Prostate Clinical Outlook Visualization System for Patients and Clinicians Considering Cyberknife Treatment—A Personalized Approach
Abstract
1. Introduction
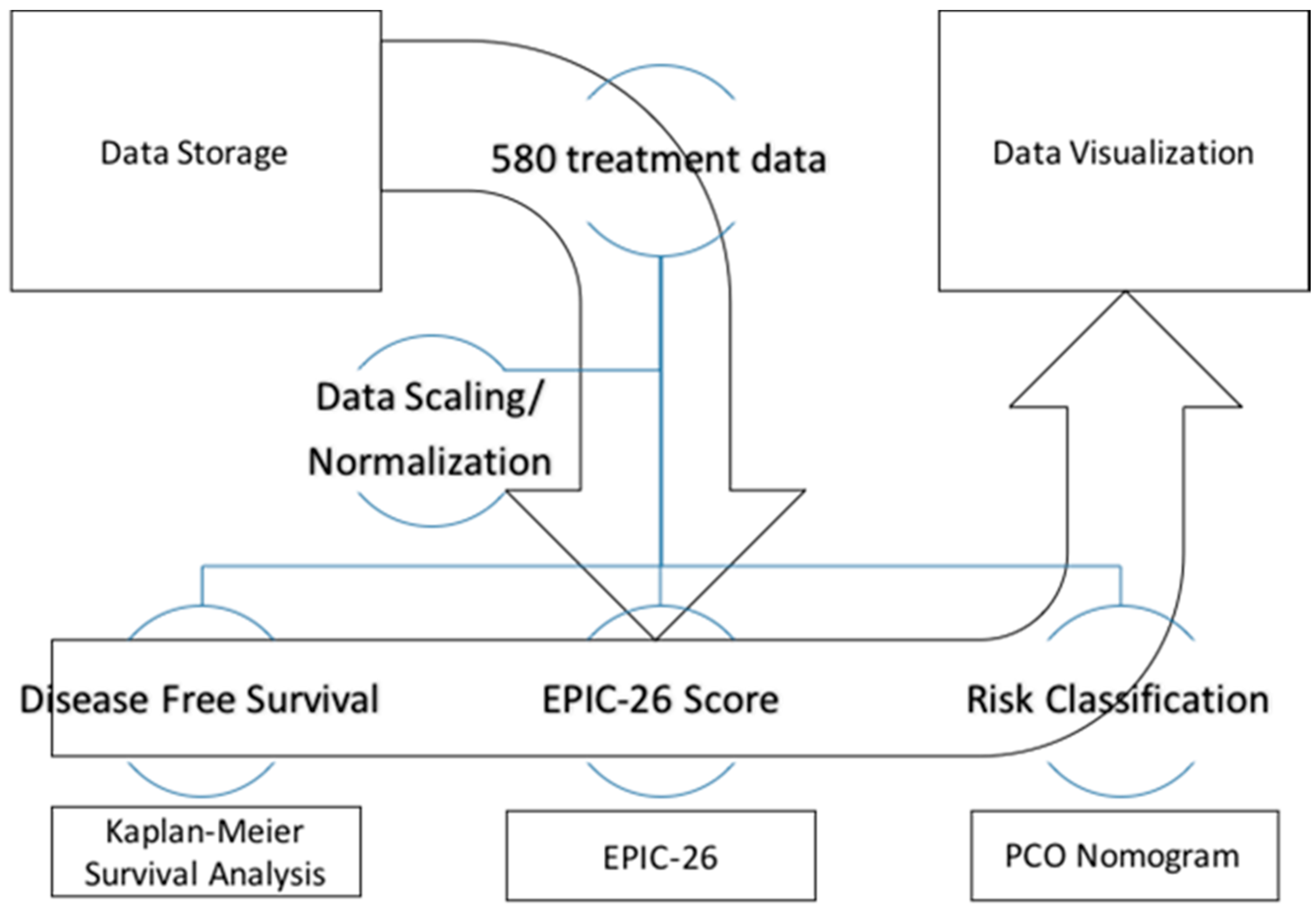
2. Materials and Methods
3. Results
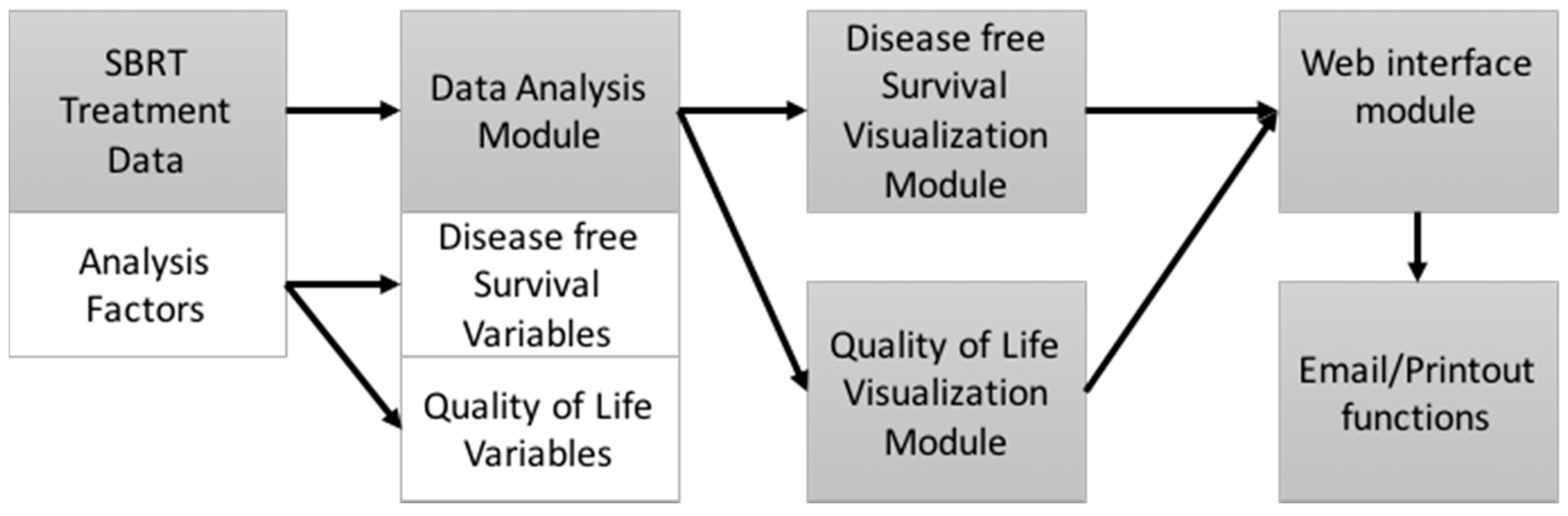
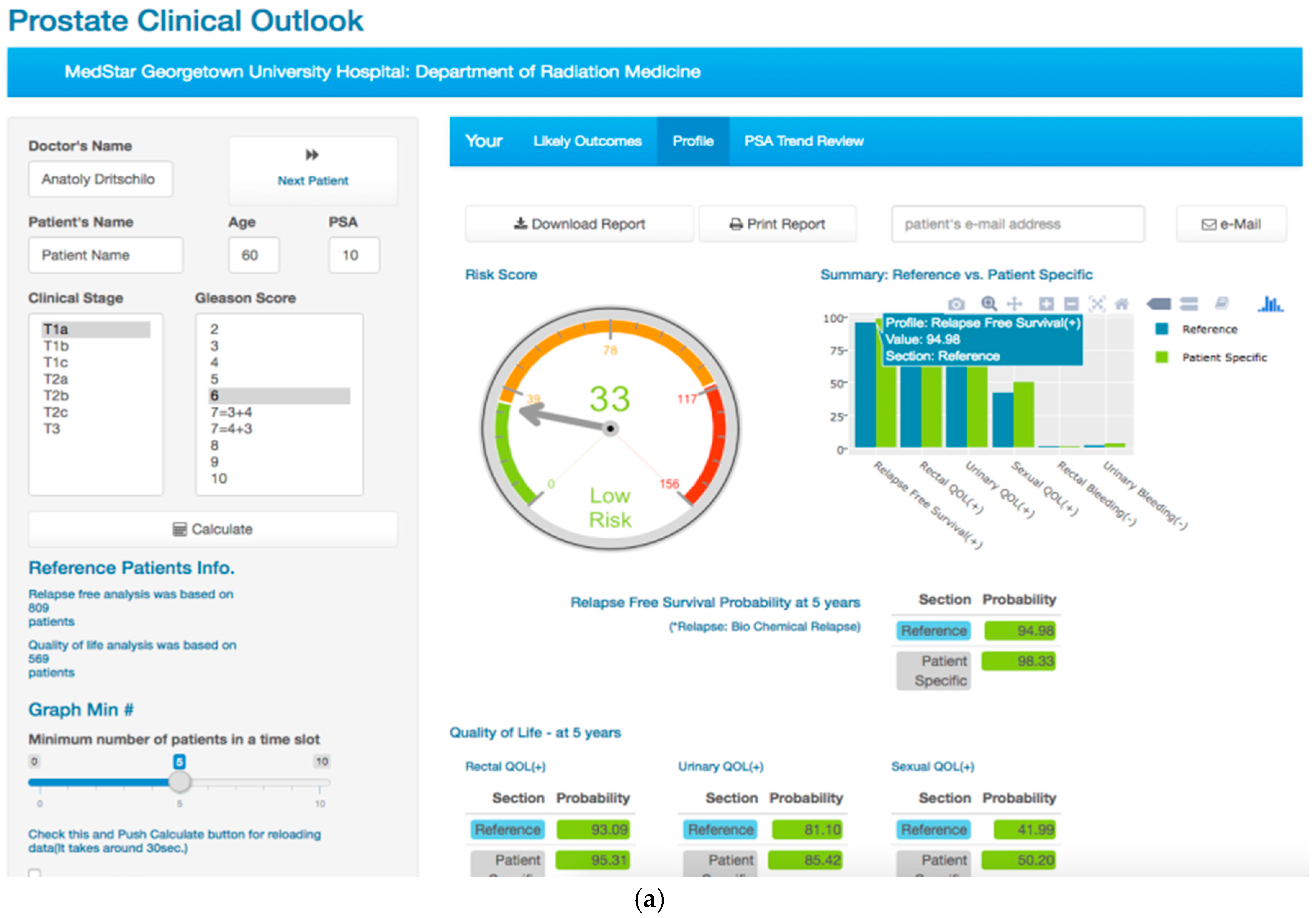
3.1. The Prostate Clinical Outlook Visualization System
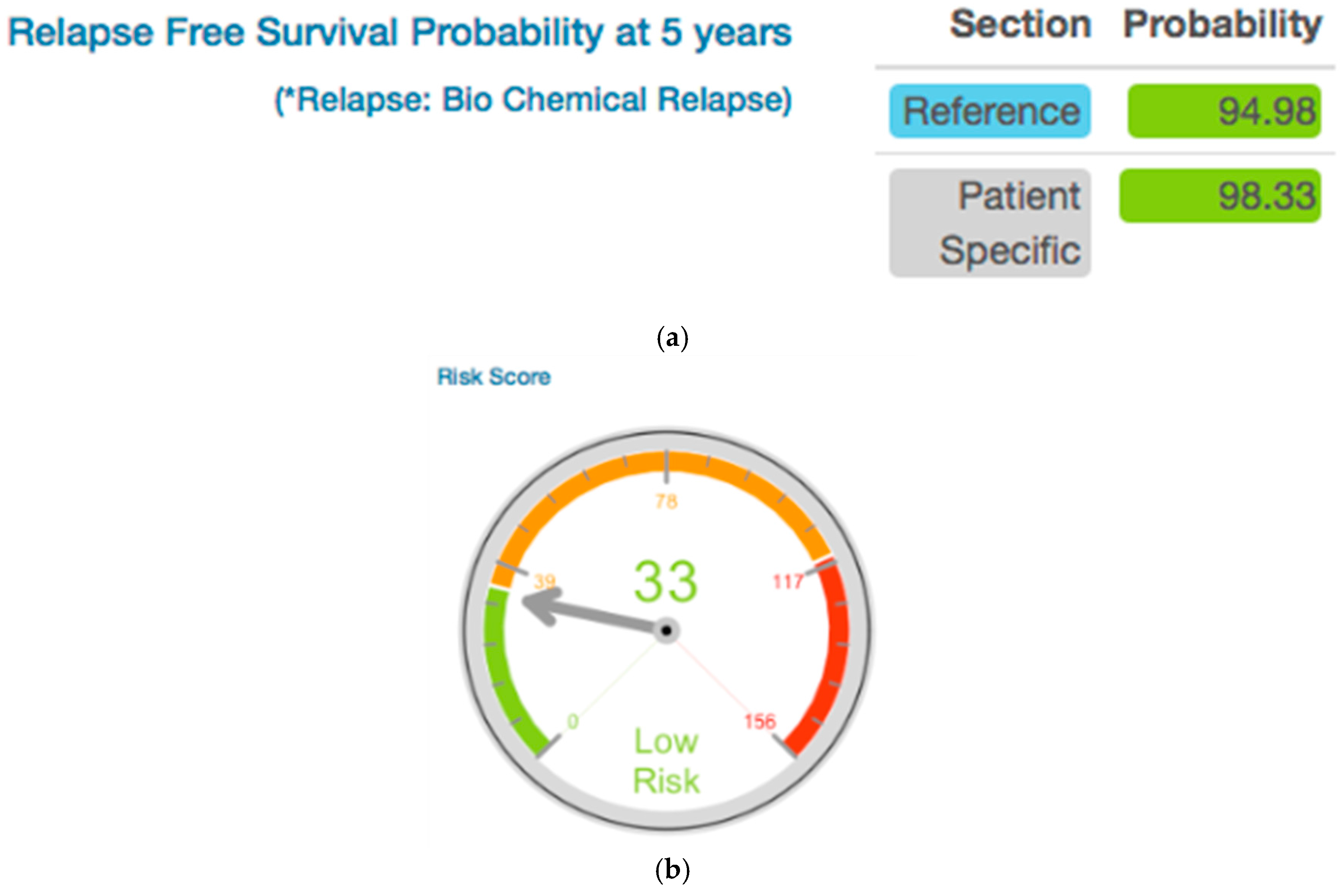
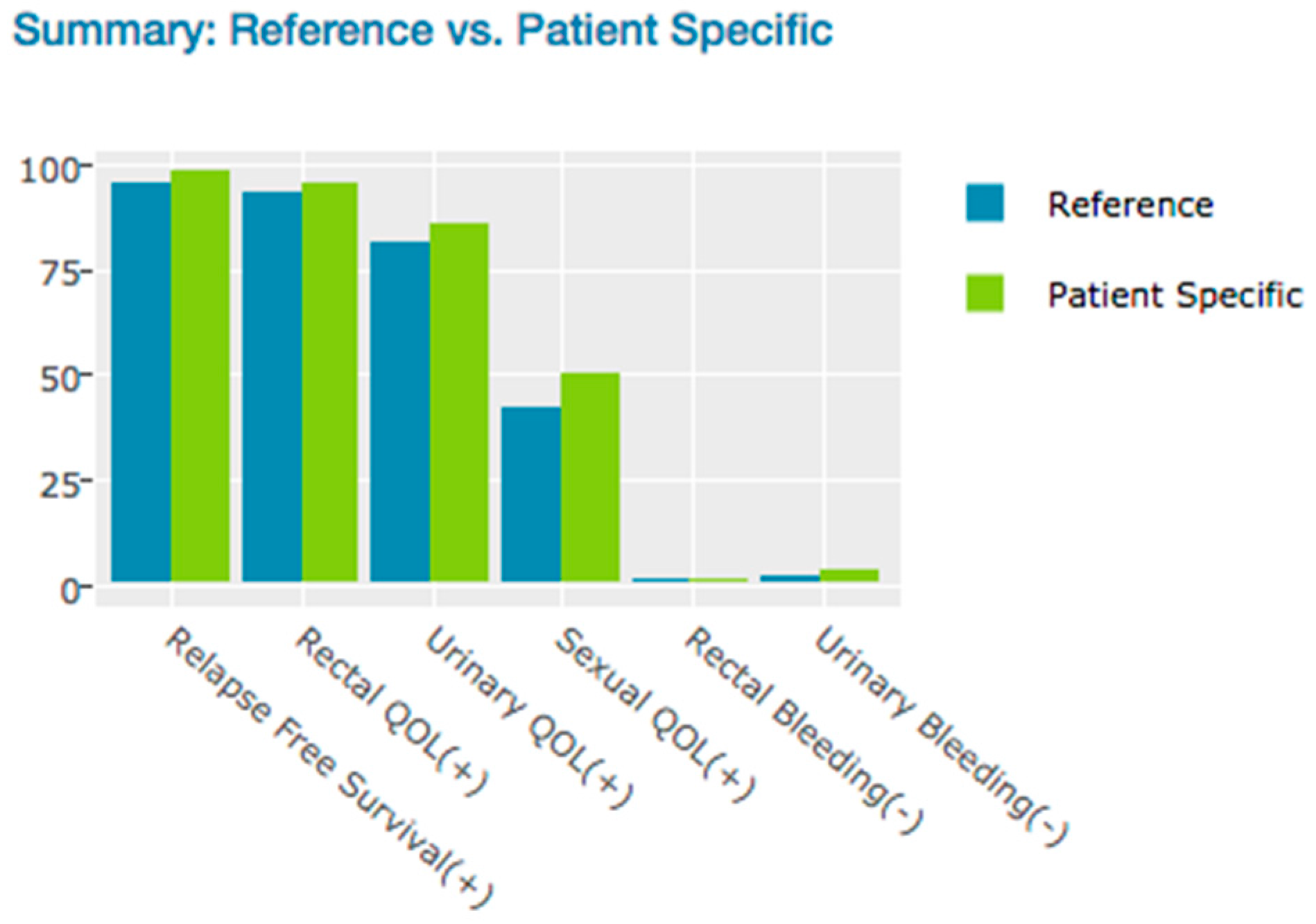
3.1.1. Likely Outcome Visualization (Patient Specific Data vs. Reference Cyberknife Treated Patient Data)
3.1.2. Disease Free Survival Likely Outcome Visualization
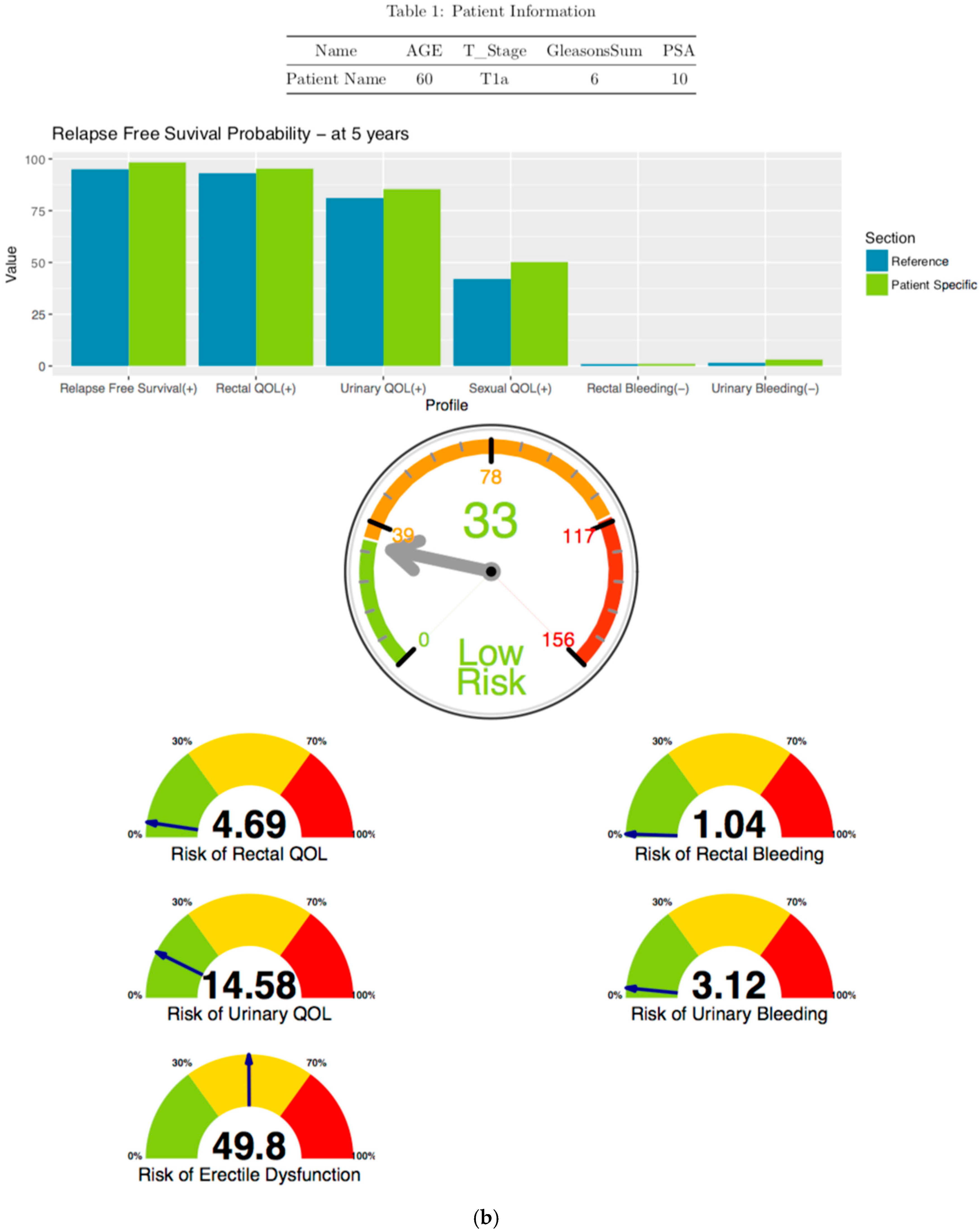
3.1.3. Risk Free Profile Visualization
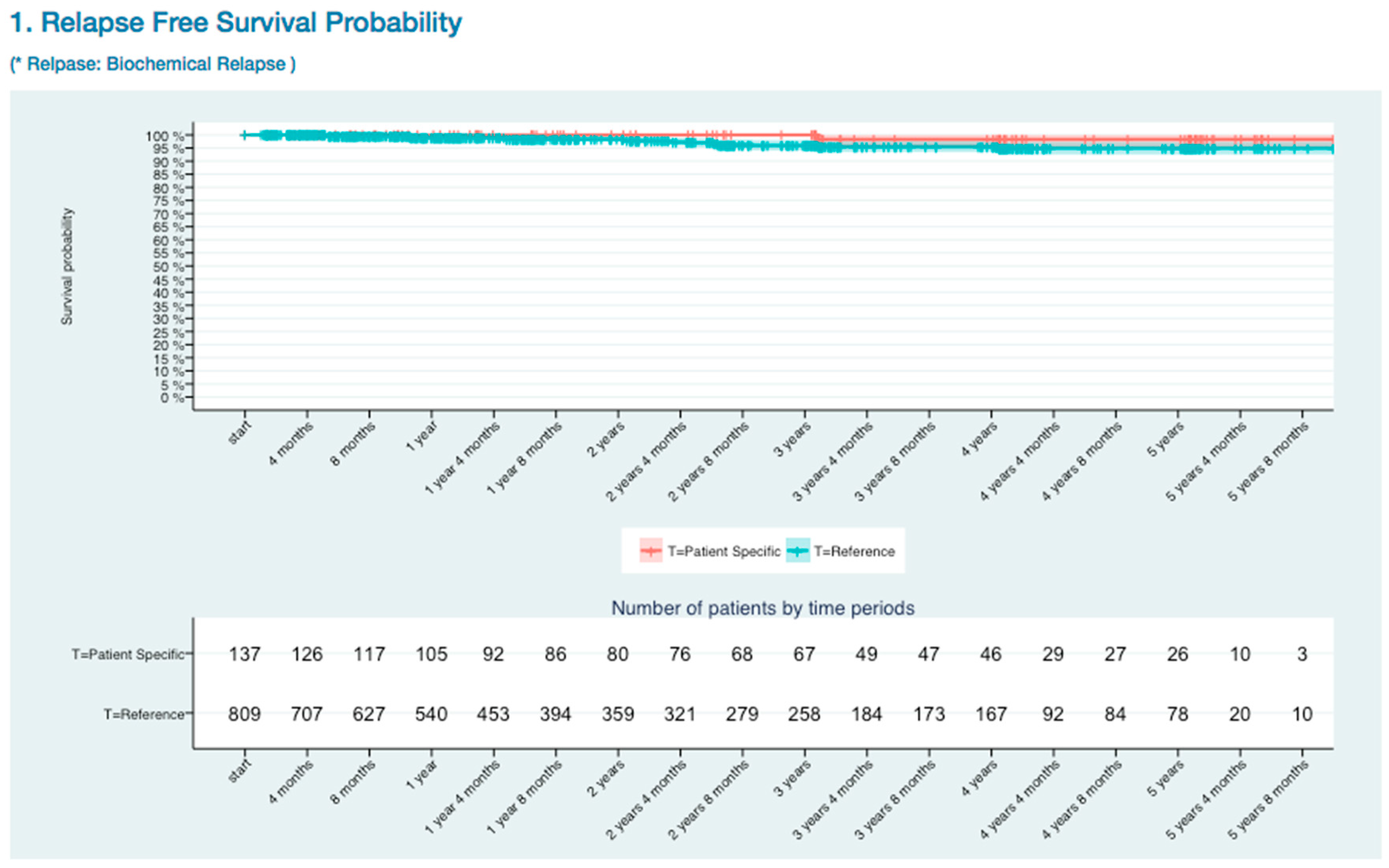
3.2. Longitudinal Trend for Visualization (Patient Specific Data vs. Reference SBRT Treated Patient Data)
3.2.1. BCR Free Survival Curve Visualization
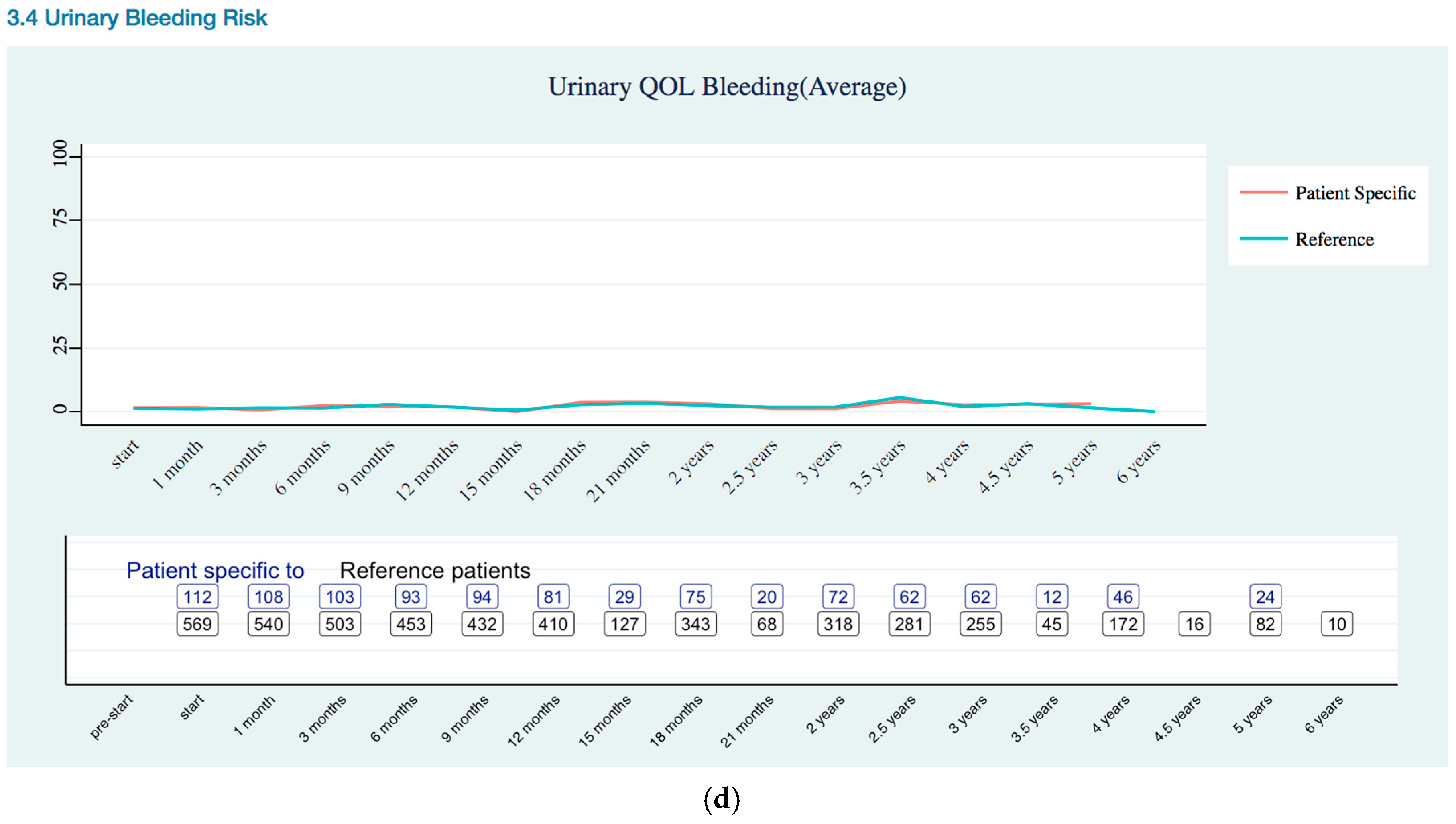
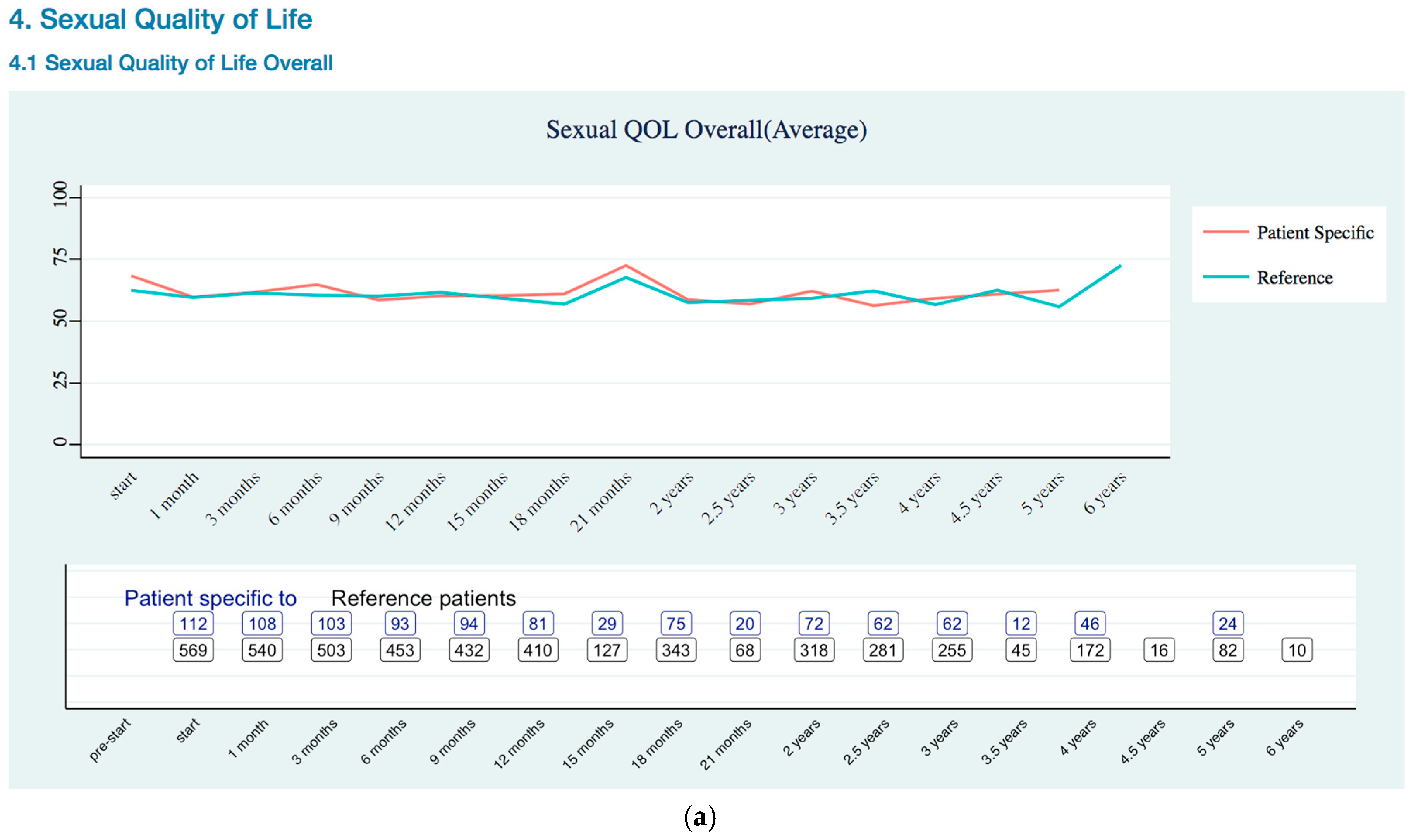
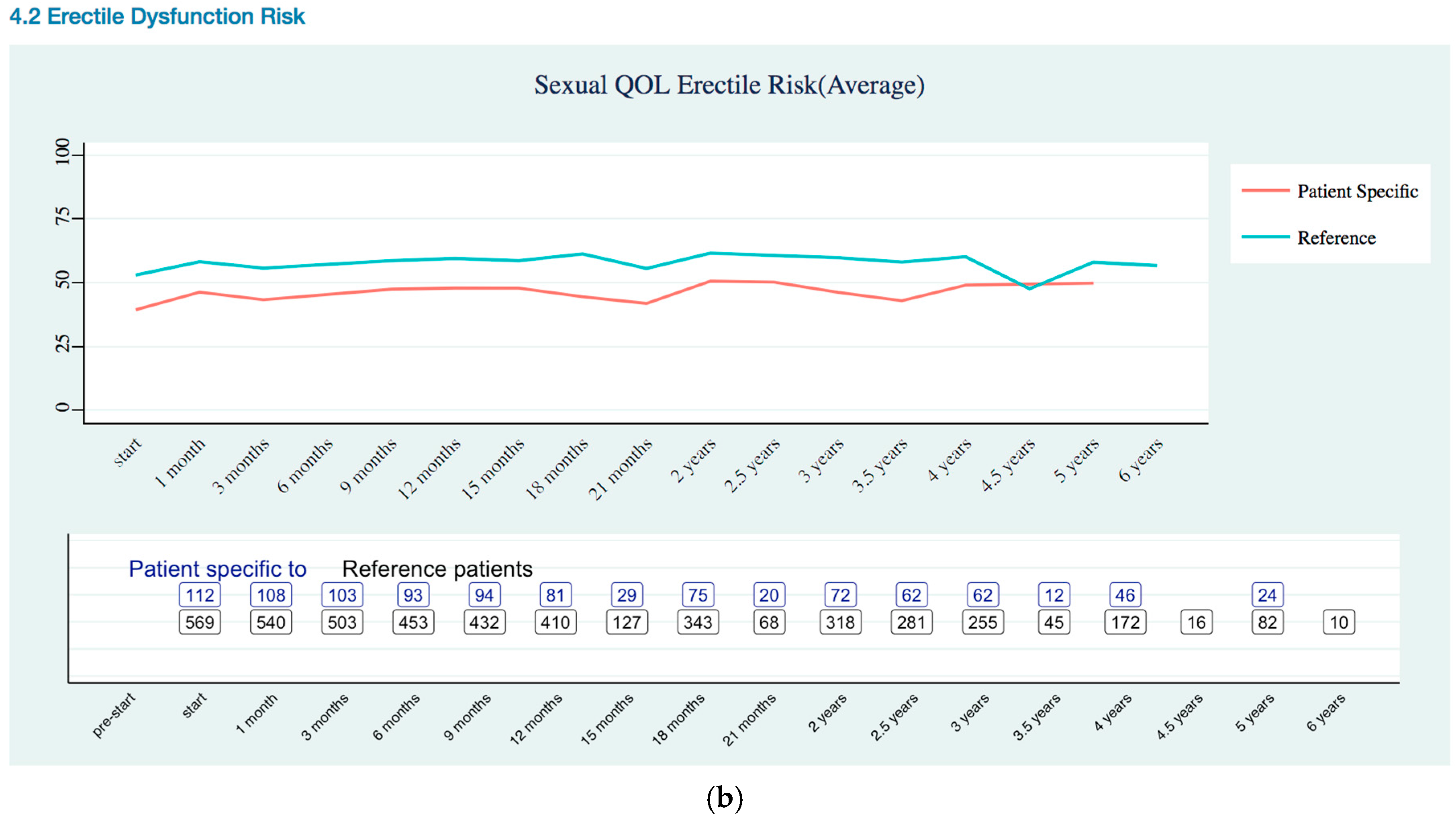
3.2.2. Quality of Life Trends (Rectal, Urinary, and Sexual)
Rectal Quality of Life Trends
Urinary Quality of Life Trends
Sexual Quality of Life Trends
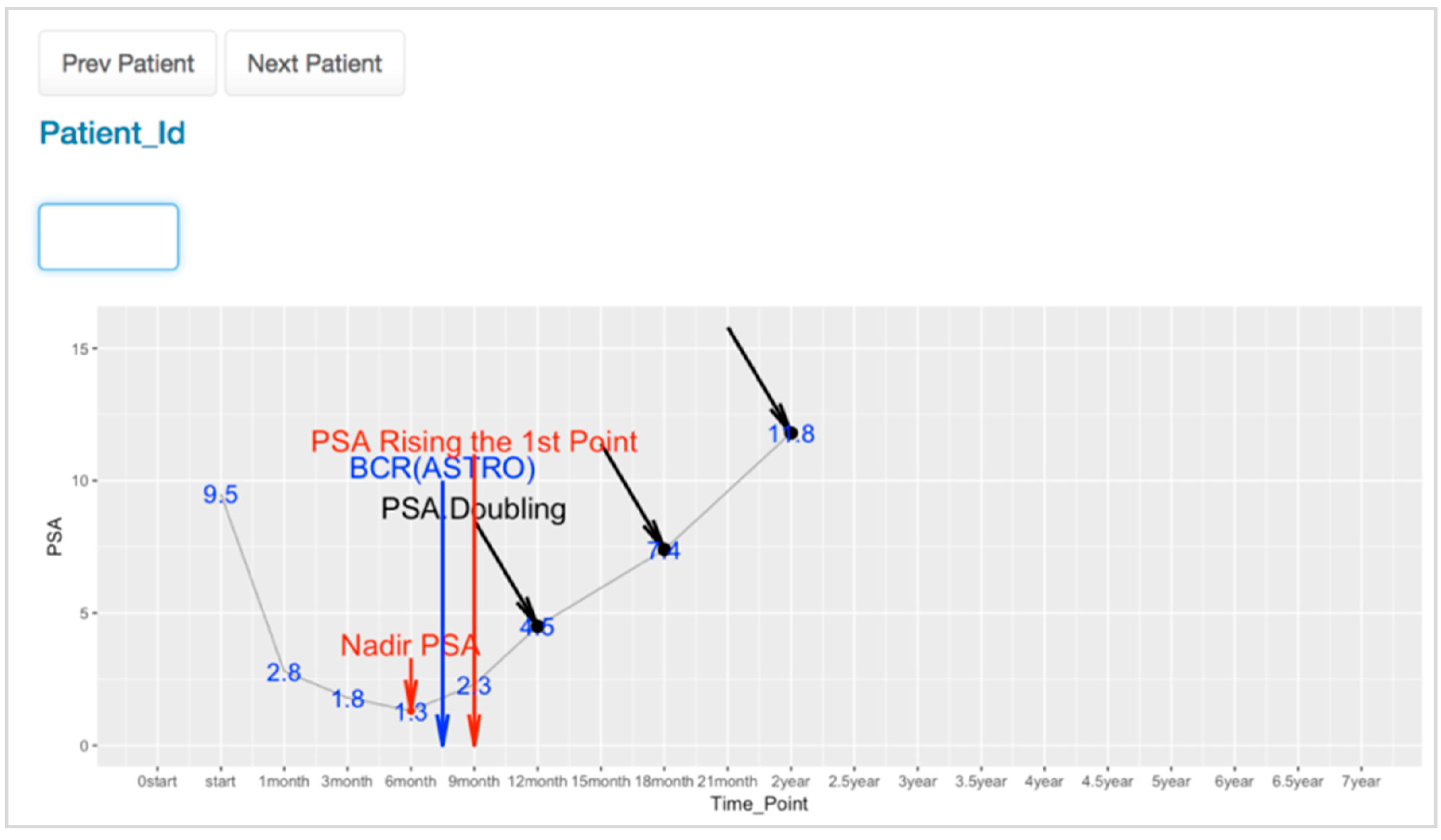
3.3. PSA Trends Visualization for Physician
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Ethical Approval
Appendix A. EPIC 26 Instrument
| No Problem | Very Small Problem | Small Problem | Moderate Problem | Big Problem | |
