Antibacterial Efficacy of Commercially Available Essential Oils Tested Against Drug-Resistant Gram-Positive Pathogens
Abstract
1. Introduction
2. Materials and Methods
2.1. Microorganisms
2.2. Essential Oils
2.3. Microbiological Assays
2.4. Statistical Analysis
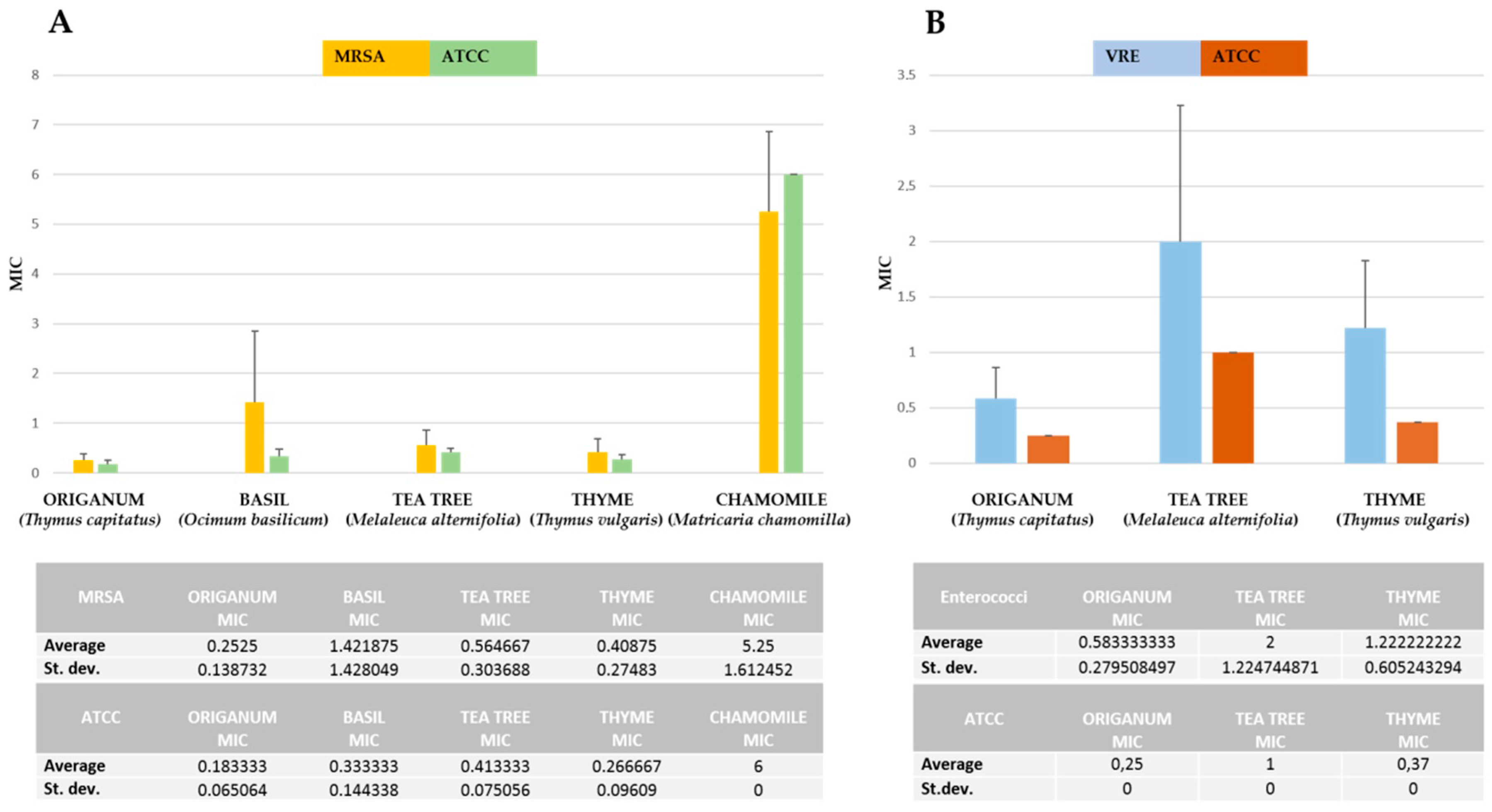
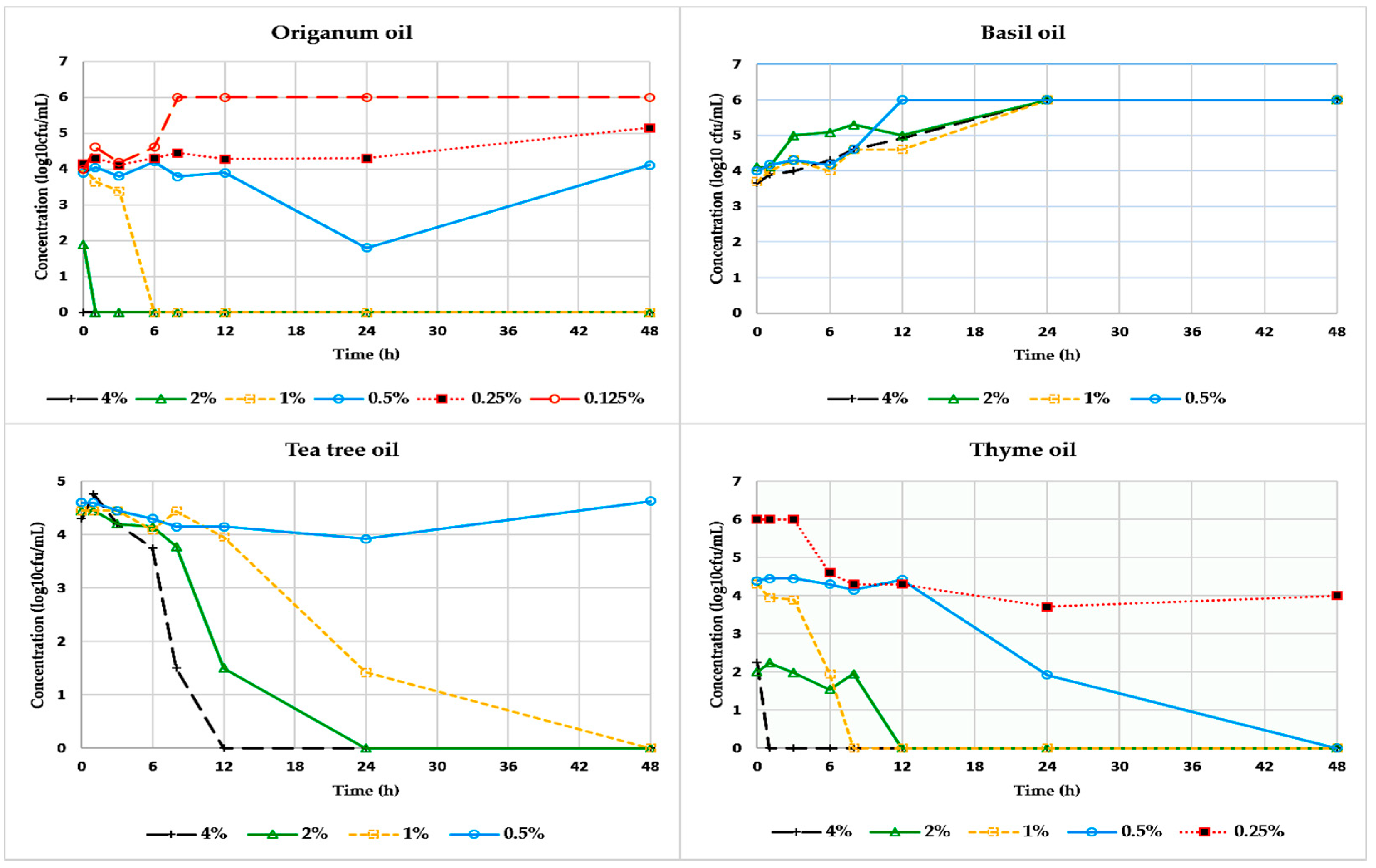
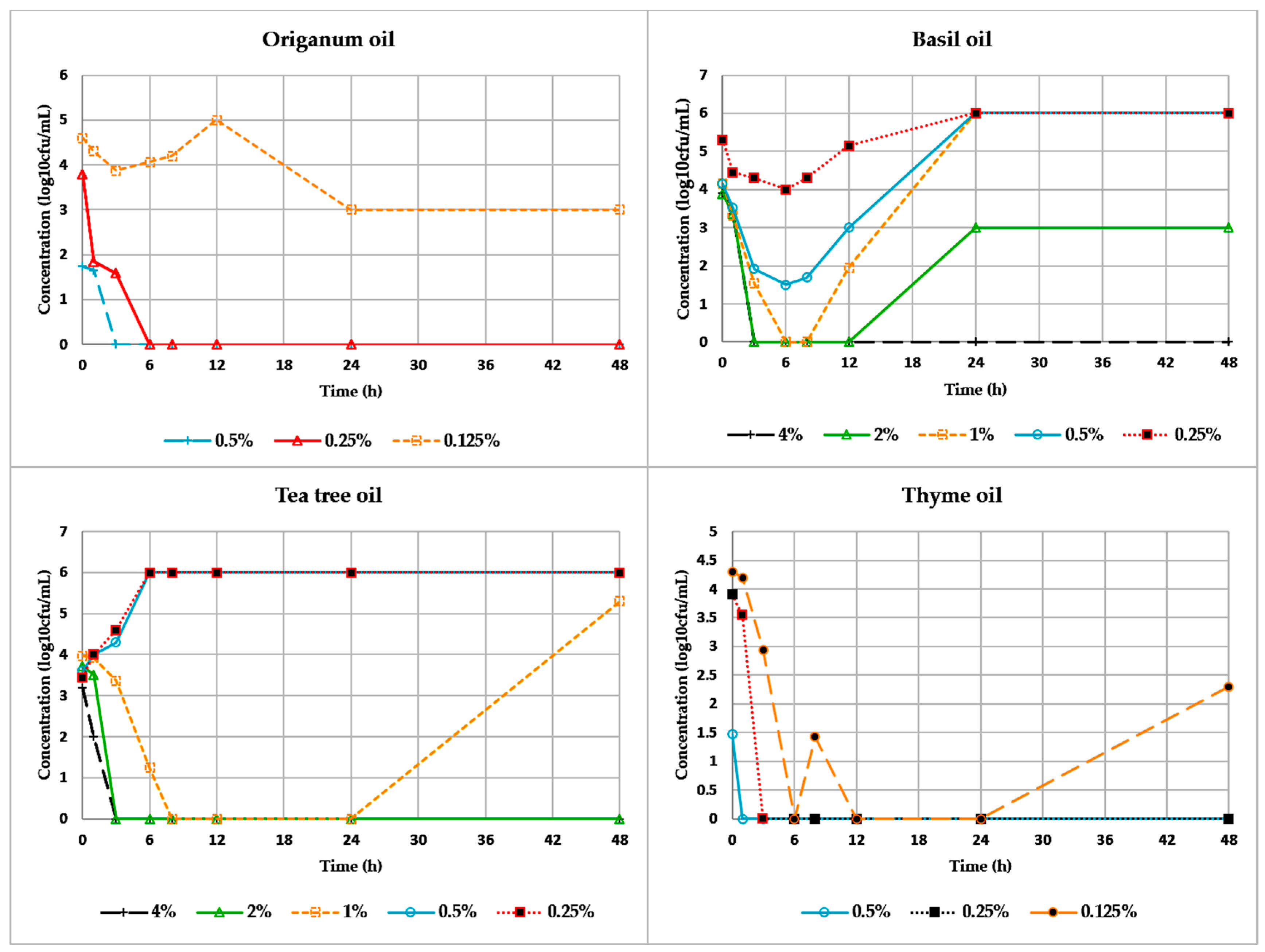
3. Results
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Gurib-Fakim, A. Medicinal Plants: Traditions of yesterday and drugs of tomorrow. Mol. Aspects Med. 2006, 27, 1–93. [Google Scholar] [CrossRef] [PubMed]
- Sakkas, H.; Papadopoulou, C. Antimicrobial activity of Basil, Oregano and Thyme essential oils. J. Microbiol. Biotechnol. 2017, 27, 429–438. [Google Scholar] [CrossRef] [PubMed]
- Teuber, M. Veterinary use and antibiotic resistance. Curr. Opin. Microbiol. 2001, 4, 493–499. [Google Scholar] [CrossRef]
- Levy, S.; Marshall, B. Antimicrobial resistance worldwide: Causes, challenges and responses. Nat. Med. 2004, 10, 122–129. [Google Scholar] [CrossRef] [PubMed]
- Wittekamp, B.H.; Bonten, M.J. Antibiotic prophylaxis in the era of multidrug-resistant bacteria. Expert Opin. Investig. Drugs 2012, 21, 767–772. [Google Scholar] [CrossRef] [PubMed]
- Michael, C.A.; Franks, A.E.; Labbate, M. The antimicrobial resistance crisis: Management through gene monitoring. Open Biol. 2016, 6, 160236. [Google Scholar] [CrossRef] [PubMed]
- Papadopoulou, C.; Dimitriou, D.; Levidiotou, S.; Panagiou, A.; Gessouli, H.; Antoniades, G. Bacterial strains isolated from eggs and their resistance to currently used antibiotics: Is there a health hazard for the consumers? Comp. Immun. Microbiol. Infect. Dis. 1997, 20, 35–40. [Google Scholar] [CrossRef]
- Rodríquez-Baño, J.; Pascual, A. Clinical significance of extended-spectrum beta-lactamases. Expert. Rev. Anti Infect. Ther. 2008, 6, 671–683. [Google Scholar] [CrossRef] [PubMed]
- Shorr, A.E. Review of studies of the impact on Gram-negative bacterial resistance on outcomes in the intensive care unit. Crit. Care Med. 2009, 37, 1463–1469. [Google Scholar] [CrossRef] [PubMed]
