Nanomaterials for the Abatement of Pharmaceuticals and Personal Care Products from Wastewater
Abstract
1. Introduction
2. Source, Fate and Effects of Selected PPCs
3. Removal of PPCPs by Conservative Methods Envisaging the Use of Nanomaterials
4. Removal of PPCPs by Non-Conservative Methods Envisaging the Use of Nanomaterials
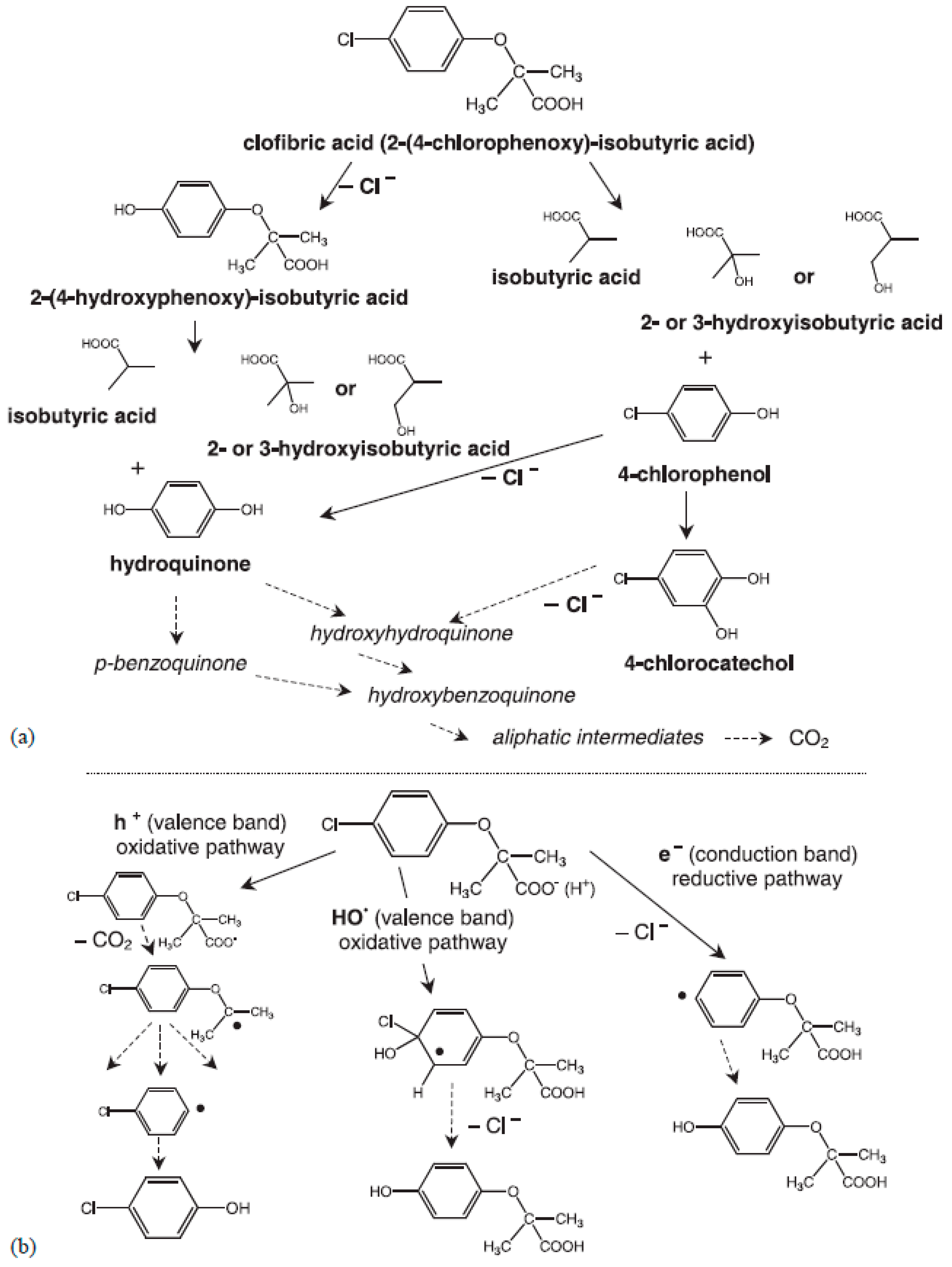
4.1. Photocatalysis and Other Advanced Oxidation Processes
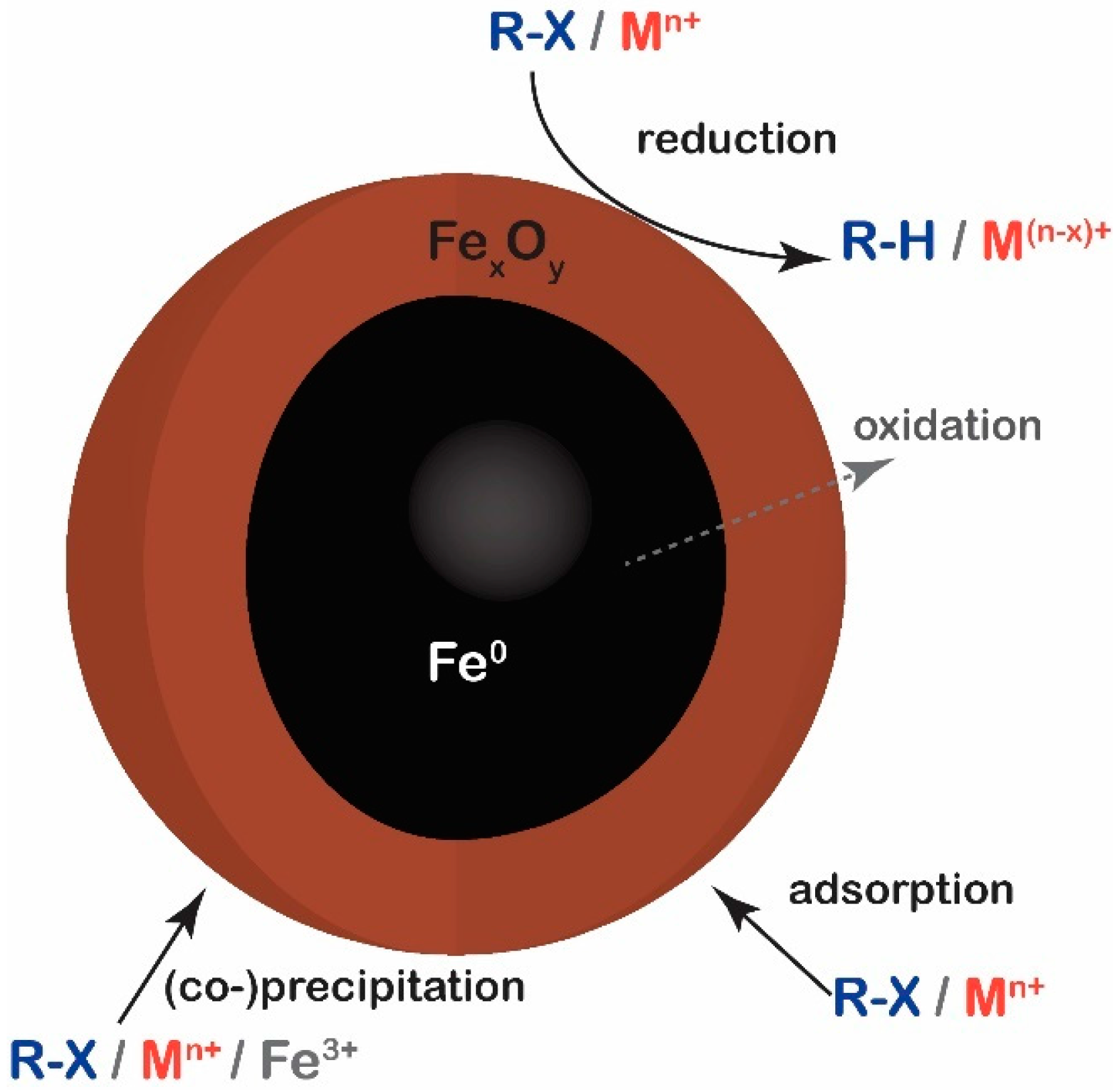
4.2. Reaction with Nanoscale Zero Valent Iron
5. Conclusions and Future Perspectives
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Luo, Y.; Guo, W.; Ngo, H.H.; Nghiem, L.D.; Hai, F.I.; Zhang, J.; Liang, S.; Wang, X.C. A review on the occurrence of micropollutants in the aquatic environment and their fate and removal during wastewater treatment. Sci. Total Environ. 2014, 473–474, 619–641. [Google Scholar] [CrossRef] [PubMed]
- Hughes, S.R.; Kay, P.; Brown, L.E. Global Synthesis and Critical Evaluation of Pharmaceutical Data Sets Collected from River Systems. Environ. Sci. Technol. 2013, 47, 661–677. [Google Scholar] [CrossRef] [PubMed]
- Cizmas, L.; Sharma, V.K.; Gray, C.M.; McDonald, T.J. Pharmaceuticals and personal care products in waters: Occurrence, toxicity, and risk. Environ. Chem. Lett. 2015, 13, 381–394. [Google Scholar] [CrossRef] [PubMed]
- Kurwadkar, S.; Zhang, X.; Ramirez, D.; Mitchell, F.L. Emerging Micro-Pollutants in the Environment: Occurrence, Fate, and Distribution; American Chemical Society: Washington, DC, USA, 2015. [Google Scholar]
- Fairbairn, D.J.; Karpuzcu, M.E.; Arnold, W.A.; Barber, B.L.; Kaufenberg, E.F.; Koskinen, W.C.; Novak, P.J.; Rice, P.J.; Swackhamer, D.L. Sources and transport of contaminants of emerging concern: A two-year study of occurrence and spatiotemporal variation in a mixed land use watershed. Sci. Total Environ. 2016, 551–552, 605–613. [Google Scholar] [CrossRef] [PubMed]
- Basile, T.; Petrella, A.; Petrella, M.; Boghetich, G.; Petruzzelli, V.; Colasuonno, S.; Petruzzelli, D. Review of Endocrine-Disrupting-Compound Removal Technologies in Water and Wastewater Treatment Plants: An EU Perspective. Ind. Eng. Chem. Res. 2011, 50, 8389–8401. [Google Scholar] [CrossRef]
- Mottaleb, M.A.; Meziani, M.J.; Matin, M.A.; Arafat, M.M.; Wahab, M.A. Emerging Micro-Pollutants Pharmaceuticals and Personal Care Products (PPCPs) Contamination Concerns in Aquatic Organisms—LC/MS and GC/MS Analysis. In Emerging Micro-Pollutants in the Environment: Occurrence, Fate, and Distribution; American Chemical Society: Washington, DC, USA, 2015; pp. 43–74. [Google Scholar]
