Luminescent Properties of Silicon Nanocrystals:Spin on Glass Hybrid Materials
Abstract
:1. Introduction
2. Experimental Details
3. Results and Discussion
3.1. Structural Properties
3.1.1. Transmission Electron Microscopy
3.1.2. Grazing Incidence X-Ray Diffraction
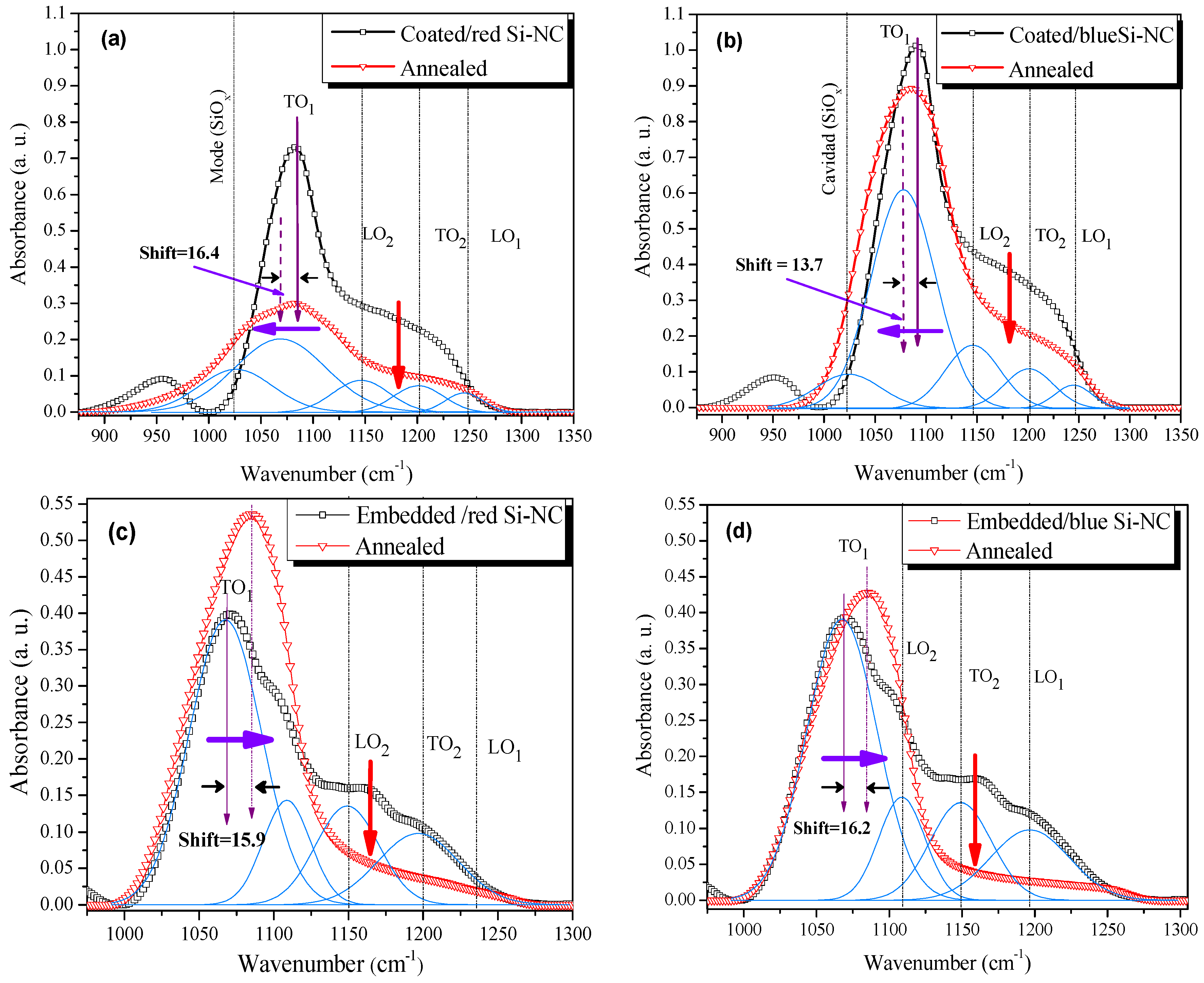
3.1.3. Fourier Transform Infrared Spectroscopy
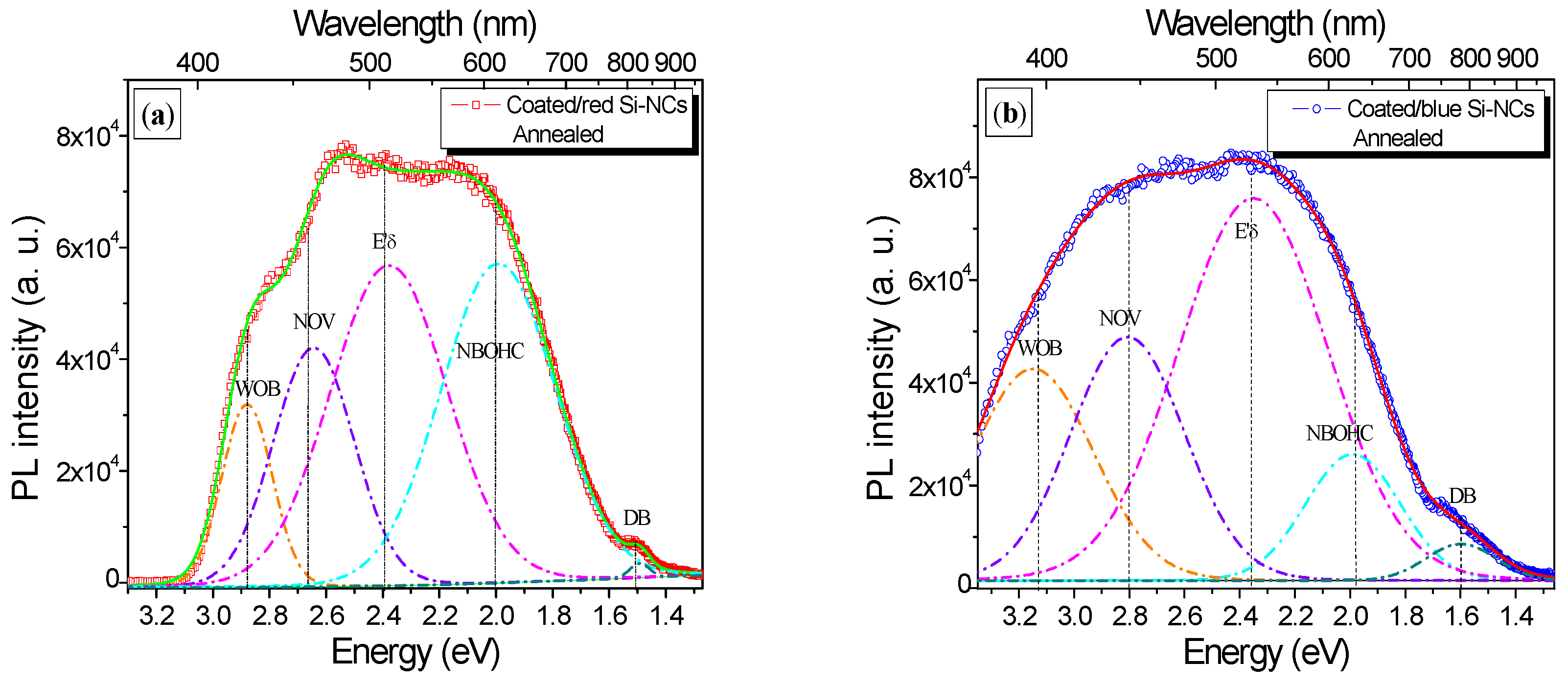
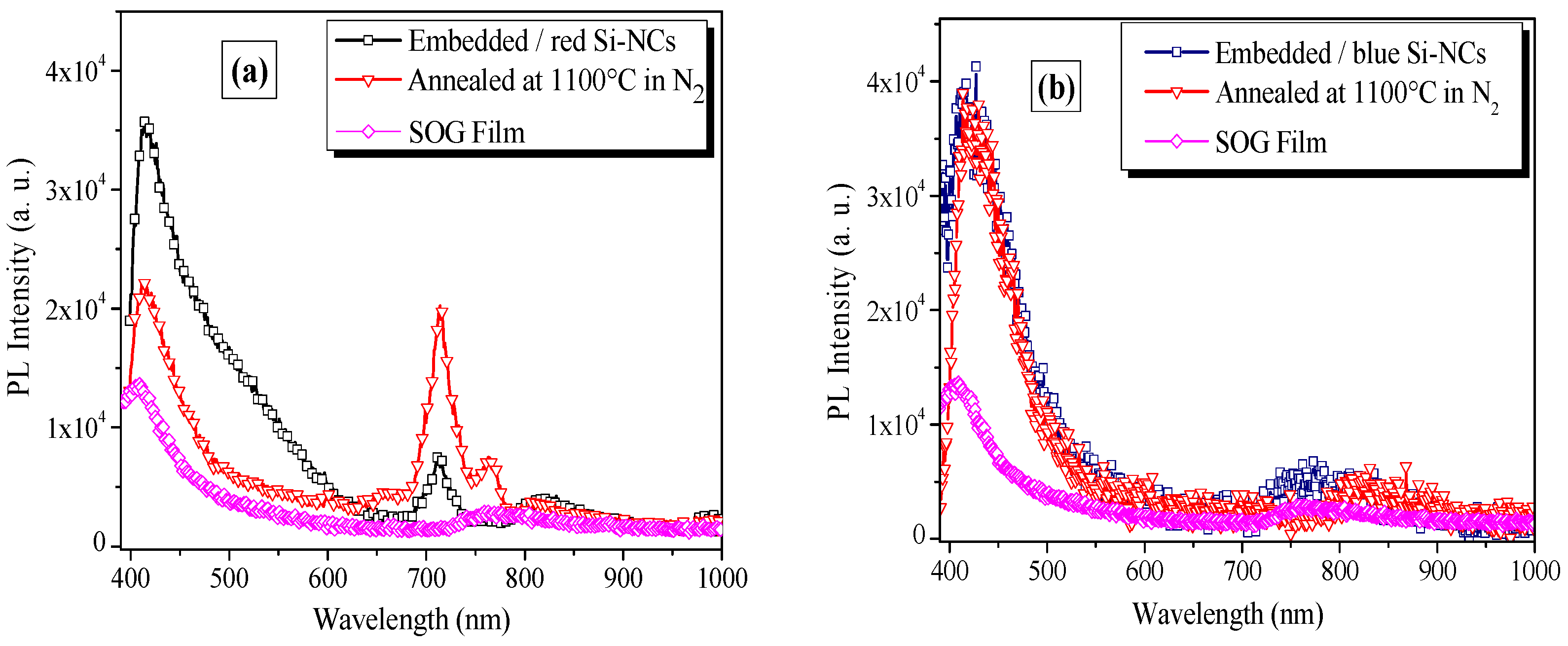
3.2. Photoluminescence Properties
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Lehmann, V.; Gösele, U. Porous silicon formation: A quantum wire effect. Appl. Phys. Lett. 1991, 58, 856–858. [Google Scholar] [CrossRef]
- Doğan, İ.; Kramer, N.J.; Westermann, R.H.; Dohnalová, K.; Smets, A.H.; Verheijen, M.A.; Gregorkiewicz, T.; van de Sanden, M.C. Ultrahigh throughput plasma processing of free standing silicon nanocrystals with lognormal size distribution. J. Appl. Phys. 2013, 113. [Google Scholar] [CrossRef]
- Bolduc, M.; Genard, G.; Yedji, M.; Barba, D.; Martin, F.; Terwagne, G.; Ross, G.G. Influence of nitrogen on the growth and luminescence of silicon nanocrystals embedded in silica. J. Appl. Phys. 2009, 105. [Google Scholar] [CrossRef]
- Quiroga-González, E.; Bensch, W.; Aceves-Mijares, M.; Yu, Z.; López-Estopier, R.; Monfil-Leyva, K. On the photoluminescence of multilayer arrays of silicon rich oxide with high silicon content prepared by low pressure chemical vapor deposition. Thin Solid Films 2011, 519, 8030–8036. [Google Scholar] [CrossRef]
- Fauchet, P.M.; Ruan, J.; Chen, H.; Pavesi, L.; Dal Negro, L.; Cazzaneli, M.; Elliman, R.G.; Smith, N.; Samoc, M.; Luther-Davies, B. Optical gain in different silicon nanocrystal systems. Opt. Mater. 2005, 27, 745–749. [Google Scholar] [CrossRef]
