Diagnostics and Dosimetry Solutions for Multidisciplinary Applications at the ELIMAIA Beamline
Abstract
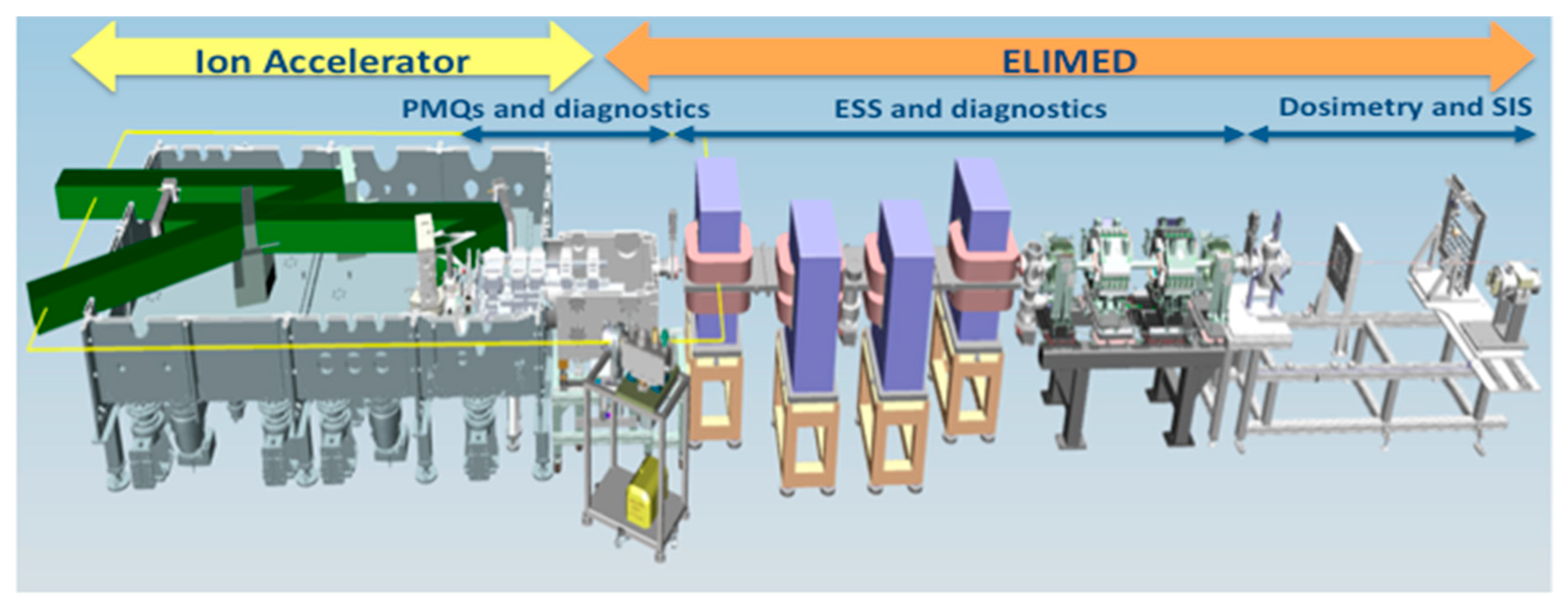
1. Introduction
2. ELIMAIA Diagnostics
- real-time diagnostics based on Thomson parabola spectrometers (TPS) and detectors working in time-of-flight (TOF) providing an on line characterization of the ion beam features;
- diagnostics operating in single shot mode using well-established detectors as radiochromic films (RCF), nuclear track detectors (typically CR39), image plates (IP), and other types of dosimeters.
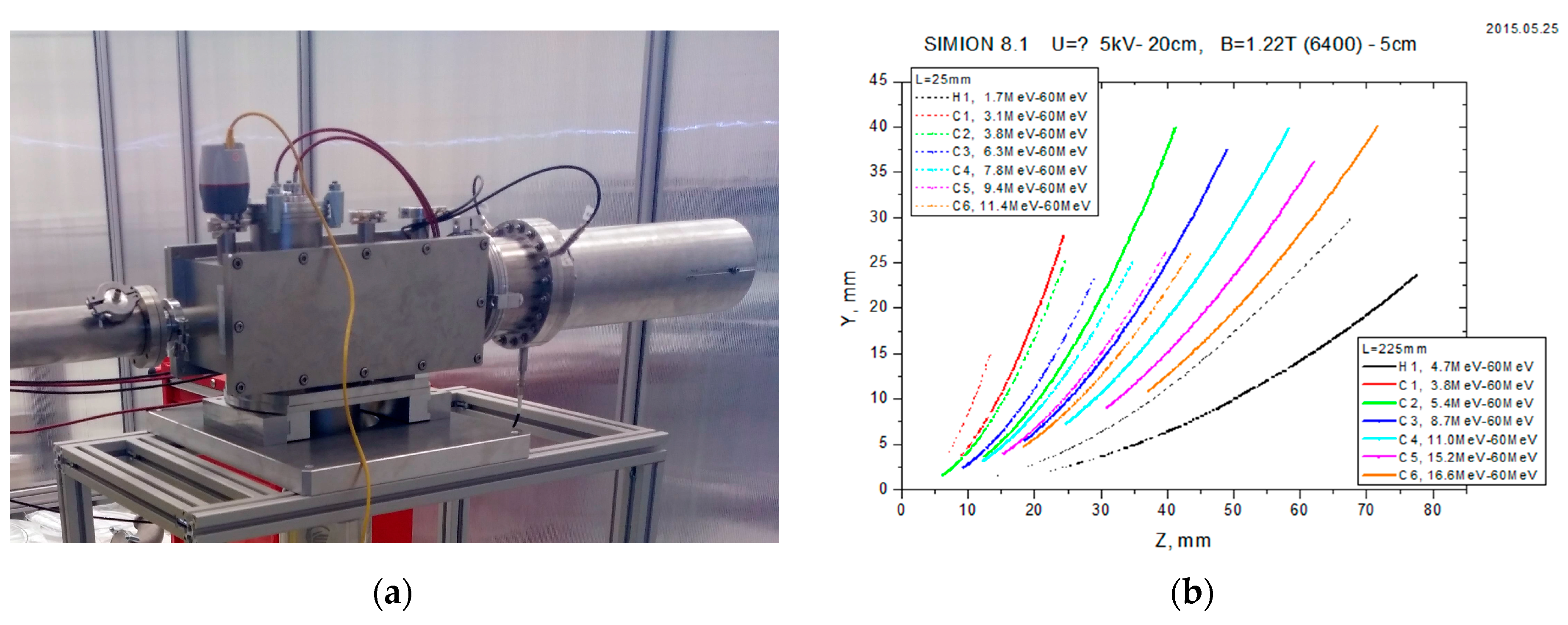
2.1. TPS: Thomson Parabola Spectrometer
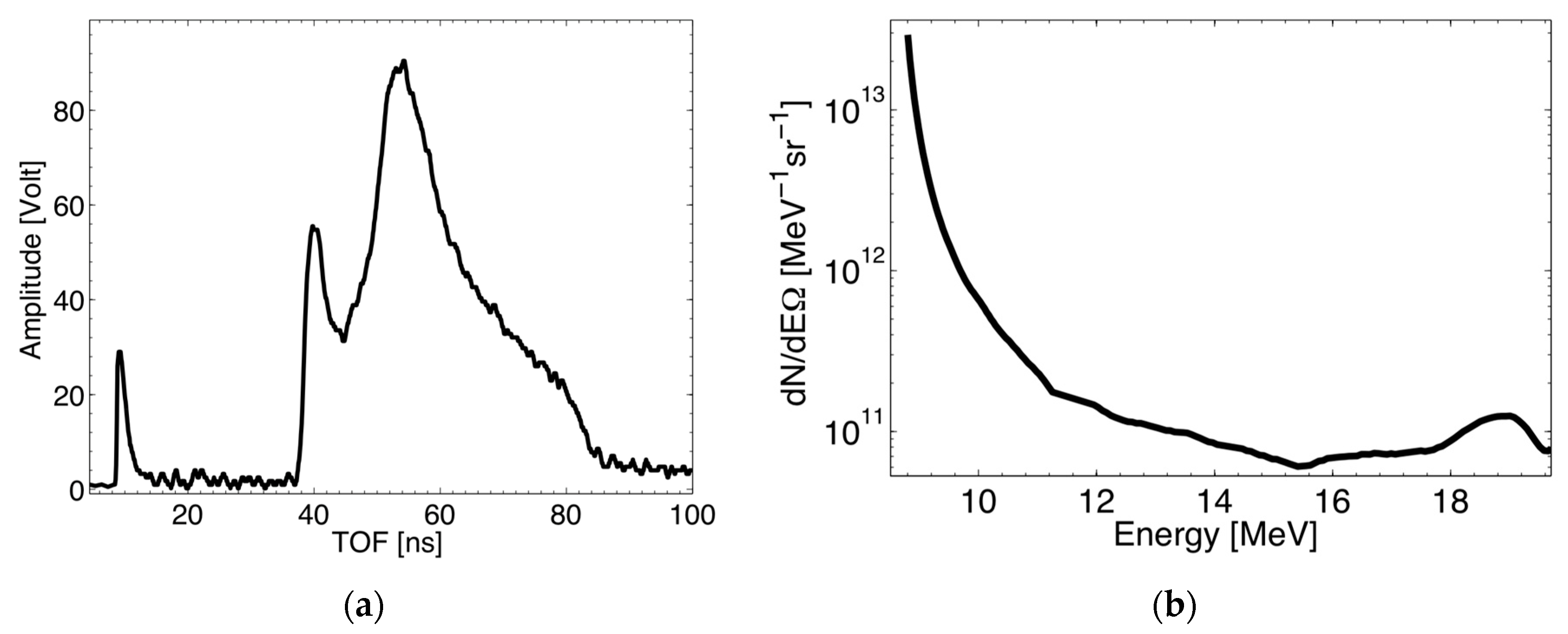
2.2. TOF: Description, Test, and Results
2.3. Emittance Diagnostics Device
2.4. Single Shot Diagnostics
3. ELIMAIA Dosimetry Description
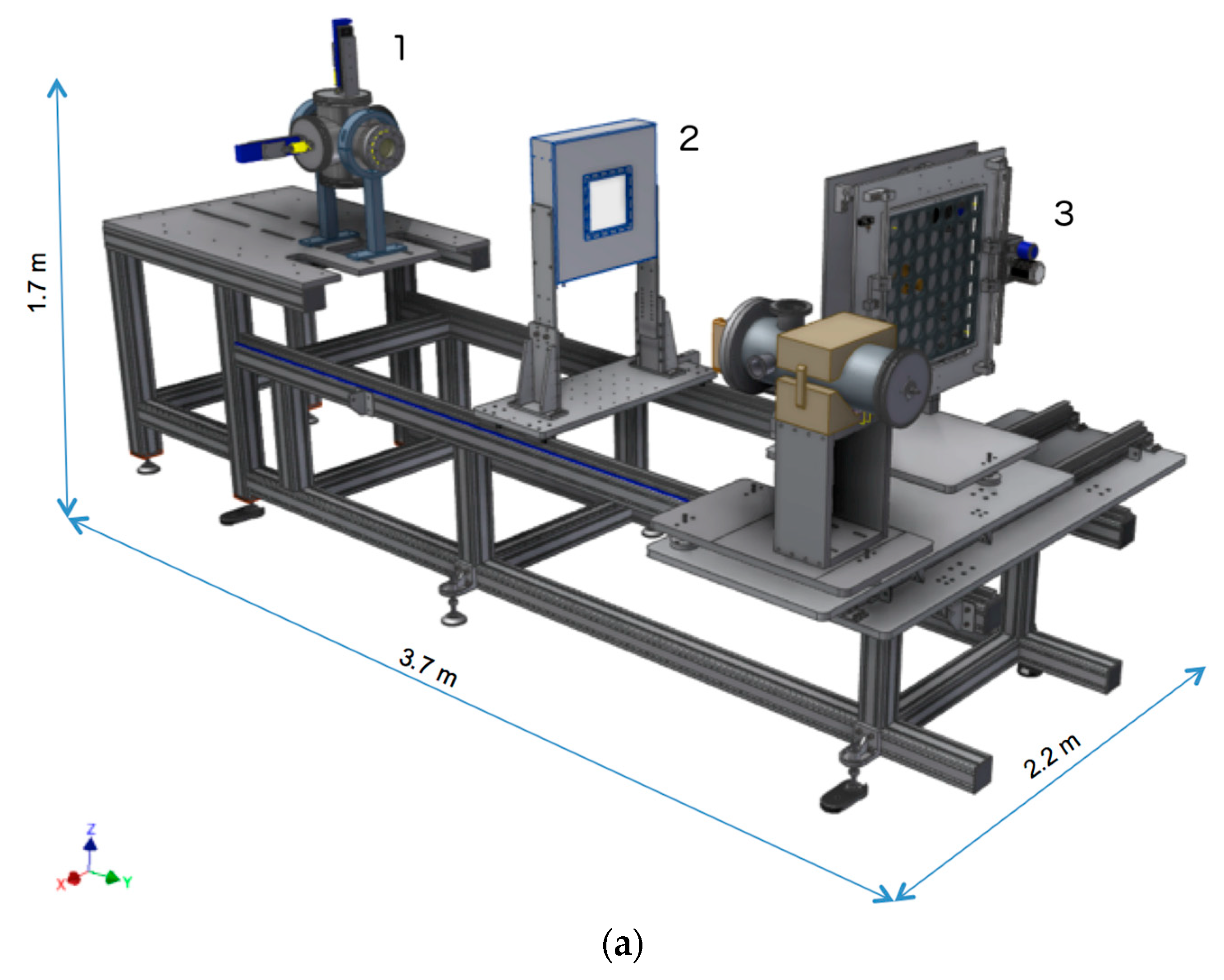
