Review on Retrospective Procedures to Correct Retinal Motion Artefacts in OCT Imaging
Abstract
1. Introduction
2. Motion and Distortion Induced Artefacts
2.1. Optical Distortions
2.2. Involuntary Eye Motion
2.2.1. Tremors
2.2.2. Drifts
2.2.3. Microsaccades
2.2.4. Pulsing and Respiration
3. Methods
3.1. Literature Search
3.1.1. Inclusion Criteria
3.1.2. Exclusion Criteria
3.2. Overview
3.3. Motion Correction Strategies
3.3.1. Prospective Approaches
3.3.2. Retrospective Approaches
3.3.3. Input Data
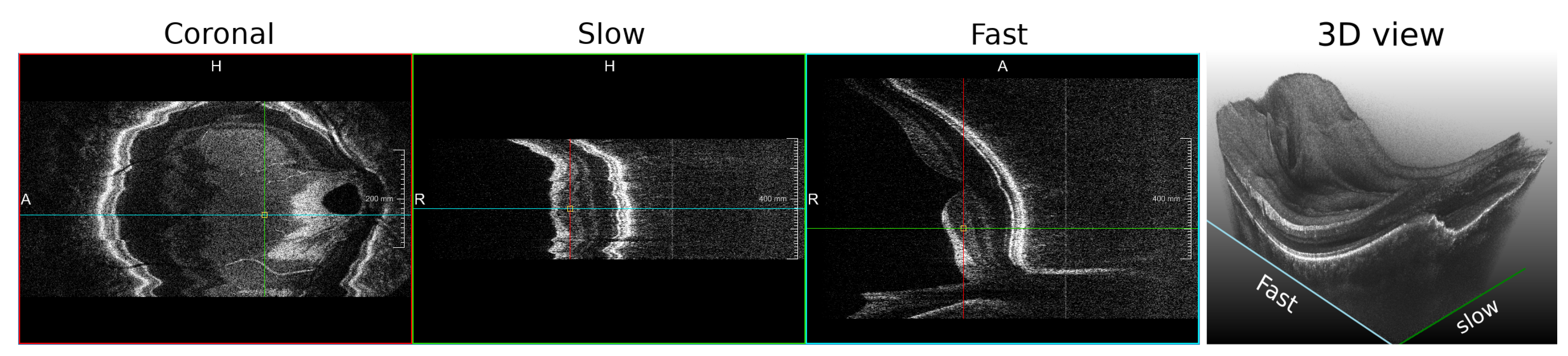
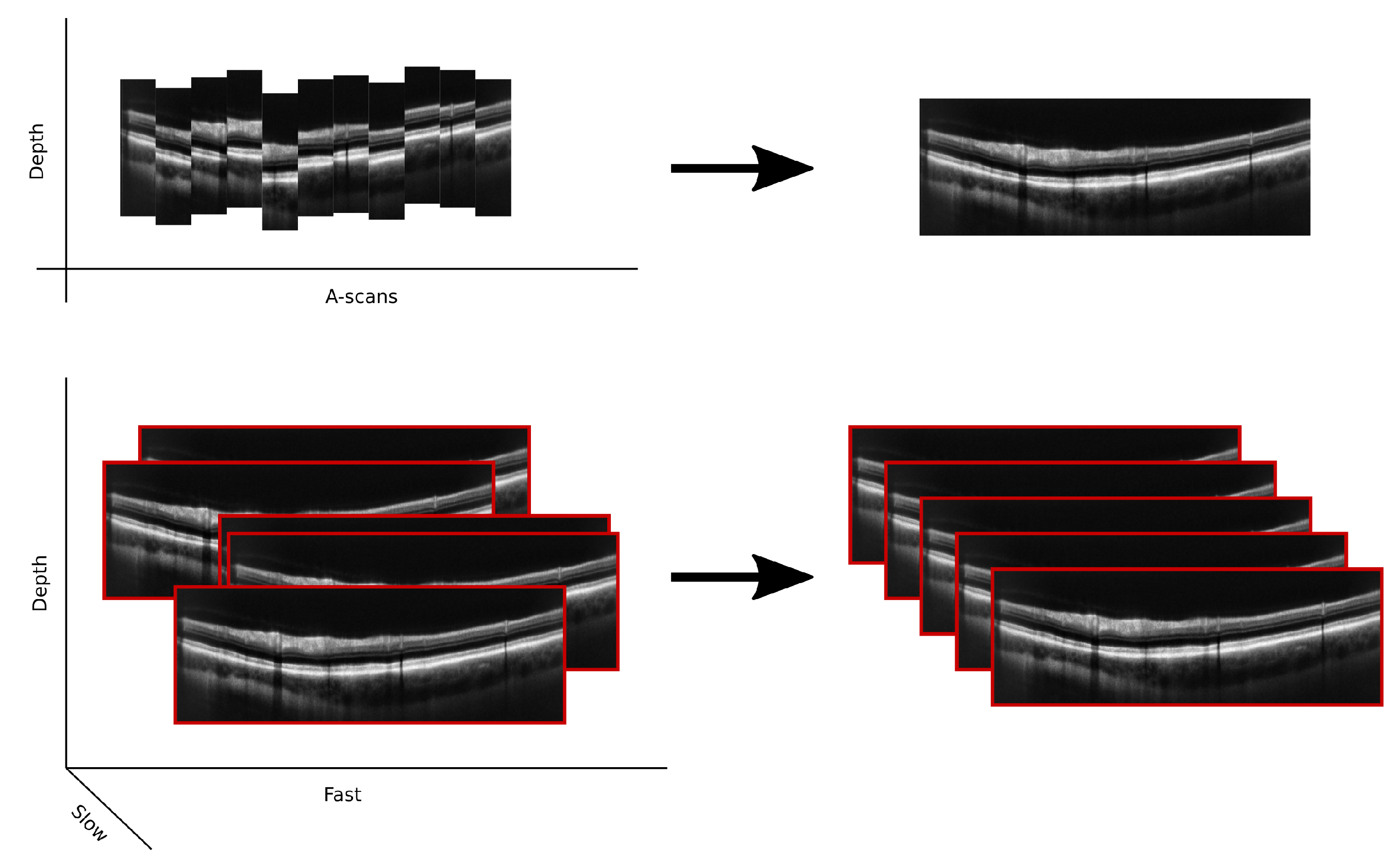
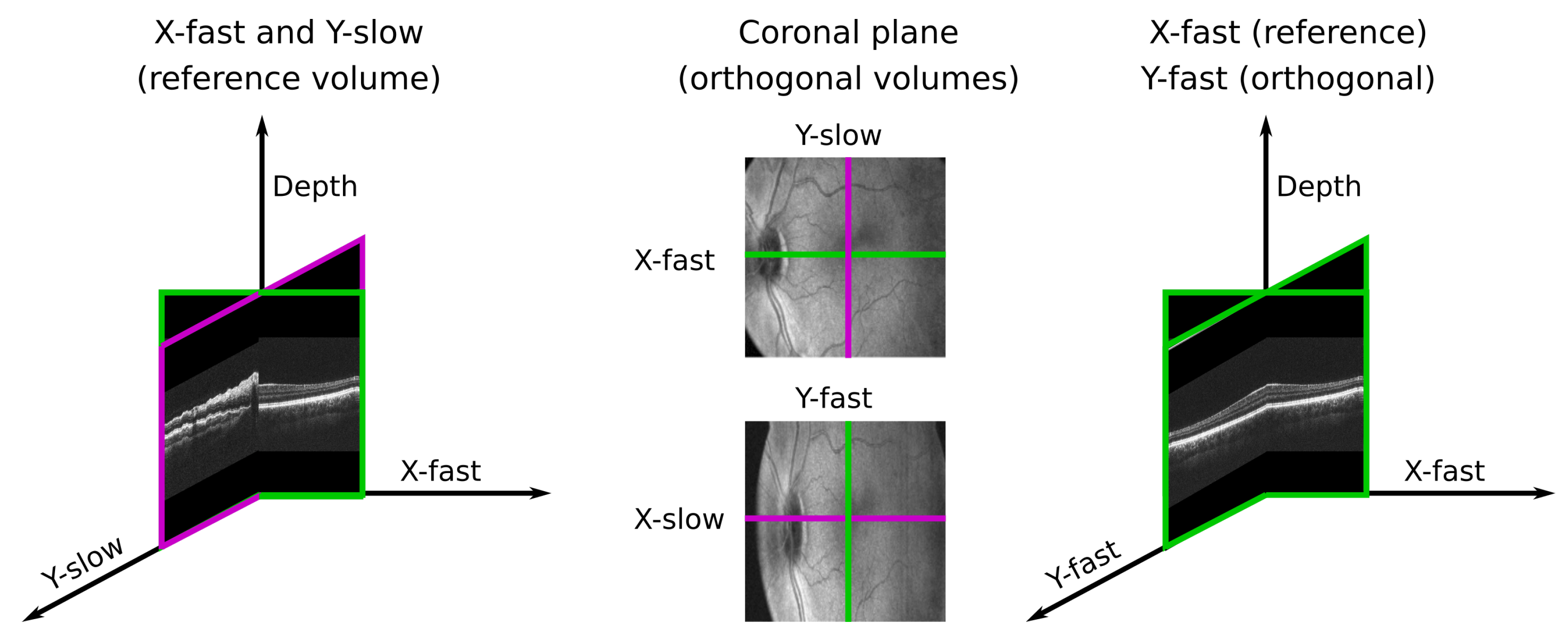
3.3.4. Handled Plane
3.4. Pre-Processing Techniques
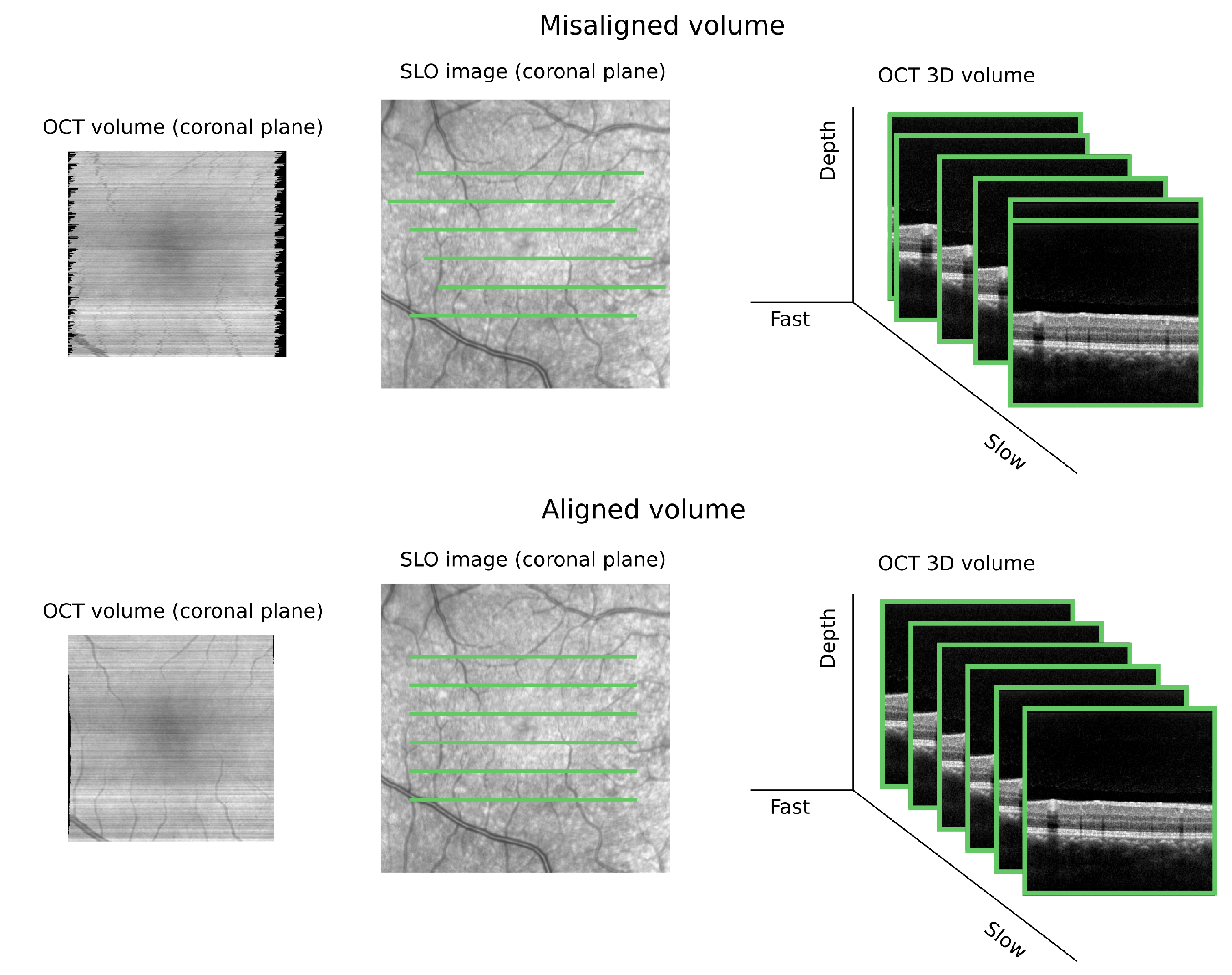
3.5. Validation Techniques
3.5.1. Qualitative Metrics
3.5.2. Quantitative Metrics
3.5.3. Simulated Data
3.5.4. Multimodality Comparison
4. Chronological Review of Retrospective Approaches
4.1. [1991–2005]
4.2. [2006–2008]
4.3. [2009–2011]
4.4. [2012–2014]
4.5. [2015–2018]
5. Discussion
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Huang, D.; Swanson, E.A.; Lin, C.P.; Schuman, J.S.; Stinson, W.G.; Chang, W.; Hee, M.R.; Flotte, T.; Gregory, K.; Puliafito, C.A.; et al. Optical coherence tomography. Science 1991, 254, 1178–1181. [Google Scholar] [CrossRef] [PubMed]
- Hitzenberger, C.K. Optical coherence tomography in Optics Express. Opt. Express 2018, 26, 24240–24259. [Google Scholar] [CrossRef] [PubMed]
- Fujimoto, J.G. Optical coherence tomography for ultrahigh resolution in vivo imaging. Nat. Biotechnol. 2003, 21, 1361. [Google Scholar] [CrossRef] [PubMed]
- Drexler, W.; Fujimoto, J. Optical Coherence Tomography: Technology and Applications; Springer International Publishing: Berlin, Germany, 2015. [Google Scholar]
- Drexler, W.; Fujimoto, J.G. State-of-the-art retinal optical coherence tomography. Prog. Retin. Eye Res. 2008, 27, 45–88. [Google Scholar] [CrossRef] [PubMed]
- Swanson, E.A.; Fujimoto, J.G. The ecosystem that powered the translation of OCT from fundamental research to clinical and commercial impact. Biomed. Opt. Express 2017, 8, 1638–1664. [Google Scholar] [CrossRef] [PubMed]
- Wieser, W.; Biedermann, B.R.; Klein, T.; Eigenwillig, C.M.; Huber, R. Multi-megahertz OCT: High quality 3D imaging at 20 million A-scans and 4.5 GVoxels per second. Opt. Express 2010, 18, 14685–14704. [Google Scholar] [CrossRef] [PubMed]
- Potsaid, B.; Baumann, B.; Huang, D.; Barry, S.; Cable, A.E.; Schuman, J.S.; Duker, J.S.; Fujimoto, J.G. Ultrahigh speed 1050 nm Swept Source Fourier Domain OCT retinal and anterior segment imaging at 100,000 to 400,000 axial scans per second. Opt. Express 2010, 18, 20029–20048. [Google Scholar] [CrossRef]
- Potsaid, B.; Gorczynska, I.; Srinivasan, V.J.; Chen, Y.; Jiang, J.; Cable, A.; Fujimoto, J.G. Ultrahigh speed spectral/Fourier domain OCT ophthalmic imaging at 70,000 to 312,500 axial scans per second. Opt. Express 2008, 16, 15149–15169. [Google Scholar] [CrossRef]
- Marschall, S.; Sander, B.; Mogensen, M.; Jørgensen, T.M.; Andersen, P.E. Optical coherence tomography—Current technology and applications in clinical and biomedical research. Anal. Bioanal. Chem. 2011, 400, 2699–2720. [Google Scholar] [CrossRef]
- He, Y.; Wei, L.; Wang, Z.; Yang, J.; Li, X.; Shi, G.; Zhang, Y. Extraction of ultra-high frequency retinal motions with a line scanning quasi-confocal ophthalmoscope. J. Opt. 2014, 17, 015301. [Google Scholar] [CrossRef]
- Martinez-Conde, S.; Macknik, S.L.; Hubel, D.H. The role of fixational eye movements in visual perception. Nat. Rev. Neurosci. 2004, 5, 229. [Google Scholar] [CrossRef] [PubMed]
- Spaide, R.F.; Fujimoto, J.G.; Waheed, N.K. Image artifacts in optical coherence angiography. Retina 2015, 35, 2163. [Google Scholar] [CrossRef] [PubMed]
- Pircher, M.; Baumann, B.; Götzinger, E.; Sattmann, H.; Hitzenberger, C.K. Simultaneous SLO/OCT imaging of the human retina with axial eye motion correction. Opt. Express 2007, 15, 16922–16932. [Google Scholar] [CrossRef] [PubMed]
- Baghaie, A.; Yu, Z.; D’souza, R.M. State-of-the-art in retinal optical coherence tomography image analysis. Quant. Imaging Med. Surg. 2015, 5, 603. [Google Scholar] [PubMed]
- Baghaie, A.; Yu, Z.; D’souza, R.M. Involuntary eye motion correction in retinal optical coherence tomography: Hardware or software solution? Med. Image Anal. 2017, 37, 129–145. [Google Scholar] [CrossRef] [PubMed]