| a. Dripping or leaking urine | 0 | 1 | 2 | 3 | 4 |
| b. Pain or burning on urination | 0 | 1 | 2 | 3 | 4 |
| c. Bleeding with urination | 0 | 1 | 2 | 3 | 4 |
| d. Weak urine stream or incomplete emptying | 0 | 1 | 2 | 3 | 4 |
| e. Need to urinate frequently during the day | 0 | 1 | 2 | 3 | 4 |
| No Problem | Very Small Problem | Small Problem | Moderate Problem | Big Problem | |
| a. Urgency to have a bowel movement | 0 | 1 | 2 | 3 | 4 |
| b. Increased frequency of bowel movements | 0 | 1 | 2 | 3 | 4 |
| c. Losing control of your stools | 0 | 1 | 2 | 3 | 4 |
| d. Bloody stools | 0 | 1 | 2 | 3 | 4 |
| e. Abdominal/ Pelvic/Rectal pain | 0 | 1 | 2 | 3 | 4 |
| Very Poor to None | Poor | Fair | Good | Very Good | |
| Your ability to have an erection? | 1 | 2 | 3 | 4 | 5 |
| Your ability to reach orgasm (climax)? | 1 | 2 | 3 | 4 | 5 |
| No Problem | Very Small Problem | Small Problem | Moderate Problem | Big Problem | |
| a. Hot flashes | 0 | 1 | 2 | 3 | 4 |
| b. Breast tenderness/enlargement | 0 | 1 | 2 | 3 | 4 |
| c. Feeling depressed | 0 | 1 | 2 | 3 | 4 |
| d. Lack of energy | 0 | 1 | 2 | 3 | 4 |
| e. Change in body weight | 0 | 1 | 2 | 3 | 4 |
Appendix B. Quality of Life Trends (Rectal, Urinary, and Sexual)
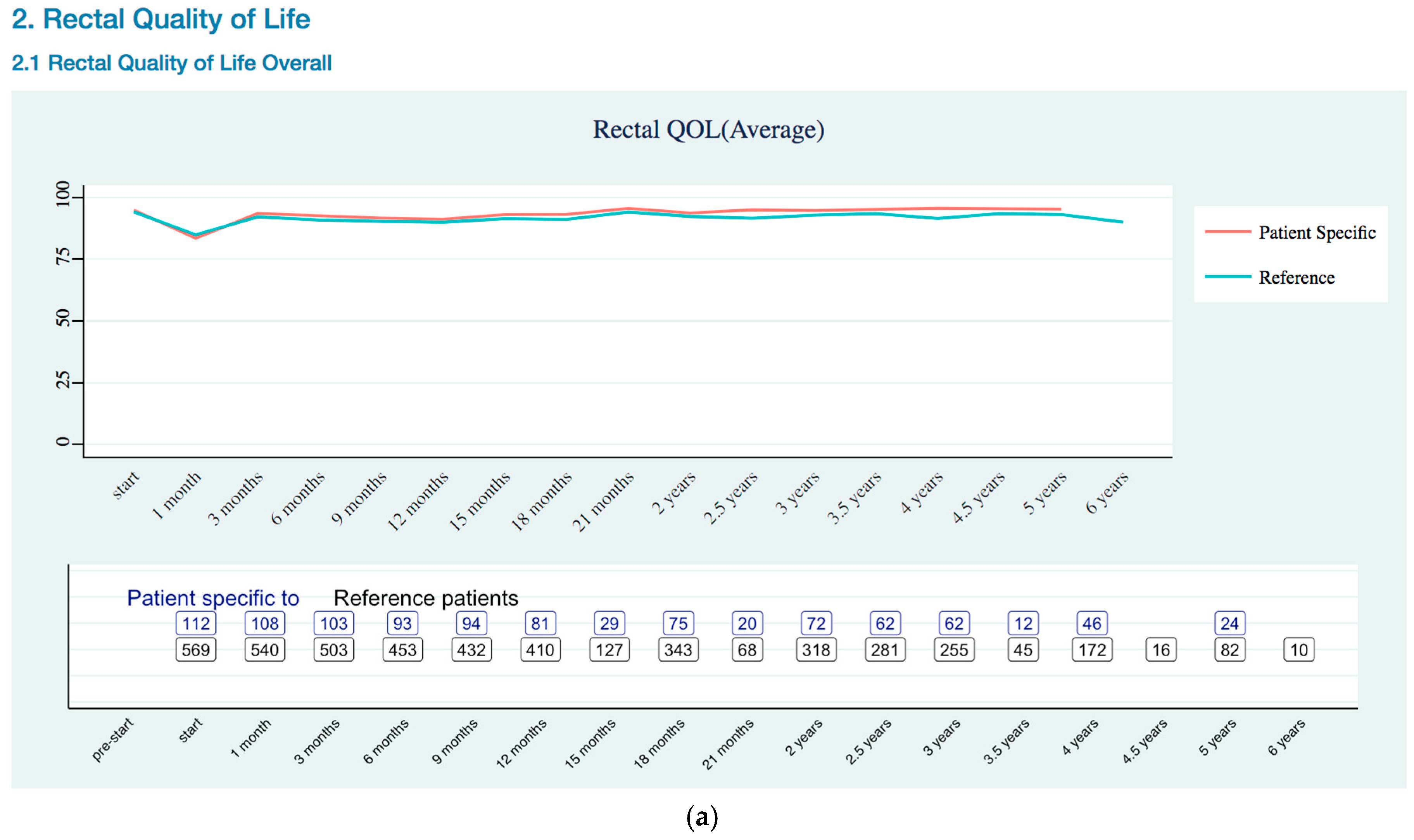
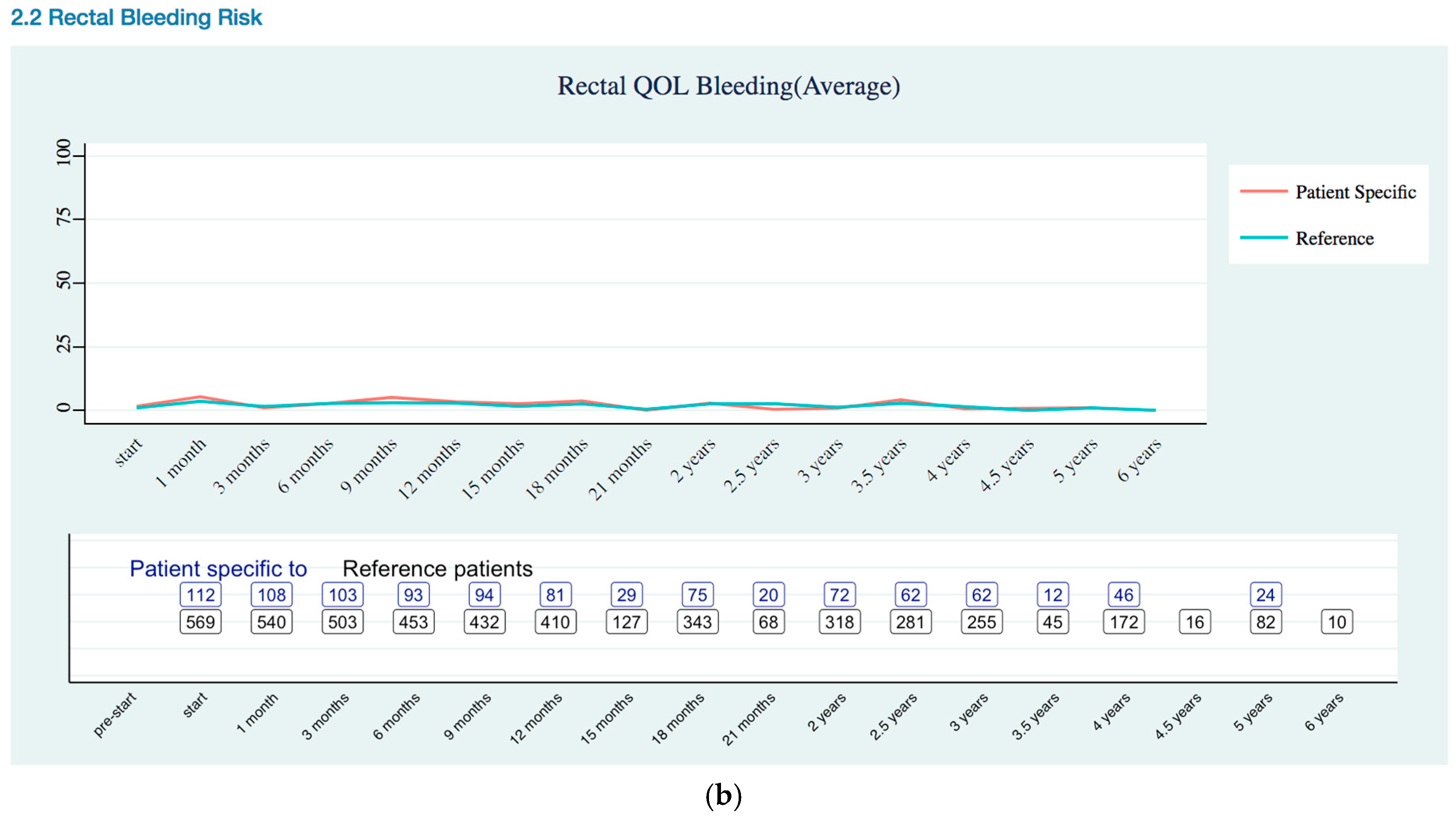
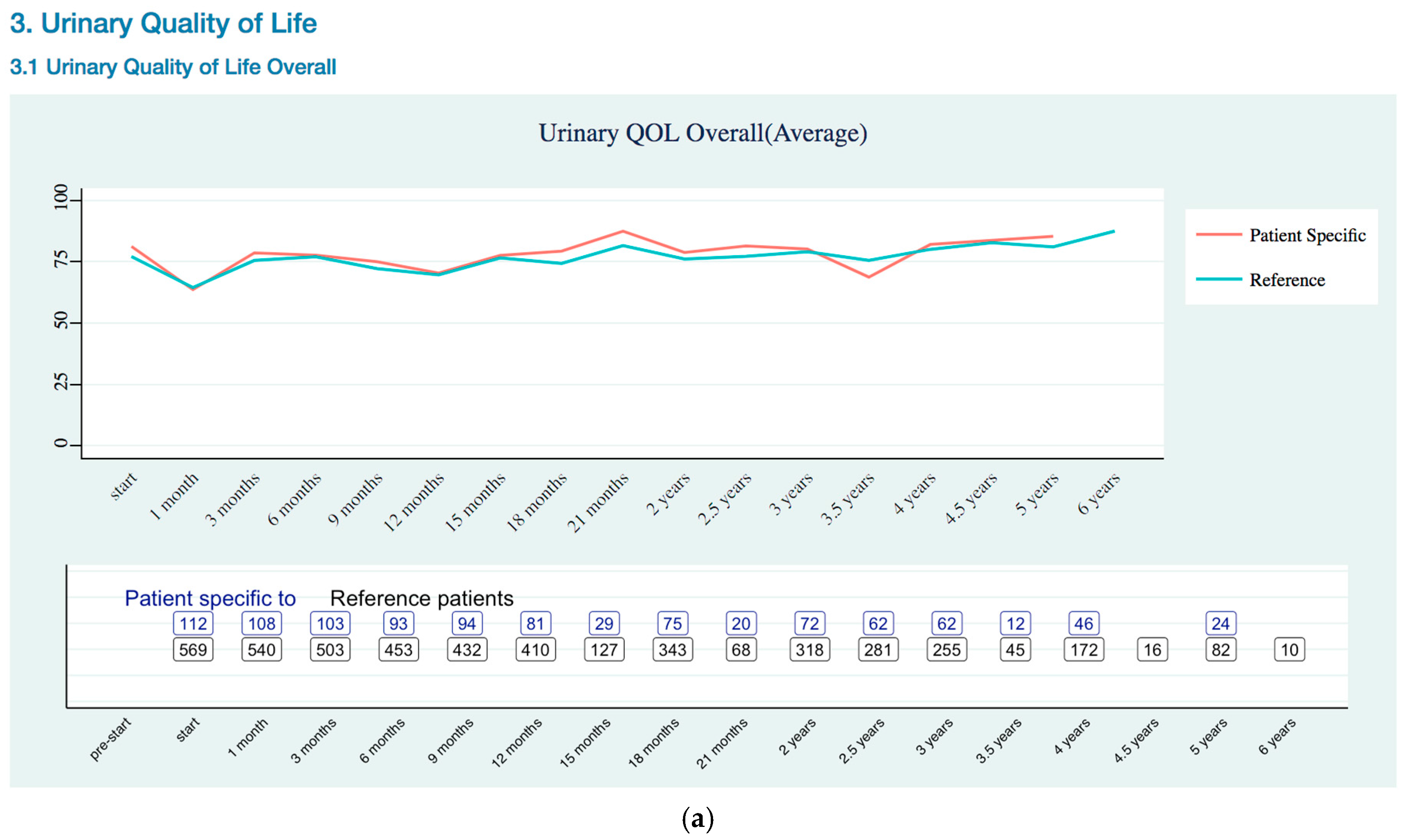
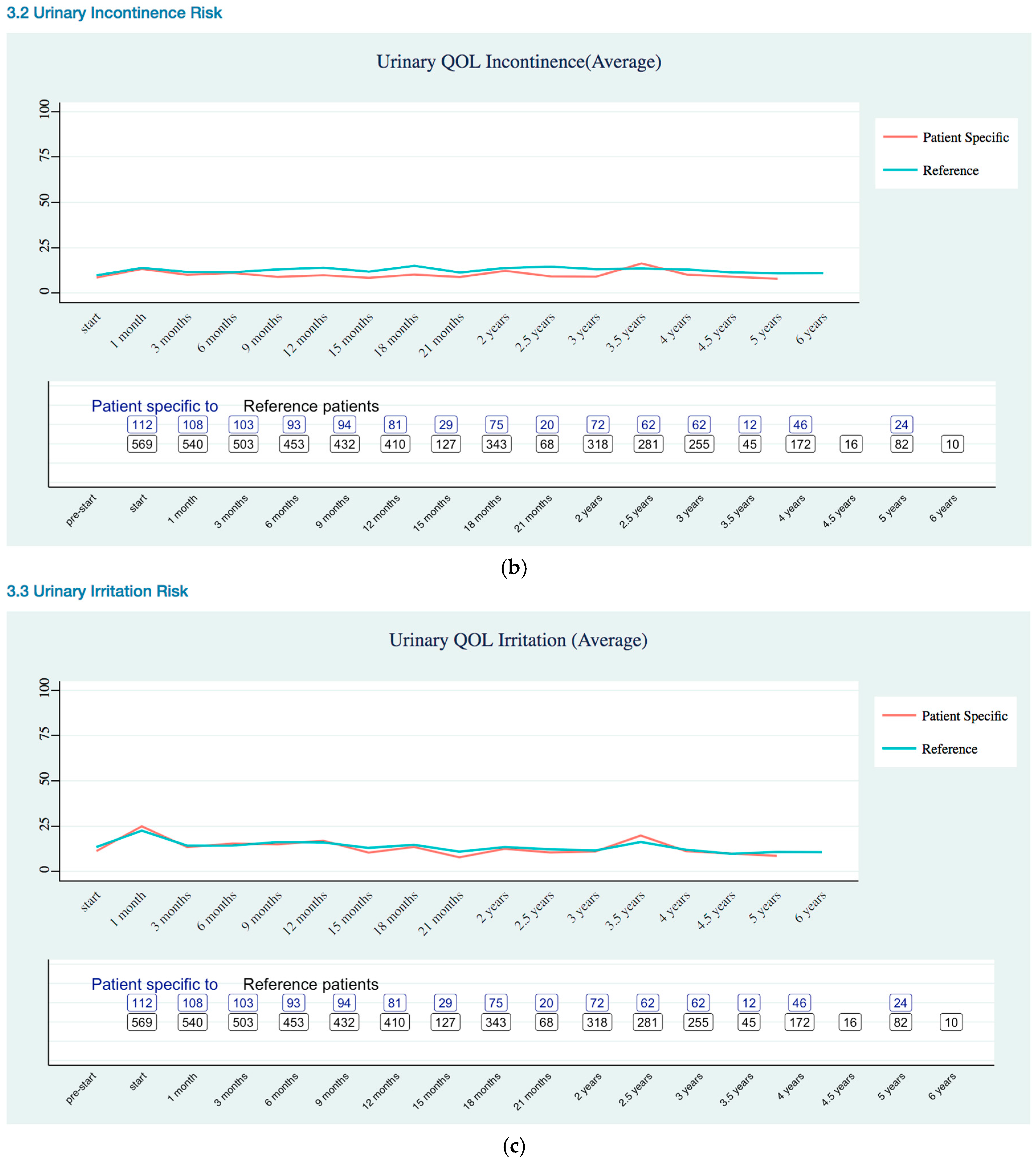
