An Overview of Smart Shoes in the Internet of Health Things: Gait and Mobility Assessment in Health Promotion and Disease Monitoring
Abstract
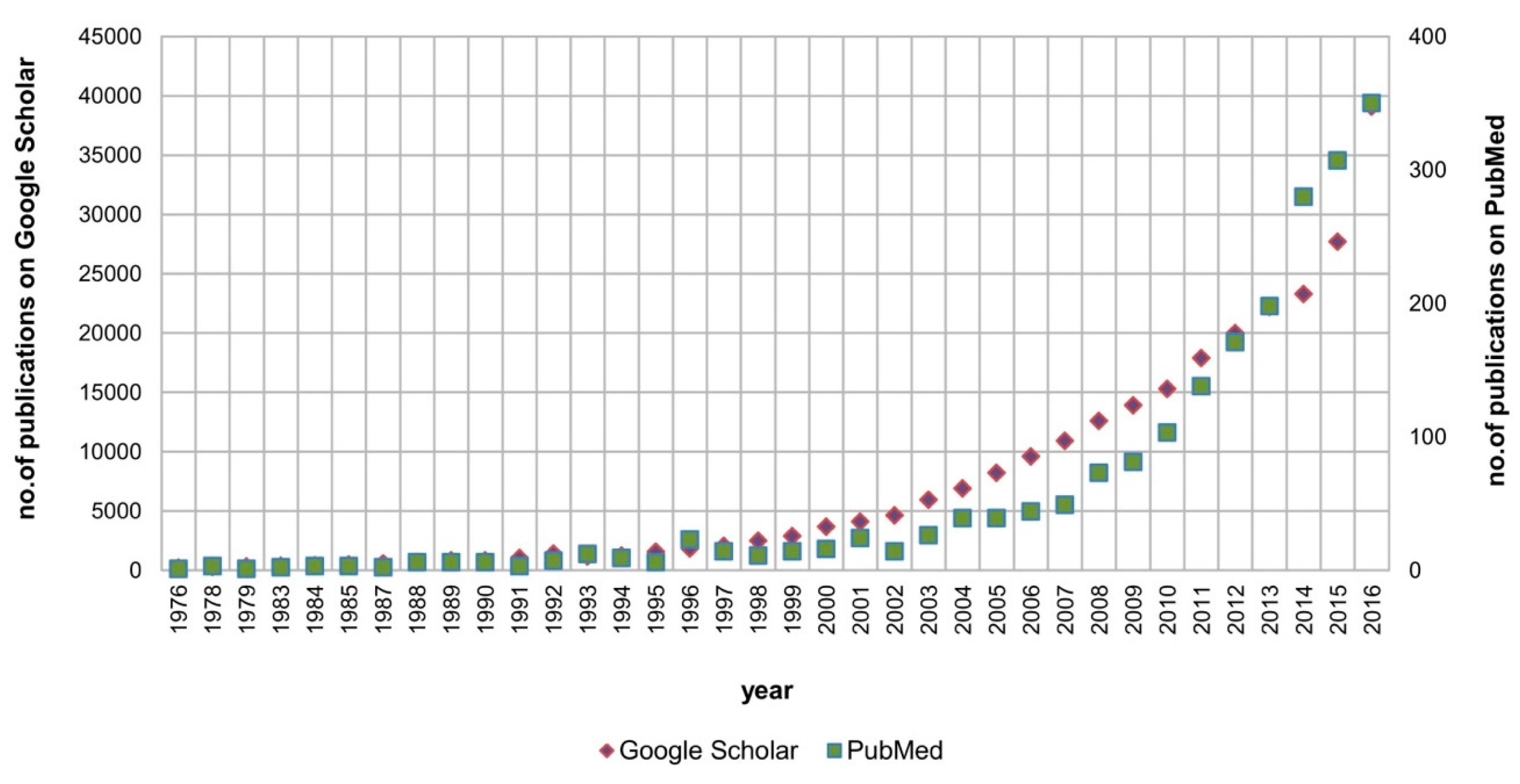
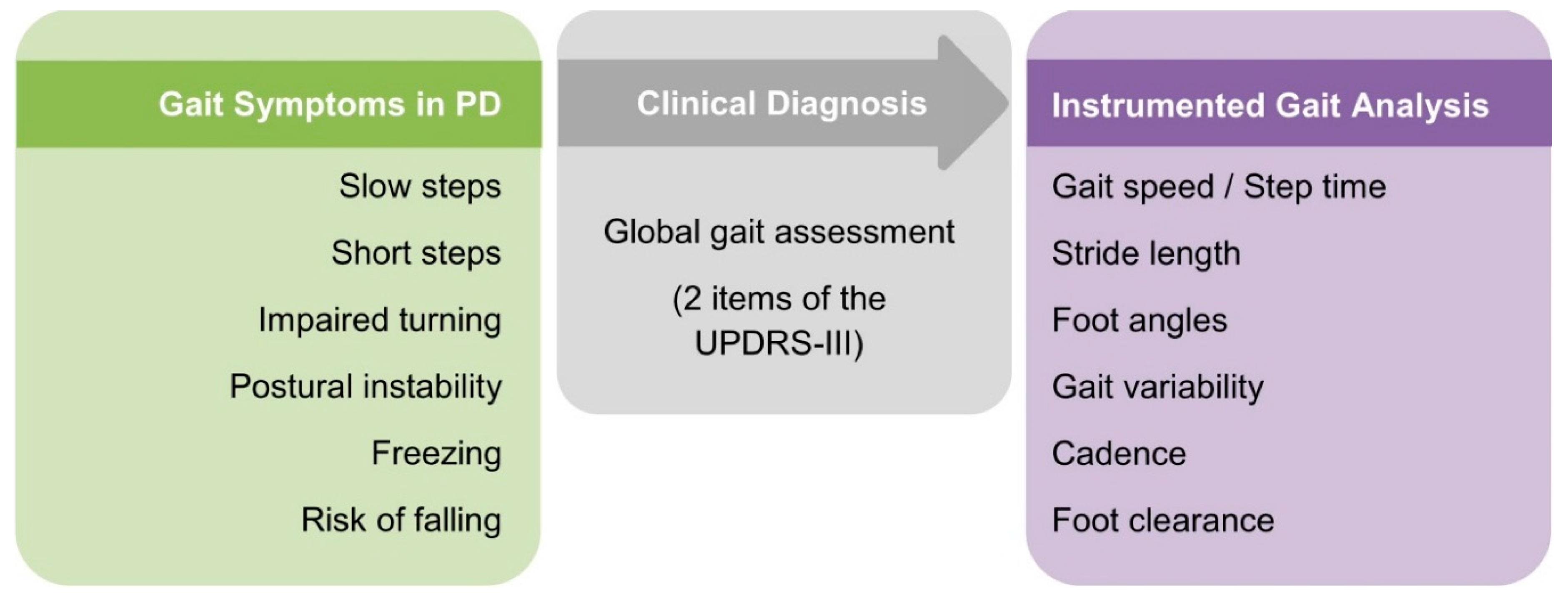
1. Introduction
2. Technological Aspects of Smart Shoes for Digital Health
2.1. Acquisition Systems
2.1.1. Energy Management Aspects
2.1.2. Data Generation
- Relative location and orientation determination using inertial-magnetic measurement units (IMMUs, consisting of an accelerometer, a gyroscope, and a magnetometer) and data fusion algorithms [18]. This data can be used, for example, for gait analysis that is explained later.
