1. Introduction
Recently, tiny but highly deformable bending actuators based on smart materials are being actively studied because they have many advantages such as being lightweight, small and simple operating mechanisms [
1,
2,
3,
4,
5]. Among various smart material-based bending actuators, electroactive polymers (EAPs), which are operated by electric energy, are widely used. These electroactive polymers can be classified into two types: ionic and nonionic polymers. An ionic polymer/metal composite (IPMC) is one of the most popular ionic polymers for bending actuators. It consists of an ion-exchangeable membrane (ex, Nafion film) and electrodes. The IPMC-based bending actuators were widely studied over the past few decades, and they are still improving nowadays [
6,
7,
8]. Indrek Must et al. presented a bending actuator using an IPMC with carbon for worm-like robots [
7]. Ajahar Khan et al. proposed a new ion-exchangeable membrane (polyaniline thorium phosphate (ThP-PANI) composite) to substitute a Nafion film, which has drawbacks such as high cost, low water-retention capability and back-relaxation, and developed a bending actuator based on the IPMC with the new ionic-exchangeable membrane [
8]. Poly vinyl alcohols (PVAs) are another candidate material for fabricating bending actuators. Fan Wang et al. introduced a PVA-2,2,6,6-tetramethylpy-peridine-1-oxyl-oxidized bacterial cellulose nanofiber (TOCN)-based bending actuator [
9]. Xuan-Lun Wang et al. enhanced the actuation performance of a bending actuator based on PVA by improving proton conductivity and ionic-exchangeable capacity [
10]. Jiahua Shi et al. tried to mix multi-wall carbon nanotubes (MWCNTs) with PVA to enhance the electromechanical property of PVA in order to develop a bending actuator [
11]. Gang Zhao et al. fabricated another type of an ionic polymer using a biopolymer (chitosan polymer) and ionic liquid, and then developed a bending actuator based on the ionic polymer [
12]. Liang Gao et al. proposed a bending actuator using a post-synthetic modification of PVA and
N1,
N1-diethylethane-1,2-diamine (DEEDA) (PVA-DEEDA) [
13]. Jian-Ke Sun et al. developed a metal-organic framework hybrid nanoporous membrane-based bending actuator [
14]. Although there are many advantages (such as light weight and low power consumption) in ionic polymers and there have been lots of attempts to enhance the performance of bending actuators based on ionic polymers, the polyelectrolyte is vulnerable to solvent evaporation, and application of the electric field is inevitably accompanied by an electrolysis reaction.
On the other hand, nonionic EAP-based bending actuators, which are composed of dielectric polymers covered with stretchable electrodes on both sides, have a fast response time. Jun Shintake et al. developed a bending actuator using a dielectric polymer for a soft gripper [
15]. Guggi Kofod et al. designed a bending actuator having a minimum-energy structure based on a dielectric polymer [
16]. When voltage is applied to the electrode, a dielectric material quickly expands in planar directions and compresses in the thickness direction. Because the nonionic EAPs do not need electrolyte, they are operated in air conditions. However, the nonionic EAPs require a prestretching process to maximize their deformation and they need stretchable electrodes for their operation. Among many nonionic EAPs, polyvinylidene fluoride (PVDF) is one of the most attractive and representative nonionic polymers for fabricating bending actuators. I. Chilibon et al. introduced a bending actuator using prepolarized piezoelectric bimorph structure consisting of two PVDF thin films [
17]. R. Mejri et al. proposed a new method to fabricate a high-performance bending actuator using an ionic liquid/PVDF blend [
18]. Babita Gaihre et al. synthesized a PPy-PVDF multilayered electroactive polymer and fabricated a bending actuator using the PPy-PVDF multilayered polymer [
19]. Tian Chen and Bo Liu enhanced the performance of the bending transducer by applying graphene quantum dots to PVDF [
20]. The PVDF-based bending actuators are widely used and researched, and furthermore they are enhanced via various methods nowadays. However, they need highly stretchable electrodes for achieving a large bending behavior.
T. Hirai et al. [
21,
22] has presented a poly vinyl chloride (PVC) gel, which could solve the above problems, as an electroactive material for bending actuators. PVC gel-based bending actuators work well in air and do not require stretchable electrodes. Another great advantage of PVC gel-based bending actuators is that they are transparent. Even though a bending actuator based on an electroactive PVC gel does not need stretchable electrodes, there are still problems to be solved in that its response time is slow and its bending angle is not large. We inserted 12 related works in the reference section.
Previously, we synthesized PVC and dibutyl adipate (DBA) to fabricate an electroactive PVC gel [
23,
24]. When a voltage was applied to the developed electroactive PVC gel, the plasticizers in the PVC matrix migrated to the anode side with dragging PVC chains, consequently deforming the PVC gel. As soon as the voltage was removed, the PVC gel returned to its original shape due to its elasticity. Thus, we can readily assume that the stiffness of a PVC gel affects the performance of the PVC gel-based bending actuator. If the stiffness of the PVC gel is too great, its deformation becomes small. In contrast, a compliant PVC gel is deformed a great deal by a small voltage. A large amount of plasticizer not only makes the electroactive gel softer, but also increases the number of moving dipoles so that large amounts of plasticizers deform the PVC gel more. In other words, the actuation performance of the PVC gel is strongly related to the amount of plasticizer added.
The crystallinity, entanglement and physical crosslinking in PVC resins are affected by their molecular weight [
25,
26]. Thus, using PVC resins of different molecular weights would change the mechanical properties of PVC gels. PVC is a semicrystalline polymer having a 3-D crosslinking network. As the molecular weight of PVC increases, the crystalline region inside the PVC also increases. For this reason, the higher the molecular weight of the PVC resin used, the greater the area within the PVC that can be plasticized. Furthermore, a PVC with high molecular weight has denser entanglement and more physical crosslinking, so excess plasticizer can be retained in the PVC chain matrix. Previously, we investigated the effect of the molecular weight of the PVC resin on the electroactive performance of plasticized PVC gels [
27]. Because a high-molecular-weight PVC can accept more plasticizer, the performance of PVC gels based on high-molecular-weight PVC resin increases. In this paper, we investigated the electrical and mechanical properties of a PVC gel-based actuator in relation to its molecular weight and plasticizer content. Then, we fabricated a high-performance PVC gel-based actuator. The dynamic mechanical property of the proposed actuator was also investigated, and bending performance examined in terms of response time and maximum bending angle.