References
- Walsh, P.C.; DeWeese, T.L.; Eisenberger, M.A. Localized prostate cancer. N. Engl. J. Med. 2007, 357, 2696–2705. [Google Scholar] [CrossRef] [PubMed]
- Gigerenzer, G.; Gaissmaier, W.; Kurz-Milcke, E.; Schwartz, L.M.; Woloshin, S. Helping doctors and patients make sense of health statistics. Psychol. Sci. Public Interest 2007, 8, 53–96. [Google Scholar] [CrossRef] [PubMed]
- Coiera, E. Clinical decision support systems. In Guide to Health Informatics; CRC Press: Boca Rato, FL, USA, 2003; Volume 2. [Google Scholar]
- Chen, L.N.; Suy, S.; Uhm, S.; Oermann, E.K.; Ju, A.W.; Chen, V.; Hanscom, H.N.; Laing, S.; Kim, J.S.; Lei, S. Stereotactic body radiation therapy (sbrt) for clinically localized prostate cancer: The georgetown university experience. Radiat. Oncol. 2013, 8, 58. [Google Scholar] [CrossRef] [PubMed]
- Lakshmanan, K.; Peter, A.P.; Mohandass, S.; Varadharaj, S.; Lakshmanan, U.; Dharmar, P. Synrio: R and shiny based application platform for cyanobacterial genome analysis. Bioinformation 2015, 11, 422. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Walter, W.; Sánchez-Cabo, F.; Ricote, M. Goplot: An R package for visually combining expression data with functional analysis. Bioinformatics 2015, 31, 2912–2914. [Google Scholar] [CrossRef] [PubMed]
