The Effects of Carbonation and Elevated Temperatures on the Properties of Magnesium-Based Mortar
Abstract
1. Introduction
2. Materials and Methods
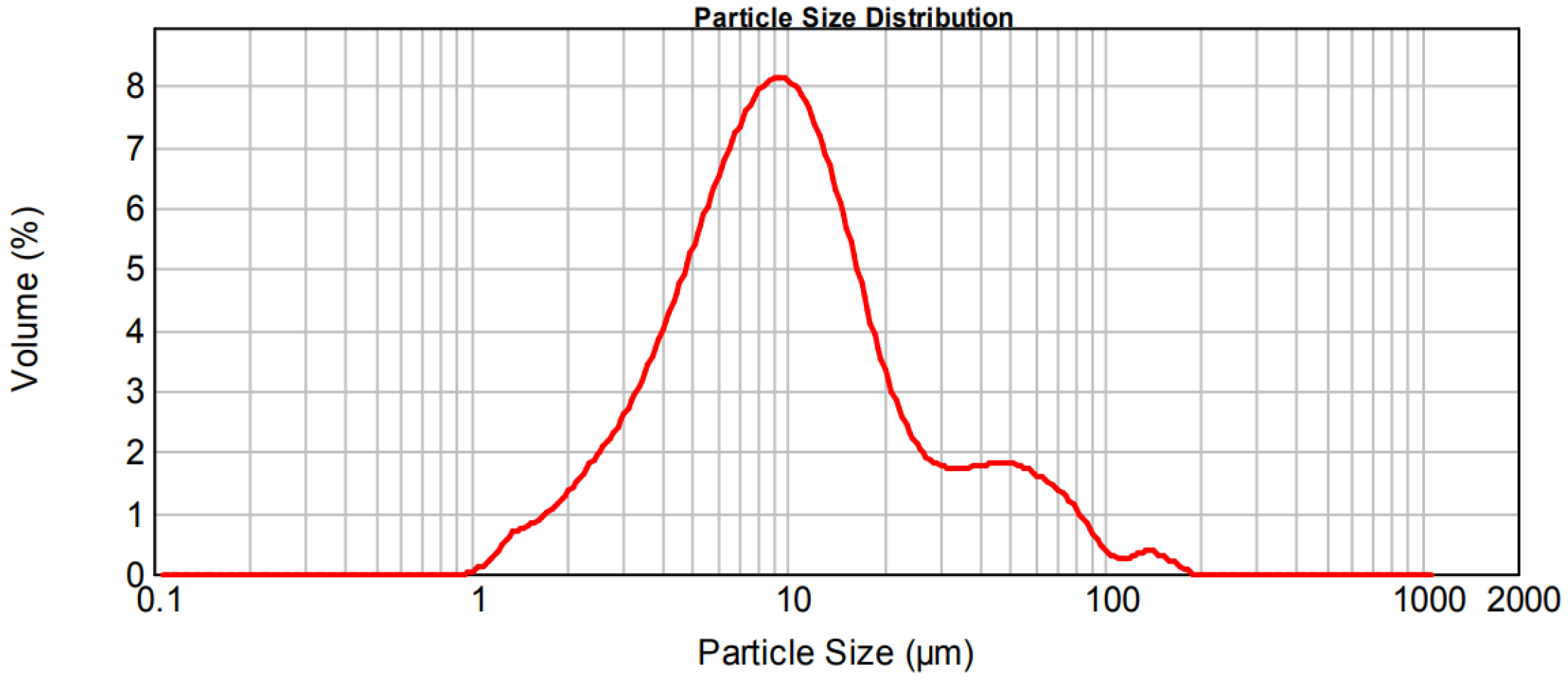
2.1. Materials and Mix Proportions
2.2. Curing Conditions
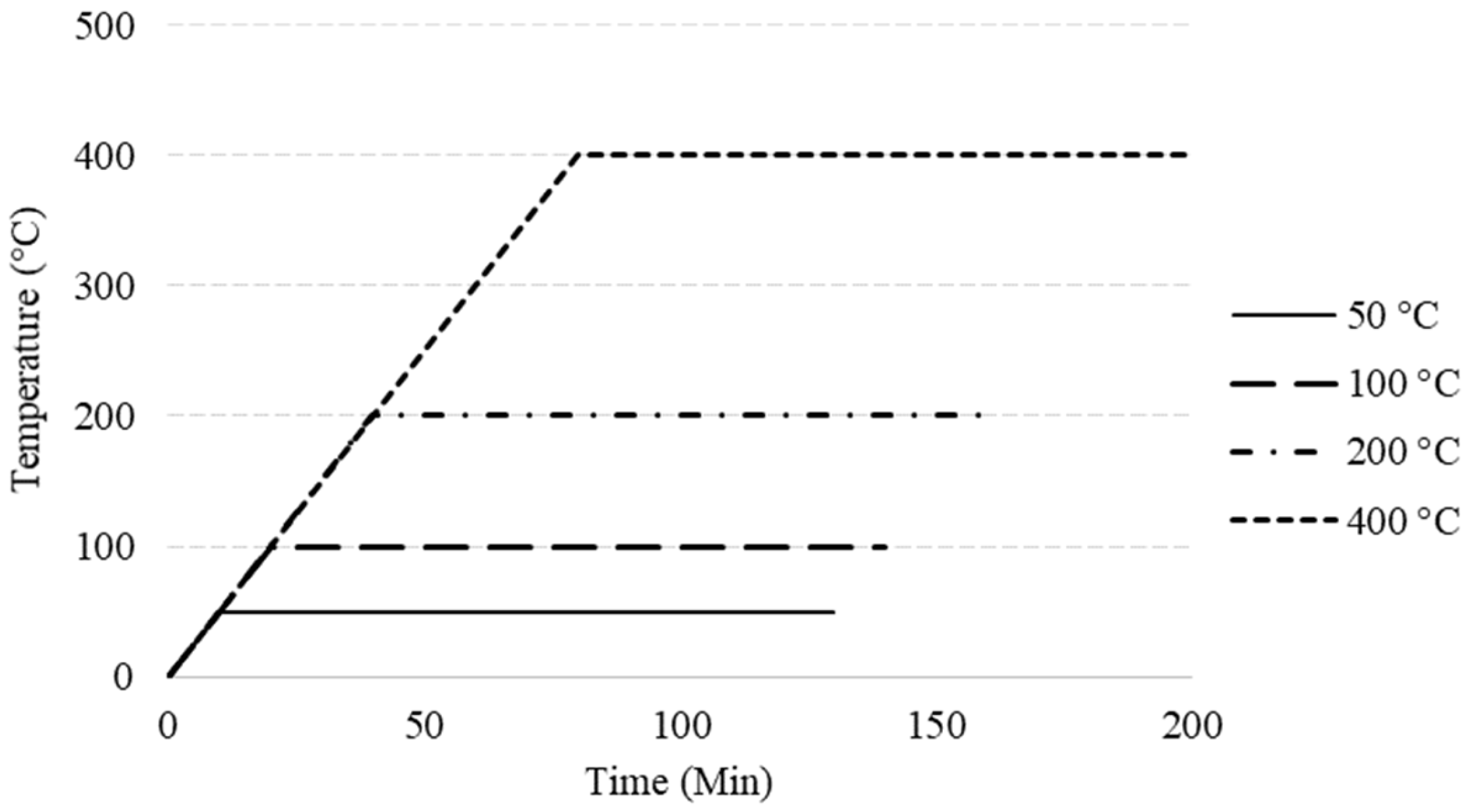
2.3. Heating Procedures
2.4. Mass Change Monitoring and Compression Tests
2.5. Powder Preparation and TGA/XRD Protocols
3. Results
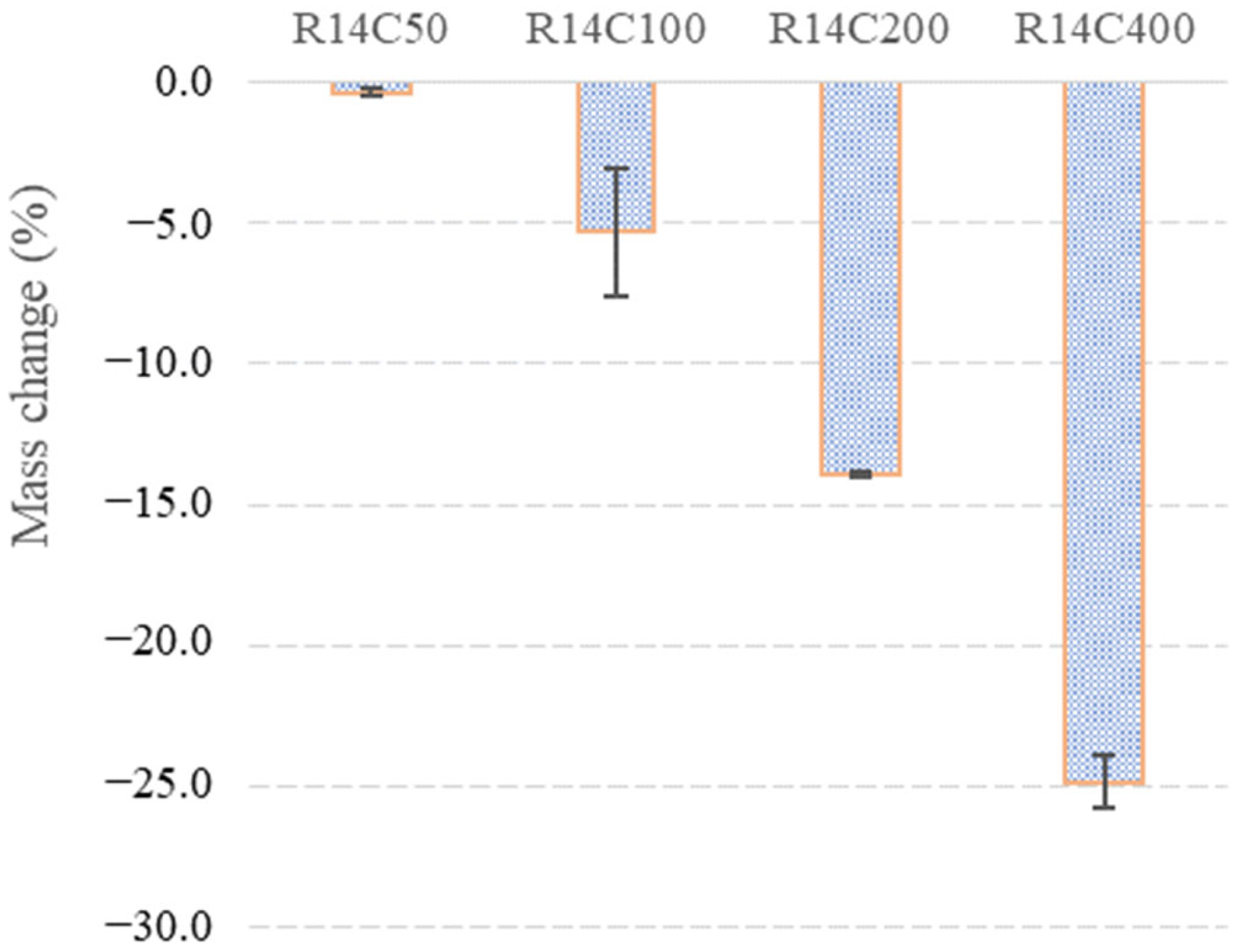
3.1. Mass Changes
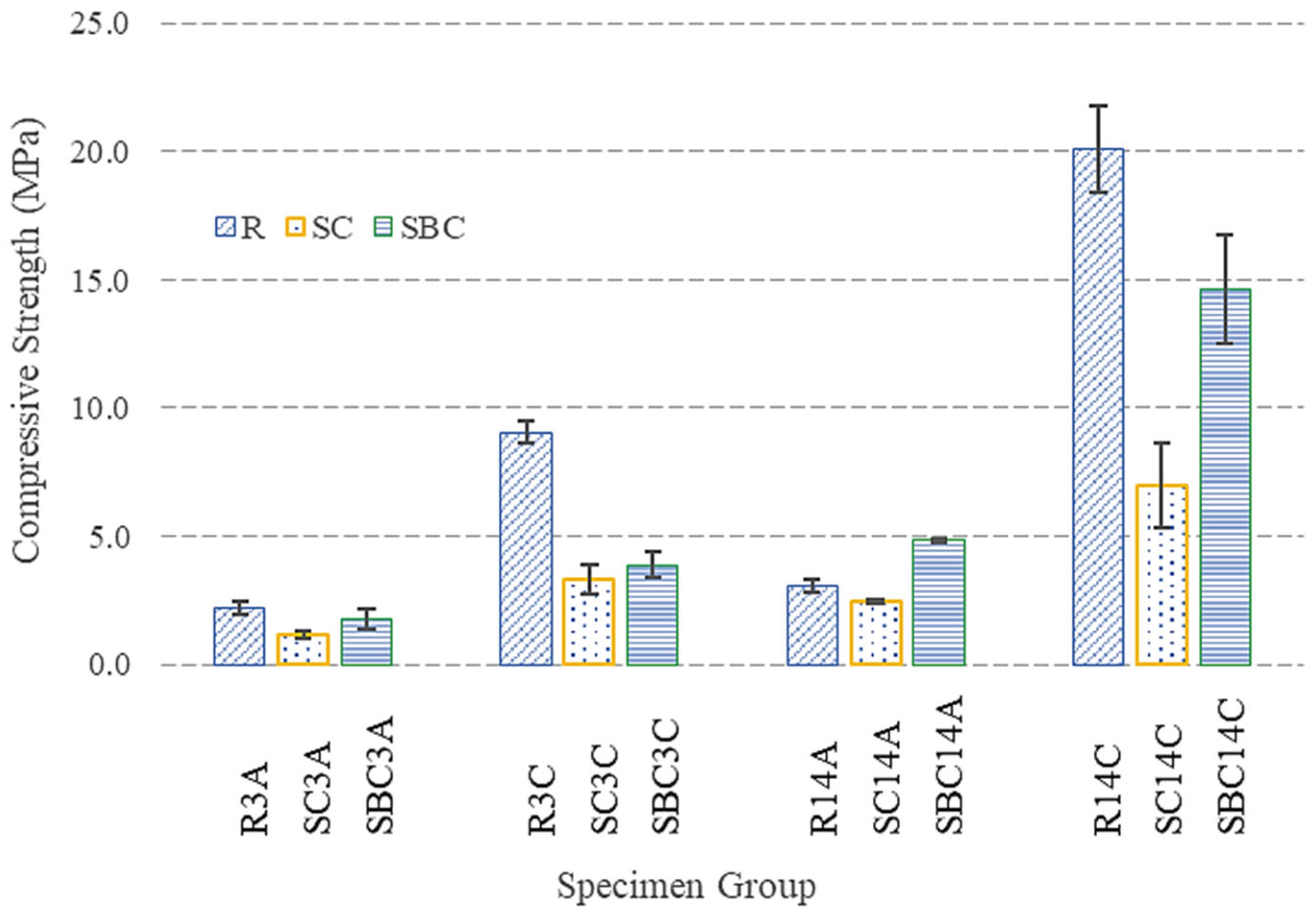
3.2. Compressive Strength Measurements
