A Review on the Thermal Hazards of the Lithium-Ion Battery and the Corresponding Countermeasures
Abstract
1. Introduction
2. Safety-Related Incidents Involving Lithium-Ion Batteries
3. The Causes of Thermal Hazards Associated with Lithium-Ion Batteries
3.1. Physical Factor
3.2. Electrical Factor
3.3. Thermal Factor
3.4. Manufacturing Defect and Aging
4. Thermal Hazards of Lithium-Ion Battery
4.1. Thermal Hazards of Typical Battery Components
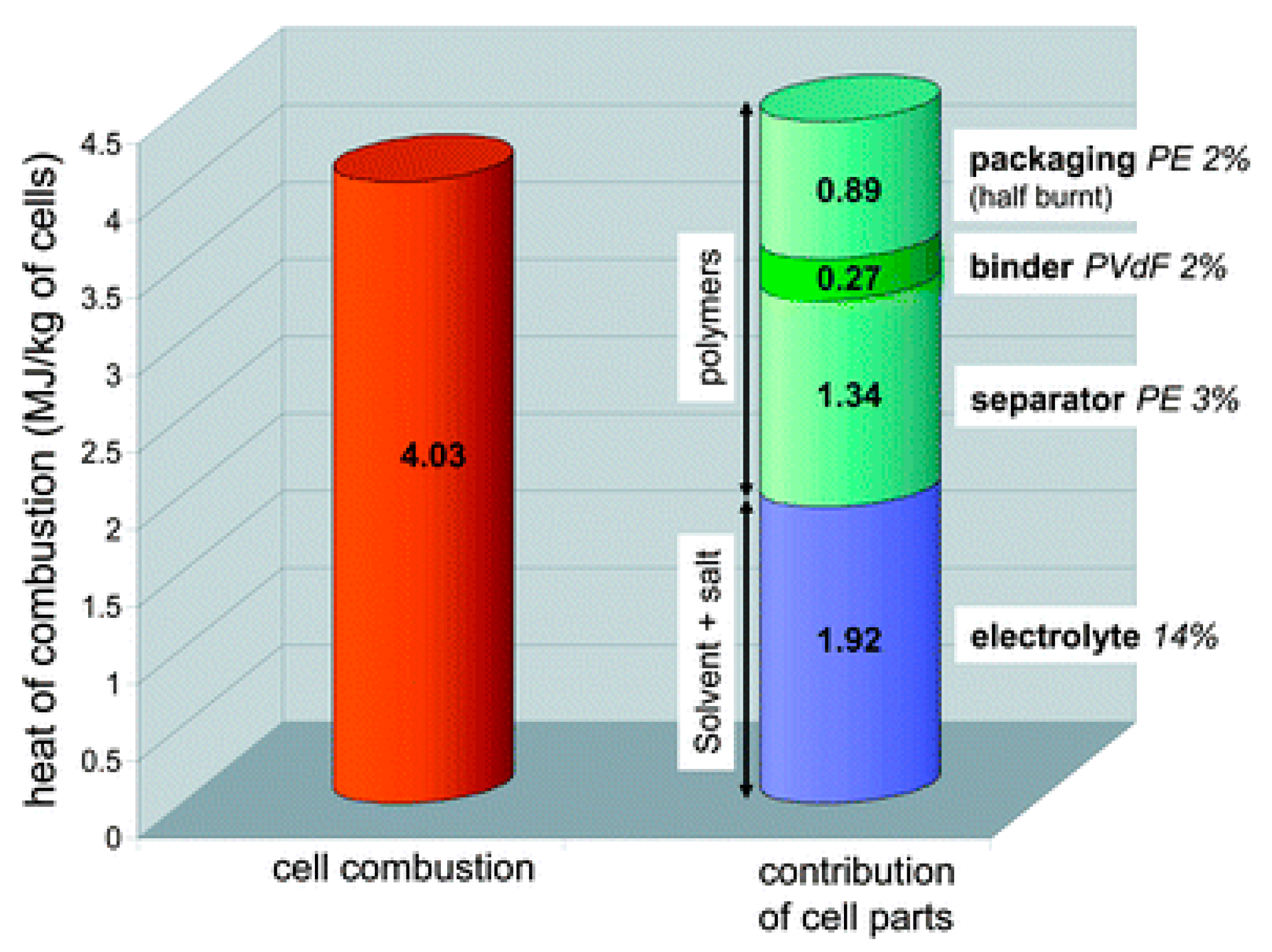
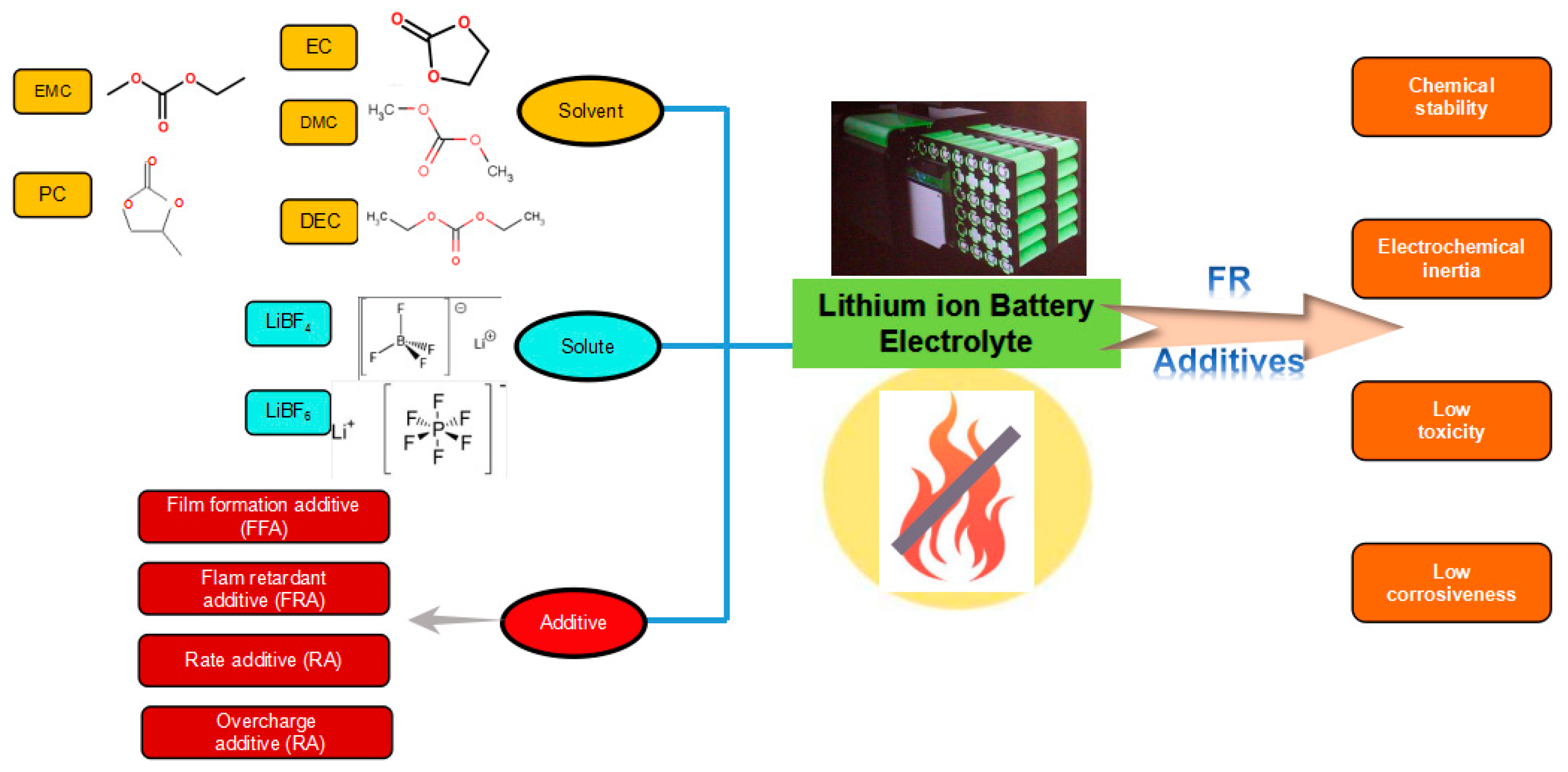
4.1.1. Thermal Hazards of the Electrolytes
- Lithium salt undergoes thermal decomposition to form PF5;
- PF5 reacts with water to generate hydrogen fluoride (HF);
- PF5 can also react with carbonate solution to form methyl fluoride (CH3F) and other substances.
4.1.2. Thermal Hazards of the Electrodes
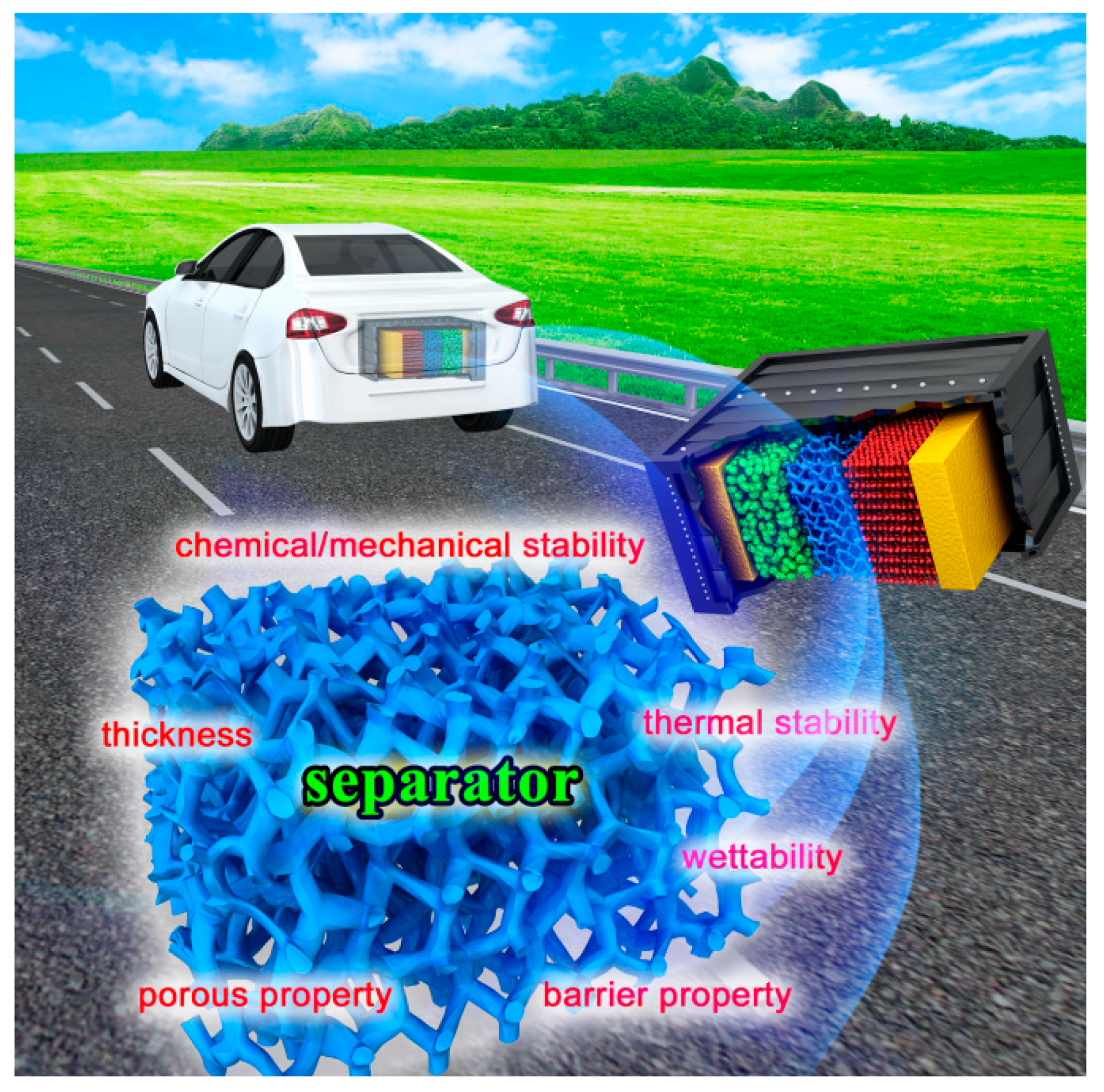
4.1.3. Thermal Hazards of the Separator
4.2. Thermal Hazards of a Single Battery and Battery Pack
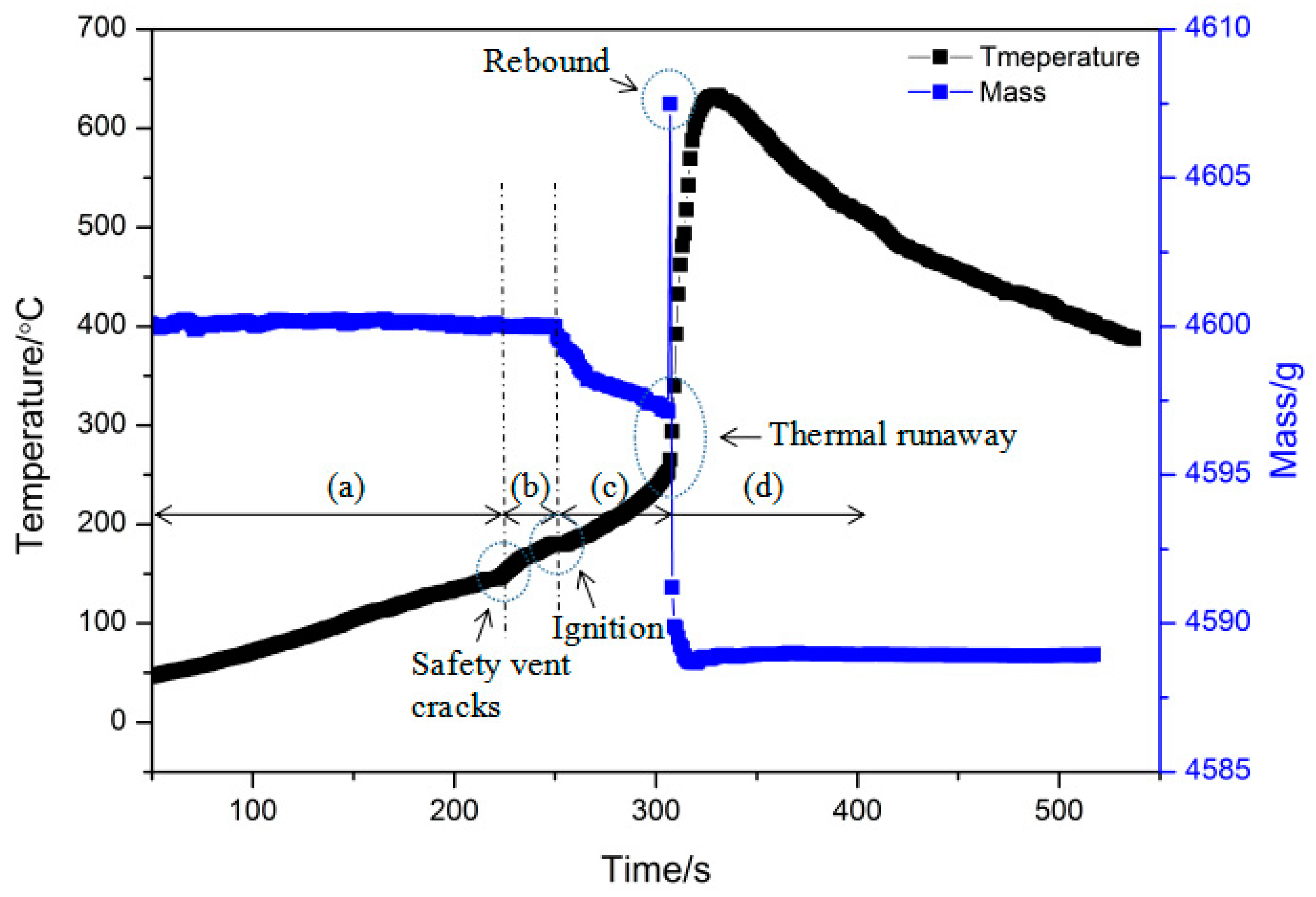
4.2.1. Thermal Hazards of a Single Battery
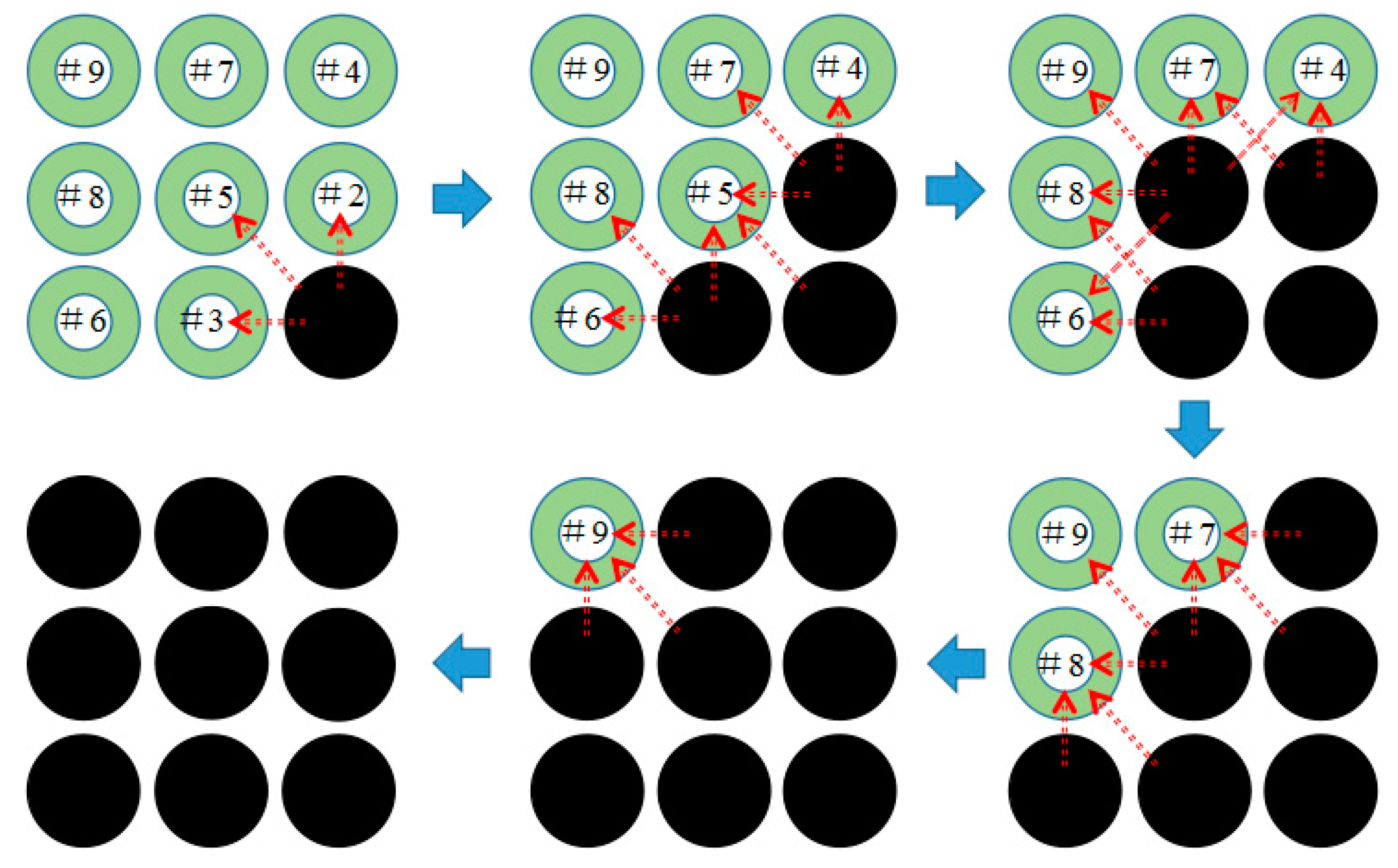
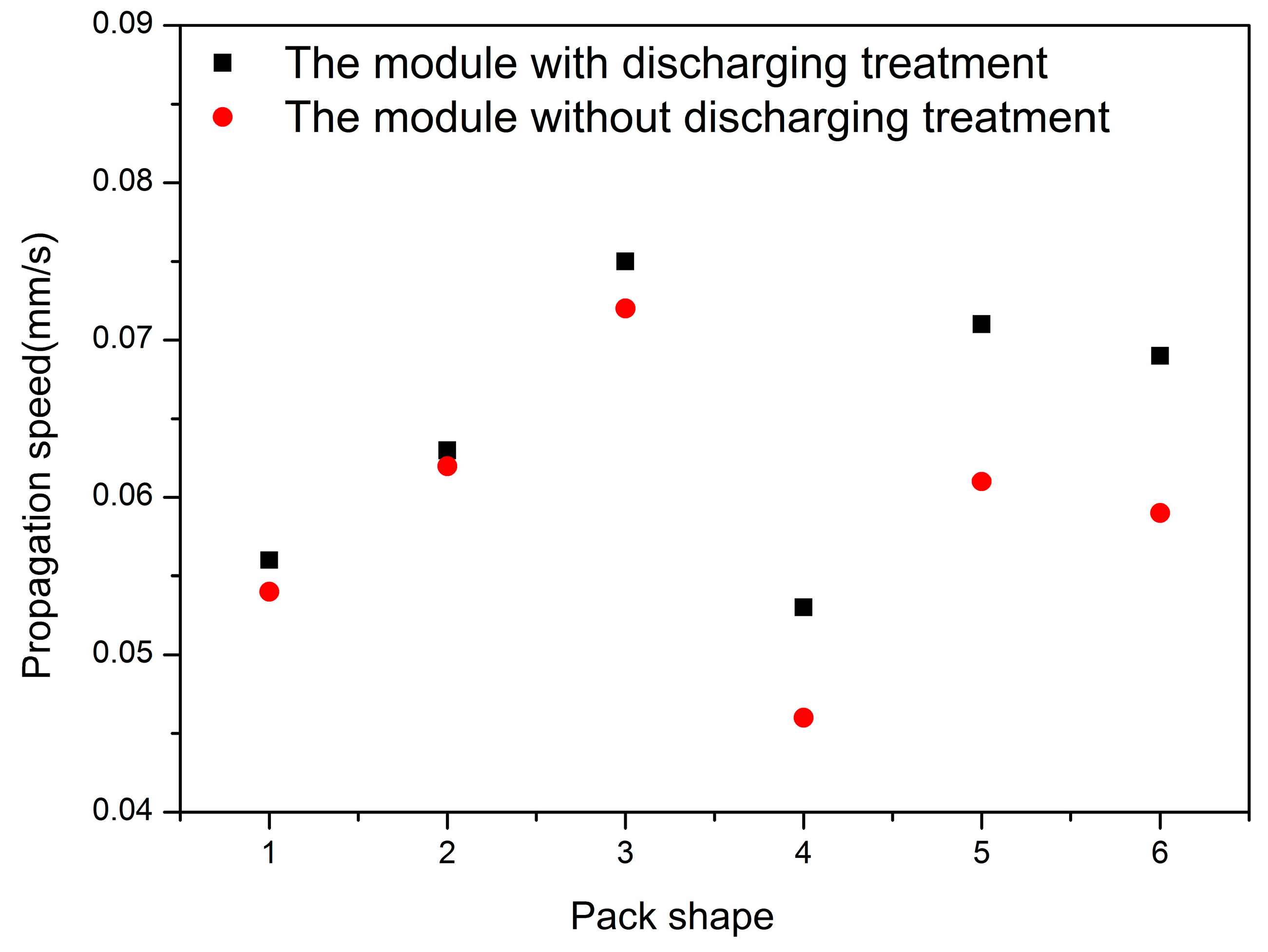
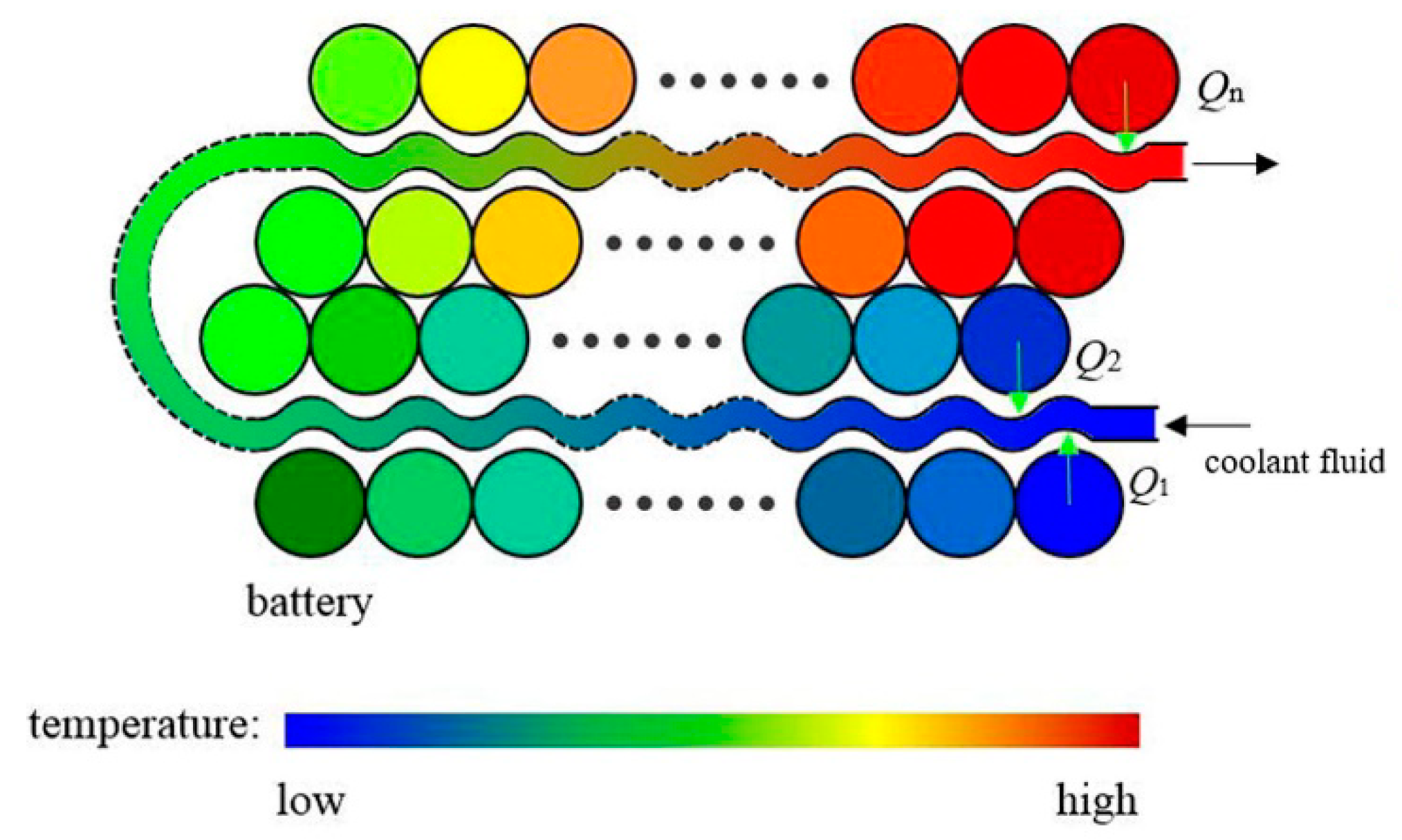
4.2.2. Thermal Hazards of a Battery Pack
4.3. Thermal Hazards of the Lithium-Ion Battery under a Low-Pressure Environment
4.4. Thermal Hazards of Typical Commercial Lithium-Ion Batteries
4.4.1. Lithium cobaltate oxide
4.4.2. Lithium iron phosphate
4.4.3. Lithium nickel manganese cobalt oxide
4.4.4. Lithium nickel cobalt aluminate oxide
5. Management and Countermeasures
5.1. Safety Devices within Battery
5.1.1. Safety Vent
5.1.2. Current Interrupt Device
5.1.3. Positive Temperature Coefficient
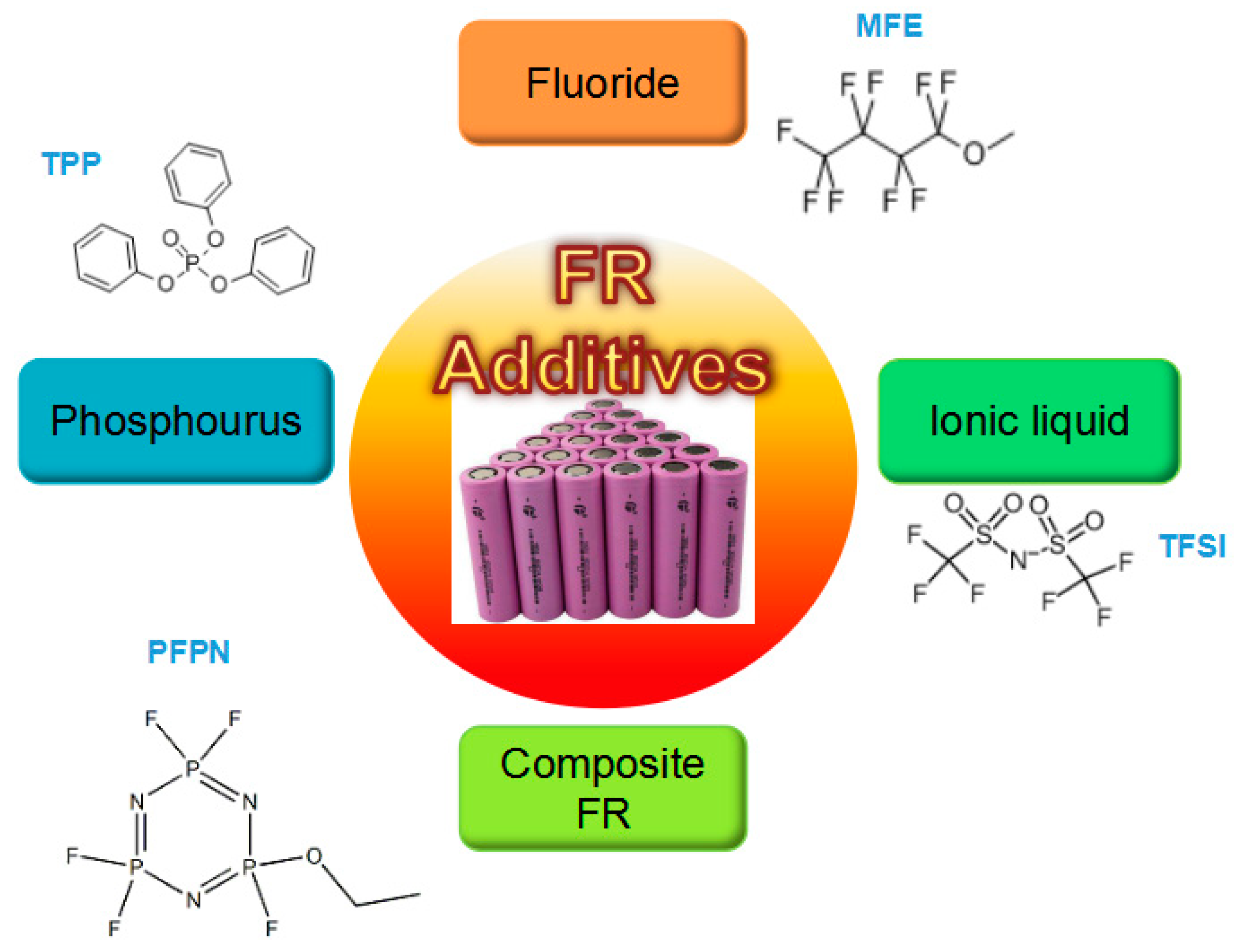
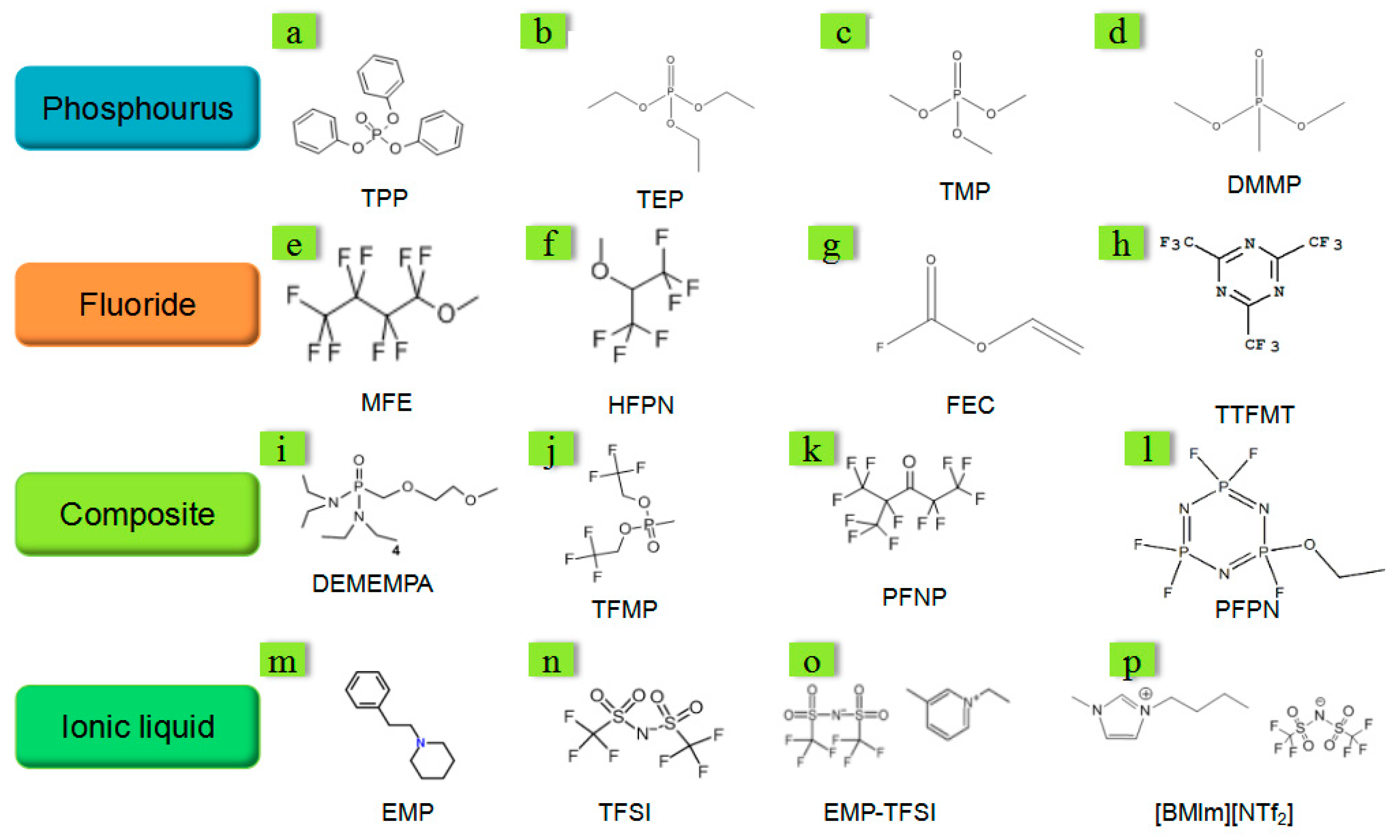
5.2. Fire Retardant
5.2.1. Fire-retardant Electrolyte
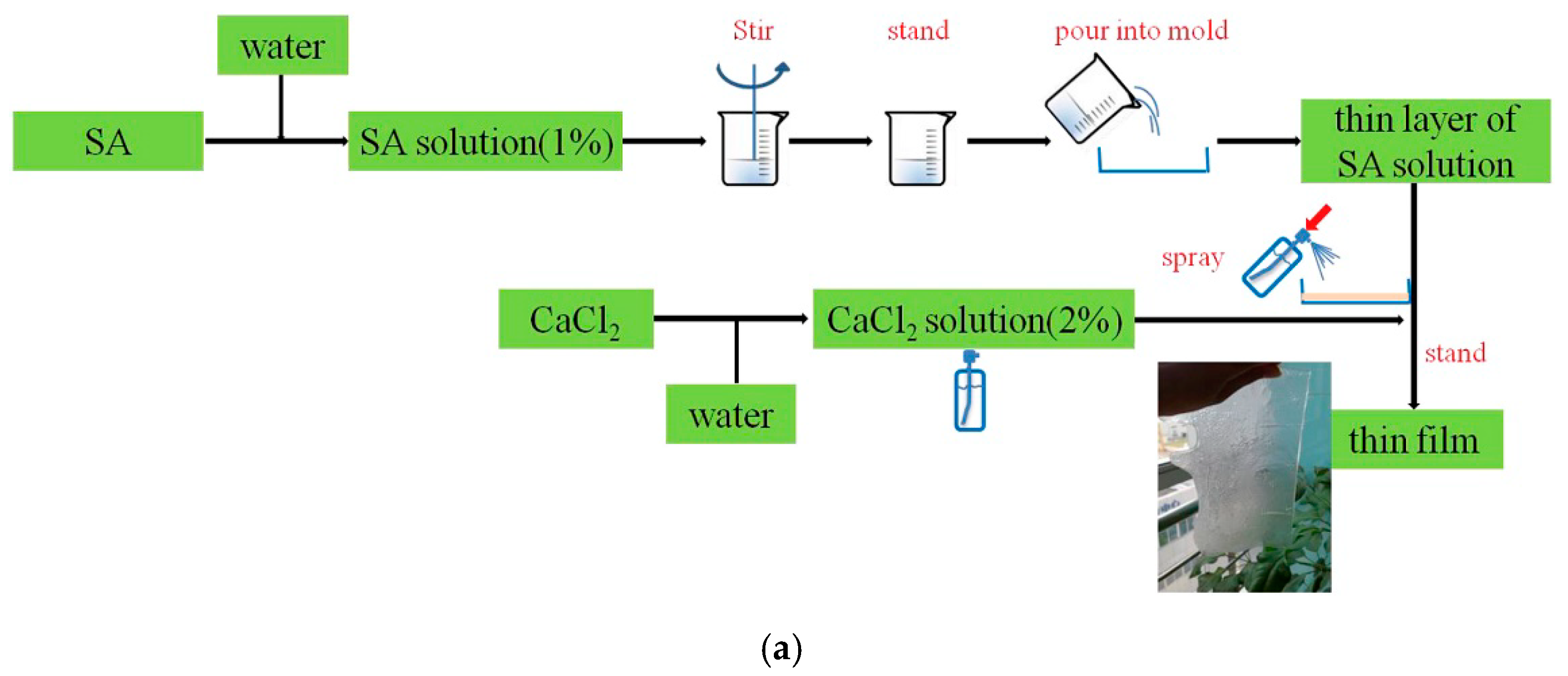
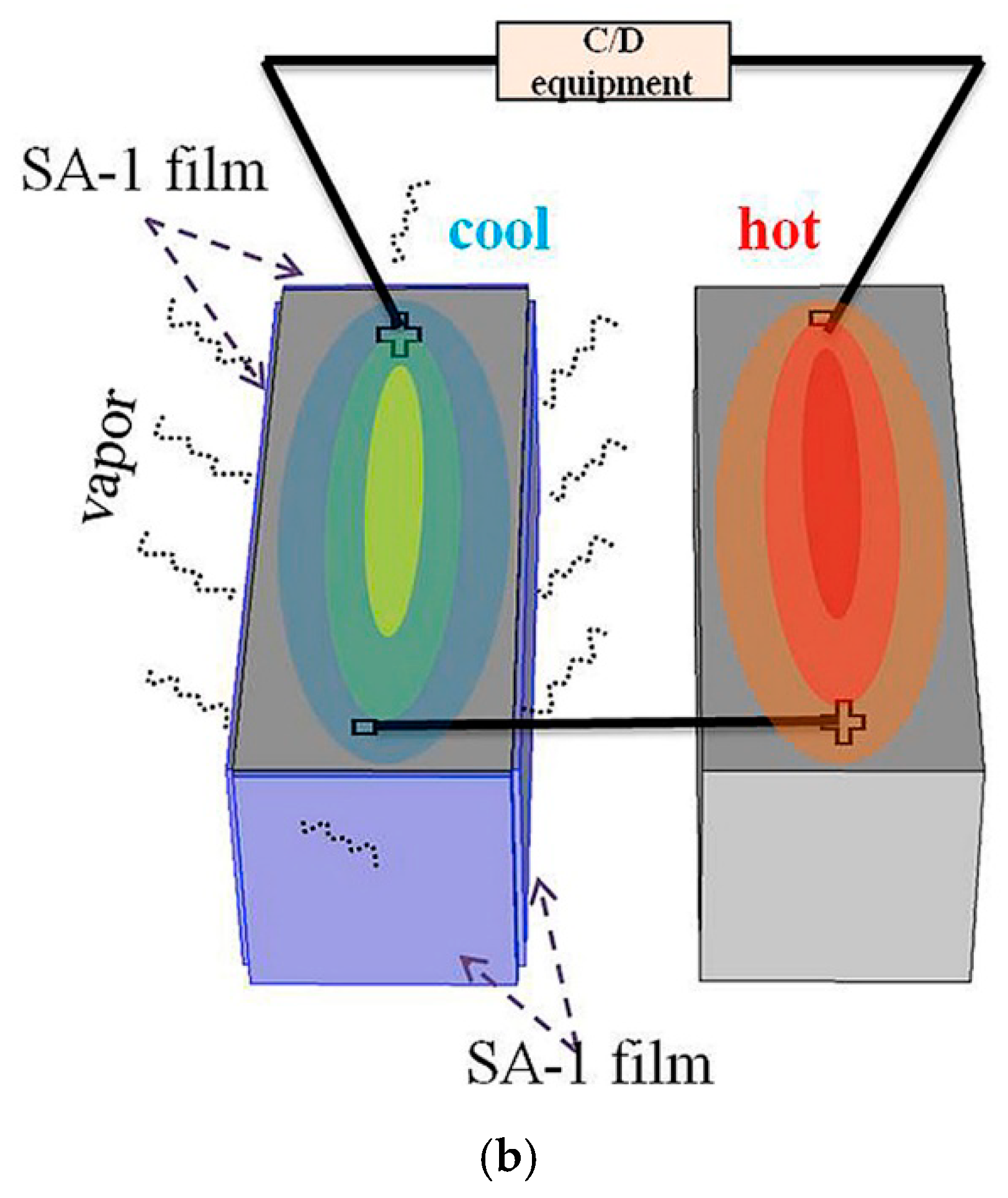
5.2.2. FR Separator
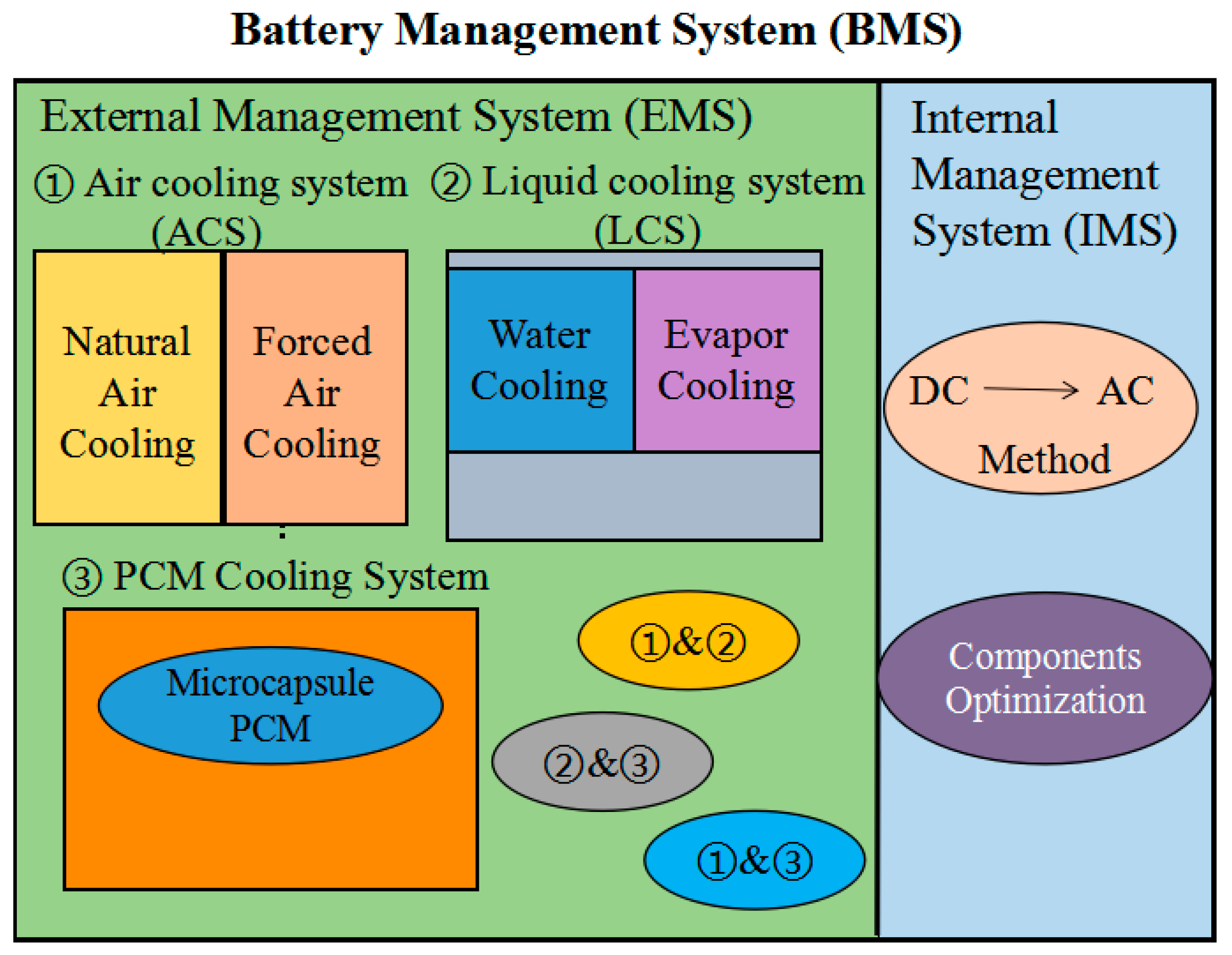
5.3. Battery Management System
5.4. Warning
5.5. Firefighting
6. Summary and Outlook
Author Contributions
Funding
Conflicts of Interest
References
- Kannan, N.; Vakeesan, D. Solar energy for future world: A review. Renew. Sustain. Energy Rev. 2016, 62, 1092–10111. [Google Scholar] [CrossRef]
- Lewis, N.S. Research opportunities to advance solar energy utilization. Science 2016, 351, aad1920. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Collazo, C.; Greaves, D.; Iglesias, G. A review of combined wave and offshore wind energy. Renew. Sustain. Energy Rev. 2015, 42, 141–153. [Google Scholar] [CrossRef]
- Lewis, M.; Neill, S.P.; Robins, P.E.; Hashemi, M.R. Resource assessment for future generations of tidal-stream energy arrays. Energy 2015, 83, 403–415. [Google Scholar] [CrossRef]
- Dyer, C.K. Fuel cells for portable applications. J. Power Sources 2002, 106, 31–34. [Google Scholar] [CrossRef]
- Saevarsdottir, G.; Tao, P.; Stefansson, H.; Harvey, W. Potential use of geothermal energy sources for the production of lithium-ion batteries. Renew. Energy 2014, 61, 17–22. [Google Scholar] [CrossRef]
- Richter, F.; Vie, P.J.S.; Kjelstrup, S.; Burheim, O.S. Measurements of ageing and thermal conductivity in a secondary NMC-hard carbon Li-ion battery and the impact on internal temperature profiles. Electrochim. Acta 2017, 250, 228–237. [Google Scholar] [CrossRef]
- Maleki, H.; Howard, J.N. Effects of overdischarge on performance and thermal stability of a Li-ion cell. J. Power Sources 2006, 160, 1395–1402. [Google Scholar] [CrossRef]
- Doughty, D.H.; Roth, E.P. A general discussion of Li ion battery safety. Electrochem. Soc. Interface 2012, 21, 37–44. [Google Scholar] [CrossRef]