- Badgeley, M.A.; Shameer, K.; Glicksberg, B.S.; Tomlinson, M.S.; Levin, M.A.; McCormick, P.J.; Kasarskis, A.; Reich, D.L.; Dudley, J.T. Ehdviz: Clinical dashboard development using open-source technologies. BMJ Open 2016, 6, e010579. [Google Scholar] [CrossRef] [PubMed]
- Hinterberg, M.A.; Kao, D.P.; Bristow, M.R.; Hunter, L.E.; Port, J.D.; Görg, C. Peax: Interactive visual analysis and exploration of complex clinical phenotype and gene expression association Pacific Symposium on Biocomputing. In Proceedings of the Pacific Symposium on Biocomputing, Puako, HI, USA, 4–8 January 2015; pp. 419–430. [Google Scholar]
- Cookson, M.S.; Aus, G.; Burnett, A.L.; Canby-Hagino, E.D.; D’Amico, A.V.; Dmochowski, R.R.; Eton, D.T.; Forman, J.D.; Goldenberg, S.L.; Hernandez, J. Variation in the definition of biochemical recurrence in patients treated for localized prostate cancer: The american urological association prostate guidelines for localized prostate cancer update panel report and recommendations for a standard in the reporting of surgical outcomes. J. Urol. 2007, 177, 540–545. [Google Scholar] [PubMed]
- Katz, A.J. Cyberknife radiosurgery for prostate cancer. Technol. Cancer Res. Treat. 2010, 9, 463–472. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, E.L.; Meier, P. Nonparametric estimation from incomplete observations. J. Am. Stat. Assoc. 1958, 53, 457–481. [Google Scholar] [CrossRef]
- Capanu, M.; Gonen, M. Building a Nomogram for Survey-Weighted Cox Models Using R; Department of Epidemiology and Biostatistics: New York, NY, USA, 2011. [Google Scholar]
