The Characteristics of Intrinsic Fluorescence of Type I Collagen Influenced by Collagenase I
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents
2.2. Analysis of Fluorescence Spectroscopy
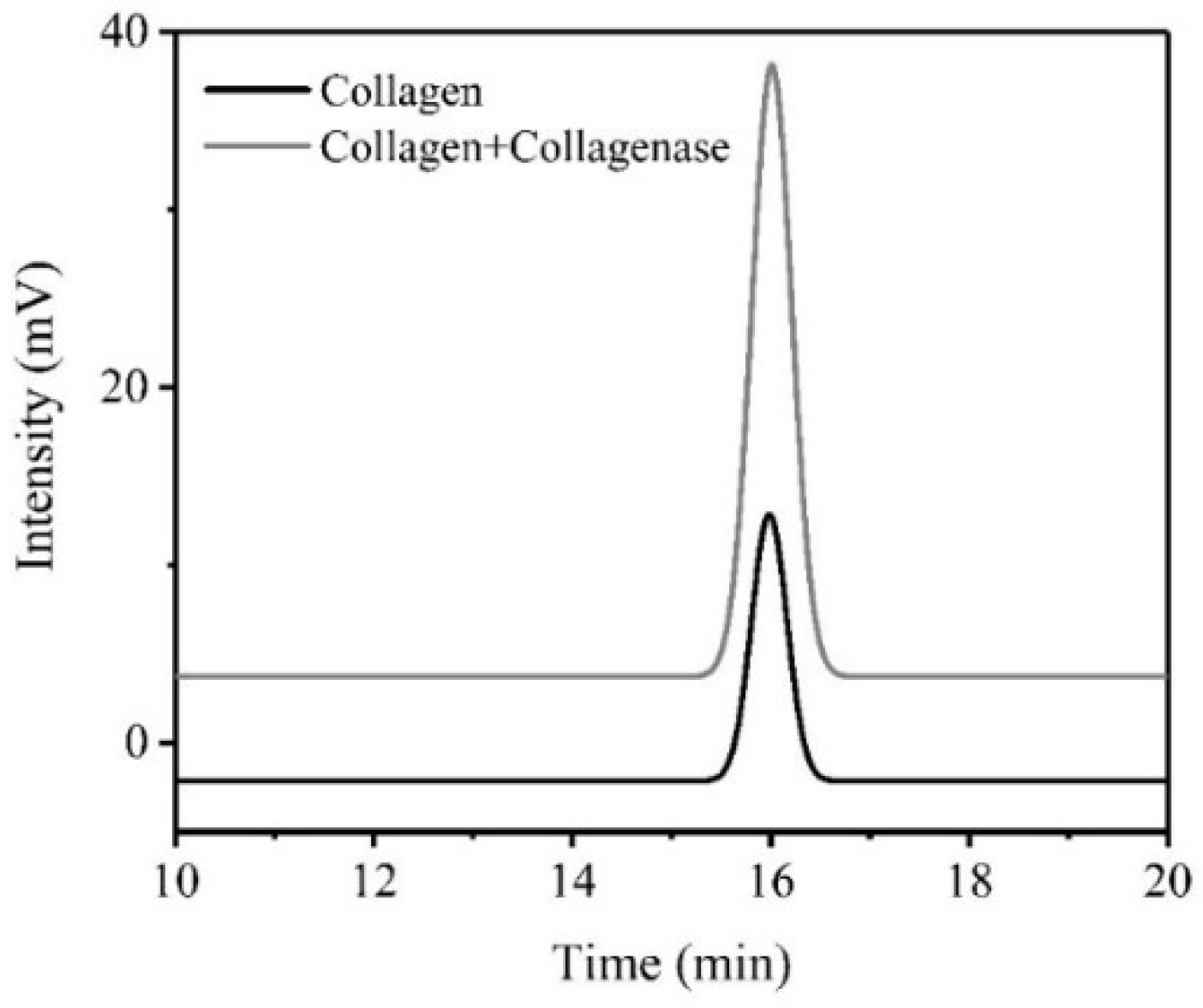
2.3. High-Performance Liquid Chromatography (HPLC) Analysis
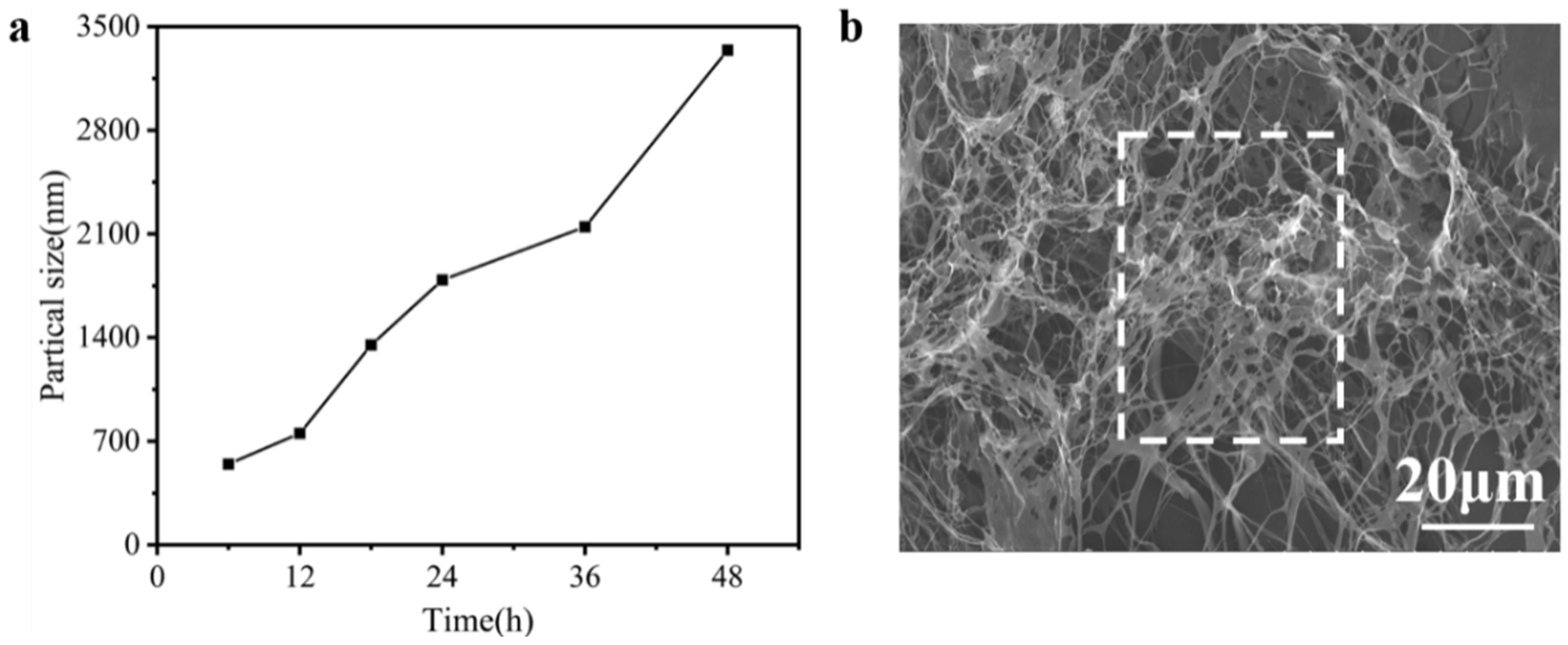
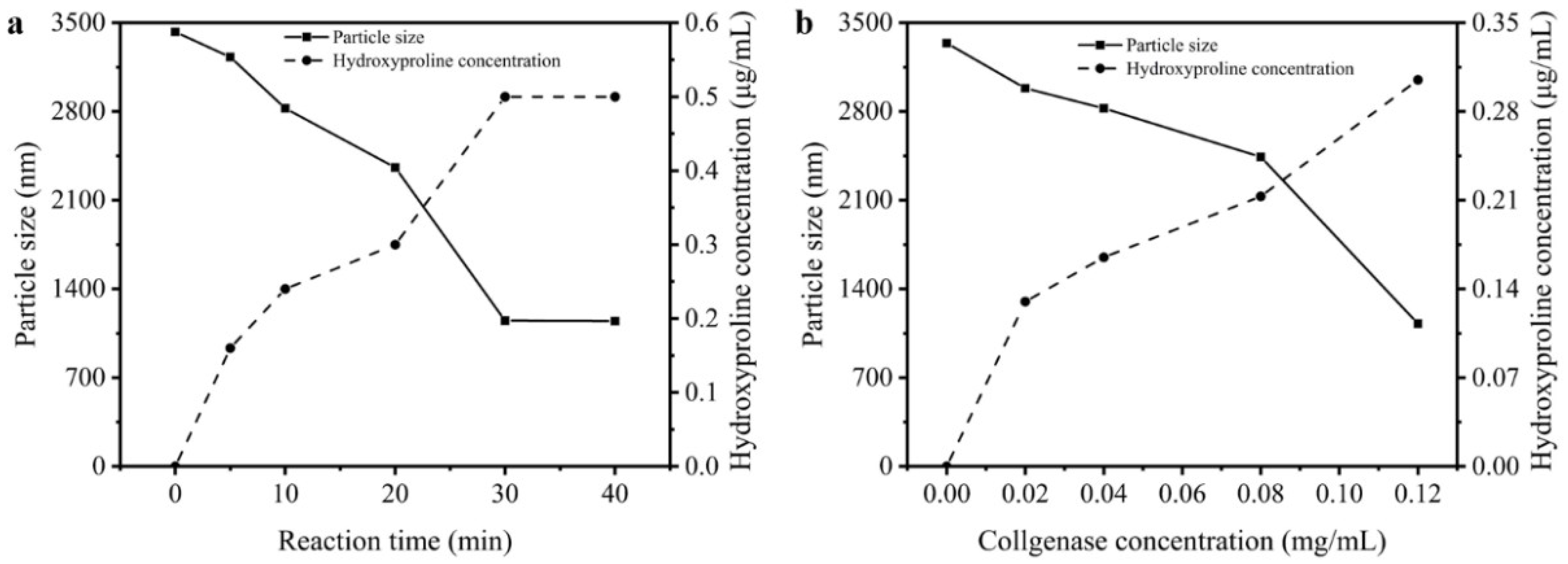
2.4. Analysis of Particle Size
2.5. Analysis of Hydroxyproline Concentration
2.6. SEM Analysis
2.7. Differential Scanning Calorimetry (DSC) Analysis
3. Results and Discussions
3.1. Fluorescence Spectroscopy Analysis
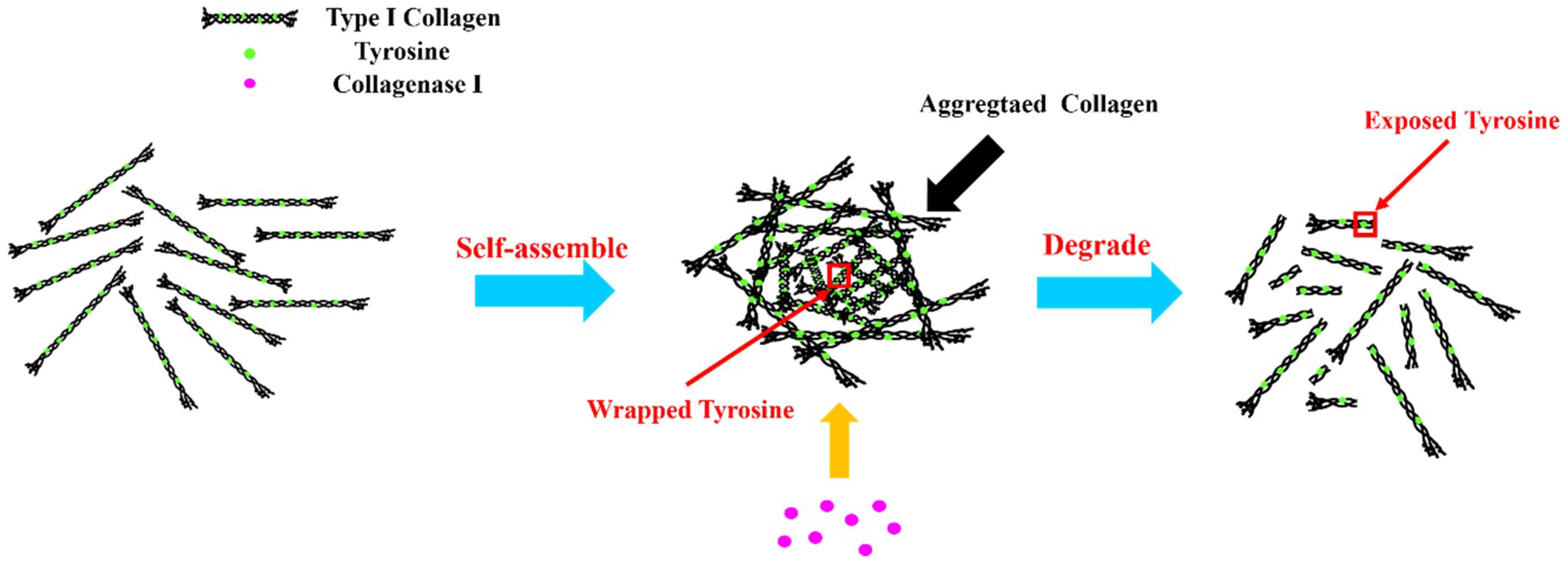
3.2. Self-Assembly of Type I Collagen
3.3. HPLC Analysis
3.4. Particle Sizes and Hydroxyproline Contents of Reaction Solution
3.5. Electron Microscope Observation of the Solution
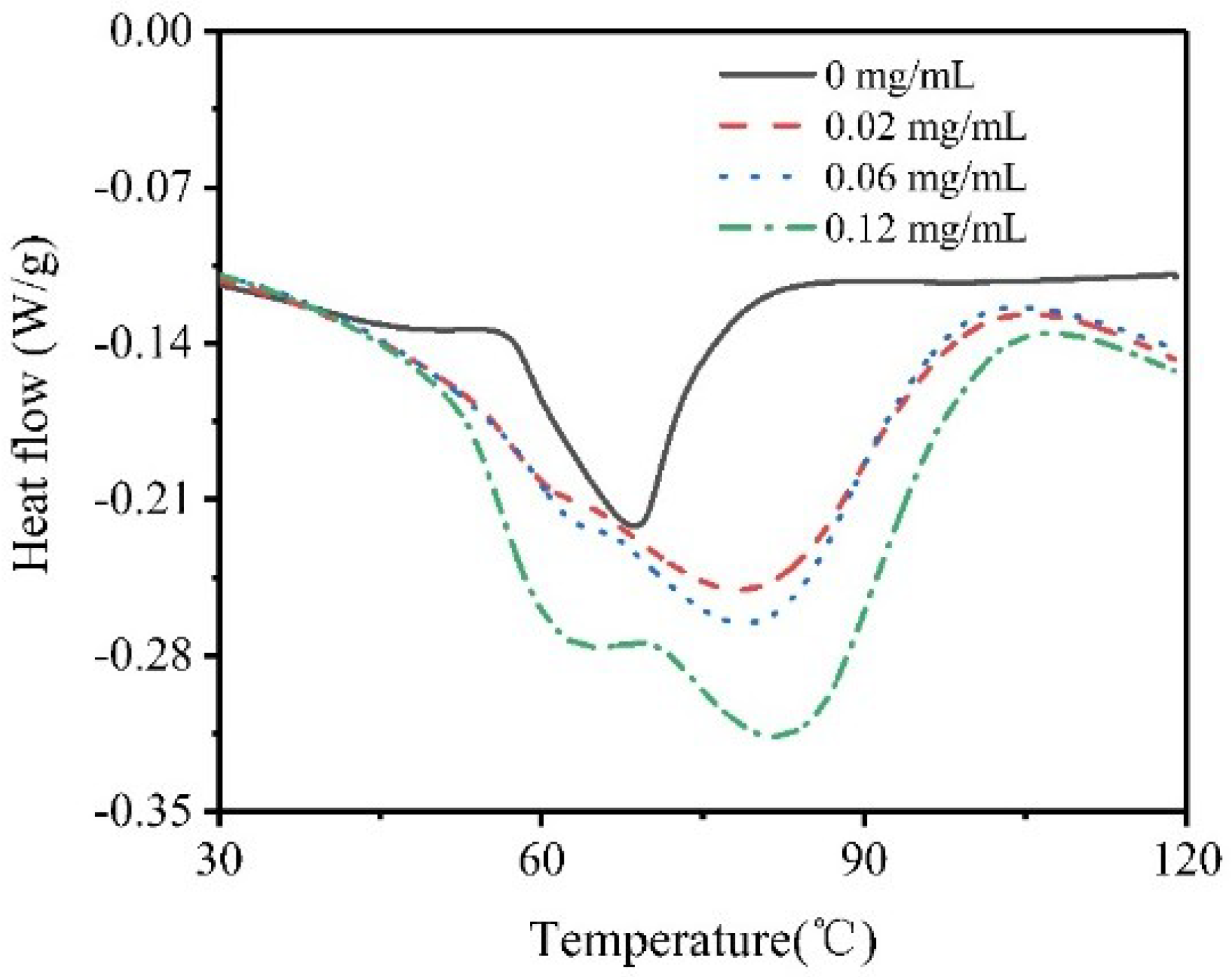
3.6. Thermal Stability of the Solution
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Chen, H.; Rhoades, E. Fluorescence characterization of denatured proteins. Curr. Opin. Struct. Biol. 2008, 18, 516–524. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.H.; Yue, J.C.; Cai, G.P. Fluorescence characterization of type I collagen from normal and silicotic rats and its quenching dynamics induced by hypocrellin B. Biopolymers 1997, 42, 219–226. [Google Scholar] [CrossRef]
