Scotch broom into a high-quality organic fertiliser: Vermicomposting as a sustainable option. Waste Manag. Res. 2018, 36, 1092–1099. [Google Scholar] [CrossRef]
- Sanchez, N.; Ruiz, R.Y.; Infante, N.; Cobo, M. Bioethanol production from Cachaza as hydrogen feedstock: Effect of ammonium sulfate during fermentation. Energies 2017, 10, 2112. [Google Scholar] [CrossRef]
- Franchi, O.; Rosenkranz, F.; Chamy, R. Key microbial populations involved in anaerobic degradation of phenol and p-cresol using different inocula. Electron. J. Biotechnol. 2018, 35, 33–38. [Google Scholar] [CrossRef]
- Chen, K.H.; Chao, Y.C. Characterization of performance of short stroke engines with valve timing for blended bioethanol internal combustion. Energies 2019, 12, 759. [Google Scholar] [CrossRef]
- Rujnić-Sokele, M.; Pilipović, A. Challenges and opportunities of biodegradable plastics: A mini review. Waste Manag. Res. 2017, 35, 132–140. [Google Scholar] [CrossRef] [PubMed]
- Gao, L.; Guo, Q.; Lin, H.; Pan, D.; Huang, X.; Lin, J.; Lin, J. Modeling of lactose enzymatic hydrolysis using Monte Carlo method. Electron. J. Biotechnol. 2019, 40, 78–83. [Google Scholar] [CrossRef]
- Berlowska, J.; Pielech-Przybylska, K.; Balcerek, M.; Cieciura, W.; Borowski, S.; Kregiel, D. Integrated bioethanol fermentation/anaerobic digestion for valorization of sugar beet pulp. Energies 2017, 10, 1255. [Google Scholar] [CrossRef]
- Berlowska, J.; Pielech-Przybylska, K.; Balcerek, M.; Dziekońska-Kubczak, U.; Patelski, P.; Dziugan, P.; Kręgiel, D. Simultaneous saccharification and fermentation of sugar beet pulp for efficient bioethanol production. BioMed. Res. Int. 2016, 9, 1–10. [Google Scholar]
- Centraal Bureau voor de Statistiek. Available online: https://statline.cbs.nl/Statweb/publication/?DM=SLNL&PA=7100oogs&D1=1-3&D2=25&D3=1&D4=6,22-24&HDR=G2,G3,T&STB=G1&VW=T (accessed on 28 May 2019).
- Centraal Bureau voor de Statistiek. Available online: https://statline.cbs.nl/Statweb/publication/?DM=SLNL&PA=80780ned&D1=459-481&D2=1&D3=l&HDR=G1,G2&STB=T&VW=C (accessed on 1 June 2019).
- Liu, Y.K.; Chen, W.C.; Huang, Y.C.; Chang, Y.K.; Chu, I.M.; Tsai, S.L.; Wei, Y.H. Production of bioethanol from Napier grass via simultaneous saccharification and co-fermentation in a modified bioreactor. J. Biosci. Bioeng. 2017, 124, 184–188. [Google Scholar] [CrossRef]
- Centraal Bureau in de Statistiek. Available online: https://statline.cbs.nl/Statweb/publication/?DM=SLNL&PA=81302ned&D1=440-443&D2=l&HDR=G1&STB=T&VW=T (accessed on 2 June 2019).
- Centraal Bureau in de Statistiek. Available online: https://statline.cbs.nl/Statweb/publication/?DM=SLNL&PA=7140GRAS&D1=a&D2=1&D3=l&HDR=G2,G1&STB=T&VW=T (accessed on 2 June 2019).
- IRS. Sugar Beet Growing in the Netherlands; Institute of Sugar Beet Research: 2005. Available online: http://applicaties.irs.nl/ccmsupload/ccmsalg/sugar%20beet%20growing%20in%20the%20netherlands%20.pdf (accessed on 19 Sugust 2019).
