Electrokinetic Characterization of Natural Stones Coated with Nanocomposites for the Protection of Cultural Heritage
Abstract
Featured Application
Abstract
1. Introduction
2. Materials and Methods
2.1 Lithotypes
2.2. Nanocomposite Products
2.3. Treatment Procedure
2.4. Testing of Stone Specimens
2.4.1. Streaming Current Measurements
2.4.2. Water Absorption Measurements
2.4.3. Static Contact Angle Measurements
3. Results and Discussion
3.1. Electrokinetic Analysis of Untreated Lithotypes
3.2. Electrokinetic Analysis of Nanocomposites
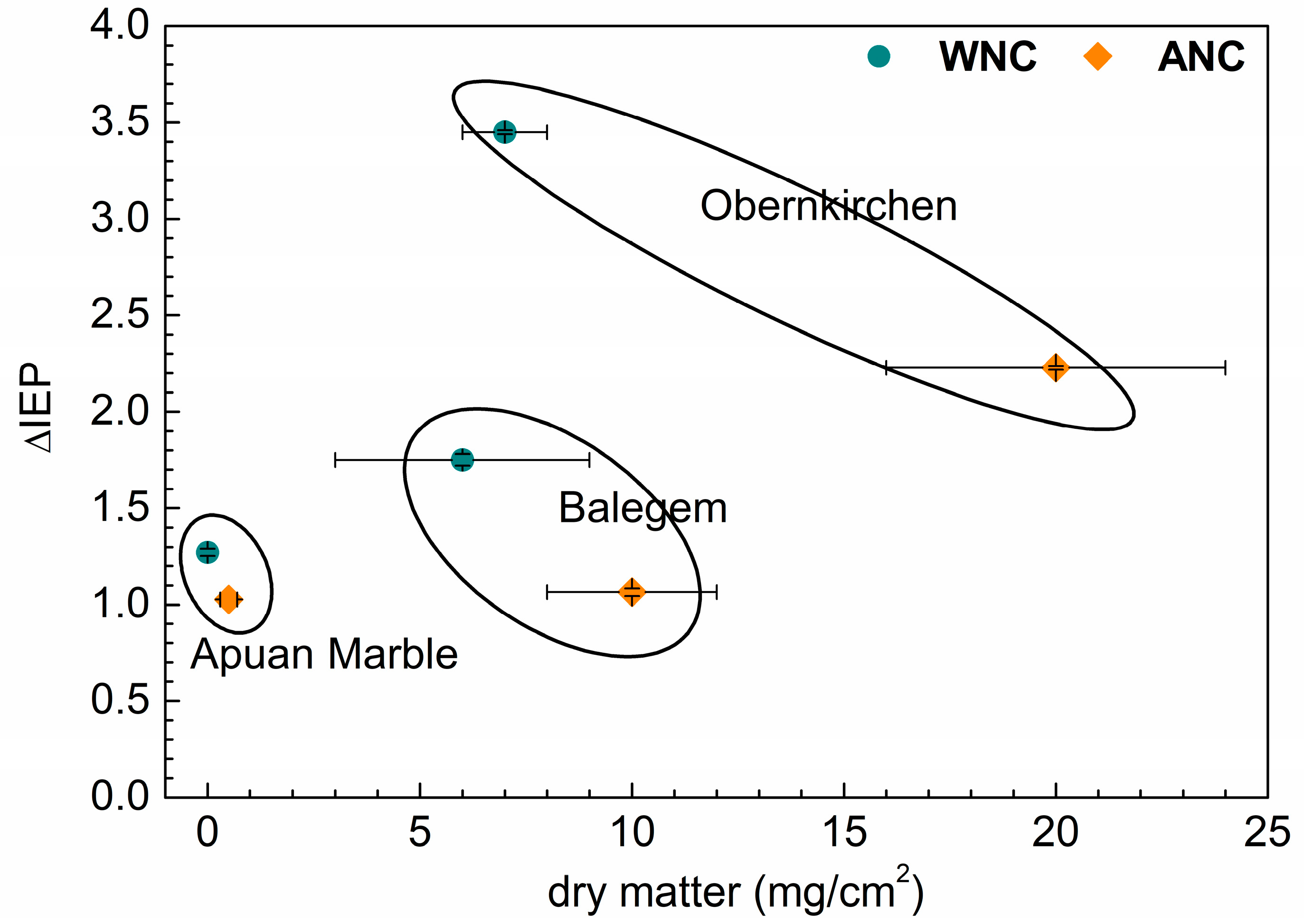
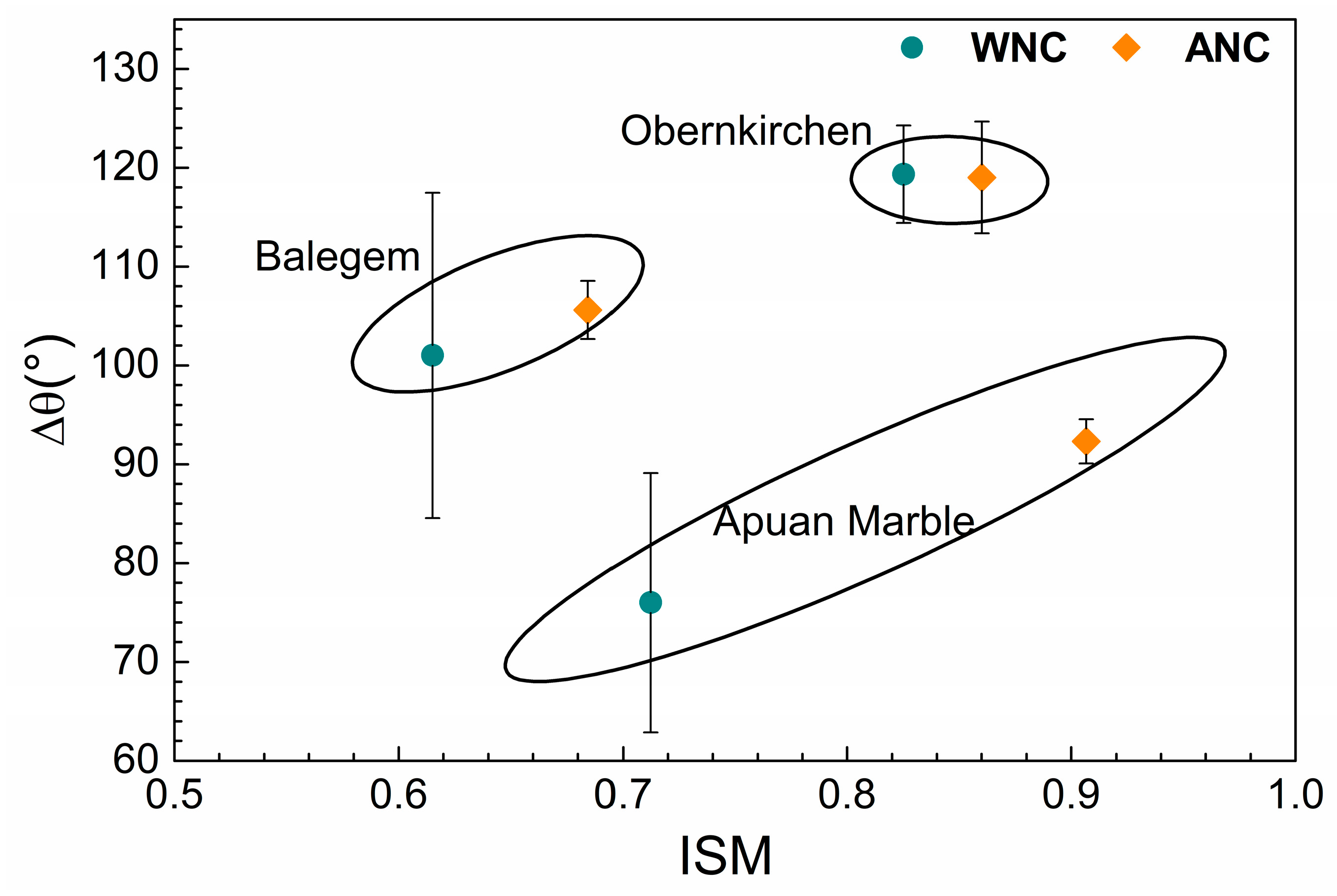
3.3. Electrokinetic Analysis of Treated Lithotypes
4. Conclusions
- electrokinetic analysis based on streaming current measurements was shown to be a sensitive and effective tool to characterize the modification of the surface properties of different natural stones upon application of coating materials;
- a correlation was found between the extent of surface modification (according to the empirical ISM parameter) and the effectiveness of the products as determined by the reduction of water absorption by capillarity and of surface wettability for the six investigated lithotype-nanocomposite coating combinations;
- significant differences were observed between the low porosity Apuan marble and the other two more porous lithotypes, as the former prevents to a large extent penetration of more viscous fluids and possibly of nanoparticles, while the difference between products is, to a good extent, levelled out in the case of the most porous lithotypes as a result of product absorption into the stone matrix.