- Su, D.; Wang, C.; Cai, S.; Mu, C.; Li, D.; Lin, W. Influence of palygorskite on the structure and thermal stability of collagen. Appl. Clay Sci. 2012, 62, 41–46. [Google Scholar] [CrossRef]
- Sun, X.-X.; Fan, J.; Hou, Y.-N.; Liang, S.; Zhang, Y.-P.; Xiao, J.-X. Fluorescence characterization of the thermal stability of collagen mimic peptides. Chin. Chem. Lett. 2017, 28, 963–967. [Google Scholar] [CrossRef]
- Yan, M.; Li, B.; Zhao, X. Determination of critical aggregation concentration and aggregation number of acid-soluble collagen from walleye pollock (Theragra chalcogramma) skin using the fluorescence probe pyrene. Food. Chem. 2010, 122, 1333–1337. [Google Scholar] [CrossRef]
- Masters, B.R. Principles of Fluorescence Spectroscopy, Third Edition. J. Biomed. Opt. 2008, 13, 029901. [Google Scholar] [CrossRef]
- Yan, L.I.; Wang, C.; Lan, W.Q. Isolation and purification of collagen from porcine skin and analysis of amino acid composition. Food Sci. Technol. 2007, 10, 060. [Google Scholar]
- Chen, R. Fluorescence Quantum Yields of Tryptophan and Tyrosine. Anal. Lett. 1967, 1, 35–42. [Google Scholar] [CrossRef]