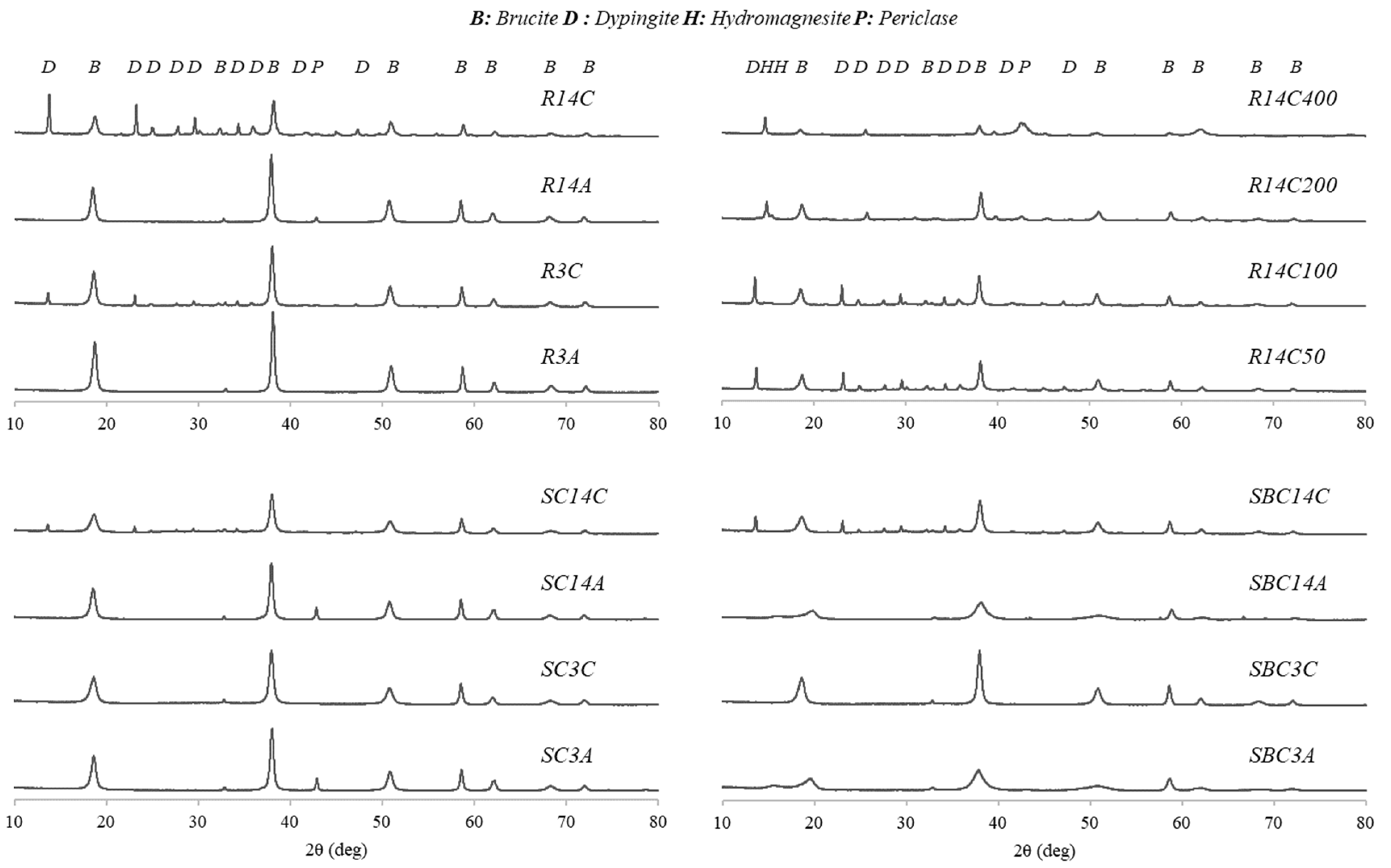
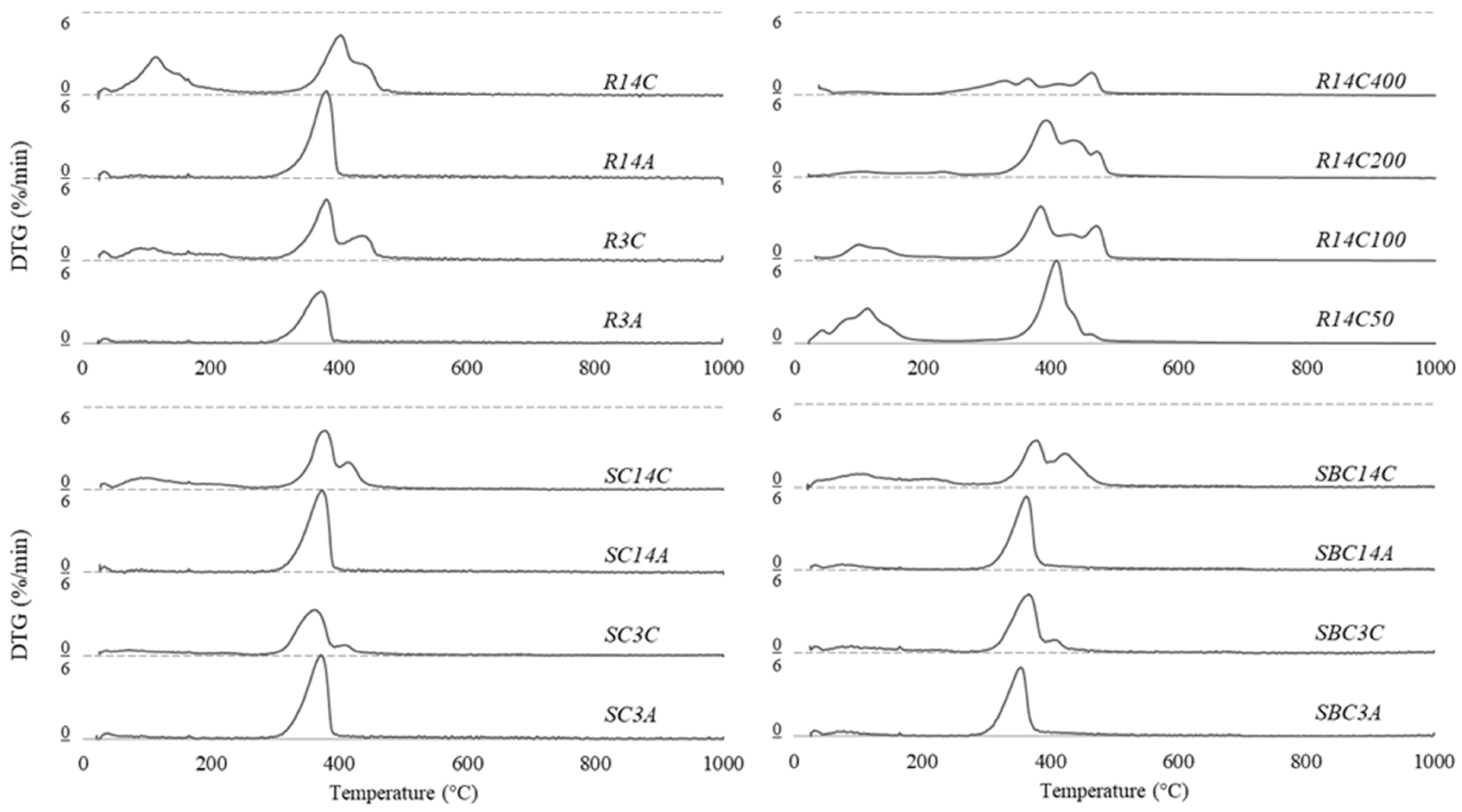
3.3. Microstructural Analyses
4. Conclusions
- Curing time causes an increase in compressive strength after both air and ACC. However, the rate of strength enhancement decreases over time. Because ACC is more difficult to apply than other methods, optimization of the curing time is necessary and can be further investigated in the future.
- After 14 days of air curing, the compressive strength of RMC-based mortar (R14A) reached 3 MPa. On the other hand, ACC improved the