1. Introduction
Recent developments in advanced dental materials [
1,
2], solid-oxide fuel-cell design to oxygen detection [
3], nuclear waste confinement [
4], optics [
5], medical prosthesis [
6,
7], and catalytic [
8] technologies have drawn attention towards the remarkable structural properties of zirconia (ZrO
2)-based ceramics. In all the current technological applications, ZrO
2-based ceramics are preferred due to their advanced mechanical properties such as high-fracture toughness and bulk modulus, corrosion resistance, chemical inertness, low chemical conductivity and biocompatibility [
9]. It is well known that three polymorphic forms of pure ZrO
2 can be found: the monoclinic state, P2
1/c, stable at temperatures below 1170 °C; the tetragonal phase, P4
2/nmc stable in the temperature range between 1170 and 2370 °C; and the cubic, Fm-3m phase, appearing at a temperature above 2370 °C [
10]. These ceramics have mechanical properties, promoting them to special applications. Under external stress, as grinding or impact, transition from the tetragonal (t) to monoclinic (m) phase can appear at normal temperatures, being followed by an increase of volume of at least 4%, causing compressive stress. This can cause material failure. Ceria (Ce
2O
3) or yttria (Y
2O
3) are used to stabilize these ceramics at room temperature and allowing t → m transformation to prevent crack propagation.
The main method used for the stabilization of the ZrO
2 tetragonal phase is the introduction of stabilization components in the zirconia lattice, such as Mg, Ce, Fe, Y,
etc. [
11,
12,
13,
14]. At nanoscale level, the metastable phase formation in ZrO
2 can be induced by including in the oxide structure some vacancy defects [
11]. Although, the stabilization effect of the oxygen vacancies in tetragonal ZrO
2 is not yet well understood (contrary to cubic ZrO
2) [
14], the concentration of oxygen vacancies in the lattice required to stabilize the tetragonal phase is found in phases like ZrO
1.97 and ZrO
1.98 for tetragonal zirconia doped by rare-earth elements, mainly cerium (Ce) and yttrium (Y).
Our aim is to investigate the influence of the phase change of ZrO2 ceramics following doping with Ce on the mechanical properties. We investigated the structure of these ceramics and the phase stabilization using X-ray and neutron diffraction, Scanning Electron Microscopy (SEM) as well as microstructure characterization methods, including micro-hardness measurements. Moreover, we employed Resonant