Numerical Analyses of the Influences of Connector Structures on the Performance of Flat-Tube SOFC
Abstract
1. Introduction
2. The Geometric Modeling and Numerical Methods
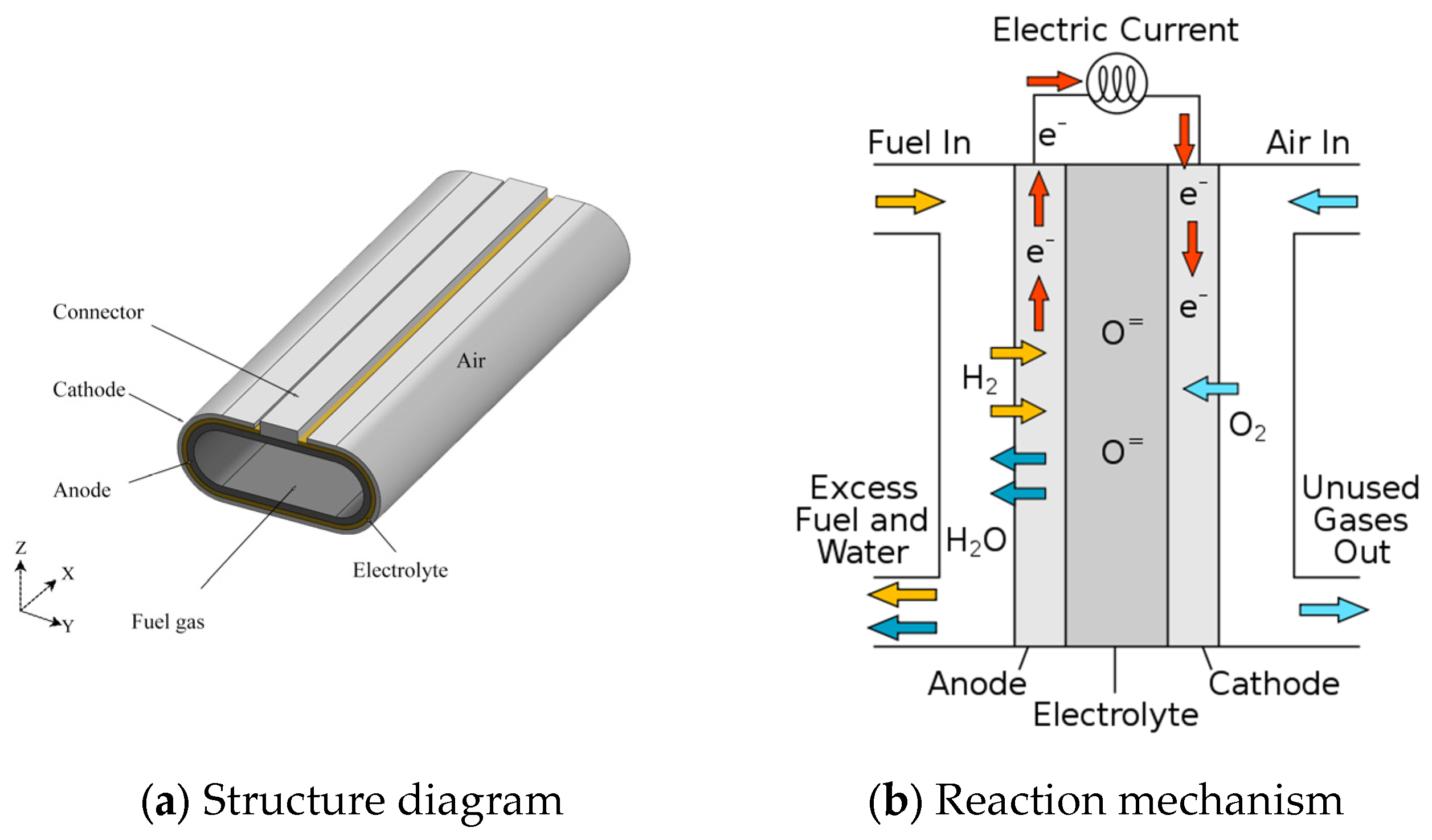
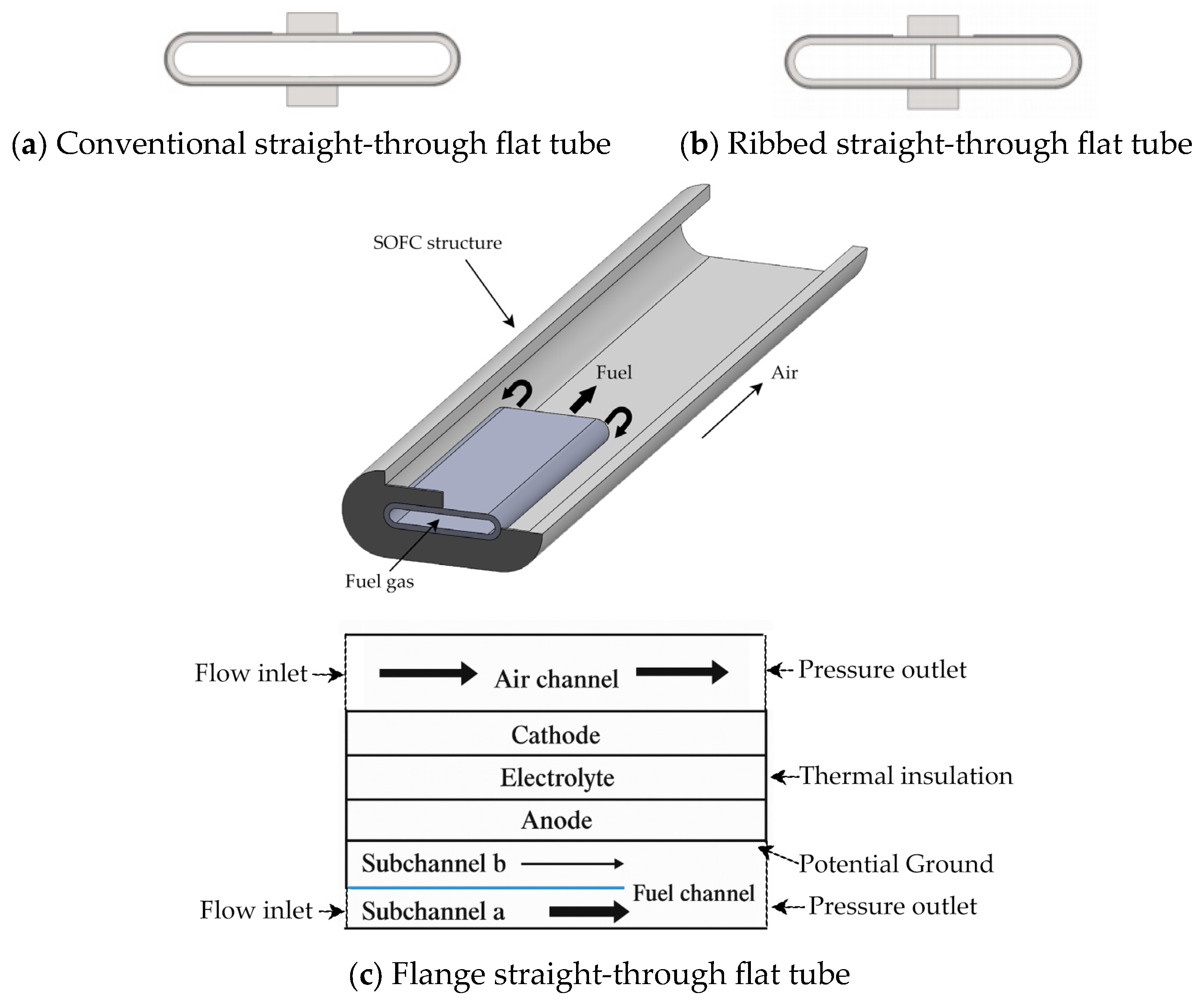
2.1. Geometric Modeling
2.2. The Numerical Methods
2.2.1. Boundary Conditions