compressive strength of the mortar, with the carbonated reference group achieving a value of 20.1 MPa after 14 days of curing (R14C), representing an increase of roughly 6.5 times. A comparable improvement was observed in the mortars employing precursors, demonstrating an approximate threefold increase in both groups.
- Sodium-based precursors had a negative impact on the carbonation and the strength development compared to the reference series. After 14 days of ACC, the lowest compressive strength was measured in the SC14C group, 7 MPa. This was attributed to the effect of sodium carbonates on the mixture pH. The decrease in pH might be the reason for the reduction in both hydration and carbonation rate which can be seen in TGA and XRD results. According to the literature, sodium bicarbonate less strongly affects the pH level of the mixture compared to sodium carbonate; therefore, its negative impact on strength was more limited in this study. The compressive strength of the SBC14C group was measured as 14.6 MPa. Moreover, air-cured samples incorporated with sodium bicarbonate yielded better strength results than the reference group.
- Temperature rise caused an increase in the material’s compressive strength at first, then resulted in a significant degradation. An increase was observed at 50 °C and it was around 35% compared to the strength of R14C. The residual strength was measured as 4.5 MPa after 400 °C heat exposure. In fact, this temperature does not cause significant deterioration in conventional PC materials. However, the decompositions in the microstructure of magnesium-derived materials are significantly completed at temperatures below 450 °C.