- Adhikari, A.S.; Glassey, E.; Dunn, A.R. Conformational dynamics accompanying the proteolytic degradation of trimeric collagen I. by collagenases. J. Am. Chem. Soc. 2012, 134, 13259–13265. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Gao, J.; Hu, X.; Guo, H.; Wang, F.; Qiao, Y.; Wang, L. Collagen/Polyethylene Oxide Nanofibrous Membranes with Improved Hemostasis and Cytocompatibility for Wound Dressing. Appl. Sci. 2018, 8, 1226. [Google Scholar] [CrossRef]
- Di, L.G.; Sweeney, S.M.; Korkko, J.; Ala-Kokko, L.; San Antonio, J.D. Mapping the ligand-binding sites and disease-associated mutations on the most abundant protein in the human, type I collagen. J. Biol. Chem. 2002, 277, 4223–4231. [Google Scholar]
- Yao, L.; He, M.; Li, D.; Tian, J.; Liu, H.; Xiao, J. Terminal aspartic acids promote the self-assembly of collagen mimic peptides into nanospheres. RSC Adv. 2018, 8, 2404–2409. [Google Scholar] [CrossRef]
- Toyoshima, T.; Matsushita, O.; Minami, J.; Nishi, N.; Okabe, A.; Itano, T. Collagen-binding domain of a Clostridium histolyticum collagenase exhibits a broad substrate spectrum both in vitro and in vivo. Connect. Tissue Res. 2001, 42, 281–290. [Google Scholar] [CrossRef] [PubMed]
- Mu, Y.; Lin, J.; Liu, R. Interaction of sodium benzoate with trypsin by spectroscopic techniques. Spectrochim. Acta A 2011, 83, 130–135. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Tang, A.G.; Mu, S. HPLC-FLD determination of serum aromatic amino acids: Application in chronic kidney disease patients. Clin. Chim. Acta 2011, 412, 1032–1035. [Google Scholar] [CrossRef] [PubMed]
