Robotics in Health Care: Perspectives of Robot-Aided Interventions in Clinical Practice for Rehabilitation of Upper Limbs
Abstract
1. Introduction
2. Neurological Rehabilitation
3. Robotics in Healthcare: Rehabilitation Domain
4. Robot-Aided Modalities for Upper Limb Training
5. Literature Review Summary
5.1. Materials and Methods
5.2. Robot-Aided Rehabilitation of Upper Limb Motor Function
5.2.1. Data Analysis Capability
5.2.2. Adaptability of Treatments
5.2.3. Intervention and Safety Strategies
5.2.4. Focus of Treatment
5.2.5. Interaction Channel and Feedback for the User
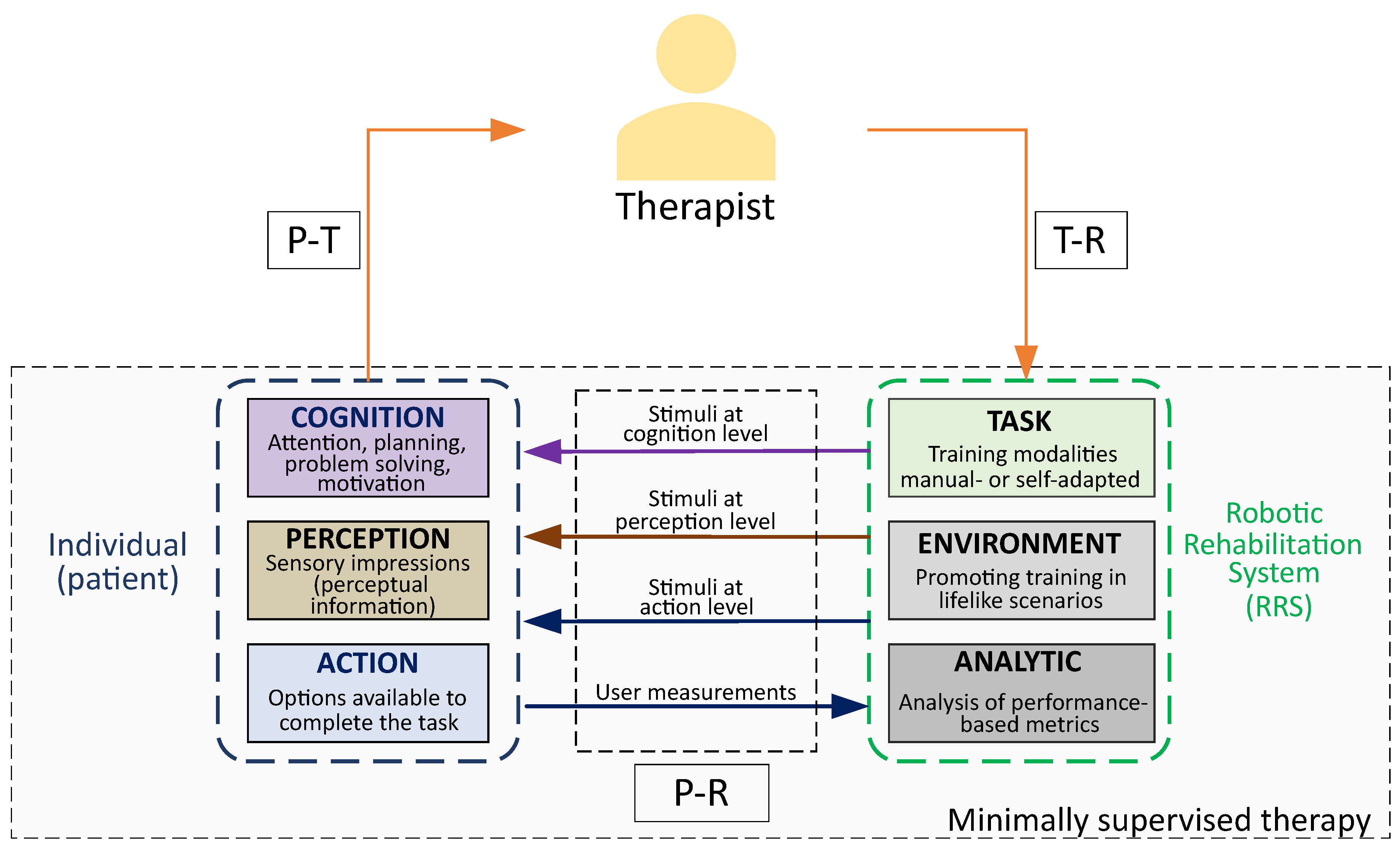
6. Framework for Robot-Aided Systems in Clinical Practice
6.1. Efficient Human–Robot Interactions
6.2. Safety in Physical Human–Robot Interaction
6.3. Scenarios for Boosting Motor Gain Assimilation
6.4. Towards More Autonomous Interventions: Self-Adaptive Versus Sizeable Systems
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- World Health Organization. Neurological Disorders: Public Health Challenges; WHO Press: Geneva, Switzerland, 2006; Available online: http://www.who.int/mental_health/neurology/neurodiso/en/ (accessed on 1 October 2018).
- Feigin, V.L.; Abajobir, A.A.; Abate, K.H.; Abd-Allah, F.; Abdulle, A.M.; Abera, S.F.; Abyu, G.Y.; Ahmed, M.B.; Aichour, A.N.; Aichour, I.; et al. Global, regional, and national burden of neurological disorders during 1990–2015: A systematic analysis for the Global Burden of Disease Study 2015. Lancet Neurol. 2017, 16, 877–897. [Google Scholar] [CrossRef]
- Dorsey, E.R.; Elbaz, A.; Nichols, E.; Abd-Allah, F.; Abdelalim, A.; Adsuar, J.C.; Ansha, M.G.; Brayne, C.; Choi, J.Y.J.; Collado-Mateo, D.; et al. Global, regional, and national burden of Parkinson’s disease, 1990–2016: A systematic analysis for the global burden of disease study 2016. Lancet Neurol. 2018, 17, 939–953. [Google Scholar] [CrossRef]
- Population Division, Department of Economic and Social Affairs, United Nations. World Population Ageing 2017; United Nations: Rome, Italy, 2017; Available online: https://www.un.org/en/development/desa/population/theme/aging/WPA2017.asp (accessed on 1 October 2018).