- The hydration of magnesium oxide produced brucite, which majorly governs the strength development of air-cured specimens, whereas carbonation led to dypingite formations contributing to the mechanical improvement of the material. On the other hand, temperature rise led to the disintegration of dypingite and the extent of decarbonation was the highest in the specimens subjected to 400 °C, resulting in the reappearance of magnesium oxide.
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ACC | Accelerated CO2 Curing |
| BET | Brunauer–Emmett–Teller |
| DTG | Differential Thermogravimetry |
| ICDD | International Centre for Diffraction Data |
| PC | Portland Cement |
| Powder Diffraction File | |
| RH | Relative Humidity |
| RMC | Reactive MgO Cement |
| TGA | Thermal Gravimetric Analysis |
| XRD | X-Ray Diffraction |
| XRF | X-Ray Fluorescence |
References
- US Geological Survey. Mineral Commodity Summaries 2021; US Geological Survey: Reston, VA, USA, 2022. [Google Scholar]
- Andrew, R.M. Global CO2 emissions from cement production. Earth Syst. Sci. Data 2022, 10, 195–217. [Google Scholar] [CrossRef]
- International Energy Agency (IEA); World Business Council for Sustainable Development (WBCSD). Technology Roadmap: Low-Carbon Transition in the Cement Industry; IEA/WBCSD: Paris, France, 2018; pp. 18–20. [Google Scholar]
- Birchal, V.S.S.; Rocha, S.D.F.; Ciminelli, V.S.T. The effect of magnesite calcination conditions on magnesia hydration. Miner. Eng. 2000, 13, 1629–1633. [Google Scholar] [CrossRef]
- Bocanegra-Bernal, M.H. Agglomeration of magnesia powders precipitated from sea water and its effects on uniaxial compaction. Mater. Sci. Eng. A 2002, 333, 176–186. [Google Scholar] [CrossRef]
- Barba, D.; Brandani, V.; Di Giacomo, G.; Foscolo, P.U. Magnesium oxide production from concentrated brines. Desalination 1980, 33, 241–250. [Google Scholar] [CrossRef]
- Dung, N.T.; Unluer, C. Carbonated MgO concrete with improved performance: The influence of temperature and hydration agent on hydration, carbonation and strength gain. Cem. Concr. Compos. 2017, 82, 152–164. [Google Scholar] [CrossRef]
- German, A.; Winnefeld, F.; Lura, P.; Rentsch, D.; Lothenbach, B. Hydrous carbonate-containing brucite (HCB) in MgO/hydromagnesite blends. Cem. Concr. Res. 2023, 173, 107304. [Google Scholar] [CrossRef]
- Lothenbach, B.; Bernard, E.; German, A.; Winnefeld, F. MgO-based binders. Ce/Papers Proc. Civ. Eng. 2023, 6, 342–356. [Google Scholar] [CrossRef]
- Kuenzel, C.; Zhang, F.; Ferrándiz-Mas, V.; Cheeseman, C.R.; Gartner, E.M. The mechanism of hydration of MgO-hydromagnesite blends. Cem. Concr. Res. 2018, 103, 123–129. [Google Scholar] [CrossRef]
- Ruan, S.; Qiu, J.; Yang, E.-H.; Unluer, C. Fiber-reinforced reactive magnesia-based tensile strain-hardening composites. Cem. Concr. Compos. 2018, 89, 52–61. [Google Scholar] [CrossRef]
- Winnefeld, F.; Epifania, E.; Montagnaro, F.; Gartner, E.M. Further studies of the hydration of MgO-hydromagnesite blends. Cem. Concr. Res. 2019, 126, 105912. [Google Scholar] [CrossRef]
- Zhou, Z.; Bernardi, E.; Lothenbach, B. Effect of nesquehonite and silica on magnesia-silicate-carbonate cements. Cem. Concr. Res. 2025, 194, 107892. [Google Scholar] [CrossRef]
- Scott, A.; Oze, C.; Shah, V.; Yang, N.; Shanks, B.; Cheeseman, C.; Marshall, A.; Watson, M. Transformation of abundant magnesium silicate minerals for enhanced CO2 sequestration. Commun. Earth Environ. 2021, 2, 25. [Google Scholar] [CrossRef]
- Forero, J.A.; Bravo, M.; Pacheco, J.; de Brito, J.; Evangelista, L. Fracture behaviour of concrete with reactive magnesium oxide as alternative binder. Appl. Sci. 2021, 11, 2891. [Google Scholar] [CrossRef]
- Unluer, C.; Al-Tabbaa, A. Impact of hydrated magnesium carbonate additives on the carbonation of reactive MgO cements. Cem. Concr. Res. 2013, 54, 87–97. [Google Scholar] [CrossRef]
