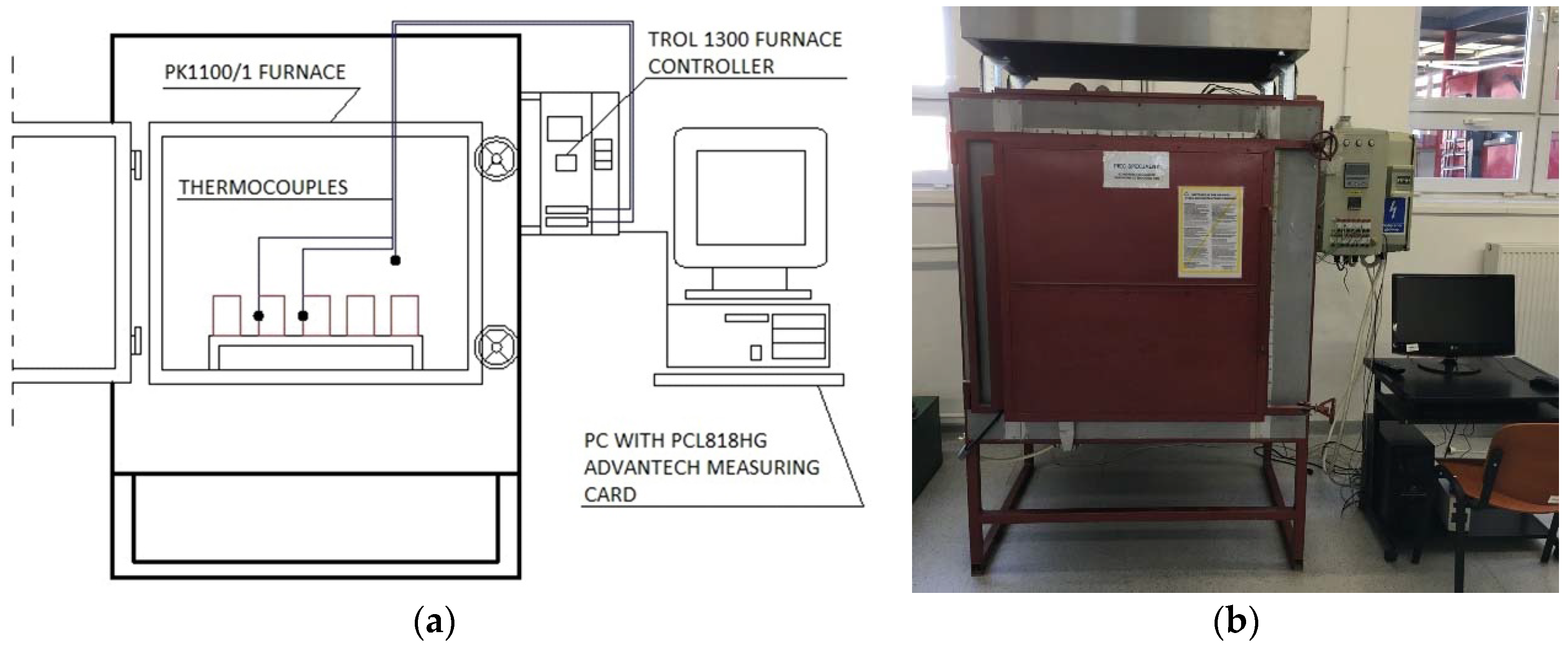
5.1. Stage 1—Initial Assesment of the TES Properties at Lower Temperature
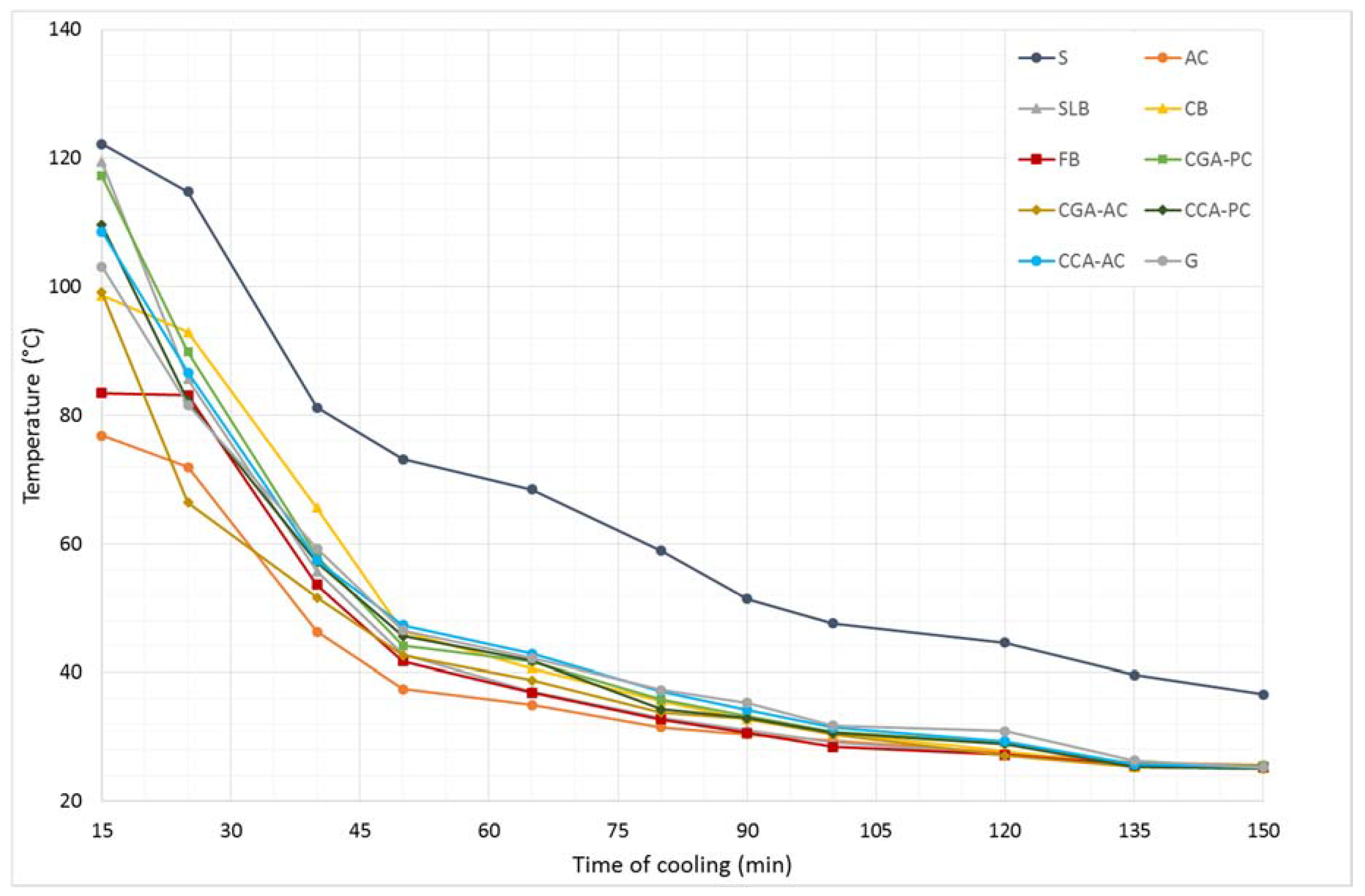
Results from the first stage of the experiment are shown in
Figure 4.
In spite of the same size of the samples and the fact that they were left in the same thermal conditions, they showed different thermal characteristics. After the heating process, the specimens had different temperatures. None of the materials, after 3 h of heating did not reach the set temperature. After 15 min of cooling, the highest temperature reached was the steel specimen (122.2 °C), while the lowest was the sample made of the aerated concrete (76.8 °C). The reason for the differences, while heating and cooling of these materials, is their structure and internal interactions between the material’s molecules. Steel is a compact material which has the highest density, and the least amount of voids and free spaces inside its volume in comparison to the other materials tested. In addition, the high thermal conductivity of steel is due to the metallic bonding between molecules. An aerated concrete is radically different in terms of its structure: it has low density and high porosity; the appearance of air voids makes it an insulating material with a low heat transfer coefficient. The porosity of the surface of the aerated concrete gives it the largest contact surface with the cooler room air of all the materials tested. Such an effect unfavorably influences the emission time of the heat stored—the sample very quickly drops in temperature.
The analysis of the temperature drops during further cooling confirms this pattern. The steel sample, despite the fact that after 15 min reached a temperature close to the concrete samples (steel: 122.2 °C; ordinary concrete: 117.2 °C), it remained warm the longest, and the temperature drops in the subsequent intervals were the lowest. The aerated concrete specimen cooled down in the fastest time.
