Conformal Titanyl Phosphate Surface Passivation for Enhancing Photocatalytic Activity
Abstract
:1. Introduction
2. Materials and Methods
2.1. Material Synthesis
2.2. Material Characterization
2.3. Photocatalytic Activity
2.4. Photoelectrochemical Measurement
3. Results and Discussion
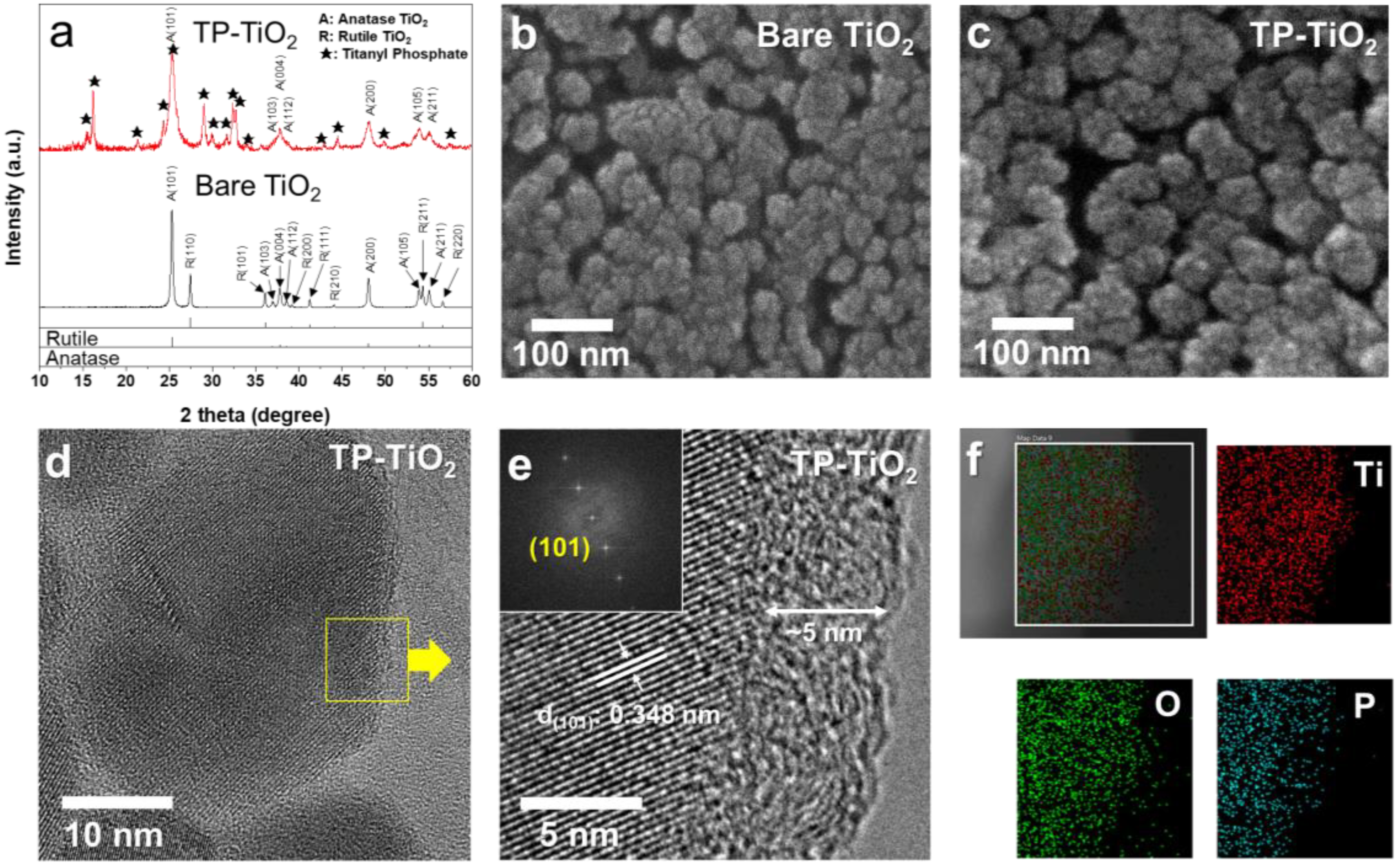
3.1. Morphology Characterization
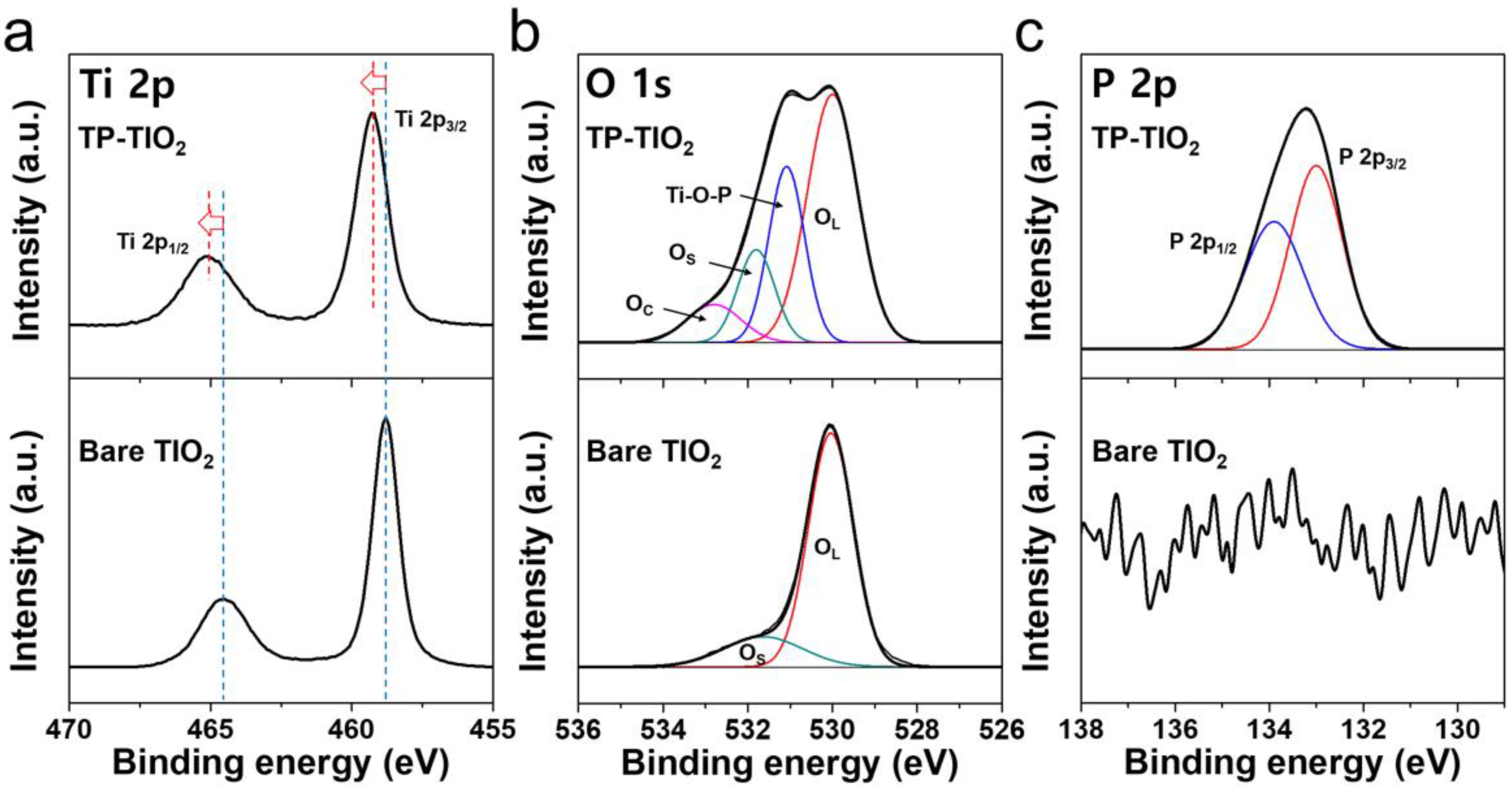
3.2. Chemical Modification
3.3. Photocatalytic Performance
4. Conclusions
Supplementary Materials
Acknowledgments
Conflicts of Interest
References
- Fujishima, A.; Honda, K. Electrochemical photolysis of water at a semiconductor electrode. Nature 1972, 238, 37–38. [Google Scholar] [CrossRef] [PubMed]
- O’Regan, B.; Grätzel, M. A low-cost, high-efficiency solar cell based on dye-sensitized colloidal TiO2 films. Nature 1991, 353, 737–740. [Google Scholar] [CrossRef]
- Bard, A.J. Photoelectrochemistry. Science 1980, 207, 139–144. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.K.; Jang, J.-r.; Choi, N.; Hong, D.; Nam, C.-H.; Yoo, P.J.; Park, J.H.; Choe, W.-S. Lysozyme-mediated biomineralization of titanium–tungsten oxide hybrid nanoparticles with high photocatalytic activity. Chem. Commun. 2014, 50, 12392–12395. [Google Scholar] [CrossRef] [PubMed]
- Crossland, E.J.W.; Noel, N.; Sivaram, V.; Leijtens, T.; Alexander-Webber, J.A.; Snaith, H.J. Mesoporous TiO2 single crystals delivering enhanced mobility and optoelectronic device performance. Nature 2013, 495, 215–219. [Google Scholar] [CrossRef] [PubMed]
- Giordano, F.; Abate, A.; Baena, J.P.C.; Saliba, M.; Matsui, T.; Im, S.H.; Zakeeruddin, S.M.; Nazeeruddin, M.K.; Hagfeldt, A.; Graetzel, M. Enhanced electronic properties in mesoporous TiO2 via lithium doping for high-efficiency perovskite solar cells. Nat. Commun. 2016, 7. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.K.; Chai, S.U.; Cho, Y.; Cai, L.L.; Kim, S.J.; Park, S.; Park, J.H.; Zheng, X.L. Ultrafast flame annealing of TiO2 paste for fabricating dye-sensitized and perovskite solar cells with enhanced efficiency. Small 2017, 13. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.K.; Shin, K.; Cho, S.M.; Lee, T.W.; Park, J.H. Synthesis of transparent mesoporous tungsten trioxide films with enhanced photoelectrochemical response: Application to unassisted solar water splitting. Energy Environ. Sci. 2011, 4, 1465–1470. [Google Scholar] [CrossRef]