- Belov, D.; Yang, M.H. Failure mechanism of Li-ion battery at overcharge conditions. J. Solid State Electrochem. 2008, 12, 885–894. [Google Scholar] [CrossRef]
- Kim, T.H.; Park, J.S.; Chang, S.K.; Choi, S.; Ryu, J.H.; Song, H.K. The current move of lithium ion batteries towards the next phase. Adv. Energy Mater. 2012, 2, 860–872. [Google Scholar] [CrossRef]
- Lisbona, D.; Snee, T. A review of hazards associated with primary lithium and lithium-ion batteries. Process Saf. Environ. Prot. 2011, 89, 434–442. [Google Scholar] [CrossRef]
- Feng, X.; Ouyang, M.; Liu, X.; Lu, L.; Xia, W.; He, X. Thermal runaway mechanism of lithium ion battery for electric vehicles: A review. Energy Storage Mater. 2018, 10, 246–267. [Google Scholar] [CrossRef]
- Wang, Q.; Ping, P.; Zhao, X.; Chu, G.; Sun, J.; Chen, C. Thermal runaway caused fire and explosion of lithium ion battery. J. Power Sources 2012, 208, 210–224. [Google Scholar] [CrossRef]
- Huang, Q.; Ma, L.; Liu, A.; Ma, X.W.; Li, J.; Wang, J.; Dahn, J.R. The reactivity of charged positive Li1-n[NixMnyCoz]O2 electrodes with electrolyte at elevated temperatures using accelerating rate calorimetry. J. Power Sources 2018, 390, 78–86. [Google Scholar] [CrossRef]
- Ma, L.; Nie, M.; Xia, J.; Dahn, J.R. A systematic study on the reactivity of different grades of charged Li [NixMnyCoz]O2 with electrolyte at elevated temperatures using accelerating rate calorimetry. J. Power Sources 2016, 327, 145–150. [Google Scholar] [CrossRef]
- Xia, L.; Li, S.L.; Ai, X.P.; Yang, H.X.; Cao, Y.L. Temperature-sensitive cathode materials for safer lithium-ion batteries. Energy Environ. Sci. 2011, 4, 2845–2848. [Google Scholar] [CrossRef]
- Zhong, H.; Kong, C.; Zhan, H.; Zhan, C.; Zhou, Y. Safe positive temperature coefficient composite cathode for lithium ion battery. J. Power Sources 2012, 216, 273–280. [Google Scholar] [CrossRef]
- Kovalenko, I.; Zdyrko, B.; Magasinski, A.; Benjamin, H. A major constituent of brown algae for use in high-capacity Li-ion batteries. Science 2011, 334, 75–79. [Google Scholar] [CrossRef]
- Ryou, M.H.; Kim, J.; Lee, I.; Kim, S.J. Mussel-inspired adhesive binders for high-performance silicon nanoparticle anodes in lithium-ion batteries. Adv. Mater. 2013, 25, 1571–1576. [Google Scholar] [CrossRef]
- Wu, H.; Chan, G.; Choi, J.W.; Ryu, I. Stable cycling of double-walled silicon nanotube battery anodes through solid–electrolyte interphase control. Nat. Nanotechnol. 2012, 7, 310–315. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.S.; Kim, T.H.; Lee, H.; Song, H.K. Electronegativity-induced enhancement of thermal stability by succinonitrile as an additive for Li ion batteries. Energy Environ. Sci. 2011, 4, 4038–4045. [Google Scholar] [CrossRef]
- Kim, G.Y.; Dahn, J.R. The effect of some nitriles as electrolyte additives in Li-ion batteries. J. Electrochem. Soc. 2015, 162, A437–A447. [Google Scholar] [CrossRef]
- Chen, R.; Liu, F.; Chen, Y.; Ye, Y.; Huang, Y. An investigation of functionalized electrolyte using succinonitrile additive for high voltage lithium-ion batteries. J. Power Sources 2016, 306, 70–77. [Google Scholar] [CrossRef]
- Zhu, Y.; Wang, F.; Liu, L.; Xiao, S. Composite of a nonwoven fabric with poly (vinylidene fluoride) as a gel membrane of high safety for lithium ion battery. Energy Environ. Sci. 2013, 6, 618–624. [Google Scholar] [CrossRef]