The analysis of the thermal behavior of other samples indicates the superiority of the concretes (including concrete with the ceramic aggregate) over the traditional ceramics in terms of the TES issues. Concrete samples as the more compact material with higher density, heat up at the same time to the higher temperatures under the same conditions (concrete samples: over 100 °C; ceramic samples: less than 100 °C).
The calculation of the thermal power is shown in
Table 7. During the analysis of the results, it is noted that the highest thermal power reached was in the steel (8.8 W), and the lowest were concretes (5.3−6.6 W). The lowest thermal power was found in the aerated concrete (1.616 W) and the fireclay brick (2.408 W).
5.2. Stage 2—TES Properties in the Fireplace Operational Conditions
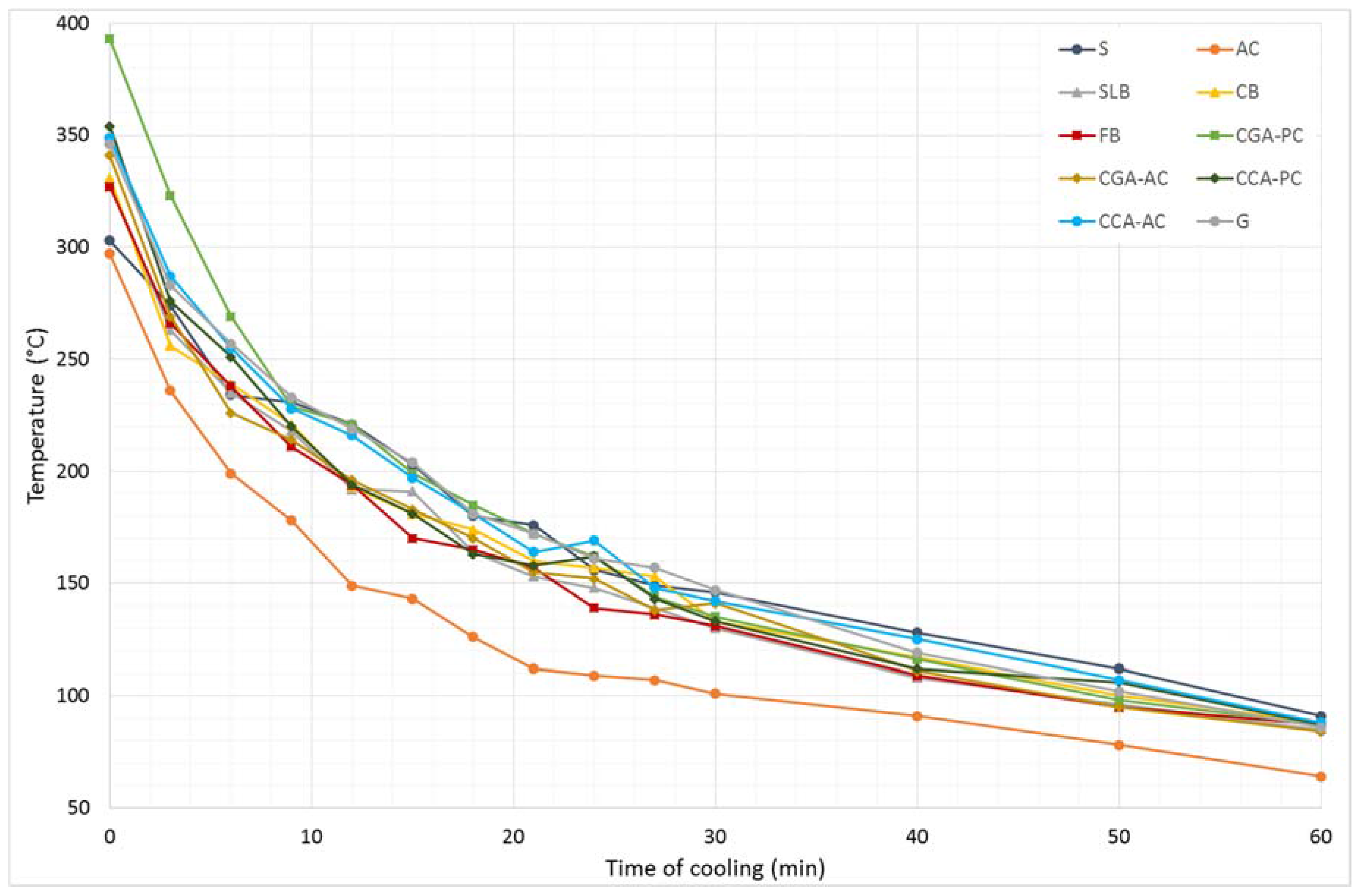
Results from the second stage of the experiment, in which samples were heated up to 400 °C, are shown in
Figure 5.