- Ledoux, G.; Gong, J.; Huisken, F.; Guillois, O.; Reynaud, C. Photoluminescence of size-separated silicon nanocrystals: Confirmation of quantum confinement. Appl. Phys. Lett. 2002, 80, 4834–4836. [Google Scholar] [CrossRef]
- Ray, M.; Hossain, S.M.; Klie, R.F.; Banerjee, K.; Ghosh, S. Free standing luminescent silicon quantum dots: evidence of quantum confinement and defect related transitions. Nanotechnology 2010, 21. [Google Scholar] [CrossRef] [PubMed]
- Valenta, J.; Janda, P.; Dohnalová, K.; Nižňansky, D.; Vácha, F.; Linnros, J. Colloidal suspensions of silicon nanocrystals: from single nanocrystals to photonic structures. Opt. Mater. 2005, 27, 1046–1049. [Google Scholar] [CrossRef]
- Švrček, V.; Rehspringer, J.L.; Slaoui, A.; Pivac, B.; Muller, J.C. Clustering/declustering of silicon nanocrystals in spin-on glass solutions. Semicond. Sci. Technol. 2005, 20, 314–319. [Google Scholar] [CrossRef]
- Chen, X.Y.; Lu, Y.F.; Wu, Y.H.; Cho, B.J.; Liu, M.H.; Dai, D.Y.; Song, W.D. Mechanisms of photoluminescence from silicon nanocrystals formed by pulsed-laser deposition in argon and oxygen ambient. J. Appl. Phys. 2003, 93, 6311–6319. [Google Scholar] [CrossRef]
- Ali, A.M.; Kobayashi, H.; Inokuma, T.; Al-Hajry, A. Morphological, luminescence and structural properties of nanocrystalline silicon thin films. Mater. Res. Bull. 2013, 48, 1027–1033. [Google Scholar] [CrossRef]
- Godefroo, S.; Hayne, M.; Jivanescu, M.; Stesmans, A.; Zacharias, M.; Lebedev, O.I.; Van Tendeloo, G.; Moshchalkov, V.V. Classification and control of the origin of photoluminescence from Si nanocrystals. Nat. Nanotechnol. 2008, 3, 174–178. [Google Scholar] [CrossRef] [PubMed]
- Dohnalová, K.; Poddubny, A.N.; Prokofiev, A.A.; De Boer, W.D.; Umesh, C.P.; Paulusse, J.M.; Zuilhof, H.; Gregorkiewicz, T. Surface brightens up Si quantum dots: Direct bandgap-like size-tunable emission. Light Sci. Appl. 2013, 2. [Google Scholar] [CrossRef]