2.2.2. Governing Equations [31]
- (1)
- Continuity Equation
- (2)
- Momentum Conservation Equation
- (3)
- Charge Transport Equation
- (4)
- Species Conservation Equation
- (5)
- Energy Equation
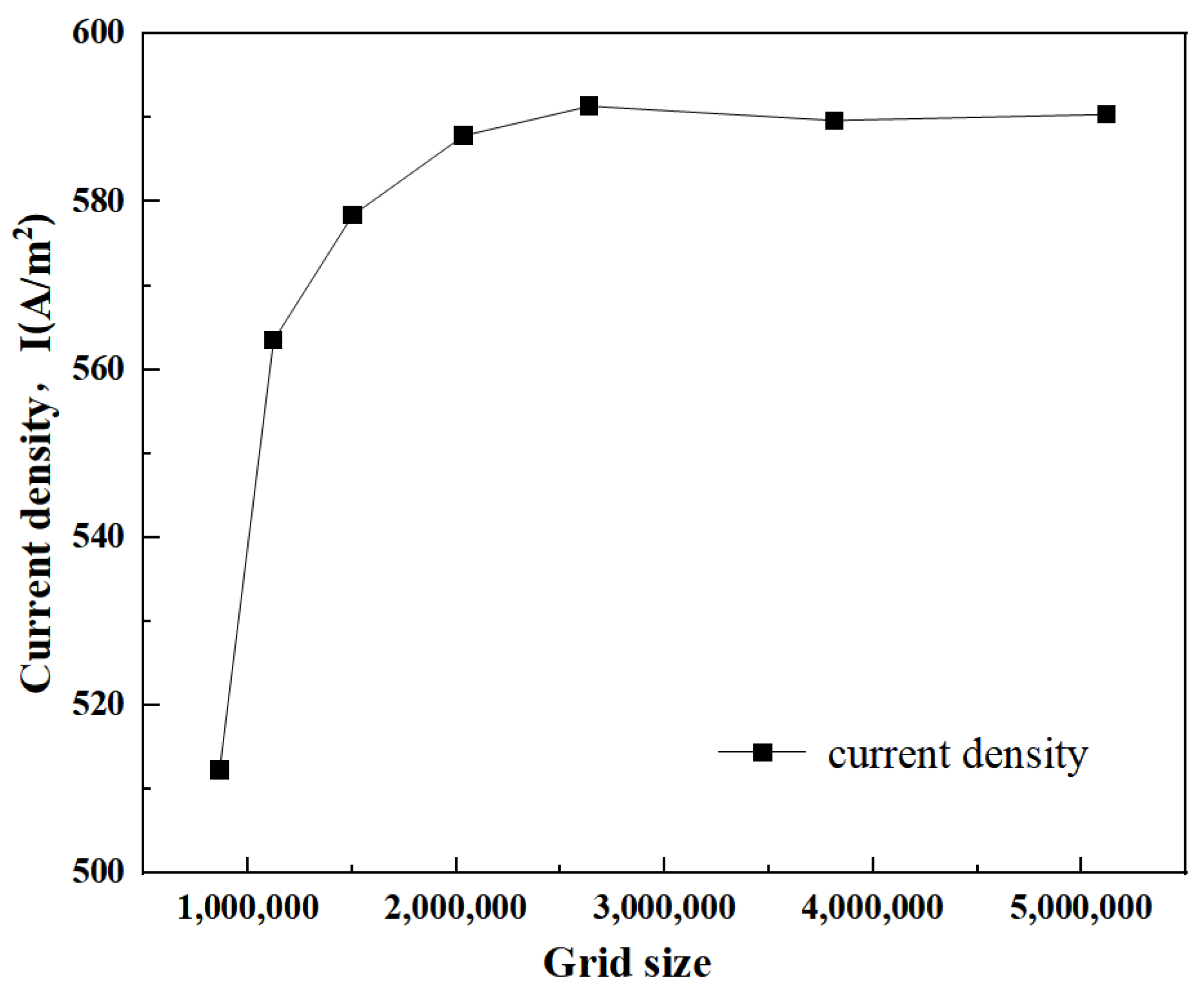
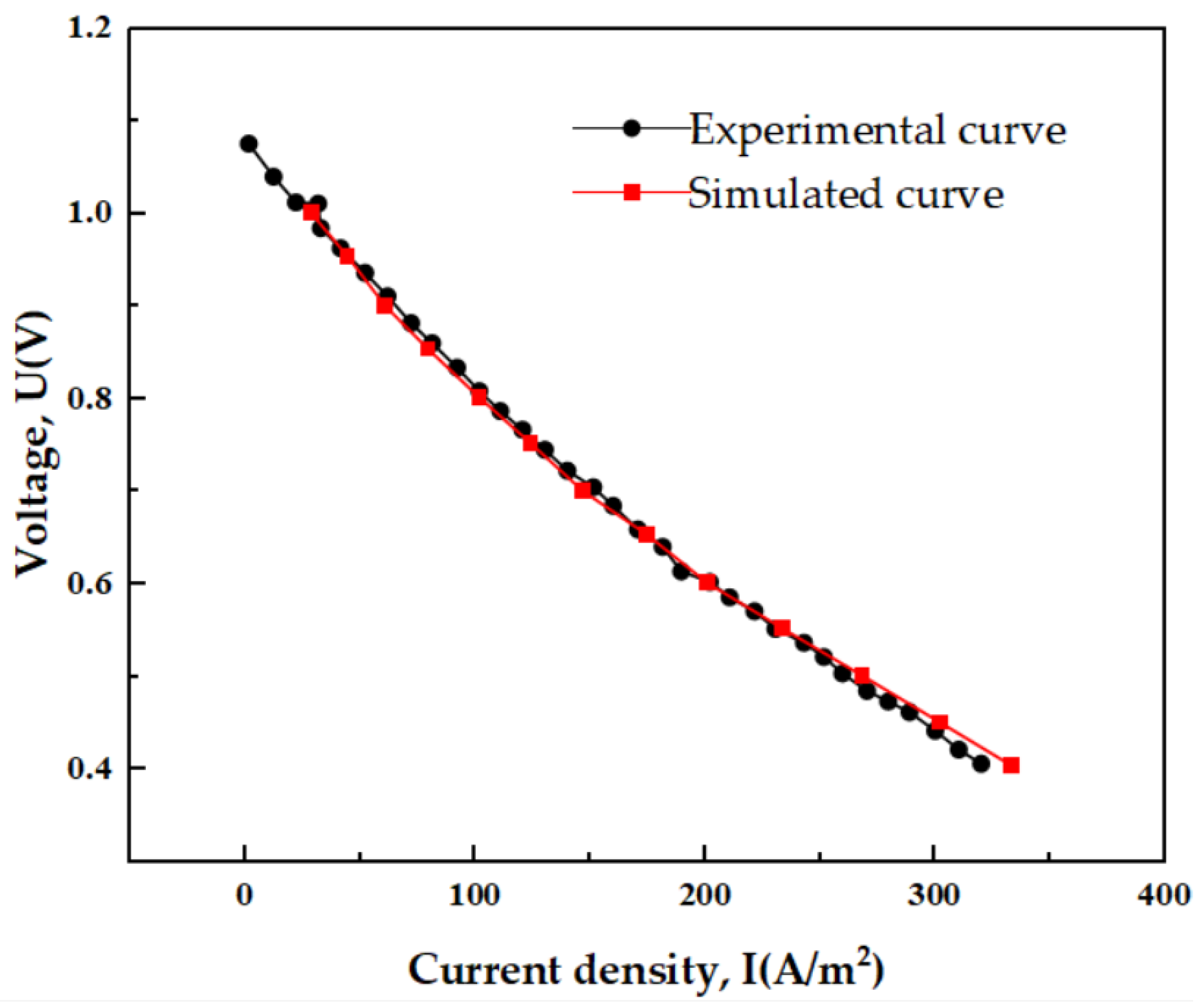
2.2.3. Verification of Grid Independence and Validation of Numerical Methods