Ultrasound Spectroscopy (RUS) as a non-destructive evaluation method in order to estimate the presence of low-density regions, state of sintering, and the presence and development of small cracks in the structure, by evaluating the complete elasticity matrix. RUS appears to be a feasible method for the inspection of zirconia ceramics based on the changes of the resonance frequencies, being easily applied for the quality control procedure of such ceramic samples; essentially monitoring of a single resonance peak within the frequency band included in experiment would be sufficient for this purpose. We emphasize the properties of zirconium-based ceramics for applications in the biomedical field, such as ceramic femoral heads used in hip implant procedures, which are very resistant to scratches resulting from debris caused by accumulation of bone parts, cement, or metal that occasionally fall between artificial joint surfaces, but are extremely fragile.
2. Materials and Methods
The Ce doped zirconia samples have been obtained through the standard ceramic technology [
15], a mixture of ceria and zirconia oxides (Zr
1-xCe
xO
2 (
x = 0–0.17)) being used in proportions established
a priori. After grinding and pressing, the samples were shaped in form of cylinders. Finally we treated cylindrical samples at a temperature of 1500 °C for six hours in air.
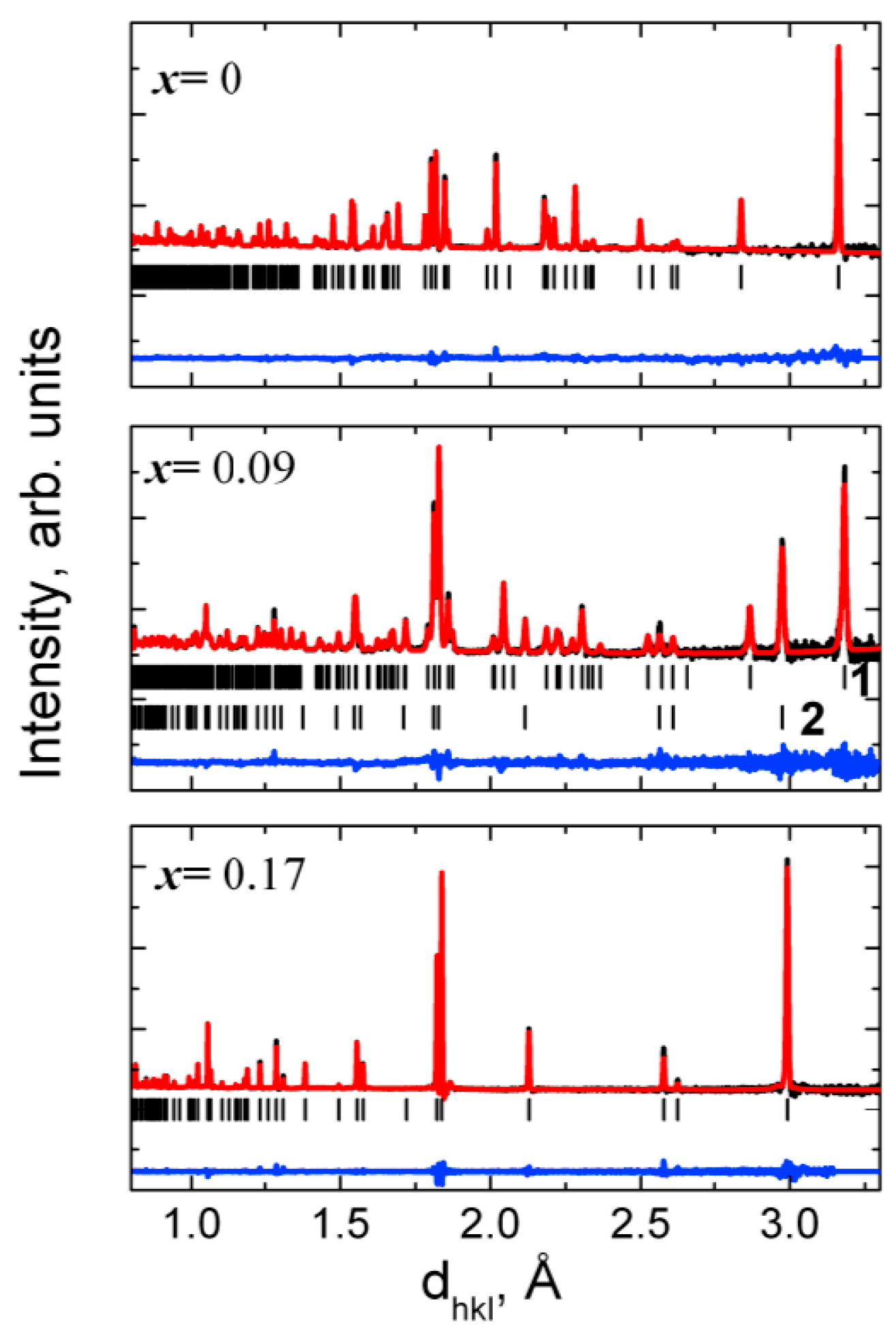
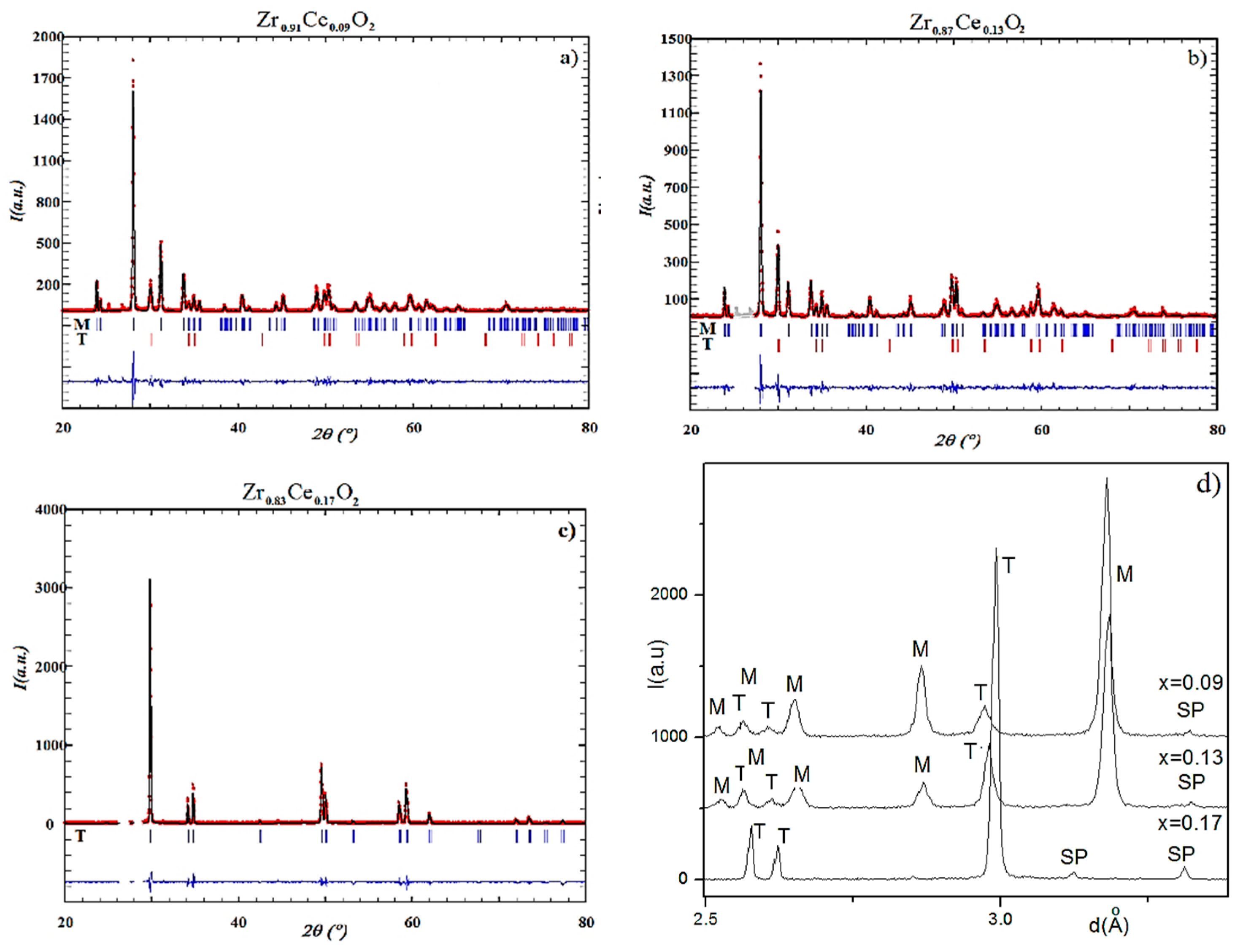
The phase composition, the lattice constants, space group, average size of coherent blocks, microstrains, and positions of cations and anions in the unit cell have been determined using XRD and ND data.
For XRD analysis the samples surface were prepared by a standard metallographic technique [
16]. XRD data were acquired with a X’pert Pro MPD PANalytical diffractometer (PANalytical Inc., Westborough, MA, USA) and a BRUKER AXS D8—Advance diffractometer (Bragg—Brentano geometry, CuKα, 2θ range of 20°–80°) (BRUKER AXS Inc., Madison, WI, USA). A powder diffraction software package, which includes the standards of the Crystallography Open Database (COD) [
17], was used in order to identify the phase composition of the samples. The microstrains and average size of crystalline blocks of the thin layer from the surface of the bulk samples were obtained using the XRD data, processed by the Rietveld method, PowderCell [
18]. LaB
6 was used as standard for XRD measurements (NIST SRM-660—a lanthanum hexaboride powder LaB
6, for line position and line shape).