- Wei, J.T.; Dunn, R.L.; Litwin, M.S.; Sandler, H.M.; Sanda, M.G. Development and validation of the expanded prostate cancer index composite (epic) for comprehensive assessment of health-related quality of life in men with prostate cancer. Urology 2000, 56, 899–905. [Google Scholar] [CrossRef]
- Szymanski, K.M.; Wei, J.T.; Dunn, R.L.; Sanda, M.G. Development and validation of an abbreviated version of the expanded prostate cancer index composite instrument for measuring health-related quality of life among prostate cancer survivors. Urology 2010, 76, 1245–1250. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.Y.; Daignault-Newton, S.; Heath, G.; Scarlett, S.; Sanda, M.G.; Chang, P.; Regan, M.M.; Michalski, J.M.; Sandler, H.M.; Feng, F.Y. Multinational prospective study of patient-reported outcomes after prostate radiation therapy: Detailed assessment of rectal bleeding. Int. J. Radiat. Oncol. Biol. Phys. 2016, 96, 770–777. [Google Scholar] [CrossRef] [PubMed]
- Bhattasali, O.; Chen, L.N.; Woo, J.; Park, J.-W.; Kim, J.S.; Moures, R.; Yung, T.; Lei, S.; Collins, B.T.; Kowalczyk, K. Patient-reported outcomes following stereotactic body radiation therapy for clinically localized prostate cancer. Radiat. Oncol. 2014, 9, 1. [Google Scholar] [CrossRef] [PubMed]