The highest temperature reached was in the CGA-PC specimen (393 °C), while the lowest—similar to the first stage— was the AC sample (297 °C). As the heating temperature increases, the differences in the final temperature of the materials also increases. In this case, the temperature of steel (303 °C), immediately after heating, was only higher from the AC. This shows that the thermal properties of the materials, besides the material’s structure and type of bonding between molecules, also depend on the temperature at which the materials operate.
The temperature drops during cooling showing that aerated concrete cooled down in the shortest time, but a different cooling pattern was observed for the steel sample (in comparison to the first stage). The S sample was characterized by the lowest rate of the temperature drop in a function of the cooling time. After 9 min of cooling the S sample had a higher temperature than all concretes tested and, after 12 min, the S specimen had the highest temperature of all the materials.
The initial temperature after heating of the concretes made with ceramic aggregate was higher than almost all materials, i.e., CCA-PC and CCA-AC, at 354 °C and 349 °C, respectively. Only the CGA-PC sample obtained higher initial temperature, at 393 °C. During the first stage, more materials (S, SLB, and CGA-PC) obtained higher initial temperatures in comparison to the ceramic concretes. The test at 400 °C shows that the TES properties of concretes based on ceramic aggregate are better when the operational temperature increases, compared to other materials.
In the case of the temperature measurement using an infrared camera, when a high-temperature object (interior of the furnace) is reflected, an adulteration of the measurement results may appear unless the ambient temperature is considered. During measurements, the angle of the observation did not exceed 30°, and the emissivity coefficients of the materials’ surface were taken into account.
The analysis of thermal images was carried out using the comparative method. The samples’ temperatures were compared, which were obtained from the thermograms made at one time for the materials. The brightest color on the thermal image during the entire cooling period was observed for the steel sample, which was in agreement with the temperature reading from the thermocouple. The darkest color was recorded for the aerated concrete sample. This fact was consistent with the temperature readings—the AC sample reached the lowest temperature during the heating process, and the temperatures recorded for it in subsequent points in time were also the lowest. The thermographic examination was carried out in order to detect possible anomalies, i.e., an intensified infrared radiation for selected materials, which would not be compatible with temperature readings obtained from the thermocouples. The thermal images of the ceramic materials, concretes, and natural stone were similar in quality. This fact may indicate that concrete composites can fulfill the functions of heat radiators that have traditionally been made in old fireplaces made of ceramic and stone materials.