- Eva, N.Y.I.; Danilov, N.A.; Averkiev, S.V.; Obrezkova, M.V.; Lunin, V.V.; Sobol’, E.N. Determination of hydroxyproline in tissues and the evaluation of the collagen content of the tissues. J. Anal. Chem. 2007, 62, 51–57. [Google Scholar] [CrossRef]
- Chen, J.P.; Chang, G.Y.; Chen, J.K. Electrospun collagen/chitosan nanofibrous membrane as wound dressing. Colloid Surf. A 2008, 313, 183–188. [Google Scholar] [CrossRef]
- Shanmugasundaram, N.; Ravichandran, P.; Reddy, P.N.; Ramamurty, N.; Pal, S.; Rao, K.P. Collagen-chitosan polymeric scaffolds for the in vitro culture of human epidermoid carcinoma cells. Biomaterials 2001, 22, 1943–1951. [Google Scholar] [CrossRef]
- Vanscyoc, W.S.; Sorensen, B.R.; Rusinova, E.; Laws, W.R.; Ross, J.B.A.; Shea, M.A. Calcium binding to calmodulin mutants monitored by domain-specific intrinsic phenylalanine and tyrosine fluorescence. Biophys. J. 2002, 83, 2767–2780. [Google Scholar] [CrossRef]
- Persikov, A.V.; Ramshaw, J.A.M.; Kirkpatrick, A.; Brodsky, B. Amino acid propensities for the collagen triple-helix. Biochemistry 2000, 39, 14960–14967. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Yang, J.; Yang, Q.; Huang, L.; Wu, H.; Chen, L.; Ding, C. Fluorescence studies on the aggregation behaviors of collagen modified with NHS-activated poly(γ-glutamic acid). Int. J. Biol. Macromol. 2018, 112, 1156–1163. [Google Scholar] [CrossRef] [PubMed]