- Arscott, W.T.; Chen, L.N.; Wilson, N.; Bhagat, A.; Kim, J.S.; Moures, R.A.; Yung, T.M.; Lei, S.; Collins, B.T.; Kowalczyk, K.; et al. Obstructive voiding symptoms following stereotactic body radiation therapy for prostate cancer. Radiat. Oncol. 2014, 9, 163. [Google Scholar] [CrossRef] [PubMed]
- Joh, D.Y.; Chen, L.N.; Porter, G.; Bhagat, A.; Sood, S.; Kim, J.S.; Moures, R.; Yung, T.; Lei, S.; Collins, B.T. Proctitis following stereotactic body radiation therapy for prostate cancer. Radiat. Oncol. 2014, 9, 1. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.N.; Suy, S.; Wang, H.; Bhagat, A.; Woo, J.A.; Moures, R.A.; Kim, J.S.; Yung, T.M.; Lei, S.; Collins, B.T. Patient-reported urinary incontinence following stereotactic body radiation therapy (sbrt) for clinically localized prostate cancer. Radiat. Oncol. 2014, 9, 1. [Google Scholar] [CrossRef] [PubMed]
- Obayomi-Davies, O.; Chen, L.N.; Bhagat, A.; Wright, H.C.; Uhm, S.; Kim, J.S.; Yung, T.M.; Lei, S.; Batipps, G.P.; Pahira, J. Potency preservation following stereotactic body radiation therapy for prostate cancer. Radiat. Oncol. 2013, 8, 1. [Google Scholar] [CrossRef]
- Kattan, M.W.; Zelefsky, M.J.; Kupelian, P.A.; Scardino, P.T.; Fuks, Z.; Leibel, S.A. Pretreatment nomogram for predicting the outcome of three-dimensional conformal radiotherapy in prostate cancer. J. Clin. Oncol. 2000, 18, 3352–3359. [Google Scholar] [CrossRef] [PubMed]