- Westphal, V.; Rollins, A.M.; Radhakrishnan, S.; Izatt, J.A. Correction of geometric and refractive image distortions in optical coherence tomography applying Fermat’s principle. Opt. Express 2002, 10, 397–404. [Google Scholar] [CrossRef] [PubMed]
- Podoleanu, A.; Charalambous, I.; Plesea, L.; Dogariu, A.; Rosen, R. Correction of distortions in optical coherence tomography imaging of the eye. Phys. Med. Biol. 2004, 49, 1277. [Google Scholar] [CrossRef] [PubMed]
- Zawadzki, R.J.; Fuller, A.R.; Choi, S.S.; Wiley, D.F.; Hamann, B.; Werner, J.S. Correction of motion artifacts and scanning beam distortions in 3D ophthalmic optical coherence tomography imaging. In Proceedings of the Ophthalmic Technologies XVII, San Jose, CA, USA, 20–25 January 2007; Volume 6426, p. 642607. [Google Scholar]
- Jesus, D.A.; Iskander, D.R. Simplifying numerical ray tracing for two-dimensional non circularly symmetric models of the human eye. Appl. Opt. 2015, 54, 10123–10127. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Ishikawa, H.; Wollstein, G.; Kagemann, L.; Schuman, J.S. Alignment of 3D optical coherence tomography scans to correct eye movement using a particle filtering. IEEE Trans. Med. Imaging 2012, 31, 1337–1345. [Google Scholar] [CrossRef]
- Salas, M.; Augustin, M.; Felberer, F.; Wartak, A.; Laslandes, M.; Ginner, L.; Niederleithner, M.; Ensher, J.; Minneman, M.P.; Leitgeb, R.A.; et al. Compact akinetic swept source optical coherence tomography angiography at 1060 nm supporting a wide field of view and adaptive optics imaging modes of the posterior eye. Biomed. Opt. Express 2018, 9, 1871–1892. [Google Scholar] [CrossRef]
- Martinez-Conde, S. Fixational eye movements in normal and pathological vision. Prog. Brain Res. 2006, 154, 151–176. [Google Scholar] [PubMed]
- Spauschus, A.; Marsden, J.; Halliday, D.M.; Rosenberg, J.R.; Brown, P. The origin of ocular microtremor in man. Exp. Brain Res. 1999, 126, 556–562. [Google Scholar] [CrossRef] [PubMed]
- Pircher, M.; Zawadzki, R.J. Review of adaptive optics OCT (AO-OCT): Principles and applications for retinal imaging. Biomed. Opt. Express 2017, 8, 2536–2562. [Google Scholar] [CrossRef] [PubMed]
- Jonnal, R.S.; Kocaoglu, O.P.; Zawadzki, R.J.; Liu, Z.; Miller, D.T.; Werner, J.S. A review of adaptive optics optical coherence tomography: Technical advances, scientific applications, and the future. Investig. Ophthalmol. Vis. Sci. 2016, 57, OCT51–OCT68. [Google Scholar] [CrossRef] [PubMed]
- Yarbus, A.L. Eye movements during perception of complex objects. In Eye Movements and Vision; Springer: Berlin, Germany, 1967; pp. 171–211. [Google Scholar]
- Srebro, R. Fixation of normal and amblyopic eyes. Arch. Ophthalmol. 1983, 101, 214–217. [Google Scholar] [CrossRef] [PubMed]
- Schulz, E. Binocular micromovements in normal persons. Graefe’s Arch. Clin. Exp. Ophthalmol. 1984, 222, 95–100. [Google Scholar] [CrossRef]
- Riggs, L.A.; Armington, J.C.; Ratliff, F. Motions of the retinal image during fixation. Opt. Soc. Am. 1954, 44, 315–321. [Google Scholar] [CrossRef]
- Møller, F.; Laursen, M.; Tygesen, J.; Sjølie, A. Binocular quantification and characterization of microsaccades. Graefe’s Arch. Clin. Exp. Ophthalmol. 2002, 240, 765–770. [Google Scholar] [CrossRef]
- Mustafi, D.; Engel, A.H.; Palczewski, K. Structure of cone photoreceptors. Prog. Retin. Eye Res. 2009, 28, 289–302. [Google Scholar] [CrossRef]
- Robinson, M.D.; Chiu, S.J.; Lo, J.; Toth, C.; Izatt, J.; Farsiu, S. New applications of super-resolution in medical imaging. Super-Resolut. Imaging 2010, 2010, 384–412. [Google Scholar]
- De Kinkelder, R.; Kalkman, J.; Faber, D.J.; Schraa, O.; Kok, P.H.; Verbraak, F.D.; van Leeuwen, T.G. Heartbeat-induced axial motion artifacts in optical coherence tomography measurements of the retina. Investig. Ophthalmol. Vis. Sci. 2011, 52, 3908–3913. [Google Scholar] [CrossRef] [PubMed]
- Kiernan, D.F.; Mieler, W.F.; Hariprasad, S.M. Spectral-domain optical coherence tomography: A comparison of modern high-resolution retinal imaging systems. Am. J. Ophthalmol. 2010, 149, 18–31. [Google Scholar] [CrossRef] [PubMed]
- Brautaset, R.; Birkeldh, U.; Alstig, P.F.; Wikén, P.; Nilsson, M. Repeatability using automatic tracing with Canon OCT-HS100 and Zeiss Cirrus HD-OCT 5000. PLoS ONE 2016, 11, e0149138. [Google Scholar] [CrossRef] [PubMed]