- Zhang, J.; Liu, Z.; Kong, Q. Renewable and superior thermal-resistant cellulose-based composite nonwoven as lithium-ion battery separator. ACS Appl. Mater. Interfaces 2012, 5, 128–134. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.M.; Ryou, M.H.; Choi, J.W. Mussel-and diatom-inspired silica coating on separators yields improved power and safety in Li-ion batteries. Chem. Mater. 2012, 24, 3481–3485. [Google Scholar] [CrossRef]
- Saw, L.H.; Ye, Y.; Tay, A.A.O. Computational fluid dynamic and thermal analysis of Lithium-ion battery pack with air cooling. Appl. Energy 2016, 177, 783–792. [Google Scholar] [CrossRef]
- Huo, Y.; Rao, Z.; Liu, X. Investigation of power battery thermal management by using mini-channel cold plate. Energy Convers. Manag. 2015, 89, 387–395. [Google Scholar] [CrossRef]
- Li, K.; Yan, J.; Chen, H. Water cooling based strategy for lithium ion battery pack dynamic cycling for thermal management system. Appl. Thermal Eng. 2018, 132, 575–585. [Google Scholar] [CrossRef]
- Zhao, J.; Rao, Z.; Huo, Y. Thermal management of cylindrical power battery module for extending the life of new energy electric vehicles. Appl. Thermal Eng. 2015, 85, 33–43. [Google Scholar] [CrossRef]
- Wang, Z.; Li, X.; Zhang, G. Experimental study of a passive thermal management system for three types of battery using copper foam saturated with phase change materials. RSC Adv. 2017, 7, 27441–27448. [Google Scholar] [CrossRef]
- Al-Zareer, M.; Dincer, I.; Rosen, M.A. Novel thermal management system using boiling cooling for high-powered lithium-ion battery packs for hybrid electric vehicles. J. Power Sources 2017, 363, 291–303. [Google Scholar] [CrossRef]
- Lee, C.Y.; Lee, S.J.; Tang, M.S. In situ monitoring of temperature inside lithium-ion batteries by flexible micro temperature sensors. Sensors 2011, 11, 9942–9950. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Yin, P.; Zhu, X. Synthesis of hematite (α-Fe2O3) nanorods: Diameter-size and shape effects on their applications in magnetism, lithium ion battery, and gas sensors. J. Phys. Chem. B 2006, 110, 17806–17812. [Google Scholar] [CrossRef] [PubMed]
- Mutyala, M.S.K.; Zhao, J.; Li, J.; Pan, H.; Yuan, C.; Li, X. In-situ temperature measurement in lithium ion battery by transferable flexible thin film thermocouples. J. Power Sources 2014, 260, 43–49. [Google Scholar] [CrossRef]
- Chen, J.; Xu, L.; Li, W.; Gou, X. α-Fe2O3 nanotubes in gas sensor and lithium-ion battery applications. Adv. Mater. 2005, 17, 582–586. [Google Scholar] [CrossRef]
- Reif, R.H.; Liffers, M.; Forrester, N.; Peal, K. Lithium Battery Safety: A look at Woods Hole Oceanographic Institution’s Program. Prof. Saf. 2010, 55, 32–37. [Google Scholar]
- Liu, Y.; Duan, Q.; Xu, J. Experimental study on the efficiency of dodecafluoro-2-methylpentan-3-one on suppressing lithium-ion battery fires. RSC Adv. 2018, 8, 42223–42232. [Google Scholar] [CrossRef]
- Williard, N.; He, W.; Hendricks, C.; Pecht, M. Lessons learned from the 787 dreamliner issue on lithium-ion battery reliability. Energies 2013, 6, 4682–4695. [Google Scholar] [CrossRef]
- Kolly, J.M.; Panagiotou, J.; Czech, B.A. The Investigation of a Lithium-Ion Battery Fire Onboard a Boeing 787 by the US National Transportation Safety Board; Safety Research Corporation of America: Dothan, AL, USA, 2013; pp. 1–18. [Google Scholar]
- Goto, N. Aircraft Serious Incident Investigation Report: All Nippon Airways Co. Ltd. JA804A; Report No. AI2014-4; Japan Transport Safety Board: Tokyo, Japan, 25 September 2014. [Google Scholar]
- Aircraft Incident Report: Auxiliary Power Unit Battery Fire, Japan Airlines Boeing 787, JA 829 J, Boston, Massachusetts, January 7, 2013; Report No. PB2014-108867; National Transportation Safety Board: Washington, DC, USA, 21 November 2014.