2. Experiment
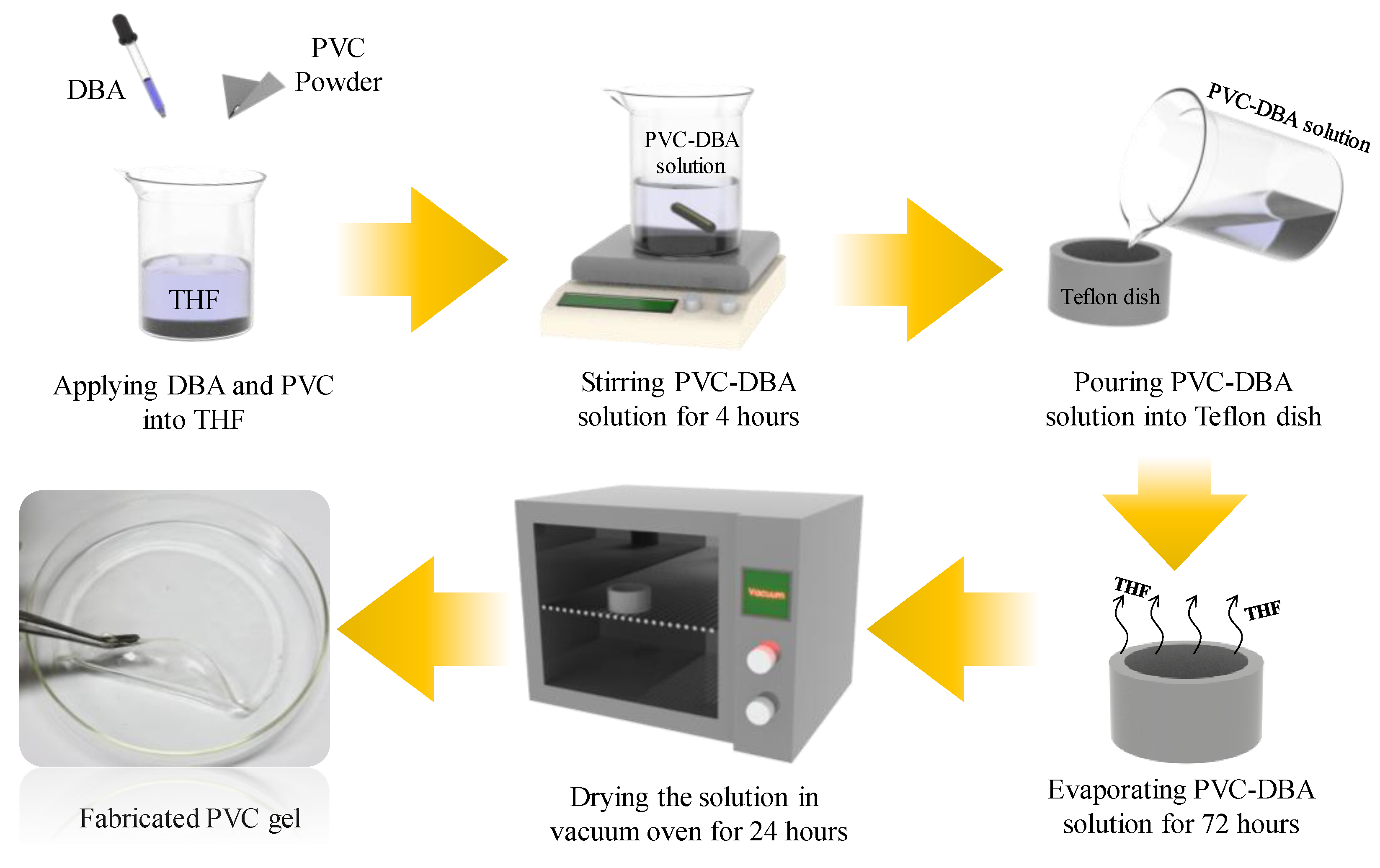
An electroactive PVC gel was fabricated using poly vinyl chloride (PVC), tetrahydrofuran (THF) as a solvent, and dibutyl adipate (DBA) as the plasticizer. The process for fabrication of the electroactive PVC gel is shown in
Figure 1. First, we poured PVC powder and DBA (Sigma-Aldrich, St. Louis, MO, USA) plasticizer into the THF (Sigma-Aldrich, St. Louis, MO, USA) solvent, and then stirred the solution (we call it PVC-DBA solution) for 4 h to fully dissolve the PVC and the DBA in the THF solvent. To obtain flat, transparent PVC gel, we poured the fully dissolved PVC-DBA solution into a Teflon dish; then we dried the solution for 72 h at room temperature. To evaporate the THF fully in the solution, we put the solution into a vacuum oven for 24 h. Finally, we obtained a transparent, electroactive, physically crosslinked and flat PVC gel, 1 mm thick.
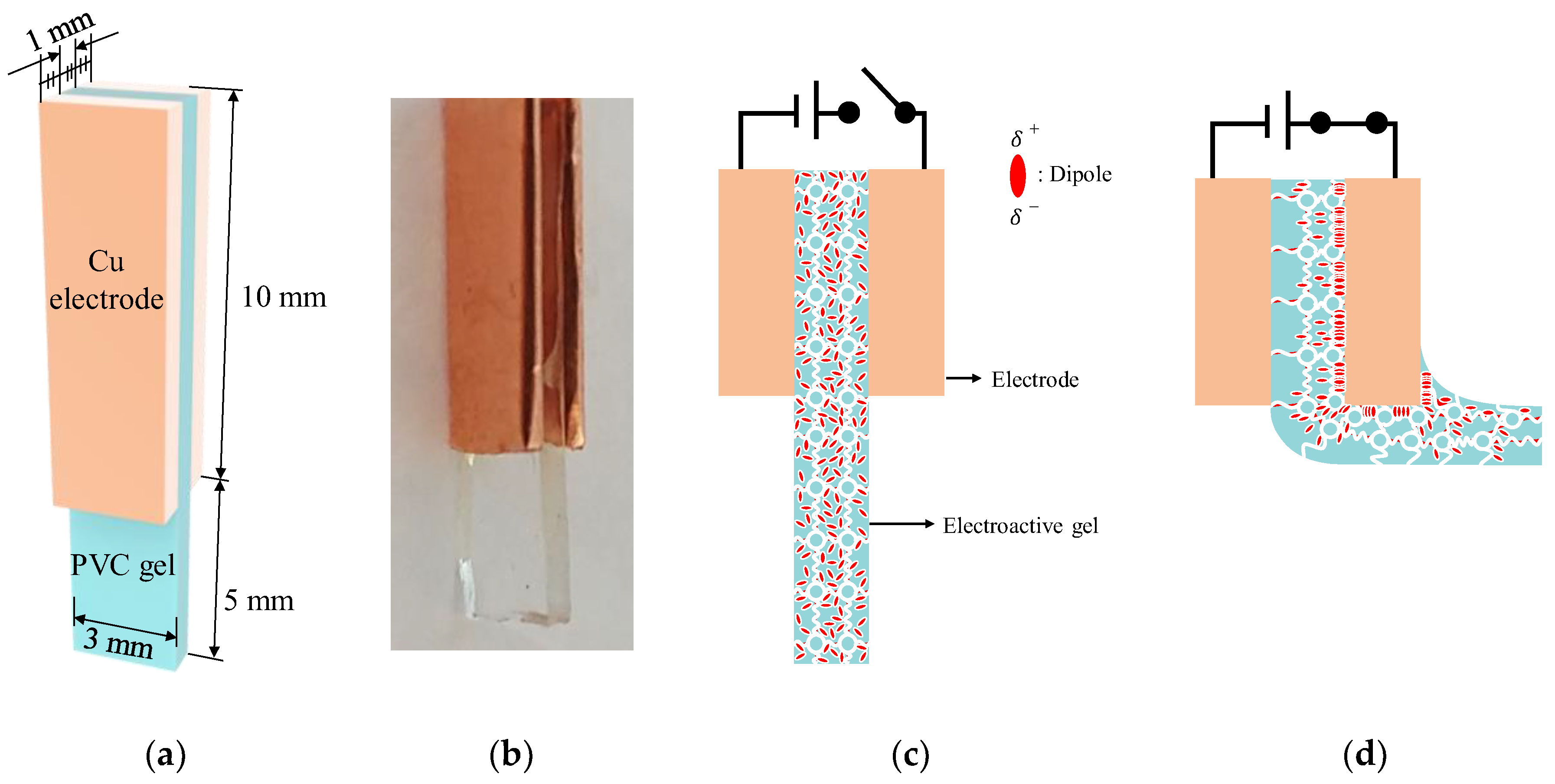
Two electrodes were attached, one on each side of the PVC gel, to fabricate a transparent bending actuator, as shown in
Figure 2a, where the PVC gel, PVC chains and dipoles in the PVC gel are colored aquamarine, white and red, respectively.
Figure 2b shows the fabricated bending actuator, and
Figure 2c,d show the voltage-off and -on states (respectively) of the proposed actuator. When we applied voltage input to the proposed actuator, the dipoles (DBA plasticizer) rotated and reoriented, then moved toward the anode, dragging the PVC chains. Thus, the PVC gel was deformed toward the anode, as shown in
Figure 2d. In the voltage-off state, electric field-induced deformation does not occur because the dipoles are randomly distributed in the PVC gel.