- Absolute location determination using satellite navigation systems (GPS, GLONASS, GALILEO) [19], which can also provide time information [20]. Relative and absolute location and orientation determination can be fused using loosely or tightly coupled data fusion algorithms [21]. This data can be used, for example, for wide-range activity tracking in a daily living outdoor context.
- Foot plantar pressure determination using various forms of pressure sensors, which provide information regarding how effectively and efficiently individuals control the distribution of the body weight during gait [22]. This data can be used, for example, for rehabilitation purposes, when a patient should not put too much weight onto a leg after surgery.
- Ambient environmental sensors, such as atmospheric pressure sensors for altitude-dependent activities (e.g., stair climbing or hiking) and local weather information (changes over minutes and hours), and light and sound sensors for context-related information generation.
- Internal status sensors, for example for battery and memory capacity (not discussed further).
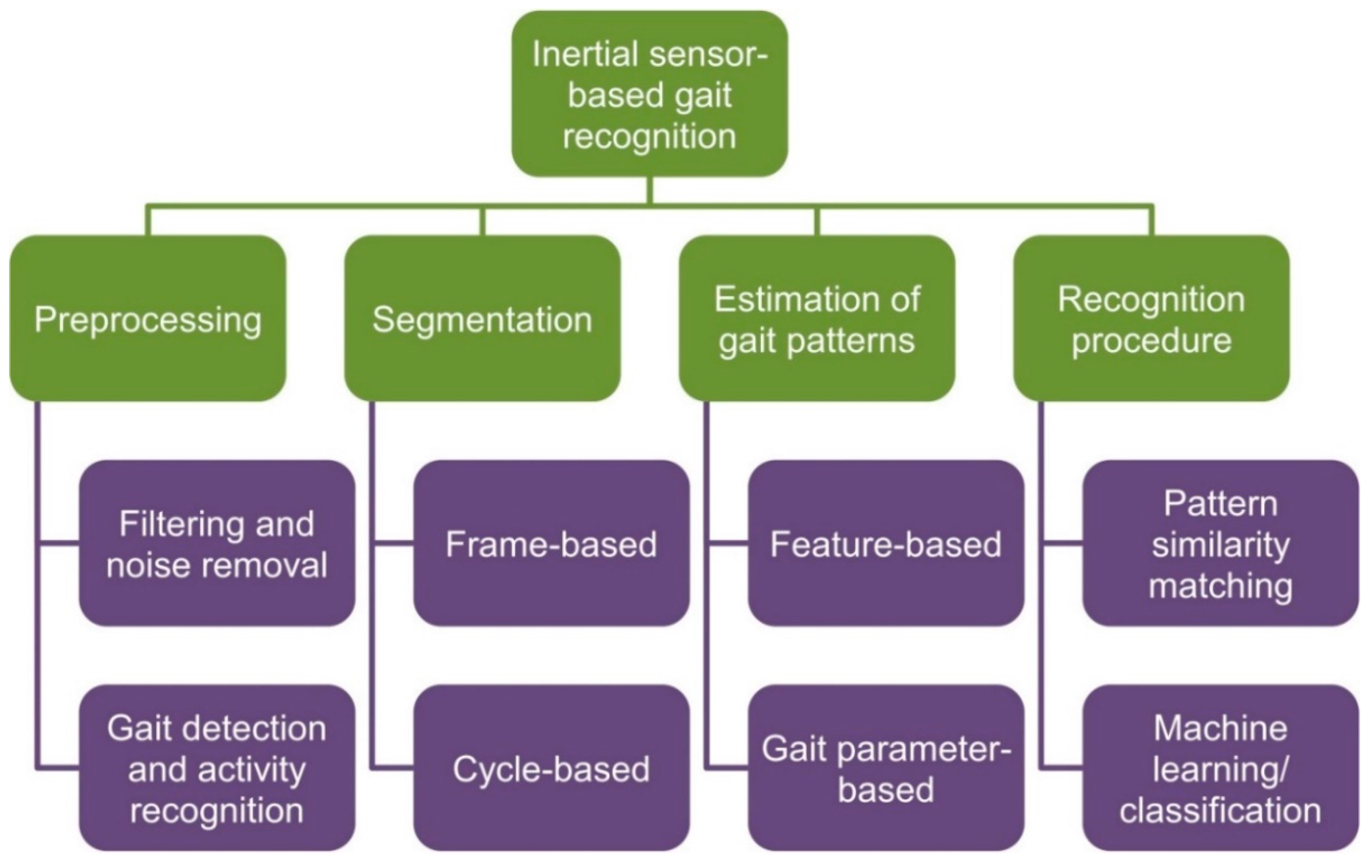
2.2. Analysis Methods
2.2.1. Preprocessing
2.2.2. Segmentation
2.2.3. Estimation of Gait Patterns
2.2.4. Recognition of Important Gait Events
2.3. Application Examples
2.3.1. Sports and Healthy Living Applications
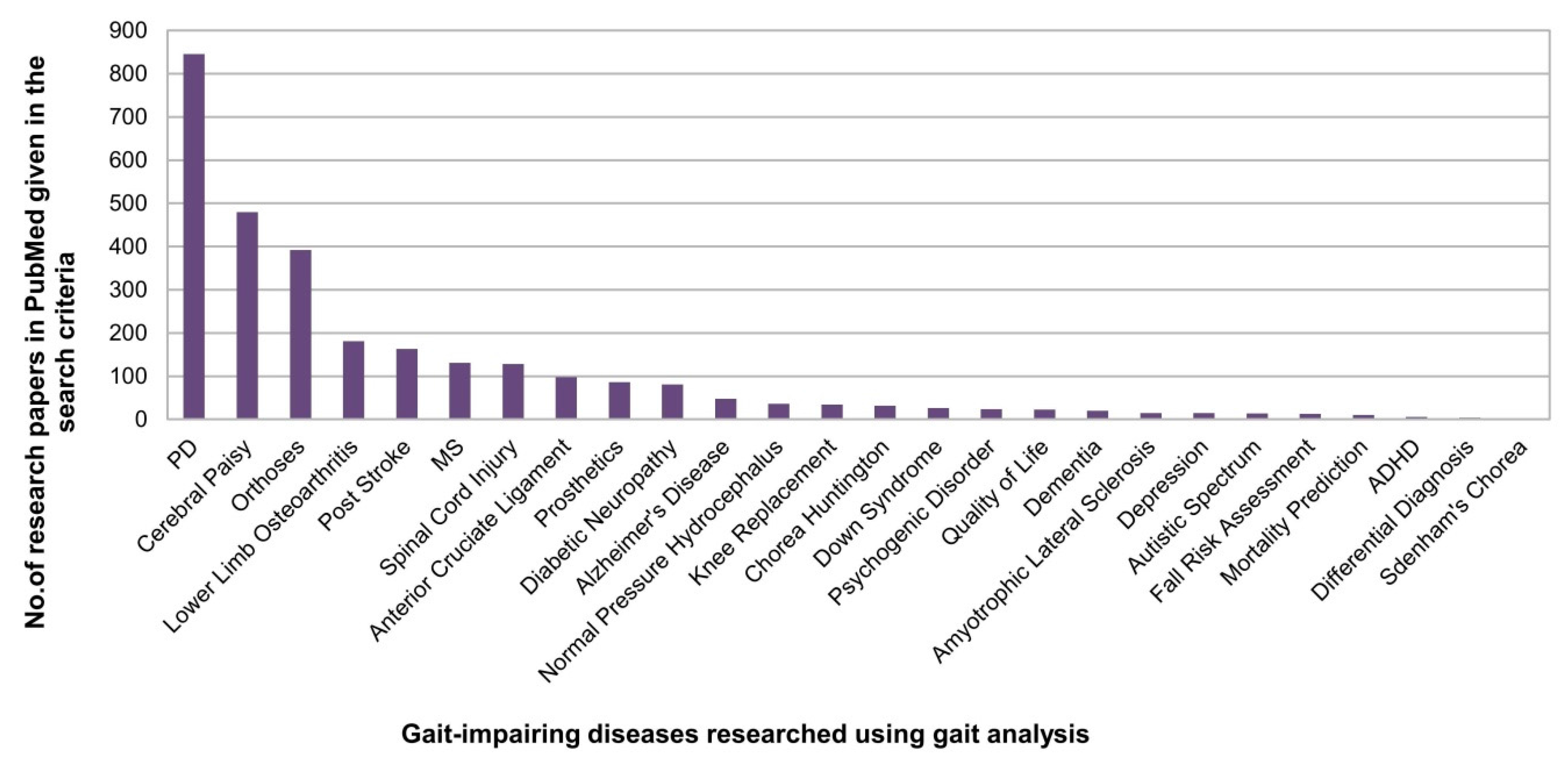
2.3.2. Medical Applications
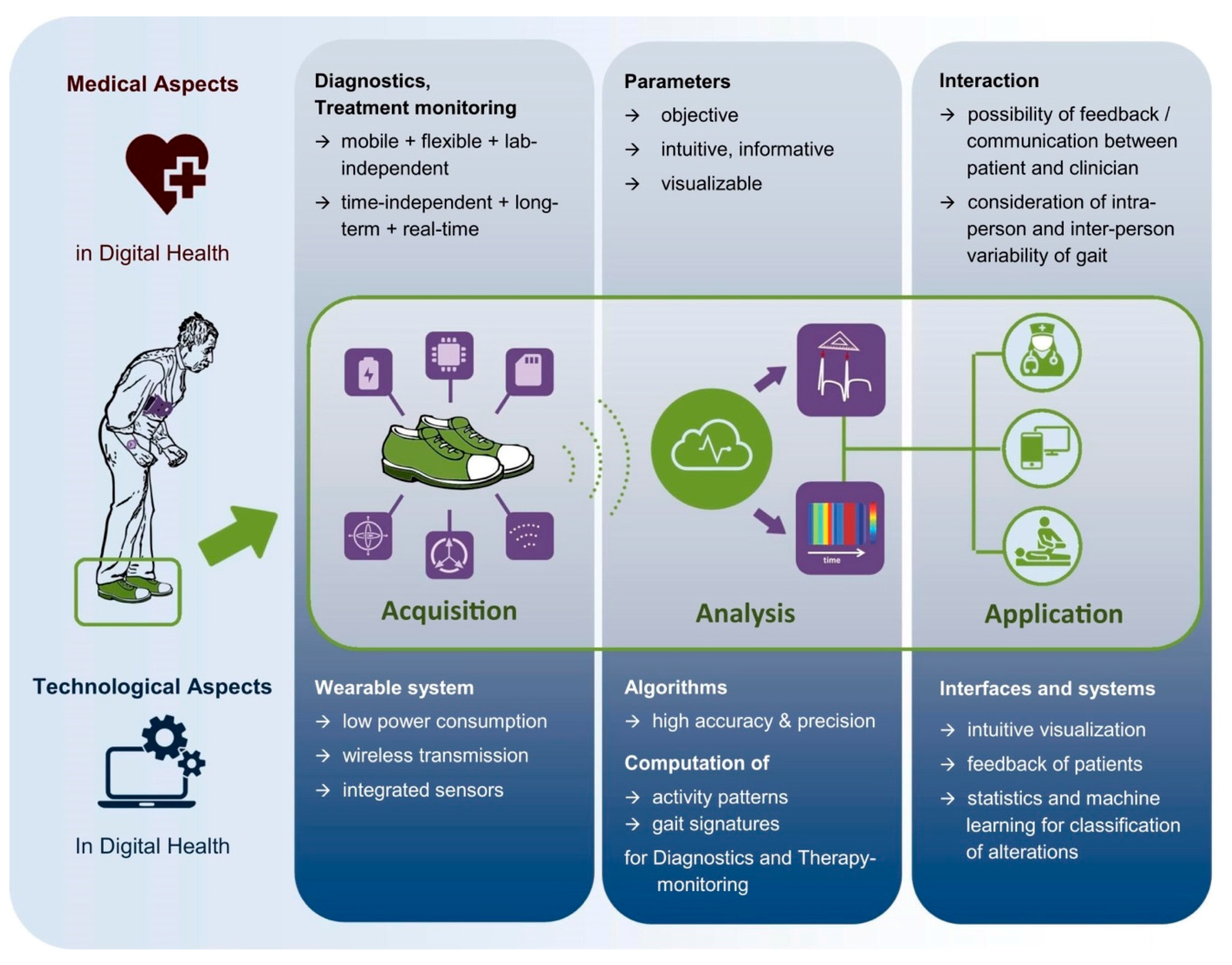
3. Medical Aspects of Smart Shoes for Digital Health
4. Discussion and Conclusions
- (1)
- address disease-specific gait characteristics by objective and quantifiable gait parameters
- (2)
- assess the validity and reproducibility of gait-related measures
- (3)
- provide secure, safe, and reliable telemedical communication platforms linking wearable sensor-based diagnostics, patient, therapists, and care givers
- (4)
- integrate the medical and technological requirements during the development phase of wearable sensors
- (5)
- target user experience, both from medical- and consumer-use perspectives; this should also be a target of academic investigations using statistics and discussion on user compliance for different types of smart shoes (materials, features, usability)
- (6)
- include regulatory and economic requirements in the development process
- (7)
- motivate researchers to comprehensively address both technological solutions and medical requirements, and demonstrate their relevance in operational clinical environments.
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Pasluosta, C.F.; Gaßner, H.; Winkler, J.; Klucken, J.; Eskofier, B.M. An emerging era in the management of parkinson’s disease: Wearable technologies and the internet of things. Biomed. Health Inform. 2015, 19, 9. [Google Scholar]
- Lord, S.; Galna, B.; Rochester, L. Moving forward on gait measurement: Toward a more refined approach. Mov. Disord. 2013, 28, 1534–1543. [Google Scholar] [CrossRef] [PubMed]
- Freeiconspng. Available online: http://freeiconspng.com/ (accessed on 24 March 2017).
- Flaticon. Available online: https://www.flaticon.com/ (accessed on 24 March 2017).
- The Noun Project. Available online: https://thenounproject.com/ (accessed on 24 March 2017).
- Parkinson, J. An Essay on the Shaking Palsy; Sherwood, Neely, Jones: London, UK, 1817. [Google Scholar]
- Chen, M.; Gonzalez, S.; Vasilakos, A.; Cao, H.; Leung, V.C.M. Body area networks: A survey. Mob. Netw. Appl. 2011, 16, 171–193. [Google Scholar] [CrossRef]
- Fortino, G.; Giannantonio, R.; Gravina, R.; Kuryloski, P.; Jafari, R. Enabling effective programming and flexible management of efficient body sensor network applications. IEEE Trans. Hum.-Mach. Syst. 2013, 43, 115–133. [Google Scholar] [CrossRef]