- Onotri, L.; Race, M.; Clarizia, L.; Guida, M.; Alfè, M.; Andreozzi, R.; Marotta, R. Solar photocatalytic processes for treatment of soil washing wastewater. Chem. Eng. J. 2017, 318, 10–18. [Google Scholar] [CrossRef]
- Malato, S.; Blanco, J.; Vidal, A.; Alarcón, D.; Maldonado, M.I.; Cáceres, J.; Gernjak, W. Applied studies in solar photocatalytic detoxification: An overview. Sol. Energy 2003, 75, 329–336. [Google Scholar] [CrossRef]
- Aguirre, M.E.; Zhou, R.; Eugene, A.J.; Guzman, M.I.; Grela, M.A. Cu2O/TiO2 heterostructures for CO2 reduction through a direct Z-scheme: Protecting Cu2O from photocorrosion. Appl. Catal. B: Environ. 2017, 217, 485–493. [Google Scholar] [CrossRef]
- Park, H.; Park, Y.; Kim, W.; Choi, W. Surface modification of TiO2 photocatalyst for environmental applications. J. Photochem. Photobio. C: Photochem. Rev. 2013, 15, 1–20. [Google Scholar] [CrossRef]
- Naldoni, A.; Allieta, M.; Santangelo, S.; Marelli, M.; Fabbri, F.; Cappelli, S.; Bianchi, C.L.; Psaro, R.; Dal Santo, V. Effect of nature and location of defects on bandgap narrowing in black TiO2 nanoparticles. J. Am. Chem. Soc. 2012, 134, 7600–7603. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.; Yin, L.C.; Wang, J.Q.; Niu, P.; Zhen, C.; Xie, Y.P.; Cheng, H.M. A red anatase TiO2 photocatalyst for solar energy conversion. Energy Environ. Sci. 2012, 5, 9603–9610. [Google Scholar] [CrossRef]
- Li, Y.G.; Zhu, L.P.; Guo, Y.M.; Song, H.; Lou, Z.R.; Ye, Z.Z. A new type of hybrid nanostructure: Complete photo-generated carrier separation and ultrahigh photocatalytic activity. J. Mater. Chem. A 2014, 2, 14245–14250. [Google Scholar] [CrossRef]
- Gao, L.; Li, Y.; Ren, J.; Wang, S.; Wang, R.; Fu, G.; Hu, Y. Passivation of defect states in anatase TiO2 hollow spheres with mg doping: Realizing efficient photocatalytic overall water splitting. Appl. Catal. B: Environ. 2017, 202, 127–133. [Google Scholar] [CrossRef]
- Tong, H.; Ouyang, S.X.; Bi, Y.P.; Umezawa, N.; Oshikiri, M.; Ye, J.H. Nano-photocatalytic materials: Possibilities and challenges. Adv. Mater. 2012, 24, 229–251. [Google Scholar] [CrossRef] [PubMed]