- Wolkin, M.V.; Jorne, J.; Fauchet, P.M.; Allan, G.; Delerue, C. Electronic States and Luminescence in Porous Silicon Quantum Dots: The Role of Oxygen. Phys. Rev. Lett. 1999, 82, 197–200. [Google Scholar] [CrossRef]
- Dohnalová, K.; Gregorkiewicz, T.; Kůsová, K. Silicon quantum dots: Surface matters. J. Phys. Condens. Matter 2014, 26. [Google Scholar] [CrossRef] [PubMed]
- Skuja, L. Optically active oxygen-deficiency-related centers in amorphous silicon dioxide. J. Non-Cryst. Solids 1998, 239, 16–48. [Google Scholar] [CrossRef]
- Spallino, L.; Vaccaro, L.; Sciortino, L.; Agnello, S.; Buscarino, G.; Cannas, M.; Gelardi, F.M. Visible-ultraviolet vibronic emission of silica nanoparticles. Phys. Chem. Chem. Phys. 2014, 16, 22028–22034. [Google Scholar] [CrossRef] [PubMed]
- Vaccaro, L.A.; Cannas, M.A.; Boscaino, R.O. Luminescence features of nonbridging oxygen hole centres in silica probed by site-selective excitation with tunable laser. Solid State Commun. 2008, 146, 148–151. [Google Scholar] [CrossRef]
- Carrada, M.; Wellner, A.; Paillard, V.; Bonafos, C.; Coffin, H.; Claverie, A. Photoluminescence of Si nanocrystal memory devices obtained by ion beam synthesis. Appl. Phys. Lett. 2005, 87. [Google Scholar] [CrossRef]
- Limpens, R.; Lesage, A.; Fujii, M.; Gregorkiewicz, T. Size confinement of Si nanocrystals in multinanolayer structures. Sci. Rep. 2015, 5. [Google Scholar] [CrossRef] [PubMed]
- Aceves-Mijares, M.; Espinosa-Torres, N.D.; Flores-Gracia, F.; González-Fernández, A.A.; López-Estopier, R.; Román-López, S.; Pedraza, G.; Domínguez, C.; Morales, A.; Falcony, C. Composition and emission characterization and computational simulation of silicon rich oxide films obtained by LPCVD. Surf. Interface Anal. 2014, 46, 216–223. [Google Scholar] [CrossRef]