- Abraham, D.P.; Roth, E.P.; Kostecki, R. Diagnostic examination of thermally abused high-power lithium-ion cells. J. Power Sources 2006, 161, 648–657. [Google Scholar] [CrossRef]
- Karter, M.J. Fire Loss in the United States During 2013; National Fire Protection Association: Quincy, MA, USA, 2014. [Google Scholar]
- The Samsung Note7 Exploded. Available online: http://www.sohu.com/a/125020048_467791/ (accessed on 23 January 2017).
- Is the Heart of the Distortion or Moral Bankruptcy? Available online: https://www.caishimv.com/party/1476152753.html/ (accessed on 11 October 2016).
- Huawei P9 Plus Exploded and the Attitude Was Worse than Samsung Note7. Available online: http://www.sohu.com/a/116273061_162764/ (accessed on 16 October 2016).
- An iPhone 7 Took Fire and Burned a Car in Australia. Available online: http://news.mydrivers.com/1/504/504106.htm/ (accessed on 21 October 2016).
- Too Scary! Suddenly, the iPhone Battery Exploded and Seven People Were Hospitalized. Available online: https://baijiahao.baidu.com/s?id=1589274155032747498&wfr=spider&for=pc/ (accessed on 11 January 2018).
- An iPhone XS Max Caught Fire for the First Time and the Users Said They Might Sue Apple. Available online: http://www.sohu.com/a/285734633_499322/ (accessed on 30 December 2018).
- A Summary Analysis on the 35 Fire Accidents of New Energy Vehicles in 2016. Available online: http://www.sohu.com/a/123335110_526255/ (accessed on 4 January 2017).
- Electric Vehicle Fire Accident Inventory in the First Half of 2017. Available online: https://www.sohu.com/a/155869592_118021/ (accessed on 10 July 2017).
- Electric Vehicle Fire Accident Inventory in the First Half of 2018. Available online: http://www.evpartner.com/news/8/detail-36915.html/ (accessed on 8 June 2018).
- Flight UPS6 Crashed in Dubai on 3 September 2010. Available online: http://m.yzwb.net/a/180910225943473.html/ (accessed on 10 September 2018).
- The Boeing 787’s Lithium Battery Troubles. Available online: http://www.360doc.com/content/13/0522/10/7536781_287205631.shtml/ (accessed on 22 May 2013).
- Why Lithium Batteries Are ‘Frowned Upon’ by Aircraft? Available online: http://www.fx361.com/page/2017/0731/2112773.shtml/ (accessed on 31 July 2017).
- Wen, J.; Yu, Y.; Chen, C. A Review on Lithium-Ion Batteries Safety Issues: Existing Problems and Possible Solutions. Mater. Express 2012, 2, 197–212. [Google Scholar] [CrossRef]
- Lyon, R.E.; Walters, R.N. Energetics of lithium ion battery failure. J. Hazard. Mater. 2016, 318, 164–172. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Stoliarov, S.I.; Denlinger, M.; Masias, A.; Snyder, K. Comprehensive calorimetry of the thermally-induced failure of a lithium ion battery. J. Power Sources 2015, 280, 516–525. [Google Scholar] [CrossRef]
- Ouyang, D.; He, Y.; Chen, M.; Liu, J.; Wang, J. Experimental study on the thermal behaviors of lithium-ion batteries under discharge and overcharge conditions. J. Thermal Anal. Calorim. 2018, 132, 65–75. [Google Scholar] [CrossRef]
- Wang, Z.; Ouyang, D.; Chen, M.; Wang, X.; Zhang, Z.; Wang, J. Fire behavior of lithium-ion battery with different states of charge induced by high incident heat fluxes. J. Thermal Anal. Calorim. 2019, 136, 2239–2247. [Google Scholar] [CrossRef]
- Zhang, J.; Lu, B.; Song, Y.; Ji, X. Diffusion induced stress in layered Li-ion battery electrode plates. J. Power Sources 2012, 209, 220–227. [Google Scholar] [CrossRef]
- Riley, L.A.; Cavanagh, A.S.; George, S.M.; Jung, Y.S.; Yan, Y.; Lee, S.H.; Dillon, A.C. Conformal Surface Coatings to Enable High Volume Expansion Li-Ion Anode Materials. ChemPhysChem 2010, 11, 2124–2130. [Google Scholar] [CrossRef] [PubMed]
- Basu, S.; Suresh, S.; Ghatak, K.; Bartolucci, S.; Gupta, T.; Hundekar, P.; Kumar, R.; Lu, T.; Datta, D.; Shi, Y. Utilizing van der Waals slippery interfaces to enhance the electrochemical stability of Silicon film anodes in lithium-ion batteries. ACS Appl. Mater. Interfaces 2018, 10, 13442–13451. [Google Scholar] [CrossRef]
- Wu, Y.; Saxena, S.; Xing, Y.; Wang, Y.; Li, C.; Yung, W.; Pecht, M. Analysis of manufacturing-induced defects and structural deformations in Lithium-ion batteries using computed tomography. Energies 2018, 11, 925. [Google Scholar] [CrossRef]
- Samsung Expands Recall to All Galaxy Note7 Devices. Available online: http://www.samsung.com/us/note7recall/ (accessed on 20 August 2017).
- Mohanty, D.; Hockaday, E.; Li, J.; Hensley, D.K.; Daniel, C.; Wood, D.L., III. Effect of electrode manufacturing defects on electrochemical performance of lithium-ion batteries: Cognizance of the battery failure sources. J. Power Sources 2016, 312, 70–79. [Google Scholar] [CrossRef]
- Cannarella, J.; Arnold, C.B. The effects of defects on localized plating in lithium-ion batteries. J. Electrochem. Soc. 2015, 162, A1365–A1373. [Google Scholar] [CrossRef]
- Kong, D.; Wen, R.; Ping, P.; Peng, R.; Zhang, J.; Chen, G. Study on degradation behavior of commercial 18650 LiAlNiCoO2 cells in over-charge conditions. Int. J. Energy Res. 2019, 43, 552–567. [Google Scholar] [CrossRef]
- Wu, W.; Wu, W.; Qiu, X.; Wang, S. Low-temperature reversible capacity loss and aging mechanism in lithium-ion batteries for different discharge profiles. Int. J. Energy Res. 2019, 43, 243–253. [Google Scholar] [CrossRef]
- Kawamura, T.; Kimura, A.; Egashira, M.; Okada, S.; Yamaki, J.I. Thermal stability of alkyl carbonate mixed-solvent electrolytes for lithium ion cells. J. Power Sources 2002, 104, 260–264. [Google Scholar] [CrossRef]
- Sloop, S.E.; Kerr, J.B.; Kinoshita, K. The role of Li-ion battery electrolyte reactivity in performance decline and self-discharge. J. Power Sources 2003, 119, 330–337. [Google Scholar] [CrossRef]
- Nagasubramanian, G. Comparison of the thermal and electrochemical properties of LiPF6 and LiN(SO2C2F5)2 salts in organic electrolytes. J. Power Sources 2003, 119, 811–814. [Google Scholar] [CrossRef]
- Wang, Q.; Sun, J.; Yao, X.; Chen, C. C80 Calorimeter Studies of the Thermal Behavior of LiPF 6 Solutions. J. Solut. Chem. 2006, 35, 179–189. [Google Scholar] [CrossRef]
- Ribière, P.; Grugeon, S.; Morcrette, M.; Boyanov, S.; Laruelle, S.; Marlair, G. Investigation on the fire-induced hazards of Li-ion battery cells by fire calorimetry. Energy Environ. Sci. 2012, 5, 5271–5280. [Google Scholar] [CrossRef]
- Takata, K.; Morita, M.; Matsuda, Y.; Matsui, K. Cycling characteristics of secondary Li electrode in LiBF4/mixed ether electrolytes. J. Electrochem. Soc. 1985, 132, 126–128. [Google Scholar] [CrossRef]
- Zhang, S.S.; Xu, K.; Jow, T.R. A new approach toward improved low temperature performance of Li-ion battery. Electrochem. Commun. 2002, 4, 928–932. [Google Scholar] [CrossRef]
- Xu, K.; Zhang, S.S.; Lee, U.; Allen, J.L.; Jow, T.R. LiBOB: Is it an alternative salt for lithium ion chemistry? J. Power Sources 2005, 146, 79–85. [Google Scholar] [CrossRef]
- Xu, W.; Angell, C.A. LiBOB and Its Derivatives: Weakly Coordinating Anions, and the Exceptional Conductivity of Their Nonaqueous Solutions. Electrochem. Solid State Lett. 2001, 4, E1–E4. [Google Scholar] [CrossRef]
- Li, Y.; Wang, Z.; Li, C.; Cao, Y.; Guo, X. Densification and ionic-conduction improvement of lithium garnet solid electrolytes by flowing oxygen sintering. J. Power Sources 2014, 248, 642–646. [Google Scholar] [CrossRef]
- Zhang, Y.; Chen, F.; Tu, R.; Shen, Q.; Zhang, L. Field assisted sintering of dense Al-substituted cubic phase Li7La3Zr2O12 solid electrolytes. J. Power Sources 2014, 268, 960–964. [Google Scholar] [CrossRef]
- Kobayashi, Y.; Miyashiro, H.; Takeuchi, T.; Shigemura, H.; Balakrishnan, N.; Tabuchi, M.; Kageyama, H.; Iwahori, T. All-solid-state lithium secondary battery with ceramic/polymer composite electrolyte. Solid State Ion. 2002, s152–s153, 137–142. [Google Scholar] [CrossRef]
- Mei, A.; Wang, X.L.; Lan, J.L.; Feng, Y.C.; Geng, H.X.; Lin, Y.H.; Nan, C.W. Role of amorphous boundary layer in enhancing ionic conductivity of lithium–lanthanum–titanate electrolyte. Electrochim. Acta 2010, 55, 2958–2963. [Google Scholar] [CrossRef]
- Nelson, K.J.; d’Eon, G.L.; Wright, A.T.B.; Ma, L.; Xia, J.; Dahn, J.R. Studies of the effect of high voltage on the impedance and cycling performance of Li [Ni0.4Mn0.4Co0.2]O2/graphite lithium-ion pouch cells. J. Electrochem. Soc. 2015, 162, A1046–A1054. [Google Scholar] [CrossRef]
- Kim, H.; Oh, S.-M.; Scrosati, B.; Sun, Y.-K. High-performance electrode materials for lithium-ion batteries for electric vehicles. Adv. Battery Technol. Electr. Veh. 2015, 191–241. [Google Scholar] [CrossRef]
- Li, J.; Cameron, A.R.; Li, H.; Glazier, S.; Xiong, D.; Chatzidakis, M.; Allen, J.; Botton, G.A.; Dahn, J.R. Comparison of Single Crystal and Polycrystalline LiNi0.5Mn0.3Co0.2O2 Positive Electrode Materials for High Voltage Li-Ion Cells. J. Electrochem. Soc. 2017, 164, A1534–A1544. [Google Scholar] [CrossRef]
- Zhang, Z.; Fouchard, D.; Rea, J.R. Differential scanning calorimetry material studies: Implications for the safety of lithium-ion cells. J. Power Sources 1998, 70, 16–20. [Google Scholar] [CrossRef]
- Wang, Q.; Sun, J.; Chen, X.; Chu, G.; Chen, C. Effects of solvents and salt on the thermal stability of charged LiCoO2. Mater. Res. Bull. 2009, 44, 543–548. [Google Scholar] [CrossRef]
- Wang, Q.; Sun, J.; Yao, X.; Chen, C. Thermal Behavior of Lithiated Graphite with Electrolyte in Lithium-Ion Batteries. J. Electrochem. Soc. 2006, 153, A329–A333. [Google Scholar] [CrossRef]
- Yamaki, J.I.; Takatsuji, H.; Kawamura, T.; Egashira, M. Thermal stability of graphite anode with electrolyte in lithium-ion cells. Solid State Ion. 2002, 148, 241–245. [Google Scholar] [CrossRef]
- Zinigrad, E.; Larush-Asraf, L.; Gnanaraj, J.S.; Gottlieb, H.E.; Sprecher, M.; Aurbach, D. Calorimetric studies of the thermal stability of electrolyte solutions based on alkyl carbonates and the effect of the contact with lithium. J. Power Sources 2005, 146, 176–179. [Google Scholar] [CrossRef]
- Bowden, P.B.; Young, R.J. Deformation mechanisms in crystalline polymers. J. Mater. Sci. 1974, 9, 2034–2051. [Google Scholar] [CrossRef]
- Liu, N.; Wu, H.; Mcdowell, M.T.; Yao, Y.; Wang, C.; Cui, Y. A yolk-shell design for stabilized and scalable Li-ion battery alloy anodes. Nano Lett. 2012, 12, 3315–3321. [Google Scholar] [CrossRef]
- Wagemaker, M.; Mulder, F.M. Properties and promises of nanosized insertion materials for Li-ion batteries. Acc. Chem. Res. 2012, 46, 1206–1215. [Google Scholar] [CrossRef]
- Zhong, Y.J.; Li, J.T.; Wu, Z.G.; Guo, X.D.; Zhong, B.H.; Sun, S.G. LiMn0.5Fe0.5PO4 solid solution materials synthesized by rheological phase reaction and their excellent electrochemical performances as cathode of lithium ion battery. J. Power Sources 2013, 234, 217–222. [Google Scholar] [CrossRef]
- Lu, C.; Qi, W.; Li, L.; Xu, J.; Chen, P.; Xu, R.; Han, L.; Yu, Q. Electrochemical performance and thermal property of electrospun PPESK/PVDF/PPESK composite separator for lithium-ion battery. J. Appl. Electrochem. 2013, 43, 711–720. [Google Scholar] [CrossRef]
- Gong, W.; Wei, S.; Ruan, S.; Shen, C. Electrospun coaxial PPESK/PVDF fibrous membranes with thermal shutdown property used for lithium-ion batteries. Mater. Lett. 2019, 244, 126–129. [Google Scholar] [CrossRef]
- Zhang, S.; Liu, P.; Chen, Y.; Jin, J.; Hu, L.; Jian, X. Preparation of thermally stable composite forward osmosis hollow fiber membranes based on copoly (phthalazinone biphenyl ether sulfone) substrates. Chem. Eng. Sci. 2017, 166, 91–100. [Google Scholar] [CrossRef]
- Wang, G.; Zhang, H.; Qian, B.; Wang, J.; Jian, X.; Qiu, J. Preparation and characterization of electrospun poly (phthalazinone ether nitrile ketone) membrane with novel thermally stable properties. Appl. Surf. Sci. 2015, 351, 169–174. [Google Scholar] [CrossRef]
- Kim, G.H.; Pesaran, A.; Spotnitz, R. A three-dimensional thermal abuse model for lithium-ion cells. J. Power Sources 2007, 170, 476–489. [Google Scholar] [CrossRef]
- Spotnitz, R.; Franklin, J. Abuse behavior of high-power, lithium-ion cells. J. Power Sources 2003, 113, 81–100. [Google Scholar] [CrossRef]
- Venugopal, G. Characterization of thermal cut-off mechanisms in prismatic lithium-ion batteries. J. Power Sources 2001, 101, 231–237. [Google Scholar] [CrossRef]
- Wang, Q.; Sun, J. Enhancing the safety of lithium ion batteries by 4-isopropyl phenyl diphenyl phosphate. Mater. Lett. 2007, 61, 3338–3340. [Google Scholar] [CrossRef]
- Ouyang, D.; Liu, J.; Chen, M.; Weng, J.; Wang, J. An experimental study on the thermal failure propagation in lithium-ion battery pack. J. Electrochem. Soc. 2018, 165, A2184–A2193. [Google Scholar] [CrossRef]
- Huang, P.; Wang, Q.; Li, K.; Ping, P.; Sun, J. The combustion behavior of large scale lithium titanate battery. Sci. Rep. 2015, 5, 7788. [Google Scholar] [CrossRef]
- Ouyang, D.; Liu, J.; Chen, M.; Wang, J. Investigation into the Fire Hazards of Lithium-Ion Batteries under Overcharging. Appl. Sci. 2017, 7, 1314. [Google Scholar] [CrossRef]
- Ping, P.; Wang, Q.S.; Huang, P.F.; Li, K.; Sun, J.H.; Kong, D.P.; Chen, C.H. Study of the fire behavior of high-energy lithium-ion batteries with full-scale burning test. J. Power Sources 2015, 285, 80–89. [Google Scholar] [CrossRef]