As in the case of the first stage of the experiment, the thermal power of the materials tested was calculated and the results are shown
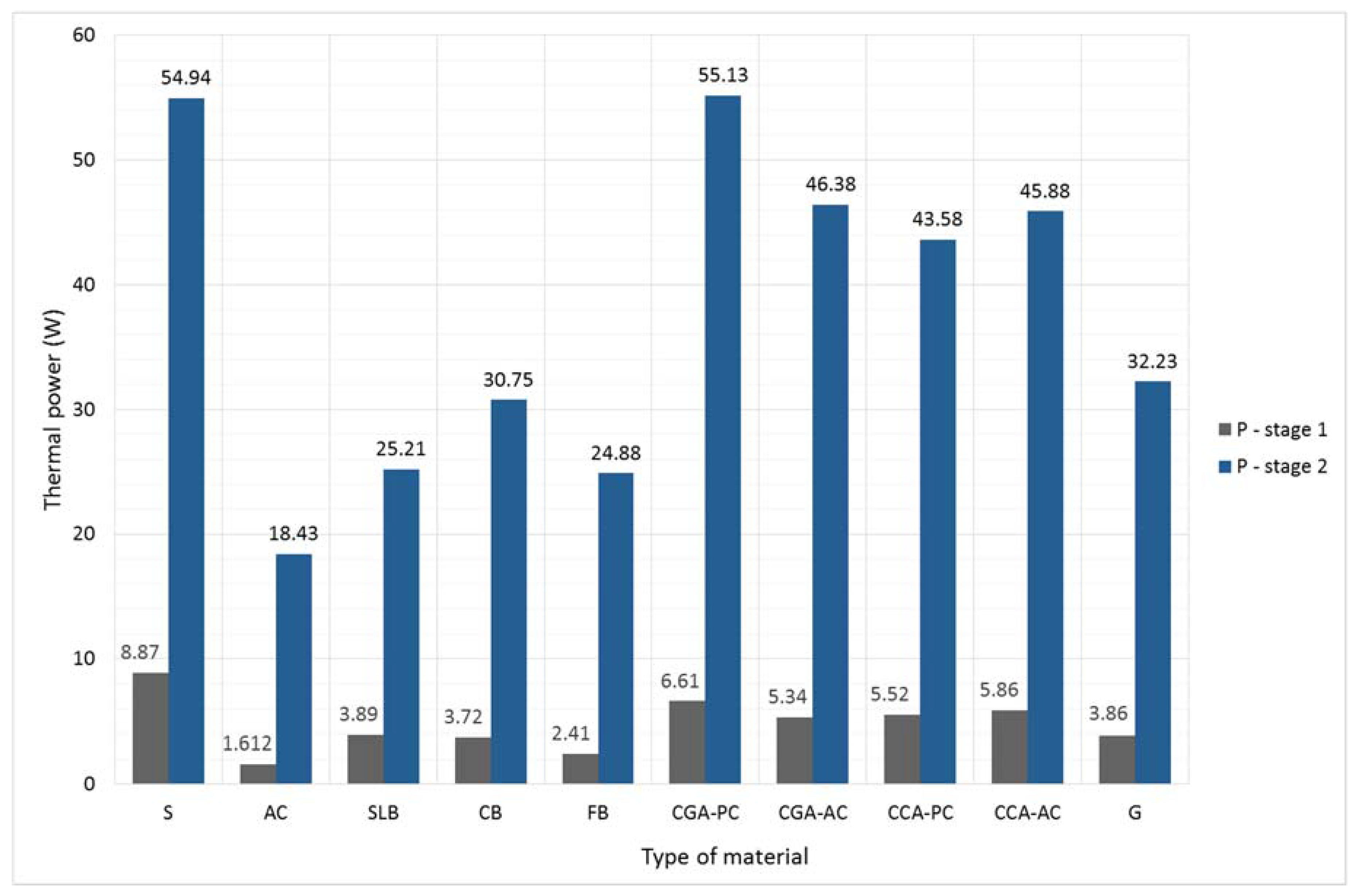
Table 8. The comparison of the thermal power between samples subjected to the low temperature load (stage 1) and the fireplace operational temperature are shown in
Figure 6.
The thermal power of the materials heated to 400 °C ranges from 6.2 (for steel) to 11.4 (for aerated concrete) times greater than the materials’ thermal power obtained during the first stage of the experiment. Previously, only the steel obtained higher thermal power than the concrete samples. In the second stage the highest thermal power was achieved by the CGA-PC concrete. All tested concretes had a thermal power greater by an average of 82% than other materials (excluding steel). In addition, the largest increases in the thermal power (between the first and the second stage of the experiment) were achieved in the concretes and the steel sample. This shows that the TES properties of these materials are better when they operate at elevated temperature conditions.
Testing at the operating temperature of the fireplace shows that the concretes (including the fireproof concrete with ceramic waste aggregate) have very good TES properties, and in combination with high durability can successfully be used as a heat accumulator in the construction industry.
After conducting the research, specimens were left in a laboratory for a period of two weeks, after which their inspection was made. The samples made of concrete with gravel aggregate (CGA-PC, CGA-AC) showed damage in the form of cracks (
Figure 7). A similar type of damage was observed for the concrete made of ceramic aggregate and Portland cement (CCA-PC). Damage occurred as a result of too high a temperature during the heating process. Other materials remained intact, including the CCA-AC concrete.
Damage after heating the concretes based on Portland cement (CGA-PC, CCA-PC) made it impossible to determine their basic mechanical properties—the compressive and tensile strength. The edges of the samples flaked off after heating, and the loss of their mass was noted. The concretes based on alumina cement (CGA-AC, CCA-AC) have a compact structure after heating, and it was possible to study their strength characteristics. For CGA-AC, the compressive strength was 28.4 MPa, and the tensile strength was 2.6 MPa. The strength characteristics for CCA-AC were 49.8 MPa and 4.2 MPa, respectively. The conclusion from this observation drew attention to the fact that, among the tested concretes, only the recycled one, for which the ceramic waste aggregate and alumina cement was used (CCA-AC), would be able to work without destruction in the thermal conditions that are present in a fireplace.