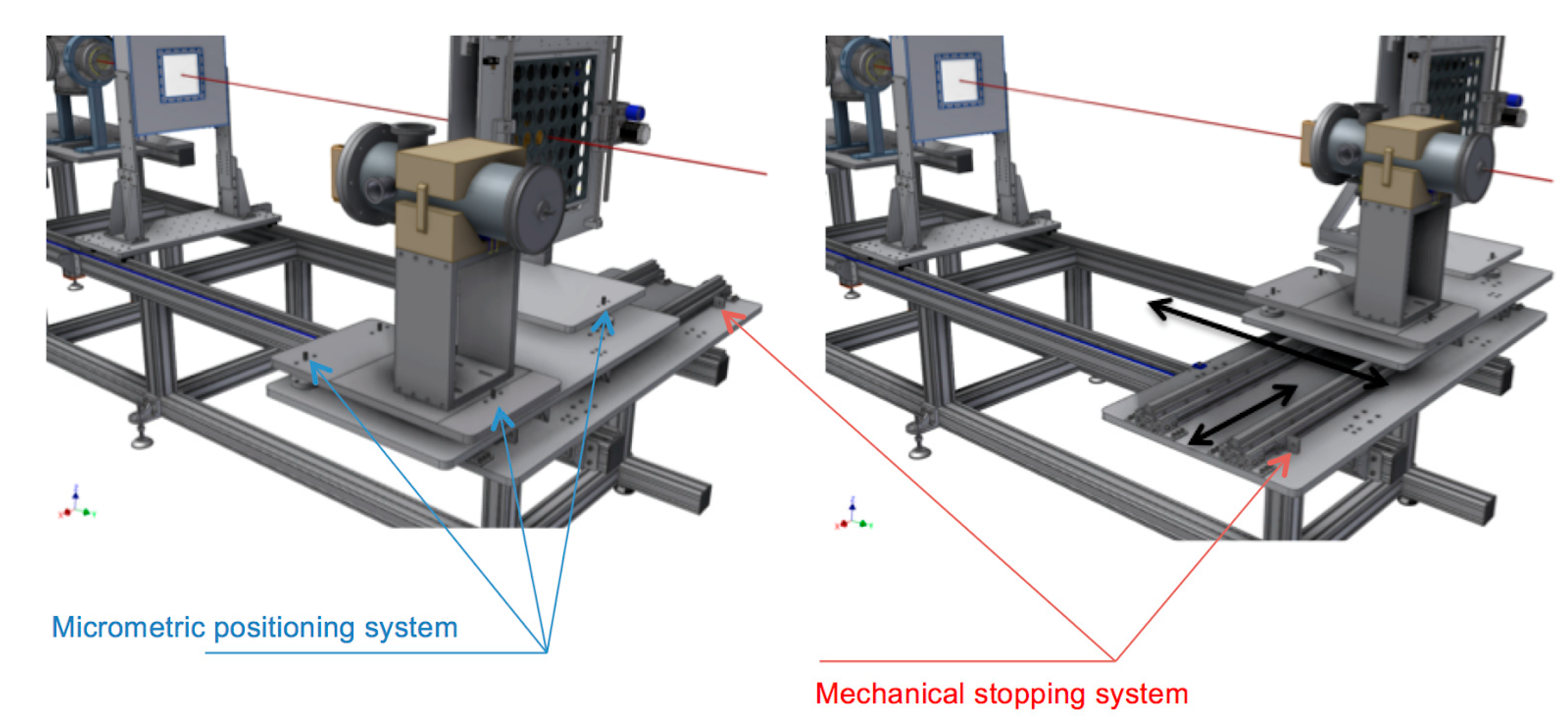
3.1. The In-Air Final Section for Dosimetry and Sample Irradiation
3.2. Detectors for Relative and Absolute Dosimetry
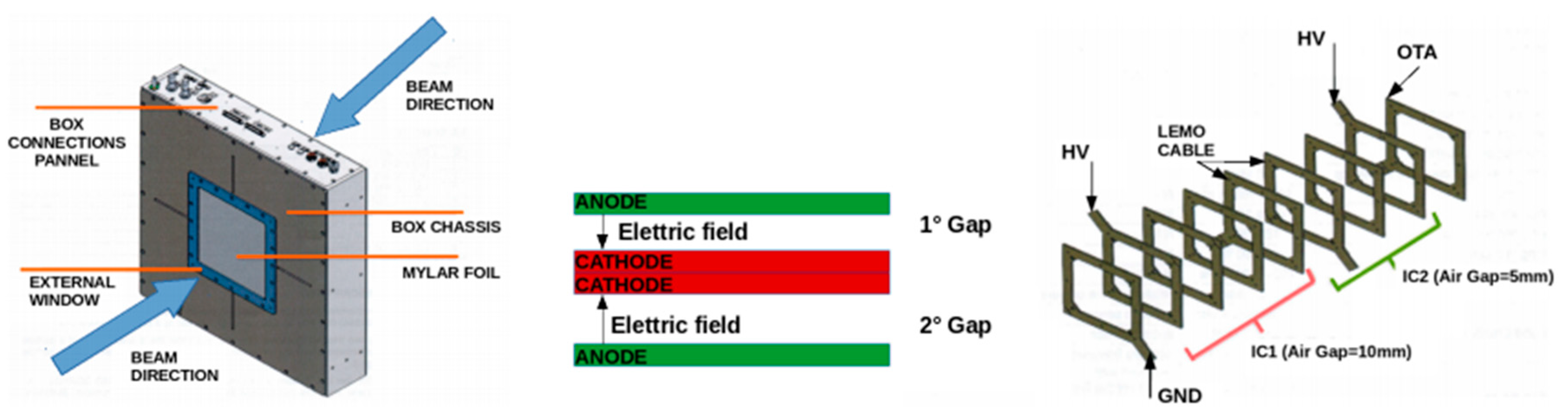
3.2.1. Secondary Electron Monitor
3.2.2. Multi-Gap Ionization Chamber
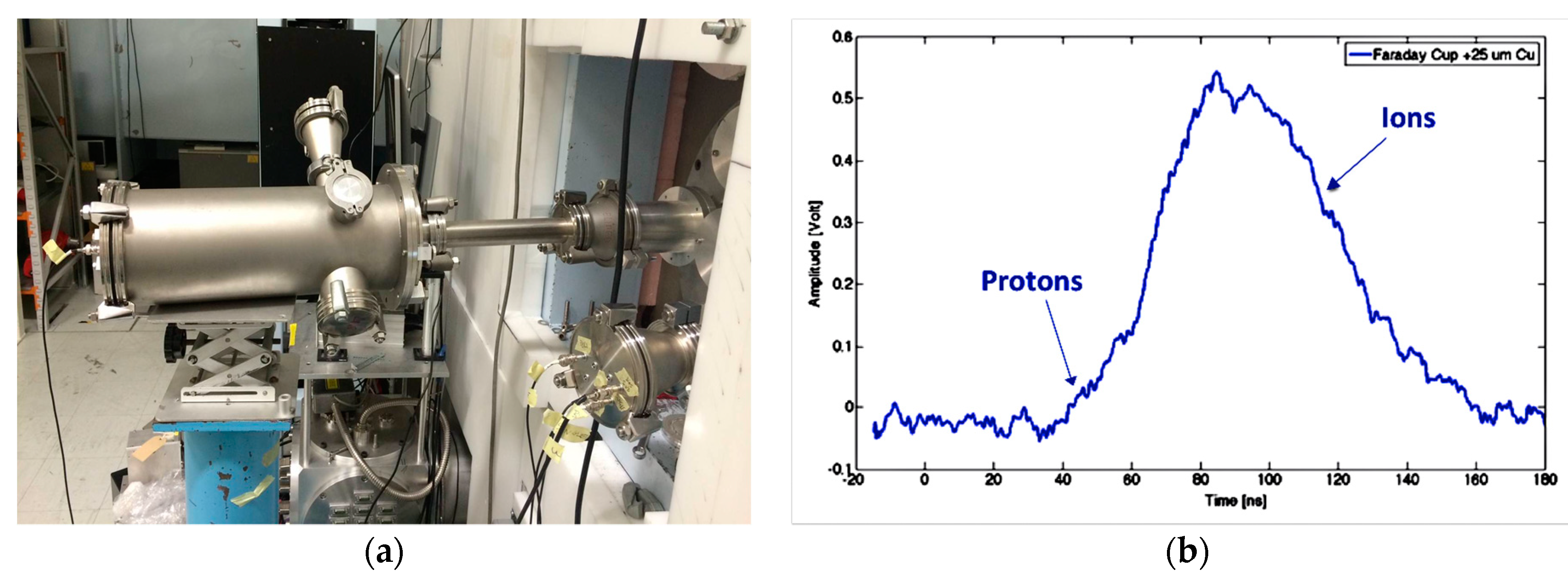
3.2.3. Faraday Cup
3.2.4. Irradiation Procedures
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
Abbreviations
| BED | Beam Emittance Detector |
| ELI | Extreme Light Infrastructure |
| ELIMAIA | ELI Multidisciplinary Applications of laser-Ion Acceleration |
| ELIMED | ELI MEDical applications |
| EMP | Electro-Magnetic Pulse |
| ESS | Energy Selection System |
| FC | Faraday Cup |
| IC | Ion Collector |
| IP | Image Plates |
| LET | Linear Energy Transfer |
| MGIC | Multi-Gap Ionization Chamber |
| PMQs | Permanent Magnet Quadrupoles |
| TOF | Time-Of-Flight |