- Hessel, C.M.; Summers, M.A.; Meldrum, A.; Malac, M.; Veinot, J.G. Direct Patterning, Conformal Coating, and Erbium Doping of Luminescent nc-Si/SiO2 Thin Films from Solution Processable Hydrogen Silsesquioxane. Adv. Mater. 2007, 19, 3513–3516. [Google Scholar] [CrossRef]
- Lin, G.R.; Lin, C.J.; Lin, C.K.; Chou, L.J.; Chueh, Y.L. Oxygen defect and Si nanocrystal dependent white-light and near-infrared electroluminescence of Si-implanted and plasma-enhanced chemical-vapor deposition-grown Si-rich SiO2. J. Appl. Phys. 2005, 97. [Google Scholar] [CrossRef]
- Švrček, V.; Sasaki, T.; Shimizu, Y.; Koshizaki, N. Colloidal blue and red luminescent silicon nanocrystals and their elaboration in pure and doped spin on glasses. Phys. E Low Dimens. Syst. Nanostruct. 2007, 40, 293–296. [Google Scholar] [CrossRef]
- Švrček, V.; Sasaki, T.; Shimizu, Y.; Koshizaki, N. Silicon nanocrystals formed by pulsed laser-induced fragmentation of electrochemically etched Si micrograins. Chem. Phys. Lett. 2006, 429, 483–487. [Google Scholar] [CrossRef]
- Svrcek, V.; Sasaki, T.; Shimizu, Y.; Koshizaki, N. Blue luminescent silicon nanocrystals prepared by nanosecond laser ablation and stabilized in electronically compatible spin on glasses. J. Appl. Phys. 2008, 103. [Google Scholar] [CrossRef]
- Švrček, V.; Slaoui, A.; Rehspringer, J.L.; Muller, J.C. Photoluminescence studies from silicon nanocrystals embedded in spin on glass thin films. J. Lumin. 2003, 101, 269–274. [Google Scholar] [CrossRef]
- Nayfeh, M.H.; Rao, S.; Nayfeh, O.M.; Smith, A.; Therrien, J. UV Photodetectors with Thin-Film Si Nanoparticle Active Medium. IEEE Trans. Nanotechnol. 2005, 4, 660–668. [Google Scholar] [CrossRef]
- Cortazar, O.; Vasquez-A, M.A.; Aceves-Mijares, M. Effect of the Annealing Atmosphere and Temperature on the Photoluminescence of Si Nanocrystal Films Covered with Spin-On Glass. J. Nanoelectron. Optoelectron. 2015, 10, 93–98. [Google Scholar] [CrossRef]