- Liska, M.; Al-Tabbaa, A. Ultra-green construction: Reactive magnesia masonry products. Proc. Inst. Civ. Eng. Waste Resour. Manag. 2009, 162, 185–196. [Google Scholar] [CrossRef]
- Shand, M.A. The Chemistry and Technology of Magnesia; John Wiley & Sons: Hoboken, NJ, USA, 2006. [Google Scholar]
- Taylor, H.F.W. Cement Chemistry, 2nd ed.; Thomas Telford: London, UK, 1997. [Google Scholar]
- Unluer, C.; Al-Tabbaa, A. Effect of aggregate size distribution on the carbonation of reactive magnesia based porous blocks. In Proceedings of the 18th Annual International Sustainable Development Research Conference, Hull, UK, 24–26 June 2012. [Google Scholar]
- Botha, A.; Strydom, C.A. Preparation of a magnesium hydroxy carbonate from magnesium hydroxide. Hydrometallurgy 2001, 62, 175–183. [Google Scholar] [CrossRef]
- Mo, L.; Liu, M.; Al-Tabbaa, A.; Deng, M.; Lau, W.Y. Deformation and mechanical properties of quaternary blended cements containing ground granulated blast furnace slag, fly ash and magnesia. Cem. Concr. Res. 2015, 71, 7–13. [Google Scholar] [CrossRef]
- Mi, T.; Yang, E.H.; Unluer, C. Investigation of the properties of reactive MgO-based cements and their effect on performance. Cem. Concr. Compos. 2023, 138, 104984. [Google Scholar] [CrossRef]
- Hay, R.; Dung, N.T.; Lesimple, A.; Unluer, C.; Celik, K. Mechanical and microstructural changes in reactive magnesium oxide cement-based concrete mixes subjected to high temperatures. Cem. Concr. Compos. 2021, 118, 103955. [Google Scholar] [CrossRef]
- Ruan, S.; Qiu, J.; Yang, E.-H.; Unluer, C. Mechanical behavior of carbonated MgO-based Engineered Cementitious Composite after high temperatures exposure. Cem. Concr. Compos. 2023, 138, 104255. [Google Scholar]
- Chen, J.; Li, X.; Wang, Y.; Zhang, H. Mechanical performance and microstructure evolution of MgO-based binder systems under elevated temperatures. J. Build. Eng. 2024, 62, 105284. [Google Scholar]
- Tamer, T.; Mazaheri, H.; Ergenç, D.; Akgül, Ç. Biochar as a bio-renewable addition to enhance carbonation of reactive magnesium oxide calcium aluminate cement blends. Mater. Struct. 2025, 58, 83. [Google Scholar] [CrossRef]
- Hay, R.; Dung, N.T.; Lesimple, A.; Unluer, C.; Celik, K. Mechanical and microstructural changes in reactive magnesium oxide cement cured under ambient and accelerated conditions. Cem. Concr. Compos. 2024, 150, 105523. [Google Scholar]
- ASTM C109/C109M; Standard Test Method for Compressive Strength of Hydraulic Cement Mortars (Using 2-in. or [50-mm] Cube Specimens). ASTM International: West Conshohocken, PA, USA, 2020.
- Wang, Y.; He, F.; Wang, J.; Hu, Q. Comparison of effects of sodium bicarbonate and sodium carbonate on the hydration and properties of Portland cement paste. Materials 2019, 12, 1033. [Google Scholar] [CrossRef]
- Ma, S.; Akca, A.H.; Esposito, D.; Kawashima, S. Influence of aqueous carbonate species on hydration and carbonation of reactive MgO cement. J. CO2 Util. 2020, 41, 101260. [Google Scholar] [CrossRef]
- Dung, N.T.; Unluer, C. Performance of reactive MgO concrete under increased CO2 dissolution. Cem. Concr. Res. 2019, 118, 92–101. [Google Scholar] [CrossRef]
- Badjatya, P.; Akca, A.H.; Fraga Alvarez, D.V.; Chang, B.; Ma, S.; Pang, X.; Wang, E.; Van Hinsberg, Q.; Esposito, D.V.; Kawashima, S. Carbon-negative cement manufacturing from seawater-derived magnesium feedstocks. Proc. Natl. Acad. Sci. USA 2022, 119, e2114680119. [Google Scholar] [CrossRef]
- Akca, A.H.; Ma, S.; Esposito, D.; Kawashima, S. Evaluation of mechanical performance of compacted magnesium hydroxide after carbonation curing. J. Mater. Civ. Eng. 2022, 34, 04022056. [Google Scholar] [CrossRef]
- Raade, G. Dypingite, a new hydrous basic carbonate of magnesium, from Norway. Am. Mineral. 1970, 55, 1457–1465. [Google Scholar]
- Ruan, S.; Unluer, C. Influence of mix design on the carbonation, mechanical properties and microstructure of reactive MgO cement-based concrete. Cem. Concr. Compos. 2017, 80, 104–114. [Google Scholar] [CrossRef]