| TPS | Thomson Parabola spectrometer |
| RCF | Radiochromic Film |
| SEM | Secondary Emission Monitor |
| SIS | Sample Irradiation System |
References
- Higginson, A.; Gray, R.J.; King, M.; Dance, R.J.; Williamson, S.D.R.; Butler, N.M.H.; Wilson, R.; Capdessus, R.; Armstrong, C.; Green, J.S.; et al. Near-100 MeV protons via a laser-driven transparency-enhanced hybrid acceleration scheme. Nat. Commun. 2018, 9, 724. [Google Scholar] [CrossRef] [PubMed]
- Wagner, F.; Deppert, O.; Brabetz, C.; Fiala, P.; Kleinschmidt, A.; Poth, P.; Schanz, V.A.; Tebartz, A.; Zielbauer, B.; Roth, M.; et al. Maximum proton energy above 85 MeV from the relativistic interaction of laser pulses with micrometer thick CH2 targets. Phys. Rev. Lett. 2016, 116, 205002. [Google Scholar] [CrossRef] [PubMed]
- Amaldi, U.; Kraft, G. Radiotherapy with beams of carbon ions. Rep. Prog. Phys. 2005, 68, 1861–1882. [Google Scholar] [CrossRef]
- Orecchia, R.; Zurlo, A.; Loasses, A.; Krengli, M.; Tosi, G.; Zurrida, S.; Zucali, P.; Veronesi, U. Particle beam therapy (Hadrontherapy): Basis for interest and clinical experience. Eur. J. Cancer 1998, 34, 459–468. [Google Scholar] [CrossRef]
- Malka, V.; Fritzler, S.; Lefebvre, E.; d’Humières, E.; Ferrand, R.; Grillon, G.; Albaret, C.; Meyroneinc, S.; Chambaret, J.; Antonetti, A.; et al. Practicability of protontherapy using compact laser systems. Med. Phys. 2004, 31, 1587–1592. [Google Scholar] [CrossRef] [PubMed]
- Hideghety, K.; Szabo, E.R.; Polanek, R.; Szabo, Z.; Ughy, B.; Brunner, S.; Tokes, T. An evaluation of the various aspects of the progress in clinical applications of laser driven ionizing radiation. J. Instrum. 2017, 12, C03038. [Google Scholar] [CrossRef]
