Supercritical Transesterification of Waste Vegetable Oil: Characteristic Comparison of Ethanol and Methanol as Solvents
Abstract
:Featured Application
Abstract
1. Introduction
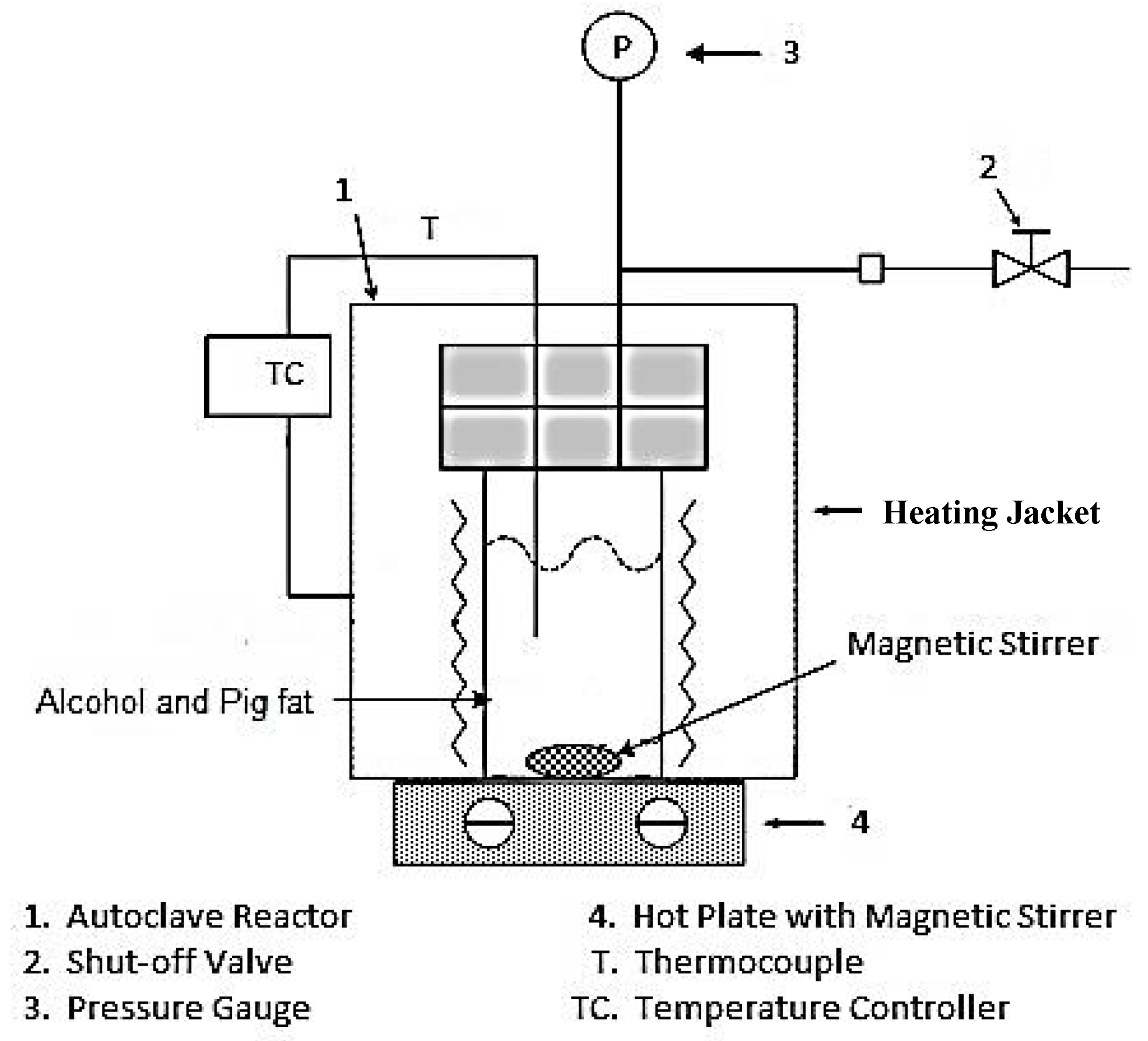
2. Raw Material, Experimental Device and Procedure
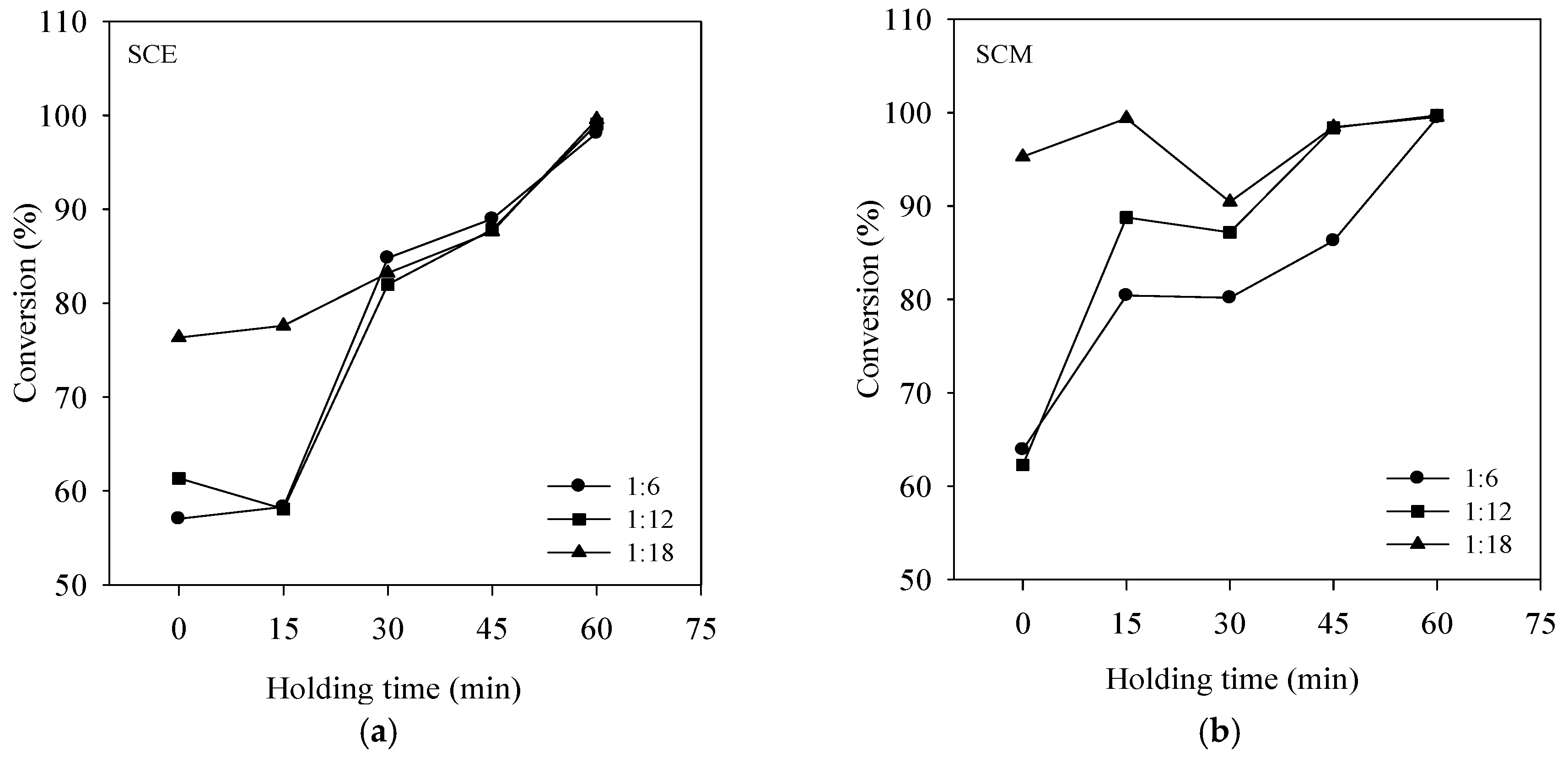
3. Results and Discussion
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Munack, A.; Schröder, O.; Krahl, J.; Bünger, J. Comparison of relevant exhaust gas emissions from biodiesel and fossil diesel fuel. International Commission of Agricultural Engineering. Agric. Eng. 2001, 3, 1–8. [Google Scholar]
- Saravanan, S.; Nagarajan, G.; Rao, G.L.N.; Sampath, S. Feasibility study of crude rice bran oil as a diesel substitute in a DI-CI engine without modifications. Energy Sustain. Dev. 2007, 11, 83–92. [Google Scholar] [CrossRef]
- Owolabi, R.; Osiyemi, N.; Amosa, M.; Ojewumi, M. Biodiesel from household/restaurant waste cooking oil (WCO). J. Chem. Eng. Process. Technol. 2011, 2, 700. [Google Scholar]