- Liu, C.; Zhang, R.; Shen, K.; Liu, T.; Wen, W.; Wang, D. An IN SITU Kinetic Study of the Dehydration of Brucite Using Synchrotron X-ray Powder Diffraction. Can. Mineral. 2018, 56, 101–108. [Google Scholar] [CrossRef]
- Ilango, N.K.; Nguyen, H.; German, A.; Winnefeld, F.; Kinnunen, P. Role of magnesium acetate in hydration and carbonation of magnesium oxide-based cements. Cem. Concr. Res. 2024, 175, 107357. [Google Scholar] [CrossRef]
- Myagmarjav, O.; Ryu, J.; Kato, Y. Dehydration kinetic study of a chemical heat storage material with lithium bromide for a magnesium oxide/water chemical heat pump. Prog. Nucl. Energy 2015, 82, 153–158. [Google Scholar] [CrossRef]
- Zhao, H.; Hanein, T.; Li, N.; Alotaibi, A.; Li, A.; Walling, S.; Kinoshita, H. Acceleration of M-S-H gel formation through the addition of alkali carbonates. In Proceedings of the 15th International Congress on the Chemistry of Cement, Prague, Czech Republic, 16–20 September 2019. [Google Scholar]
- Devasahayam, S.; Strezov, V. Thermal decomposition of magnesium carbonate with biomass and plastic wastes for simultaneous production of hydrogen and carbon avoidance. J. Clean. Prod. 2018, 174, 1089–1095. [Google Scholar] [CrossRef]
- Unluer, C.; Al-Tabbaa, A. Characterization of Light and Heavy Hydrated Magnesium Carbonates Using Thermal Analysis. J. Therm. Anal. Calorim. 2014, 115, 595–607. [Google Scholar] [CrossRef]
- Yamamoto, G.; Kyono, A.; Okada, S. Thermal decomposition process of dypingite Mg5(CO3)4(OH)2·5H2O. Mater. Lett. 2022, 308, 131125. [Google Scholar] [CrossRef]


| Group | w/b | RMC | Water | 0.12 M Na2CO3 | 0.12 M NaHCO3 | River Sand | Air Curing (Days) | CO2 Curing (Days) | Heat Exposure (°C) | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| (kg/m3) | 3 | 14 | 3 | 14 | 50 | 100 | 200 | 400 | ||||||
| R3A | 1.3 | 430 | 560 | 860 | + | |||||||||
| R14A | + | |||||||||||||
| R3C | + | |||||||||||||
| R14C | + | |||||||||||||
| R14C50 | + | + | ||||||||||||
| R14C100 | + | + | ||||||||||||
| R14C200 | + | + | ||||||||||||
| R14C400 | + | + | ||||||||||||
| SC3A | 1.3 | 430 | 567 | 860 | + | |||||||||
| SC14A | + | |||||||||||||
| SC3C | + | |||||||||||||
| SC14C | + | |||||||||||||
| SBC3A | 1.3 | 430 | 565.5 | 860 | + | |||||||||
| SBC14A | + | |||||||||||||
| SBC3C | + | |||||||||||||
| SBC14C | + | |||||||||||||
| Sample | Mass Reduction (%) | Residual Mass (%) | |
|---|---|---|---|
| 0–250 °C | 250–500 °C | ||
| R3A | 1.7 | 18.2 | 78.3 |
| R3C | 11.5 | 28.4 | 57.5 |
| R14A | 2.1 | 27.0 | 68.3 |
| R14C | 22.6 | 31.0 | 44.4 |
| SC3A | 2.3 | 26.2 | 69.1 |
| SC3C | 5.4 | 20.4 | 72.0 |
| SC14A | 2.0 | 26.0 | 69.6 |
| SC14C | 11.0 | 26.5 | 60.7 |
| SBC3A | 3.3 | 21.9 | 72.6 |
| SBC3C | 6.8 | 23.9 | 67.2 |
| SBC14A | 3.6 | 22.9 | 71.1 |
| SBC14C | 13.7 | 29.0 | 55.3 |
| R14C50 | 20.6 | 30.7 | 45.8 |
| R14C100 | 10.6 | 32.9 | 46.8 |
| R14C200 | 7.1 | 36.4 | 53.8 |
| R14C400 | 3.7 | 18.2 | 69.7 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aytimur, N.; Ulukaya, S.; Akca, A.H. The Effects of Carbonation and Elevated Temperatures on the Properties of Magnesium-Based Mortar. Appl. Sci. 2025, 15, 9264. https://doi.org/10.3390/app15179264
Aytimur N, Ulukaya S, Akca AH. The Effects of Carbonation and Elevated Temperatures on the Properties of Magnesium-Based Mortar. Applied Sciences. 2025; 15(17):9264. https://doi.org/10.3390/app15179264
Chicago/Turabian StyleAytimur, Neslişah, Serhan Ulukaya, and Abdullah Huzeyfe Akca. 2025. "The Effects of Carbonation and Elevated Temperatures on the Properties of Magnesium-Based Mortar" Applied Sciences 15, no. 17: 9264. https://doi.org/10.3390/app15179264
APA StyleAytimur, N., Ulukaya, S., & Akca, A. H. (2025). The Effects of Carbonation and Elevated Temperatures on the Properties of Magnesium-Based Mortar. Applied Sciences, 15(17), 9264. https://doi.org/10.3390/app15179264