- Taylor, K.L.; Williams, R.M.; Davis, K.; Luta, G.; Penek, S.; Barry, S.; Kelly, S.; Tomko, C.; Schwartz, M.; Krist, A.H.; et al. Decision making in prostate cancer screening using decision aids vs usual care: A randomized clinical trial. JAMA Int. Med. 2013, 173, 1704–1712. [Google Scholar] [CrossRef] [PubMed]
- Tomko, C.; Davis, K.M.; Luta, G.; Krist, A.H.; Woolf, S.H.; Taylor, K.L. A comparison of web-based versus print-based decision aids for prostate cancer screening: Participants’ evaluation and utilization. J. Gen. Int. Med. 2015, 30, 33–42. [Google Scholar] [CrossRef] [PubMed]
- Prostate Clinical Outlook Visualization System. Available online: http://code.osehra.org/journal/journal/view/1003 (accessed on 28 February 2018).
- Goertzel, G. Clinical decision support system. Ann. N. Y. Acad. Sci. 1969, 161, 689–693. [Google Scholar] [CrossRef] [PubMed]
| Category | Frequency | Percent | |
|---|---|---|---|
| Age at diagnosis (Mean, Range) | Under 59 | 48 | 8.3 |
| 60–69 | 230 | 39.7 | |
| 70–79 | 247 | 42.6 | |
| Over 80 | 55 | 9.5 | |
| 69.63 (44–94) | |||
| T-stage | T1(T1b, T1c) | 396 | 68.3 |
| T2a | 92 | 15.9 | |
| T2b | 64 | 11 | |
| T2c+T3 | 28 | 4.8 | |
| Gleason score | Low | 236 | 40.7 |
| Intermediate-a (3 + 4) | 210 | 36.2 | |
| Intermediate-b (4 + 3) | 90 | 15.5 | |
| High | 44 | 7.6 | |
| Pre-treatment prostate specific antigen (PSA) | ≤10.0 ng/mL | 446 | 76.9 |
| >10.0 and ≤20.0 ng/mL | 105 | 18.1 | |