- Van Gerpen, J. Biodiesel processing and production. Fuel Process Technol. 2005, 86, 1097–1107. [Google Scholar] [CrossRef]
- Srivastava, A.; Prasad, R. Triglycerides-based diesel fuels. Renew. Sustain. Energ. Rev. 2000, 4, 111–133. [Google Scholar] [CrossRef]
- Demirbas, A. Progress and recent trends in biodiesel fuels. Energ. Convers. Manag. 2009, 50, 14–34. [Google Scholar] [CrossRef]
- Alcantara, R.; Amores, J.; Canoira, L.T.; Fidalgo, E.; Franco, M.; Navarro, A. Catalytic production of biodiesel from soy-bean oil, used frying oil and tallow. Biomass Bioenergy 2000, 18, 515–527. [Google Scholar] [CrossRef]
- Mendes, A. Production of Biodiesel from Corn Oil and Ethanol by Homogeneous Alkali Catalyzed Transesterification. Available online: http://www.diva--portal.org/smash/record.jsf?pid=diva2%3A414004&dswid=-3878 (accessed on 12 June 2016).
- Khaligh, N.G.; Hamid, S.B.A.; Mihankhah, T. Eco-friendly Biodiesel Production from Waste Olive Oil by Transesterification using Nano-tube TiO2. In In Proceedings of the International Conference of Social Science, Medicine and Nursing, Istanbul, Turkey, 5–6 June 2015. [Google Scholar]
- Royon, D.; Daz, M.; Ellenrieder, G.; Locatelli, S. Enzymatic production of biodiesel from cotton seed oil using t-butanol as a solvent. Bioresour. Technol. 2007, 98, 648–653. [Google Scholar] [CrossRef] [PubMed]
- Rashid, U.; Anwar, F. Production of biodiesel through optimized alkaline-catalyzed transesterification of rapeseed oil. Fuel 2008, 87, 265–273. [Google Scholar] [CrossRef]
- Shu, Q.; Gao, J.; Nawaz, Z.; Liao, Y.; Wang, D.; Wang, J. Synthesis of biodiesel from waste vegetable oil with largee amounts of free fatty acids using a carbon-based solid acid catalyst. Appl. Energy 2010, 87, 2589–2596. [Google Scholar] [CrossRef]
- Alexander, C.; Hurt, C. Biofuels and Their Impact on Food Prices. 2007. Available online: https://www.extension.purdue.edu/extmedia/id/id-346-w.pdf (accessed on 1 April 2014).