As we mentioned before, the performance of the PVC gel-based bending actuator is closely related not only to the plasticizer content but also to the molecular weight of the PVC. When the PVC gel is fabricated using a high-molecular-weight PVC resin, it can contain more plasticizer than a low-molecular-weight PVC-based gel. Furthermore, because a high-molecular-weight PVC gel has more entanglement and more physical crosslinking than a lower-molecular-weight PVC gel, the density of the PVC chains in the high-molecular-weight PVC gel is higher than in a PVC gel based on low-molecular-weight PVC. Under voltage input, the PVC chains and the plasticizer move toward the anode. At that time, the plasticizers in the high-molecular-weight PVC gel can accelerate the movement of PVC chains toward an anode. Because there are many more PVC chains near an anode in the high-molecular-weight PVC gel than in the lower-molecular-weight PVC gel, more PVC chains can be influenced by applied electric field [
27]. For these reasons, we expect that the molecular weight of the PVC will affect the deformation of the PVC gel-based bending actuator and its response time.
To investigate the molecular weight effect of the PVC gel-based bending actuator, we prepared three commercial PVCs (PVC
L: Wako; PVC
M: Sigma-Aldrich; and PVC
H: Scientific Polymer), with molecular weights of 116,000, 239,000 and 282,000, respectively. Furthermore, to observe the plasticizer effect, we fabricated PVC gels with different weight ratios of PVC resin to DBA plasticizer: 1:0, 1:3, 1:5, 1:7, 1:9, 1:11 and 1:13 denoted as PVC0 (PVC film), PVC3, PVC5, PVC7, PVC9, PVC11 and PVC13, respectively. In this preparation, for PVC
L, we could only obtain PVC
L3 and PVC
L5 because the PVC
L has low entanglement, low physical crosslinking, and a small crystalline region. If the ratio of PVC
L to plasticizer was more than 1:5, we could not obtain a PVC gel. Hence, we prepared 17 PVC gels as shown in
Table 1 (PVC
L0, PVC
L3, PVC
L5, PVC
M0, PVC
M3, PVC
M5, PVC
M7, PVC
M9, PVC
M11, PVC
M13, PVC
H0, PVC
H3, PVC
H5, PVC
H7, PVC
H9, PVC
H11 and PVC
H13).
3. Results and Discussion
Because the crystalline region of PVC is a turbid state, increase of the PVC molecular weight decreases the optical transmittance [
23,
24]. To show this effect, we prepared three PVC films (1 mm thick) with different molecular weights and measured their transmittance on indium tin oxide (ITO) glass using UV-vis spectroscopy (HP 8452, HP, Palo Alto, CA, USA).
Figure 3a shows the transmittance of the prepared pure PVC films. The optical transmittances of the PVC
L, PVC
M and PVC
H films at 550 nm were measured and found to be 48.9%, 46.1% and 37.6%, respectively. This result shows that the optical transmittance of pure PVC films decreases as we increase the PVC’s molecular weight.
The transmittance of the PVC gels was also measured. The measurement system and the procedure were the same as for the pure PVC films. As we mentioned before, we prepared 17 PVC gels. The prepared 17 PVC gels were divided into three groups (PVCL-based gel, PVCM-based gel and PVCH-based gel). We selected the PVC gel with the greatest amount of plasticizer for each PVC gel group. The selected PVC gels were PVCL5, PVCM13 and PVCH13. Normally, the plasticizers extend the gap between PVC chains and increase the free