- Goel, S. Antibiotics in the Environment: A Review. In Emerging Micro-Pollutants in the Environment: Occurrence, Fate, and Distribution; American Chemical Society: Washington, DC, USA, 2015; pp. 19–42. [Google Scholar]
- Tarr, H.L.A.; Boyd, J.W.; Bissett, H.M. Antibiotics in Food Processing, Experimental Preservation of Fish and Beef with Antibiotics. J. Agric. Food Chem. 1954, 2, 372–375. [Google Scholar] [CrossRef]
- Gilbert, N. Rules tighten on use of antibiotics on farms. Nature 2012, 481, 125. [Google Scholar] [CrossRef] [PubMed]
- Henriksson, P.J.G.; Troell, M.; Rico, A. Antimicrobial use in aquaculture: Some complementing facts. Proc. Natl. Acad. Sci. USA 2015, 112, E3317. [Google Scholar] [CrossRef] [PubMed]
- Venter, H.; Henningsen, M.L.; Begg, S.L. Antimicrobial resistance in healthcare, agriculture and the environment: The biochemistry behind the headlines. Essays Biochem. 2017, 61, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Van Boeckel, T.P.; Brower, C.; Gilbert, M.; Grenfell, B.T.; Levin, S.A.; Robinson, T.P.; Teillant, A.; Laxminarayan, R. Global trends in antimicrobial use in food animals. Proc. Natl. Acad. Sci. USA 2015, 112, 5649–5654. [Google Scholar] [CrossRef] [PubMed]
- Heberer, T.; Mechlinski, A.; Fanck, B.; Knappe, A.; Massmann, G.; Pekdeger, A.; Fritz, B. Field Studies on the Fate and Transport of Pharmaceutical Residues in Bank Filtration. Ground Water Monit. Remediat. 2004, 24, 70–77. [Google Scholar] [CrossRef]
- Ding, S.; Chu, W.; Bond, T.; Wang, Q.; Gao, N.; Xu, B.; Du, E. Formation and estimated toxicity of trihalomethanes, haloacetonitriles, and haloacetamides from the chlor(am)ination of acetaminophen. J. Hazard. Mater. 2018, 341, 112–119. [Google Scholar] [CrossRef] [PubMed]
- Ali, A.M.; Rønning, H.T.; Al Arif, W.M.; Kallenborn, R.; Kallenborn, R. Occurrence of pharmaceuticals and personal care products in effluent-dominated Saudi Arabian coastal waters of the Red Sea. Chemosphere 2017, 175, 505–513. [Google Scholar] [CrossRef] [PubMed]
- Calamari, D.; Zuccato, E.; Castiglioni, S.; Bagnati, R.; Fanelli, R. Strategic survey of therapeutic drugs in the rivers Po and lambro in Northern Italy. Environ. Sci. Technol. 2003, 37, 1241–1248. [Google Scholar] [CrossRef]
- Archer, E.; Petrie, B.; Kasprzyk-Hordern, B.; Wolfaardt, G.M. The fate of pharmaceuticals and personal care products (PPCPs), endocrine disrupting contaminants (EDCs), metabolites and illicit drugs in a WWTW and environmental waters. Chemosphere 2017, 174, 437–446. [Google Scholar] [CrossRef] [PubMed]