- Phan, A.N.; Phan, T.M. Biodiesel production from waste cooking oils. Fuel 2008, 87, 3490–3496. [Google Scholar] [CrossRef]
- Meng, X.; Chen, G.; Wang, Y. Biodiesel production from waste cooking oil via alkali catalyst and its engine test. Fuel Process. Technol. 2008, 89, 851–857. [Google Scholar] [CrossRef]
- Chhetri, A.B.; Watts, K.C.; Islam, M.R. Waste cooking oil as an alternate feedstock for biodiesel production. Energies 2008, 1, 3–18. [Google Scholar] [CrossRef]
- Da Silva, N.D.L.; Rivera, E.C.; Batistella, C.B.; Maciel Filho, R.; Maciel, M.R.W. Biodiesel production from vegetable oils: Operational strategies for large scale systems. Comput. Aided Chem. Eng. 2008, 25, 1101–1106. [Google Scholar]
- Ngamprasertsith, S.; Sawangkeaw, R. Transesterification in supercritical conditions. In Biodiesel-Feedstocks and Processing Technologies; InTechOpen: Rijeka, Croatia, 2011; pp. 247–268. [Google Scholar]
- Meher, L.; Sagar, D.V.; Naik, S. Technical aspects of biodiesel production by transesterification—A review. Renew. Sustain. Energ. Rev. 2006, 10, 248–268. [Google Scholar] [CrossRef]
- Felizardo, P.; Correia, M.J.N.; Raposo, I.; Mendes, J.F.; Berkemeier, R.; Bordado, J.M. Production of biodiesel from waste frying oils. Waste Manag. 2006, 26, 487–494. [Google Scholar] [CrossRef] [PubMed]
- Chouhan, A.S.; Sarma, A. Modern heterogeneous catalysts for biodiesel production: A comprehensive review. Renew. Sustain. Energy Rev. 2011, 15, 4378–4399. [Google Scholar] [CrossRef]
- Di Serio, M.; Tesser, R.; Pengmei, L.; Santacesaria, E. Heterogeneous catalysts for biodiesel production. Energy Fuels 2007, 22, 207–217. [Google Scholar] [CrossRef]
- Warabi, Y.; Kusdiana, D.; Saka, S. Reactivity of triglycerides and fatty acids of rapeseed oil in supercritical alcohols. Bioresour. Technol. 2004, 91, 283–287. [Google Scholar] [CrossRef]
- Köckritz, A.; Martin, A. Oxidation of unsaturated fatty acid derivatives and vegetable oils. Eur. J. Lipid Sci. Technol. 2008, 110, 812–824. [Google Scholar] [CrossRef]
- Demirbaş, A. Biodiesel from vegetable oils via transesterification in supercritical methanol. Energy Convers. Manag. 2002, 43, 2349–2356. [Google Scholar] [CrossRef]
- Martini, N.; Schell, J.S. Plant oils as fuels: Present state of science and future developments. J. Taiwan Inst. Chem. Eng. 2016. [Google Scholar] [CrossRef]
- Demirbas, A. Biodiesel from waste cooking oil via base-catalytic and supercritical methanol transesterification. Energy Convers. Manag. 2009, 50, 923–927. [Google Scholar] [CrossRef]