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Snethlage, R. Stone conservation. In Stone in Architecture: Properties, Durability, 5th ed.; Siegesmund, S., Snethlage, R., Eds.; Springer: Berlin/Heidelberg, Germany, 2014; pp. 415–550. ISBN 978-3-662-49573-5. [Google Scholar]
- Della Volpe, C.; Penati, A.; Peruzzi, R.; Siboni, S.; Toniolo, L.; Colombo, C. Combined effect of roughness and heterogeneity on contact angles: The case of polymer coating for stone protection. J. Adhes. Sci. Technol. 2000, 14, 273–299. [Google Scholar] [CrossRef]
- De Ferri, L.; Lottici, P.P.; Lorenzi, A.; Montenero, A.; Salvioli-Mariani, E. Study of silica nanoparticles—Polysiloxane hydrophobic treatments for stone-based monument protection. J. Cult. Herit. 2011, 12, 356–363. [Google Scholar] [CrossRef]
- Fermo, P.; Cappelletti, G.; Cozzi, N.; Padeletti, G.; Kaciulis, S.; Brucale, M.; Merlini, M. Hydrophobizing coatings for cultural heritage. A detailed study of resin/stone surface interaction. Appl. Phys. A 2014, 116, 341–348. [Google Scholar] [CrossRef]
- Crupi, V.; Fazio, B.; Gessini, A.; Kis, Z.; La Russa, M.F.; Majolino, D.; Masciovecchio, C.; Ricca, M.; Rossi, B.; Ruffolo, S.A.; et al. TiO2–SiO2–PDMS nanocomposite coating with self-cleaning effect for stone material: Finding the optimal amount of TiO2. Constr. Build. Mater. 2018, 166, 464–471. [Google Scholar] [CrossRef]
- Lettieri, M.; Masieri, M. Surface characterization and effectiveness evaluation of anti-graffiti coatings on highly porous stone materials. Appl. Surf. Sci. 2014, 288, 466–477. [Google Scholar] [CrossRef]
- La Russa, M.F.; Rovella, N.; De Buergo, M.A.; Belfiore, C.M.; Pezzino, A.; Crisci, G.M.; Ruffolo, S.A. Nano-TiO2 coatings for cultural heritage protection: The role of the binder on hydrophobic and self-cleaning efficacy. Prog. Org. Coat. 2016, 91, 1–8. [Google Scholar] [CrossRef]
- Xiong, Q.; Baychev, T.G.; Jivkov, A.P. Review of pore network modelling of porous media: Experimental characterisations, network constructions and applications to reactive transport. J. Contam. Hydrol. 2016, 192, 101–117. [Google Scholar] [CrossRef] [PubMed]
- Doehne, E.; Price, C.A. Stone Conservation: An Overview of Current Research, 2nd ed.; The Getty Conservation Institute: Los Angeles, CA, USA, 2010; ISBN 978-1-60606-046-9. [Google Scholar]
- Andreotti, S.; Franzoni, E.; Fabbri, P. Poly(hydroxyalkanoate)s-based hydrophobic coatings for the protection of stone in cultural heritage. Materials 2018, 11, 165. [Google Scholar] [CrossRef] [PubMed]
- Luxbacher, T. The Zeta Guide: Principles of the Streaming Potential Technique; Anton Paar GmbH: Graz, Austria, 2014. [Google Scholar]
- Von Smoluchowski, M. Contribution à la théorie de l’endosmose électrique et de quelques phénomènes corrélatifs. Bull. Acad. Sci. Cracovie. 1903, 8, 182–200. [Google Scholar]