The structural analysis of the Zr
1−xCe
xO
2 (
x = 0.0, 0.09, 0.13, 0.17) ceramic samples was performed also using the time-of-flight (TOF) High Resolution Fourier Diffractometer (HRFD) at the IBR-2 pulsed reactor in Joint Institute for Nuclear Research, Dubna, Russia [
19]. At the HFRD diffractometer, the correlation technique of data acquisition is used, providing a very high resolution of Δ
d/
d ~ 0.001, practically constant in a wide interval of
dhkl spacings [
19]. We collected the high resolution diffraction patterns by using a detector placed at backscattering angles 2θ = ±152°,
dhkl = 0.6–3.6 Å. It is important to mention that via X-ray diffraction, a layer of typically 10 μm thickness has been investigated, whereas in the case of neutron diffraction, a thickness of tens of centimeters of a sample has been investigated.
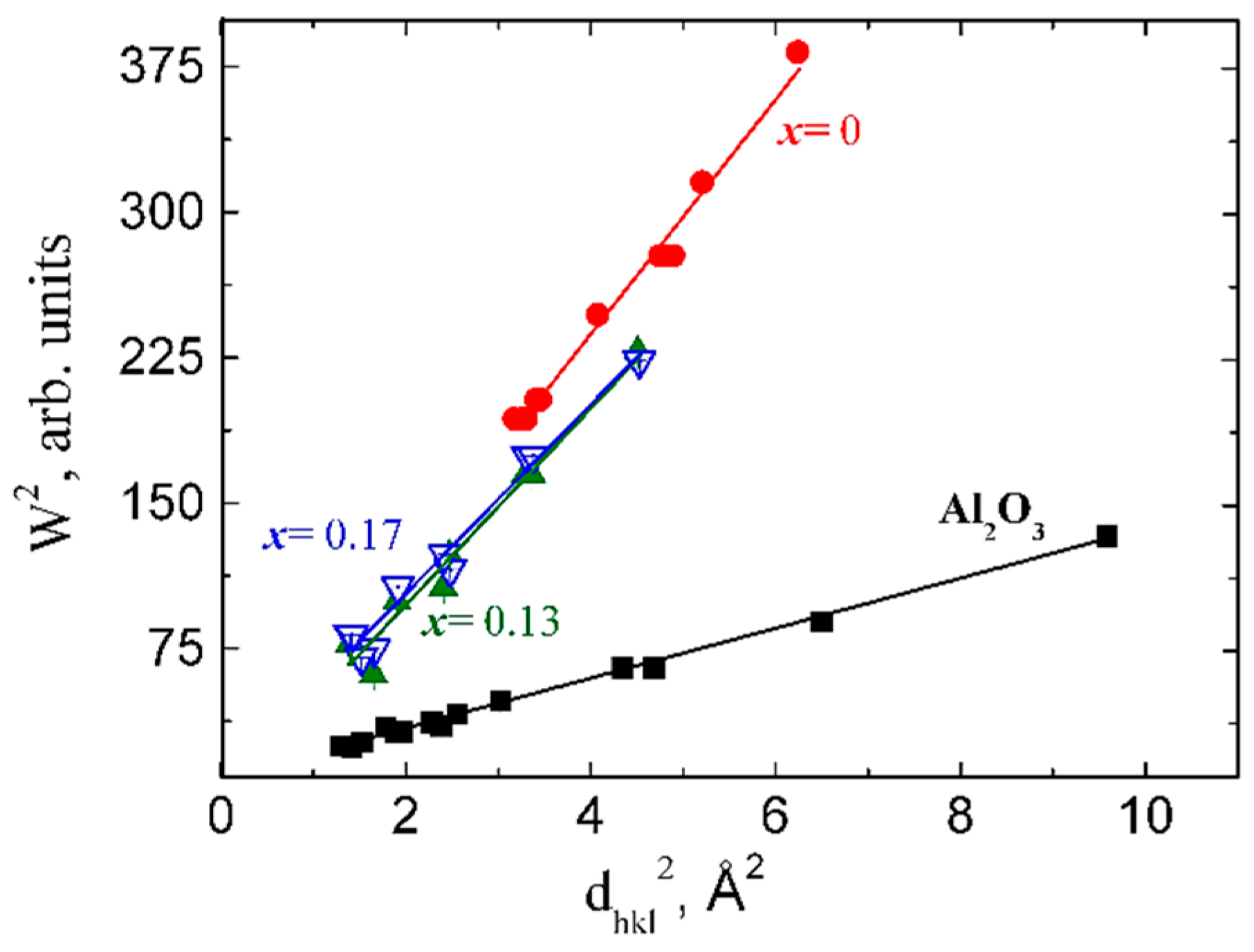
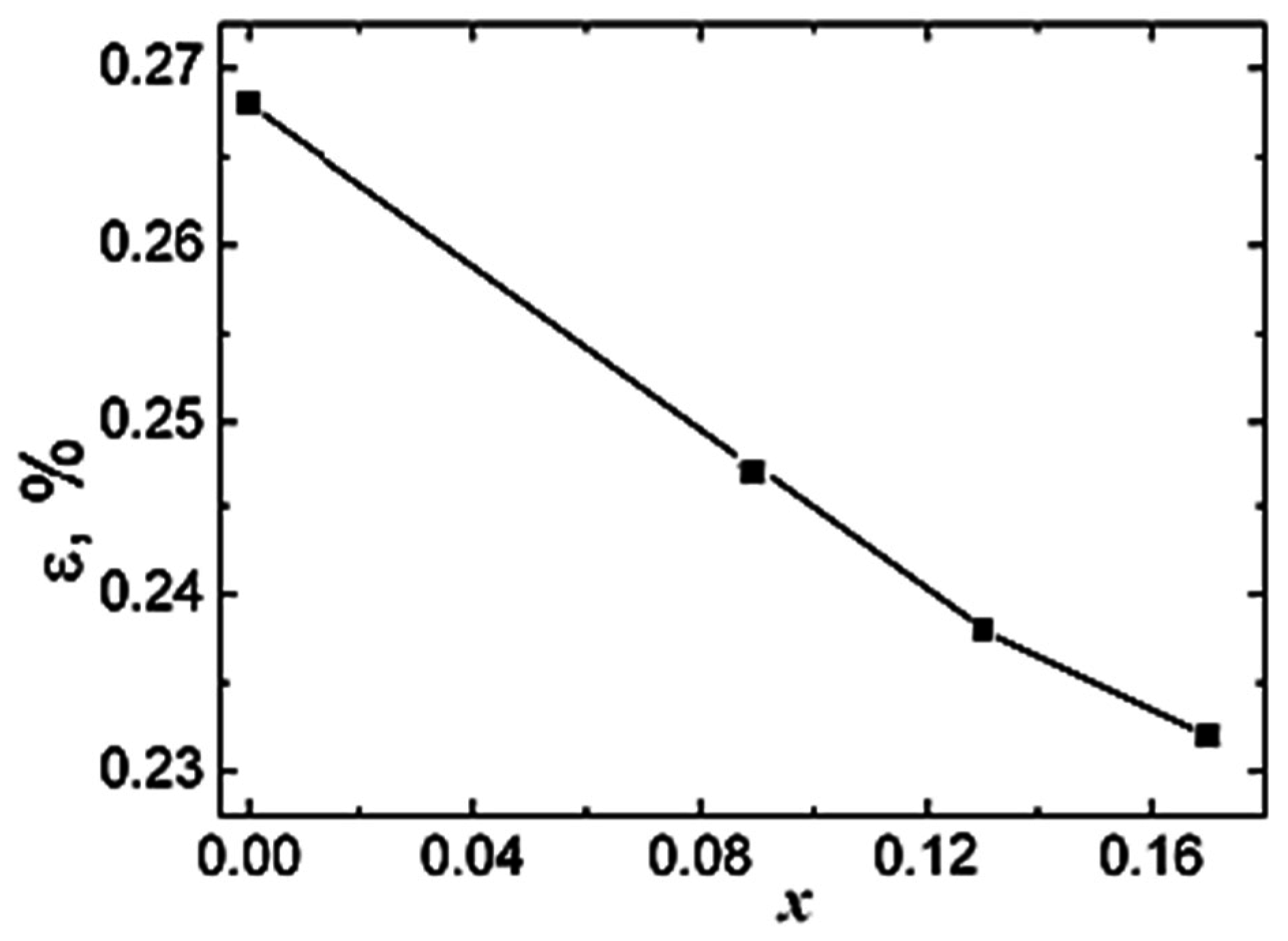
Information about microstructural parameters, such as crystalline size (average size of crystalline blocks) and microstrains were obtained from the lines broadening. From the dependence between the square of Full Width at Half Maximum (W) of neutron peaks and the square of the interplanar spacing,
d2, described by the Equation (1) [