- Lam, M.K.; Lee, K.T.; Mohamed, A.R. Homogeneous, heterogeneous and enzymatic catalysis for transesterification of high free fatty acid oil (waste cooking oil) to biodiesel: A review. Biotechnol. Adv. 2010, 28, 500–518. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Dube, M.; McLean, D.; Kates, M. Biodiesel production from waste cooking oil: 1. Process design and technological assessment. Bioresour. Technol. 2003, 89, 1–16. [Google Scholar] [CrossRef]
- Sanjel, N.; Gu, J.H.; Oh, S.C. Transesterification Kinetics of Waste Vegetable Oil in Supercritical Alcohols. Energies 2014, 7, 2095–2106. [Google Scholar] [CrossRef]
- Saka, S.; Kusdiana, D. Biodiesel fuel from rapeseed oil as prepared in supercritical methanol. Fuel 2001, 80, 225–231. [Google Scholar] [CrossRef]
- Hoffmann, M.M.; Conradi, M.S. Are there hydrogen bonds in supercritical methanol and ethanol? J. Phys. Chem. B 1998, 102, 263–271. [Google Scholar] [CrossRef]
- Madras, G.; Kolluru, C.; Kumar, R. Synthesis of biodiesel in supercritical fluids. Fuel 2004, 83, 2029–2033. [Google Scholar] [CrossRef]
- Tan, K.T.; Gui, M.M.; Lee, K.T.; Mohamed, A.R. An optimized study of methanol and ethanol in supercritical alcohol technology for biodiesel production. J. Supercrit. Fluids 2010, 53, 82–87. [Google Scholar] [CrossRef]
- Tan, K.T.; Gui, M.M.; Lee, K.T.; Mohamed, A.R. Supercritical alcohol technology in biodiesel production: A comparative study between methanol and ethanol. Energy Sour. Part A. 2010, 33, 156–163. [Google Scholar] [CrossRef]
- Minami, E.; Saka, S. Kinetics of hydrolysis and methyl esterification for biodiesel production in two-step supercritical methanol process. Fuel 2006, 85, 2479–2483. [Google Scholar] [CrossRef]
- Schwab, A.W.; Dykstra, G.J.; Selke, E.; Sorenson, S.C.; Pryde, E.H. Diesel Fuel from Thermal Decomposition of Soybean Oil. JAOCS 1988, 65, 1781–1786. [Google Scholar] [CrossRef]
- Tomasevic, A.; Siler-Marinkovic, S. Methanolysis of used frying oil. Fuel Process. Technol. 2003, 81, 1–6. [Google Scholar] [CrossRef]
- Zhou, W.; Konar, S.K.; Boocock, D.G. Ethyl esters from the single-phase base-catalyzed ethanolysis of vegetable oils. J. Am. Oil Chem. Soc. 2003, 80, 367–371. [Google Scholar] [CrossRef]
- Poudel, J.; Shah, M.; Karki, S.; Oh, S.C. Qualitative Analysis of Transesterification of Waste Pig Fat in Supercritical Alcohols. Energies 2017, 10, 265. [Google Scholar] [CrossRef]
- Barlow, S.J.; Bondarenko, G.V.; Gorbaty, Y.E.; Yamaguchi, T.; Poliakoff, M. An IR study of hydrogen bonding in liquid and supercritical alcohols. J. Phys. Chem. A 2002, 106, 10452–10460. [Google Scholar] [CrossRef]
- Zhang, Y.; Yang, J.; Yu, Y.; Li, Y. Structural and hydrogen bond analysis for supercritical ethanol: A molecular simulation study. J. Supercrit. Fluids 2005, 36, 145–153. [Google Scholar] [CrossRef]