- Gousia, P.; Economou, V.; Sakkas, H.; Leveidiotou, S.; Papadopoulou, C. Antimicrobial resistance of major foodborne pathogens from major meat products. Foodborne Pathog. Dis. 2010, 8, 27–38. [Google Scholar] [CrossRef] [PubMed]
- Wieler, L.H.; Ewers, C.; Guenther, S.; Walther, B.; Lübke-Becker, A. Methicillin-resistant staphylococci (MRS) and extended-spectrum beta-lactamases (ESBL)-producing Enterobacteriaceae in companion animals: Nosocomial infections as one reason for the rising prevalence of these potential zoonotic pathogens in clinical samples. Int. J. Med. Microbiol. 2011, 301, 635–641. [Google Scholar] [PubMed]
- Coenen, S. Infectious diseases in primary care; managing the interface between the person and the community. Eur. J. Gen. Pract. 2012, 18, 117–121. [Google Scholar] [CrossRef] [PubMed]
- Economou, V.; Gousia, P.; Kansouzidou, A.; Sakkas, H.; Karanis, P.; Papadopoulou, C. Prevalence, antimicrobial resistance and relation to indicator and pathogenic microorganisms of Salmonella enterica isolated from surface waters within an agricultural landscape. Int. J. Hyg. Environ. Health 2013, 216, 435–444. [Google Scholar] [CrossRef] [PubMed]
- Gousia, P.; Economou, V.; Bozidis, P.; Papadopoulou, C. Vancomycin-resistance phenotypes, Vancomycin-resistance genes, and resistance to antibiotics of Enterococci isolated from food of animal origin. Foodborne Pathog. Dis. 2015, 12. [Google Scholar] [CrossRef] [PubMed]
- Prabuseenivasan, S.; Jayakumar, M.; Ignacimuthu, S. In vitro antibacterial activity of some plant essential oils. BMC Complement. Altern. Med. 2006, 6, 39. [Google Scholar] [CrossRef] [PubMed]
- Dadalioglu, I.; Evrendilek, G.A. Chemical compositions and antibacterial effects of essential oils of Turkish oregano (Origanum minutiflorum), bay Laurel (Laurus nobilis), Spanish levander (Lavandula stoechas L.), and fennel (Foeniculum vulgare) on common foodborne pathogens. J. Agric. Food Chem. 2004, 52, 8255–8260. [Google Scholar] [CrossRef] [PubMed]