20,
21]
we extracted the microstrain values (ε).
Here, C1, C2 are the refining constants of the resolution function of HRFD obtained from a standard Al2O3 (SRM-676 of NIST, USA); C3 ~ ε2, where ε is the microstrain; C4 ~ 1/L where L is the size of the coherently scattering blocks. The values of ε and L can be determined from the true physical characteristics of line broadening, whose value is defined as the difference between broadening of peaks of the experimental sample and the standard Al2O3.
RUS is a complex method that allows the determination of elastic constant and the elements of elasticity matrix for samples with certain shapes [
22,
23,
24,
25,
26]. Sample geometry affects data acquisition [
26]. For cylinders with a high ratio of length to diameter we have few excitation modes and the spectra are simple [
26,
27]. In the case of the studied samples the ratio is around the unit and the spectrum requires more analysis than a long bar. The increasing of spectral complexity supposes adjacent analyses before concluding about these samples. In order to determine a parameter, more resonance frequencies must be searched [
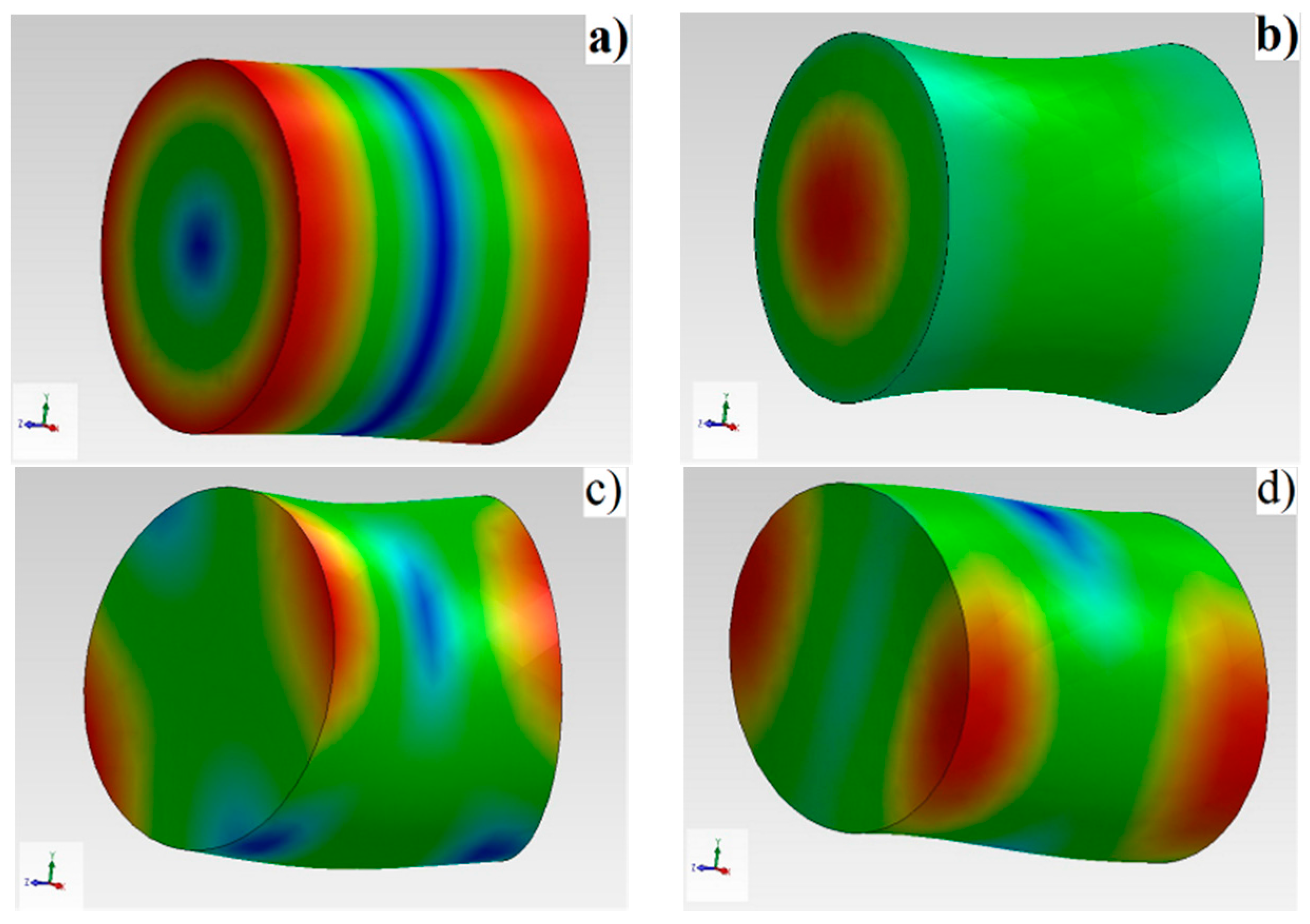