- Vos, T.; Abajobir, A.A.; Abate, K.H.; Abbafati, C.; Abbas, K.M.; Abd-Allah, F.; Abdulkader, R.S.; Abdulle, A.M.; Abebo, T.A.; Abera, S.F.; et al. Global, regional, and national incidence, prevalence, and years lived with disability for 328 diseases and injuries for 195 countries, 1990–2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet 2017, 390, 1211–1259. [Google Scholar] [CrossRef]
- Chua, K.S.G.; Kuah, C.W.K. Innovating With Rehabilitation Technology in the Real World: Promises, Potentials, and Perspectives. Am. J. Phys. Med. Rehabil. 2017, 96, S150–S156. [Google Scholar] [CrossRef] [PubMed]
- Van der Loos, H.M.; Reinkensmeyer, D.J.; Guglielmelli, E. Rehabilitation and Health Care Robotics; Springer Handbook of Robotics; Siciliano, B., Khatib, O., Eds.; Springer International Publishing: Cham, Switzerland, 2016; pp. 1685–1728. [Google Scholar]
- Lo, A.C.; Guarino, P.D.; Richards, L.G.; Haselkorn, J.K.; Wittenberg, G.F.; Federman, D.G.; Ringer, R.J.; Wagner, T.H.; Krebs, H.I.; Volpe, B.T.; et al. Robot-assisted therapy for long-term upper-limb impairment after stroke. N. Engl. J. Med. 2010, 362, 1772–1783. [Google Scholar] [CrossRef] [PubMed]
- Kwakkel, G.; Kollen, B.J.; Krebs, H.I. Effects of robot-assisted therapy on upper limb recovery after stroke: A systematic review. Neurorehabilit. Neural Repair 2008, 22, 111–121. [Google Scholar] [CrossRef] [PubMed]
- Norouzi-Gheidari, N.; Archambault, P.S.; Fung, J. Effects of robot-assisted therapy on stroke rehabilitation in upper limbs: Systematic review and meta-analysis of the literature. J. Rehabil. Res. Dev. 2012, 49, 479–496. [Google Scholar] [CrossRef] [PubMed]
- Veerbeek, J.M.; Langbroek-Amersfoort, A.C.; van Wegen, E.E.H.; Meskers, C.G.M.; Kwakkel, G. Effects of Robot-Assisted Therapy for the Upper Limb After Stroke: A Systematic Review and Meta-analysis. Neurorehabilit. Neural Repair 2017, 31, 107–121. [Google Scholar] [CrossRef] [PubMed]
- Oña, E.D.; Cano-de la Cuerda, R.; Sánchez-Herrera, P.; Balaguer, C.; Jardón, A. A review of robotics in neurorehabilitation: Towards an automated process for upper limb. J. Healthc. Eng. 2018, 2018, 9758939. [Google Scholar] [CrossRef]
- Huang, H.; Wolf, S.L.; He, J. Recent developments in biofeedback for neuromotor rehabilitation. J. NeuroEng. Rehabil. 2006, 3, 11. [Google Scholar] [CrossRef]
- Poli, P.; Morone, G.; Rosati, G.; Masiero, S. Robotic technologies and rehabilitation: New tools for stroke patients’ therapy. BioMed Res. Int. 2013, 2013, 153872. [Google Scholar] [CrossRef] [PubMed]
- Timmermans, A.A.; Seelen, H.A.; Willmann, R.D.; Kingma, H. Technology-assisted training of arm-hand skills in stroke: Concepts on reacquisition of motor control and therapist guidelines for rehabilitation technology design. J. NeuroEng. Rehabil. 2009, 6, 1. [Google Scholar] [CrossRef] [PubMed]
- Díaz, I.; Gil, J.J.; Sánchez, E. Lower-limb robotic rehabilitation: Literature review and challenges. J. Robot. 2011, 759764. [Google Scholar] [CrossRef]
- Lv, X.; Wu, Z. Review of robot-assisted gait rehabilitation after stroke. J. Rehabil. Robot. 2013, 2013, 3–8. [Google Scholar]
- Qian, Z.; Bi, Z. Recent development of rehabilitation robots. Adv. Mech. Eng. 2015, 7. [Google Scholar] [CrossRef]
- Marchal-Crespo, L.; Reinkensmeyer, D.J. Review of control strategies for robotic movement training after neurologic injury. J. NeuroEng. Rehabil. 2009, 6, 20. [Google Scholar] [CrossRef] [PubMed]
- Yan, T.; Cempini, M.; Oddo, C.M.; Vitiello, N. Review of assistive strategies in powered lower-limb orthoses and exoskeletons. Robot. Auton. Syst. 2015, 64, 120–136. [Google Scholar] [CrossRef]
- Basteris, A.; Nijenhuis, S.M.; Stienen, A.H.; Buurke, J.H.; Prange, G.B.; Amirabdollahian, F. Training modalities in robot-mediated upper limb rehabilitation in stroke: A framework for classification based on a systematic review. J. Neuroeng. Rehabil. 2014, 11, 111. [Google Scholar] [CrossRef]
- Bamdad, M.; Zarshenas, H.; Auais, M.A. Application of BCI systems in neurorehabilitation: A scoping review. Disabil. Rehabil. Assist. Technol. 2015, 10, 355–364. [Google Scholar] [CrossRef]
- Cervera, M.A.; Soekadar, S.R.; Ushiba, J.; Millán, J.d.R.; Liu, M.; Birbaumer, N.; Garipelli, G. Brain-computer interfaces for post-stroke motor rehabilitation: A meta-analysis. Ann. Clin. Transl. Neurol. 2018, 5, 651–663. [Google Scholar] [CrossRef]
- Li, M.; Xu, G.; Xie, J.; Chen, C. A review: Motor rehabilitation after stroke with control based on human intent. Proc. Inst. Mech. Eng. Part H J. Eng. Med. 2018, 232, 344–360. [Google Scholar] [CrossRef] [PubMed]
- Barnes, M. Principles of neurological rehabilitation. J. Neurol. Neurosurg. Psychiatry 2003, 74, iv3–iv7. [Google Scholar] [CrossRef] [PubMed]
- Shumway-Cook, A.; Woollacott, M.H. Motor Control: Translating Research into Clinical Practice, 4 ed.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2007. [Google Scholar]
- Wade, D. Principles of neurological rehabilitation. In Brain’s Diseases of the Nervous System, 12th ed.; Donaghy, M., Ed.; Oxford University Press: Oxford, UK, 2009; Chapter 6; pp. 165–179. [Google Scholar]
- Hubbard, I.J.; Parsons, M.W.; Neilson, C.; Carey, L.M. Task-specific training: Evidence for and translation to clinical practice. Occup. Ther. Int. 2009, 16, 175–189. [Google Scholar] [CrossRef] [PubMed]
- The Partnership for Robotics in Europe SPARC. Robotics 2020 Multi-Annual Roadmap; Call 2 ICT24 (2015)—Horizon 2020; SPARC: Brussels, Belgium, 2015. [Google Scholar]
- International Organization for Standardization. ISO 8549-1:1989. Prosthetics and orthotics. Vocabulary Part 1: General Terms for External Limb Prostheses and External Orthoses. 2011. Available online: http://www.iso.org/iso/home.htm (accessed on 1 February 2018).
- Ottobock. Ottobock North America Consumer Home | Ottobock US. 1919. Available online: http://www.ottobockus.com/ (accessed on 1 February 2019).
- Dario, P.; Guglielmelli, E.; Allotta, B.; Carrozza, M. Robotics for medical applications. Robot. Autom. Mag. IEEE 1996, 3, 44–56. [Google Scholar] [CrossRef]
- Brown University. Neuromotor Prosthetics. 2002. Available online: http://biomed.brown.edu/Courses/BI108/BI108_2002_Groups/neuromotor/webpage/Index.htm (accessed on 1 February 2019).
- Li, Y.; Alam, M.; Guo, S.; Ting, K.; He, J. Electronic bypass of spinal lesions: Activation of lower motor neurons directly driven by cortical neural signals. J. Neuroeng. Rehabil. 2014, 11, 107. [Google Scholar] [CrossRef] [PubMed]
- Park, S.; Borton, D.A.; Kang, M.; Nurmikko, A.V.; Song, Y.K. An implantable neural sensing microsystem with fiber-optic data transmission and power delivery. Sensors 2013, 13, 6014–6031. [Google Scholar] [CrossRef] [PubMed]
- Serruya, M.D.; Kahana, M.J. Techniques and devices to restore cognition. Behav. Brain Res. 2008, 192, 149–165. [Google Scholar] [CrossRef]
- Rehabilitation Engineering Laboratory. Tenoexo: Robotic Hand Orthosis for Therapy and Assistance in Activities of Daily Living. 2017. Available online: https://relab.ethz.ch/research/current-research-projects/robotic-hand-orthosis-for-therapy-and-assistance-in-activities-of-daily-living.html (accessed on 10 June 2019).
- Hocoma. Armeo Therapy Concept. 2011. Available online: https://www.hocoma.com/solutions/arm-hand/ (accessed on 10 Jun 2019).