- Minero, C.; Mariella, G.; Maurino, V.; Pelizzetti, E. Photocatalytic transformation of organic compounds in the presence of inorganic anions. 1. Hydroxyl-Mediated and Direct Electron-Transfer reactions of Phenol on a Titanium Dioxide−Fluoride system. Langmuir 2000, 16, 2632–2641. [Google Scholar] [CrossRef]
- Körösi, L.; Papp, S.; Bertóti, I.; Dékány, I. Surface and bulk composition, structure, and photocatalytic activity of phosphate-modified TiO2. Chem. Mater. 2007, 19, 4811–4819. [Google Scholar] [CrossRef]
- He, T.; Weng, Y.; Yu, P.; Liu, C.; Lu, H.; Sun, Y.; Zhang, S.; Yang, X.; Liu, G. Bio-template mediated in situ phosphate transfer to hierarchically porous TiO2 with localized phosphate distribution and enhanced photoactivities. J. Phys. Chem. C 2014, 118, 4607–4617. [Google Scholar] [CrossRef]
- Shao, G.-S.; Wang, F.-Y.; Ren, T.-Z.; Liu, Y.; Yuan, Z.-Y. Hierarchical mesoporous phosphorus and nitrogen doped titania materials: Synthesis, characterization and visible-light photocatalytic activity. Appl. Catal. B: Environ. 2009, 92, 61–67. [Google Scholar] [CrossRef]
- Jing, L.; Zhou, J.; Durrant, J.R.; Tang, J.; Liu, D.; Fu, H. Dynamics of photogenerated charges in the phosphate modified TiO2 and the enhanced activity for photoelectrochemical water splitting. Energy Environ. Sci. 2012, 5, 6552–6558. [Google Scholar] [CrossRef]
- Zhao, D.; Chen, C.C.; Wang, Y.F.; Ji, H.W.; Ma, W.H.; Zang, L.; Zhao, J.C. Surface modification of TiO2 by phosphate: Effect on photocatalytic activity and mechanism implication. J. Phys. Chem. C 2008, 112, 5993–6001. [Google Scholar] [CrossRef]
- Yu, J.C.; Zhang, L.Z.; Zheng, Z.; Zhao, J.C. Synthesis and characterization of phosphated mesoporous titanium dioxide with high photocatalytic activity. Chem. Mater. 2003, 15, 2280–2286. [Google Scholar] [CrossRef]
- Ortiz-Islas, E.; Gomez, R.; Lopez, T.; Navarrete, J.; Aguilar, D.H.; Quintana, P. Effect of the crystallite size in the structural and textural properties of sulfated and phosphated titania. Appl. Surf. Sci. 2005, 252, 807–812. [Google Scholar] [CrossRef]
- Connor, P.A.; McQuillan, A.J. Phosphate adsorption onto TiO2 from aqueous solutions: An in situ internal reflection infrared spectroscopic study. Langmuir 1999, 15, 2916–2921. [Google Scholar] [CrossRef]