- Giani, A.; Pellegrini, M.; Invernizzi, A.; Cigada, M.; Staurenghi, G. Aligning scan locations from consecutive spectral-domain optical coherence tomography examinations: A comparison among different strategies. Investig. Ophthalmol. Vis. Sci. 2012, 53, 7637–7643. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ferguson, R.D.; Hammer, D.X.; Paunescu, L.A.; Beaton, S.; Schuman, J.S. Tracking optical coherence tomography. Opt. Lett. 2004, 29, 2139–2141. [Google Scholar] [CrossRef]
- Hammer, D.X.; Ferguson, R.D.; Magill, J.C.; Paunescu, L.A.; Beaton, S.; Ishikawa, H.; Wollstein, G.; Schuman, J.S. Active retinal tracker for clinical optical coherence tomography systems. J. Biomed. Opt. 2005, 10, 024038. [Google Scholar] [CrossRef] [PubMed]
- Vienola, K.V.; Braaf, B.; Sheehy, C.K.; Yang, Q.; Tiruveedhula, P.; Arathorn, D.W.; de Boer, J.F.; Roorda, A. Real-time eye motion compensation for OCT imaging with tracking SLO. Biomed. Opt. Express 2012, 3, 2950–2963. [Google Scholar] [CrossRef] [PubMed]
- Sugita, M.; Zotter, S.; Pircher, M.; Makihira, T.; Saito, K.; Tomatsu, N.; Sato, M.; Roberts, P.; Schmidt-Erfurth, U.; Hitzenberger, C.K. Motion artifact and speckle noise reduction in polarization sensitive optical coherence tomography by retinal tracking. Biomed. Opt. Express 2014, 5, 106–122. [Google Scholar] [CrossRef] [PubMed]
- Hajnal, J.V.; Hill, D.L. Medical Image Registration; CRC Press: Boca Raton, FL, USA, 2001. [Google Scholar]
- Klein, S.; Staring, M.; Murphy, K.; Viergever, M.A.; Pluim, J.P. Elastix: A toolbox for intensity-based medical image registration. IEEE Trans. Med. Imaging 2010, 29, 196–205. [Google Scholar] [CrossRef]
- Avants, B.B.; Epstein, C.L.; Grossman, M.; Gee, J.C. Symmetric diffeomorphic image registration with cross-correlation: Evaluating automated labeling of elderly and neurodegenerative brain. Med. Image Anal. 2008, 12, 26–41. [Google Scholar] [CrossRef]
- Maes, F.; Collignon, A.; Vandermeulen, D.; Marchal, G.; Suetens, P. Multimodality image registration by maximization of mutual information. IEEE Trans. Med. Imaging 1997, 16, 187–198. [Google Scholar] [CrossRef] [PubMed]
- Metz, C.; Klein, S.; Schaap, M.; van Walsum, T.; Niessen, W.J. Nonrigid registration of dynamic medical imaging data using nD+t B-splines and a groupwise optimization approach. Med. Image Anal. 2011, 15, 238–249. [Google Scholar] [CrossRef] [PubMed]
- Peli, E.; Augliere, R.A.; Timberlake, G.T. Feature-based registration of retinal images. IEEE Trans. Med. Imaging 1987, 6, 272–278. [Google Scholar] [CrossRef] [PubMed]
- Vogel, C.R.; Arathorn, D.W.; Roorda, A.; Parker, A. Retinal motion estimation in adaptive optics scanning laser ophthalmoscopy. Opt. Express 2006, 14, 487–497. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Gerendas, B.S.; Waldstein, S.M.; Langs, G.; Simader, C.; Schmidt-Erfurth, U. Stable registration of pathological 3D-OCT scans using retinal vessels. In Proceedings of the Ophthalmic Medical Image Analysis First International Workshop, Boston, MA, USA, 14 September 2014; pp. 1–8. [Google Scholar]
- Cheng, J.; Lee, J.A.; Xu, G.; Quan, Y.; Ong, E.P.; Wong, K.; Wing, D. Motion Correction in Optical Coherence Tomography for Multi-modality Retinal Image Registration. In Proceedings of the Ophthalmic Medical Image Analysis Third International Workshop, Athens, Greece, 17–21 October 2016. [Google Scholar]
- Anantrasirichai, N.; Nicholson, L.; Morgan, J.E.; Erchova, I.; Mortlock, K.; North, R.V.; Albon, J.; Achim, A. Adaptive-weighted bilateral filtering and other pre-processing techniques for optical coherence tomography. Comput. Med. Imaging Graph. 2014, 38, 526–539. [Google Scholar] [CrossRef] [PubMed]
- Dice, L.R. Measures of the amount of ecologic association between species. Ecology 1945, 26, 297–302. [Google Scholar] [CrossRef]
- Swanson, E.A.; Izatt, J.A.; Hee, M.R.; Huang, D.; Lin, C.; Schuman, J.; Puliafito, C.; Fujimoto, J.G. In vivo retinal imaging by optical coherence tomography. Opt. Lett. 1993, 18, 1864–1866. [Google Scholar] [CrossRef] [PubMed]
- Schuman, J.S.; Pedut-Kloizman, T.; Hertzmark, E.; Hee, M.R.; Wilkins, J.R.; Coker, J.G.; Puliafito, C.A.; Fujimoto, J.G.; Swanson, E.A. Reproducibility of nerve fiber layer thickness measurements using optical coherence tomography. Ophthalmology 1996, 103, 1889–1898. [Google Scholar] [CrossRef]
- Ishikawa, H.; Stein, D.M.; Wollstein, G.; Beaton, S.; Fujimoto, J.G.; Schuman, J.S. Macular segmentation with optical coherence tomography. Investig. Ophthalmol. Vis. Sci. 2005, 46, 2012–2017. [Google Scholar] [CrossRef]