3. Results and Discussions
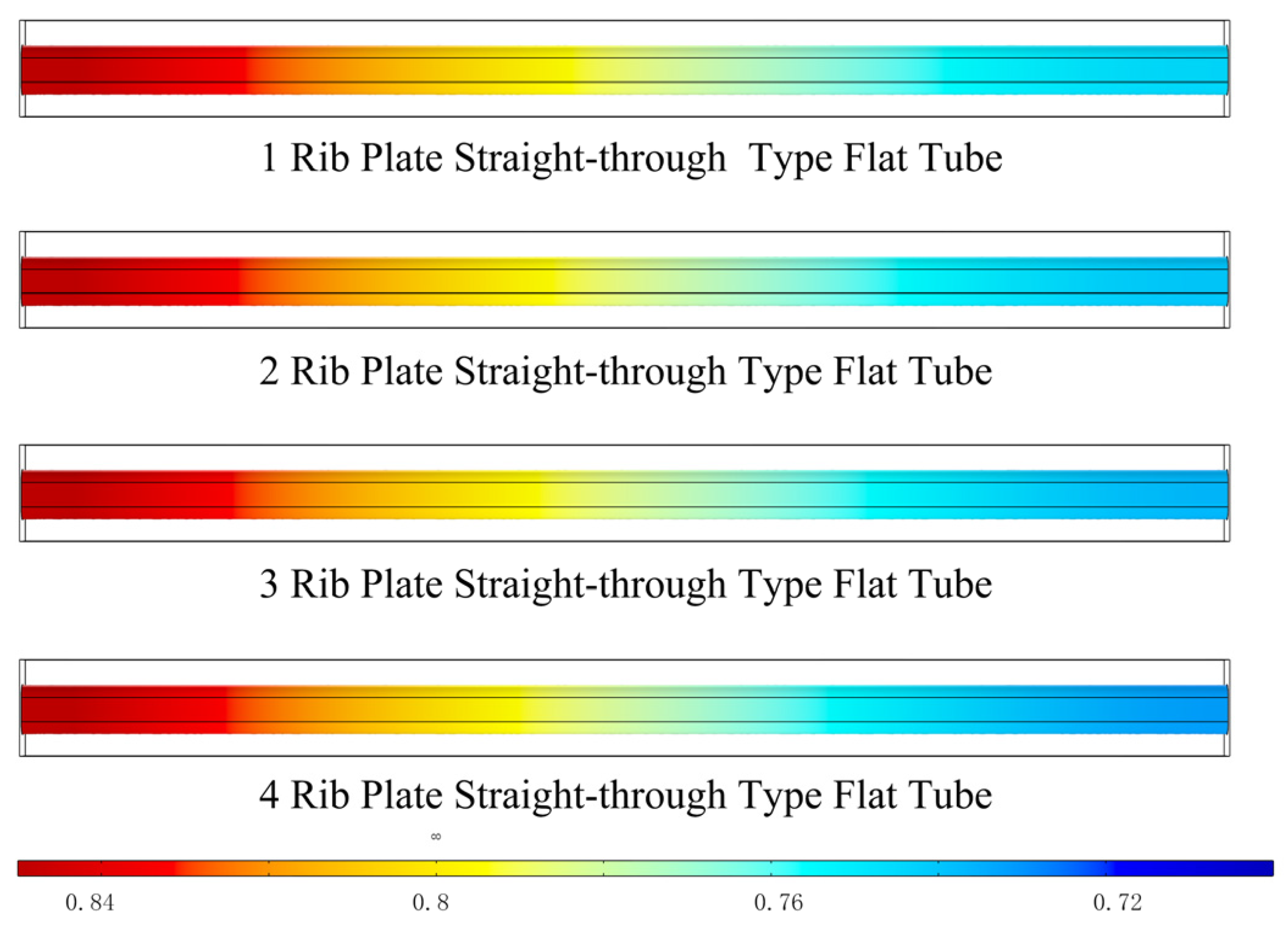
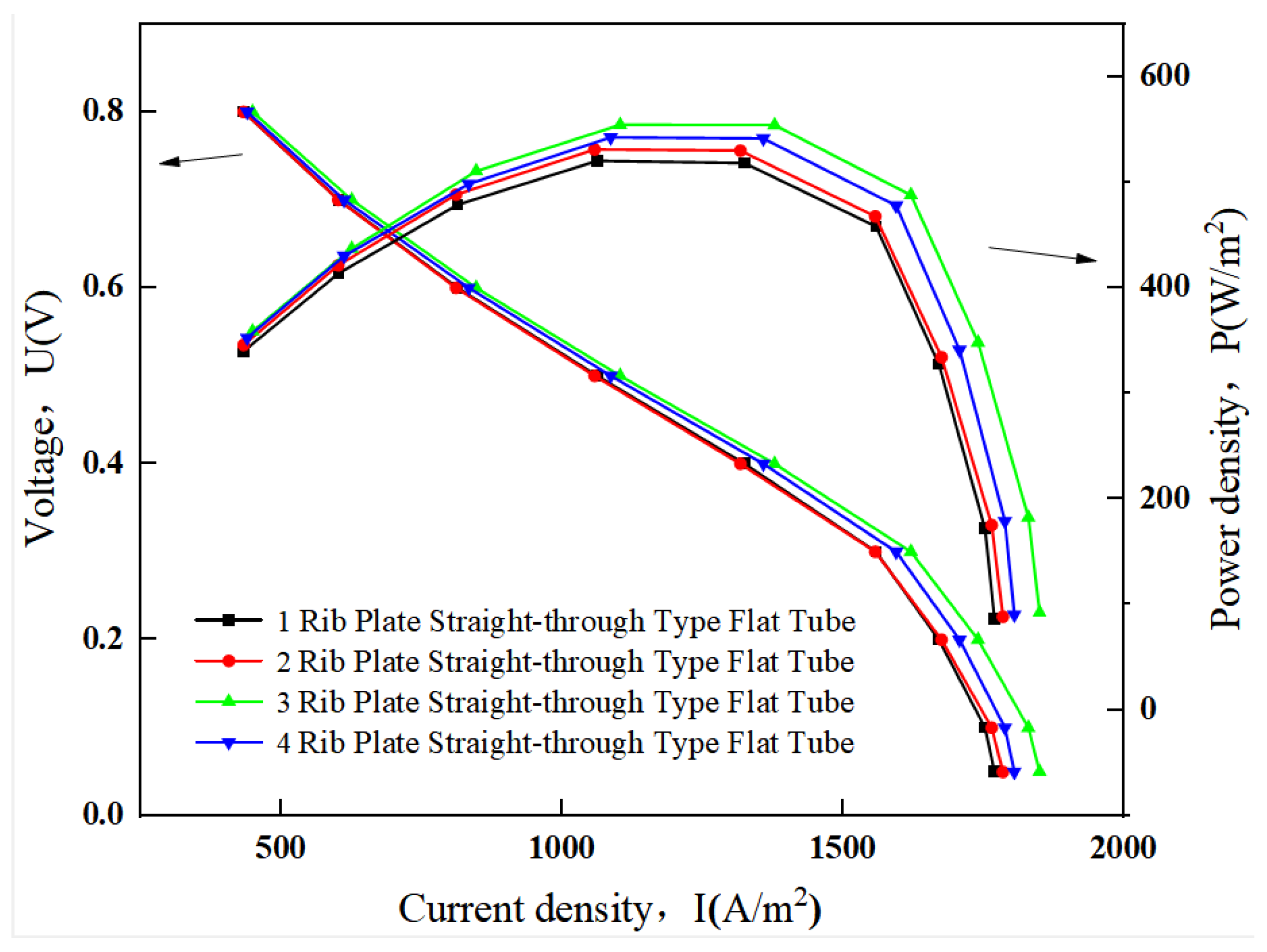
3.1. Effects of the Number of Ribbed Connectors