- Chen, M.; Zhou, D.; Chen, X.; Zhang, W.; Liu, J.; Yuen, R.; Wang, J. Investigation on the thermal hazards of 18650 lithium ion batteries by fire calorimeter. J. Therm. Anal. Calorim. 2015, 122, 755–763. [Google Scholar] [CrossRef]
- Fu, Y.; Lu, S.; Shi, L.; Cheng, X.; Zhang, H. Ignition and combustion characteristics of lithium ion batteries under low atmospheric pressure. Energy 2018, 161, 38–45. [Google Scholar] [CrossRef]
- Ouyang, D.; Chen, M.; Liu, J.; Wei, R.; Weng, J.; Wang, J. Investigation of a commercial lithium-ion battery under overcharge/over-discharge failure conditions. RSC Adv. 2018, 8, 33414–33424. [Google Scholar] [CrossRef]
- Jhu, C.Y.; Wang, Y.W.; Shu, C.M.; Chang, J.C.; Wu, H.C. Thermal explosion hazards on 18650 lithium ion batteries with a VSP2 adiabatic calorimeter. J. Hazard. Mater. 2011, 192, 99–107.87. [Google Scholar] [CrossRef]
- Larsson, F.; Andersson, P.; Blomqvist, P.; Lorén, A.; Mellander, B.E. Characteristics of lithium-ion batteries during fire tests. J. Power Sources 2014, 271, 414–420. [Google Scholar] [CrossRef]
- Golubkov, A.W.; Fuchs, D.; Wagner, J.; Wiltsche, H.; Stangl, C.; Fauler, G.; Voitic, G.; Thaler, A.; Hacher, V. Thermal-runaway experiments on consumer Li-ion batteries with metal-oxide and olivin-type cathodes. RSC Adv. 2014, 4, 3633–3642. [Google Scholar] [CrossRef]
- Sun, J.; Li, J.; Zhou, T.; Yang, K.; Wei, S.; Tang, N.; Dang, N.; Li, H.; Qiu, X.; Chen, L. Toxicity, a serious concern of thermal runaway from commercial Li-ion battery. Nano Energy 2016, 27, 313–319. [Google Scholar] [CrossRef]
- Yang, H.; Zhuang, G.V.; Ross, P.N. Thermal stability of LiPF6 salt and Li-ion battery electrolytes containing LiPF6. J. Power Sources 2006, 161, 573–579. [Google Scholar] [CrossRef]
- Maleki, H.; Deng, G.; Anani, A. Thermal Stability Studies of Li-Ion Cells and Components. J. Electrochem. Soc. 1999, 146, 3224–3229. [Google Scholar] [CrossRef]
- Gray, F.M. Solid Polymer Electrolytes; VCH: New York, NY, USA, 1991. [Google Scholar]
- Braun, J.; Stöss, H.; Zober, A. Intoxication following the inhalation of hydrogen fluoride. Arch. Toxicol. 1984, 56, 50–54. [Google Scholar] [CrossRef] [PubMed]
- Ouyang, D.; Chen, M.; Wei, R.; Wang, Z.; Wang, J. A study on the fire behaviors of 18650 battery and batteries pack under discharge. J. Therm. Anal. Calorim. 2019, 136, 1915–1926. [Google Scholar] [CrossRef]
- Chen, M.; Liu, J.; He, Y.; Yuen, R.; Wang, J. Study of the fire hazards of lithium-ion batteries at different pressures. Appl. Therm. Eng. 2017, 125, 1061–1074. [Google Scholar] [CrossRef]
- Wagner, M.W.; Liebenow, C.; Besenhard, J.O. Effect of polysulfide-containing electrolyte on the film formation of the negative electrode. J. Power Sources 1997, 68, 328–332. [Google Scholar] [CrossRef]
- Wrodnigg, G.H.; Besenhard, J.O.; Winter, M. Cyclic and acyclic sulfites: New solvents and electrolyte additives for lithium ion batteries with graphitic anodes? J. Power Sources 2001, 97, 592–594. [Google Scholar] [CrossRef]
- Lee, J.H.; Paik, U.; Hackley, V.A.; Choi, Y.M. Effect of carboxymethyl cellulose on aqueous processing of natural graphite negative electrodes and their electrochemical performance for lithium batteries. J. Electrochem. Soc. 2005, 152, A1763–A1769. [Google Scholar] [CrossRef]
- Ouyang, D.; Liu, J.; Chen, M.; Weng, J.; Wang, J. Thermal Failure Propagation in Lithium-Ion Battery Modules with Various Shapes. Appl. Sci. 2018, 8, 1263. [Google Scholar] [CrossRef]
- Lopez, C.F.; Jeevarajan, J.A.; Mukherjee, P.P. Experimental analysis of thermal runaway and propagation in lithium-ion battery modules. J. Electrochem. Soc. 2015, 162, A1905–A1915. [Google Scholar] [CrossRef]
- Lamb, J.; Orendorff, C.J.; Steele, L.A.M.; Spangler, S.W. Failure propagation in multi-cell lithium ion batteries. J. Power Sources 2015, 283, 517–523. [Google Scholar] [CrossRef]
- Larsson, F.; Anderson, J.; Andersson, P.; Mellander, B.E. Thermal modelling of cell-to-cell fire propagation and cascading thermal runaway failure effects for lithium-ion battery cells and modules using fire walls. J. Electrochem. Soc. 2016, 163, A2854–A2865. [Google Scholar] [CrossRef]
- Feng, X.; He, X.; Ouyang, M.; Lu, L.; Wu, P.; Kulp, C.; Prasser, S. Thermal runaway propagation model for designing a safer battery pack with 25 Ah LiNixCoyMnzO2 large format lithium ion battery. Appl. Energy 2015, 154, 74–91. [Google Scholar] [CrossRef]
- Wilke, S.; Schweitzer, B.; Khateeb, S.; Al-Hallaj, S. Preventing thermal runaway propagation in lithium ion battery packs using a phase change composite material: An experimental study. J. Power Sources 2017, 340, 51–59. [Google Scholar] [CrossRef]
- Spinner, N.S.; Field, C.R.; Hammond, M.H.; Williams, B.A.; Myers, K.M.; Lubrano, A.L.; Rose- Pehrsson, S.L.; Tuttle, S.G. Physical and chemical analysis of lithium-ion battery cell-to-cell failure events inside custom fire chamber. J. Power Sources 2015, 279, 713–721. [Google Scholar] [CrossRef]
- Gao, S.; Feng, X.; Lu, L. Thermal Runaway Propagation Assessment of Different Battery Pack Designs Using the TF5 Draft as Framework. J. Electrochem. Soc. 2019, 166, A1653–A1659. [Google Scholar] [CrossRef]
- Feng, X.; Sun, J.; Ouyang, M.; Wang, F.; He, X.; Lu, L.; Peng, H. Characterization of penetration induced thermal runaway propagation process within a large format lithium ion battery module. J. Power Sources 2015, 275, 261–273. [Google Scholar] [CrossRef]
- Alshaer, W.G.; Nada, S.A.; Rady, M.A.; Le Bot, C.; Del Barrio, E.P. Numerical investigations of using carbon foam/PCM/Nano carbon tubes composites in thermal management of electronic equipment. Energy Convers. Manag. 2015, 89, 873–884. [Google Scholar] [CrossRef]
- Şahan, N.; Fois, M.; Paksoy, H. Improving thermal conductivity phase change materials—A study of paraffin nanomagnetite composites. Sol. Energy Mater. Sol. Cells 2015, 137, 61–67. [Google Scholar] [CrossRef]
- Feng, X.; Lu, L.; Ouyang, M.; Li, J.; He, X. A 3D thermal runaway propagation model for a large format lithium ion battery module. Energy 2016, 115, 194–208. [Google Scholar] [CrossRef]
- Coman, P.T.; Darcy, E.C.; Veje, C.T.; White, R.E. Numerical analysis of heat propagation in a battery pack using a novel technology for triggering thermal runaway. Appl. Energy 2017, 203, 189–200. [Google Scholar] [CrossRef]
- Coman, P.T.; Rayman, S.; White, R.E. A lumped model of venting during thermal runaway in a cylindrical Lithium Cobalt Oxide lithium-ion cell. J. Power Sources 2016, 307, 56–62. [Google Scholar] [CrossRef]
- Richard, M.N.; Dahn, J.R. Accelerating rate calorimetry study on the thermal stability of lithium intercalated graphite in electrolyte. I. Experimental. J. Electrochem. Soc. 1999, 146, 2068–2077. [Google Scholar] [CrossRef]
- Richard, M.N.; Dahn, J.R. Accelerating rate calorimetry study on the thermal stability of lithium intercalated graphite in electrolyte. II. Modeling the results and predicting differential scanning calorimeter curves. J. Electrochem. Soc. 1999, 146, 2078–2084. [Google Scholar] [CrossRef]
- Guo, G.; Long, B.; Cheng, B. Three-dimensional thermal finite element modeling of lithium-ion battery in thermal abuse application. J. Power Sources 2010, 195, 2393–2398. [Google Scholar] [CrossRef]
- Hatchard, T.D.; MacNeil, D.D.; Basu, A.; Dahn, J.R. Thermal model of cylindrical and prismatic lithium-ion cells. J. Electrochem. Soc. 2001, 148, A755–A761. [Google Scholar] [CrossRef]
- Yeow, K.F.; Teng, H. Characterizing thermal runaway of lithium-ion cells in a battery system using finite element analysis approach. Sae Int. J. Altern. Powertrains 2013, 2, 179–186. [Google Scholar] [CrossRef]
- Spotnitz, R.M.; Weaver, J.; Yeduvaka, G.; Doughty, D.H.; Roth, E.P. Simulation of abuse tolerance of lithium-ion battery packs. J. Power Sources 2007, 163, 1080–1086. [Google Scholar] [CrossRef]
- Kieler, A. At Least 17 Lithium-Ion Batteries Have Exploded on Planes This Year. Available online: https://consumerist.com/2017/06/09/at-least-17-lithium-ion-batteries-have-exploded-on-planes-this-year/ (accessed on 6 September 2017).
- Yafei, W.; Lizhong, Y.; Xiaodong, Z.; Jiakun, D.; Yupeng, Z.; Zhihua, D. Experiment study of the altitude effects on spontaneous ignition characteristics of wood. Fuel 2010, 89, 1029–1034. [Google Scholar] [CrossRef]
- Niu, Y.; He, Y.; Hu, X.; Zhou, D.; Lin, C.; Yin, J.; Yao, W. Experimental study of burning rates of cardboard box fires near sea level and at high altitude. Proc. Combust. Inst. 2013, 34, 2565–2573. [Google Scholar] [CrossRef]
- Yao, W.; Hu, X.; Rong, J.; Wang, J.; Zhang, H. Experimental study of large-scale fire behavior under low pressure at high altitude. J. Fire Sci. 2013, 31, 481–494. [Google Scholar] [CrossRef]
- Li, Z.; He, Y.; Zhang, H.; Wang, J. Combustion characteristics of n-heptane and wood crib fires at different altitudes. Proc. Combust. Inst. 2009, 32, 2481–2488. [Google Scholar] [CrossRef]
- McAllister, S.; Fernandez-Pello, C.; Urban, D.; Ruff, G. The combined effect of pressure and oxygen concentration on piloted ignition of a solid combustible. Combust. Flame 2010, 157, 1753–1759. [Google Scholar] [CrossRef]
- Zarzecki, M.; Quintiere, J.G.; Lyon, R.E.; Rossmann, T.; Diez, F.J. The effect of pressure and oxygen concentration on the combustion of PMMA. Combust. Flame 2013, 160, 1519–1530. [Google Scholar] [CrossRef]
- Weng, W.G.; Hasemi, Y.; Fan, W.C. Predicting the pyrolysis of wood considering char oxidation under different ambient oxygen concentrations. Combust. Flame 2006, 145, 723–729. [Google Scholar] [CrossRef]
- Liu, J.; He, Y.; Zhou, Z.; Yao, W.; Yuen, R.; Wang, J. The burning behaviors of pool fire flames under low pressure. Fire Mater. 2016, 40, 318–334. [Google Scholar] [CrossRef]
- Hu, X.; He, Y.; Li, Z.; Wang, J. Combustion characteristics of n-heptane at high altitudes. Proc. Combust. Inst. 2011, 33, 2607–2615. [Google Scholar] [CrossRef]
- Tu, R.; Fang, J.; Zhang, Y.M.; Zhang, J.; Zeng, Y. Effects of low air pressure on radiation-controlled rectangular ethanol and n-heptane pool fires. Proc. Combust. Inst. 2013, 34, 2591–2598. [Google Scholar] [CrossRef]
- Tao, C.; He, Y.; Li, Y.; Wang, X. Effects of oblique air flow on burning rates of square ethanol pool fires. J. Hazard. Mater. 2013, 260, 552–562. [Google Scholar] [CrossRef] [PubMed]
- Yin, J.; Yao, W.; Liu, Q.; Zhou, Z.; Wu, N.; Zhang, H.; Lin, C.H.; Wu, T.; Meier, O.C. Experimental study of n-Heptane pool fire behavior in an altitude chamber. Int. J. Heat Mass Transf. 2013, 62, 543–552. [Google Scholar] [CrossRef]
- Jiusheng, Y.; Wei, Y.; Quanyi, L.; Wu, N.; Zhou, Z.; Wu, Y.; Zhang, H. Experimental study of n-heptane pool fire behaviors under dynamic pressures in an altitude chamber. Procedia Eng. 2013, 52, 548–556. [Google Scholar] [CrossRef]
- Yao, J.; Liu, J.; Chen, X.; Li, H.; Niu, Y.; Zhou, Z.; Wang, J. Experimental study of small scale n-heptane pool fire with water bath in an altitude chamber. Int. J. Heat Mass Transf. 2015, 90, 1153–1159. [Google Scholar] [CrossRef]
- Fang, J.; Tu, R.; Guan, J.; Wang, J.; Zhang, Y. Influence of low air pressure on combustion characteristics and flame pulsation frequency of pool fires. Fuel 2011, 90, 2760–2766. [Google Scholar] [CrossRef]
- Liu, J.; He, Y.; Zhou, Z.; Yuen, R.; Wang, J. Investigation of enclosure effect of pressure chamber on the burning behavior of a hydrocarbon fuel. Appl. Therm. Eng. 2016, 101, 202–216. [Google Scholar] [CrossRef]
- Zeng, Y.; Fang, J.; Tu, R.; Wang, J.; Zhang, Y. Study on burning characteristics of small-scale ethanol pool fire in closed and open space under low air pressure. In ASME 2011 International Mechanical Engineering Congress and Exposition; American Society of Mechanical Engineers: New York, NY, USA, 2011; pp. 1423–1430. [Google Scholar]
- Chen, M.; Liu, J.; Lin, X.; Huang, Q.; Yuen, R.; Wang, J. Combustion characteristics of primary lithium battery at two altitudes. J. Therm. Anal. Calorim. 2016, 124, 865–870. [Google Scholar] [CrossRef]
- Mizushima, K.; Jones, P.; Wiseman, P.; Goodenough, J.B. LixCoO2 (0 < x < −1): A new cathode material for batteries of high energy density. Mater. Res. Bull. 1980, 15, 783–789. [Google Scholar]
- Thackeray, M.; David, W.; Bruce, P.; Goodenough, J.B. Lithium insertion into manganese spinels. Mater. Res. Bull. 1983, 18, 461–472. [Google Scholar] [CrossRef]
- Padhi, A.K.; Nanjundaswamy, K.S.; Goodenough, J.B. Phospho-olivines as positive-electrode materials for rechargeable lithium batteries. J. Electrochem. Soc. 1997, 144, 1188–1194. [Google Scholar] [CrossRef]
- Kraytsberg, A.; Ein-Eli, Y. Higher, Stronger, Better… A Review of 5 Volt Cathode Materials for Advanced Lithium-Ion Batteries. Adv. Energy Mater. 2012, 2, 922–939. [Google Scholar] [CrossRef]
- Takahashi, Y.; Tode, S.; Kinoshita, A.; Fujimoto, H.; Nakane, I.; Fujitani, S. Development of Lithium-Ion Batteries with a LiCoO2 Cathode Toward High Capacity by Elevating Charging Potential. J. Electrochem. Soc. 2008, 155, A537–A541. [Google Scholar] [CrossRef]