- Bulanov, S.V.; Wilkens, J.J.; Esirkepov, T.Zh.; Korn, G.; Kraft, G.; Molls, M.; Khoroshkov, V.S. Laser ion acceleration for hadron therapy. Phys. Usp. 2014, 57, 1149–1179. [Google Scholar] [CrossRef]
- Ledingham, K.W.D.; Bolton, P.R.; Shikazono, N.; Ma, C.M.C. Towards Laser Driven Hadron Cancer Radiotherapy: A Review of Progress. Appl. Sci. 2014, 4, 402–443. [Google Scholar] [CrossRef]
- Margarone, D.; Cirrone, G.A.P.; Cuttone, G.; Amico, A.; Andò, L.; Borghesi, M.; Bulanov, S.S.; Bulanov, S.V.; Chatain, D.; Fajstavr, A.; et al. ELIMAIA: A laser-driven ion accelerator for multidisciplinary applications. Quantum Beam Sci. 2018, 2, 8. [Google Scholar] [CrossRef]
- The 3rd ELIMED Workshop MEDical and Multidisciplinary Applications of Laser-Driven Ion Beams at ELI-Beamlines (III ELIMED). Available online: http://iopscience.iop.org/journal/1748-0221/page/extraproc58 (accessed on 9 August 2018).