- Kumar, D.; Murthy, G.S. Impact of pretreatment and downstream processing technologies on economics and energy in cellulosic ethanol production. Biotechnol. Biofuels 2011, 4, 1–19. [Google Scholar] [CrossRef]
- Smuga-Kogut, M.; Walendzik, B.; Szymanowska-Powalowska, D.; Kobus-Cisowska, J.; Wojdalski, J.; Wieczorek, M.; Cielecka-Piontek, J. Comparison of bioethanol preparation from triticale straw using the ionic liquid and sulfate methods. Energies 2019, 12, 1155. [Google Scholar] [CrossRef]
- Lemões, J.S.; Lemons e Silva, C.F.; Farias Avila, S.P.; Scherrer Montero, C.R.; dos Anjos e Silva, S.D.; Samios, D.; do Carmo Ruaro Peralba, M. Chemical pretreatment of Arundo donax L. for second-generation ethanol production. Electron. J. Biotechnol. 2018, 31, 67–74. [Google Scholar] [CrossRef]
- Raud, M.; Rooni, V.; Kikas, T. The efficiency of nitrogen and flue gas as operating gases in explosive decompression pretreatment. Energies 2018, 11, 2074. [Google Scholar] [CrossRef]
- Portero-Barahona, P.; Carvajal-Barriga, E.J.; Martín-Gil, J.; Martín-Ramos, P. Sugarcane bagasse hydrolysis enhancement by microwave-assisted sulfolane pretreatment. Energies 2019, 12, 1703. [Google Scholar] [CrossRef]
- Duque, A.; Manzanares, P.; González, A.; Ballesteros, M. Study of the application of alkaline extrusion to the pretreatment of Eucalyptus biomass as first step in a bioethanol production process. Energies 2018, 11, 2961. [Google Scholar] [CrossRef]
- Dziekońska-Kubczak, U.; Berłowska, J.; Dziugan, P.; Patelski, P.; Pielech-Przybylska, K.; Balcerek, M. Nitric acid pretreatment of Jerusalem artichoke stalks for enzymatic saccharification and bioethanol production. Energies 2018, 11, 2153. [Google Scholar] [CrossRef]
- Łukajtis, R.; Kucharska, K.; Hołowacz, I.; Rybarczyk, P.; Wychodnik, K.; Słupek, E.; Nowak, P.; Kamiński, M. Comparison and optimization of Saccharification conditions of alkaline pre-treated triticale straw for acid and enzymatic hydrolysis followed by ethanol fermentation. Energies 2018, 11, 639. [Google Scholar] [CrossRef]
- Dziekońska-Kubczak, U.; Berłowska, J.; Dziugan, P.; Patelski, P.; Balcerek, M.; Pielech-Przybylska, K.; Robak, K. Two-stage pretreatment to improve saccharification of oat straw and Jerusalem artichoke biomass. Energies 2019, 12, 1715. [Google Scholar] [CrossRef]
- Trakulvichean, S.; Chaiprasert, P.; Otmakhova, J.; Songkasiri, W. Comparison of fermented animal feed and mushroom growth media as two value-added options for waste Cassava pulp management. Waste Manag. Res. 2017, 35, 1210–1219. [Google Scholar] [CrossRef]
- Farris, P.W.; Bendle, N.T.; Pfeifer, P.E.; Reibstein, D.J. Marketing and Finance, 1st ed.Pearson Education: Cranbury, NJ, USA, 2010; pp. 337–355. [Google Scholar]
- Sun, Y.; Zhang, J.P.; Yang, G.; Yao, M.S. Clean production of corn Stover pulp using KOH+NH 4OH solution and its kinetics during Delignification. Chem. Ind. Chem. Eng. Q. 2012, 18, 137–145. [Google Scholar] [CrossRef]
- Safarian, S.; Unnthorsson, R. An assessment of the sustainability of lignocellulosic bioethanol production from wastes in Iceland. Energies 2018, 11, 1493. [Google Scholar] [CrossRef]
- Gabriel Rullo, P.; Costa-Castelló, R.; Roda, V.; Feroldi, D. Energy management strategy for a bioethanol isolated hybrid system: Simulations and experiments. Energies 2018, 11, 1362. [Google Scholar] [CrossRef]