- Solomakos, N.; Govaris, A.; Koidis, P.; Botsoglou, N. Antimicrobial effect of thyme essential oil, nisin, and their combination against Listeria monocytogenes in minced beef during refrigerated storage. Food Microbiol. 2008, 25, 120–127. [Google Scholar] [CrossRef] [PubMed]
- Tiwari, B.K.; Valdramidis, V.P.; O’Donnell, C.P.; Muthukumarappan, K.; Bourke, P.; Cullen, P.J. Application of natural antimicrobials for food preservation. J. Agric. Food Chem. 2009, 57, 5987–6000. [Google Scholar] [CrossRef] [PubMed]
- Fratianni, F.; De Martino, L.; Melone, A.; De Feo, V.; Coppola, R.; Nazzaro, F. Preservation of chicken breast meat treated with thyme and balm essential oils. J. Food Sci. 2010, 75, M528–M535. [Google Scholar] [CrossRef] [PubMed]
- Hyldgaard, M.; Mygind, T.; Meyer, R.L. Essential oils in food preservation: Mode of action, synergies and interactions with food matrix components. Front. Microbiol. 2012, 3, 1–24. [Google Scholar] [CrossRef] [PubMed]
- Yossa, N.; Patel, J.; Millner, P.; Lo, Y.M. Essential oils reduce Escherichia coli O157:H7 and Salmonella on spinach leaves. J. Food Prot. 2012, 75, 488–496. [Google Scholar] [CrossRef] [PubMed]
- Penalver, P.; Huerta, B.; Borge, C.; Astorga, R.; Romero, R.; Perea, A. Antimicrobial activity of five essential oils against origin strains of the Enterobacteriaceae family. APMIS 2005, 113, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Windisch, W.; Schedle, K.; Plitzner, C.; Kroismayr, A. Use of phytogenic products as feed additives for swine and poultry. J. Anim. Sci. 2008, 86, E140–E148. [Google Scholar] [CrossRef] [PubMed]
- Huyghebaert, G.; Ducatelle, R.; Van Immerseel, F. An update on alternatives to antimicrobial growth promoters for broilers. Vet. J. 2011, 187, 182–188. [Google Scholar] [CrossRef] [PubMed]
- Maenner, K.; Vahjen, W.; Simon, O. Studies of the effects of essential-oil-based feed additives on performance, ileal nutrient digestibility and selected bacterial groups in the gastrointestinal tract of piglets. J. Anim. Sci. 2011, 89, 2106–2112. [Google Scholar] [CrossRef] [PubMed]