- Yin, R.-Z.; Kim, Y.-S.; Shin, S.-J.; Jung, I.; Kim, J.S.; Jeong, S.K. In situ XRD investigation and thermal properties of Mg doped LiCoO2 for lithium ion batteries. J. Electrochem. Soc. 2012, 159, A253–A258. [Google Scholar] [CrossRef]
- Ahn, S.; Kim, H.-S.; Yang, S.; Do, J.Y.; Kim, B.H.; Kim, K. Thermal stability and performance studies of LiCo1/3Ni1/3Mn1/3O2 with phosphazene additives for Li-ion batteries. J. Electroceram. 2009, 23, 289–294. [Google Scholar] [CrossRef]
- Zhang, L.; Ma, Y.; Cheng, X.; Du, C.; Guan, T.; Cui, Y.; Sun, S.; Zuo, P.; Gao, Y.; Yin, G. Capacity fading mechanism during long-term cycling of over-discharged LiCoO2/mesocarbon microbeads battery. J. Power Sources 2015, 293, 1006–1015. [Google Scholar] [CrossRef]
- Ji, Y.; Zhang, P.; Lin, M.; Zhao, W.; Zhang, Z.; Zhao, Y.; Yang, Y. Toward a stable electrochemical interphase with enhanced safety on high-voltage LiCoO2 cathode: A case of phosphazene additives. J. Power Sources 2017, 359, 391–399. [Google Scholar] [CrossRef]
- Deng, Y.; Kang, T.; Ma, Z.; Tan, X.; Song, X.; Wang, Z.; Pang, P.; Shu, D.; Zuo, X.; Nan, J. Safety influences of the Al and Ti elements modified LiCoO2 materials on LiCoO2/graphite batteries under the abusive conditions. Electrochim. Acta 2019, 295, 703–709. [Google Scholar] [CrossRef]
- Yamada, A.; Chung, S.-C.; Hinokuma, K. Optimized LiFePO4 for lithium battery cathodes. J. Electrochem. Soc. 2001, 148, A224–A229. [Google Scholar] [CrossRef]
- Jiang, J.; Dahn, J. ARC studies of the thermal stability of three different cathode materials: LiCoO2; Li[Ni0.1Co0.8Mn0.1]O2; and LiFePO4, in LiPF6 and LiBoB EC/DEC electrolytes. Electrochem. Commun. 2004, 6, 39–43. [Google Scholar] [CrossRef]
- Bazzi, K.; Nazri, M.; Naik, V.; Garg, V.K.; Oliveira, A.C.; Nazri, G.A.; Naik, R. Enhancement of electrochemical behavior of nanostructured LiFePO4/Carbon cathode material with excess Li. J. Power Sources 2016, 306, 17–23. [Google Scholar] [CrossRef]
- Johnson, I.D.; Lübke, M.; Wu, O.Y.; Makwana, N.M.; Smales, G.J.; Islam, H.U.; Dedigama, R.Y.; Gruar, R.I.; Tighe, C.J.; Scanlon, D.O.; et al. Pilot-scale continuous synthesis of a vanadium-doped LiFePO4/C nanocomposite high-rate cathodes for lithium-ion batteries. J. Power Sources 2016, 302, 410–418. [Google Scholar] [CrossRef]
- Huang, Y.; Liu, H.; Gong, L.; Hou, Y.; Li, Q. A simple route to improve rate performance of LiFePO4/reduced graphene oxide composite cathode by adding Mg2+ via mechanical mixing. J. Power Sources 2017, 347, 29–36. [Google Scholar] [CrossRef]
- Ohzuku, T.; Makimura, Y. Layered lithium insertion material of LiCo1/3Ni1/3Mn1/3O2 for lithium-ion batteries. Chem. Lett. 2001, 30, 642–643. [Google Scholar] [CrossRef]
- Liu, Z.; Yu, A.; Lee, J.Y. Synthesis and characterization of LiNi1−x−yCoxMnyO2 as the cathode materials of secondary lithium batteries. J. Power Sources 1999, 81, 416–419. [Google Scholar] [CrossRef]
- Myung, S.-T.; Maglia, F.; Park, K.-J.; Yoon, C.S.; Lamp, P.; Kim, S.J.; Sun, Y.K. Nickel-rich layered cathode materials for automotive lithium-ion batteries: Achievements and perspectives. ACS Energy Lett. 2016, 2, 196–223. [Google Scholar] [CrossRef]
- Ma, L.; Xia, J.; Xia, X.; Dahn, J.R. The impact of vinylene carbonate, fluoroethylene carbonate and vinyl ethylene carbonate electrolyte additives on electrode/electrolyte reactivity studied using accelerating rate calorimetry. J. Electrochem. Soc. 2014, 161, A1495–A1498. [Google Scholar] [CrossRef]
- Ma, L.; Xia, J.; Dahn, J. Ternary electrolyte additive mixtures for Li-ion cells that promote long lifetime and less reactivity with charged electrodes at elevated temperatures. J. Electrochem. Soc. 2015, 162, A1170–A1174. [Google Scholar] [CrossRef]
- Liu, X.; Ren, D.; Hsu, H.; Feng, X.; Xu, G.L.; Zhuang, M.; Gao, H.; Lu, L.; Han, X.; Chu, Z.; et al. Thermal runaway of lithium-ion batteries without internal short circuit. Joule 2018, 2, 2047–2064. [Google Scholar] [CrossRef]
- Hwang, S.; Chang, W.; Kim, S.M.; Su, D.; Kim, D.H.; Lee, J.Y.; Chung, K.Y.; Stach, E.A. Investigation of changes in the surface structure of LixNi0.8Co0.15Al0.05O2 cathode materials induced by the initial charge. Chem. Mater. 2014, 26, 1084–1092. [Google Scholar] [CrossRef]
- Cai, L.; Liu, Z.; An, K.; Liang, C. Probing Li-Ni Cation Disorder in Li1−xNi1+x−yAlyO2 Cathode Materials by Neutron Diffraction. J. Electrochem. Soc. 2012, 159, A924–A928. [Google Scholar] [CrossRef]
- Lee, M.J.; Noh, M.; Park, M.H.; Jo, M.; Kim, H.; Nam, H.; Cho, J. The role of nanoscale-range vanadium treatment in LiNi0.8Co0.15Al0.05O2 cathode materials for Li-ion batteries at elevated temperatures. J. Mater. Chem. A 2015, 3, 13453–13460. [Google Scholar] [CrossRef]
- Muto, S.; Tatsumi, K.; Kojima, Y.; Oka, H.; Kondo, H.; Horibuchi, K.; Ukyo, Y. Effect of Mg-doping on the degradation of LiNiO2-based cathode materials by combined spectroscopic methods. J. Power Sources 2012, 205, 449–455. [Google Scholar] [CrossRef]
- Huang, B.; Li, X.; Wang, Z.; Guo, H.; Xiong, X. Synthesis of Mg-doped LiNi0.8Co0.15Al0.05O2 oxide and its electrochemical behavior in high-voltage lithium-ion batteries. Ceram. Int. 2014, 40, 13223–13230. [Google Scholar] [CrossRef]
- Wang, Q.; Mao, B.; Stoliarov, S.I. A review of lithium ion battery failure mechanisms and fire prevention strategies. Prog. Energy Combust. Sci. 2019, 73, 95–131. [Google Scholar] [CrossRef]
- Kong, L.; Li, C.; Jiang, J.; Pecht, M.G. Li-ion battery fire hazards and safety strategies. Energies 2018, 11, 2191. [Google Scholar] [CrossRef]
- Mier, F.A.; Hargather, M.J.; Ferreira, S.R. Experimental Quantification of Vent Mechanism Flow Parameters in 18650 Format Lithium Ion Batteries; Sandia National Lab.(SNL-NM): Albuquerque, NM, USA, 2019.
- Balakrishnan, P.G.; Ramesh, R.; Kumar, T.P. Safety mechanisms in lithium-ion batteries. J. Power Sources 2006, 155, 401–414. [Google Scholar] [CrossRef]
- Meyer, J. Glass transition temperature as a guide to selection of polymers suitable for PTC materials. Polym. Eng. Sci. 1973, 13, 462–468. [Google Scholar] [CrossRef]
- Johnson, B.A.; White, R.E. Characterization of commercially available lithium-ion batteries. J. Power Sources 1998, 70, 48–54. [Google Scholar] [CrossRef]
- Huo, W.; Qu, Y. Effects of Bi1/2Na1/2TiO3 on the Curie temperature and the PTC effects of BaTiO3-based positive temperature coefficient ceramics. Sens. Actuators A Phys. 2006, 128, 265–269. [Google Scholar] [CrossRef]
- Ding, S.W.; Jia, G.; Wang, J.; He, Z.Y. Electrical properties of Y-and Mn-doped BaTiO3-based PTC ceramics. Ceram. Int. 2008, 34, 2007–2010. [Google Scholar] [CrossRef]
- Zhao, S.; Li, G.; Liu, H.; Dai, K.; Zheng, G.; Yan, X.; Liu, C.; Chen, J.; Shen, C.; Guo, Z. Positive Temperature Coefficient (PTC) Evolution of Segregated Structural Conductive Polypropylene Nanocomposites with Visually Traceable Carbon Black Conductive Network. Adv. Mater. Interfaces 2017, 4, 1700265. [Google Scholar] [CrossRef]
- Zhao, S.; Lou, D.; Zhan, P.; Li, G.; Dai, K.; Guo, J.; Zheng, G.; Liu, C.; Shen, C.; Guo, Z. Heating-induced negative temperature coefficient effect in conductive graphene/polymer ternary nanocomposites with a segregated and double-percolated structure. J. Mater. Chem. C 2017, 5, 8233–8242. [Google Scholar] [CrossRef]
- Dunn, B.; Kamath, H.; Tarascon, J.M. Electrical energy storage for the grid: A battery of choices. Science 2011, 334, 928–935. [Google Scholar] [CrossRef]
- Mönnighoff, X.; Murmann, P.; Weber, W.; Winter, M.; Nowak, S. Post-mortem investigations of fluorinated flame retardants for lithium ion battery electrolytes by gas chromatography with chemical ionization. Electrochim. Acta 2017, 246, 1042–1051. [Google Scholar] [CrossRef]
- Wang, Q.; Jiang, L.; Yu, Y.; Sun, J. Progress of enhancing the safety of lithium ion battery from the electrolyte aspect. Nano Energy 2019, 55, 93–114. [Google Scholar] [CrossRef]
- Murmann, P.; Mönnighoff, X.; von Aspern, N.; Janssen, P.; Kalinovich, N.; Shevchuk, M.; Kazakova, O.; RöschenthaleR, G.V.; Cekic-Laskovic, I.; Winter, M. Influence of the fluorination degree of organophosphates on flammability and electrochemical performance in lithium ion batteries: Studies on fluorinated compounds deriving from triethyl phosphate. J. Electrochem. Soc. 2016, 163, A751–A757. [Google Scholar] [CrossRef]
- Xu, K.; Ding, M.S.; Zhang, S.; Allen, J.L.; Jow, T.R. An attempt to formulate nonflammable lithium ion electrolytes with alkyl phosphates and phosphazenes. J. Electrochem. Soc. 2002, 149, A622–A626. [Google Scholar] [CrossRef]
- Laoutid, F.; Bonnaud, L.; Alexandre, M.; Lopez-Cuesta, J.M.; Dubois, P. New prospects in flame retardant polymer materials: From fundamentals to nanocomposites. Mater. Sci. Eng. R Rep. 2009, 63, 100–125. [Google Scholar] [CrossRef]
- Xiang, H.F.; Xu, H.Y.; Wang, Z.Z.; Chen, C. Dimethyl methylphosphonate (DMMP) as an efficient flame retardant additive for the lithium-ion battery electrolytes. J. Power Sources 2007, 173, 562–564. [Google Scholar] [CrossRef]
- Hu, J.; Jin, Z.; Zhong, H.; Zhan, H.; Zhou, Y.; Li, Z. A new phosphonamidate as flame retardant additive in electrolytes for lithium ion batteries. J. Power Sources 2012, 197, 297–300. [Google Scholar] [CrossRef]
- Ding, M.S.; Xu, K.; Jow, T.R. Effects of tris (2, 2, 2-trifluoroethyl) phosphate as a flame-retarding cosolvent on physicochemical properties of electrolytes of LiPF6 in EC-PC-EMC of 3, 3, 4 weight ratios. J. Electrochem. Soc. 2002, 149, A1489–A1498. [Google Scholar] [CrossRef]
- Zeng, Z.; Jiang, X.; Wu, B.; Xiao, L.; Ai, X.; Yang, H.; Cao, Y. Bis (2, 2, 2-trifluoroethyl) methylphosphonate: An novel flame-retardant additive for safe lithium-ion battery. Electrochim. Acta 2014, 129, 300–304. [Google Scholar] [CrossRef]
- Moreno, M.; Simonetti, E.; Appetecchi, G.B.; Carewska, M.; Montaninoc, M.; Kim, G.T.; Loeffler, N.; Passerini, S. Ionic Liquid Electrolytes for Safer Lithium Batteries I. Investigation around Optimal Formulation. J. Electrochem. Soc. 2017, 164, A6026–A6031. [Google Scholar] [CrossRef]
- Chawla, N.; Bharti, N.; Singh, S. Recent advances in non-flammable electrolytes for safer lithium-ion batteries. Batteries 2019, 5, 19. [Google Scholar] [CrossRef]
- Shi, P.; Zheng, H.; Liang, X.; Sun, Y.; Cheng, S.; Chen, C.; Xiang, H. A highly concentrated phosphate-based electrolyte for high-safety rechargeable lithium batteries. Chem. Commun. 2018, 54, 4453–4456. [Google Scholar] [CrossRef] [PubMed]
- Safa, M.; Chamaani, A.; Chawla, N.; El-Zahab, B. Polymeric ionic liquid gel electrolyte for room temperature lithium battery applications. Electrochim. Acta 2016, 213, 587–593. [Google Scholar] [CrossRef]
- Guo, Q.; Han, Y.; Wang, H.; Xiong, S.; Sun, W.; Zheng, C.; Xie, K. Flame Retardant and Stable Li1.5Al0.5Ge1.5(PO4)3-Supported Ionic Liquid Gel Polymer Electrolytes for High Safety Rechargeable Solid-State Lithium Metal Batteries. J. Phys. Chem. C 2018, 122, 10334–10342. [Google Scholar] [CrossRef]
- Rectenwald, M.F.; Gaffen, J.R.; Rheingold, A.L.; Morgan, A.B.; Protasiewicz, J.D. Phosphoryl-Rich Flame-Retardant Ions (FRIONs): Towards Safer Lithium-Ion Batteries. Angew. Chem. 2014, 126, 4257–4260. [Google Scholar] [CrossRef]
- Dagger, T.; Meier, V.; Hildebrand, S.; Brüggemann, D.; Winter, M.; Schappacher, F.M. Safety Performance of 5 Ah Lithium Ion Battery Cells Containing the Flame Retardant Electrolyte Additive (Phenoxy) Pentafluorocyclotriphosphazene. Energy Technol. 2018, 6, 2001–2010. [Google Scholar] [CrossRef]
- Wu, B.; Pei, F.; Wu, Y.; Mao, R.; Ai, X.; Yang, H.; Cao, Y. An electrochemically compatible and flame-retardant electrolyte additive for safe lithium ion batteries. J. Power Sources 2013, 227, 106–110. [Google Scholar] [CrossRef]
- Li, X.; Li, W.; Chen, L.; Lu, Y.; Su, Y.; Bao, L.; Wang, J.; Chen, R.; Chen, S.; Wu, F. Ethoxy (pentafluoro) cyclotriphosphazene (PFPN) as a multi-functional flame retardant electrolyte additive for lithium-ion batteries. J. Power Sources 2018, 378, 707–716. [Google Scholar] [CrossRef]
- Wu, H.; Zhuo, D.; Kong, D. Improving battery safety by early detection of internal shorting with a bifunctional separator. Nat. Commun. 2014, 5, 5193. [Google Scholar] [CrossRef]
- Ryou, M.H.; Lee, D.J.; Lee, J.N.; Lee, J.K.; Park, J.K.; Choi, J.W. Excellent cycle life of lithium-metal anodes in lithium-ion batteries with mussel-inspired polydopamine-coated separators. Adv. Energy Mater. 2012, 2, 645–650. [Google Scholar] [CrossRef]
- Kim, J.H.; Kim, J.H.; Choi, K.H.; Yu, H.K.; Kim, J.H.; Lee, J.S.; Lee, S.Y. Inverse opal-inspired, nanoscaffold battery separators: A new membrane opportunity for high-performance energy storage systems. Nano Lett. 2014, 14, 4438–4448. [Google Scholar] [CrossRef]
- Orendorff, C.J. The role of separators in lithium-ion cell safety. Electrochem. Soc. Interface 2012, 21, 61–65. [Google Scholar] [CrossRef]
- Xiang, Y.; Li, J.; Lei, J.; Liu, D.; Xie, Z.; Qu, D.; Li, K.; Deng, T.; Tang, H. Advanced separators for lithium-ion and lithium–sulfur batteries: A review of recent progress. ChemSusChem 2016, 9, 3023–3039. [Google Scholar] [CrossRef] [PubMed]