- Raveendranathan, N.; Galzarano, S.; Loseu, V.; Gravina, R.; Giannantonio, R.; Sgroi, M.; Jafari, R.; Fortino, G. From modeling to implementation of virtual sensors in body sensor networks. IEEE Sens. J. 2012, 12, 583–593. [Google Scholar] [CrossRef]
- Meckler, S.; Goridko, V.; Ylli, K.; Hoffmann, D.; Willmann, A.; Hehn, T.; Schwaab, M.; Ehrbrecht, B.; Manoli, Y. Pedestrian localization based on a shoe-integrated energy autarkic mobile cyber-physical system. In Proceedings of the VDE-Kongress, Mannheim, Germany, 7–8 November 2016; pp. 1–6. [Google Scholar]
- Jensen, U.; Kugler, P.; Ring, M.; Eskofier, B.M. Approaching the accuracy–cost conflict in embedded classification system design. Pattern Anal. Appl. 2016, 3, 839–855. [Google Scholar] [CrossRef]
- Ochoa, C.E.; Aries, M.B.C.; van Loenen, E.J.; Hensen, J.L.M. Considerations on design optimization criteria for windows providing low energy consumption and high visual comfort. Appl. Energy 2012, 95, 238–245. [Google Scholar] [CrossRef]
- Park, W.K.; Choi, C.S.; Lee, I.W.; Jang, J. Energy efficient multi-function home gateway in always-on home environment. IEEE Trans. Consum. Electron. 2010, 56, 106–111. [Google Scholar] [CrossRef]
- Pantelopoulos, A.; Bourbakis, N.G. Prognosis—Awearable health-monitoring system for people at risk: Methodology and modeling. IEEE Trans. Inform. Technol. Biomed. 2010, 14, 613–621. [Google Scholar] [CrossRef] [PubMed]
- Georgiou, O.; Mimis, K.; Halls, D.; Thompson, W.; Gibbins, D. How many wi-fi aps does it take to light a lightbulb? IEEE Access 2016, 4, 3732–3746. [Google Scholar] [CrossRef]
- Tan, Y.K.; Panda, S.K. Energy harvesting from hybrid indoor ambient light and thermal energy sources for enhanced performance ofwireless sensor nodes. IEEE Trans. Ind. Electron. 2011, 58, 4424–4435. [Google Scholar] [CrossRef]
- Elouze, N.; Rekhis, S.; Boudriga, N. Forensic investigation of digital crimes in healthcare applications. In Data Mining Trends and Applications in Criminal Science and Investigations; IGI Global: Hershey, PA, USA, 2016. [Google Scholar]
- Bergamini, E.; Ligorio, G.; Summa, A.; Vannozzi, G.; Cappozzo, A.; Sabatini, A.M. Estimating orientation using magnetic and inertial sensors and different sensor fusion approaches: Accuracy assessment in manual and locomotion tasks. Sensors 2014, 14, 18625–18649. [Google Scholar] [CrossRef] [PubMed]
- Madgwick, S.O.H.; Harrison, A.J.L.; Vaidyanathan, R. Estimation of imu and marg orientation using a gradient descent algorithm. In Proceedings of the IEEE International Conference on Rehabilitation Robotics, Zurich, Switzerland, 29 June–1 July 2011; pp. 1–7. [Google Scholar]
- Mahony, R.; Hamel, T.; Pflimlin, J.M. Nonlinear complementary filters on the special orthogonal group. IEEE Trans. Autom. Control 2008, 53, 1203–1217. [Google Scholar] [CrossRef]
- Daniec, K.; Jędrasiak, K.; Koteras, R.; Nawrat, A. Embedded micro inertial navigation system. Appl. Mech. Mater. 2013, 249–250, 1234–1246. [Google Scholar] [CrossRef]
- Noshadi, H.; Dabiri, F.; Ahmadian, S.; Amini, N.; Sarrafzadeh, M. Hermes: Mobile system for instability analysis and balance assessment. ACM Trans. Embed. Comput. Syst. 2013, 12, 1–24. [Google Scholar]
- Mariani, B.; Hoskovec, C.; Rochat, S.; Buela, C.; Penders, J.; Aminian, K. 3d gait assessment in young and elderly subjects using foot-worn inertial sensors. J. Biomech. 2010, 43, 2999–3006. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Yang, Z.; Dong, T. A review of wearable technologies for elderly care that can accurately track indoor position, recognize physical activities and monitor vital signs in real time. Sensors 2017, 17, 341. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Lach, J.; Lo, B.; Yang, G.Z. Toward pervasive gait analysis with wearable sensors: A systematic review. Biomed. Health Inform. 2016, 20, 1521–1537. [Google Scholar] [CrossRef] [PubMed]