- Doyle, M.P.; Erickson, M.C. Opportunities for mitigating pathogen contamination during on-farm food production. Int. J. Food Microbiol. 2012, 152, 54–74. [Google Scholar] [CrossRef] [PubMed]
- Banes-Marshall, L.; Cawley, P.; Phillips, C.A. In vitro activity of Melaleuca alternifolia (tea tree) oil against bacterial and Candida spp. Isolates from clinical specimens. Br. J. Biomed. Sci. 2001, 58, 139–145. [Google Scholar] [PubMed]
- Warnke, H.; Becker, S.T.; Podschun, R.; Sivananthan, S.; Springer, I.N.; Russo, P.A.J.; Wiltfang, J.; Fickenscher, H.; Sherry, E. The battle against multi-resistant strains: Renaissance of antimicrobial essential oils as a promising force to fight hospital-acquired infections. J. Craniomaxillofac. Surg. 2009, 37, 392–397. [Google Scholar] [CrossRef] [PubMed]
- Alexopoulos, A.; Kimbaris, A.C.; Plessas, S.; Mantzourani, I.; Theodoridou, I.; Stavropoulou, E.; Polissiou, M.G.; Bezirtzoglou, E. Antibacterial activities of essential oils from eight Greek aromatic plants against clinical isolates of Staphylococcus aureus. Anaerobe 2011, 17, 399–402. [Google Scholar] [CrossRef] [PubMed]
- Sienkiewicz, M.; Łysakowska, M.; Ciećwierz, J.; Denys, P.; Kowalczyk, E. Antibacterial activity of thyme and lavender essential oils. Med. Chem. 2011, 7, 674–689. [Google Scholar] [CrossRef] [PubMed]
- Crandall, P.G.; Ricke, S.C.; O’Bryan, C.A.; Parrish, N.M. In vitro effects of citrus oils against Mycobacterium turberculosis and non-tuberculous Mycobacteria of clinical importance. J. Environ. Sci. Health B 2012, 47, 736–741. [Google Scholar] [CrossRef] [PubMed]
- Ferrini, A.M.; Mannoni, V.; Aureli, P.; Salvatore, G.; Piccirilli, E.; Ceddia, T.; Pontieri, E.; Sessa, R.; Oliva, B. Melaleuca alternifolia essential oil possesses potent anti-staphylococcal activity extended to strains resistant to antibiotics. Int. J. Immunopathol. Pharmacol. 2006, 19, 539–544. [Google Scholar] [CrossRef] [PubMed]