volume of the PVC gel. Due to these effects, the crystalline region of a PVC polymer is changed into an amorphous region with entanglement and physical crosslinking. Therefore, the transmittance of the PVC gel becomes higher than that of pure PVC films. Furthermore, comparing two PVC gels (PVCM13 and PVCH13) with the same plasticizer content but different molecular weights, the transparency of PVCM13 (95.1%) was almost the same as that of PVCH13 (95.4%) at 550 nm. This result implies that most of the crystalline regions in the PVC resin changed into amorphous regions during gel formation.
Strain–stress relationships of three pure PVC films (PVC
L0, PVC
M0 and PVC
H0) were measured using a Universal Tensile Machine (UTM, Tinius Olsen, H5KT) to confirm that PVCs with different molecular weights had different mechanical properties. For the experiment, dumbbell-shaped PVC films according to ASTM D638 type-V were prepared. The crosshead speed of the UTM was determined to be 50 mm/min. In
Figure 4, we can observe the mechanical properties (including the toughness and the break point) of the three prepared pure PVC films. This result indicates that the crystalline region of the PVC film increases with increasing molecular weight (same as the result for the transmittance). In addition, the strain and the break point of the PVC film increase as the molecular weight of the PVC increases. Because the entanglement and physical crosslinking in the amorphous region of the PVC increase with increasing molecular weight of the PVC, the PVC can stand more applied stress. Taken together, the increase of the PVC molecular weight causes a larger crystalline region, with more entanglement and physical crosslinking, in the PVC.
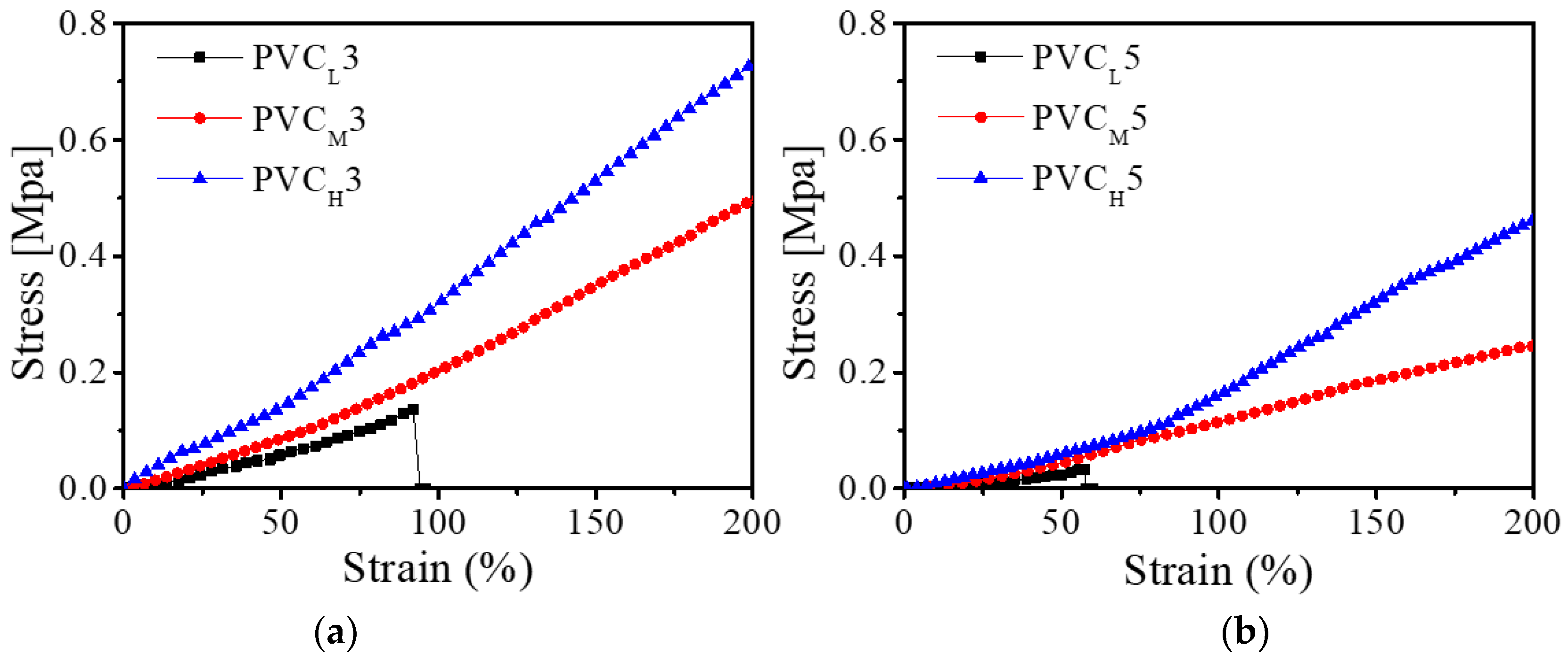
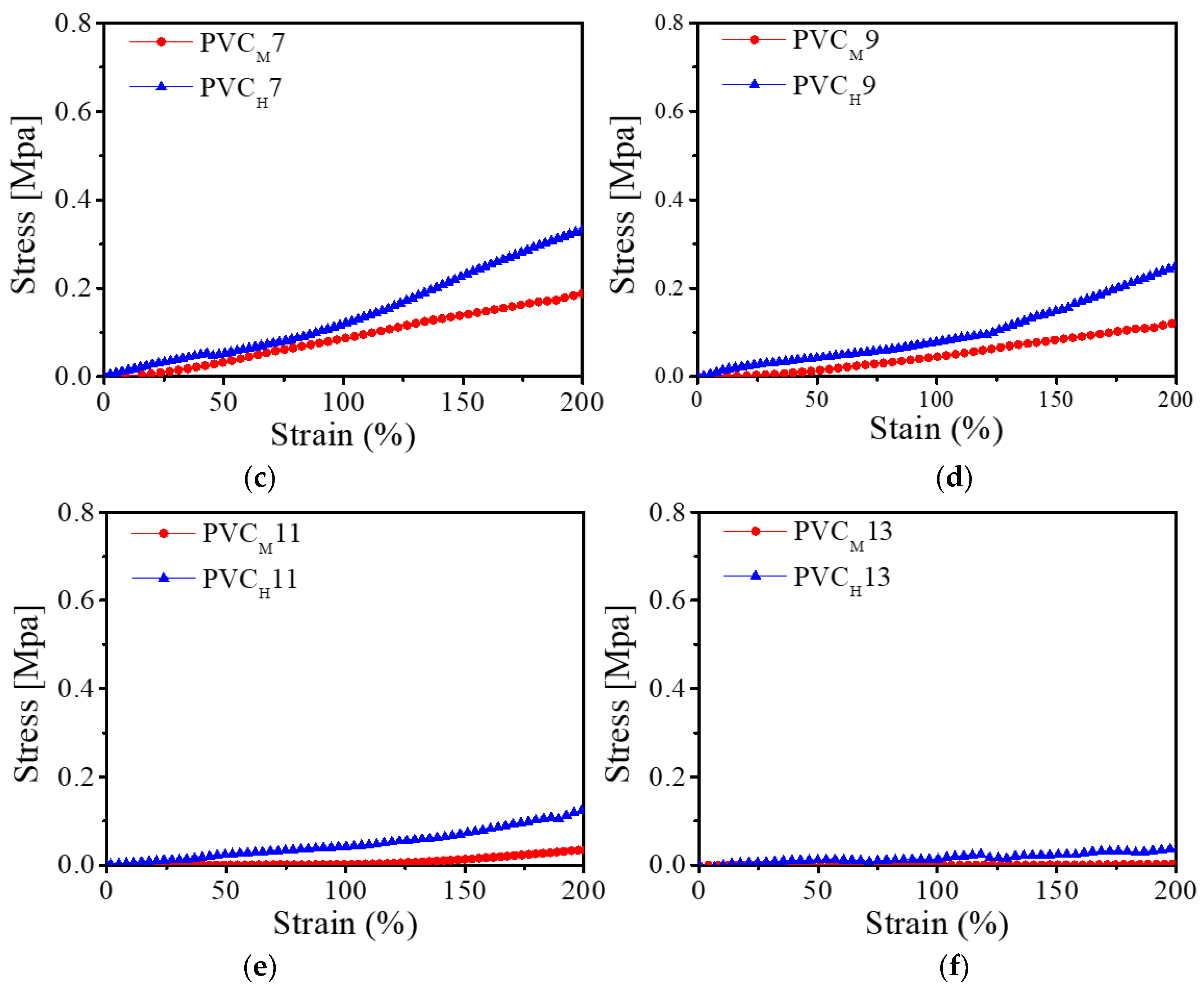
The mechanical property of the PVC gel is one of the most important factors affecting the actuation performance of the PVC gel-based bending actuator. Therefore, an experiment to investigate the mechanical properties of PVC gels was conducted using the same UTM as the one we used in the previous experiments. We prepared 14 PVC gel samples: two PVC gels with low molecular weight (PVCL3 and PVCL5), six PVC gels with medium molecular weight (PVCM), and six PVC gels with high molecular weight (PVCH).
Figure 5 shows the strain–stress relationships of the PVC gels with different molecular weights and different mixing weight ratios of plasticizer. From
Figure 5, we found that the increase of plasticizer in a PVC gel makes the PVC gel soften, and higher molecular weight leads to increased stiffness of the PVC gel. That is, these are two major factors (the weight ratio of PVC to DBA plasticizer and the molecular weight of PVC resin) for adjusting the stiffness of the PVC gel.
The dielectric property is also an important factor in the proposed bending actuator because the proposed actuator has electrically induced actuation behavior. The dielectric constant of a PVC gel was investigated by changing the molecular weight of the PVC resin and the amount of the plasticizer. An SI-1260 impedance/gain–phase analyzer connected with a 1296 dielectric interface (Solartron Analytical Co., Farnborough, UK) was used for the dielectric constant. The input frequency is swept from 1 Hz to 106 Hz with 10 points/decade under 2 V input voltage.