- Ristić, T.; Hribernik, S.; Fras-Zemljič, L. Electrokinetic properties of fibres functionalised by chitosan and chitosan nanoparticles. Cellulose 2015, 22, 3811–3823. [Google Scholar] [CrossRef]
- Ripoll, L.; Bordes, C.; Marote, P.; Etheve, S.; Elaissari, A.; Fessi, H. Electrokinetic properties of bare or nanoparticle-functionalized textile fabrics. Colloids Surf. A 2012, 397, 24–32. [Google Scholar] [CrossRef]
- Déon, S.; Fievet, P.; Doubad, C.O. Tangential streaming potential/current measurements for the characterization of composite membranes. J. Membr. Sci. 2012, 413–421. [Google Scholar] [CrossRef]
- Wang, Y.; Guo, L.; Ren, L.; Yin, S.; Ge, J.; Gao, Q.; Luxbacher, T.; Luo, S. A study on the performance of hyaluronic acid immobilized chitosan film. Biomed. Mater. 2009, 4, 035009. [Google Scholar] [CrossRef] [PubMed]
- Heberling, F.; Trainor, T.P.; Lützenkirchen, J.; Eng, P.; Denecke, M.A.; Bosbach, D. Structure and reactivity of the calcite-water interface. J. Colloid Interface Sci. 2011, 354, 843–857. [Google Scholar] [CrossRef] [PubMed]
- Kershner, R.J.; Bullard, J.W.; Cima, M.J. Zeta potential orientation dependence of sapphire substrates. Langmuir 2004, 20, 4101–4108. [Google Scholar] [CrossRef] [PubMed]
- Laumann, S.; Micić, V.; Lowry, G.V.; Hofmann, T. Carbonate minerals in porous media decrease mobility of polyacrylic acid modified zero-valent iron nanoparticles used for groundwater remediation. Environ. Poll. 2013, 179, 53–60. [Google Scholar] [CrossRef] [PubMed]
- Adamczyk, Z.; Sadlej, K.; Wajnryb, E.; Nattich, M.; Ekiel-Jezewska, M.L.; Bławzdziewicz, J. Streaming potential studies of colloid, polyelectrolyte and protein deposition. Adv. Colloid Interface Sci. 2010, 153, 1–29. [Google Scholar] [CrossRef] [PubMed]
- Szymczyk, A.; Dirir, Y.I.; Picot, M.; Nicolas, I.; Barrière, F. Advanced electrokinetic characterization of composite porous membranes. J. Membr. Sci. 2013, 429, 44–51. [Google Scholar] [CrossRef]
- Cerepi, A.; Loisy, C.; Toullec, R.; Burlot, R.; Galaup, S.; Schmutz, M. Electrical behaviour of saturated and unsaturated geological carbonate porous systems. Stud. Surf. Sci. Catal. 2006, 160, 713–719. [Google Scholar]
- Vdović, N.; Bišćan, J. Electrokinetics of natural and synthetic calcite suspensions. Colloids Surf. A 1998, 137, 7–14. [Google Scholar] [CrossRef]
- Kanellopoulou, D.G.; Koutsoukos, P.G. The calcitic marble/water interface: Kinetics of dissolution and inhibition with potential implications in stone conservation. Langmuir 2003, 19, 5691–5699. [Google Scholar] [CrossRef]
- Zhao, Z.; Wang, G. Surface electrical property and adsorption performance of Maifan stone. J. Nat. Sci. Heilongjiang Univ. 2007, 24, 357–360. [Google Scholar]
- Falchi, L.; Balliana, E.; Izzo, F.C.; Agostinetto, L.; Zendri, E. Distribution of nanosilica dispersions in Lecce stone. Sciences at Cà Foscari 2013, 1, 39–46. [Google Scholar]