- Poudel, J.; Oh, S.C. A kinetic analysis of wood degradation in supercritical alcohols. Ind. Eng. Chem. Res. 2012, 51, 4509–4514. [Google Scholar] [CrossRef]
- Gui, M.M.; Lee, K.T.; Bhatia, S. Supercritical ethanol technology for the production of biodiesel: Process optimization studies. J. Supercrit. Fluids 2009, 49, 286–292. [Google Scholar] [CrossRef]

| Sample | FA | FFA | |||||
|---|---|---|---|---|---|---|---|
| 13:2 | 16:1 | 18:1 | 18:2 | 18:3 | Others | ||
| Virgin Oil | 6.49 | 9.86 | 2.41 | 13.58 | 58.48 | 9.19 | 0.28 |
| WVO | 3.38 | 9.89 | 2.41 | 36.70 | 43.21 | 4.42 | 3.15 |
| Esters | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 0 min | |||||||||||||
| Temperature (°C) | Glycerol | C10:2 | C12:0 | C15:0 | C17:0 | C17:1 | C18:2 | C18:3 | C19:0 | C19:1 | C21:1 | C28:0 | Non-esters |
| 250 | - | 1.79 | 0.57 | - | 16.61 | - | 40.48 | 4.64 | - | 35.89 | - | - | 0.02 |
| 270 | - | 0.64 | 0.45 | - | 16.09 | - | 42.15 | 4.68 | - | 35.80 | - | - | 0.19 |
| 290 | - | - | 2.68 | 0.30 | 15.37 | 1.03 | 37.04 | 4.89 | - | 37.26 | - | - | 1.43 |
| 15 min | |||||||||||||
| 250 | - | - | 0.34 | 0.20 | 16.05 | - | 42.47 | 4.80 | - | 35.93 | - | - | 0.21 |
| 270 | - | 0.13 | 0.26 | 0.20 | 16.59 | - | 40.36 | 4.93 | - | 36.39 | 1.14 | - | 1.14 |
| 290 | - | 0.07 | 3.36 | 0.28 | 14.64 | 1.06 | 38.19 | 5.30 | - | 36.18 | - | - | 0.92 |
| 30 min | |||||||||||||
| 250 | - | 0.19 | 0.47 | 0.21 | 16.10 | - | 41.52 | 4.72 | - | 35.68 | - | - | 1.11 |
| 270 | - | 0.11 | - | 0.20 | 16.52 | - | 39.57 | 5.42 | - | 36.57 | 1.27 | - | 1.61 |
| 290 | 0.66 | 0.09 | 3.02 | 0.26 | 14.64 | 0.92 | 38.48 | 5.35 | - | 36.31 | - | 0.07 | 0.27 |
| 45 min | |||||||||||||
| 250 | - | 0.41 | 0.14 | 0.20 | 16.53 | - | 39.93 | 5.44 | - | 36.35 | 1.01 | - | 1.00 |
| 270 | 0.22 | 0.11 | - | 0.21 | 16.58 | - | 38.69 | 5.41 | 0.32 | 36.69 | 1.54 | - | 1.77 |
| 290 | 1.14 | - | 2.72 | 0.27 | 14.21 | 1.18 | 39.58 | 5.45 | - | 35.45 | - | - | |
| 60 min | |||||||||||||
| 250 | - | 0.29 | 0.23 | 0.21 | 16.79 | - | 39.75 | 5.07 | - | 36.74 | 0.91 | - | 0.92 |
| 270 | 0.37 | 0.20 | - | 0.20 | 16.75 | - | 38.79 | 5.39 | - | 36.87 | 1.32 | - | 1.43 |
| 290 | 1.27 | 0.08 | 1.19 | 0.27 | 13.97 | 1.08 | 39.84 | 5.34 | 0.81 | 35.76 | - | - | 0.39 |
| Esters | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 0 min | ||||||||||
| Temperature (°C) | Glycerol | C10:2 | C12:0 | C16:0 | C18:0 | C20:0 | C20:1 | C20:2 | C20:3 | Non-esters |
| 250 | - | 11.20 | 13.78 | - | 13.79 | - | 24.21 | 34.22 | 2.80 | |
| 270 | - | 0.57 | - | - | 16.00 | 2.71 | 33.45 | 43.45 | 3.81 | 0.01 |