- Huo, W.; Huang, J.; Wang, Y.; Wu, J.; Cheng, L. Control of upper-limb power-assist exoskeleton based on motion intention recognition. In Proceedings of the 2011 IEEE International Conference on Robotics and Automation (ICRA), Shanghai, China, 9–13 May 2011; pp. 2243–2248. [Google Scholar]
- Khan, A.M.; Yun, D.w.; Han, J.S.; Shin, K.; Han, C.S. Upper extremity assist exoskeleton robot. In Proceedings of the 2014 RO-MAN: The 23rd IEEE International Symposium on Robot and Human Interactive Communication, Edinburgh, UK, 25–29 August 2014; pp. 892–898. [Google Scholar]
- Wang, S.; van Dijk, W.; van der Kooij, H. Spring uses in exoskeleton actuation design. In Proceedings of the 2011 IEEE International Conference on Rehabilitation Robotics (ICORR), Zurich, Switzerland, 29 June–1 July 2011; pp. 1–6. [Google Scholar]
- Noda, T.; Teramae, T.; Ugurlu, B.; Morimoto, J. Development of an upper limb exoskeleton powered via pneumatic electric hybrid actuators with bowden cable. In Proceedings of the 2014 IEEE/RSJ International Conference on Intelligent Robots and Systems (IROS 2014), Chicago, IL, USA, 14–18 September 2014; pp. 3573–3578. [Google Scholar]
- Vitiello, N.; Lenzi, T.; Roccella, S.; De Rossi, S.M.; Cattin, E.; Giovacchini, F.; Vecchi, F.; Carrozza, M. NEUROExos: A powered elbow exoskeleton for physical rehabilitation. Robot. IEEE Trans. 2013, 29, 220–235. [Google Scholar] [CrossRef]
- Yang, J.; Shi, J.; Xie, H. Research on SMA actuated tendon driven hand exoskeleton with bidirectional finger joint motion coupling for rehabilitation usage. In Proceedings of the 2015 IEEE International Conference on Cyber Technology in Automation, Control, and Intelligent Systems (CYBER), Shenyang, China, 8–12 June 2015; pp. 336–340. [Google Scholar]
- Tang, T.; Zhang, D.; Xie, T.; Zhu, X. An exoskeleton system for hand rehabilitation driven by shape memory alloy. In Proceedings of the 2013 IEEE International Conference on Robotics and Biomimetics (ROBIO), Shenzhen, China, 12–14 December 2013; pp. 756–761. [Google Scholar]
- Bionik. InMotion Interactive Therapy. 2011. Available online: https://www.bioniklabs.com/products/inmotion-arm (accessed on 10 Jun 2019).
- Oña, E.D.; Barroso-de María, G.; Balaguer, C.; Jardón, A. Towards an Affordable Assistive Device for Personal Autonomy Recovery in Tasks Required of Manual Dexterity. IEEE Access 2018, 6, 26338–26349. [Google Scholar]
- Baker, M.; McDonough, M.; McMullin, E.; Swift, M.; BuSha, B. Orthotic hand-assistive exoskeleton. In Proceedings of the 2011 IEEE 37th Annual Northeast Bioengineering Conference (NEBEC), Troy, NY, USA, 1–3 April 2011; pp. 1–2. [Google Scholar]
- Nycz, C.J.; Bützer, T.; Lambercy, O.; Arata, J.; Fischer, G.S.; Gassert, R. Design and characterization of a lightweight and fully portable remote actuation system for use with a hand exoskeleton. IEEE Robot. Autom. Lett. 2016, 1, 976–983. [Google Scholar] [CrossRef]
- Ellis, M.D.; Sukal-Moulton, T.M.; Dewald, J.P.A. Impairment-Based 3-D Robotic Intervention Improves Upper Extremity Work Area in Chronic Stroke: Targeting Abnormal Joint Torque Coupling with Progressive Shoulder Abduction Loading. IEEE Trans. Robot. 2009, 25, 549–555. [Google Scholar] [CrossRef] [PubMed]
- Ellis, M.D.; Kottink, A.I.; Prange, G.B.; Rietman, J.S.; Buurke, J.H.; Dewald, J.P. Quantifying loss of independent joint control in acute stroke with a robotic evaluation of reaching workspace. In Proceedings of the 2011 Annual International Conference of the IEEE Engineering in Medicine and Biology Society, Boston, MA, USA, 30 August–3 September 2011; pp. 8231–8234. [Google Scholar]
- Ellis, M.D.; Sukal, T.; DeMott, T.; Dewald, J.P. ACT 3D exercise targets gravity-induced discoordination and improves reaching work area in individuals with stroke. In Proceedings of the 2007 IEEE 10th International Conference on Rehabilitation Robotics, Noordwijk, The Netherlands, 12–15 June 2007; pp. 890–895. [Google Scholar]
- Kahn, L.E.; Zygman, M.L.; Rymer, W.Z.; Reinkensmeyer, D.J. Robot-assisted reaching exercise promotes arm movement recovery in chronic hemiparetic stroke: A randomized controlled pilot study. J. Neuroeng. Rehabil. 2006, 3, 12. [Google Scholar] [CrossRef] [PubMed]
- Kahn, L.E.; Lum, P.S.; Rymer, W.Z.; Reinkensmeyer, D.J. Robot-assisted movement training for the stroke-impaired arm: Does it matter what the robot does? J. Rehabil. Res. Dev. 2014, 43, 619–630. [Google Scholar] [CrossRef]
- Reinkensmeyer, D.J.; Dewald, J.P.; Rymer, W.Z. Guidance-based quantification of arm impairment following brain injury: A pilot study. IEEE Trans. Rehabil. Eng. 1999, 7, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Vergaro, E.; Casadio, M.; Squeri, V.; Giannoni, P.; Morasso, P.; Sanguineti, V. Self-adaptive robot training of stroke survivors for continuous tracking movements. J. Neuroeng. Rehabil. 2010, 7, 13. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Casadio, M.; Sanguineti, V.; Morasso, P.G.; Arrichiello, V. Braccio di Ferro: A new haptic workstation for neuromotor rehabilitation. Technol. Health Care 2006, 14, 123–142. [Google Scholar] [PubMed]
- Chemuturi, R.; Amirabdollahian, F.; Dautenhahn, K. Adaptive training algorithm for robot-assisted upper-arm rehabilitation, applicable to individualised and therapeutic human-robot interaction. J. Neuroeng. Rehabil. 2013, 10, 102. [Google Scholar] [CrossRef]
- Chemuturi, R.; Amirabdollahian, F.; Dautenhahn, K. A study to understand lead-lag performance of subject vs rehabilitation system. In Proceedings of the 3rd Augmented Human International Conference, Megeve, France, 8–9 March 2012; p. 3. [Google Scholar]
- Bosecker, C.; Dipietro, L.; Volpe, B.; Igo Krebs, H. Kinematic robot-based evaluation scales and clinical counterparts to measure upper limb motor performance in patients with chronic stroke. Neurorehabilit. Neural Repair 2010, 24, 62–69. [Google Scholar] [CrossRef]
- Dipietro, L.; Krebs, H.; Volpe, B.; Stein, J.; Bever, C.; Mernoff, S.; Fasoli, S.; Hogan, N. Learning, not adaptation, characterizes stroke motor recovery: evidence from kinematic changes induced by robot-assisted therapy in trained and untrained task in the same workspace. IEEE Trans. Neural Syst. Rehabil. Eng. 2012, 20, 48–57. [Google Scholar] [CrossRef]