- Paraschiv-Ionescu, A.; Perruchoud, C.; Buchser, E.; Aminian, K. Barcoding human physical activity to assess chronic pain conditions. PLoS ONE 2012, 7, 12. [Google Scholar] [CrossRef] [PubMed]
- Rampp, A.; Barth, J.; Schuelein, S.; Gaßmann, K.G.; Klucken, J.; Eskofier, B. Inertial sensor-based stride parameter calculation from gait sequences in geriatric patients. IEEE Trans. Biomed. Eng. 2015, 62, 1089–1097. [Google Scholar] [CrossRef] [PubMed]
- Klucken, J.; Barth, J.; Kugler, P.; Schlachetzki, J.; Henze, T.; Marxreiter, F.; Kohl, Z.; Steidl, R.; Hornegger, J.; Eskofier, B.; et al. Unbiased and mobile gait analysis detects motor impairment in parkinson’s disease. PLoS ONE 2013, 8, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Schulein, S.; Barth, J.; Rampp, A.; Rupprecht, R.; Eskofier, B.M.; Winkler, J.; Gassmann, K.G.; Klucken, J. Instrumented gait analysis: A measure of gait improvement by a wheeled walker in hospitalized geriatric patients. J. Neuroeng. Rehabil. 2017, 14, 18. [Google Scholar] [CrossRef] [PubMed]
- Sprager, S.; Juric, M.B. Inertial sensor-based gait recognition: A review. Sensors 2015, 15, 39. [Google Scholar] [CrossRef] [PubMed]
- Brønd, J.C.; Arvidsson, D. Sampling frequency affects the processing of actigraph raw acceleration data to activity counts. J. Appl. Physiol. 2016, 120, 362–369. [Google Scholar] [CrossRef] [PubMed]
- Bastian, T.; Maire, A.; Dugas, J.; Ataya, A.; Villars, C.; Gris, F.; Perrin, E.; Caritu, Y.; Doron, M.; Blanc, S.; et al. Automatic identification of physical activity types and sedentary behaviors from 3-axial accelerometer: Lab-based calibrations are not enough. J. Appl. Physiol. 2015, 118, 716–722. [Google Scholar] [CrossRef] [PubMed]
- Ferraris, F.; Grimaldi, U.; Parvis, M. Procedure for effortless in-field calibration of three-axis rate gyros and accelerometers. Sens. Mater. 1995, 7, 311–330. [Google Scholar]
- Tong, K.; Granat, M.H. A practical gait analysis system using gyroscopes. Med. Eng. Phys. 1999, 21, 87–94. [Google Scholar] [CrossRef]
- Sabatini, A.M.; Ligorio, G.; Mannini, A. Fourier-based integration of quasi-periodic gait accelerations for drift-free displacement estimation using inertial sensors. BioMed. Eng. OnLine 2015, 14, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Sabatini, A.M.; Martelloni, C.; Scapellato, S.; Cavallo, F. Assessment of walking features from foot inertial sensing. IEEE Trans. Biomed. Eng. 2005, 52, 9. [Google Scholar] [CrossRef] [PubMed]
- Mannini, A.; Sabatini, A.M. Gait phase detection and discrimination between walking–jogging activities using hidden markov models applied to foot motion data from a gyroscope. Gait Posture 2012, 36, 657–661. [Google Scholar] [CrossRef] [PubMed]
- Jarchi, D.; Wong, C.; Kwasnicki, R.M.; Heller, B.; Tew, G.A.; Yang, G.Z. Gait parameter estimation from a miniaturized ear-worn sensor using singular spectrum analysis and longest common subsequence. IEEE Trans. Biomed. Eng. 2014, 61, 1261–1273. [Google Scholar] [CrossRef] [PubMed]
- Barth, J.; Oberndorfer, C.; Pasluosta, C.; Schuelein, S.; Gaßner, H.; Reinfelder, S.; Kugler, P.; Schuldhaus, D.; Winkler, J.; Klucken, J.; et al. Stride segmentation during free walk movements using multi-dimensional subsequence dynamic time warping on inertial sensor data. Sensors 2015, 15, 22. [Google Scholar] [CrossRef] [PubMed]
- Ten Holt, G.A.; Reinders, M.J.T.; Hendriks, E.A. Multi-dimensional dynamic time warping for gesture recognition. In Proceedings of the Thirteenth Annual Conference of the Advanced School for Computing and Imaging, Heijen, The Netherlands, 13–15 June 2007; pp. 1–8. [Google Scholar]