- Arora, P.; Zhang, Z. Battery separators. Chem. Rev. 2004, 104, 4419–4462. [Google Scholar] [CrossRef] [PubMed]
- Ryou, M.H.; Lee, J.N.; Lee, D.J.; Kim, W.K.; Jeong, Y.K.; Choi, J.W.; Park, J.K.; Lee, Y.M. Effects of lithium salts on thermal stabilities of lithium alkyl carbonates in SEI layer. Electrochim. Acta 2012, 83, 259–263. [Google Scholar] [CrossRef]
- Yeon, D.; Lee, Y.; Ryou, M.H.; Lee, Y.M. New flame-retardant composite separators based on metal hydroxides for lithium-ion batteries. Electrochim. Acta 2015, 157, 282–289. [Google Scholar] [CrossRef]
- Lee, T.; Kim, W.K.; Lee, Y.; Ryou, M.H.; Lee, Y.M. Effect of Al2O3 coatings prepared by RF sputtering on polyethylene separators for high-power lithium ion batteries. Macromol. Res. 2014, 22, 1190–1195. [Google Scholar] [CrossRef]
- Lee, J.Y.; Shin, S.H.; Moon, S.H. Flame retardant coated polyolefin separators for the safety of lithium ion batteries. Korean J. Chem. Eng. 2016, 33, 285–289. [Google Scholar] [CrossRef]
- Zhang, J.; Yue, L.; Kong, Q.; Liu, Z.; Zhou, X.; Zhang, C.; Xu, Q.; Zhang, B.; Ding, G.; Qin, B.; et al. Sustainable, heat-resistant and flame-retardant cellulose-based composite separator for high-performance lithium ion battery. Sci. Rep. 2014, 4, 3935. [Google Scholar] [CrossRef]
- Wu, W.; Wang, S.; Wu, W.; Chen, K.; Hong, S.; Lai, Y. A critical review of battery thermal performance and liquid based battery thermal management. Energy Convers. Manag. 2019, 182, 262–281. [Google Scholar] [CrossRef]
- Ramadass, P.; Haran, B.; White, R.; Popov, B. Capacity fade of Sony 18650 cells cycled at elevated temperatures: Part II. Capacity fade analysis. J. Power Sources 2002, 112, 614–620. [Google Scholar] [CrossRef]
- Smart, M.C.; Ratnakumar, B.V.; Whitcanack, L.D.; Chin, K.B.; Surampudi, S.; Croft, H.; Tice, D.; Staniewicz, R. Improved low-temperature performance of lithium-ion cells with quaternary carbonate-based electrolytes. J. Power Sources 2003, 119, 349–358. [Google Scholar] [CrossRef]
- Ouyang, M.; Chu, Z.; Lu, L.; Li, J.; Han, X.; Feng, X.; Liu, G. Low temperature aging mechanism identification and lithium deposition in a large format lithium iron phosphate battery for different charge profiles. J. Power Sources 2015, 286, 309–320. [Google Scholar] [CrossRef]
- Feng, X.; Xu, C.; He, X.; Wang, L.; Zhang, G.; Ouyang, M. Mechanisms for the evolution of cell variations within a LiNixCoyMnzO2/graphite lithium-ion battery pack caused by temperature non-uniformity. J. Clean. Prod. 2018, 205, 447–462. [Google Scholar] [CrossRef]
- Lu, L.; Han, X.; Li, J.; Hua, J.; Ouyang, M. A review on the key issues for lithium-ion battery management in electric vehicles. J. Power Sources 2013, 226, 272–288. [Google Scholar] [CrossRef]
- Choi, K.W.; Yao, N.P. Heat Transfer in Lead-Acid Batteries Designed for Electric-Vehicle Propulsion Application. J. Electrochem. Soc. 1979, 126, 1321–1328. [Google Scholar] [CrossRef]
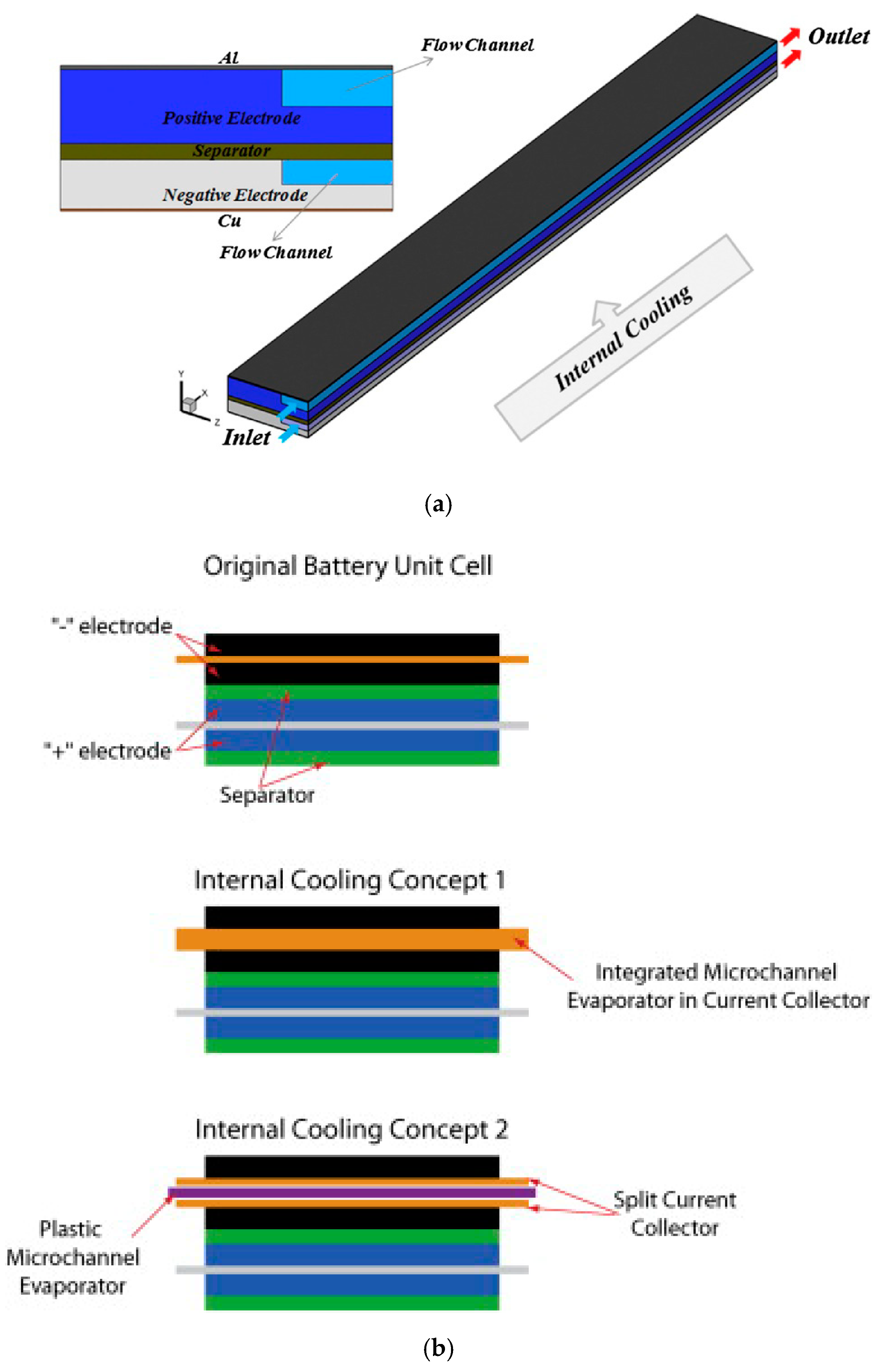
- Mohammadian, S.K.; He, Y.L.; Zhang, Y. Internal cooling of a lithium-ion battery using electrolyte as coolant through microchannels embedded inside the electrodes. J. Power Sources 2015, 293, 458–466. [Google Scholar] [CrossRef]
- Bandhauer, T.M.; Garimella, S. Passive, internal thermal management system for batteries using microscale liquid–vapor phase change. Appl. Therm. Eng. 2013, 61, 756–769. [Google Scholar] [CrossRef]
- Stuart, T.A.; Hande, A. HEV battery heating using AC currents. J. Power Sources 2004, 129, 368–378. [Google Scholar] [CrossRef]
- Ruan, H.; Jiang, J.; Sun, B.; Zhang, W.; Gao, W.; Wang, L.; Ma, Z. A rapid low-temperature internal heating strategy with optimal frequency based on constant polarization voltage for lithium-ion batteries. Appl. Energy 2016, 177, 771–782. [Google Scholar] [CrossRef]
- Wang, S.; Li, K.; Tian, Y.; Wang, J.; Wu, Y.; Ji, S. Improved thermal performance of a large laminated lithium-ion power battery by reciprocating air flow. Appl. Therm. Eng. 2019, 152, 445–454. [Google Scholar] [CrossRef]
- Liu, Y.; Ouyang, C.; Jiang, Q.; Liang, B. Design and parametric optimization of thermal management of lithium-ion battery module with reciprocating air-flow. J. Cent. South Univ. 2015, 22, 3970–3976. [Google Scholar] [CrossRef]
- Zhang, J.; Kang, H.; Wu, K.; Li, J.; Wang, Y. The impact of enclosure and boundary conditions with a wedge-shaped path and air cooling for battery thermal management in electric vehicles. Int. J. Energy Res. 2018, 42, 4054–4069. [Google Scholar] [CrossRef]
- Nelson, P.; Dees, D.; Amine, K.; Henriksen, G. Modeling thermal management of lithium-ion PNGV batteries. J. Power Sources 2002, 110, 349–356. [Google Scholar] [CrossRef]
- Shang, Z.; Qi, H.; Liu, X.; Ouyang, C.; Wang, Y. Structural optimization of lithium-ion battery for improving thermal performance based on a liquid cooling system. Int. J. Heat Mass Transf. 2019, 130, 33–41. [Google Scholar] [CrossRef]
- Zhao, C.; Sousa, A.C.M.; Jiang, F. Minimization of thermal non-uniformity in lithium-ion battery pack cooled by channeled liquid flow. Int. J. Heat Mass Transf. 2019, 129, 660–670. [Google Scholar] [CrossRef]
- Wang, C.; Zhang, G.; Li, X.; Huang, J.; Wang, Z.; Lv, Y.; Meng, L.; Situ, W.; Rao, M. Experimental examination of large capacity liFePO4 battery pack at high temperature and rapid discharge using novel liquid cooling strategy. Int. J. Energy Res. 2018, 42, 1172–1182. [Google Scholar] [CrossRef]
- Ren, Y.; Yu, Z.; Song, G. Thermal management of a Li-ion battery pack employing water evaporation. J. Power Sources 2017, 360, 166–171. [Google Scholar] [CrossRef]
- Ianniciello, L.; Biwolé, P.H.; Achard, P. Electric vehicles batteries thermal management systems employing phase change materials. J. Power Sources 2018, 378, 383–403. [Google Scholar] [CrossRef]
- Fan, Y.F.; Zhang, X.X.; Wang, X.C.; Li, J.; Zhu, Q.B. Super-cooling prevention of microencapsulated phase change material. Thermochim. Acta 2004, 413, 1–6. [Google Scholar] [CrossRef]
- Wang, W.; Zhang, X.; Xin, C.; Rao, Z. An experimental study on thermal management of lithium ion battery packs using an improved passive method. Appl. Therm. Eng. 2018, 134, 163–170. [Google Scholar] [CrossRef]
- Huang, P.; Verma, A.; Robles, D.J.; Wang, Q.; Mukherjee, P.; Sun, J. Probing the cooling effectiveness of phase change materials on lithium-ion battery thermal response under overcharge condition. Appl. Therm. Eng. 2018, 132, 521–530. [Google Scholar] [CrossRef]
- Ling, Z.; Wen, X.; Zhang, Z.; Fang, X.; Gao, X. Thermal management performance of phase change materials with different thermal conductivities for Li-ion battery packs operated at low temperatures. Energy 2018, 144, 977–983. [Google Scholar] [CrossRef]
- Zhang, S.; Wu, W.; Wang, S. Experimental investigations of Alum/expanded graphite composite phase change material for thermal energy storage and its compatibility with metals. Energy 2018, 161, 508–516. [Google Scholar] [CrossRef]
- Wang, W.; Yang, X.; Fang, Y.; Ding, J.; Yan, J. Preparation and thermal properties of polyethylene glycol/expanded graphite blends for energy storage. Appl. Energy 2009, 86, 1479–1483. [Google Scholar] [CrossRef]
- Ruiz, J.; Ganatra, Y.; Bruce, A.; Howarter, J.; Marconnet, A.M. Investigation of aluminum foams and graphite fillers for improving the thermal conductivity of paraffin wax-based phase change materials. In Proceedings of the 2017 16th IEEE Intersociety Conference on Thermal and Thermomechanical Phenomena in Electronic Systems (ITherm), Orlando, FL, USA, 30 May–2 June 2017; pp. 384–389. [Google Scholar]
- Kim, D.; Jung, J.; Kim, Y.; Lee, M.; Seo, J.; Khan, S.B. Structure and thermal properties of octadecane/expanded graphite composites as shape-stabilized phase change materials. Int. J. Heat Mass Transf. 2016, 95, 735–741. [Google Scholar] [CrossRef]
- Sheng, N.; Dong, K.; Zhu, C.; Akiyama, T.; Nomura, T. Thermal conductivity enhancement of erythritol phase change material with percolated aluminum filler. Mater. Chem. Phys. 2019, 229, 87–91. [Google Scholar] [CrossRef]
- Zhang, L.; Zhou, K.; Wei, Q.; Ma, L.; Ye, W.; Li, H.; Zhou, B.; Yu, Z.; Lin, C.; Luo, J.; et al. Thermal conductivity enhancement of phase change materials with 3D porous diamond foam for thermal energy storage. Appl. Energy 2019, 233, 208–219. [Google Scholar] [CrossRef]
- Darkwa, J.; Zhou, T. Enhanced laminated composite phase change material for energy storage. Energy Convers. Manag. 2011, 52, 810–815. [Google Scholar] [CrossRef]
- Hasse, C.; Grenet, M.; Bontemps, A.; Dendievel, R.; Sallée, H. Realization, test and modelling of honeycomb wallboards containing a Phase Change Material. Energy Build. 2011, 43, 232–238. [Google Scholar] [CrossRef]
- Yang, Y.; Pang, Y.; Liu, Y.; Guo, H. Preparation and thermal properties of polyethylene glycol/expanded graphite as novel form-stable phase change material for indoor energy saving. Mater. Lett. 2018, 216, 220–223. [Google Scholar] [CrossRef]
- Ling, Z.; Wang, F.; Fang, X.; Gao, X.; Zhang, Z. A hybrid thermal management system for lithium ion batteries combining phase change materials with forced-air cooling. Appl. Energy 2015, 148, 403–409. [Google Scholar] [CrossRef]
- Fathabadi, H. High thermal performance lithium-ion battery pack including hybrid active–passive thermal management system for using in hybrid/electric vehicles. Energy 2014, 70, 529–538. [Google Scholar] [CrossRef]
- Xie, Y.; Tang, J.; Shi, S.; Xing, Y.; Wu, H.; Hu, Z.; Wen, D. Experimental and numerical investigation on integrated thermal management for lithium-ion battery pack with composite phase change materials. Energy Convers. Manag. 2017, 154, 562–575. [Google Scholar] [CrossRef]
- Song, L.; Zhang, H.; Yang, C. Thermal analysis of conjugated cooling configurations using phase change material and liquid cooling techniques for a battery module. Int. J. Heat Mass Transf. 2019, 133, 827–841. [Google Scholar] [CrossRef]
- Ling, Z.; Cao, J.; Zhang, W.; Fang, X.; Gao, X. Compact liquid cooling strategy with phase change materials for Li-ion batteries optimized using response surface methodology. Appl. Energy 2018, 228, 777–788. [Google Scholar] [CrossRef]
- Zheng, Y.; Shi, Y.; Huang, Y. Optimisation with adiabatic interlayers for liquid-dominated cooling system on fast charging battery packs. Appl. Therm. Eng. 2019, 147, 636–646. [Google Scholar] [CrossRef]
- Xie, J.; Chen, J. Battery Early Warning and Monitoring System. U.S. Patent 8,952,823, 10 February 2015. [Google Scholar]
- Hermann, W.A. Method for Detecting Battery Thermal Events Via Battery Pack Isolation Monitoring. U.S. Patent 8,168,315, 1 May 2012. [Google Scholar]
- Hill, D.; Gully, B.; Agarwal, A.; Nourai, A.; Thrun, L.; Swartz, S.; Koslowske, M.; Cumming, S.; Butkowski, J.; Moore, B. Detection of off gassing from Li-ion batteries. In Proceedings of the 2013 IEEE Energytech, Cleveland, OH, USA, 21–23 May 2013; pp. 1–7. [Google Scholar]
- Somov, A.; Spirjakin, D.; Ivanov, M.; Khromushin, I.; Passerone, R.; Baranov, A.; Savkin, A. Combustible gases and early fire detection: An autonomous system for wireless sensor networks. In Proceedings of the 1st International Conference on Energy-Efficient Computing and Networking; ACM: New York, NY, USA, 2010; pp. 85–93. [Google Scholar]
- Lecocq, A.; Eshetu, G.G.; Grugeon, S.; Martin, N.; Laruelle, S.; Marlair, G. Scenario-based prediction of Li-ion batteries fire-induced toxicity. J. Power Sources 2016, 316, 197–206. [Google Scholar] [CrossRef]
- Larsson, F.; Andersson, P.; Blomqvist, P.; Mellander, B.E. Toxic fluoride gas emissions from lithium-ion battery fires. Sci. Rep. 2017, 7, 10018. [Google Scholar] [CrossRef]
- Andersson, P.; Blomqvist, P.; Lorén, A.; Larsson, F. Using Fourier transform infrared spectroscopy to determine toxic gases in fires with lithium-ion batteries. Fire Mater. 2016, 40, 999–1015. [Google Scholar] [CrossRef]
- Keane, R.E.; Cary, G.J.; Davies, I.D.; Flannigan, M.D.; Gardner, R.H.; Lavorel, S.; Lenihan, J.M.; Li, C.; Rupp, T.S. A classification of landscape fire succession models: Spatial simulations of fire and vegetation dynamics. Ecol. Model. 2004, 179, 3–27. [Google Scholar] [CrossRef]
- Summer, S.M. Flammability Assessment of Lithium-Ion and Lithium-Ion Polymer Battery Cells Designed for Aircraft Power Usage, US Department of Transportation; Federal Aviation Administration: Washington, DC, USA, 2010.
- Ditch, B.; de Vries, J. Flammability Characterization of Lithium-Ion Batteries in Bulk Storage; FM Global: Johnston, RI, USA, 2013. [Google Scholar]
- Egelhaaf, M.; Kress, D.; Wolpert, D.; Lange, T.; Justen, R.; Wilstermann, H. Fire Fighting of Li-Ion Traction Batteries. Sae Int. J. Altern. Powertrains 2013, 2, 37–48. [Google Scholar] [CrossRef]
- Long, R.T.; Blum, A.F.; Bress, T.J.; Cotts, B.R. Best Practices for Emergency Response to Incidents Involving Electric Vehicles Battery Hazards: A Report on Full-Scale Testing Results; National Fire Protection Research Foundation: Quincy, MA, USA, 2013. [Google Scholar]
- Maloney, T. Extinguishment of Lithium-Ion and Lithium-Metal Battery Fires, US Department of Transportation; Federal Aviation Administration: Washington, DC, USA, 2014; pp. 46–51.