- Comedi, D.; Zalloum, O.H.; Irving, E.A.; Wojcik, J.; Roschuk, T.; Flynn, M.J.; Mascher, P. X-ray-diffraction study of crystalline Si nanocluster formation in annealed silicon-rich silicon oxides. J. Appl. Phys. 2006, 99. [Google Scholar] [CrossRef]
- Vásquez-A., M.A.; Águila Rodríguez, G.; García-Salgado, G.; Romero-Paredes, G.; Peña-Sierra, R. FTIR and photoluminescence studies of porous silicon layers oxidized in controlled water vapor conditions. Rev. Mex. Fís. 2007, 53, 431–435. [Google Scholar]
- Lau, W.S. Infrared Characterization for Microelectronics; World Scientific: Singapore, 1999. [Google Scholar]
- Chou, J.S.; Lee, S.C. Effect of porosity on infrared-absorption spectra of silicon dioxide. J. Appl. Phys. 1995, 77, 1805–1807. [Google Scholar] [CrossRef]
- Hayashi, S.; Tanimoto, S.; Yamamoto, K. Analysis of surface oxides of gas-evaporated Si small particles with infrared spectroscopy, high-resolution electron microscopy, and X-ray photoemission spectroscopy. J. Appl. Phys. 1990, 68, 5300–5308. [Google Scholar] [CrossRef]
- Daldosso, N.; Das, G.; Larcheri, S.; Mariotto, G.; Dalba, G.; Pavesi, L.; Irrera, A.; Priolo, F.; Iacona, F.; Rocca, F. Silicon nanocrystal formation in annealed silicon-rich silicon oxide films prepared by plasma enhanced chemical vapor deposition. J. Appl. Phys. 2007, 101. [Google Scholar] [CrossRef]
- Borghesi, A.; Piaggi, A.; Sassella, A.; Stella, A.; Pivac, B. Infrared study of oxygen precipitate composition in silicon. Phys. Rev. B 1992, 46, 4123–4127. [Google Scholar] [CrossRef]
- Chayani, M.; Caquineau, H.; Despax, B.; Bandet, J.; Berjoan, R. Variations in the physico-chemical properties of near-stoichiometric silica deposited from SiH4–N2O and SiH4–N2O–He radiofrequency discharges. Thin Solid Films 2005, 471, 53–62. [Google Scholar] [CrossRef]