- Lezzerini, M.; Marroni, M.; Raneri, S.; Tamayo, S.; Narbona, B.; Fernández, B.; Weber, J.; Ghaffari, E.; Ban, M.; Rohatsch, A. Part I: Natural stone test methods. In D1.5—Mapping of Stones and Their Decay; Nano-Cathedral Project Grant Agreement No. 646178-Confidential Deliverable; Nano-Cathedral: Pisa, Italy, 2017; pp. 8–16. [Google Scholar]
- Giesche, H. Mercury porosimetry: A general (practical) overview. Part. Part. Syst. Charact. 2006, 23, 9–19. [Google Scholar] [CrossRef]
- Roveri, M.; Gherardi, F.; Goidanich, S.; Niccolai, L.; Dami, V.; Toniolo, L. Use of Nanomaterials for the Protection of Historic Stone Architecture: Laboratory Methods for the Evaluation and Investigation of Photocatalytic Activity. In Proceedings of the 4th WTA International PhD Symposium, Delft, The Netherlands, 13–16 September 2017; Quist, W.J., Granneman, S.J.C., van Hees, R.P.J., Eds.; TU Delft: Delft, The Netherlands, 2017. [Google Scholar]
- Gherardi, F.; Roveri, M.; Goidanich, S.; Toniolo, L. Photocatalytic nanocomposites for the protection of european architectural heritage. Materials 2018, 11, 11010065. [Google Scholar] [CrossRef]
- EN 16581:2014. Surface protection for porous inorganic materials: Laboratory test methods for the evaluation of the performance of water repellent products. In Conservation of Cultural Heritage; European Committee for Standardization: Brussels, Belgium, 2014. [Google Scholar]
- Yaroshchuk, A.; Luxbacher, T. Interpretation of electrokinetic measurements with porous films: Role of electric conductance and streaming current within porous structure. Langmuir 2010, 26, 10882–10889. [Google Scholar] [CrossRef] [PubMed]
- EN 15801:2009. Test methods—Determination of water absorption by capillarity. In Conservation of Cultural Property; European Committee for Standardization: Brussels, Belgium, 2009. [Google Scholar]
- Peruzzi, R.; Poli, T.; Toniolo, L. The experimental test for the evaluation of protective treatments: A critical survey of the “capillary absorption index”. J. Cult. Herit. 2003, 4, 251–254. [Google Scholar] [CrossRef]
- EN 15802:2009. Test methods—Determination of static contact angle. In Conservation of Cultural Property; European Committee for Standardization: Brussels, Belgium, 2009. [Google Scholar]
- Schnitzer, C.; Ripperger, S. Influence of surface roughness on streaming potential method. Chem. Eng. Technol. 2008, 31, 1696–1700. [Google Scholar] [CrossRef]
- Kosmulski, M. Compilation of PZC and IEP of sparingly soluble metal oxides and hydroxides from literature. Adv. Colloid Interface Sci. 2009, 152, 14–25. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Silva, M.; Uribe-Salas, A.; Mirnezami, M.; Finch, J.A. The point of zero charge of phyllosilicate minerals using the Mular-Roberts titration technique. Miner. Eng. 2010, 23, 383–389. [Google Scholar] [CrossRef]