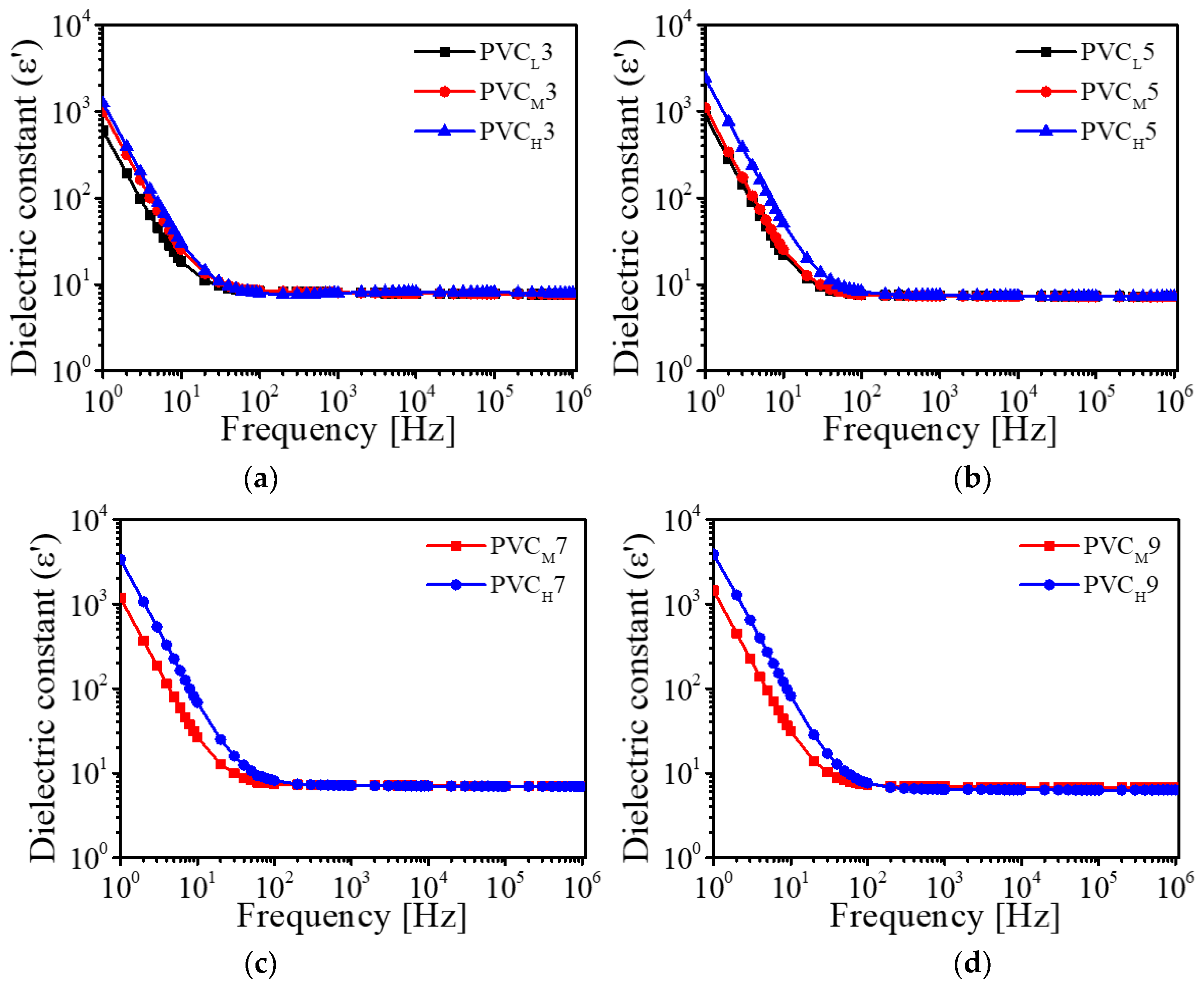
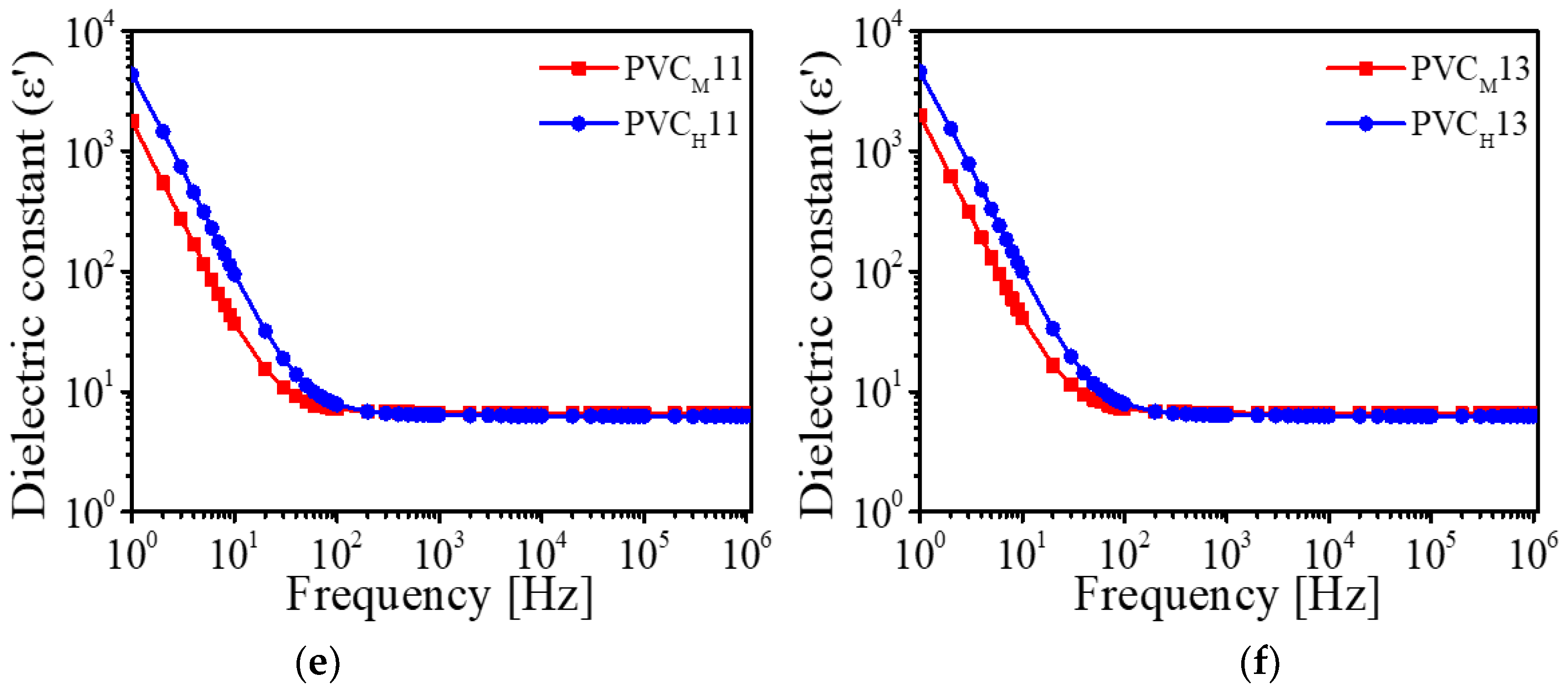
Figure 6 shows the results of dielectric constants of the prepared PVC gels as a function of input frequency. The dielectric constants are increased in the low-frequency range (1–20 Hz). It can be considered that, in a nonionic PVC gel physically crosslinked with plasticizer, DBA molecules are charged and polarized under the electric field, and then the polarized DBA easily facilitates the free dipole reorientation of the PVC chain in the PVC gel network [
28]. This dipole reorientation of a PVC gel makes its dielectric constant high. In the high-frequency range (20 Hz–10