- Jackson, A.; Culmer, P.; Levesley, M.; Makower, S.; Cozens, J.; Bhakta, B. Effector force requirements to enable robotic systems to provide assisted exercise in people with upper limb impairment after stroke. In Proceedings of the 2011 IEEE International Conference on Rehabilitation Robotics, Zurich, Switzerland, 27 June–1 July 2011; pp. 1–6. [Google Scholar]
- O’Connor, R.J.; Jackson, A.; Makower, S.G.; Cozens, A.; Levesley, M. A proof of concept study investigating the feasibility of combining iPAM robot assisted rehabilitation with functional electrical stimulation to deliver whole arm exercise in stroke survivors. J. Med Eng. Technol. 2015, 39, 411–418. [Google Scholar] [CrossRef][Green Version]
- Colombo, R.; Pisano, F.; Micera, S.; Mazzone, A.; Delconte, C.; Carrozza, M.C.; Dario, P.; Minuco, G. Robotic techniques for upper limb evaluation and rehabilitation of stroke patients. IEEE Trans. Neural Syst. Rehabil. Eng. 2005, 13, 311–324. [Google Scholar] [CrossRef] [PubMed]
- Micera, S.; Sergi, P.N.; Zaccone, F.; Cappiello, G.; Carrozza, M.C.; Dario, P.; Guglielmelli, E.; Colombo, R.; Pisano, F.; Minuco, G. A low-cost biomechatronic system for the restoration and assessment of upper limb motor function in hemiparetic subjects. In Proceedings of the First IEEE/RAS-EMBS International Conference on Biomedical Robotics and Biomechatronics, BioRob 2006, Pisa, Italy, 20–22 February 2006; pp. 25–30. [Google Scholar]
- Colombo, R.; Pisano, F.; Delconte, C.; Mazzone, A.; Grioni, G.; Castagna, M.; Bazzini, G.; Imarisio, C.; Maggioni, G.; Pistarini, C. Comparison of exercise training effect with different robotic devices for upper limb rehabilitation: A retrospective study. Eur. J. Phys. Rehabil. Med. 2017, 53, 240–248. [Google Scholar] [PubMed]
- Lum, P.S.; Burgar, C.G.; Van der Loos, M.; Shor, P.C.; Majmundar, M.; Yap, R. MIME robotic device for upper-limb neurorehabilitation in subacute stroke subjects: A follow-up study. J. Rehabil. Res. Dev. 2006, 43, 631. [Google Scholar] [CrossRef] [PubMed]
- Burgar, C.G.; Lum, P.S.; Scremin, A.; Garber, S.L.; Van der Loos, H.; Kenney, D.; Shor, P. Robot-assisted upper-limb therapy in acute rehabilitation setting following stroke: Department of Veterans Affairs multisite clinical trial. J. Rehabil. Res. Dev. 2011, 48, 445–458. [Google Scholar] [CrossRef] [PubMed]
- Rosati, G.; Gallina, P.; Masiero, S. Design, implementation and clinical tests of a wire-based robot for neurorehabilitation. IEEE Trans. Neural Syst. Rehabil. Eng. 2007, 15, 560–569. [Google Scholar] [CrossRef] [PubMed]
- Masiero, S.; Armani, M.; Rosati, G. Upper-limb robot-assisted therapy in rehabilitation of acute stroke patients: Focused review and results of new randomized controlled trial. J. Rehabil. Res. Dev. 2011, 48, 355–366. [Google Scholar] [CrossRef] [PubMed]
- Masiero, S.; Armani, M.; Ferlini, G.; Rosati, G.; Rossi, A. Randomized Trial of a Robotic Assistive Device for the Upper Extremity During Early Inpatient Stroke Rehabilitation. Neurorehabilit. Neural Repair 2014, 28, 377–386. [Google Scholar] [CrossRef]
- Rosati, G.; Masiero, S.; Rossi, A. On the use of cable-driven robots in early inpatient stroke rehabilitation. In Advances in Italian Mechanism Science; Springer: Berlin/Heidelberg, Germany, 2017; pp. 551–558. [Google Scholar]
- Peter, O.; Tavaszi, I.; Toth, A.; Fazekas, G. Exercising daily living activities in robot-mediated therapy. J. Phys. Ther. Sci. 2017, 29, 854–858. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Sale, P.; Lombardi, V.; Franceschini, M. Hand robotics rehabilitation: Feasibility and preliminary results of a robotic treatment in patients with hemiparesis. Stroke Res. Treat. 2012, 2012, 820931. [Google Scholar] [CrossRef]
- Hwang, C.H.; Seong, J.W.; Son, D.S. Individual finger synchronized robot-assisted hand rehabilitation in subacute to chronic stroke: A prospective randomized clinical trial of efficacy. Clin. Rehabil. 2012, 26, 696–704. [Google Scholar] [CrossRef]
- Huang, X.; Naghdy, F.; Du, H.; Naghdy, G.; Todd, C. Reinforcement learning neural network (RLNN) based adaptive control of fine hand motion rehabilitation robot. In Proceedings of the 2015 IEEE Conference on Control Applications (CCA), Sydney, Australia, 21–23 September 2015; pp. 941–946. [Google Scholar]
- Hesse, S.; Schulte-Tigges, G.; Konrad, M.; Bardeleben, A.; Werner, C. Robot-assisted arm trainer for the passive and active practice of bilateral forearm and wrist movements in hemiparetic subjects. Arch. Phys. Med. Rehabil. 2003, 84, 915–920. [Google Scholar] [CrossRef]
- Schmidt, H.; Hesse, S.; Werner, C.; Bardeleben, A. Upper and lower extremity robotic devices to promote motor recovery after stroke -recent developments. In Proceedings of the 26th Annual International Conference of the IEEE Engineering in Medicine and Biology Society, San Francisco, CA, USA, 1–4 September 2004; Volume 2, pp. 4825–4828. [Google Scholar]
- Prange, G.B.; Jannink, M.J.; Groothuis-Oudshoorn, C.G.; Hermens, H.J.; IJzerman, M.J. Systematic review of the effect of robot-aided therapy on recovery of the hemiparetic arm after stroke. J. Rehabil. Res. Dev. 2006, 43, 171. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, C.D.; Der-Yeghiaian, L.; Le, V.H.; Cramer, S.C. A robotic device for hand motor therapy after stroke. In Proceedings of the 9th International Conference on Rehabilitation Robotics, ICORR 2005, Chicago, IL, USA, 28 June–1 July 2005; pp. 17–20. [Google Scholar]
- Takahashi, C.D.; Der-Yeghiaian, L.; Le, V.; Motiwala, R.R.; Cramer, S.C. Robot-based hand motor therapy after stroke. Brain 2008, 131, 425–437. [Google Scholar] [CrossRef] [PubMed]
- Takebayashi, T.; Takahashi, K.; Amano, S.; Uchiyama, Y.; Gosho, M.; Domen, K.; Hachisuka, K. Assessment of the Efficacy of ReoGo-J Robotic training against other rehabilitation therapies for Upper-Limb Hemiplegia after Stroke: Protocol for a Randomized Controlled Trial. Front. Neurol. 2018, 9, 730. [Google Scholar] [CrossRef] [PubMed]
- Meyer-Rachner, P.; Passon, A.; Klauer, C.; Schauer, T. Compensating the effects of FES-induced muscle fatigue by rehabilitation robotics during arm weight support. Curr. Dir. Biomed. Eng. 2017, 3, 31–34. [Google Scholar]