- Ravishankar, S.; Zhu, L.; Law, B.; Joens, L.; Friedman, M. Plant-derived compounds inanctivate antibiotic-resistant Campylobacter jejuni strains. J. Food Prot. 2008, 71, 1145–1149. [Google Scholar] [CrossRef] [PubMed]
- Tohidpour, A.; Sattari, M.; Omidbaigi, R.; Yadegar, A.; Nazemi, J. Antibacterial effect of essential oils from two medicinal plants against Methicillin-resistant Staphylococcus aureus (MRSA). Phytomedicine 2010, 17, 142–145. [Google Scholar] [CrossRef] [PubMed]
- Sakkas, H.; Gousia, P.; Economou, V.; Sakkas, V.; Petsios, S.; Papadopoulou, C. In vitro antimicrobial activity of five essential oils against multi-drug resistant Gram-negative clinical isolates. J. Intercult. Ethnopharmacol. 2016, 5, 212–218. [Google Scholar] [CrossRef] [PubMed]
- Clinical and Laboratory Standards Institute (CLSI). Performance Standards for Antimicrobial Susceptibility Testing, 27th ed.; CLSI: Wayne, PA, USA, 2017. [Google Scholar]
- Hammer, K.A.; Carson, C.F.; Riley, T.V. Susceptibility of transient and commensal skin flora to the essential oil of Melaleuca alternifolia (tea tree oil). Am. J. Infect. Control 1996, 24, 186–189. [Google Scholar] [CrossRef]
- Clinical and Laboratory Standards Institute (CLSI). Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria that Grow Aerobically, 9th ed.; Approved Standard; CLSI: Wayne, PA, USA, 2012. [Google Scholar]
- Moreira, M.R.; Ponce, A.G.; Del Valle, C.E.; Roura, S.I. Inhibitory parameters of essential oils to reduce a foodborne pathogen. LWT 2005, 38, 565–570. [Google Scholar] [CrossRef]
- Economou, V.; Gousia, P. Agriculture and food animals as a source of antimicrobial-resistant bacteria. Infect. Drug. Resist. 2015, 1, 49–51. [Google Scholar] [CrossRef] [PubMed]
- Cowan, M.M. Plant Products as Antimicrobial Agents. Clin. Microbiol. Rev. 1999, 12, 564–582. [Google Scholar] [CrossRef] [PubMed]
- Canillac, N.; Mourey, A. Effects of several environmental factors on the anti-Listeria monocytogenes activity of an essential oil of Picea excelsa. Int. J. Food Microbiol. 2004, 92, 95–103. [Google Scholar] [CrossRef] [PubMed]