- Menter, J.M. Temperature dependence of collagen fluorescence. Photochem. Photobiol. 2006, 5, 403–410. [Google Scholar] [CrossRef] [PubMed]
- Wu, K.; Liu, W.; Li, G. The aggregation behavior of native collagen in dilute solution studied by intrinsic fluorescence and external probing. Spectrochim. Acta A 2013, 102, 186–193. [Google Scholar] [CrossRef] [PubMed]
- Bauer, R.; Wilson, J.J.; Dan, D.; Matsushita, O.; Sakon, J. Structural Comparison of ColH and ColG Collagen-Binding Domains from Clostridium histolyticum. J. Bacteriol. 2013, 195, 318–327. [Google Scholar] [CrossRef] [PubMed]
- Eckhard, U.; Huesgen, P.F.; Brandstetter, H.; Overall, C.M. Proteomic protease specificity profiling of clostridial collagenases reveals their intrinsic nature as dedicated degraders of collagen. J. Proteom. 2014, 100, 102–114. [Google Scholar] [CrossRef] [PubMed]
- Madden, A.S.; Hochella, M.F., Jr. A test of geochemical reactivity as a function of mineral size: Manganese oxidation promoted by hematite nanoparticles. Geochim. Cosmochim. Acta 2005, 69, 389–398. [Google Scholar] [CrossRef]
- Paige, M.F.; Goh, M.C. Ultrastructure and assembly of segmental long spacing collagen studied by atomic force microscopy. Micron 2001, 32, 355–361. [Google Scholar] [CrossRef]