- Passon, A.; Klewe, T.; Seel, T.; Schauer, T. A new approach for a patient-cooperative upper limb fes support based on vector fields. IFAC-PapersOnLine 2017, 50, 9954–9960. [Google Scholar] [CrossRef]
- Passon, A.; Seel, T.; Massmann, J.; Schauer, T.; Freernan, C. Iterative learning vector field for FES-supported cyclic upper limb movements in combination with robotic weight compensation. In Proceedings of the 2018 IEEE/RSJ International Conference on Intelligent Robots and Systems (IROS), Madrid, Spain, 1–5 October 2018; pp. 5169–5174. [Google Scholar]
- Johnson, M.; Wisneski, K.; Anderson, J.; Nathan, D.; Smith, R. Development of ADLER: The activities of daily living exercise robot. In Proceedings of the First IEEE/RAS-EMBS International Conference on Biomedical Robotics and Biomechatronics, BioRob 2006, Pisa, Italy, 20–22 February 2006; pp. 881–886. [Google Scholar]
- Johnson, M.J.; Wisneski, K.J.; Anderson, J.; Nathan, D.; Strachota, E.; Kosasih, J.; Johnston, J.; Smith, R.O. Task-oriented and purposeful robot-assisted therapy. In Rehabilitation Robotics; IntechOpen: Rijeka, Croatia, 2007. [Google Scholar]
- Nathan, D.E.; Johnson, M.J.; McGuire, J. Feasibility of integrating FES grasp assistance with a task-oriented robot-assisted therapy environment: A case study. In Proceedings of the 2008 2nd IEEE RAS & EMBS International Conference on Biomedical Robotics and Biomechatronics, Scottsdale, AZ, USA, 19–22 October 2008; pp. 807–812. [Google Scholar]
- Montagner, A.; Frisoli, A.; Borelli, L.; Procopio, C.; Bergamasco, M.; Carboncini, M.C.; Rossi, B. A pilot clinical study on robotic assisted rehabilitation in VR with an arm exoskeleton device. In Proceedings of the 2007 Virtual Rehabilitation, Venice, Italy, 27–29 September 2007; pp. 57–64. [Google Scholar]
- Frisoli, A.; Bergamasco, M.; Carboncini, M.C.; Rossi, B. Robotic assisted rehabilitation in virtual reality with the L-EXOS. Stud. Health Technol. Inform. 2009, 145, 40–54. [Google Scholar]
- Frisoli, A.; Chisari, C.; Sotgiu, E.; Procopio, C.; Fontana, M.; Rossi, B.; Bergamasco, M. Rehabilitation training and evaluation with the L-EXOS in chronic stroke. In Proceedings of the International Conference on Smart Homes and Health Telematics, Artiminio, Italy, 12–15 June 2012; Springer: Berlin/Heidelberg, Germany, 2012; pp. 242–245. [Google Scholar]
- Page, S.J.; Hill, V.; White, S. Portable upper extremity robotics is as efficacious as upper extremity rehabilitative therapy: A randomized controlled pilot trial. Clin. Rehabil. 2013, 27, 494–503. [Google Scholar] [CrossRef]
- Kim, G.J.; Rivera, L.; Stein, J. Combined Clinic-Home Approach for Upper Limb Robotic Therapy after Stroke: A Pilot Study. Arch. Phys. Med. Rehabil. 2015, 96, 2243–2248. [Google Scholar] [CrossRef]
- Dunaway, S.; Dezsi, D.B.; Perkins, J.; Tran, D.; Naft, J. Case report on the use of a custom myoelectric elbow–wrist–hand orthosis for the remediation of upper extremity paresis and loss of function in chronic stroke. Mil. Med. 2017, 182, e1963–e1968. [Google Scholar] [CrossRef]
- Rahman, T.; Sample, W.; Jayakumar, S.; King, M.M.; Wee, J.Y.; Seliktar, R.; Alexander, M.; Scavina, M.; Clark, A. Passive exoskeletons for assisting limb movement. J. Rehabil. Res. Dev. 2006, 43, 583. [Google Scholar] [CrossRef] [PubMed]
- Ragonesi, D.; Agrawal, S.; Sample, W.; Rahman, T. Series elastic actuator control of a powered exoskeleton. In Proceedings of the 2011 Annual International Conference of the IEEE Engineering in Medicine and Biology Society, Boston, MA, USA, 30 August–3 September 2011; pp. 3515–3518. [Google Scholar]
- Gijbels, D.; Lamers, I.; Kerkhofs, L.; Alders, G.; Knippenberg, E.; Feys, P. The Armeo Spring as training tool to improve upper limb functionality in multiple sclerosis: A pilot study. J. NeuroEng. Rehabil. 2011, 8, 5. [Google Scholar] [CrossRef]
- Zariffa, J.; Kapadia, N.; Kramer, J.; Taylor, P.; Alizadeh-Meghrazi, M.; Zivanovic, V.; Willms, R.; Townson, A.; Curt, A.; Popovic, M.; et al. Feasibility and efficacy of upper limb robotic rehabilitation in a subacute cervical spinal cord injury population. Spinal Cord 2012, 50, 220. [Google Scholar] [CrossRef] [PubMed]
- El-Shamy, S.M. Efficacy of Armeo® robotic therapy versus conventional therapy on upper limb function in children with hemiplegic cerebral palsy. Am. J. Phys. Med. Rehabil. 2018, 97, 164–169. [Google Scholar] [CrossRef] [PubMed]
- Kutner, N.G.; Zhang, R.; Butler, A.J.; Wolf, S.L.; Alberts, J.L. Quality-of-life change associated with robotic-assisted therapy to improve hand motor function in patients with subacute stroke: A randomized clinical trial. Phys. Ther. 2010, 90, 493–504. [Google Scholar] [CrossRef] [PubMed]
- Linder, S.M.; Rosenfeldt, A.B.; Bay, R.C.; Sahu, K.; Wolf, S.L.; Alberts, J.L. Improving quality of life and depression after stroke through telerehabilitation. Am. J. Occup. Ther. 2015, 69, 6902290020. [Google Scholar] [CrossRef] [PubMed]
- Cherry, C.O.; Chumbler, N.R.; Richards, K.; Huff, A.; Wu, D.; Tilghman, L.M.; Butler, A. Expanding stroke telerehabilitation services to rural veterans: A qualitative study on patient experiences using the robotic stroke therapy delivery and monitoring system program. Disabil. Rehabil. Assist. Technol. 2017, 12, 21–27. [Google Scholar] [CrossRef]
- Schabowsky, C.N.; Godfrey, S.B.; Holley, R.J.; Lum, P.S. Development and pilot testing of HEXORR: Hand EXOskeleton Rehabilitation Robot. J. NeuroEng. Rehabil. 2010, 7, 36. [Google Scholar] [CrossRef]
- Godfrey, S.B.; Holley, R.J.; Lum, P.S. Clinical effects of using HEXORR (Hand Exoskeleton Rehabilitation Robot) for movement therapy in stroke rehabilitation. Am. J. Phys. Med. Rehabil. 2013, 92, 947–958. [Google Scholar] [CrossRef]
- Godfrey, S.B.; Holley, R.J.; Lum, P.S. Evaluation of HEXORR Tone Assistance Mode Against Spring Assistance. IEEE Trans. Neural Syst. Rehabil. Eng. 2015, 23, 610–617. [Google Scholar] [CrossRef]
- Bouzit, M.; Burdea, G.; Popescu, G.; Boian, R. The Rutgers Master II-new design force-feedback glove. IEEE/ASME Trans. Mech. 2002, 7, 256–263. [Google Scholar] [CrossRef]
- Merians, A.; Jack, D.; Boian, R.; Tremaine, M.; Burdea, G.; Adamovich, S.; Recce, M.; Poizner, H. Virtual reality-augmented rehabilitation for patients following stroke. Phys. Ther. 2002, 82, 898–915. [Google Scholar]