- Vdović, N. Electrokinetic behaviour of calcite—The relation with other calcite properties. Chem. Geol. 2001, 177, 241–248. [Google Scholar] [CrossRef]
- Hayes, R.A.; Böhmer, M.R.; Fokkink, L.G.J. Study of silica nanoparticle adsorption using optical reflectometry and streaming potential techniques. Langmuir 1999, 15, 2865–2870. [Google Scholar] [CrossRef]
- Dussaud, A.D.; Breen, P.C.; Koczo, K. Characterization of the deposition of silicone copolymers on keratin fibers by streaming potential measurements. Colloids Surf. A 2013, 434, 102–109. [Google Scholar] [CrossRef]
- Lee, C.H.; Park, S.H.; Chung, W.; Kim, J.Y.; Kim, S.H. Preparation and characterization of surface modified silica nanoparticles with organo-silane compounds. Colloids Surf. A 2011, 384, 318–322. [Google Scholar] [CrossRef]
- Kosmulski, M. The significance of the difference in the point of zero charge between rutile and anatase. Adv. Colloid Interface Sci. 2002, 99, 255–264. [Google Scholar] [CrossRef]
- Cassie, A.B.D.; Baxter, S. Wettability of porous surfaces. Trans. Faraday Soc. 1944, 40, 546–551. [Google Scholar] [CrossRef]
- Brugnara, M.; Degasperi, E.; Volpe, C.D.; Maniglio, D.; Penati, A.; Siboni, S.; Toniolo, L.; Poli, T.; Invernizzi, S.; Castelvetro, V. The application of the contact angle in monument protection: New materials and methods. Colloids Surf. A 2004, 241, 299–312. [Google Scholar] [CrossRef]
- Morga, M.; Adamczyk, Z.; Kosior, D. Silica monolayer formation and stability determined by in situ streaming potential measurements. Electrochim. Acta 2016, 206, 409–418. [Google Scholar] [CrossRef]
- Schabikowski, M.; Zalewska, M.; Kata, D.; Graule, T. The effect of CuO coatings on the electrokinetic properties of stonewool fibres determined by streaming potential measurements. Ceram. Int. 2016, 42, 13944–13951. [Google Scholar] [CrossRef]




| Stone Type | Open Porosity (vol%) | Average Pore Diameter (μm) | RMS Roughness (nm) |
|---|---|---|---|
| Apuan Marble | 0.7a | 0.08a | 6 1 |
| Balegem | 9.9 ± 0.8 | 0.28 ± 0.06 | 38 ± 8 |
| Obernkirchen | 24.1 ± 0.1 | 0.8 ± 0.3 | 10 ± 9 |
| Main Features of Formulation | WNC (Water-Based Nanocomposite) | ANC (Alcohol-Based Nanocomposite) |
|---|---|---|
| Solvent | water | 2-propanol |
| Main component 1 | aminoalkyl-trialkoxysilane (oligomers) | monomeric alkylalkoxysilane |
| Concentration (wt/wt) 1 | 15% | 40% |
| Density (g/cm3) | 1.03 | 0.84 |
| Viscosity (mPa⋅s) | 12 | 6 |
| pH | 4.5 | n.a. |
| TiO2 (ppm) 1 | 9600 | 1200 |
| TiO2 particle size (nm) | 106 | 25 |
| Stone Type | WNC | ANC |
|---|---|---|
| Apuan marble | <0.01 | 0.5 ± 0.2 |
| Balegem | 6 ± 3 | 10 ± 2 |
| Obernkirchen | 7 ± 1 | 20 ± 4 |
| Stone Type | Calcite + Dolomite 1 (mol%) | ζ (pH 7) (mV) | ζ (pH 5) (mV) | IEP (pH Units) |
|---|---|---|---|---|
| Apuan marble | 99.6 | −60 ± 4 | −47 ± 4 | 3.7 ± 0.1 |