6 Hz), the dielectric constants of the PVC gels were almost saturated because the dipoles stop rotation.
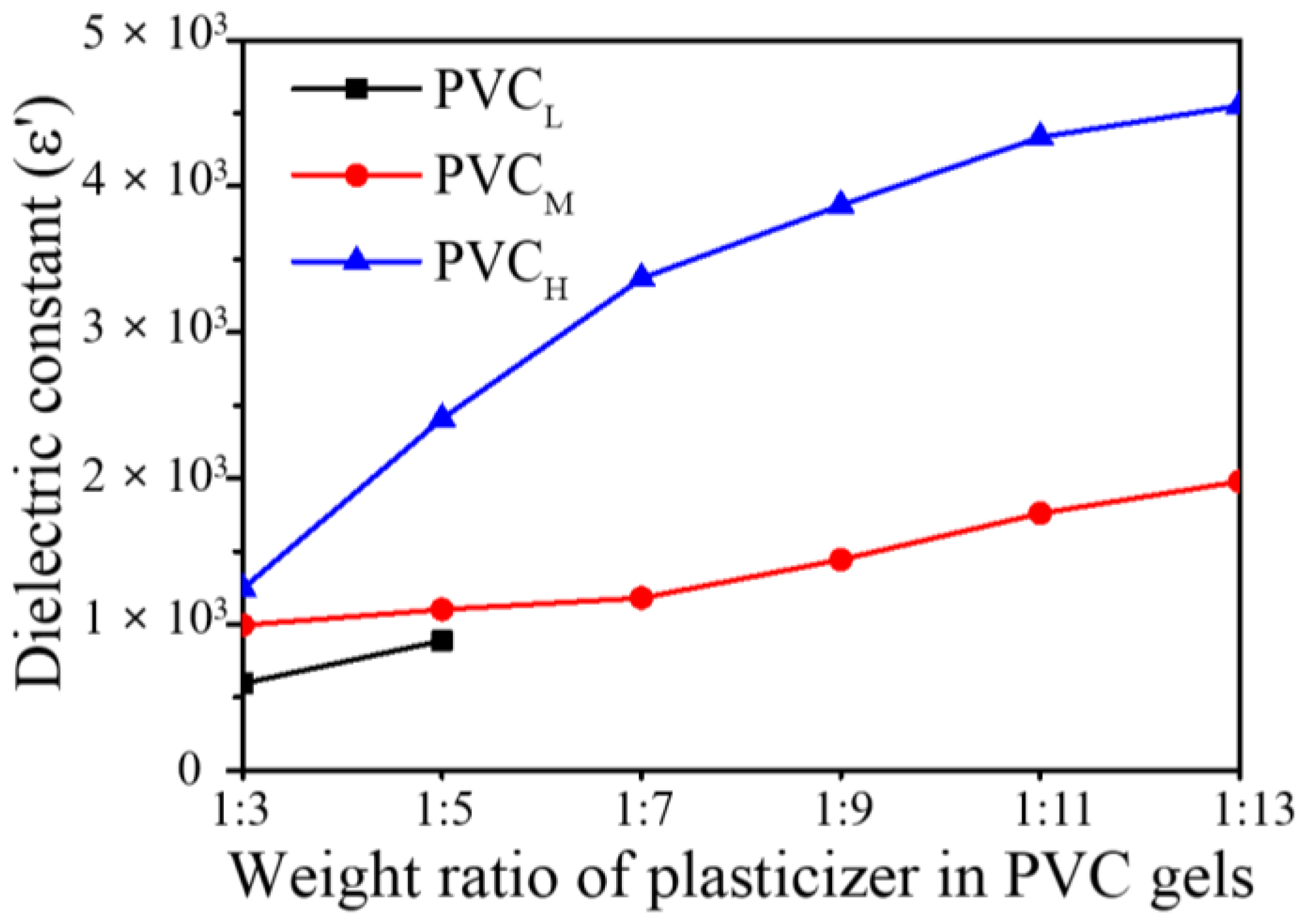
We plotted the dielectric constants of the prepared PVC gels at 1 Hz (
Figure 7). As shown in
Figure 7, as the amount of plasticizer increases, the number of dipoles increases, and as a result, the dielectric constant also increases. Furthermore, the dielectric constant is proportional to the molecular weight of the PVC resin (
Figure 6). It means that the use of PVC resin with high molecular weight makes entanglement, physical crosslinking and crystallization regions in a PVC gel increase.
The leakage current is another important factor in electrically induced actuation of the proposed bending actuator. The leakage current of the prepared PVC gels was measured by a potentiostat/galvanostat (Bio-logic Science Instruments, SP300) in applied electric fields of 500 V/mm at room temperature.
Figure 8 shows the results of the prepared samples’ leakage currents. It seems that the leakage current in the PVC gel is proportional to the contents of the plasticizer and is inversely proportional to the molecular weight of the PVC. However, there are little differences in leakage currents from the prepared PVC gel samples.
As we mentioned before, we prepared three PVC gel groups (PVCL-based gel, PVCM-based gel and PVCH-based gel). In each group, we need to select the best mixing ratio of PVC and a plasticizer using two important properties (dielectric and mechanical properties). The increase of a plasticizer makes a PVC gel soft and it makes its dielectric constant high. Although the increase of the dielectric constant can improve the performance of the PVC gel, the increase of the compliance can disturb the precise movement of the PVC gel-based bending actuator. Thus, we can say that there is a trade-off relationship between the dielectric constant of a PVC gel and its stiffness. Considering the dielectric/mechanical properties of the proposed PVC gel, we selected PVCL5, PVCM9 and PVCH11 gels. We inserted the following work in the reference section.
We conducted an experiment to investigate the dynamic mechanical properties of those three PVC gels (PVC
L5, PVC
M9 and PVC
H11) using a TA Instruments RSA-G2 dynamic mechanical analyzer. Specimens of the three PVC gels were prepared with dimensions of 10 mm (W) × 50 mm (H) × 0.2 mm (T). In this experiment, we obtained the dynamic mechanical properties of the PVC gels in the time domain and frequency domain. In the case of the time domain, the PVC gels were strained 10 times (2% at 1 Hz, sinusoidal), and then the PVC gels were strained with 2% at 1–50 Hz (sinusoidal) in the frequency domain.
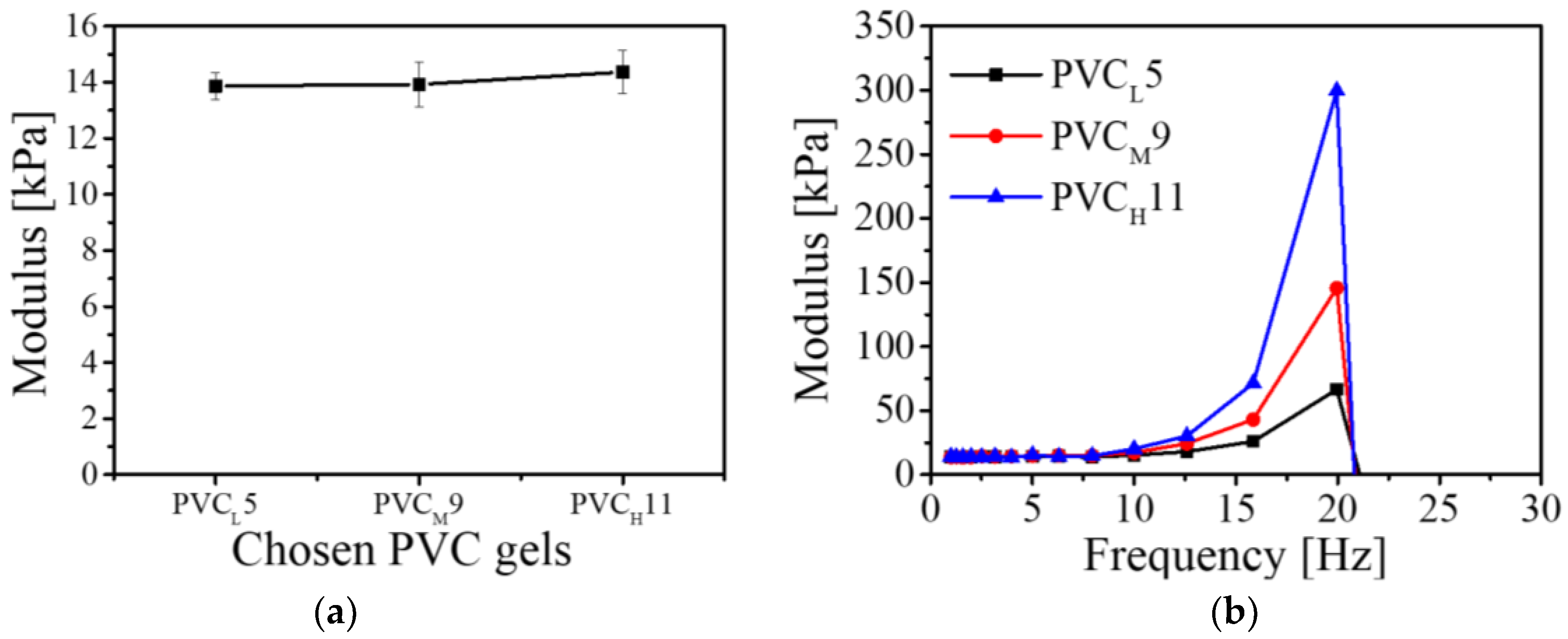
Figure 9a shows the result of the dynamic mechanical analysis in the time domain. The points are the average of the modulus, and the error bars show the standard deviation. The average storage moduli of the PVC
L5, PVC
M9 and PVC
H11 gels were 13.866, 13.923 and 14.366 kPa, respectively. Although the three PVC gels had different plasticizer contents, there was no significant discrepancy in the storage modulus.