- Alayo, M.I.; Pereyra, I.; Scopel, W.L.; Fantini, M.C. On the nitrogen and oxygen incorporation in plasma-enhanced chemical vapor deposition (PECVD) SiOxNy films. Thin Solid Films 2002, 402, 154–161. [Google Scholar] [CrossRef]
- Kumar, V. Nanosilicon; Elsevier: Amsterdam, The Netherlands, 2011. [Google Scholar]
- Neamen, D.A. Semiconductor Physics and Devices; McGraw-Hill Higher Education: New York, NY, USA, 2003. [Google Scholar]
- Dinh, L.N.; Chase, L.L.; Balooch, M.; Siekhaus, W.J.; Wooten, F. Optical properties of passivated Si nanocrystals and SiOx nanostructures. Phys. Rev. B 1996, 54, 5029–5037. [Google Scholar] [CrossRef]
- Nishikawa, H.; Watanabe, E.; Ito, D.; Sakurai, Y.; Nagasawa, K.; Ohki, Y. Visible photoluminescence from Si clusters in γ-irradiated amorphous SiO2. J. Appl. Phys. 1996, 80, 3513–3517. [Google Scholar] [CrossRef]
- Vaccaro, L.; Popescu, R.; Messina, F.; Camarda, P.; Schneider, R.; Gerthsen, D.; Gelardi, F.M.; Cannas, M. Self-limiting and complete oxidation of silicon nanostructures produced by laser ablation in water. J. Appl. Phys. 2016, 120. [Google Scholar] [CrossRef]
- Nishikawa, H.; Watanabe, E.; Ito, D.; Takiyama, M.; Ieki, A.; Ohki, Y. Photoluminescence study of defects in ion-implanted thermal SiO2 films. J. Appl. Phys. 1995, 78, 842–846. [Google Scholar] [CrossRef]
- Zhao, X.; Schoenfeld, O.; Kusano, J.I.; Aoyagi, Y.; Sugano, T. Observation of Direct Transitions in Silicon Nanocrystallites. Jpn. J. Appl. Phys. 1994, 33, L899–L901. [Google Scholar] [CrossRef]
- Chou, S.T.; Tsai, J.H.; Sheu, B.C. The photoluminescence in Si+-implanted SiO2 films with rapid thermal anneal. J. Appl. Phys. 1998, 83, 5394–5398. [Google Scholar] [CrossRef]
- Nishikawa, H.; Nakamura, R.; Tohmon, R.; Ohki, Y.; Sakurai, Y.; Nagasawa, K.; Hama, Y. Generation mechanism of photoinduced paramagnetic centers from preexisting precursors in high-purity silicas. Phys. Rev. B 1990, 41, 7828–7834. [Google Scholar] [CrossRef]