- Kim, J.K.; Bae, S.; Kim, W.; Jeong, M.J.; Lee, S.H.; Lee, C.-L.; Choi, W.K.; Hwang, J.Y.; Park, J.H.; Son, D.I. Nano carbon conformal coating strategy for enhanced photoelectrochemical responses and long-term stability of ZnO quantum dots. Nano Energy 2015, 13, 258–266. [Google Scholar] [CrossRef]
- Linsebigler, A.L.; Lu, G.Q.; Yates, J.T. Photocatalysis on TiO2 surfaces—principles, mechanisms, and selected results. Chem. Rev. 1995, 95, 735–758. [Google Scholar] [CrossRef]
- Zhang, Y.Y.; Chen, J.Z.; Li, X.J. Preparation and photocatalytic performance of anatase/rutile mixed-phase TiO2 nanotubes. Catal. Lett. 2010, 139, 129–133. [Google Scholar] [CrossRef]
- Nolan, N.T.; Seery, M.K.; Pillai, S.C. Spectroscopic investigation of the anatase-to-rutile transformation of sol-gel-synthesized TiO2 photocatalysts. J. Phys. Chem. C 2009, 113, 16151–16157. [Google Scholar] [CrossRef]
- Antonelli, D.M.; Ying, J.C. Synthesis of hexagonally packed mesoporous TiO2 by a modified sol–gel method. Angew. Chem. Int. Ed. 1995, 34, 2014–2017. [Google Scholar] [CrossRef]
- Yermakov, A.Y.; Zakharova, G.S.; Uimin, M.A.; Kuznetsov, M.V.; Molochnikov, L.S.; Konev, S.F.; Konev, A.S.; Minin, A.S.; Mesilov, V.V.; Galakhov, V.R.; et al. Surface magnetism of cobalt-doped anatase TiO2 nanopowders. J. Phys. Chem. C 2016, 120, 28857–28866. [Google Scholar] [CrossRef]
- Jun, P.; Duong, T.; Zhou, X.Z.; Shen, H.P.; Wu, Y.L.; Mulmudi, H.K.; Wan, Y.M.; Zhong, D.Y.; Li, J.T.; Tsuzuki, T.; Weber, K.J.; et al. Efficient indium-doped Tix electron transport layers for high-performance perovskite solar cells and perovskite-silicon tandems. Adv. Energy Mater. 2017, 7, 1601768. [Google Scholar] [CrossRef]
- Boukhvalov, D.W.; Korotion, D.M.; Efremov, A.V.; Kurmaev, E.Z.; Borchers, C.; Zhidkov, I.S.; Gunderov, D.V.; Valiev, D.V.; Gavrilov, N.V.; Cholakh, S.O. Modification of titanium and titanium dioxide surfaces by ion implantation: Combined XPS and DFT study. Phys. Status Solidi B 2015, 252, 748–754. [Google Scholar] [CrossRef]
- Duan, X.; Liu, J.; Chen, Y.; Li, Z.; Zhu, P.; Jiang, H. Modification of RbTiOPO4 (001) crystal surface induced by Ar+ ion bombardment: X-ray photoelectron spectroscopy and first-principles studies. Vacuum 2018, 147, 38–44. [Google Scholar] [CrossRef]