25]. This requires a lot of computational calculation time and repeated tests. The same number of normal modes for a short cylinder compared with a long cylinder requires a narrower frequency range.
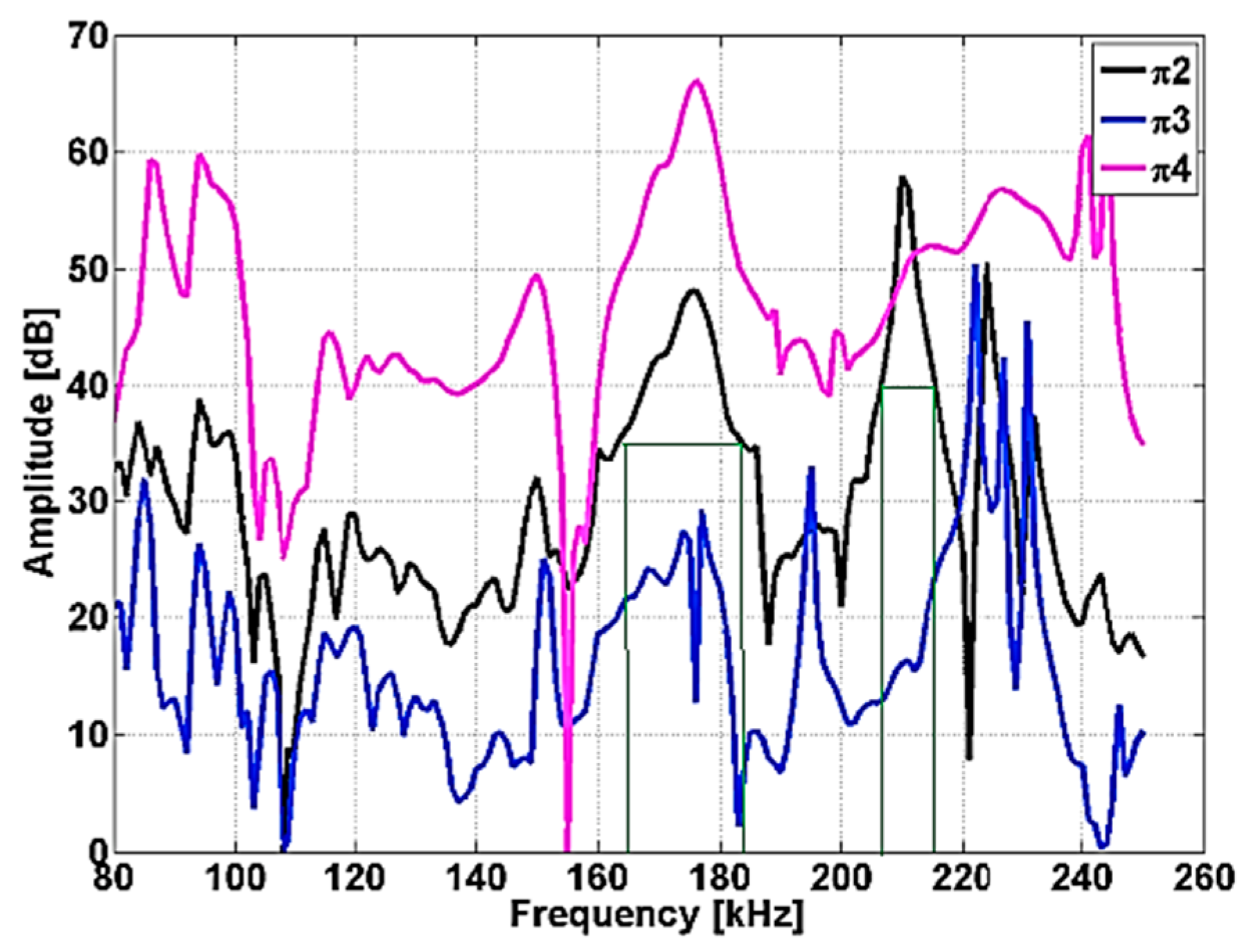
In the case of cylindrical samples, having diameter approximately equal with height, the first mode is the fundamental torsional mode allowing the direct determination of shear modulus. RUS involves scanning the resonance structure of a compact specimen (in our case a ceramic cylinder) with the aim of determining its mechanical properties [
28,
29]. In principle, RUS is composed of three parts. Successful use of this method implies the obtaining of a resonant ultrasound spectrum, meaning obtaining the frequency answer for a certain excitation for the analyzed sample. It is necessary to develop a computing method which can help in predicting the resonant ultrasound spectrum for the estimated parameters. The third component represents the iterative refinement of the estimated parameters, in agreement with the measured spectrum. Each of the steps mentioned above have multiple solutions. In the classical RUS experiment, a sample is placed between two US transducers. The primary advantages of this contact technique are its relative simplicity and low cost.