- Jørgensen, T.M.; Thomadsen, J.; Christensen, U.; Soliman, W.; Sander, B.A. Enhancing the signal-to-noise ratio in ophthalmic optical coherence tomography by image registration—Method and clinical examples. J. Biomed. Opt. 2007, 12, 041208. [Google Scholar] [CrossRef]
- Fuller, A.; Zawadzki, R.; Choi, S.; Wiley, D.; Werner, J.; Hamann, B. Segmentation of three-dimensional retinal image data. IEEE Trans. Vis. Comput. Graph. 2007, 13, 1719–1726. [Google Scholar] [CrossRef] [PubMed]
- Khanifar, A.A.; Koreishi, A.F.; Izatt, J.A.; Toth, C.A. Drusen ultrastructure imaging with spectral domain optical coherence tomography in age-related macular degeneration. Ophthalmology 2008, 115, 1883–1890. [Google Scholar] [CrossRef] [PubMed]
- Ricco, S.; Chen, M.; Ishikawa, H.; Wollstein, G.; Schuman, J. Correcting motion artifacts in retinal spectral domain optical coherence tomography via image registration. In Proceedings of the International Conference on Medical Image Computing and Computer-Assisted Intervention, London, UK, 20–24 September 2009; pp. 100–107. [Google Scholar]
- Niemeijer, M.; Garvin, M.K.; Lee, K.; van Ginneken, B.; Abràmoff, M.D.; Sonka, M. Registration of 3D spectral OCT volumes using 3D SIFT feature point matching. In Proceedings of the Medical Imaging 2009: Image Processing, Lake Buena Vista, FL, USA, 7–12 February 2009; Volume 7259, p. 72591I. [Google Scholar]
- Garvin, M.K.; Abramoff, M.D.; Wu, X.; Russell, S.R.; Burns, T.L.; Sonka, M. Automated 3D intraretinal layer segmentation of macular spectral-domain optical coherence tomography images. IEEE Trans. Med. Imaging 2009, 28, 1436–1447. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Ishikawa, H.; Tolliver, D.; Wollstein, G.; Miller, G.; Bilonick, R.; Kagemann, L.; Schuman, J. Shape context algorithm applied to correct eye movement artifacts on three-dimensional (3D) spectral domain optical coherence tomography (SD-OCT). Investig. Ophthalmol. Vis. Sci. 2009, 50, 1104. [Google Scholar]
- Tolliver, D.; Ishikawa, H.; Schuman, J.; Miller, G. An in-painting method for combining multiple SD-OCT scans with applications to Z-motion recovery, noise reduction and longitudinal studies. Investig. Ophthalmol. Vis. Sci. 2009, 50, 1100. [Google Scholar]
- Kolar, R.; Tasevsky, P. Registration of 3D retinal optical coherence tomography data and 2D fundus images. In International Workshop on Biomedical Image Registration; Springer: Berlin, Germany, 2010; pp. 72–82. [Google Scholar]
- Gibson, E.; Young, M.; Sarunic, M.V.; Beg, M.F. Optic nerve head registration via hemispherical surface and volume registration. IEEE Trans. Biomed. Eng. 2010, 57, 2592–2595. [Google Scholar] [CrossRef]
- Xu, J.; Ishikawa, H.; Wollstein, G.; Schuman, J.S. 3D OCT eye movement correction based on particle filtering. In Proceedings of the 2010 Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Buenos Aires, Argentina, 31 August–4 September 2010; pp. 53–56. [Google Scholar]
- Antony, B.; Abramoff, M.D.; Tang, L.; Ramdas, W.D.; Vingerling, J.R.; Jansonius, N.M.; Lee, K.; Kwon, Y.H.; Sonka, M.; Garvin, M.K. Automated 3D method for the correction of axial artifacts in spectral-domain optical coherence tomography images. Biomed. Opt. Express 2011, 2, 2403–2416. [Google Scholar] [CrossRef] [PubMed]
- Song, X.; Estrada, R.; Chiu, S.J.; Dhalla, A.H.; Toth, C.A.; Izatt, J.A.; Farsiu, S. Segmentation-based registration of retinal optical coherence tomography images with pathology. Investig. Ophthalmol. Vis. Sci. 2011, 52, 1309. [Google Scholar]
- Li, Y.; Gregori, G.; Lam, B.L.; Rosenfeld, P.J. Automatic montage of SD-OCT data sets. Opt. Express 2011, 19, 26239–26248. [Google Scholar] [CrossRef]
- Niemeijer, M.; Lee, K.; Garvin, M.K.; Abràmoff, M.D.; Sonka, M. Registration of 3D spectral OCT volumes combining ICP with a graph-based approach. In Proceedings of the Medical Imaging 2012: Image Processing, San Diego, CA, USA, 4–9 February 2012; Volume 8314, p. 83141A. [Google Scholar]
- Kraus, M.F.; Potsaid, B.; Mayer, M.A.; Bock, R.; Baumann, B.; Liu, J.J.; Hornegger, J.; Fujimoto, J.G. Motion correction in optical coherence tomography volumes on a per A-scan basis using orthogonal scan patterns. Biomed. Opt. Express 2012, 3, 1182–1199. [Google Scholar] [CrossRef]