3.1.1. Distribution of Gas Molar Fractions
3.1.2. SOFC Polarization Curve and Power Density
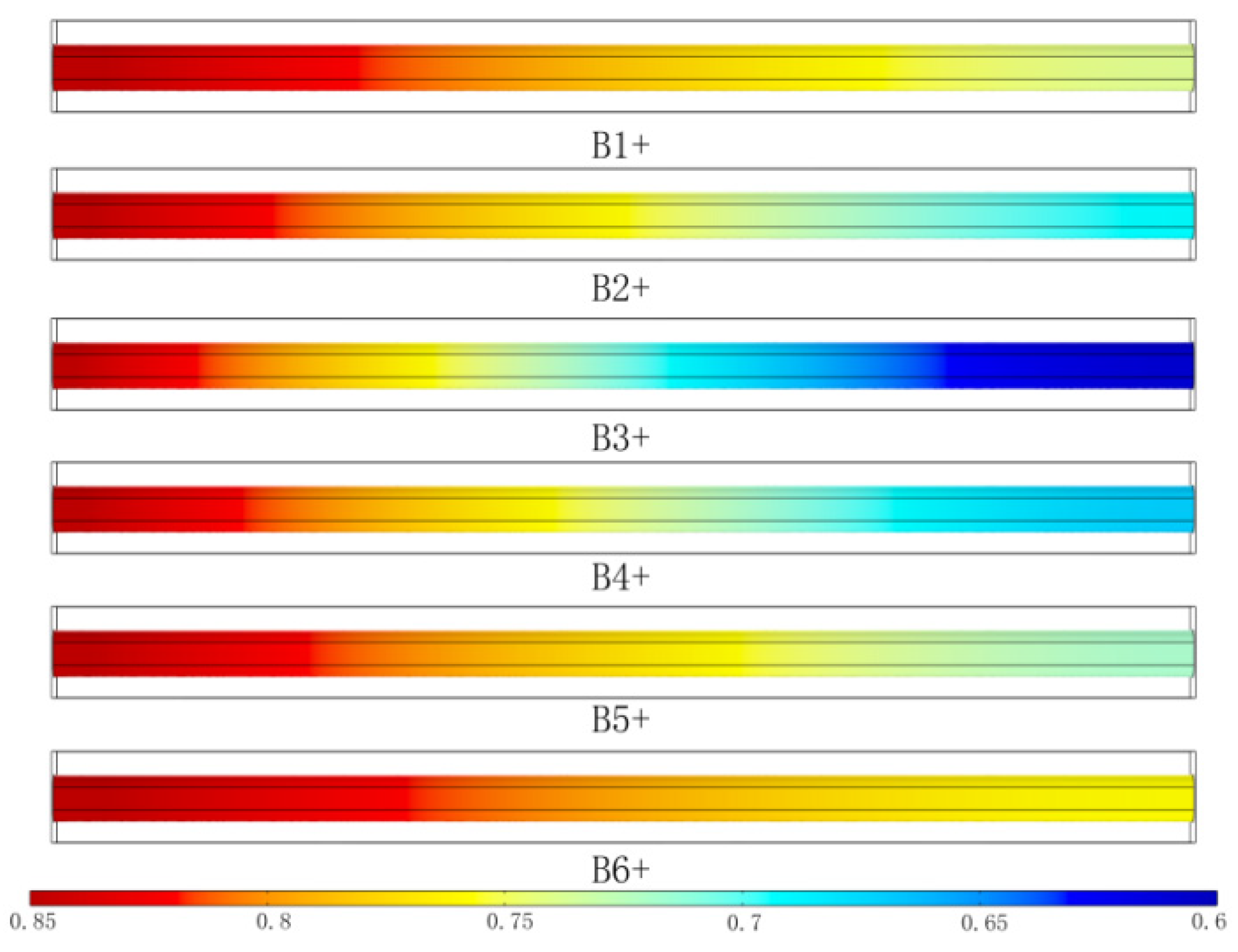
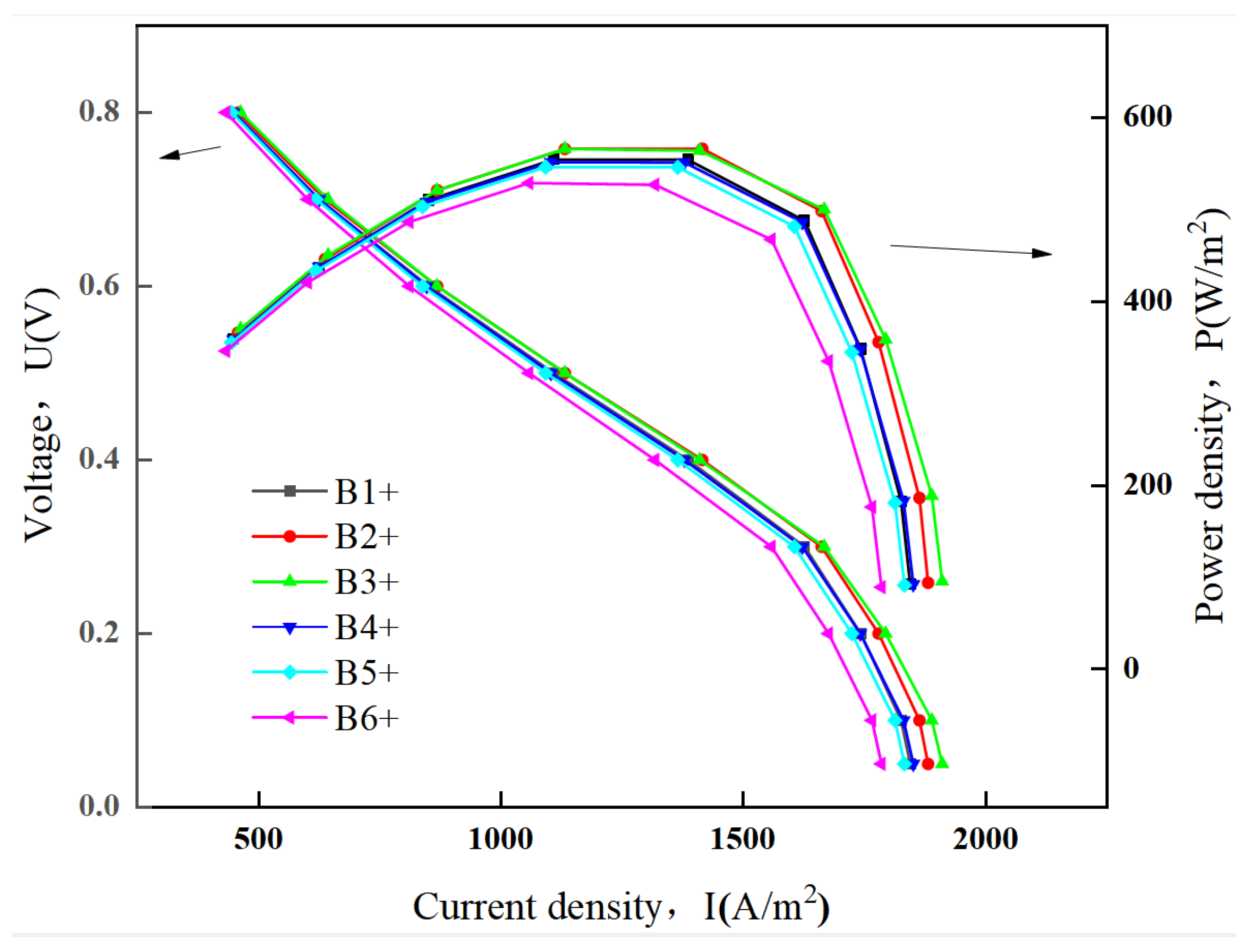