- Ebele, A.J.; Abou-Elwafa Abdallah, M.; Harrad, S. Pharmaceuticals and personal care products (PPCPs) in the freshwater aquatic environment. Emerg. Contam. 2017, 3, 1–16. [Google Scholar] [CrossRef]
- Magureanu, M.; Mandache, N.B.; Parvulescu, V.I. Degradation of pharmaceutical compounds in water by non-thermal plasma treatment. Water Res. 2015, 81, 124–136. [Google Scholar] [CrossRef] [PubMed]
- Klaminder, J.; Jonsson, M.; Fick, J.; Sundelin, A.; Brodin, T. The conceptual imperfection of aquatic risk assessment tests: Highlighting the need for tests designed to detect therapeutic effects of pharmaceutical contaminants. Environ. Res. Lett. 2014, 9. [Google Scholar] [CrossRef]
- Fritsch, E.B.; Connon, R.E.; Werner, I.; Davies, R.E.; Beggel, S.; Feng, W.; Pessah, I.N. Triclosan impairs swimming behavior and alters expression of excitation-contraction coupling proteins in fathead minnow (Pimephales promelas). Environ. Sci. Technol. 2013, 47, 2008–2017. [Google Scholar] [CrossRef] [PubMed]
- Pinto, P.I.S.; Guerreiro, E.M.; Power, D.M. Triclosan interferes with the thyroid axis in the zebrafish (Danio rerio). Toxicol. Res. 2013, 2, 60–69. [Google Scholar] [CrossRef]
- Yangali-Quintanilla, V.; Maeng, S.K.; Fujioka, T.; Kennedy, M.; Li, Z.; Amy, G. Nanofiltration vs. reverse osmosis for the removal of emerging organic contaminants in water reuse. Desalination Water Treat. 2011, 34, 50–56. [Google Scholar] [CrossRef]
- Bellona, C.; Drewes, J.E.; Xu, P.; Amy, G. Factors affecting the rejection of organic solutes during NF/RO treatment—A literature review. Water Res. 2004, 38, 2795–2809. [Google Scholar] [CrossRef] [PubMed]
- Nghiem, L.D.; Schäfer, A.I.; Elimelech, M. Removal of Natural Hormones by Nanofiltration Membranes: Measurement, Modeling and Mechanisms. Environ. Sci. Technol. 2004, 38, 1888–1896. [Google Scholar] [CrossRef] [PubMed]
- Schäfer, A.I.; Nghiem, L.D.; Waite, T.D. Removal of the natural hormone estrone from aqueous solutions using nanofiltration and reverse osmosis. Environ. Sci. Technol. 2003, 37, 182–188. [Google Scholar] [CrossRef] [PubMed]
- Nghiem, L.D.; Manis, A.; Soldenhoff, K.; Schäfer, A.I. Estrogenic hormone removal from wastewater using NF/RO membranes. J. Membr. Sci. 2004, 242, 37–45. [Google Scholar] [CrossRef]
- Huang, C.H.; Sedlak, D.L. Analysis of estrogenic hormones in municipal wastewater effluent and surface water using enzyme-linked immunosorbent assay and gas chromatography/tandem mass spectrometry. Environ. Toxicol. Chem. 2001, 20, 133–139. [Google Scholar] [CrossRef] [PubMed]