| 290 | - | - | 0.34 | - | 16.15 | - | 37.11 | 43.37 | 3.03 | - |
| 15 min | ||||||||||
| 250 | - | 0.54 | - | - | 15.15 | 3.36 | 32.07 | 43.75 | 5.13 | - |
| 270 | - | - | 0.07 | - | 15.53 | 2.72 | 34.38 | 43.74 | 3.37 | 0.19 |
| 290 | - | - | - | - | 16.33 | - | 36.78 | 43.20 | 3.50 | 0.19 |
| 30 min | ||||||||||
| 250 | - | 0.33 | - | - | 15.90 | 2.69 | 33.46 | 42.55 | 5.03 | 0.04 |
| 270 | - | - | - | - | 16.45 | - | 37.22 | 42.89 | 3.30 | 0.14 |
| 290 | - | - | 0.61 | 0.13 | 16.53 | - | 37.06 | 40.76 | 4.91 | - |
| 45 min | ||||||||||
| 250 | - | 0.21 | 0.37 | - | 16.33 | - | 36.41 | 41.36 | 5.08 | 0.24 |
| 270 | - | - | - | 0.18 | 16.16 | - | 37.07 | 40.54 | 5.24 | 0.81 |
| 290 | - | - | - | 0.16 | 16.08 | - | 37.16 | 40.71 | 4.90 | 0.99 |
| 60 min | ||||||||||
| 250 | - | 0.18 | 0.70 | 0.23 | 15.96 | - | 36.61 | 40.89 | 5.10 | 0.33 |
| 270 | 0.83 | - | - | 0.16 | 16.11 | - | 37.22 | 39.81 | 5.36 | 0.51 |
| 290 | 0.93 | - | 0.08 | 0.22 | 15.78 | - | 37.19 | 40.63 | 4.86 | 0.31 |
| Esters | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 0 min | ||||||||||||||
| M.R | Glycerol | C10:2 | C12:0 | C15:0 | C17:0 | C17:1 | C18:0 | C18:2 | C18:3 | C19:0 | C19:1 | C21:0 | C28:0 | Non-esters |
| 1:6 | - | - | 2.68 | 0.30 | 15.37 | 1.03 | - | 37.04 | 4.89 | - | 37.26 | - | - | 1.43 |
| 1:12 | - | 0.17 | 1.21 | 0.25 | 14.98 | 0.82 | - | 37.70 | 5.12 | - | 37.63 | - | 0.02 | 2.12 |
| 1:18 | 2.64 | 0.55 | 1.12 | 0.28 | 14.93 | 0.97 | - | 36.16 | 4.82 | - | 37.05 | 0.98 | - | 1.48 |
| 15 min | ||||||||||||||
| 1:6 | - | 0.07 | 3.36 | 0.28 | 14.64 | 1.06 | - | 38.19 | 5.30 | - | 36.18 | - | - | 0.92 |
| 1:12 | 1.71 | 0.11 | 1.09 | 0.25 | 14.67 | 0.76 | - | 37.53 | 5.05 | 1.78 | 35.40 | - | - | 1.65 |
| 1:18 | 2.60 | 0.15 | 0.99 | 0.26 | 14.45 | 0.92 | - | 37.33 | 5.26 | - | 36.20 | - | - | 1.84 |
| 30 min | ||||||||||||||
| 1:6 | 0.66 | 0.09 | 3.02 | 0.26 | 14.64 | 0.92 | - | 38.48 | 5.35 | - | 36.31 | - | 0.07 | 0.27 |
| 1:12 | 1.79 | 0.09 | 0.69 | 0.26 | 14.33 | 0.96 | - | 39.65 | 5.24 | - | 35.98 | - | - | 1.01 |
| 1:18 | 2.56 | 0.08 | 0.70 | 0.26 | 14.09 | 0.85 | - | 37.49 | 5.39 | 1.84 | 35.83 | - | 0.13 | 0.91 |
| 45 min | ||||||||||||||
| 1:6 | 1.14 | - | 2.72 | 0.27 | 14.21 | 1.18 | - | 39.58 | 5.45 | - | 35.45 | - | - | - |
| 1:12 | 2.44 | 0.05 | 1.28 | 0.26 | 13.66 | 1.03 | 0.33 | 39.77 | 5.39 | - | 35.53 | - | 0.06 | 0.26 |
| 1:18 | 2.58 | 0.08 | 0.22 | 0.26 | 14.07 | 0.79 | 0.21 | 37.71 | 5.40 | 2.30 | 35.94 | - | - | 0.44 |
| 60 min | ||||||||||||||
| 1:6 | 1.27 | 0.08 | 1.19 | 0.27 | 13.97 | 1.08 | 39.84 | 5.34 | 0.81 | 35.76 | - | - | 0.39 | |
| 1:12 | 2.44 | - | 0.60 | 0.27 | 13.95 | 0.98 | 0.23 | 39.60 | 5.07 | 0.79 | 35.69 | - | - | 0.38 |