- Zhang, X.; Qin, J.; Xue, Y.; Yu, P.; Zhang, B.; Wang, L.; Liu, R. Effect of aspect raTi and surface defects on the photocatalytic activity of ZnO nanorods. Sci. Rep. 2014, 4, 4596. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.K.; Shi, X.J.; Jeong, M.J.; Park, J.; Han, H.S.; Kim, S.H.; Guo, Y.; Heinz, T.F.; Fan, S.H.; Lee, C.L.; et al. Enhancing Mo:Bivo4 solar water splitting with patterned Au nanospheres by plasmon-induced energy transfer. Adv. Energy Mater. 2018, 8. [Google Scholar] [CrossRef]
- Liu, J.; Liu, Y.; Liu, N.; Han, Y.; Zhang, X.; Huang, H.; Lifshitz, Y.; Lee, S.-T.; Zhong, J.; Kang, Z. Metal-free efficient photocatalyst for stable visible water splitting via a two-electron pathway. Science 2015, 347, 970–974. [Google Scholar] [CrossRef] [PubMed]
- Dimitrijevic, N.M.; Vijayan, B.K.; Poluektov, O.G.; Rajh, T.; Gray, K.A.; He, H.; Zapol, P. Role of water and carbonates in photocatalytic transformation of CO2 to CH4 on titania. J. Am. Chem. Soc. 2011, 133, 3964–3971. [Google Scholar] [CrossRef] [PubMed]
- Pilli, S.K.; Deutsch, T.G.; Furtak, T.E.; Brown, L.D.; Turner, J.A.; Herring, A.M. BiVO4/CuWO4 heterojunction photoanodes for efficient solar driven water oxidation. Phys. Chem. Chem. Phys. 2013, 15, 3273–3278. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.K.; Cho, Y.; Jeong, M.J.; Levy-Wendt, B.; Shin, D.; Yi, Y.; Wang, D.H.; Zheng, X.L.; Park, J.H. Rapid formation of a disordered layer on monoclinic BiVO4: Co-catalyst-free photoelectrochemical solar water splitting. Chemsuschem 2018, 11, 933–940. [Google Scholar] [CrossRef] [PubMed]

© 2018 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, J.K. Conformal Titanyl Phosphate Surface Passivation for Enhancing Photocatalytic Activity. Appl. Sci. 2018, 8, 1402. https://doi.org/10.3390/app8081402
Kim JK. Conformal Titanyl Phosphate Surface Passivation for Enhancing Photocatalytic Activity. Applied Sciences. 2018; 8(8):1402. https://doi.org/10.3390/app8081402
Chicago/Turabian StyleKim, Jung Kyu. 2018. "Conformal Titanyl Phosphate Surface Passivation for Enhancing Photocatalytic Activity" Applied Sciences 8, no. 8: 1402. https://doi.org/10.3390/app8081402
APA StyleKim, J. K. (2018). Conformal Titanyl Phosphate Surface Passivation for Enhancing Photocatalytic Activity. Applied Sciences, 8(8), 1402. https://doi.org/10.3390/app8081402