- Kimura, K.; Toshima, S.; Amy, G.; Watanabe, Y. Rejection of neutral endocrine disrupting compounds (EDCs) and pharmaceutical active compounds (PhACs) by RO membranes. J. Membr. Sci. 2004, 245, 71–78. [Google Scholar] [CrossRef]
- Pelekani, C.; Snoeyink, V. Competitive adsorption in natural water: Role of activated carbon pore size. Water Res. 1999, 33, 1209–1219. [Google Scholar] [CrossRef]
- Crittenden, J.C.; Sanongraj, S.; Bulloch, J.L.; Hand, D.W.; Rogers, T.N.; Speth, T.F.; Ulmer, M. Correlation of aqueous-phase adsorption isotherms. Environ. Sci. Technol. 1999, 33, 2926–2933. [Google Scholar] [CrossRef]
- Eisenman, G. The Molecular Basis of Ionic Selectivity in Macroscopic Systems. In Mass Transfer and Kinetics of Ion Exchange; Liberti, L., Helfferich, F.G., Eds.; Springer: Dordrecht, The Netherlands, 1983; pp. 121–155. ISBN 978-94-009-6899-8. [Google Scholar]
- Newcombe, G.; Drikas, M.; Hayer, R. Influence of characterised natural organic material on activated carbon adsorption: II. Effect on pore volume distribution and adsorption of 2-methylisoborneol. Water Res. 1997, 31, 1065–1073. [Google Scholar] [CrossRef]
- Freyria, F.S.; Armandi, M.; Compagnoni, M.; Ramis, G.; Rossetti, I.; Bonelli, B. Catalytic and Photocatalytic Processes for the Abatement of N-Containing Pollutants from Wastewater. Part 2: Organic Pollutants. J. Nanosci. Nanotechnol. 2017, 17, 3654–3672. [Google Scholar] [CrossRef]
- Compagnoni, M.; Ramis, G.; Freyria, F.S.; Armandi, M.; Bonelli, B.; Rossetti, I. Photocatalytic Processes for the Abatement of N-Containing Pollutants from Waste Water. Part 1: Inorganic Pollutants. J. Nanosci. Nanotechnol. 2017, 17, 3632–3653. [Google Scholar] [CrossRef]
- Bonelli, B.; Esposito, S.; Freyria, F.S. Mesoporous Titania: Synthesis, properties and comparison with non-porous titania. In Titanium Dioxide; Janus, M., Ed.; InTech: Rijeka, Croatia, 2017; ISBN 978-953-51-5493-8. [Google Scholar]
- Kaniou, S.; Pitarakis, K.; Barlagianni, I.; Poulios, I. Photocatalytic oxidation of sulfamethazine. Chemosphere 2005, 60, 372–380. [Google Scholar] [CrossRef] [PubMed]
- Chatzitakis, A.; Berberidou, C.; Paspaltsis, I.; Kyriakou, G.; Sklaviadis, T.; Poulios, I. Photocatalytic degradation and drug activity reduction of Chloramphenicol. Water Res. 2008, 42, 386–394. [Google Scholar] [CrossRef] [PubMed]
- Elmolla, E.S.; Chaudhuri, M. Degradation of amoxicillin, ampicillin and cloxacillin antibiotics in aqueous solution by the UV/ZnO photocatalytic process. J. Hazard. Mater. 2010, 173, 445–449. [Google Scholar] [CrossRef] [PubMed]