- Heuser, A.; Kourtev, H.; Winter, S.; Fensterheim, D.; Burdea, G.; Hentz, V.; Forducey, P. Telerehabilitation using the Rutgers Master II glove following carpal tunnel release surgery: Proof-of-concept. IEEE Trans. Neural Syst. Rehabil. Eng. 2007, 15, 43–49. [Google Scholar] [CrossRef]
- Allington, J.; Spencer, S.J.; Klein, J.; Buell, M.; Reinkensmeyer, D.J.; Bobrow, J. Supinator extender (SUE): A pneumatically actuated robot for forearm/wrist rehabilitation after stroke. In Proceedings of the 2011 Annual International Conference of the IEEE Engineering in Medicine and Biology Society, Boston, MA, USA, 30 August–3 September 2011; pp. 1579–1582. [Google Scholar]
- Rocon, E.; Belda-Lois, J.M.; Ruiz, A.F.; Manto, M.; Moreno, J.C.; Pons, J.L. Design and Validation of a Rehabilitation Robotic Exoskeleton for Tremor Assessment and Suppression. IEEE Trans. Neural Syst. Rehabil. Eng. 2007, 15, 367–378. [Google Scholar] [CrossRef] [PubMed]
- Nef, T.; Riener, R. Shoulder actuation mechanisms for arm rehabilitation exoskeletons. In Proceedings of the 2008 2nd IEEE RAS EMBS International Conference on Biomedical Robotics and Biomechatronics, Scottsdale, AZ, USA, 19–22 October 2008; pp. 862–868. [Google Scholar]
- Guidali, M.; Duschau-Wicke, A.; Broggi, S.; Klamroth-Marganska, V.; Nef, T.; Riener, R. A robotic system to train activities of daily living in a virtual environment. Med. Biol. Eng. Comput. 2011, 49, 1213. [Google Scholar] [CrossRef] [PubMed]
- Nef, T.; Klamroth-Marganska, V.; Keller, U.; Riener, R. Three-dimensional multi-degree-of-freedom arm therapy robot (ARMin). In Neurorehabilitation Technology; Springer: London, UK, 2016; pp. 351–374. [Google Scholar]
- Loureiro, R.C.V.; Harwin, W.S. Reach & Grasp Therapy: Design and Control of a 9-DOF Robotic Neuro-rehabilitation System. In Proceedings of the 2007 IEEE 10th International Conference on Rehabilitation Robotics, Noordwijk, The Netherlands, 12–15 June 2007; pp. 757–763. [Google Scholar]
- Loureiro, R.C.; Lamperd, B.; Collin, C.; Harwin, W.S. Reach & grasp therapy: Effects of the Gentle/G System assessing sub-acute stroke whole-arm rehabilitation. In Proceedings of the 2009 IEEE International Conference on Rehabilitation Robotics, Kyoto, Japan, 23–26 June2009; pp. 755–760. [Google Scholar]
- Huang, J.; Tu, X.; He, J. Design and Evaluation of the RUPERT Wearable Upper Extremity Exoskeleton Robot for Clinical and In-Home Therapies. IEEE Trans. Syst. Man, Cybern. Syst. 2016, 46, 926–935. [Google Scholar] [CrossRef]
- Balasubramanian, S.; Wei, R.; Perez, M.; Shepard, B.; Koeneman, E.; Koeneman, J.; He, J. RUPERT: An exoskeleton robot for assisting rehabilitation of arm functions. In Proceedings of the 2008 Virtual Rehabilitation, Vancouver, BC, Canada, 25–27 August 2008; pp. 163–167. [Google Scholar]
- Loureiro, R.; Amirabdollahian, F.; Topping, M.; Driessen, B.; Harwin, W. Upper limb robot mediated stroke therapy—GENTLE/s approach. Auton. Robot. 2003, 15, 35–51. [Google Scholar] [CrossRef]
- Coote, S.; Murphy, B.; Harwin, W.; Stokes, E. The effect of the GENTLE/s robot-mediated therapy system on arm function after stroke. Clin. Rehabil. 2008, 22, 395–405. [Google Scholar] [CrossRef]
- Krebs, H.I.; Hogan, N.; Aisen, M.L.; Volpe, B.T. Robot-aided neurorehabilitation. IEEE Trans. Rehabil. Eng. 1998, 6, 75–87. [Google Scholar] [CrossRef]
- Krebs, H.I.; Palazzolo, J.J.; Dipietro, L.; Ferraro, M.; Krol, J.; Rannekleiv, K.; Volpe, B.T.; Hogan, N. Rehabilitation robotics: Performance-based progressive robot-assisted therapy. Auton. Robot. 2003, 15, 7–20. [Google Scholar] [CrossRef]
- Jackson, A.; Culmer, P.; Makower, S.; Levesley, M.; Richardson, R.; Cozens, A.; Williams, M.M.; Bhakta, B. Initial patient testing of iPAM-a robotic system for stroke rehabilitation. In Proceedings of the 2007 IEEE 10th International Conference on Rehabilitation Robotics, Noordwijk, The Netherlands, 12–15 June 2007; pp. 250–256. [Google Scholar]
- Toth, A.; Fazekas, G.; Arz, G.; Jurak, M.; Horvath, M. Passive robotic movement therapy of the spastic hemiparetic arm with REHAROB: Report of the first clinical test and the follow-up system improvement. In Proceedings of the 9th International Conference on Rehabilitation Robotics, ICORR 2005, Chicago, IL, USA, 28 June–1 July 2005; pp. 127–130. [Google Scholar]
- Fazekas, G.; Horvath, M.; Troznai, T.; Toth, A. Robot-mediated upper limb physiotherapy for patients with spastic hemiparesis: A preliminary study. J. Rehabil. Med. 2007, 39, 580–582. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, R.; Reinkensmeyer, D.; Shah, P.; Liu, J.; Rao, S.; Smith, R.; Cramer, S.; Rahman, T.; Bobrow, J. Monitoring functional arm movement for home-based therapy after stroke. In Proceedings of the 26th Annual International Conference of the IEEE Engineering in Medicine and Biology Society, San Francisco, CA, USA, 1–4 September 2004; Volume 2, pp. 4787–4790. [Google Scholar]
- Sanchez, R.J.; Liu, J.; Rao, S.; Shah, P.; Smith, R.; Rahman, T.; Cramer, S.C.; Bobrow, J.E.; Reinkensmeyer, D.J. Automating Arm Movement Training Following Severe Stroke: Functional Exercises With Quantitative Feedback in a Gravity-Reduced Environment. IEEE Trans. Neural Syst. Rehabil. Eng. 2006, 14, 378–389. [Google Scholar] [CrossRef] [PubMed]
- Nef, T.; Mihelj, M.; Colombo, G.; Riener, R. ARMin—Robot for rehabilitation of the upper extremities. In Proceedings of the 2006 IEEE International Conference on Robotics and Automation, Orlando, FL, USA, 15–19 May 2006; pp. 3152–3157. [Google Scholar]
- Staubli, P.; Nef, T.; Klamroth-Marganska, V.; Riener, R. Effects of intensive arm training with the rehabilitation robot ARMin II in chronic stroke patients: Four single-cases. J. Neuroeng. Rehabil. 2009, 6, 46. [Google Scholar] [CrossRef] [PubMed]
- Nef, T.; Guidali, M.; Riener, R. ARMin III—Arm therapy exoskeleton with an ergonomic shoulder actuation. Appl. Bionics Biomech. 2009, 6, 127–142. [Google Scholar] [CrossRef]
- Treger, I.; Faran, S.; Ring, H. Robot-assisted therapy for neuromuscular training of sub-acute stroke patients. A feasibility study. Eur. J. Phys. Rehabil. Med. 2008, 44, 431–435. [Google Scholar] [PubMed]
- Kumar, S.; Wöhrle, H.; Trampler, M.; Simnofske, M.; Peters, H.; Mallwitz, M.; Kirchner, E.; Kirchner, F. Modular Design and Decentralized Control of the Recupera Exoskeleton for Stroke Rehabilitation. Appl. Sci. 2019, 9, 626. [Google Scholar] [CrossRef]