Figure 9b shows the result of the dynamic mechanical analysis (DMA) in the frequency domain. As with the time domain result, there was no significant discrepancy among the three PVC gel samples in the low-frequency range (1–10 Hz), but the moduli of the PVC gels dramatically increased above 10 Hz and their rates of increase were distinct. In particular, the modulus of the PVC
H11 gel, which had the longest PVC chains among the three samples, showed a remarkable increase. The maximum modulus of the PVC
H11 gel reached 299 kPa, while the moduli of PVC
L5 and PVC
M9 were 66 and 145 kPa, respectively. It seems that the modulus of the PVC gel is proportional to the PVC chain length. We found that the valid frequency range of all three PVC gel samples was about 20 Hz.
We fabricated three bending actuators based on the PVC
L5, PVC
M9 and PVC
H11 gels, and then quantitatively evaluated their actuation performance in terms of response times and deformation angles. To measure these, we designed an experimental environment using a graph paper and a digital camera (1 V3, Nikon, Tokyo, Japan), as shown in
Figure 10. To measure the bending angle of the actuator, we installed a wall behind the bending actuator and attached the graph paper to the wall. When we applied a voltage to a PVC gel-based bending actuator, the bending deformation occurred rapidly. At that time, we recorded the bending angle (
) using the camera (120 fps) and saved it to memory. After that, the time it took for the bending angle of the proposed actuator to increase by 10° was calculated through image processing.
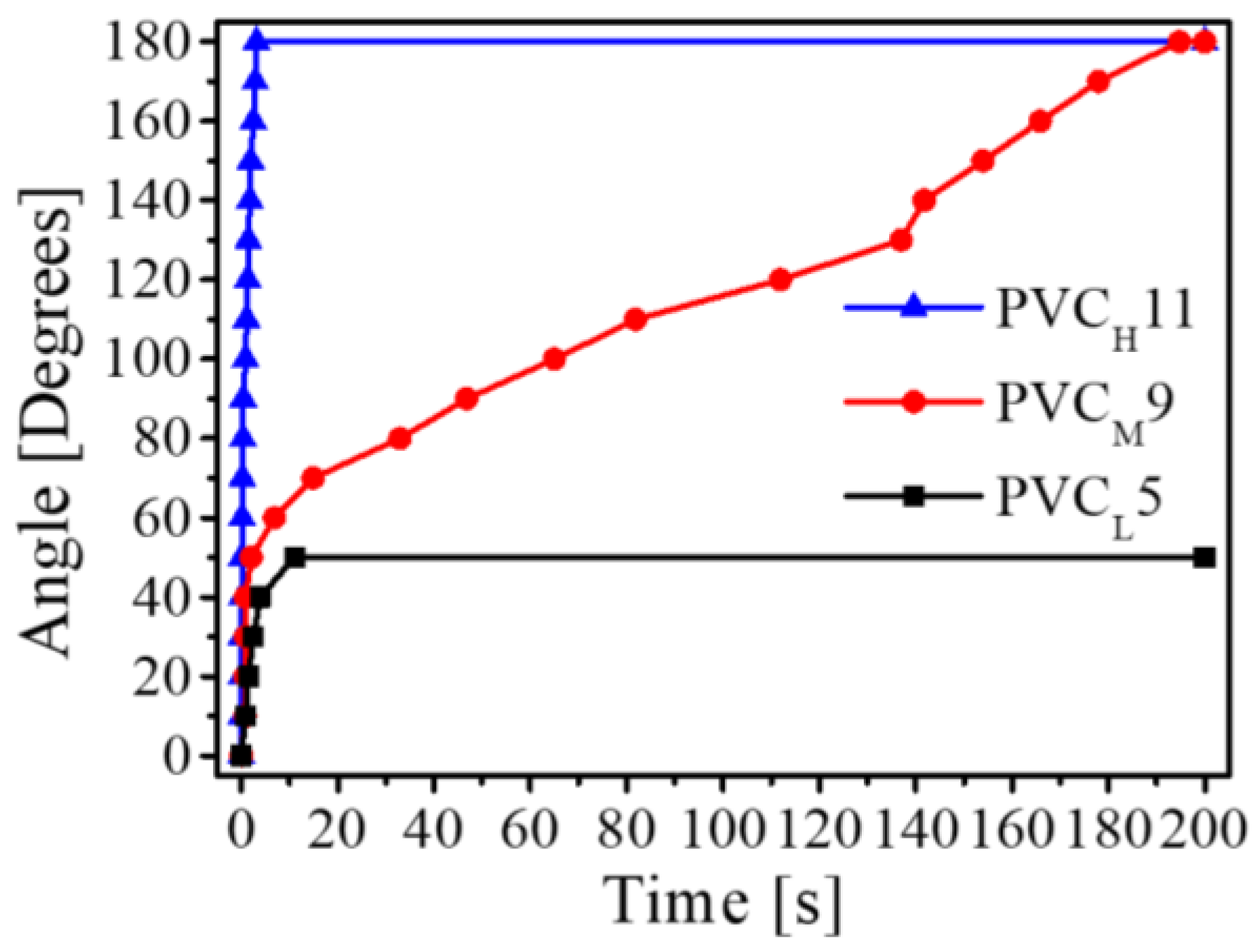
Figure 11 shows the measured bending angles of the three transparent actuators over time. The maximum bending angle of the PVC
L5 gel-based actuator was only about 50°, and its elapsed time was 11 s. On the other hand, the maximum bending angle of the PVC
M9- and PVC
H11-based bending actuators was 180°. However, the elapsed time of the PVC
H11 gel-based bending actuator (3.154 s) was much faster than that of the PVC
M9 gel-based bending actuator (194.79 s).
The transparencies of the PVC
M9 and PVC
H11 gels were measured using UV-vis spectroscopy. As a result, the PVC
H11 gel was found to have transparency >92% at 550 nm (
Figure 12). From this result, the PVC
H11 gel-based bending actuator, which had the largest bending angle and the quickest response time, was selected as the most transparent and highly deformable bending actuator.