- He, H.; Liu, G.; Mo, P.; Li, B.; Wu, J.; Ding, X. Correction of motion artifact in 3d retinal optical coherence tomography imaging. In Proceedings of the 2013 6th International Congress on Image and Signal Processing (CISP), Hangzhou, China, 16–18 December 2013; Volume 1, pp. 261–265. [Google Scholar]
- Chen, M.; Lang, A.; Sotirchos, E.; Ying, H.S.; Calabresi, P.A.; Prince, J.L.; Carass, A. Deformable registration of macular OCT using A-mode scan similarity. In Proceedings of the 2013 IEEE 10th International Symposium on Biomedical Imaging (ISBI), San Francisco, CA, USA, 7–11 April 2013; pp. 476–479. [Google Scholar]
- Zheng, Y.; Xiao, R.; Wang, Y.; Gee, J.C. A generative model for OCT retinal layer segmentation by integrating graph-based multi-surface searching and image registration. In Proceedings of the International Conference on Medical Image Computing and Computer-Assisted Intervention, Nagoya, Japan, 22–26 September 2013; Springer: Berlin, Germany, 2013; pp. 428–435. [Google Scholar]
- Hendargo, H.C.; Estrada, R.; Chiu, S.J.; Tomasi, C.; Farsiu, S.; Izatt, J.A. Automated non-rigid registration and mosaicing for robust imaging of distinct retinal capillary beds using speckle variance optical coherence tomography. Biomed. Opt. Express 2013, 4, 803–821. [Google Scholar] [CrossRef] [PubMed]
- LaRocca, F.; Nankivil, D.; Farsiu, S.; Izatt, J.A. Handheld simultaneous scanning laser ophthalmoscopy and optical coherence tomography system. Biomed. Opt. Express 2013, 4, 2307–2321. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Lang, A.; Ying, H.S.; Calabresi, P.A.; Prince, J.L.; Carass, A. Analysis of macular OCT images using deformable registration. Biomed. Opt. Express 2014, 5, 2196–2214. [Google Scholar] [CrossRef] [PubMed]
- Kraus, M.F.; Liu, J.J.; Schottenhamml, J.; Chen, C.L.; Budai, A.; Branchini, L.; Ko, T.; Ishikawa, H.; Wollstein, G.; Schuman, J.; et al. Quantitative 3D-OCT motion correction with tilt and illumination correction, robust similarity measure and regularization. Biomed. Opt. Express 2014, 5, 2591–2613. [Google Scholar] [CrossRef] [PubMed]
- Montuoro, A.; Wu, J.; Waldstein, S.; Gerendas, B.; Langs, G.; Simader, C.; Schmidt-Erfurth, U. Motion artefact correction in retinal optical coherence tomography using local symmetry. In Proceedings of the International Conference on Medical Image Computing and Computer-Assisted Intervention, Boston, MA, USA, 14–18 September 2014; Springer: Berlin, Germany, 2014; pp. 130–137. [Google Scholar]
- Lee, S.; Lebed, E.; Sarunic, M.V.; Beg, M.F. Exact surface registration of retinal surfaces from 3-d optical coherence tomography images. IEEE Trans. Biomed. Eng. 2015, 62, 609–617. [Google Scholar] [CrossRef] [PubMed]
- Lang, A.; Carass, A.; Al-Louzi, O.; Bhargava, P.; Solomon, S.D.; Calabresi, P.A.; Prince, J.L. Combined registration and motion correction of longitudinal retinal OCT data. In Proceedings of the Medical Imaging 2016: Image Processing, San Diego, CA, USA, 27 February–3 March 2016; Volume 9784, p. 97840X. [Google Scholar]
- Lezama, J.; Mukherjee, D.; McNabb, R.P.; Sapiro, G.; Kuo, A.N.; Farsiu, S. Segmentation guided registration of wide field-of-view retinal optical coherence tomography volumes. Biomed. Opt. Express 2016, 7, 4827. [Google Scholar] [CrossRef]
- Fu, H.; Xu, Y.; Wong, D.W.K.; Liu, J. Eye movement correction for 3D OCT volume by using saliency and center bias constraint. In Proceedings of the 2016 IEEE Region 10 Conference (TENCON), Singapore, 22–25 November 2016; pp. 1536–1539. [Google Scholar]
- Chen, Y.; Hong, Y.J.; Makita, S.; Yasuno, Y. Three-dimensional eye motion correction by Lissajous scan optical coherence tomography. Biomed. Opt. Express 2017, 8, 1783–1802. [Google Scholar] [CrossRef]
- Fercher, A.F. In vivo optical coherence tomography. Am. J. Ophthalmol. 1993, 116, 113–114. [Google Scholar] [CrossRef]
- Lam, B.S.Y.; Yan, H. A novel vessel segmentation algorithm for pathological retina images based on the divergence of vector fields. IEEE Trans. Med. Imaging 2008, 27, 237–246. [Google Scholar] [CrossRef]
- Periaswamy, S.; Farid, H. Elastic registration in the presence of intensity variations. IEEE Trans. Med. Imaging 2003, 22, 865–874. [Google Scholar] [CrossRef]
- Sakoe, H.; Chiba, S. Dynamic programming algorithm optimization for spoken word recognition. IEEE Trans. Acoust. Speech Signal Process. 1978, 26, 43–49. [Google Scholar] [CrossRef]