- Pouretedal, H.R.; Hasanali, M.A. Photocatalytic degradation of some b-lactam antibiotics in aqueous suspension of ZnS nanoparticles. Desalination Water Treat. 2013, 51, 2617–2623. [Google Scholar] [CrossRef]
- Holmberg, J.P.; Ahlberg, E.; Bergenholtz, J.; Hassellöv, M.; Abbas, Z. Surface charge and interfacial potential of titanium dioxide nanoparticles: Experimental and theoretical investigations. J. Colloid Interface Sci. 2013, 407, 168–176. [Google Scholar] [CrossRef] [PubMed]
- Elmolla, E.S.; Chaudhuri, M. Photocatalytic degradation of amoxicillin, ampicillin and cloxacillin antibiotics in aqueous solution using UV/TiO2 and UV/H2O2/TiO2 photocatalysis. Desalination 2010, 252, 46–52. [Google Scholar] [CrossRef]
- Doll, T.E.; Frimmel, F.H. Kinetic study of photocatalytic degradation of carbamazepine, clofibric acid, iomeprol and iopromide assisted by different TiO2 materials—Determination of intermediates and reaction pathways. Water Res. 2004, 38, 955–964. [Google Scholar] [CrossRef] [PubMed]
- Das, R.; Sarkar, S.; Chakraborty, S.; Choi, H.; Bhattacharjee, C. Remediation of antiseptic components in wastewater by photocatalysis using TiO2 nanoparticles. Ind. Eng. Chem. Res. 2014, 53, 3012–3020. [Google Scholar] [CrossRef]
- Choina, J.; Fischer, C.; Flechsig, G.U.; Kosslick, H.; Tuan, V.A.; Tuyen, N.D.; Tuyen, N.A.; Schulz, A. Photocatalytic properties of Zr-doped titania in the degradation of the pharmaceutical ibuprofen. J. Photochem. Photobiol. A Chem. 2014, 274, 108–116. [Google Scholar] [CrossRef]
- Augugliaro, V.; García-López, E.; Loddo, V.; Malato-Rodríguez, S.; Maldonado, I.; Marcì, G.; Molinari, R.; Palmisano, L. Degradation of lincomycin in aqueous medium: Coupling of solar photocatalysis and membrane separation. Sol. Energy 2005, 79, 402–408. [Google Scholar] [CrossRef]
- Méndez-Arriaga, F.; Otsu, T.; Oyama, T.; Gimenez, J.; Esplugas, S.; Hidaka, H.; Serpone, N. Photooxidation of the antidepressant drug Fluoxetine (Prozac®) in aqueous media by hybrid catalytic/ozonation processes. Water Res. 2011, 45, 2782–2794. [Google Scholar] [CrossRef] [PubMed]
- Piumetti, M.; Freyria, F.S.; Armandi, M.; Geobaldo, F.; Garrone, E.; Bonelli, B. Fe- and V-doped mesoporous titania prepared by direct synthesis: Characterization and role in the oxidation of AO7 by H2O2 in the dark. Catal. Today 2014, 227, 71–79. [Google Scholar] [CrossRef]
- Freyria, F.S.; Compagnoni, M.; Ditaranto, N.; Rossetti, I.; Piumetti, M.; Ramis, G.; Bonelli, B. Pure and Fe-doped mesoporous titania catalyse the oxidation of acid orange 7 by H2O2 under different illumination conditions: Fe doping improves photocatalytic activity under simulated solar light. Catalysts 2017, 7. [Google Scholar] [CrossRef]