| >20.0 ng/mL | 29 | 5.0 | |
| Race | Asian | 21 | 3.6 |
| Black | 226 | 39 | |
| Hispanic | 13 | 2.2 | |
| White | 308 | 53.1 | |
| Other | 12 | 2.1 | |
| BCR | Patients with BCR | 17 | 2.9 |
| Patients with non-BCR | 563 | 97.1 | |
| Total | 580 | 100 | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Park, J.; Rho, M.J.; Dritschilo, A.; Choi, I.Y.; Mun, S.K. Prostate Clinical Outlook Visualization System for Patients and Clinicians Considering Cyberknife Treatment—A Personalized Approach. Appl. Sci. 2018, 8, 471. https://doi.org/10.3390/app8030471
Park J, Rho MJ, Dritschilo A, Choi IY, Mun SK. Prostate Clinical Outlook Visualization System for Patients and Clinicians Considering Cyberknife Treatment—A Personalized Approach. Applied Sciences. 2018; 8(3):471. https://doi.org/10.3390/app8030471
Chicago/Turabian StylePark, Jihwan, Mi Jung Rho, Anatoly Dritschilo, In Young Choi, and Seong K. Mun. 2018. "Prostate Clinical Outlook Visualization System for Patients and Clinicians Considering Cyberknife Treatment—A Personalized Approach" Applied Sciences 8, no. 3: 471. https://doi.org/10.3390/app8030471
APA StylePark, J., Rho, M. J., Dritschilo, A., Choi, I. Y., & Mun, S. K. (2018). Prostate Clinical Outlook Visualization System for Patients and Clinicians Considering Cyberknife Treatment—A Personalized Approach. Applied Sciences, 8(3), 471. https://doi.org/10.3390/app8030471