- Lowe, D.G. Object recognition from local scale-invariant features. In Proceedings of the Proceedings of the Seventh IEEE International Conference on Computer Vision, Kerkyra, Greece, 20–27 September 1999; Volume 2, pp. 1150–1157. [Google Scholar]
- Rohde, G.K.; Aldroubi, A.; Dawant, B.M. The adaptive bases algorithm for intensity-based nonrigid image registration. IEEE Trans. Med. Imaging 2003, 22, 1470–1479. [Google Scholar] [CrossRef]
- Myronenko, A.; Song, X. Point set registration: Coherent point drift. IEEE Trans. Pattern Anal. Mach. Intell. 2010, 32, 2262–2275. [Google Scholar] [CrossRef] [PubMed]
- Dabov, K.; Foi, A.; Katkovnik, V.; Egiazarian, K. Image denoising by sparse 3D transform-domain collaborative filtering. IEEE Trans. Image Process. 2007, 16, 2080–2095. [Google Scholar] [CrossRef] [PubMed]
- Yuille, A.L.; Grzywacz, N.M. The motion coherence theory. In Proceedings of the 1988 Second International Conference on Computer Vision, Tampa, FL, USA, 5–8 December 1988; pp. 344–353. [Google Scholar]
- Dubuisson, M.P.; Jain, A.K. A modified Hausdorff distance for object matching. In Proceedings of the 12th International Conference on Pattern Recognition, Jerusalem, Israel, 9–13 October 1994; pp. 566–568. [Google Scholar]
- Zhu, S.; Ma, K.K. A new diamond search algorithm for fast block-matching motion estimation. IEEE Trans. Image Process. 2000, 9, 287–290. [Google Scholar] [CrossRef]
- Lee, J.A.; Cheng, J.; Xu, G.; Ong, E.P.; Lee, B.H.; Wong, D.W.K.; Liu, J. Registration of color and OCT fundus images using low-dimensional step pattern analysis. In Proceedings of the International Conference on Medical Image Computing and Computer-Assisted Intervention, Munich, Germany, 5–9 October 2015; pp. 214–221. [Google Scholar]
- Fischler, M.A.; Bolles, R.C. Random sample consensus: A paradigm for model fitting with applications to image analysis and automated cartography. Commun. ACM 1981, 24, 381–395. [Google Scholar] [CrossRef]
- Chen, Y.; Hong, Y.J.; Makita, S.; Yasuno, Y. Eye-motion-corrected optical coherence tomography angiography using Lissajous scanning. Biomed. Opt. Express 2018, 9, 1111–1129. [Google Scholar] [CrossRef]
- Jesus, D.A.; Breda, J.B.; Van Keer, K.; Sousa, A.R.; Pinto, L.A.; Stalmans, I. Quantitative automated circumpapillary microvascular density measurements: A new angioOCT-based methodology. Eye 2018, 33, 320. [Google Scholar] [CrossRef]
- DeBuc, D.C.; Somfai, G.M. Early detection of retinal thickness changes in diabetes using optical coherence tomography. Med. Sci. Monit. 2010, 16, MT15–MT21. [Google Scholar]
- Barry, R.J.; Tasiopoulou, A.; Murray, P.I.; Patel, P.J.; Sagoo, M.S.; Denniston, A.K.; Keane, P.A. Characteristic optical coherence tomography findings in patients with primary vitreoretinal lymphoma: A novel aid to early diagnosis. Br. J. Ophthalmol. 2018, 102, 1362–1366. [Google Scholar] [CrossRef]
- Hart, N.J.; Koronyo, Y.; Black, K.L.; Koronyo-Hamaoui, M. Ocular indicators of Alzheimer’s: Exploring disease in the retina. Acta Neuropathol. 2016, 132, 767–787. [Google Scholar] [CrossRef] [PubMed]
- Grinvald, A.; Nelson, D.A.; Vanzetta, I. Characterization of Arteriosclerosis by Optical Imaging. U.S. Patent 8,521,260, 27 August 2013. [Google Scholar]
- Pircher, M.; Götzinger, E.; Sattmann, H.; Leitgeb, R.A.; Hitzenberger, C.K. In vivo investigation of human cone photoreceptors with SLO/OCT in combination with 3D motion correction on a cellular level. Opt. Express 2010, 18, 13935–13944. [Google Scholar] [CrossRef] [PubMed]
- Kocaoglu, O.P.; Ferguson, R.D.; Jonnal, R.S.; Liu, Z.; Wang, Q.; Hammer, D.X.; Miller, D.T. Adaptive optics optical coherence tomography with dynamic retinal tracking. Biomed. Opt. Express 2014, 5, 2262–2284. [Google Scholar] [CrossRef] [PubMed]
- Kolb, J.P.; Klein, T.; Kufner, C.L.; Wieser, W.; Neubauer, A.S.; Huber, R. Ultra-widefield retinal MHz-OCT imaging with up to 100 degrees viewing angle. Biomed. Opt. Express 2015, 6, 1534–1552. [Google Scholar] [CrossRef] [PubMed]
- Zang, P.; Liu, G.; Zhang, M.; Dongye, C.; Wang, J.; Pechauer, A.D.; Hwang, T.S.; Wilson, D.J.; Huang, D.; Li, D.; et al. Automated motion correction using parallel-strip registration for wide-field en face OCT angiogram. Biomed. Opt. Expressf 2016, 7, 2823–2836. [Google Scholar] [CrossRef]
- Chen, F.K.; Viljoen, R.D.; Bukowska, D.M. Classification of image artefacts in optical coherence tomography angiography of the choroid in macular diseases. Clin. Exp. Ophthalmol. 2016, 44, 388–399. [Google Scholar] [CrossRef] [PubMed]