- Edison, C. Considerations for ESS Fire Safety; DNV GL: Oslo, Norway, 2017. [Google Scholar]
- Rao, H.; Huang, Z.; Zhang, H.; Xiao, S. Study of fire tests and fire safety measures on lithiumion battery used on ships. In Proceedings of the 2015 International Conference on Transportation Information and Safety (ICTIS), Wuhan, China, 25–28 June 2015; pp. 865–870. [Google Scholar]
- Wang, Q.; Shao, G.; Duan, Q.; Chen, M.; Li, Y.; Wu, K.; Liu, B.; Peng, P.; Sun, J. The efficiency of heptafluoropropane fire extinguishing agent on suppressing the lithium titanate battery fire. Fire Technol. 2016, 52, 387–396. [Google Scholar] [CrossRef]
- Luo, W.; Zhu, S.; Gong, J.; Zhou, Z. Research and Development of Fire Extinguishing Technology for Power Lithium Batteries. Procedia Eng. 2018, 211, 531–537. [Google Scholar] [CrossRef]


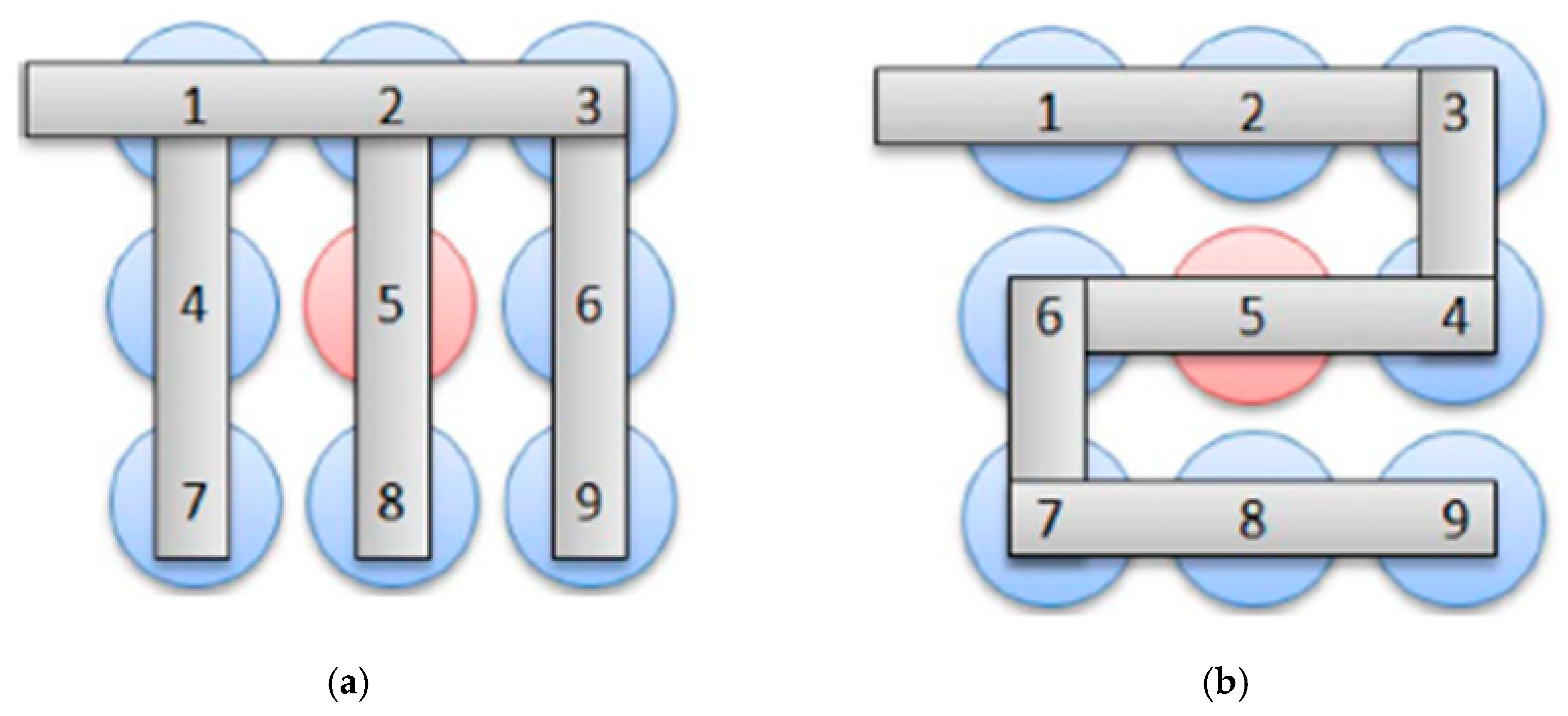

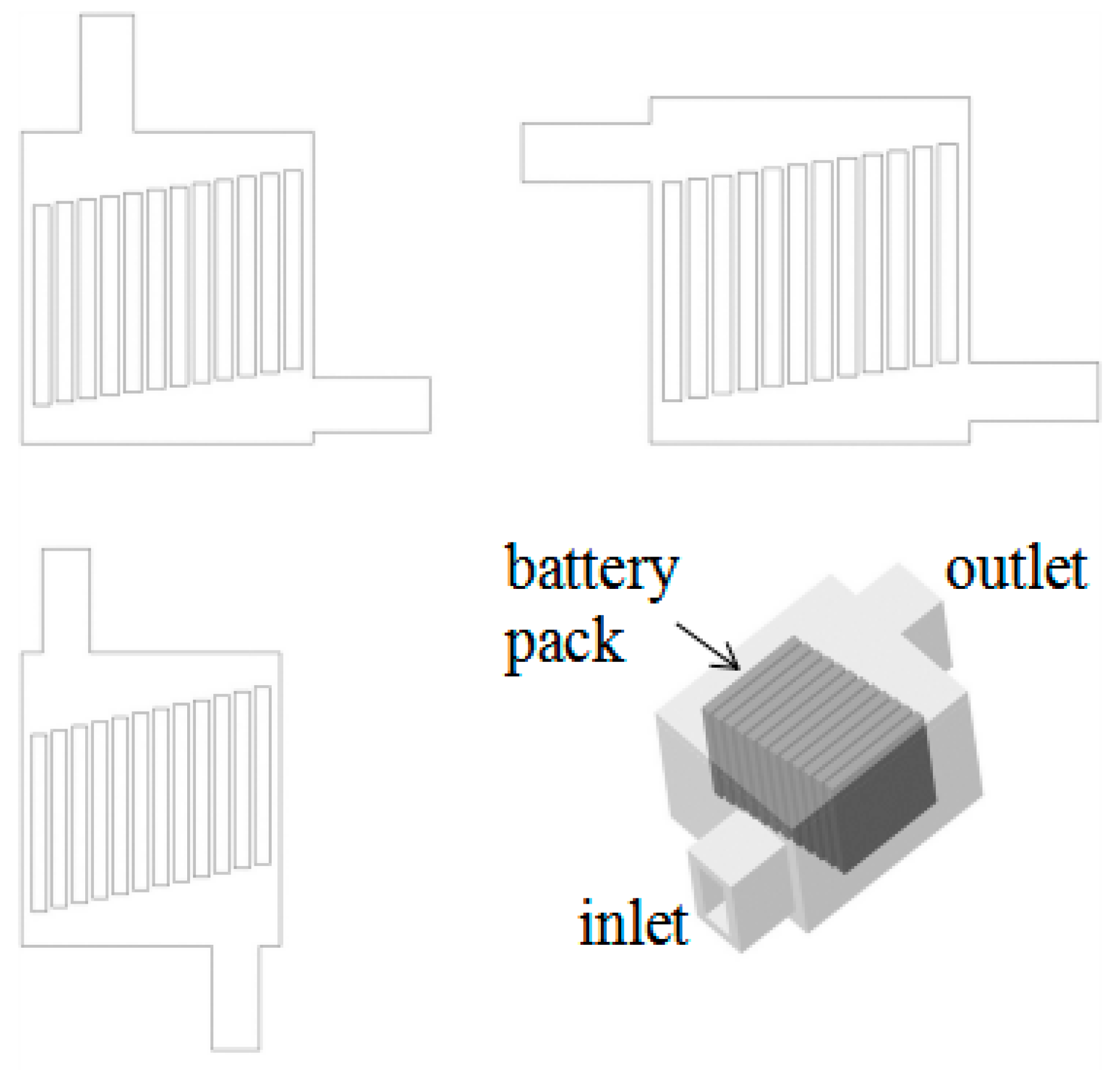
| Classification | No. | Date | Location | Accident Reply |
|---|---|---|---|---|
| Mobile telephone | 1 | 2016.8.24 | Korea | The first explosion of a Note 7 in the world [46] |
| 2 | 2016.9.18 | China | The first explosion of a Note 7 in China [46] | |
| 3 | 2016.10.10 | China | An iPhone 7 exploded and hurt the user [47] | |
| 4 | 2016.10.14 | China | A Huawei P9 exploded during charging [48] | |
| 5 | 2016.10.17 | Australia | An iPhone 7 caught fire which then, burned a car [49] | |
| 6 | 2018.1.9 | Switzerland | An iPhone exploded when replacing the battery, which caused an injury and seven poisonings [50] | |
| 7 | 2018.1.10 | Spain | An iPhone exploded which caused thick smoke inside the store [50] | |
| 8 | 2018.12.30 | America | An iPhone XS Max self-ignited and burned the user [51] | |
| EV | 1 | 2016.1.1 | Norway | A Tesla Model S caught fire during charging [52] |
| 2 | 2016.5.14 | China | An EV bus caught fire because of battery pack short-circuit [52] | |
| 3 | 2016.9.7 | Netherlands | A Tesla Model S crashed to a tree which caused the battery to catch fire, resulting in the death of the driver [52] | |
| 4 | 2017.1.15 | China | An EV bus self-ignited during driving [53] | |
| 5 | 2017.2.19 | China | A Tesla Model X caught fire after crashing [53] | |
| 6 | 2017.5.1 | China | An EV bus self-ignited during charging [53] | |
| 7 | 2018.3.24 | America | A Tesla Model S caught fire whilst stationary [54] | |
| 8 | 2018.5.21 | China | An EV bus self-ignited during driving [54] | |
| Airplane | 1 | 2010.9.3 | The United Arab Emirates | A Boeing 787 crashed due to the battery catching fire, which caused two deaths [55] |
| 2 | 2013.1.7 | America | The battery pack caught fire and filled the cabin of a Boeing 787 with smoke [56] | |
| 3 | 2013.1.16 | Japan | The battery pack caught fire during a Boeing 787 flight from Yamaguchi-Ube to Tokyo [56] | |
| 4 | 2014.4 | Australia | A Boeing 737 caught fire due to the short-circuit of the battery inside a trunk [57] |
| Type | Advantages | Disadvantages |
|---|---|---|
| Phosphorus | Low cost; Simple synthesis process | Poor electrochemical compatibility; Low retardant efficiency |
| Fluoride | Good electrochemical compatibility; Simple synthesis process | Low retardant efficiency; High cost |
| Ionic liquid | Appropriate to low-temperature environment | Complicated synthesis process; Low thermal stability; Low retardant efficiency |
| Composite | High retardant efficiency; Good electrochemical compatibility | Complicated synthesis process; High cost |
| Authors/Reference | CPCM/Proportions | Melting Temperature (°C) | Latent Heat (kJ/kg) | Thermal Conductivity (W/m·K) |
|---|---|---|---|---|
| Zhang et al. [254] | Alum/EG | 87.9 | 214.9 | 0.95–5.88 |
| Wang et al. [255] | Polyethylene glycol (PEG)/EG | 61.5 | 161.2 | 0.30–1.32 |
| Javieradrain et al. [256] | Alum foam/EG/parraffin | 60.1 | 190.0 | 0.20–1.20 |
| Kim et al. [257] | Octadecane (OD)/Expanded graphite (WEPG) | 30.1 | 198.8 | / |
| Sheng et al. [258] | Alum/Erythritol | 118.0 | / | 4.00–30.00 |
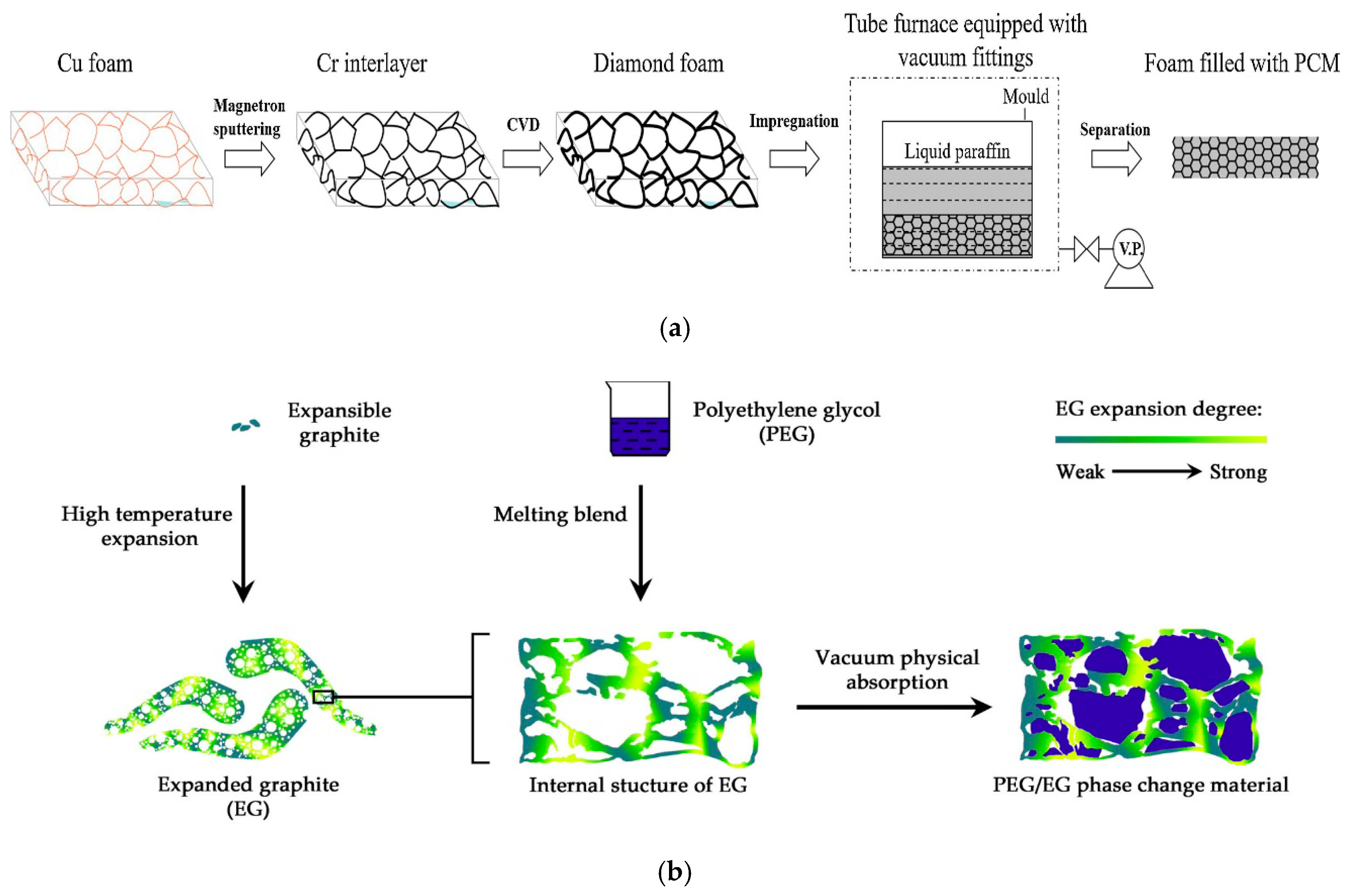
| Zhang et al. [259] (Figure 22a) | Diamond foam (DF)/parraffin | 90.0 | 124.7 | 0.25–6.70 |
| Darkwa et al. [260] | Alum/Hexadecane | 24.9 | 167.0 | 0.15–1.25 |
| Hasse et al. [261] | Parraffin | 27.9 | 170.1 | 0.19 |
| Yang et al. [262] (Figure 22b) | PEG/EG | 25.9 | 98.6 | / |
| BMS | Advantages | Disadvantages | |
|---|---|---|---|
| ACS | NC | Low cost; Simple structure; Easy to integrate; Little electricity consumption | Low heat transfer coefficient; Dependent on ambient temperature; Uneven temperature distribution |
| FC | Low cost; Easy to maintain | Low efficiency; Dependent on ambient environment; Insufficient for extreme condition | |
| LCS | Liquid cooling | Low cost; Easy to maintain | Risk of leakage |
| Vapor cooling | Higher efficiency; Low operate cost; Better uniformity | Higher cost for structure design; High cost for circulation | |
| PCM-CS | Organic | High efficiency; Uniform temperature distribution; Appropriate to extreme conditions | Risk of leakage; Volume difference with phase change; Risk of supercooling |
| Inorganic | |||
| Eutectic | |||
| Class | Description |
|---|---|
| A | Fires in ordinary combustible materials such as wood, cloth, paper, rubber and many plastics. |
| B | Fires in flammable liquids, combustible liquids, petroleum greases, tars, oils, oil-based paints, solvents, lacquers, alcohols and flammable gases. |
| C | Fires that involve energized electrical equipment. |
| D | Fires in combustible metals such as magnesium, titanium, zirconium, sodium, lithium and potassium. |
| K | Fires in cooking appliances that involve combustible cooking media. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ouyang, D.; Chen, M.; Huang, Q.; Weng, J.; Wang, Z.; Wang, J. A Review on the Thermal Hazards of the Lithium-Ion Battery and the Corresponding Countermeasures. Appl. Sci. 2019, 9, 2483. https://doi.org/10.3390/app9122483
Ouyang D, Chen M, Huang Q, Weng J, Wang Z, Wang J. A Review on the Thermal Hazards of the Lithium-Ion Battery and the Corresponding Countermeasures. Applied Sciences. 2019; 9(12):2483. https://doi.org/10.3390/app9122483
Chicago/Turabian StyleOuyang, Dongxu, Mingyi Chen, Que Huang, Jingwen Weng, Zhi Wang, and Jian Wang. 2019. "A Review on the Thermal Hazards of the Lithium-Ion Battery and the Corresponding Countermeasures" Applied Sciences 9, no. 12: 2483. https://doi.org/10.3390/app9122483
APA StyleOuyang, D., Chen, M., Huang, Q., Weng, J., Wang, Z., & Wang, J. (2019). A Review on the Thermal Hazards of the Lithium-Ion Battery and the Corresponding Countermeasures. Applied Sciences, 9(12), 2483. https://doi.org/10.3390/app9122483