- Chorianopoulos, N.G.; Evergetis, E.T.; Aligiannis, N.; Mitakou, S.; Nychas, G.J.E.; Haroutian, S.A. Correlation between chemical composition of Greek essential oils and their antibacterial activity against food-borne pathogens. Nat. Prod. Commun. 2007, 2, 419–426. [Google Scholar]
- Νostro, A.; Blanco, A.; Cannatelli, M.; Enea, V.; Flamini, G.; Morelli, I.; Roccaro-Sudano, A.; Alonzo, V. Susceptibility of methicillin-resistant staphylococci to oregano essential oil, carvacrol and thymol. FEMS Microbiol. Lett. 2004, 230, 191–195. [Google Scholar] [CrossRef]
- Preuss, H.; Echard, B.; Enig, M.; Brook, I.; Elliot, T. Minimum inhibitory concentrations of herbal essential oils and monolaurin for gram-positive and gram-negative bacteria. Mol. Cell. Biochem. 2005, 272, 29–34. [Google Scholar] [CrossRef] [PubMed]
- Hammer, K.A.; Carson, C.F.; Riley, T.V. Antimicrobial activity of essential oils and other plant extracts. J. Appl. Microbiol. 1999, 86, 985–990. [Google Scholar] [CrossRef] [PubMed]
- Carson, C.; Mee, B.; Riley, T. Mechanism of action of Melaleuca alternifolia (Tea Tree) oil on Staphylococcus aureus determined by time-kill, lysis, leakage and salt tolerance assays and electron microscopy. Antimicrob. Agents Chemother. 2002, 46, 1914–1920. [Google Scholar] [CrossRef] [PubMed]
- Mann, C.M.; Markham, J.L. A new method for determining the minimum inhibitory concentration of essential oils. J. Appl. Microbiol. 1998, 84, 538–544. [Google Scholar] [CrossRef] [PubMed]
- Nelson, R. Selection of resistance to the essential oil of Melaleuca alternifolia in Staphylococcus aureus. J. Antimicrob. Chemother. 2000, 45, 547–551. [Google Scholar] [CrossRef]
- Nazzaro, F.; Fratianni, F.; De Martino, L.; Coppola, R.; De Feo, V. Effect of Essential Oils on Pathogenic Bacteria. Pharmaceuticals 2013, 6, 1451–1474. [Google Scholar] [CrossRef] [PubMed]