- Hwang, T.S.; Gao, S.S.; Liu, L.; Lauer, A.K.; Bailey, S.T.; Flaxel, C.J.; Wilson, D.J.; Huang, D.; Jia, Y. Automated quantification of capillary nonperfusion using optical coherence tomography angiography in diabetic retinopathy. JAMA Ophthalmol. 2016, 134, 367–373. [Google Scholar] [CrossRef]
- Zhang, M.; Wang, J.; Pechauer, A.D.; Hwang, T.S.; Gao, S.S.; Liu, L.; Liu, L.; Bailey, S.T.; Wilson, D.J.; Huang, D.; et al. Advanced image processing for optical coherence tomographic angiography of macular diseases. Biomed. Opt. Express 2015, 6, 4661–4675. [Google Scholar] [CrossRef]
- Liu, L.; Gao, S.S.; Bailey, S.T.; Huang, D.; Li, D.; Jia, Y. Automated choroidal neovascularization detection algorithm for optical coherence tomography angiography. Biomed. Opt. Express 2015, 6, 3564–3576. [Google Scholar] [CrossRef]
- Liu, L.; Jia, Y.; Takusagawa, H.L.; Pechauer, A.D.; Edmunds, B.; Lombardi, L.; Davis, E.; Morrison, J.C.; Huang, D. Optical coherence tomography angiography of the peripapillary retina in glaucoma. JAMA Ophthalmol. 2015, 133, 1045–1052. [Google Scholar] [CrossRef]
- Jia, Y.; Bailey, S.T.; Hwang, T.S.; McClintic, S.M.; Gao, S.S.; Pennesi, M.E.; Flaxel, C.J.; Lauer, A.K.; Wilson, D.J.; Hornegger, J.; et al. Quantitative optical coherence tomography angiography of vascular abnormalities in the living human eye. Proc. Natl. Acad. Sci. USA 2015, 112, E2395–E2402. [Google Scholar] [CrossRef] [PubMed]
- Sotiras, A.; Davatzikos, C.; Paragios, N. Deformable medical image registration: A survey. IEEE Trans. Med. Imaging 2013, 32, 1153. [Google Scholar] [CrossRef] [PubMed]
- Johnson, H.J.; McCormick, M.M.; Ibanez, L. The ITK Software Guide: Design and Functionality; Kitware, Inc.: Clifton Park, NY, USA, 2015. [Google Scholar]
- Avants, B.B.; Tustison, N.J.; Stauffer, M.; Song, G.; Wu, B.; Gee, J.C. The Insight ToolKit image registration framework. Front. Neuroinform. 2014, 8, 44. [Google Scholar] [CrossRef] [PubMed]
- Tustison, N.J. Explicit B-spline regularization in diffeomorphic image registration. Front. Neuroinform. 2013, 7, 39. [Google Scholar] [CrossRef] [PubMed]
- Sharp, G.; Kandasamy, N.; Singh, H.; Folkert, M. GPU-based streaming architectures for fast cone-beam CT image reconstruction and demons deformable registration. Phys. Med. Biol. 2007, 52, 5771. [Google Scholar] [CrossRef] [PubMed]
- Shackleford, J.A.; Kandasamy, N.; Sharp, G. On developing B-spline registration algorithms for multi-core processors. Phys. Med. Biol. 2010, 55, 6329. [Google Scholar] [CrossRef] [PubMed]
- Ou, Y.; Sotiras, A.; Paragios, N.; Davatzikos, C. DRAMMS: Deformable registration via attribute matching and mutual-saliency weighting. Med. Image Anal. 2011, 15, 622–639. [Google Scholar] [CrossRef]
- Modat, M.; Ridgway, G.R.; Taylor, Z.A.; Lehmann, M.; Barnes, J.; Hawkes, D.J.; Fox, N.C.; Ourselin, S. Fast free-form deformation using graphics processing units. Comput. Methods Programs Biomed. 2010, 98, 278–284. [Google Scholar] [CrossRef]

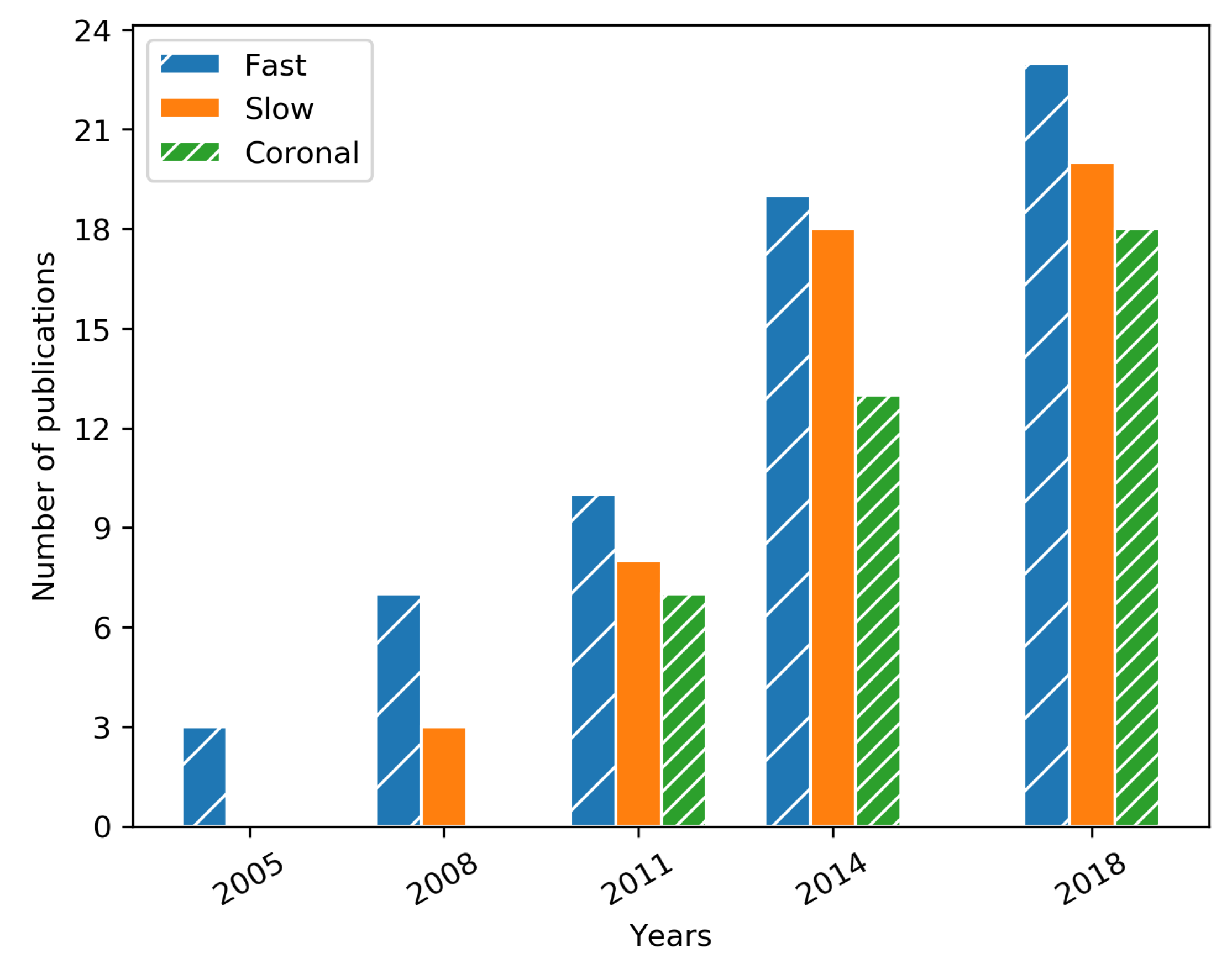
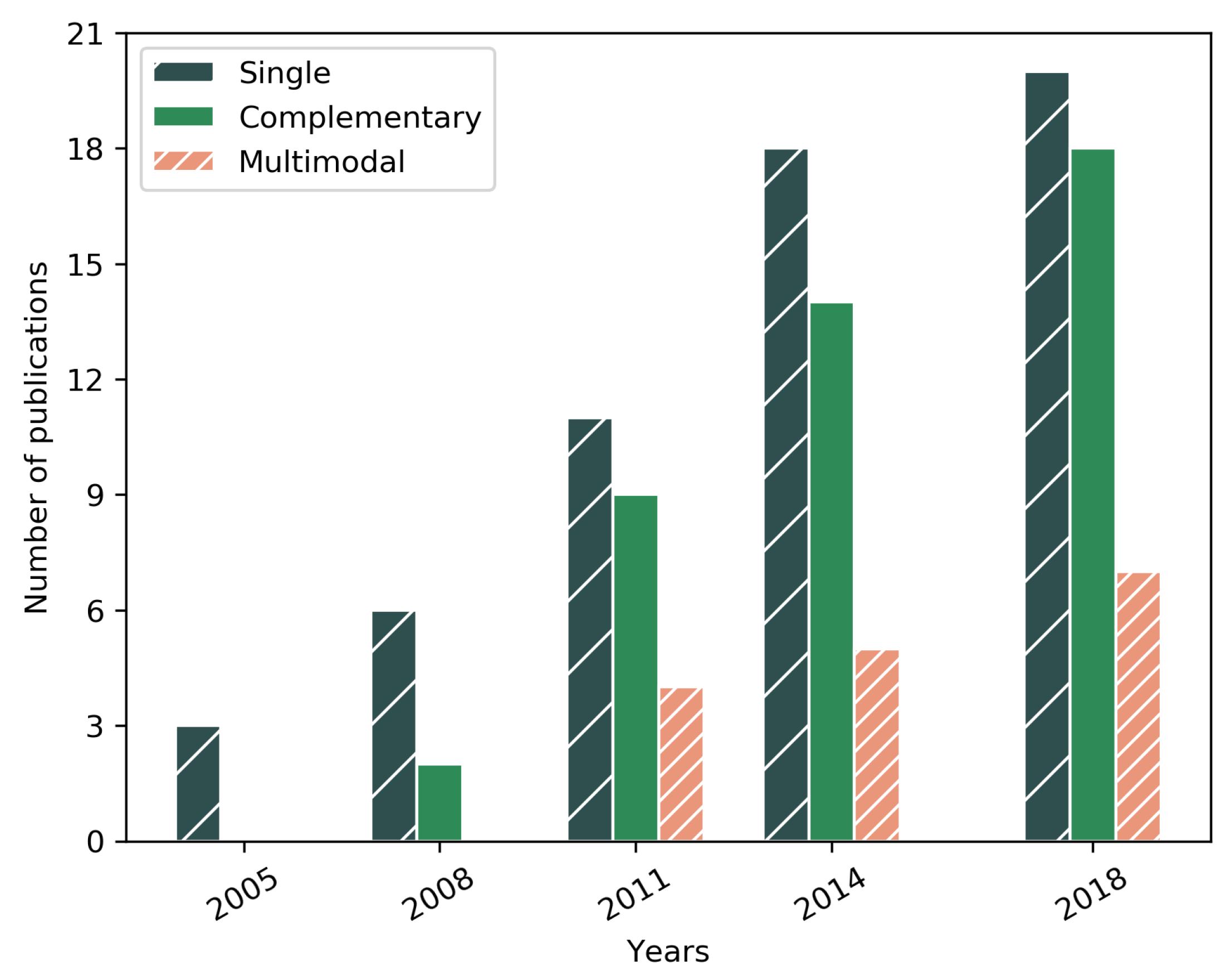
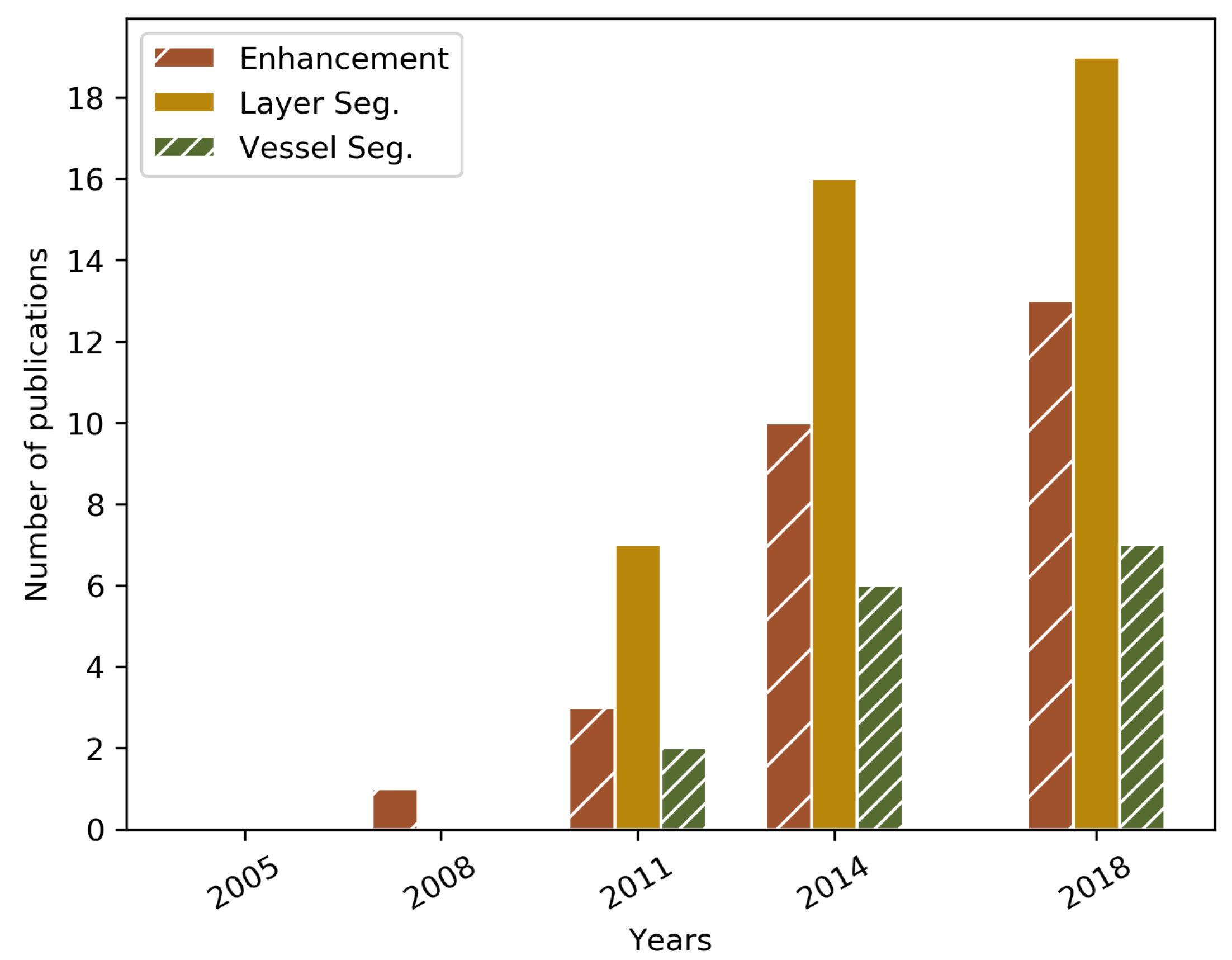
| Year | Reference | Research Topic | Handled Plane | Method | Preproc. | Validation | OCT Type | Dataset |
|---|---|---|---|---|---|---|---|---|
| 1993 | Swanson [53] | OCT retinal imaging: proof of concept | | | − | − | G | − | |
| 1996 | Schuman [54] | Layer thickness repeatability | | | − | − | G | 10P, 11H | |
| 2005 | Ishikawa [55] | Layer segmentation | | | − | − | A | 24P, 23H | |
| 2007 | Zawadzki [19] | Motion correction | , | ⊥ | − | Qualitative | H | 2− |
| Jorgensen [56] | Image enhanc. | , | | | − | Qualitative | A | − | |
| Fuller [57] | Layer segmentation | | | − | − | H | − | ||
| 2008 | Potsaid [9] | Hardware development | ⊥ | Enhanc. | Qualitative | I | − | |
| Khanifar [58] | Drusen imaging | | | − | Qualitative | J | 31P | ||
| 2009 | Ricco [59] | Motion correction | C | |; SLO | Vessel Seg. | Qualitative, Multimodal | SD-OCT | 116P, 48H scans |
| Niemeijer1 [60] | Motion correction | C | ‖ | Layer Seg. Enhanc. | Qualitative, Simulated | B | 6H | |
| Garvin [61] | Layer segmentation | | | Layer Seg. | Qualitative | B | 41H | ||
| Xu [62] | Motion correction | C | |; SLO | − | Qualitative | B | 24P, 19H | |
| Tolliver [63] | Motion correction | ⊥ | − | Quantitative | B | − | ||