- González, I.; Fontecha, J.; Hervás, R.; Bravo, J. An ambulatory system for gait monitoring based on wireless sensorized insoles. Sensors 2015, 15, 16589–16613. [Google Scholar] [CrossRef] [PubMed]
- Sejdic, E.; Lowry, K.A.; Bellanca, J. A comprehensive assessment of gait accelerometry signals in time, frequency and time-frequency domains. J. Neural Syst. 2014, 22, 603–612. [Google Scholar] [CrossRef] [PubMed]
- Trojaniello, D.; Cereatti, A.; Della Croce, U. Accuracy, sensitivity and robustness of five different methods for the estimation of gait temporal parameters using a single inertial sensor mounted on the lower trunk. Gait Posture 2014, 40, 487–492. [Google Scholar] [CrossRef] [PubMed]
- Arminian, K. Temporal feature estimation during walking using miniature accelerometers: An analysis of gait improvement after hip arthroplasty. Med. Biol. Eng. Comput. 1999, 37, 686–691. [Google Scholar] [CrossRef]
- Shin, S.H.; Park, C.G. Adaptive step length estimation algorithm using optimal parameters and movement status awareness. Med. Eng. Phys. 2011, 33, 1064–1071. [Google Scholar] [CrossRef] [PubMed]
- Tadano, S.; Takeda, R.; Tohyama, H. Gait characterization for osteoarthritis patients using wearable gait sensors (h-gait systems). J. Biomech. 2016, 49, 684–690. [Google Scholar] [CrossRef] [PubMed]
- Sant’Anna, A.; Wickstroem, N.; Eklund, H.; Zuegner, R. Assessment of gait symmetry and gait normality using intertial sensors: In-lab and in-situ evaluation. In Communications in Computer and Information Science; Springer: Berlin/Heidelberg, Germany, 2012; pp. 239–254. [Google Scholar]
- Mazilu, S.; Blanke, U.; Hardegger, M.; Troester, G.; Gazit, E.; Dorfman, M.; Hausdorff, J.M. Gaitassist: A wearable assistant for gait training and rehabilitation in parkinson’s disease. In Proceedings of the IEEE International Conference on Pervasive Computing and Communications Demonstrations, Budapest, Hungary, 24–28 March 2014; pp. 135–137. [Google Scholar]
- McCamley, J.; Donati, M.; Grimpampi, E.; Mazzà, C. An enhanced estimate of reinitial contact and final contact instants of time using lower trunk inertial sensor data. Gait Posture 2012, 36, 316–318. [Google Scholar] [CrossRef] [PubMed]
- Federolf, P.A.; Boyer, K.A.; Andriacchi, T.P. Application of principal component analysis in clinical gait research: Identification of systematic differences between healthy and medical knee-osteoarthritic gait. J. Biomech. 2013, 46, 2173–2178. [Google Scholar] [CrossRef] [PubMed]
- Sprager, S. Gait identification using cumulants of accelerometer data. In Proceedings of the Sensors, Signals, Visualization, Imaging, Simulation and Materials, Baltimore, MD, USA, 7–9 November 2009; pp. 94–99. [Google Scholar]
- Frank, J.; Mannor, S.; Pineau, J. Time series anaylsis using geometric template matching. Trans. pattern Anal. Mach. Intell. 2013, 35, 740–754. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Yuao, T. Curve aligning approach for gait authentication based on a wearabe accelerometer. Physiol. Meas. 2012, 33, 1111–1120. [Google Scholar] [CrossRef] [PubMed]
- Lord, S.; Galna, B.; Verghese, J.; Coleman, S.; Burn, D.; Rochester, L. Independent domains of gait in older adults and associated motor and nonmotor attributes: Validation of a factor analysis approach. J. Gerontol. A Biol. Sci. Med. Sci. 2013, 68, 820–827. [Google Scholar] [CrossRef] [PubMed]
- Hannink, J.; Kautz, T.; Pasluosta, C.; Gassmann, K.G.; Klucken, J.; Eskofier, B.M. Sensor-based gait parameter extraction with deep convolutional neural networks. IEEE J. Biomed. Health Inform. 2016, 21, 85–93. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Li, Q. Inertial sensor-based methods in walking speed estimation: A systematic review. Sensors 2012, 12, 6102–6116. [Google Scholar] [CrossRef] [PubMed]