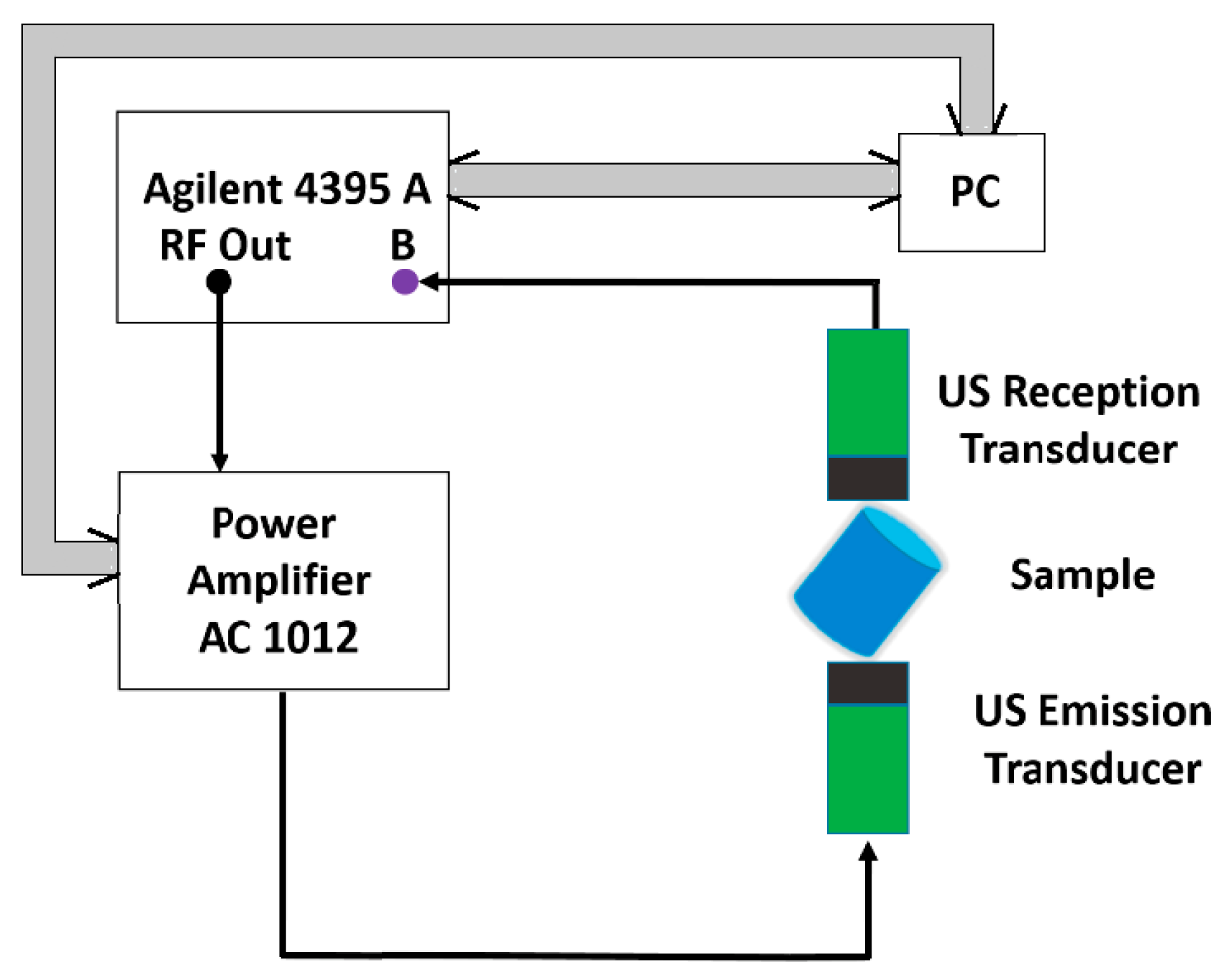
For the RUS method, the samples have been supported by two identical piezoelectric ultrasonic transducers, for emission and reception respectively, placed at the opposite ends of the cylindrical sample (see
Figure 1). The measurements are carried out via transducers with contact. The coupling of the transducer with the specimen influences which modes are measured. When the sample is pinned on its edge, more modes are excited and the modes are better defined than when the transducers are placed on the ends of the cylinder. The probe is fixed between the emission and reception transducers in order to accomplish the condition of a stress free surface. The equipment allows the setting so that for the established position of the cylindrical sample, the contact on the edge assures the excitation of a maximum number of possible resonances for the fixed geometry.
A Network/Spectrum/Impedance Analyzer 4395A (Agilent Technologies, Santa Clara, CA, USA), generates a sweep frequency ranging between 80 and 250 kHz with a 1 kHz step. The signal is amplified using a Power amplifier AG 1012 (T&C Power Conversion Inc., Rochester, NY, USA) and applied to the US emission transducer is applied to the B port of the 4395 A Agilent, the spectrum being acquired by a PC used to program the functioning of the equipment as well with a numerical code developed in Matlab 2012b (The MathWorks Inc., Natick, MA, USA, 2012) via a PCIB interface (National Instruments, Mopac Expwy, Austin, TX, USA).