- Klavarioti, M.; Mantzavinos, D.; Kassinos, D. Removal of residual pharmaceuticals from aqueous systems by advanced oxidation processes. Environ. Int. 2009, 35, 402–417. [Google Scholar] [CrossRef] [PubMed]
- Oller, I.; Fernández-Ibáñez, P.; Maldonado, M.I.; Pérez-Estrada, L.; Gernjak, W.; Pulgarín, C.; Passarinho, P.C.; Malato, S. Solar heterogeneous and homogeneous photocatalysis as a pre-treatment option for biotreatment. Res. Chem. Intermed. 2007, 33, 407–420. [Google Scholar] [CrossRef]
- Homem, V.; Santos, L. Degradation and removal methods of antibiotics from aqueous matrices—A review. J. Environ. Manag. 2011, 92, 2304–2347. [Google Scholar] [CrossRef] [PubMed]
- Shan, A.Y.; Ghazi, T.I.M.; Rashid, S.A. Immobilisation of titanium dioxide onto supporting materials in heterogeneous photocatalysis: A review. Appl. Catal. A Gen. 2010, 389, 1–8. [Google Scholar] [CrossRef]
- Murgolo, S.; Petronella, F.; Ciannarella, R.; Comparelli, R.; Agostiano, A.; Curri, M.L.; Mascolo, G. UV and solar-based photocatalytic degradation of organic pollutants by nano-sized TiO2 grown on carbon nanotubes. Catal. Today 2015, 240, 114–124. [Google Scholar] [CrossRef]
- Karaolia, P.; Michael-Kordatou, I.; Hapeshi, E.; Drosou, C.; Bertakis, Y.; Christofilos, D.; Armatas, G.S.; Sygellou, L.; Schwartz, T.; Xekoukoulotakis, N.P.; et al. Removal of antibiotics, antibiotic-resistant bacteria and their associated genes by graphene-based TiO2 composite photocatalysts under solar radiation in urban wastewaters. Appl. Catal. B Environ. 2018, 224, 810–824. [Google Scholar] [CrossRef]
- Freyria, F.S.; Bonelli, B.; Sethi, R.; Armandi, M.; Belluso, E.; Garrone, E. Reactions of Acid Orange 7 with Iron Nanoparticles in Aqueous Solutions. J. Phys. Chem. C 2011, 115, 24143–24152. [Google Scholar] [CrossRef]
- Freyria, F.S.; Esposito, S.; Armandi, M.; Deorsola, F.; Garrone, E.; Bonelli, B. Role of pH in the aqueous phase reactivity of zerovalent iron nanoparticles with acid orange 7, a model molecule of azo dyes. J. Nanomater. 2017, 2017. [Google Scholar] [CrossRef]
- Tosco, T.; Petrangeli Papini, M.; Cruz Viggi, C.; Sethi, R. Nanoscale zerovalent iron particles for groundwater remediation: A review. J. Clean. Prod. 2014, 77, 10–21. [Google Scholar] [CrossRef]
- Tiraferri, A.; Chen, K.L.; Sethi, R.; Elimelech, M. Reduced aggregation and sedimentation of zero-valent iron nanoparticles in the presence of guar gum. J. Colloid Interface Sci. 2008. [Google Scholar] [CrossRef] [PubMed]