- Ngo, T.T.; Makihara, Y.; Nagahara, H.; Mukaigawa, Y.; Yagi, Y. The largest inertial sensor-based gait database and performance evaluation of gait-based personal authentication. Pattern Recognit. 2014, 47, 228–237. [Google Scholar] [CrossRef]
- Mannini, A.; Trojaniello, D.; Cereatti, A.; Sabatini, A.M. A machine learning framework for gait classification using inertial sensors: Application to elderly, post-stroke and huntington’s disease patients. Sensors 2016, 16, 134. [Google Scholar] [CrossRef] [PubMed]
- Nukala, B.T.; Shibuya, N.; Rodriguez, A.; Tsay, J. An efficient and robust fall detection system using wireless gait analysis sensor with artificial neural network (ann) and support vector machine (svm). Open J. Appl. Biosens. 2013, 3, 29–39. [Google Scholar] [CrossRef]
- Nickel, C.; Busch, C.; Rangarajan, S. Using hidden markov models for accelerometer-based biometric gait recognition. In Proceedings of the IEEE 7th International Colloquium on Signal Processing and Its Applications, Penang, Malaysia, 4–6 March 2011; Volume 7, pp. 58–63. [Google Scholar]
- Allen, F.R.; Ambikairajah, E.; Lovell, N.H. Classification of a known sequence of motions and postures from accelerometry data using adapted gaussian mixture models. Physiol. Meas. 2006, 27, 935–951. [Google Scholar] [CrossRef] [PubMed]
- Preece, S.J.; Goulermas, J.Y.; Kenney, L.P.J. Acticity identification using body-mounted sensors—A review of classification techniques. Physiol. Meas. 2009, 1–33. [Google Scholar] [CrossRef] [PubMed]
- Mendes, J.J.A.; Vieira, M.E.M.; Pires, M.B.; Stevan, S.L. Sensor fusion and smart sensor in sports and biomedical applications. Sensors 2016, 16, 1569. [Google Scholar] [CrossRef] [PubMed]
- Bonacci, J.; Vicenzino, B.; Spratford, W.; Collins, P. Take your shoes off to reduce patellofemoral joint stress during running. Br. J. Sports Med. 2014, 48, 425–428. [Google Scholar] [CrossRef] [PubMed]
- Snijders, A.H.; van de Warrenburg, B.P.; Giladi, N.; Bloem, B.R. Neurological gait disorders in elderly people: Clinical approach and classification. Lancet Neurol. 2007, 6, 63–74. [Google Scholar] [CrossRef]
- Soh, S.E.; Morris, M.E.; McGinley, J.L. Determinants of health-related quality of life in parkinson’s disease: A systematic review. Parkinsonism Relat. Disord. 2011, 17, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Sousa, A. Influence of long-term wearing of unstable shoes on compensatory control of posture: An electromyography-based analysis. Gait Posture 2014, 39, 98–104. [Google Scholar] [CrossRef] [PubMed]
- Stolze, H.; Klebe, S.; Baecker, C.; Zechlin, C.; Friege, L.; Pohle, S.; Deuschl, G. Prevalence of gait disorders in hospitalized neurological patients. Mov. Disord. 2005, 20, 89–94. [Google Scholar] [CrossRef] [PubMed]
- Cheung, R.T.H.; Rainbow, M.J. Landing pattern and vertical loading rates during first attempt of barefoot running in habitual shod runners. Hum. Mov. Sci. 2014, 34, 120–127. [Google Scholar] [CrossRef] [PubMed]
- Cronin, N.J. The effects of high heeled shoes on female gait: A review. J. Electromyogr. Kinesiol. 2014, 24, 258–263. [Google Scholar] [CrossRef] [PubMed]
- Forghany, S.; Nester, C.J.; Richards, B.; Hatton, A.L.; Liu, A. Rollover footwear affects lower limb biomechanics during walking. Gait Posture 2014, 39, 205–212. [Google Scholar] [CrossRef] [PubMed]
- Godinho, C.; Domingos, J.; Cunha, G.; Santos, A.T.; Fernandes, R.M.; Abreu, D.; Gonçalves, N.; Matthews, H.; Isaacs, T.; Duffen, J.; et al. A systematic review of the characteristics and validity of monitoring technologies to assess parkinson’s disease. J. Neuroeng. Rehabil. 2016, 13, 24. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Ferro, Á.; Elshehabi, M.; Godinho, C.; Salkovic, D.; Hobert, M.A.; Domingos, J.; van Uem, J.M.T.; Ferreira, J.J.; Maetzler, W. New methods for the assessment of parkinson's disease (2005 to 2015): A systematic review. Mov. Disord. 2016, 31, 1283–1292. [Google Scholar] [CrossRef] [PubMed]