The command of the power amplifier is made using the same PC via a RS232 interface (National Instruments). For the determination of the elasticity and shear moduli of the samples, the propagation speed of the longitudinal and transversal ultrasound waves were measured using a transmission procedure. The emission transducer is applied at one base of the sample via a delay line from Perspex® (Perspex Acrylic Brand, Darwen, Lancashire, UK) 20 mm in length, while the reception transducer is applied on the other base.
Figure 1.
RUS Experimental setup schematic block diagram. Thin lines correspond to coaxial cable for sinusoidal wave transmission; thick lines are GPIB cable for digitalized data.
Figure 1.
RUS Experimental setup schematic block diagram. Thin lines correspond to coaxial cable for sinusoidal wave transmission; thick lines are GPIB cable for digitalized data.
A G5KB GE transducer (General Electric Measurement & Control, Boston, MA, USA) with a central frequency of 5 MHz has been used for the measurement of the propagation speed of the longitudinal waves, the coupling being made using a ZG-F Krautkramer (General Electric Measurement & Control, Boston, MA, USA), whereas for the transversal waves, a MB4Y GE transducer (General Electric Measurement & Control, Boston, MA, USA) was used, with a central frequency of 4 MHz, the coupling being made with honey. The generation of emission impulses and the reception of signals delivered by the reception transducer has been made using a PR 5073 Pulser Receiver former Panametrics NDT USA (Olympus Corporation Waltham, MA, USA).
The densities of the samples were obtained by means of Archimedes’ method, by using water as fluid. The measurements of microhardness were performed by Vickers method, with a load of 50 g and a dwell time of 20 s.
4. Conclusions
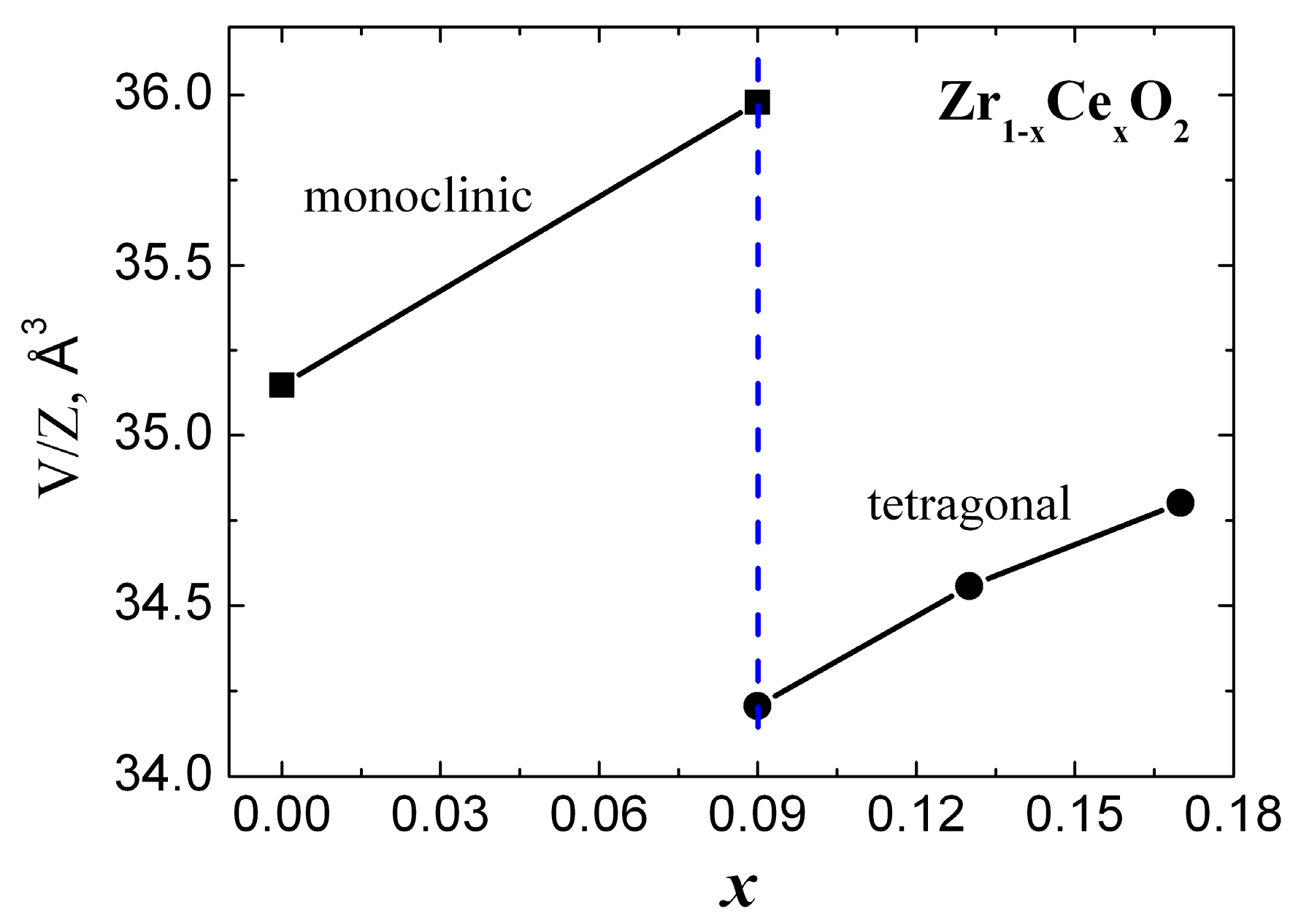
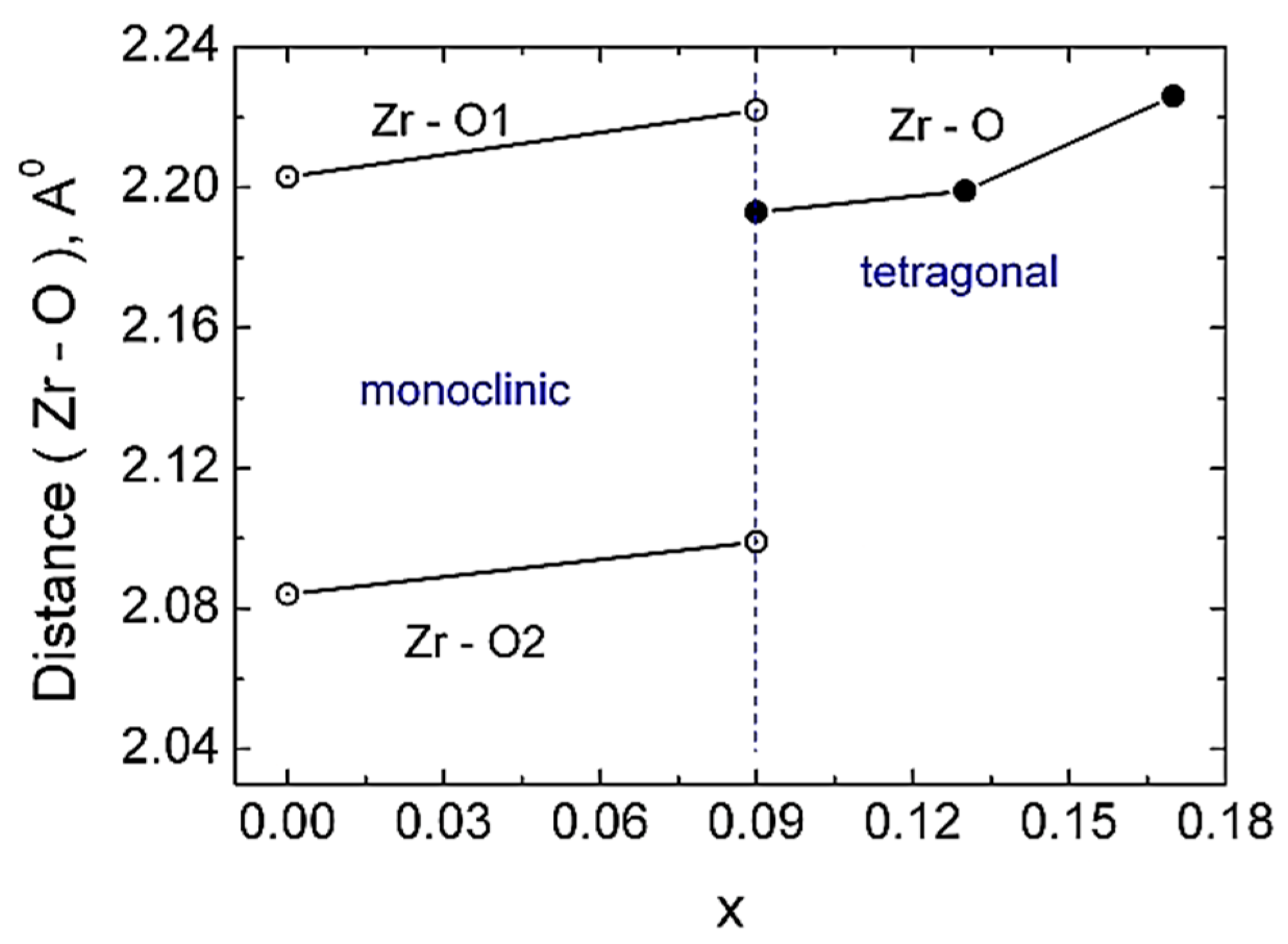
The substitution of the Zr with Ce in Zr1-xCexO2 leads to a change of the phase composition, a gradual transition from the monoclinic to tetragonal structure. Concerning the substitution of Zr with Ce we observed a difference of the phase composition between the surface layer of the sample and the phase composition of the bulk samples. We attributed this difference to the various oxygen concentration in the surface layer and in the bulk sample. The investigations performed by means of neutron diffractometry, which “viewed” a large volume of the samples, indicated a transition from the monoclinic to tetragonal phase for x = 0.09. On other hand, XRD investigations, which “viewed” only a thin