- Winter, J.; Hunter, S.; Sim, J.; Crome, P. Hands-on therapy interventions for upper limb motor dysfunction following stroke. Cochrane Database Syst. Rev. 2011, 6, CD006609. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.; Deshpande, A.D. An upper-body rehabilitation exoskeleton Harmony with an anatomical shoulder mechanism: Design, modeling, control, and performance evaluation. Int. J. Robot. Res. 2017, 36, 414–435. [Google Scholar] [CrossRef]
- Bernstein, N. The Co-Ordination and Regulation of Movements, 1st ed.; Pergamon Press: Oxford, NY, USA, 1967. [Google Scholar]
- Hidler, J.; Lum, P.S. The road ahead for rehabilitation robotics. J. Rehabil. Res. Dev. 2011, 48, 7–10. [Google Scholar] [CrossRef]
- Hernandez, M.; Oña, E.D.; Garcia-Haro, J.M.; Jardon, A.; Balaguer, C. Towards an Automatic Spasticity Assessment by Means of Collaborative Robots. In Proceedings of the 2018 IEEE/RSJ International Conference on Intelligent Robots and Systems (IROS), Madrid, Spain, 1–5 October 2018; pp. 1–9. [Google Scholar]
- Roderick, S.; Carignan, C. Designing safety-critical rehabilitation robots. In Rehabilitation Robotics; Kommu, S.S., Ed.; Itech Education and Publishing: Vienna, Austria, 2007; Volume 1, Chapter 4; pp. 43–64. [Google Scholar]
- Barattini, P.; Vicentini, F.; Virk, G.S.; Haidegger, T. Human-Robot Interaction: Safety, Standardization, and Benchmarking; CRC Press: Boca Raton, FL, USA, 2019. [Google Scholar]
- Fasoli, S.E.; Krebs, H.I.; Stein, J.; Frontera, W.R.; Hughes, R.; Hogan, N. Robotic therapy for chronic motor impairments after stroke: Follow-up results. Arch. Phys. Med. Rehabil. 2004, 85, 1106–1111. [Google Scholar] [CrossRef]
- Bozzacchi, C.; Giusti, M.A.; Pitzalis, S.; Spinelli, D.; Di Russo, F. Similar cerebral motor plans for real and virtual actions. PLoS ONE 2012, 7, e47783. [Google Scholar] [CrossRef] [PubMed]
- Oña, E.D.; Balaguer, C.; Jardón, A. Towards a framework for rehabilitation and assessment of upper limb motor function based on serious games. In Proceedings of the 2018 IEEE 6th International Conference on Serious Games and Applications for Health (SeGAH), Vienna, Austria, 16–18 May 2018; pp. 1–7. [Google Scholar]
- Oña, E.D.; Balaguer, C.; Cano-de la Cuerda, R.; Collado-Vázquez, S.; Jardón, A. Effectiveness of serious games for leap motion on the functionality of the upper limb in Parkinson’s disease: A feasibility study. Comput. Intell. Neurosci. 2018, 2018, 7148427. [Google Scholar] [CrossRef] [PubMed]
- Pollock, A.; Baer, G.; Campbell, P.; Choo, P.L.; Forster, A.; Morris, J.; Pomeroy, V.M.; Langhorne, P. Physical rehabilitation approaches for the recovery of function and mobility following stroke. Cochrane Database Syst. Rev. 2014, 4, CD001920. [Google Scholar] [CrossRef] [PubMed]
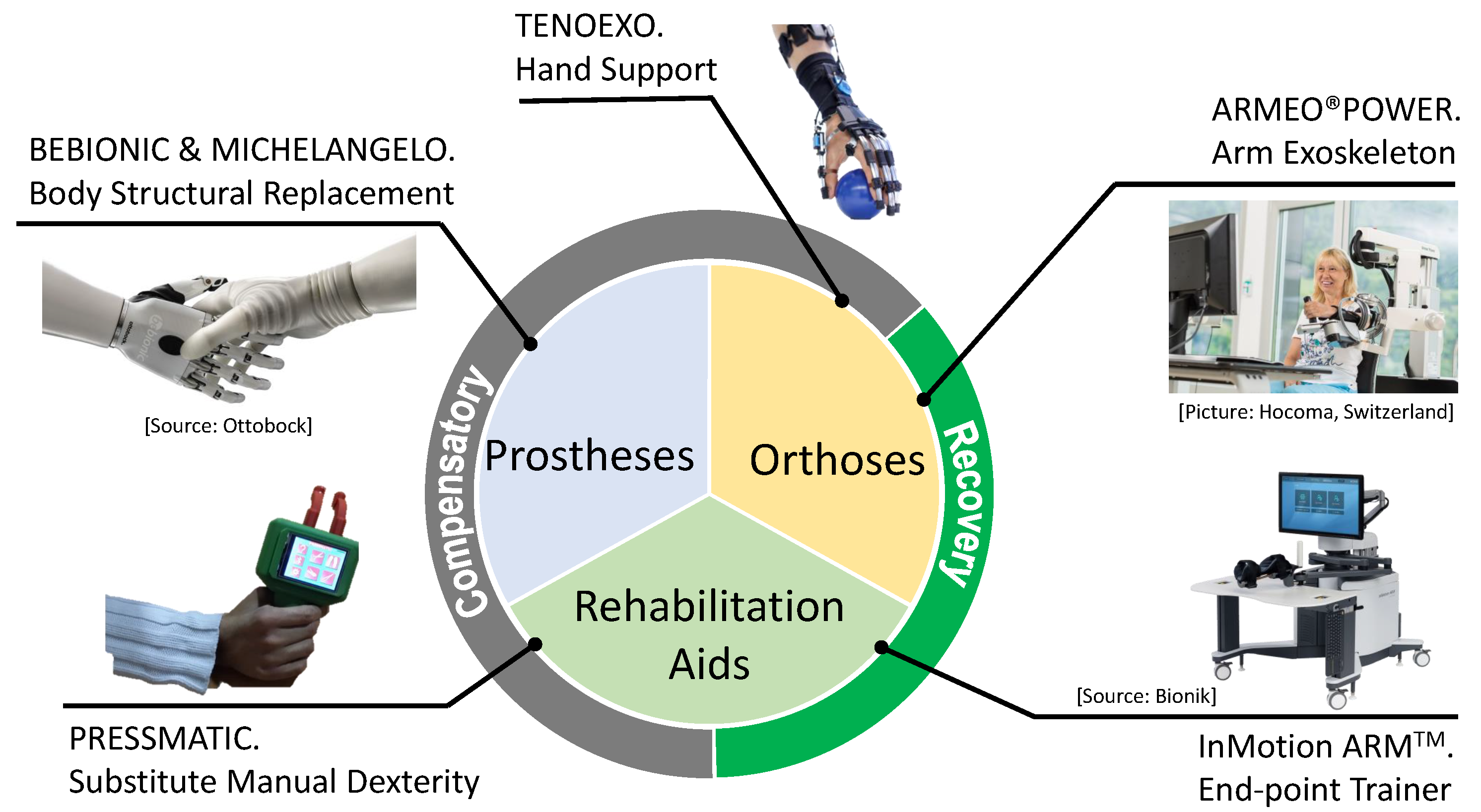
| System | Market Available† | Type of Device | Data Analysis Capability | Methods for Therapy Adaptability | Focus of Rehabilitation | Type of pHRI† | Safety Strategy | Channel For Presenting Tasks (Environment) | User Feedback |
|---|---|---|---|---|---|---|---|---|---|
| ACT-3D (2007) [50,51,52] | ✗ | End-point;2 DoF; electrical engines | Low | Variation (progressive) of abduction loading therapy | Shoulder; elbow | A | Software limits in force | 2D-VR (flat screen) showing an arm avatar | Haptic; audio |
| ARM-GUIDE (1999) [53,54,55] | ✗ | End-point;3 DoF; electrical engines | High (tone, spasticity, incoordination) | Modification of targets for reaching task | Shoulder; elbow | P; AA; R | Back stops; software limits in force | 2D-VR (flat screen) showing the target point | Haptic (off-axis force generation) |
| BRACCIO DI FERRO (2006) [56,57] | ✗ | End-point;2 DoF; electrical engines | High (performance evaluator | Adaptive controller to set the force loading automatically based on user’s performance | Shoulder; elbow | AA; R; GC | Back stops; emergency push button | 2D-VR (flat screen) displaying a path to follow | Visual; haptic (attractive force field) |
| GENTLE/A (2012) [58,59] | ✗ | End-point;3 DoF; magnetic mechanism | High (lead-lag performance) | Self-adaptation of duration to execute movements according to the user’s performance; definition of exercise path | Shoulder; elbow | P; AA;A; G | Software limits in force | 3D-VR (flat screen) showing several ping-pong balls in a 3D configuration | Visual; haptic; audio |
| INMOTION-ARM™ (2010) [60,61] | ✓ | End-point;6 DoF; electrical engines | High (software specific) | Adaptive therapy protocols; selection of exercise (games); progress measurement to determine medical necessity; | Shoulder; elbow | P; AA;R; GC | Backdrivable hardware; software limits in force | 2D-VR (flat screen) with a variety of games/task | Visual; haptic; audio |
| IPAM MkII (2011) [62,63] | ✗ | End-point;6 DoF; pneumatic engines | High (specific software) | Automatic generation of exercises (automated tasks) | Shoulder; elbow | P; R; A | Compliance control; emergency push button; software limits in force | 2D-VR (flat screen) 4 scenarios: beach, gym, city, or countryside | Visual; audio; haptic |
| MEMOS (2006) [64,65,66] | ✗ | End-point;2 DoF; electrical engines | Moderate | Variable gain of force loading | Shoulder; elbow | P; R; A | Emergency push button | 2D-VR (flat screen) displaying the target points | Visual; audio; haptic |
| MIME (2006) [67,68] | ✗ | End-point;6 DoF; electrical engines | Low | Variety of therapeutic modalities | Shoulder; elbow | P; AA;R; B | Back stops; emergency push button | Physical (real physical objects) promoting 3D reaching tasks | Visual (direct visualization of targets); haptic |
| NEREBOT (2007) [69,70,71,72] | ✗ | End-point;3 DoF; cable driven | Moderate | Exercise customization (via-points and setting of robot parameters) | Shoulder; elbow; forearm | P | Backdrivable hardware; back stops (magnetic attachment) | No user interface (hands-on movements) | Visual (direct visualization of movements) |
| REHAROB v2 (2017) [73] | ✗ | End-point;7 DoF; electrical engines | Moderate | Exercise programming available via a graphical user interface (includes a program simulation interface) | Shoulder; elbow; forearm | P | Emergency push button; software limits in ROM | No user interface (hands-on movements) | Visual; haptic |