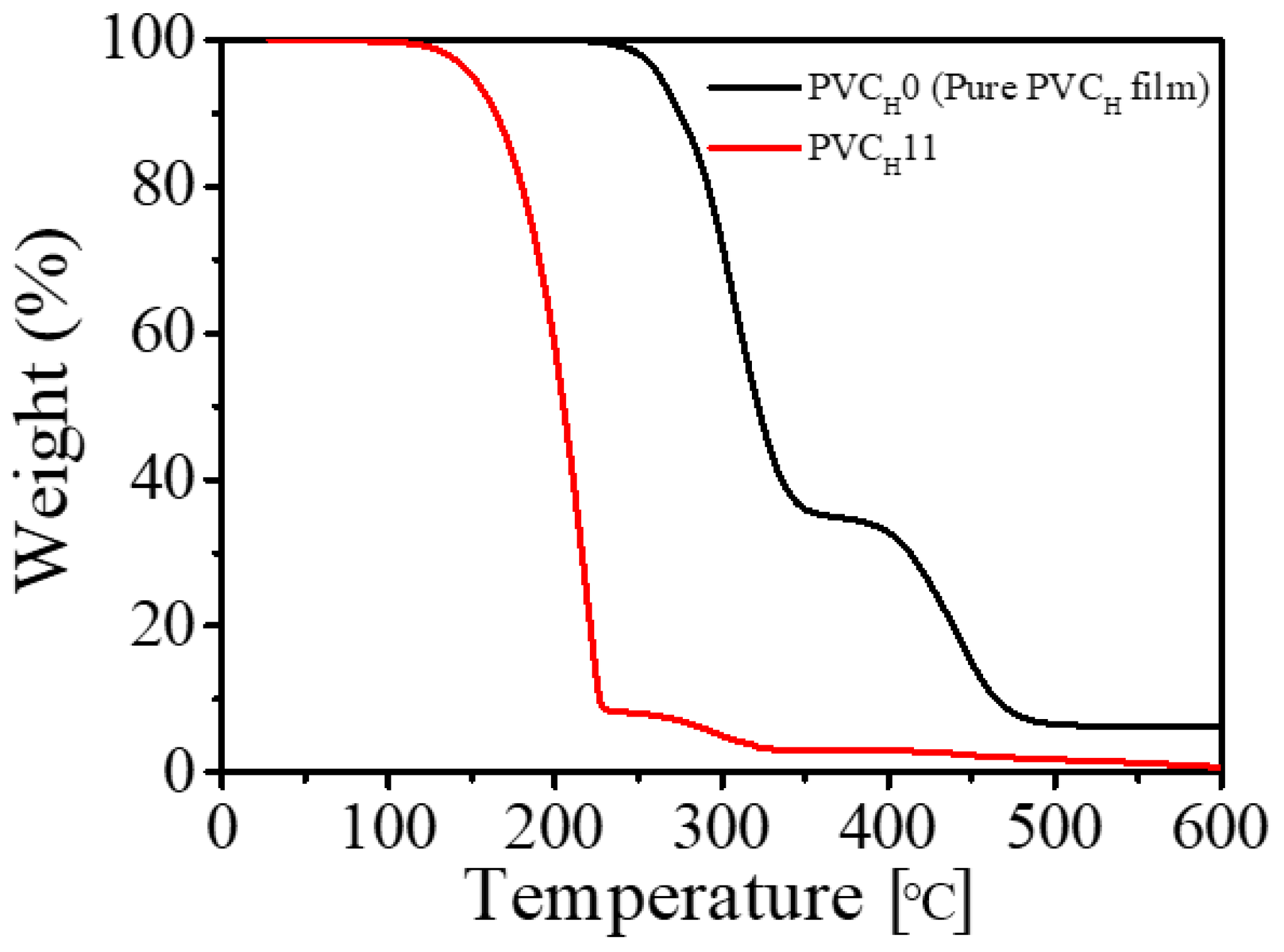
The thermal stabilities of a pure PVC
H film and a PVC
H11 gel were studied by thermogravimetric analysis (TGA) (
Figure 13). The TGA thermogram of a pure PVC
H film showed two degradation steps with onset decomposition at 220 °C. The initial weight-loss step starting at around 220–370 °C was attributed to the dehydrochlorination in the PVC chains, leading to the formation of long sequences of polyenes, after which the sample had lost 65% of its weight. The second degradation step is in the range of 370–475 °C, and can be accounted for by the main PVC chains with conjugated double bonds resulting from dehydrochlorination. However, the plasticized PVC
H11 had slightly reduced thermal stability, and practically, a major steep weight-loss range of 110–320 °C was noticed compared with that of the pure PVC film. This is because the free volume induced by the presence of DBA plasticizer allows a better diffusion of the hydrochloric acid (HCl) gas produced during dehydrochlorination of PVC [
29,
30]. We inserted the two related works in the reference section.
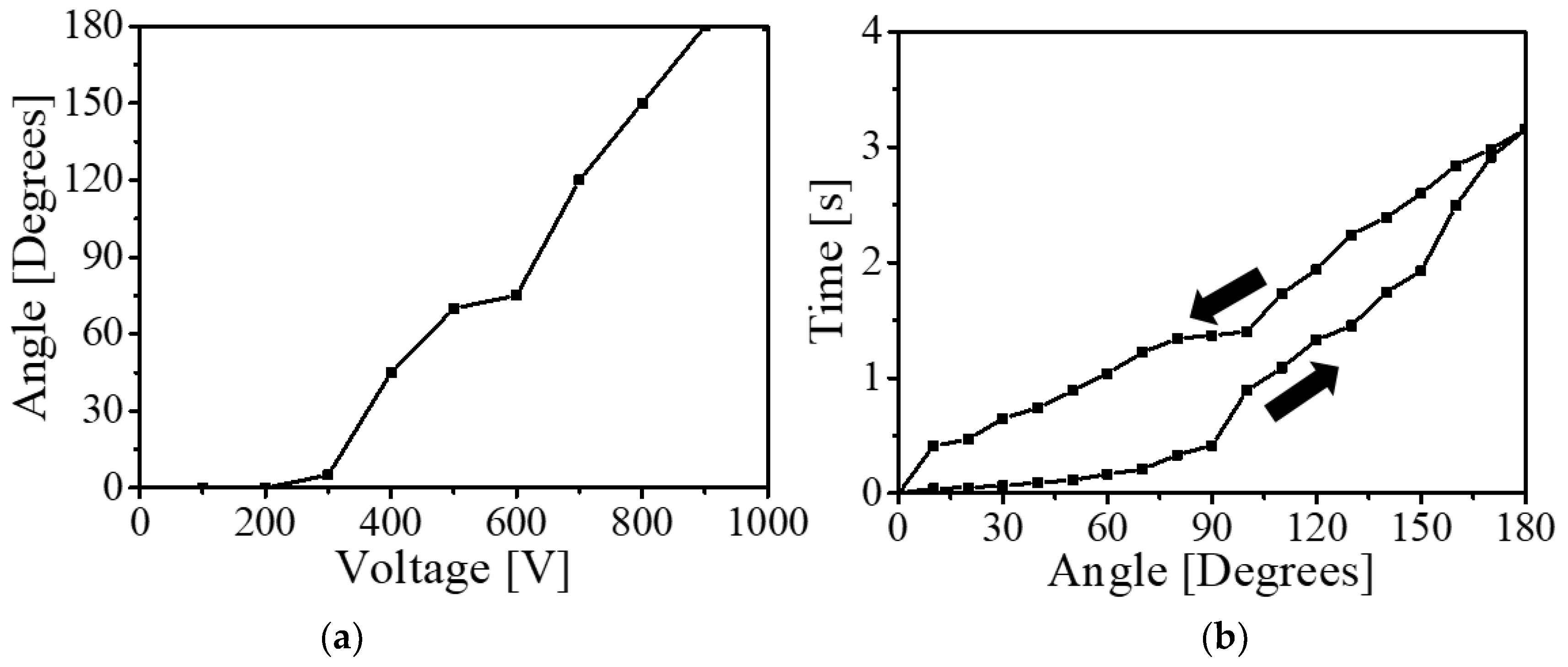
The maximum bending angle of the PVC
H11 gel-based actuator was acquired as a function of input voltage. The proposed PVC
H11 gel-based actuator is hard to bend in lower-voltage input (below 300 V) and it starts to actuate as we increase the input voltage. When the input voltage meets 900 V, the bending angle of the actuator becomes 180° (
Figure 14a). Furthermore, we investigated whether the proposed actuator has hysteresis behavior (
Figure 14b). In this experiment, we applied 1 kV to the PVC
H11 gel-based actuator, and we removed the input voltage as soon as the PVC
H11 gel-based bending actuator reached 180°. The actuator quickly reached 90° within 0.415 s, and the bending angle of the actuator gradually increased until the angle reached 180° (3.154 s). After removing the voltage input, the proposed actuator returned to its initial shape.
To verify the cycling performance of the proposed actuator, square AC input voltage (0 to 1 kV
pp) was applied to the PVC
H11 gel-based bending actuator. The input voltage of 1 kV was applied to the proposed bending actuator over 5 s and then 0 V was provided to the actuator for 5 s. This procedure was conducted 25,000 times (about three days).
Figure 15 shows the results of the proposed actuator’s cycling performance. There is no significant difference in the maximum bending angle of the PVC
H11 gel-based proposed actuator. From the result, the actuation cycles of the actuator were reproducible, indicating that the proposed actuator showed long-term durability. Taking all experimental results together, we found that the PVC
H11 gel-based actuator has the highest transparency and the best performance.