- Cirrone, G.A.P.; Cuttone, G.; Romano, F.; Schillaci, F.; Scuderi, V.; Amato, A.; Candiano, G.; Costa, M.; Gallo, G.; Larosa, G.; et al. Design and status of the ELIMED beam line for laser-driven ion beams. Appl. Sci. 2015, 5, 427–445. [Google Scholar] [CrossRef]
- Macchi, A.; Borghesi, M.; Passoni, M. Ion acceleration by superintense laser-plasma interaction. Rev. Mod. Phys. 2013, 85, 751. [Google Scholar] [CrossRef]
- Margarone, D.; Klimo, O.; Kim, I.J.; Prokůpek, J.; Limpouch, J.; Jeong, T.M.; Mocek, T.; Pšikal, J.; Kim, H.T.; Proška, J.; et al. Laser-Driven Proton Acceleration Enhancement by Nanostructured Foils. Phys. Rev. Lett. 2012, 109, 234801. [Google Scholar] [CrossRef] [PubMed]
- Klimo, O.; Psikal, J.; Limpouch, J.; Proska, J.; Novotny, F.; Ceccotti, T.; Floquet, V.; Kawata, S. Short pulse laser interaction with micro-structured targets: Simulations of laser absorption and ion acceleration. New J. Phys. 2011, 13, 053028. [Google Scholar] [CrossRef]
- Giuffrida, L.; Svensson, K.; Psikal, J.; Margarone, D.; Lutoslawski, P.; Scuderi, V.; Milluzzo, G.; Kaufman, J.; Wiste, T.; Dalui, M. Nano and micro structured targets to modulate the spatial profile of laser driven proton beams. J. Instrum. 2017, 12, C03040. [Google Scholar] [CrossRef]
- Margarone, D.; Velyhan, A.; Dostal, J.; Ullschmied, J.; Perin, J.P.; Chatain, D.; Garcia, S.; Bonnay, P.; Pisarczyk, T.; Dudzak, R.; et al. Proton Acceleration Driven by a Nanosecond Laser from a Cryogenic Thin Solid-Hydrogen Ribbon. Phys. Rev. X 2016, 6, 041030. [Google Scholar] [CrossRef]
- Opera Electromagnetic FEA Simulation Software. Available online: https://operafea.com/ (accessed on 9 August 2018).
- Margarone, D.; Krása, J.; Giuffrida, L.; Picciotto, A.; Torrisi, L.; Nowak, T.; Musumeci, P.; Velyhan, A.; Prokůpek, J.; Láska, L.; et al. Full characterization of laser-accelerated ion beams using Faraday cup, silicon carbide, and single-crystal diamond detectors. J. Appl. Phys. 2011, 109, 103302. [Google Scholar] [CrossRef]
- Marinelli, M.; Milani, E.; Prestopino, G.; Verona, C.; Verona-Rinati, G.; Cutroneo, M.; Torrisi, L.; Margarone, D.; Velyhan, A.; Krasa, J.; et al. Analysis of laser-generated plasma ionizing radiation by synthetic single crystal diamond detectors. Appl. Surf. Sci. 2013, 272, 104–108. [Google Scholar] [CrossRef]
- Milluzzo, G.; Scuderi, V.; Amico, A.G.; Borghesi, M.; Cirrone, G.A.P.; Cuttone, G.; De Napoli, M.; Doria, D.; Dostal, J.; Larosa, G.; et al. Laser-accelerated ion beam diagnostics with TOF detectors for the ELIMED beam line. J. Instrum. 2017, 12, C02025. [Google Scholar] [CrossRef]
- Scuderi, V.; Milluzzo, G.; Alejo, A.; Amico, A.G.; Booth, N.; Cirrone, G.A.P.; Doria, D.; Green, J.; Kar, S.; Larosa, G.; et al. Time of Flight based diagnostics for high energy laser driven ion beams. J. Instrum. 2017, 12, C03086. [Google Scholar] [CrossRef]
- Nurnberg, F.; Schollmeier, M.; Brambrink, E.; Blažević, A.; Carroll, D.C.; Flippo, K.; Gautier, D.C.; Geißel, M.; Harres, K.; Hegelich, B.M.; et al. Radiochromic film imaging spectroscopy of laser-accelerated proton beams. Rev. Sci. Instrum. 2009, 80, 033301. [Google Scholar] [CrossRef] [PubMed]
- Hey, D.S.; Key, M.H.; Mackinnon, A.J.; MacPhee, A.G.; Patel, P.K.; Freeman, R.R.; Van Woerkom, L.D.; Castaneda, C.M. Use of GafChromic film to diagnose laser generated proton beams. Rev. Sci. Instrum. 2008, 79, 053501. [Google Scholar] [CrossRef] [PubMed]