- Maetzler, W.; Klucken, J.; Horne, M. A clinical view on the development of technology-based tools in managing parkinson’s disease. Mov. Disord. 2016, 31, 1263–1271. [Google Scholar] [CrossRef] [PubMed]
- Klucken, J.; Friedl, K.E.; Eskofier, B.M.; Hausdorff, J.M. Guest editorial: Enabling technologies for parkinson’s disease management. IEEE J. Biomed. Health Inform. 2015, 19, 1775–1776. [Google Scholar] [CrossRef] [PubMed]
- Del Din, S.; Godfrey, A.; Mazzà, C.; Lord, S.; Rochester, L. Free-living monitoring of parkinson's disease: Lessons from the field. Mov. Disord. 2016, 31, 1293–1313. [Google Scholar] [CrossRef] [PubMed]
- Caudron, S.; Guerraz, M.; Eusebio, A.; Gros, J.P.; Azulay, J.P.; Vaugoyeau, M. Evaluation of a visual biofeedback on the postural control in parkinson's disease. Clin. Neurophysiol. 2013, 44, 77–86. [Google Scholar] [CrossRef] [PubMed]
- Horak, F.; King, L.; Mancini, M. Role of body-worn movement monitor technology for balance and gait rehabilitation. Am. Phys. Ther. Assoc. 2014, 95, 461–470. [Google Scholar] [CrossRef] [PubMed]
- Schwenk, M.; Mohler, J.; Wendel, C.; D’Huyvetter, K.; Fain, M.; Taylor-Piliae, R.; Najafi, B. Wearable sensor-based in-home assessment of gait, balance, and physical activity for discrimination of frailty status: Baseline results of the arizona frailty cohort study. Gerontology 2015, 61, 10. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.I.; Park, E.; Huang, A.; Mortazavi, B.; Garst, J.H.; Jahanforouz, N.; Espinal, M.; Siero, T.; Pollack, S.; Afridi, M.; et al. Objectively quantifying walking ability in degenerative spinal disorder patients using sensor equipped smart shoes. Med. Eng. Phys. 2016, 38, 442–449. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.I.; Campion, A.; Huang, A.; Park, E.; Garst, J.H.; Jahanforouz, N.; Espinal, M.; Siero, T.; Pollack, S.; Afridi, M.; et al. Identifying predictors for postoperative clinical outcome in lumbar spinal stenosis patients using smart-shoe technology. J. Neuroeng. Rehabil. 2017, 14, 77. [Google Scholar] [CrossRef] [PubMed]
- Perrier, A.; Vuillerme, N.; Luboz, V.; Bucki, M.; Cannard, F.; Diot, B.; Colin, D.; Rin, D.; Bourg, J.P.; Payan, Y. Smart diabetic socks: Embedded device for diabetic foot prevention. Innov. Res. BioMed. Eng. 2014, 32, 5. [Google Scholar] [CrossRef]
- Ma, Y.; Amini, N.; Ghasemzadeh, H. Wearable sensors for gait pattern examination in glaucoma patients. Microprocess. Microsyst. 2016, 46, 67–74. [Google Scholar] [CrossRef]
- Bertolucci, F.; Di Martino, S.; Orsucci, D.; Ienco, E.C.; Siciliano, G.; Rossi, B.; Mancuso, M.; Chisari, C. Robotic gait training improves motor skills and quality of life in hereditary spastic paraplegia. NeuroRehabilitation 2015, 36, 93–99. [Google Scholar] [PubMed]
- Ossig, C.; Antonini, A.; Buhmann, C.; Classen, J.; Csoti, I.; Falkenburger, B.; Schwarz, M.; Winkler, J.; Storch, A. Wearable sensor-based objective assessment of motor symptoms in parkinson's disease. J. Neural Transm. (Vienna) 2016, 123, 57–64. [Google Scholar] [CrossRef] [PubMed]
- Nonnekes, J.; Goselink, R.; Weerdesteyn, V.; Bloem, B.R. The retropulsion test: A good evaluation of postural instability in parkinson's disease? J. Parkinson Dis. 2015, 5, 43–47. [Google Scholar]
- Bregou Bourgeois, A.; Mariani, B.; Aminian, K.; Zambelli, P.Y.; Newman, C.J. Spatio-temporal gait analysis in children with cerebral palsy using, foot-worn inertial sensors. Gait Posture 2014, 39, 436–442. [Google Scholar] [CrossRef] [PubMed]