- Cantero, C.A.T.; Lopez, G.L.; Alvarado, V.M.; Jimenez, R.F.E.; Morales, J.Y.R.; Coronado, E.M.S. Control structures evaluation for a salt extractive distillation pilot plant: Application to bio-ethanol dehydration. Energies 2017, 10, 1276. [Google Scholar] [CrossRef]
| Component (Mass %) | SBP | GS |
|---|---|---|
| Ash | 5.1 | 8.9 |
| Cellulose | 15.1 | 28.8 |
| Extractives | n.a. | 0.5 |
| Fats | n.a. | n.a. |
| Hemicellulose | 18.2 | 18.8 |
| Lignin | 0.9 | 13.4 |
| Pectin solids | 20.8 | n.a. |
| Proteins | 10.3 | 4.7 |
| Trash | n.a. | 0.5 |
| Water | 29.6 | 7.0 |
| Dry matter content (%) | 70.4 | 92.5 |
| Process | Efficiency (%) |
|---|---|
| Cellulose hydrolysis in pretreatment section | 10 |
| Hemicellulose hydrolysis in pretreatment section | 70 |
| Cellulose hydrolysis in saccharification and fermentation section | 72 |
| Hemicellulose hydrolysis in saccharification and fermentation section | 72 |
| Fermentation hexose sugars | 90 |
| Fermentation pentose sugars | 87 |
| Process | Efficiency (%) |
|---|---|
| Cellulose hydrolysis in pretreatment section | 13 |
| Hemicellulose hydrolysis in pretreatment section | 60 |
| Cellulose hydrolysis in saccharification and fermentation section | 79 |
| Hemicellulose hydrolysis in saccharification and fermentation section | 80 |
| Fermentation hexose sugars | 95 |
| Fermentation pentose sugars | 70 |
| Plant Summary | Sugar Beet Pulp (SBP) | Grass Straw (GS) | ||
|---|---|---|---|---|
| Treated | Untreated | Treated | Untreated | |
| Total Capital Investment (€) | 2,584,000 | 1,931,000 | 39,450,000 | 1,993,000 |
| Capital Investment Charged to This Project (€) | 2,584,000 | 1,931,000 | 39,450,000 | 1,993,000 |
| Operation Cost (€/year) | 1,075,000 | 456,000 | 2,202,000 | 420,000 |
| Revenues (€/year) | 1,117,000 | 964,000 | 1,593,603 | 859,000 |
| Batch Size (kg) | 13,073 | - | 30,198 | - |
| Cost Basis Annual Production Rate (kg/year) | 1,424,990 | 1,228,939 | 1,962,890 | 1,094,616 |
| Conversion (%) | 14.25 | 19.63 | ||
| Unit Production Cost (€/kg) | 0.75 | 0.37 | 1.12 | 0.38 |
| Payback Time (years) | - | 5.09 | - | 5.85 |
| IRR (After Taxes) (%) | - | 23.05 | - | 19.30 |
| NPV (at 5% Interest) (€) | −671,000 | 3,367,000 | −10,543,000 | 2,763,000 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Achinas, S.; Leenders, N.; Krooneman, J.; Euverink, G.J.W. Feasibility Assessment of a Bioethanol Plant in the Northern Netherlands. Appl. Sci. 2019, 9, 4586. https://doi.org/10.3390/app9214586
Achinas S, Leenders N, Krooneman J, Euverink GJW. Feasibility Assessment of a Bioethanol Plant in the Northern Netherlands. Applied Sciences. 2019; 9(21):4586. https://doi.org/10.3390/app9214586
Chicago/Turabian StyleAchinas, Spyridon, Nienke Leenders, Janneke Krooneman, and Gerrit Jan Willem Euverink. 2019. "Feasibility Assessment of a Bioethanol Plant in the Northern Netherlands" Applied Sciences 9, no. 21: 4586. https://doi.org/10.3390/app9214586
APA StyleAchinas, S., Leenders, N., Krooneman, J., & Euverink, G. J. W. (2019). Feasibility Assessment of a Bioethanol Plant in the Northern Netherlands. Applied Sciences, 9(21), 4586. https://doi.org/10.3390/app9214586