- Tuğay, E.; Turan, R. Investigation of Photoluminescence Mechanisms from SiO2/Si:SiO2/SiO2 Structures in Weak Quantum Confined Regime by Deconvolution of Photoluminescence Spectra. J. Nanosci. Nanotechnol. 2016, 16, 4052–4064. [Google Scholar] [CrossRef] [PubMed]
- Fauchet, P.M. Photoluminescence and electroluminescence from porous silicon. J. Lumin. 1996, 70, 294–309. [Google Scholar] [CrossRef]
- Dohnalová, K.; Kůsová, K.; Pelant, I. Time-resolved photoluminescence spectroscopy of the initial oxidation stage of small silicon nanocrystals. Appl. Phys. Lett. 2009, 94. [Google Scholar] [CrossRef]






© 2017 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vásquez-Agustín, M.A.; Cortazar-Martínez, O.; González-Fernández, A.A.; Andraca-Adame, J.A.; Morales-Sánchez, A.; Aceves-Mijares, M. Luminescent Properties of Silicon Nanocrystals:Spin on Glass Hybrid Materials. Appl. Sci. 2017, 7, 72. https://doi.org/10.3390/app7010072
Vásquez-Agustín MA, Cortazar-Martínez O, González-Fernández AA, Andraca-Adame JA, Morales-Sánchez A, Aceves-Mijares M. Luminescent Properties of Silicon Nanocrystals:Spin on Glass Hybrid Materials. Applied Sciences. 2017; 7(1):72. https://doi.org/10.3390/app7010072
Chicago/Turabian StyleVásquez-Agustín, Marco Antonio, Orlando Cortazar-Martínez, Alfredo Abelardo González-Fernández, José Alberto Andraca-Adame, Alfredo Morales-Sánchez, and Mariano Aceves-Mijares. 2017. "Luminescent Properties of Silicon Nanocrystals:Spin on Glass Hybrid Materials" Applied Sciences 7, no. 1: 72. https://doi.org/10.3390/app7010072
APA StyleVásquez-Agustín, M. A., Cortazar-Martínez, O., González-Fernández, A. A., Andraca-Adame, J. A., Morales-Sánchez, A., & Aceves-Mijares, M. (2017). Luminescent Properties of Silicon Nanocrystals:Spin on Glass Hybrid Materials. Applied Sciences, 7(1), 72. https://doi.org/10.3390/app7010072