| 1:18 | 2.60 | 0.07 | 0.32 | 0.26 | 14.24 | 0.88 | 0.20 | 39.75 | 4.91 | 0.48 | 35.87 | - | - | 0.42 |
| Esters | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 0 min | |||||||||||
| M.R | Glycerol | C10:2 | C12:0 | C16:0 | C17:0 | C18:0 | C19:1 | C20:1 | C20:2 | C20:3 | Non-esters |
| 1:6 | - | - | 0.34 | - | - | 16.15 | - | 37.11 | 43.37 | 3.03 | 0.00 |
| 1:12 | - | 0.45 | 0.04 | - | - | 16.50 | - | 37.63 | 42.10 | 2.96 | 0.32 |
| 1:18 | - | 0.78 | - | - | - | 17.75 | - | 37.25 | 39.83 | 4.15 | 0.24 |
| 15 min | |||||||||||
| 1:6 | - | - | - | - | - | 16.33 | - | 36.78 | 43.20 | 3.50 | 0.19 |
| 1:12 | 1.44 | 0.18 | 0.08 | 0.23 | 0.30 | 15.75 | 0.72 | 37.38 | 38.86 | 4.67 | 0.39 |
| 1:18 | - | - | - | 0.28 | - | 13.31 | - | 35.63 | 41.60 | 5.53 | 3.65 |
| 30 min | |||||||||||
| 1:6 | - | - | 0.61 | 0.13 | - | 16.53 | - | 37.06 | 40.76 | 4.91 | 0.00 |
| 1:12 | 2.00 | 0.11 | - | 0.24 | 1.70 | 13.90 | 4.15 | 36.54 | 35.77 | 4.38 | 1.21 |
| 1:18 | 3.94 | - | - | 0.21 | - | 13.18 | - | 35.49 | 41.06 | 5.41 | 0.71 |
| 45 min | |||||||||||
| 1:6 | - | - | - | 0.16 | - | 16.08 | - | 37.16 | 40.71 | 4.90 | 0.99 |
| 1:12 | 2.15 | - | 0.32 | 0.25 | 0.13 | 14.65 | - | 36.33 | 39.64 | 5.13 | 1.40 |
| 1:18 | 3.95 | - | - | 0.21 | - | 13.43 | - | 35.55 | 41.43 | 5.42 | 0.01 |
| 60 min | |||||||||||
| 1:6 | 0.93 | - | 0.08 | 0.22 | - | 15.78 | - | 37.19 | 40.63 | 4.86 | 0.31 |
| 1:12 | 4.21 | - | - | 0.23 | - | 13.58 | - | 35.66 | 39.74 | 5.73 | 0.85 |
| 1:18 | 4.20 | - | 0.17 | 0.21 | - | 13.23 | - | 35.38 | 40.37 | 5.62 | 0.82 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Karki, S.; Sanjel, N.; Poudel, J.; Choi, J.H.; Oh, S.C. Supercritical Transesterification of Waste Vegetable Oil: Characteristic Comparison of Ethanol and Methanol as Solvents. Appl. Sci. 2017, 7, 632. https://doi.org/10.3390/app7060632
Karki S, Sanjel N, Poudel J, Choi JH, Oh SC. Supercritical Transesterification of Waste Vegetable Oil: Characteristic Comparison of Ethanol and Methanol as Solvents. Applied Sciences. 2017; 7(6):632. https://doi.org/10.3390/app7060632
Chicago/Turabian StyleKarki, Sujeeta, Nawaraj Sanjel, Jeeban Poudel, Ja Hyung Choi, and Sea Cheon Oh. 2017. "Supercritical Transesterification of Waste Vegetable Oil: Characteristic Comparison of Ethanol and Methanol as Solvents" Applied Sciences 7, no. 6: 632. https://doi.org/10.3390/app7060632
APA StyleKarki, S., Sanjel, N., Poudel, J., Choi, J. H., & Oh, S. C. (2017). Supercritical Transesterification of Waste Vegetable Oil: Characteristic Comparison of Ethanol and Methanol as Solvents. Applied Sciences, 7(6), 632. https://doi.org/10.3390/app7060632