layer have shown that the phase composition of the samples corresponding to x = 0.09 and 0.13 represents a mixture between a monoclinic phase and a tetragonal phase, the tetragonal phase concentration increasing with the increase of Ce concentration. The unit cell volumes of the tetragonal and monoclinic phases and Zr-O distances increase with the increase of Ce concentration, for the inner part of the samples. The calculated and measured densities increase also with the increase of Ce concentration. A maximum of the average size of coherent blocks and of the microstrain appear in the tetragonal phase for the sample with x = 0.13. The increase of Ce concentration leads also to an increase of the volumes of both monoclinic and tetragonal unit cells. From the variation of unit cell volume of the surface layer, compared with those corresponding to the unit cell volume of the sample core, we conclude that a small but systematic decrease of the large radii cation concentration takes place at the surface layer. An increase of the relative density and of the mechanical parameters (elasticity and shear moduli) was obtained with the increase of Ce concentration.
RUS is a reliable technique, which emphasizes the eigenfrequency intervals which exhibit a slight change as a function of samples composition, for monitoring structural modifications related to sample density. Slight material anisotropy leads to splitting of the higher modes but not of the fundamental torsion mode. In the case of the studied samples, whose ratio is around the unit, the interpretation is favorable because the torsional mode is the lowest one, well separated from the others for ν > 0, allowing immediate extraction of the shear modulus and its damping. The initiation of fracture of ceramic elements can be due to the presence of low density zone-containing dispersed high density agglomerates in the volume and any deformation which will be immediately apparent through changes in the resonance modes, with deviations from the normal spectrum. RUS can be used for quality control of certain ceramic elements of hip prosthesis, such as femoral heads. If the elements are incorrectly sintered, with a density smaller than the prescribed value and the elastic and shear moduli smaller, important modifications appear in the shape of the spectrum and the resonance frequencies.
Future research in this area should concentrate on the SEM analysis concerning the ceramic samples, whose aspects change dramatically with increasing concentrations of Ce ions, an effect which needs to be further analyzed in a future study.