- Comba, S.; Sethi, R. Stabilization of highly concentrated suspensions of iron nanoparticles using shear-thinning gels of xanthan gum. Water Res. 2009. [Google Scholar] [CrossRef] [PubMed]
- Suanon, F.; Sun, Q.; Li, M.; Cai, X.; Zhang, Y.; Yan, Y.; Yu, C.P. Application of nanoscale zero valent iron and iron powder during sludge anaerobic digestion: Impact on methane yield and pharmaceutical and personal care products degradation. J. Hazard. Mater. 2017, 321, 47–53. [Google Scholar] [CrossRef] [PubMed]
- Cao, Z.; Liu, X.; Xu, J.; Zhang, J.; Yang, Y.; Zhou, J.; Xu, X.; Lowry, G.V. Removal of Antibiotic Florfenicol by Sulfide-Modified Nanoscale Zero-Valent Iron. Environ. Sci. Technol. 2017, 51, 11269–11277. [Google Scholar] [CrossRef] [PubMed]
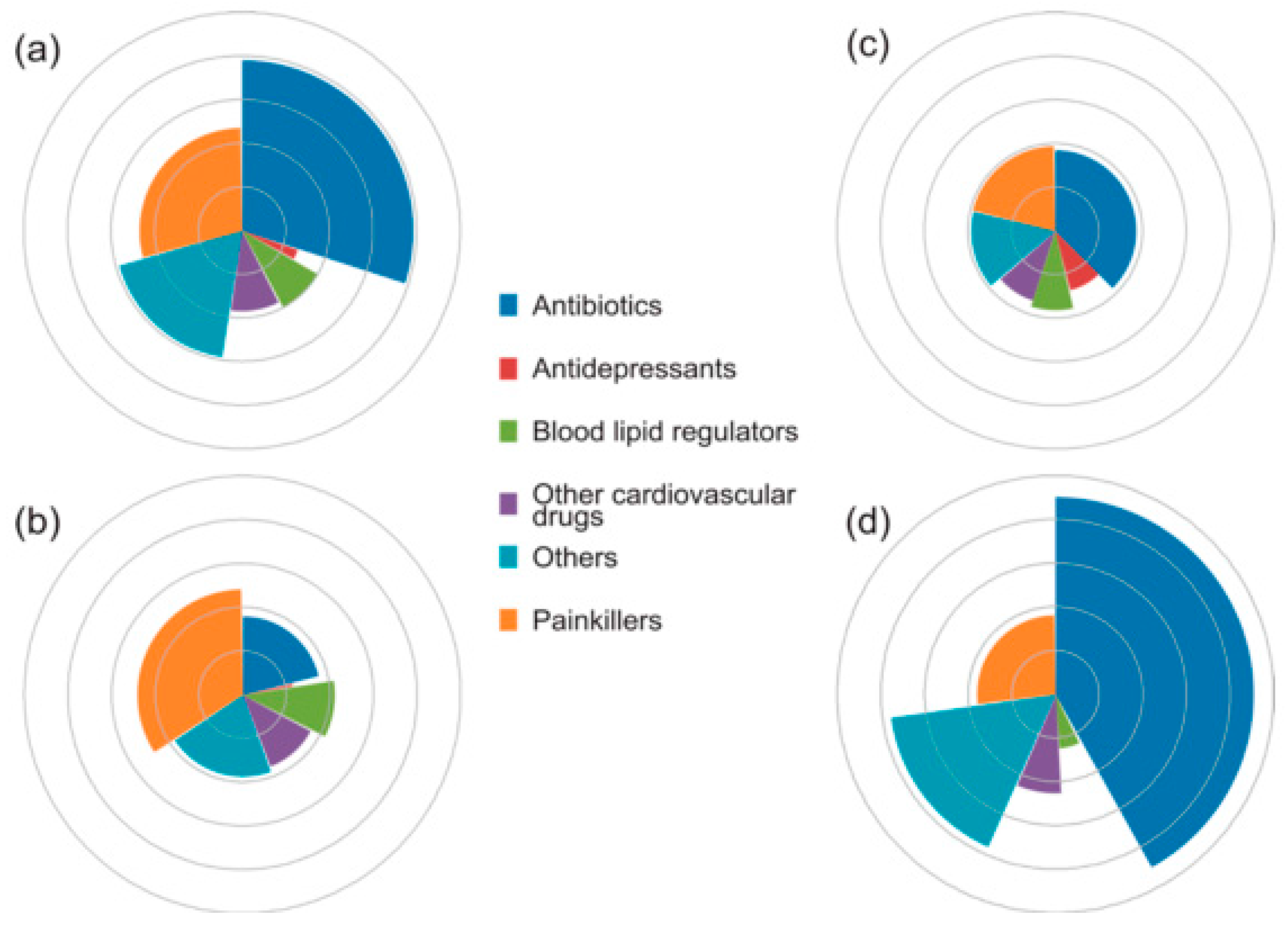

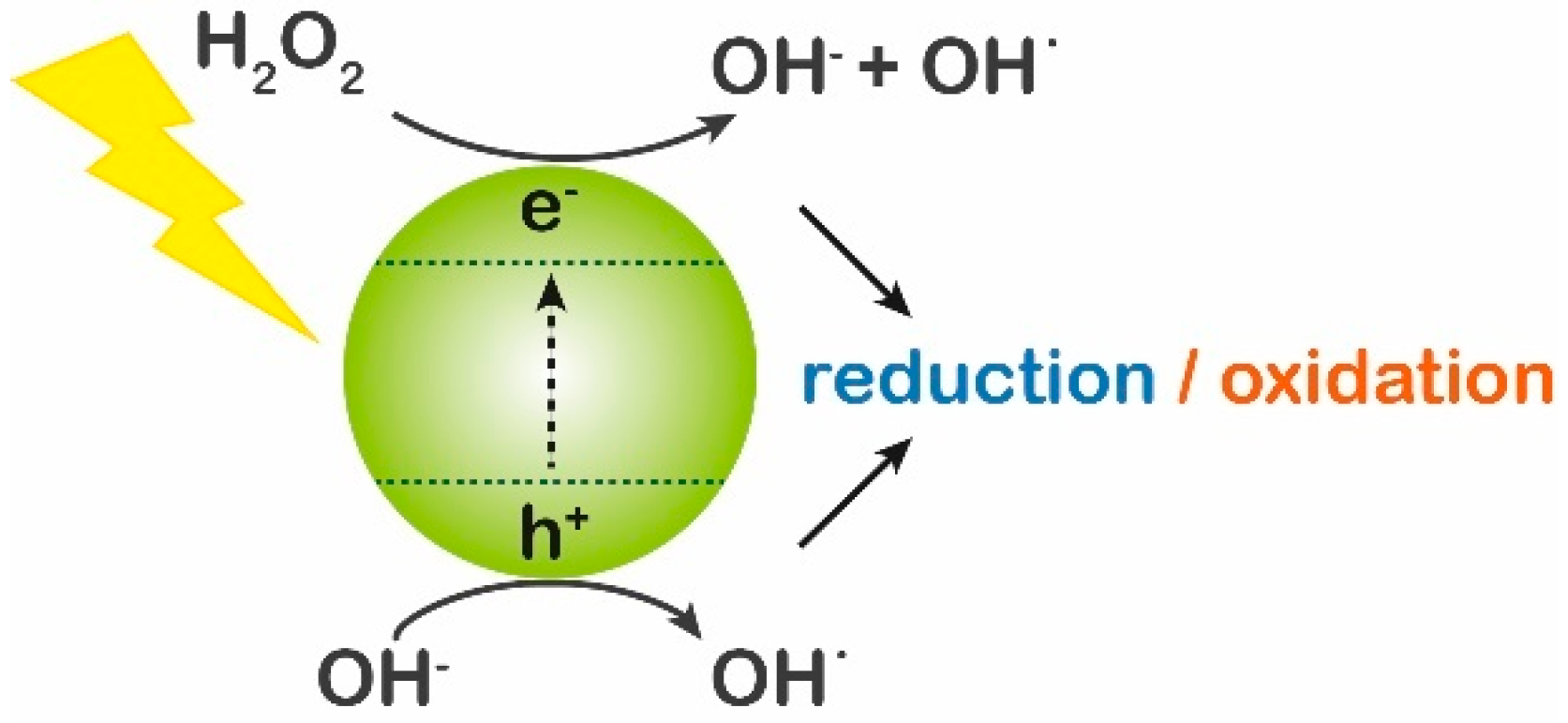
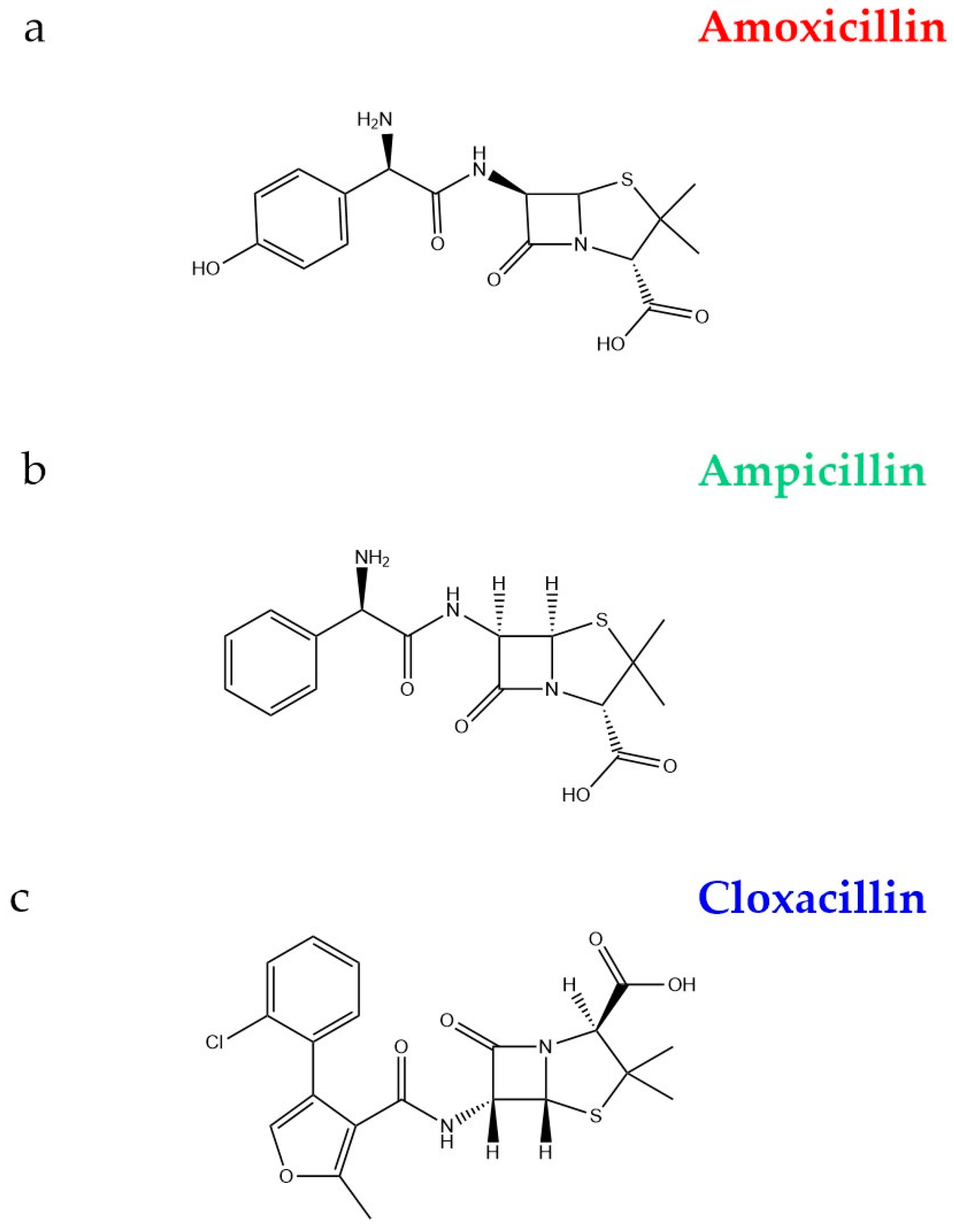
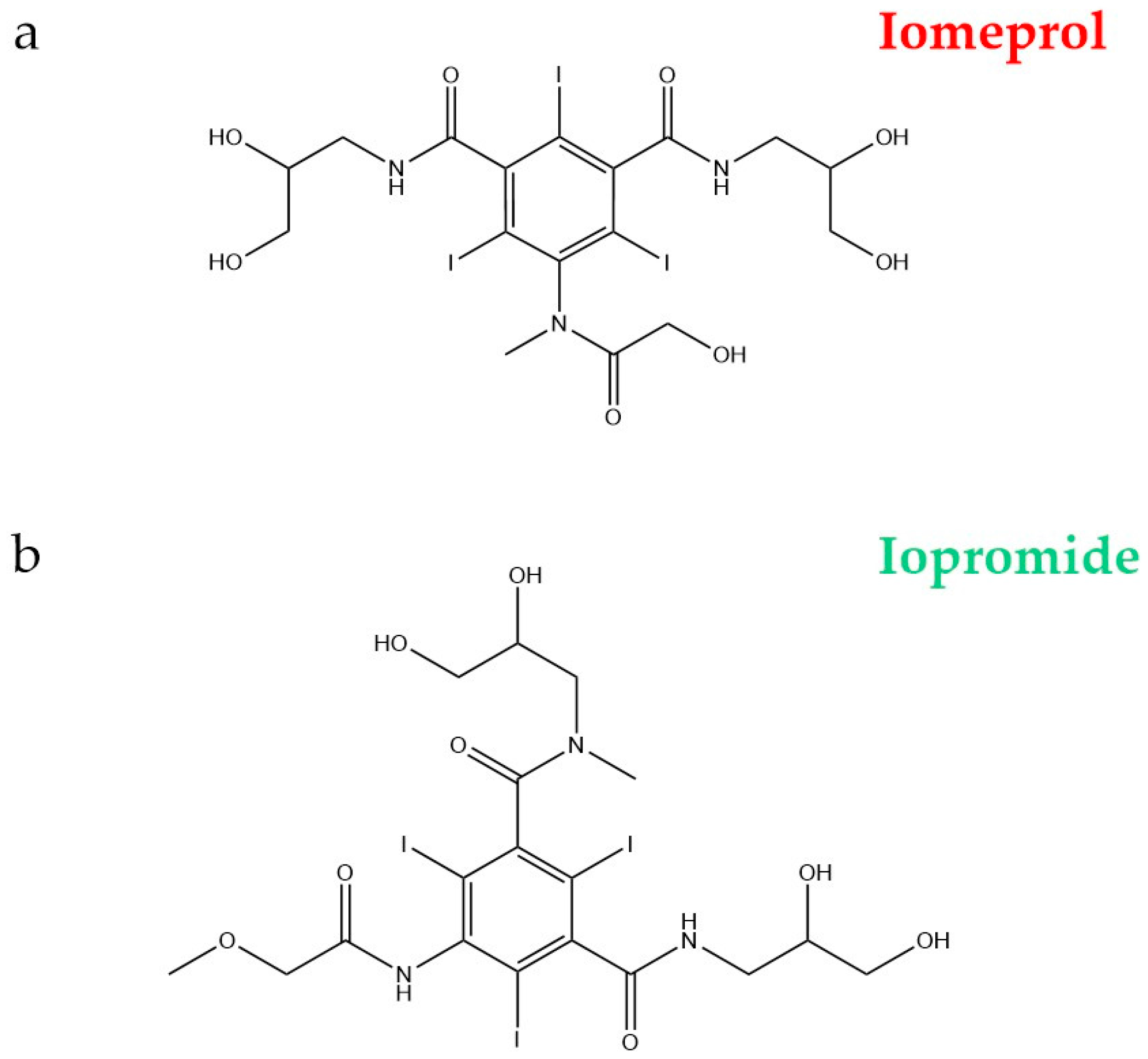

| Main Categories | Families | Examples |
|---|---|---|
| Drugs and Pharmaceuticals | antibiotics/antibacterials | amoxicillin, metronidazole, ofloxacin |
| steroids | estrone, 17β-estradiol, testosterone | |
| β-blocker | propranolol, salbutamol, atenolol | |
| nonsteroidal anti-inflammatory drugs (NSAID) | aspirin, ibuprofen, naproxen, ketoprofen | |
| antiepileptic/anticonvulsants | gabapentin, carbamazepine | |
| antidepressant/hypnotic | diazepam, venlafaxine, amitriptyline, dosulepin, meprobanate | |
| analgesic | morphine, propoxyphene, paracetamol | |
| hypertension | valsartan | |
| lipid regulation | bezafibrate, simvastatin, clofibric acid | |
| erectile dysfunction | sildenafil | |
| Stimulant and generally illegal drugs | hallucinogen | 3,4-Methylenedioxymethamphetamine (MDMA), 3,4-Methylenedioxy-N-ethylamphetamine (MDEA), 3,4-Methylenedioxyamphetamine (MDA) |
| stimulant | amphetamine, cocaine, benzylpiperazine | |
| human indicator | caffeine, nicotine | |
| Personal care products | preservative | methylparaben, propylparaben |
| sunscreen agent | 1-benzophenone, homosalate | |
| disinfectants/antiseptic | chloramines, chlorine, chlorine dioxide, chlorhexidine digluconate | |
| fragrances | musk xylol, tonalide | |
| Pesticides/Herbicides | organohalogenated compounds | Dichlorodiphenyltrichloroethane (DDT), lindane, vinclozolin, clopyralid |
| nitrogen containing | simazine, phenylurea |
| Characteristics | P25 (TiO2) | Hombikat UV100 | Mesoporous Titania (TiO2) |
|---|---|---|---|
| Crystallographic phase (w/w) | 75 ± 5% anatase 25 ± 5% rutile | 100% anatase | 100% anatase |
| Diameter of particles (nm) * | 15 ± 30 | <10 | <20 |