- Trombetta, D.; Castelli, F.; Sarpietro, M.G.; Venuti, V.; Cristani, M.; Daniele, C.; Saija, A.; Mazzanti, G.; Bisignano, G. Mechanisms of antibacterial action of three monoterpenes. Antimicrob. Agents Chemother. 2005, 49, 2474–2478. [Google Scholar] [CrossRef] [PubMed]
- Lopez Romero, J.; González-Rios, H.; Borges, A.; Simões, M. Antibacterial effects and mode of action of selected essential oils components against Escherichia coli and Staphylococcus aureus. J. Evid. Based Complement. Altern. Med. 2015. [Google Scholar] [CrossRef] [PubMed]
- Burt, S.A.; Reinders, R.D. Antibacterial activity of selected plant essential oils against Escherichia coli O157:H7. Lett. Appl. Microbiol. 2003, 36, 162–167. [Google Scholar] [CrossRef] [PubMed]
- Mrozik, A.; Piotrowska-Seget, Z.; Labuzek, S. Changes in whole cell-derived fatty acids induced by naphthalene in bacteria from genus Pseudomonas. Microbiol. Res. 2004, 1, 87–95. [Google Scholar] [CrossRef] [PubMed]
| GC-17A Gas Chromatograph with GCMS-QP5000 | |
|---|---|
| Column: | DB-5-MS, 30 × 0.32 mm, 0.25 μm, containing 5% phenyl-methylpolysiloxane |
| Oven: | 55 °C, 5 °C/min to 200 °C, 1 °C/min to 210 (2 min), 20 °C/min to 270 °C (3 min) |
| Injector: | 240 °C (splitless mode) |
| Ion source: | 240 °C |
| Interface: | 290 °C |
| Scan range: | m/z 50–450 (full scan 70 eV) |
| Carrier gas: | Helium 1.5 mL/min |
| Major Identified Components | |
| Basil oil | estragole |
| Chamomile blue oil | bisabolol and trans-b-farnesene |
| Origanum oil | carvacrol and thymol |
| Tea tree oil | terpinen-4-ol and p-cymene |
| Thyme oil | thymol, p-cymene, and linalool |
| Antibiotics ‡/Bacteria | S. aureus (n = 16) | Enterococcus spp. (n = 9) |
|---|---|---|
| PEN | 16 (100%) | 9 (100%) |
| AMP | - | 9 (100%) |
| VAN | 3 (18.7%) | 9 (100%) |
| TEC | 4 (25%) | 9 (100%) |
| ERY | 11 (68.7%) | - |
| CC | 11 (68.7%) | - |
| GEN | 4 (25%) | - |
| FOX | 16 (100%) | - |
| CIP | 11 (68.7%) | 7 (77.7%) |
| QD | 4 (25%) | 9 (100%) |
| LZD | - | 6 (66.6%) |
| TET | - | 5 (55.5%) |