- O’Leary, L.E.R.; Fallas, J.A.; Bakota, E.L.; Kang, M.K.; Hartgerink, J.D. Multi-hierarchical self-assembly of a collagen mimetic peptide from triple helix to nanofibre and hydrogel. Nat. Chem. 2011, 3, 821. [Google Scholar] [CrossRef] [PubMed]
- Ottani, V.; Martini, D.; Franchi, M.; Ruggeri, A.; Raspanti, M. Hierarchical structures in fibrillar collagens. Micron 2002, 33, 587–596. [Google Scholar] [CrossRef]
- Ottani, V.; Raspanti, M.; Ruggeri, A. Collagen structure and functional implications. Micron 2001, 32, 251–260. [Google Scholar] [CrossRef]
- Mo, X.M.; Li, Y.; Tang, A.G.; Ren, Y.P. Simultaneous determination of phenylalanine and tyrosine in peripheral capillary blood by HPLC with ultraviolet detection. Clin. Biochem. 2013, 46, 1074–1078. [Google Scholar] [CrossRef] [PubMed]
- Yamazaki, C.M.; Asada, S.; Kitagawa, K.; Koide, T. Artificial collagen gels via self-assembly of de novo designed peptides. Biopolymers 2010, 90, 816–823. [Google Scholar] [CrossRef] [PubMed]
- Pietrucha, K. Changes in denaturation and rheological properties of collagen-hyaluronic acid scaffolds as a result of temperature dependencies. Int. J. Biol. Macromol. 2005, 36, 299–304. [Google Scholar] [CrossRef] [PubMed]