| Specific surface area (BET area, m2g−1) | 60 ± 10 | >250 | 150 ± 40 |
| Catalyst | SSA (m2·g−1) | Contaminant | Conditions at Room Temperature | Reference |
|---|---|---|---|---|
| Degussa P25 (commercial) | - | prozac | 75 W High pressure Hg lamp (max 360 nm) + O3 at pH 11 | [47] |
| Degussa P25 (commercial) | - | α-methylphenylglycine | UV at pH 6 | [51] |
| TiO2 (commercial) | - | amoxicillin, ampicillin and cloxacillin | UV (365 nm) at pH 11 and + H2O2 at pH 5 | [42] |
| Degussa P25 + NF membrane (both commercial) | 50 | lyncomycin | UV light at pH 6.3 | [46] |
| TiO2 and Aeroxide P25 (commercial) | 45–55/35–65 | chlorhexidine | UV at pH 10.5 | [44] |
| P25 and Hombikat UV100 (both commercial) | Table 2 | carbamazepine, clofibric acid, iomeprol, iopromide | Solar Simulator at pH 6.5 | [43] |
| TiO2, P25 and ZnO (all commercial) | - | sulfamethazine | 9 W lamp Osram Dulux S (320–400 nm, max 365 nm) at pH 4.8 | [38] |
| TiO2, P25 and ZnO (all commercial) | - | chloramphenicol | 9 W lamp Osram Dulux S (320–400 nm, max 365 nm) at pH 5.1 + H2O2 | [39] |
| Zr-TiO2 | 100 | ibuprofen | 4UV-vis solarium lamp | [45] |
| TiO2 on SWCNT (commercial) | - | 22 different PPCPs | UV and solar simulated light | [54] |
| graphene based TiO2 | 45–48 | sulfamethoxazole, erythromycin and clarithromycin | 1 kW Xenon lamp at pH 5.2–6.2 | [55] |
| ZnO (commercial) | - | amoxicillin, ampicillin and cloxacillin | 6 W UV lamp (max 365 nm) at pH 11 | [40] |
| ZnS | - | β-lactam | 500 W halogen lamp (visible lamp) at pH 4.5 | [41] |
| NZVI | 23.3 | 19 different PPCPs | In dark, at pH 7 | [61] |
| S-NZVI | 62.5 | florfenicol | In dark, at pH 7 | [62] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Freyria, F.S.; Geobaldo, F.; Bonelli, B. Nanomaterials for the Abatement of Pharmaceuticals and Personal Care Products from Wastewater. Appl. Sci. 2018, 8, 170. https://doi.org/10.3390/app8020170
Freyria FS, Geobaldo F, Bonelli B. Nanomaterials for the Abatement of Pharmaceuticals and Personal Care Products from Wastewater. Applied Sciences. 2018; 8(2):170. https://doi.org/10.3390/app8020170
Chicago/Turabian StyleFreyria, Francesca Stefania, Francesco Geobaldo, and Barbara Bonelli. 2018. "Nanomaterials for the Abatement of Pharmaceuticals and Personal Care Products from Wastewater" Applied Sciences 8, no. 2: 170. https://doi.org/10.3390/app8020170
APA StyleFreyria, F. S., Geobaldo, F., & Bonelli, B. (2018). Nanomaterials for the Abatement of Pharmaceuticals and Personal Care Products from Wastewater. Applied Sciences, 8(2), 170. https://doi.org/10.3390/app8020170