| AMADEO® (2012) [74,75,76] | ✓ | End-point;5 DoF; electrical engines | High | Adjustable therapy and assessment modes | Hand (fingers) | P; AA; A | Software limits in force, speed and ROM; back stops (magnetic attachment) | 2D-VR (flat screen) with serious gaming in one- and two-dimensional movements | Visual (video games); haptic |
| BI-MANU-TRACK (2003) [77,78,79] | ✗ | End-point;2 DoF; electrical engines | Low | Selection of (two) operation modes | Forearm; wrist | A; B | Emergency push button; software limits in force (mechanical breaks) | Digital display showing the number of cycles | Visual (direct visualization of movements) |
| HWARD (2005) [80,81] | ✗ | End-point;3 DoF; pneumatic engines | Moderate | Selection of standardized training protocols | Wrist; hand | P; AA; R | Backdrivable hardware; emergency push button; software limits of force; software shutdown | 2D-VR (flat screen) | Visual; audio; haptic |
| ReoGo™-J (2008) [82] | ✓ | End-point;3 DoF; electrical engines | High | Library with several exercises and games | Shoulder; elbow; wrist; hand | P; A; G | N/A | 2D-VR (flat screen) presenting several real scenarios | Visual; audio; haptic |
| DIEGO® (2017) [83,84,85] | ✓ | End-point;4 DoF; cable driven | High | Selection of therapy games. Intelligent gravity compensation (IGC); cooperative sequences of movement | Shoulder; elbow | P; B; AS;A; GC | Backdrivable hardware | 3D-VR (fully immersive) with interactive games | Visual; audio; haptic (training with objects) |
| ADLER (2006) [86,87,88] | ✗ | End-point;6 DoF; electrical engines | High (specific software) | Selection of training modes.; movement programming available via pre-defined trajectories | Forearm; Wrist | A; AA; R | Backdrivable hardware; emergency push button; software limits in force | 2D-VR (Flat screen) displaying the target points | Visual; haptic |
| L-EXOS (2007) [89,90,91] | ✗ | Exoskeleton;5 DoF; cable driven | Low | Selection of different trajectories in the same virtual environment | Shoulder; elbow | A; AA; GC | Compliance control;back stops | 3D-VR (flat screen) | Visual (physical objects) |
| MYOPRO (2006) [92,93,94] | ✓ | Exoskeleton;2 DoF; electrical engines | Low | Distributed control mode for training different muscles | Elbow | AA | Software limits in forces | Physical (quotidian environments due to its portability) | Visual; haptic (physical objects); EMG |
| WREX (2004) [95,96] | ✓ | Exoskeleton;4 DoF; elastic bands | Low | Variation (manual) of force loadings | Shoulder; elbow | AA; GC | Compliance control | Physical (quotidian environments due to its portability) | Visual; haptic (physical objects); EMG |
| ARMEO®SPRING (2006) [97,98,99] | ✓ | Exoskeleton;5 DoF; electrical engines | High | Selection of therapy games; self-directed therapy option | Shoulder; elbow; forearm; wrist; hand | P; GC | Software limits in forces | 3D-VR (flat screen) | Visual; audio; haptic |
| Mentor Pro™ (2004) [100,101,102] | ✓ | Exoskeleton;1 DoF; pneumatic engines | Low | Selection of difficult/comfort levels; selection of (three) control modes | Wrist; hand; fingers | A | Compliance control; back stops | Physical (for increasing ROM in a real scenario) | Haptic, EMG |
| HEXORR (2010) [103,104,105] | ✗ | Exoskeleton;2 DoF; electrical engines | Low | Selection of multiple exercises | Hand | P; AA;A; GC | Software limits in velocities; back stops | 2D-VR (flat screen) with basic graphics | Haptic |
| RUTGERS-MASTER-II (2002) [106,107,108] | ✗ | Exoskeleton;20DoF; pneumatic engines | Moderate | Selection of exercises and setting of parameters | Hand (fingers) | P; AA; R | Compliance control; software limits in forces; back stops | 3D-VR (flat screen) training with games | Audio; visual; haptic |
| SUPINATOR- EXTENDER (2011) [109] | ✗ | Exoskeleton;2 DoF; pneumatic engines | Low | N/A | Forearm; wrist | AA | Compliance control; emergency push button | N/A | Haptic |
| WOTAS (2005) [110] | ✗ | Exoskeleton;3 DoF; Electrical engines | Low | Selection of assistance modes | Elbow; forearm; wrist | AS | Software limits in forces; back stops | Physical (keeping a target in a real scenario) | Haptic |
| ARMEO®POWER (2008) [111,112,113] | ✓ | Exoskeleton;6 DoF; electrical engines | High | Selection of several VR therapy tasks | Shoulder; elbow; forearm; wrist; hand | A; P; AA;R; GC | Backdrivable hardware; software limits in forces and loads | 3D-VR (flat screen) training with games | Audio; visual; haptic |
| GENTLE/G (2007) [114,115] | ✗ | Exoskeleton;9 DoF; cable driven | Moderate | Distributed control for training arm and/or hand; grasp therapy option | Shoulder; elbow; hand | P; AA;A; G | Compliance control; software limits in forces | 3D-VR (flat screen) showing tasks in real environments | Audio; visual; haptic; scoreboard; rewards |
| RUPERT (2008) [116,117] | ✗ | Exoskeleton;5 DoF; pneumatic engines | Low | Progressively challenging tasks | Shoulder; elbow, forearm; wrist | P; AA; A | Backdrivable hardware; compliance control | 3D-VR (flat screen) showing the target point | Visual |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Oña, E.D.; Garcia-Haro, J.M.; Jardón, A.; Balaguer, C. Robotics in Health Care: Perspectives of Robot-Aided Interventions in Clinical Practice for Rehabilitation of Upper Limbs. Appl. Sci. 2019, 9, 2586. https://doi.org/10.3390/app9132586
Oña ED, Garcia-Haro JM, Jardón A, Balaguer C. Robotics in Health Care: Perspectives of Robot-Aided Interventions in Clinical Practice for Rehabilitation of Upper Limbs. Applied Sciences. 2019; 9(13):2586. https://doi.org/10.3390/app9132586
Chicago/Turabian StyleOña, Edwin Daniel, Juan Miguel Garcia-Haro, Alberto Jardón, and Carlos Balaguer. 2019. "Robotics in Health Care: Perspectives of Robot-Aided Interventions in Clinical Practice for Rehabilitation of Upper Limbs" Applied Sciences 9, no. 13: 2586. https://doi.org/10.3390/app9132586
APA StyleOña, E. D., Garcia-Haro, J. M., Jardón, A., & Balaguer, C. (2019). Robotics in Health Care: Perspectives of Robot-Aided Interventions in Clinical Practice for Rehabilitation of Upper Limbs. Applied Sciences, 9(13), 2586. https://doi.org/10.3390/app9132586