| Balegem | 53.1 | −57 ± 6 | −48 ± 8 | 3.0 ± 0.3 |
| Obernkirchen | 1.2 | −88 ± 10 | −73 ± 5 | 2.1 ± 0.2 |
| Calcite monocrystal | 100 | - | −39 | 3.6 |
| Coating | IEP (pH Units) |
|---|---|
| WNC | 10.3 ± 0.7 |
| ANC | 7.9 ± 0.8 |
| Stone Type | WNC | ANC | ||||||
|---|---|---|---|---|---|---|---|---|
| Untreated 1 | Treated | Untreated 1 | Treated | |||||
| ζ | IEP | ζ | IEP | ζ | IEP | ζ | IEP | |
| Apuan marble | −58 ± 2 | 3.7 ± 0.2 | 24 ± 3 | 8.4 ± 0.6 | −62 ± 3 | 3.7 ± 0.1 | 7 ± 6 | 7.51 ± 0.06 |
| Balegem | −54 ± 5 | 2.8 ± 0.3 | 9 ± 5 | 7.7 ± 0.2 | −61 ± 2 | 3.1 ± 0.1 | −22 ± 10 | 6.4 ± 0.3 |
| Obernkirchen | −93 2 | 2.0 ± 0.1 | 43 2 | 8.9 ± 0.3 | −85 ± 10 | 2.2 ± 0.2 | 1 ± 1 | 7.1 ± 0.3 |
| Stone Type | ISM | |
|---|---|---|
| WNC | ANC | |
| Apuan marble | 0.71 ± 0.08 | 0.91 ± 0.01 |
| Balegem | 0.62 ± 0.09 | 0.68 ± 0.07 |
| Obernkirchen | 0.83 ± 0.03 | 0.86 ± 0.04 |
| Stone Type | WNC | ANC | ||||
|---|---|---|---|---|---|---|
| Untreated | Treated | Untreated | Treated | |||
| Integralnt | Integralt | ICrel | Integralnt | Integralt | ICrel | |
| Apuan marble | 1.82 ± 0.07 | 1.3 ± 0.1 | 0.69 ± 0.05 | 1.8 ± 0.2 | 0.17 ± 0.09 | 0.09 ± 0.05 |
| Balegem | 98 ± 13 | 15 ± 4 | 0.15 ± 0.04 | 76 ± 4 | 5.2 ± 0.4 | 0.068 ± 0.003 |
| Obernkirchen | 121 ± 4 | 15 ± 5 | 0.12 ± 0.04 | 124 ± 4 | 6 ± 1 | 0.05 ± 0.01 |
| Stone Type | WNC | ANC | ||||
|---|---|---|---|---|---|---|
| Untreated | Treated | Untreated | Treated | |||
| θnt | θt | δθ | θnt | θt | Δθ | |
| Apuan marble | 53 ± 4 | 129 ± 13 | 76 ± 13 | 51 ± 1 | 141 ± 2 | 92 ± 2 |
| Balegem | 32 ± 3 | 134 ± 15 | 101 ± 16 | 36 ± 1 | 141 ± 2 | 106 ± 3 |
| Obernkirchen | 21 ± 3 | 140 ± 4 | 119 ± 5 | 17 ± 7 | 137 ± 1 | 119 ± 6 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Roveri, M.; Raneri, S.; Bianchi, S.; Gherardi, F.; Castelvetro, V.; Toniolo, L. Electrokinetic Characterization of Natural Stones Coated with Nanocomposites for the Protection of Cultural Heritage. Appl. Sci. 2018, 8, 1694. https://doi.org/10.3390/app8091694
Roveri M, Raneri S, Bianchi S, Gherardi F, Castelvetro V, Toniolo L. Electrokinetic Characterization of Natural Stones Coated with Nanocomposites for the Protection of Cultural Heritage. Applied Sciences. 2018; 8(9):1694. https://doi.org/10.3390/app8091694
Chicago/Turabian StyleRoveri, Marco, Simona Raneri, Sabrina Bianchi, Francesca Gherardi, Valter Castelvetro, and Lucia Toniolo. 2018. "Electrokinetic Characterization of Natural Stones Coated with Nanocomposites for the Protection of Cultural Heritage" Applied Sciences 8, no. 9: 1694. https://doi.org/10.3390/app8091694
APA StyleRoveri, M., Raneri, S., Bianchi, S., Gherardi, F., Castelvetro, V., & Toniolo, L. (2018). Electrokinetic Characterization of Natural Stones Coated with Nanocomposites for the Protection of Cultural Heritage. Applied Sciences, 8(9), 1694. https://doi.org/10.3390/app8091694