- Lees, A.J.; Hardy, J.; Revesz, T. Parkinson’s disease. Lancet 2009, 373, 2055–2066. [Google Scholar] [CrossRef]
- Goetz, C.G.; Tilley, B.C.; Shaftman, S.R.; Stebbins, G.T.; Fahn, S.; Martinez-Martin, P.; Poewe, W.; Sampaio, C.; Stern, M.B.; Dodel, R.; et al. Movement disorder society-sponsored revision of the unified parkinson's disease rating scale (mds-updrs): Scale presentation and clinimetric testing results. Mov. Disord. 2008, 23, 2129–2170. [Google Scholar] [CrossRef] [PubMed]
- van Uem, J.M.; Marinus, J.; Canning, C.; van Lummel, R.; Dodel, R.; Liepelt-Scarfone, I.; Berg, D.; Morris, M.E.; Maetzler, W. Health-related quality of life in patients with parkinson's disease--a systematic review based on the icf model. Neurosci. Biobehav. Rev. 2016, 61, 26–34. [Google Scholar] [CrossRef] [PubMed]
- Maetzler, W.; Liepelt, I.; Berg, D. Progression of parkinson’s disease in the clinical phase: Potential markers. Lancet. Neurol. 2009, 8, 1158–1171. [Google Scholar] [CrossRef]
- Espay, A.J.; Bonato, P.; Nahab, F.B.; Maetzler, W.; Dean, J.M.; Klucken, J.; Eskofier, B.M.; Merola, A.; Horak, F.; Lang, A.E.; et al. Technology in parkinson’s disease: Challenges and opportunities. Mov. Disord. 2016, 31, 1272–1282. [Google Scholar] [CrossRef] [PubMed]
- Qiang, J.K.; Marras, C. Telemedicine in parkinson’s disease: A patient perspective at a tertiary care centre. Parkinsonism Relat. Disord. 2015, 21, 525–528. [Google Scholar] [CrossRef] [PubMed]
- Zampieri, C.; Salarian, A.; Carlson-Kuhta, P.; Nutt, J.G.; Horak, F.B. Assessing mobility at home in people with early parkinson's disease using an instrumented timed up and go test. Parkinsonism Relat. Disord. 2011, 17, 277–280. [Google Scholar] [CrossRef] [PubMed]
- Salarian, A.; Horak, F.B.; Zampieri, C.; Carlson-Kuhta, P.; Nutt, J.G.; Aminian, K. Itug, a sensitive and reliable measure of mobility. IEEE Trans. Neural Syst. Rehabil. Eng. 2010, 18, 303–310. [Google Scholar] [CrossRef] [PubMed]
- Forsaa, E.B.; Larsen, J.P.; Wentzel-Larsen, T.; Herlofson, K.; Alves, G. Predictors and course of health-related quality of life in parkinson's disease. Mov. Disord. 2008, 23, 1420–1427. [Google Scholar] [CrossRef] [PubMed]
- Muro-de-la-Herran, A.; Garcia-Zapirain, B.; Mendez-Zorrilla, A. Gait analysis methods: An overview of wearable and non-wearable systems, highlighting clinical applications. Sensors 2014, 14, 3362–3394. [Google Scholar] [CrossRef] [PubMed]


© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Eskofier, B.M.; Lee, S.I.; Baron, M.; Simon, A.; Martindale, C.F.; Gaßner, H.; Klucken, J. An Overview of Smart Shoes in the Internet of Health Things: Gait and Mobility Assessment in Health Promotion and Disease Monitoring. Appl. Sci. 2017, 7, 986. https://doi.org/10.3390/app7100986
Eskofier BM, Lee SI, Baron M, Simon A, Martindale CF, Gaßner H, Klucken J. An Overview of Smart Shoes in the Internet of Health Things: Gait and Mobility Assessment in Health Promotion and Disease Monitoring. Applied Sciences. 2017; 7(10):986. https://doi.org/10.3390/app7100986
Chicago/Turabian StyleEskofier, Bjoern M., Sunghoon Ivan Lee, Manuela Baron, André Simon, Christine F. Martindale, Heiko Gaßner, and Jochen Klucken. 2017. "An Overview of Smart Shoes in the Internet of Health Things: Gait and Mobility Assessment in Health Promotion and Disease Monitoring" Applied Sciences 7, no. 10: 986. https://doi.org/10.3390/app7100986
APA StyleEskofier, B. M., Lee, S. I., Baron, M., Simon, A., Martindale, C. F., Gaßner, H., & Klucken, J. (2017). An Overview of Smart Shoes in the Internet of Health Things: Gait and Mobility Assessment in Health Promotion and Disease Monitoring. Applied Sciences, 7(10), 986. https://doi.org/10.3390/app7100986