- Breschi, E.; Borghesi, M.; Galimberti, M.; Giulietti, D.; Gizzi, L.A.; Romagnani, L. A new algorithm for spectral and spatial reconstruction of proton beams from dosimetric measurements. Nucl. Instrum. Methods Phys. Res. Sect. A 2004, 522, 190–195. [Google Scholar] [CrossRef]
- Kirby, D.; Green, S.; Fiorini, F.; Parker, D.; Romagnani, L.; Doria, D.; Kar, S.; Lewis, C.; Borghesi, M.; Palmans, H. Radiochromic film spectroscopy of laser-accelerated proton beams using the FLUKA code and dosimetry traceable to primary standards. Laser Part Beams 2011, 29, 231–239. [Google Scholar] [CrossRef]
- Sadowski, M.; Al-Mashhadani, E.M.; SzydIowski, A.; Czyżewski, T.; Gtowacka, L.; Jaskóła, M.; Wieluński, M. Investigation on the response of CR-39 and PM-355 track detectors to fast protons in the energy range 0.2-4.5 MeV. Nucl. Instrum. Methods Phys. Res. Sect. B 1994, 86, 311–316. [Google Scholar] [CrossRef]
- Hegelich, B.M.; Albright, B.J.; Cobble, J.; Flippo, K.; Letzring, S.; Paffett, M.; Ruhl, H.; Schreiber, J.; Schulze, R.K.; Fernández, J.C. Laser acceleration of quasi-monoenergetic MeV ion beams. Nature 2006, 439, 441–444. [Google Scholar] [CrossRef] [PubMed]
- Jeong, T.W.; Singh, P.K.; Scullion, C.; Ahmed, H.; Hadjisolomou, P.; Jeon, C.; Yun, H.; Kakolee, K.F.; Borghesi, M.; Ter-Avetisyan, S. CR-39 track detector for multi-MeV ion spectroscopy. Sci. Rep. 2017, 7, 2152. [Google Scholar] [CrossRef] [PubMed]
- Sinenian, N.; Rosenberg, M.J.; Manuel, M.; McDuffee, S.C.; Casey, D.T.; Zylstra, A.B.; Rinderknecht, H.G.; Johnson, M.G.; Séguin, F.H.; Frenje, J.A.; et al. The response of cr-39 nuclear track detector to 1–9 mev protons. Rev. Sci. Instrum. 2011, 82, 103303. [Google Scholar] [CrossRef] [PubMed]
- Gaillard, S.; Fuchs, J.; Renard-Le Galloudec, N.; Cowan, T.E. Study of saturation of CR39 nuclear track detectors at high ion fluence and of associated artifact patterns. Rev. Sci. Instrum 2007, 78, 013304. [Google Scholar] [CrossRef] [PubMed]
- Romano, F.; Schillaci, F.; Cirrone, G.A.P.; Cuttone, G.; Scuderi, V.; Allegra, L.; Amato, A.; Amico, A.; Candiano, G.; De Luca, G.; et al. The ELIMED transport and dosimetry beamline for laser-drivenion beams. Nucl. Instrum. Methods Phys. Res. Sect. A 2016, 829, 153–158. [Google Scholar] [CrossRef]
- Cirrone, G.A.P.; Romano, F.; Scuderi, V.; Amato, A.; Candiano, G.; Cuttone, G.; Giovo, D.; Korn, G.; Krasa, J.; Leanza, R.; et al. Transport and dosimetric solutions for the ELIMED laser-driven beam line. Nucl. Instrum. Methods Phys. Res. Sect. A 2015, 796, 99–103. [Google Scholar] [CrossRef]
- Sternglass, E.J. Theory of Secondary Electron Emission by High-Speed Ions. Phys. Rev. 1957, 108, 1. [Google Scholar] [CrossRef]
- Hasselkamp, D.; Rothard, H.; Groeneveld, K.O.; Kemmler, J.; Varga, P.; Winter, H. Particle Induced Electron Emission II; Springer: Berlin, Germany, 1992. [Google Scholar]
- Badano, L.; Ferrando, O.; Klatka, T.; Koziel, M.; Molinari, G.; Abbas, K.; Braccini, S.; Bulgheroni, A.; Caccia, M.; Gibson, P.N.; et al. Laboratory and in-beam tests of a novel real-time beam monitor for hadrontherapy. IEEE Trans. Nucl. Sci. 2005, 52, 830–833. [Google Scholar] [CrossRef]
- Palmans, H.; Thomas, R.; Kacperek, A. Ion recombination correction in the Clatterbridge Centre of Oncology clinical proton beam. Phys. Med. Biol. 2006, 51, 903. [Google Scholar] [CrossRef] [PubMed]
- Boag, J.W. Ionization chambers. Radiat. Dosim. 1966, 2, 2–72. [Google Scholar]
- Boag, J.W. Ionization measurements at very high intensities—Part I. British J. Radiol. 1950, 2, 601–611. [Google Scholar] [CrossRef] [PubMed]