| Essential Oils/Bacterial Strains | Ocimum basilicum | Thymus capitatus | Melaleuca alternifolia | Thymus vulgaris | Matricaria chamomilla | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| MIC | MBC | MIC | MBC | MIC | MBC | MIC | MBC | MIC | MBC | |
| MRSA | 0.25–4 | 0.25–4 | 0.06–0.5 | 0.06–0.5 | 0.12–1 | 0.06–2 | 0.06–1 | 0.06–1 | 2->4 | 2->4 |
| VRE | ≥4 | >4 | 0.25–1 | 0.5–1 | 1–4 | 0.75–4 | 0.5–2 | 0.32–4 | >4 | >4 |
| SA 29213 | 0.25 | 0.25 | 0.25 | 0.25 | 0.5 | 0.5 | 0.25–0.5 | 0.25 | >4 | >4 |
| SA 43300 | 0.5 | 0.5 | 0.12 | 0.12 | 0.25–0.5 | 0.25 | 0.25 | 0.25 | >4 | >4 |
| SE 12228 | 0.25 | 0.25 | 0.12–0.25 | 0.12 | 0.25–0.5 | 0.25–0.5 | 0.12–0.25 | 0.12–0.25 | >4 | >4 |
| EF 51299 | 4 | 4 | 0.25 | 0.25 | 1 | 1 | 0.25–0.5 | 0.25–0.5 | >4 | >4 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sakkas, H.; Economou, V.; Gousia, P.; Bozidis, P.; Sakkas, V.A.; Petsios, S.; Mpekoulis, G.; Ilia, A.; Papadopoulou, C. Antibacterial Efficacy of Commercially Available Essential Oils Tested Against Drug-Resistant Gram-Positive Pathogens. Appl. Sci. 2018, 8, 2201. https://doi.org/10.3390/app8112201
Sakkas H, Economou V, Gousia P, Bozidis P, Sakkas VA, Petsios S, Mpekoulis G, Ilia A, Papadopoulou C. Antibacterial Efficacy of Commercially Available Essential Oils Tested Against Drug-Resistant Gram-Positive Pathogens. Applied Sciences. 2018; 8(11):2201. https://doi.org/10.3390/app8112201
Chicago/Turabian StyleSakkas, Hercules, Vangelis Economou, Panagiota Gousia, Petros Bozidis, Vasilios A. Sakkas, Stefanos Petsios, George Mpekoulis, Afrodite Ilia, and Chrissanthy Papadopoulou. 2018. "Antibacterial Efficacy of Commercially Available Essential Oils Tested Against Drug-Resistant Gram-Positive Pathogens" Applied Sciences 8, no. 11: 2201. https://doi.org/10.3390/app8112201
APA StyleSakkas, H., Economou, V., Gousia, P., Bozidis, P., Sakkas, V. A., Petsios, S., Mpekoulis, G., Ilia, A., & Papadopoulou, C. (2018). Antibacterial Efficacy of Commercially Available Essential Oils Tested Against Drug-Resistant Gram-Positive Pathogens. Applied Sciences, 8(11), 2201. https://doi.org/10.3390/app8112201