| 2010 | Robinson [33] | Motion correction | C | ‖ | − | Qualitative | SD-OCT | − |
| Kolar [64] | Multimodal registration | C | |; FP, SLO | Enhanc. Layer Seg. | Qualitative | C | − | |
| Gibson [65] | 3D-OCT ONH registration | , C | ‖ | ONH Seg. | Qualitative, Quantitative | Custom SD-OCT | − | |
| Xu [66] | 3D-OCT registration | , | | | − | Qualitative | B | − | |
| 2011 | Antony [67] | Motion correction | , | ⊥; FP | Layer Seg. | Qualitative, Multimodal | E | − |
| Song [68] | Motion correction | ⊥ | Layer Seg. Vessel Seg. | Qualitative, Quantitative | SD-OCT | − | ||
| Li [69] | OCT montage | C | ‖ | RPE Seg. | Quantitative | B | 3P, 3H | |
| 2012 | Niemeijer [70] | 3D-OCT registration | C, , | ‖ | Layer Seg. Vessel Seg. | Qualitative, Simulated | F | 5P |
| Kraus [71] | Motion correction | C, , | ⊥ | Enhanc. | Qualitative, Quantitative | K, L | − | |
| Xu [21] | Registration | , | | | − | Qualitative, Simulated | B | 49P, 25H | |
| 2013 | He [72] | Motion correction | , | | | − | Qualitative | Custom SD-OCT | − |
| Chen [73] | Motion correction | , | | | Enhanc. Layer Seg. | Qualitative, Quantitative | C | 15− | |
| Zheng [74] | Layer segmentation | | | Layer Seg. | Quantitative | E | 5− | ||
| Hendargo [75] | Motion correction, OCT montage | C | ‖, ⊥ | Enhanc. Layer Seg. Vessel Seg. | Qualitative | M | 1H | |
| LaRocca [76] | Handheld SLO-OCT device | C, , | ⊥; SLO | Enhanc. Layer Seg. Vessel Seg. | Qualitative, Multimodal | N | − | |
| 2014 | Chen [77] | Motion correction | , | | | Enhanc. Layer Seg. | Qualitative, Quantitative | C | 26P, 19H |
| Kraus [78] | Motion correction | C, , | ⊥ | Enhanc. Layer Seg. | Qualitative, Quantitative | D | 73− | |
| Montuoro [79] | Motion correction | , , | | | Layer Seg. | Qualitative, Simulated | B, E | 100 scans | |
| Wu [49] | Motion correction | C | | | Enhanc. Vessel Seg. RPE Seg. | Qualitative, Quantitative | SD-OCT | 15P | |
| 2015 | Lee [80] | OCT surface registration | , C | ‖ | Layer Seg. | Qualitative, Simulated | O | 3P, 3H |
| 2016 | Lang [81] | Motion correction, OCT registration | C | ‖ | Enhanc. Vessel Seg. Layer Seg. | Qualitative, Quantitative | SD-OCT | 39H |
| Lezama [82] | Motion correction, OCT registration | C, , | ⊥ | Enhanc. Layer Seg. | Quantitative | P | 12− | |
| Cheng [50] | Motion correction, Multimodal registration | C | |; FP | − | Multimodal, Quantitative | F | 18− scans | |
| Fu [83] | Motion correction | | | − | Qualitative, Simulated | F | 7− | ||
| 2017 | Chen [84] | Motion correction, Scanning protocol | C, , | ‖; SLO | Enhanc. | Qualitative, Multimodal | Q | 1− |
| Code | Device | OCT Type | Acquisition Rate (A-Scan s) | Axial Resolution in Air (m) |
|---|---|---|---|---|
| A | Stratus OCT3 | TD | 400 | 10 |
| B | Cirrus HD-OCT | SD | 27k | 5 |
| C | Spectralis OCT | SD | 40k | 7 |
| D | RTVue | SD | 26k | 6 |
| E | Topcon SD-OCT1000 | SD | 18k | 5 |
| F | Topcon DRI OCT-1 | SS | 100k | 8 |
| G | Custom | TD | 40 | 14 |
| H | Custom | SD | 18k | 4.5 (retina) |
| I | Custom | SD | 70–312.5k | 3.6–11.6 |
| J | Custom | SD | 20k | × |
| K | Custom | SD | 70k | 3 |
| L | Custom | SS | 200k | 7 |
| M | Custom | SS | 100k | × |
| N | Custom | SD | 20k | 7 |
| O | Custom | SS | 100k | 2.7 |
| P | Custom | SS | 100k | 4.7 |
| Q | Custom | SS | 100k | 8.5 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sánchez Brea, L.; Andrade De Jesus, D.; Shirazi, M.F.; Pircher, M.; van Walsum, T.; Klein, S. Review on Retrospective Procedures to Correct Retinal Motion Artefacts in OCT Imaging. Appl. Sci. 2019, 9, 2700. https://doi.org/10.3390/app9132700
Sánchez Brea L, Andrade De Jesus D, Shirazi MF, Pircher M, van Walsum T, Klein S. Review on Retrospective Procedures to Correct Retinal Motion Artefacts in OCT Imaging. Applied Sciences. 2019; 9(13):2700. https://doi.org/10.3390/app9132700
Chicago/Turabian StyleSánchez Brea, Luisa, Danilo Andrade De Jesus, Muhammad Faizan Shirazi, Michael Pircher, Theo van Walsum, and Stefan Klein. 2019. "Review on Retrospective Procedures to Correct Retinal Motion Artefacts in OCT Imaging" Applied Sciences 9, no. 13: 2700. https://doi.org/10.3390/app9132700
APA StyleSánchez Brea, L., Andrade De Jesus, D., Shirazi, M. F., Pircher, M., van Walsum, T., & Klein, S. (2019). Review on Retrospective Procedures to Correct Retinal Motion Artefacts in OCT Imaging. Applied Sciences, 9(13), 2700. https://doi.org/10.3390/app9132700