- Guarachi, L.F.; Sacchi, R.; Giordanengo, S.; Marchetto, F.; Talpacci, E.; Monaco, V.; Stasi, M.; Donetti, M.; Vignati, A.; Anvar, M.V.; et al. TH-CD-BRA-08: Multi-Gap Ionization Chamber for High-Flux Charged Particle Beams. Med. Phys. 2016, 42, 3727. [Google Scholar] [CrossRef]
- Cambria, R.; Hérault, J.; Brassart, N.; Silari, M.; Chauvel, P. Proton beam dosimetry: A comparison between the Faraday cup and an ionization chamber. Phys. Med. Biol. 1997, 42, 1185–1196. [Google Scholar] [CrossRef] [PubMed]
- Cuttone, G.; Raffaele, L.; Tonghi, L.; Fattibene, P. First Dosimetry Intercomparison Results for the CATANA Project. Phys. Med. 1999, 15, 121–130. [Google Scholar]
- Milluzzo, G.; Pipek, J.; Amico, A.G.; Cirrone, G.A.P.; Cuttone, G.; Korn, G.; Larosa, G.; Leanza, R.; Margarone, D.; Petringa, G.; et al. Geant4 simulation of the ELIMED transport and dosimetry beam line for high-energy laser-driven ion beam multidisciplinary applications. Nucl. Instrum. Methods Phys. Res. Sect. A 2018, arXiv:1802.03745. [Google Scholar] [CrossRef]
- Karsch, L.; Beyreuther, E.; Burris-Mog, T.; Kraft, S.; Richter, C.; Zeil, K.; Pawelke, J. Dose rate dependence for different dosimeters and detectors: TLD, OSL, EBT films, and diamond detectors. Med. Phys. 2012, 39, 2447–2455. [Google Scholar] [CrossRef] [PubMed]
- Jaccard, M.; Petersson, K.; Buchillier, T.; Germond, J.F.; Durán, M.T.; Vozenin, M.C.; Bourhis, J.; Bochud, F.O.; Bailat, C. High dose-per-pulse electron beam dosimetry: Usability and dose-rate independence of EBT3 Gafchromic films. Med. Phys. 2017, 44, 725–735. [Google Scholar] [CrossRef] [PubMed]
- Carnicer, A.; Angellier, G.; Gérard, A.; Garnier, N.; Dubois, C.; Amblard, R.; Hérault, J. Development and validation of radiochromic film dosimetry and Monte Carlo simulation tools for acquisition of absolute, high-spatial resolution longitudinal dose distributions in ocular proton therapy. Radiat. Meas. 2013, 59, 225–232. [Google Scholar] [CrossRef]







© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Scuderi, V.; Amato, A.; Amico, A.G.; Borghesi, M.; Cirrone, G.A.P.; Cuttone, G.; Fajstavr, A.; Giuffrida, L.; Grepl, F.; Korn, G.; et al. Diagnostics and Dosimetry Solutions for Multidisciplinary Applications at the ELIMAIA Beamline. Appl. Sci. 2018, 8, 1415. https://doi.org/10.3390/app8091415
Scuderi V, Amato A, Amico AG, Borghesi M, Cirrone GAP, Cuttone G, Fajstavr A, Giuffrida L, Grepl F, Korn G, et al. Diagnostics and Dosimetry Solutions for Multidisciplinary Applications at the ELIMAIA Beamline. Applied Sciences. 2018; 8(9):1415. https://doi.org/10.3390/app8091415
Chicago/Turabian StyleScuderi, Valentina, Antonino Amato, Antonio Giuseppe Amico, Marco Borghesi, Giuseppe Antonio Pablo Cirrone, Giacomo Cuttone, Antonin Fajstavr, Lorenzo Giuffrida, Filip Grepl, Georg Korn, and et al. 2018. "Diagnostics and Dosimetry Solutions for Multidisciplinary Applications at the ELIMAIA Beamline" Applied Sciences 8, no. 9: 1415. https://doi.org/10.3390/app8091415
APA StyleScuderi, V., Amato, A., Amico, A. G., Borghesi, M., Cirrone, G. A. P., Cuttone, G., Fajstavr, A., Giuffrida, L., Grepl, F., Korn, G., Larosa, G., Leanza, R., Margarone, D., Milluzzo, G., Petringa, G., Pipek, J., Russo, A., Schillaci, F., Velyhan, A., & Romano, F. (2018). Diagnostics and Dosimetry Solutions for Multidisciplinary Applications at the ELIMAIA Beamline. Applied Sciences, 8(9), 1415. https://doi.org/10.3390/app8091415