- He, L.; Mu, C.; Shi, J.; Zhang, Q.; Shi, B.; Lin, W. Modification of collagen with a natural cross-linker, procyanidin. Int. J. Biol. Macromol. 2011, 48, 354–359. [Google Scholar] [CrossRef] [PubMed]
- Cucos, A.; Budrugeac, P. Simultaneous TG/DTG–DSC–FTIR characterization of collagen in inert and oxidative atmospheres. J. Therm. Anal. Calorim. 2014, 115, 2079–2087. [Google Scholar] [CrossRef]


© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shen, Y.; Zhu, D.; Lu, W.; Liu, B.; Li, Y.; Cao, S. The Characteristics of Intrinsic Fluorescence of Type I Collagen Influenced by Collagenase I. Appl. Sci. 2018, 8, 1947. https://doi.org/10.3390/app8101947
Shen Y, Zhu D, Lu W, Liu B, Li Y, Cao S. The Characteristics of Intrinsic Fluorescence of Type I Collagen Influenced by Collagenase I. Applied Sciences. 2018; 8(10):1947. https://doi.org/10.3390/app8101947
Chicago/Turabian StyleShen, Yiming, Deyi Zhu, Wenhui Lu, Bing Liu, Yanchun Li, and Shan Cao. 2018. "The Characteristics of Intrinsic Fluorescence of Type I Collagen Influenced by Collagenase I" Applied Sciences 8, no. 10: 1947. https://doi.org/10.3390/app8101947
APA StyleShen, Y., Zhu, D., Lu, W., Liu, B., Li, Y., & Cao, S. (2018). The Characteristics of Intrinsic Fluorescence of Type I Collagen Influenced by Collagenase I. Applied Sciences, 8(10), 1947. https://doi.org/10.3390/app8101947