3.2. The Effects of the Ribbed Connector Arrangement Distance
3.2.1. Distribution of Gas Molar Fractions
3.2.2. SOFC Polarization Curve and Power Density
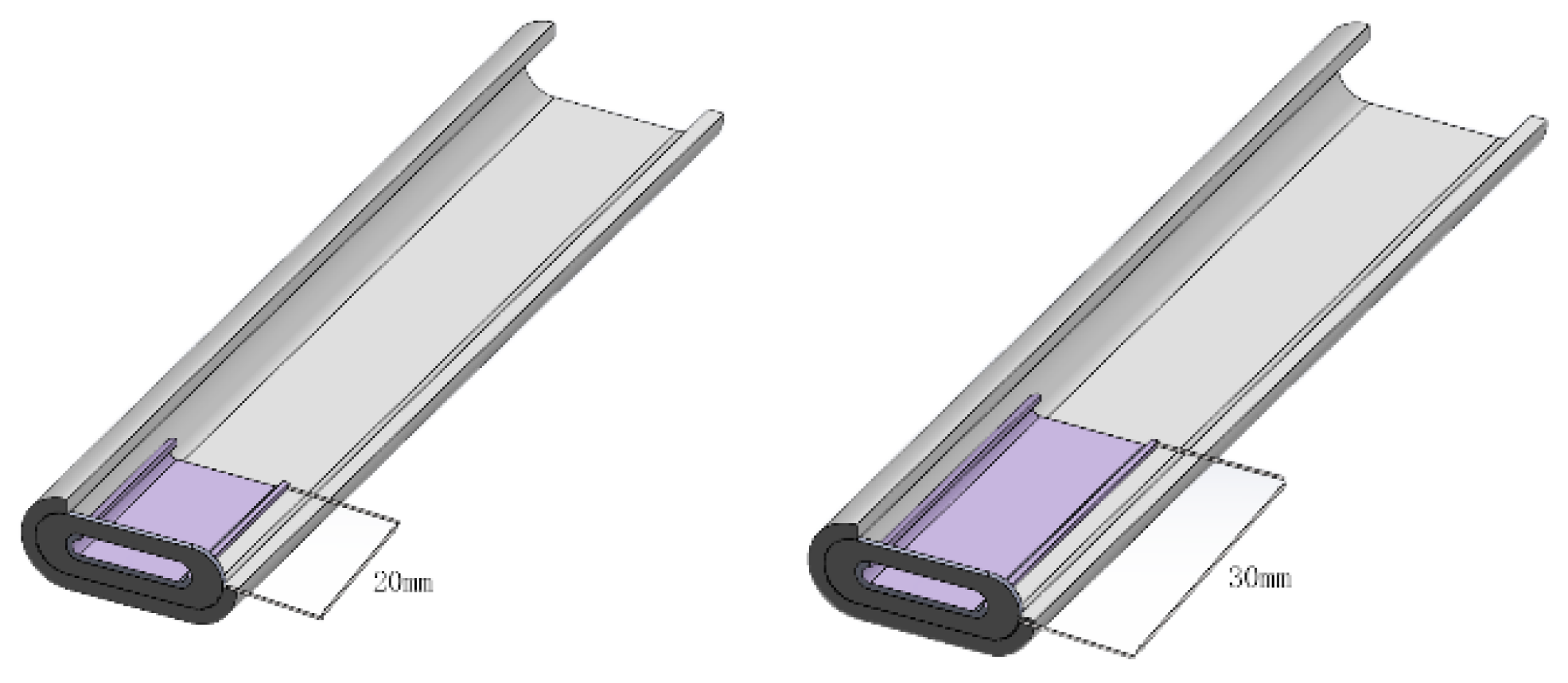
3.3. Effects of Flange-Type Connector Length
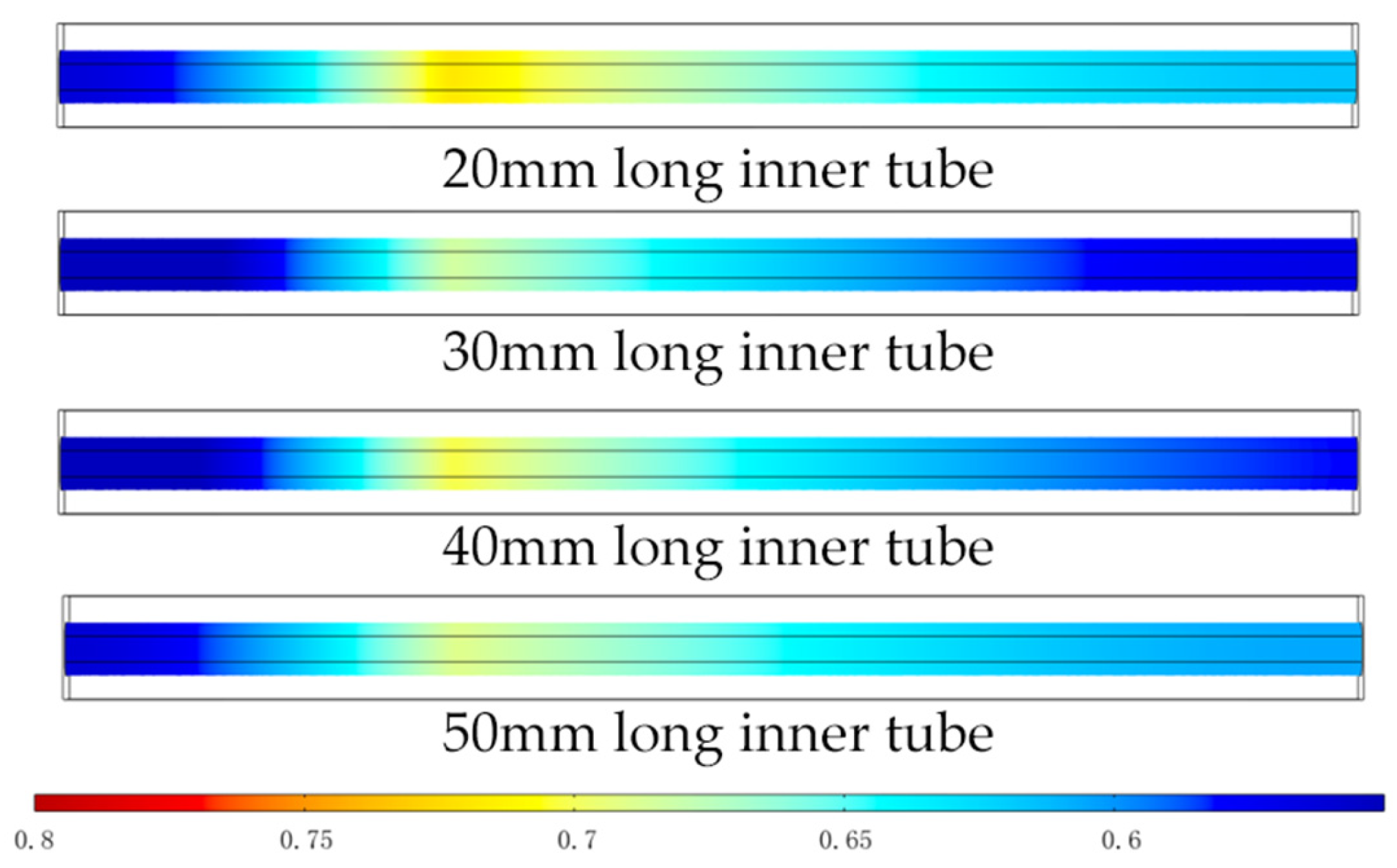
3.3.1. Distribution of Gas Molar Fractions
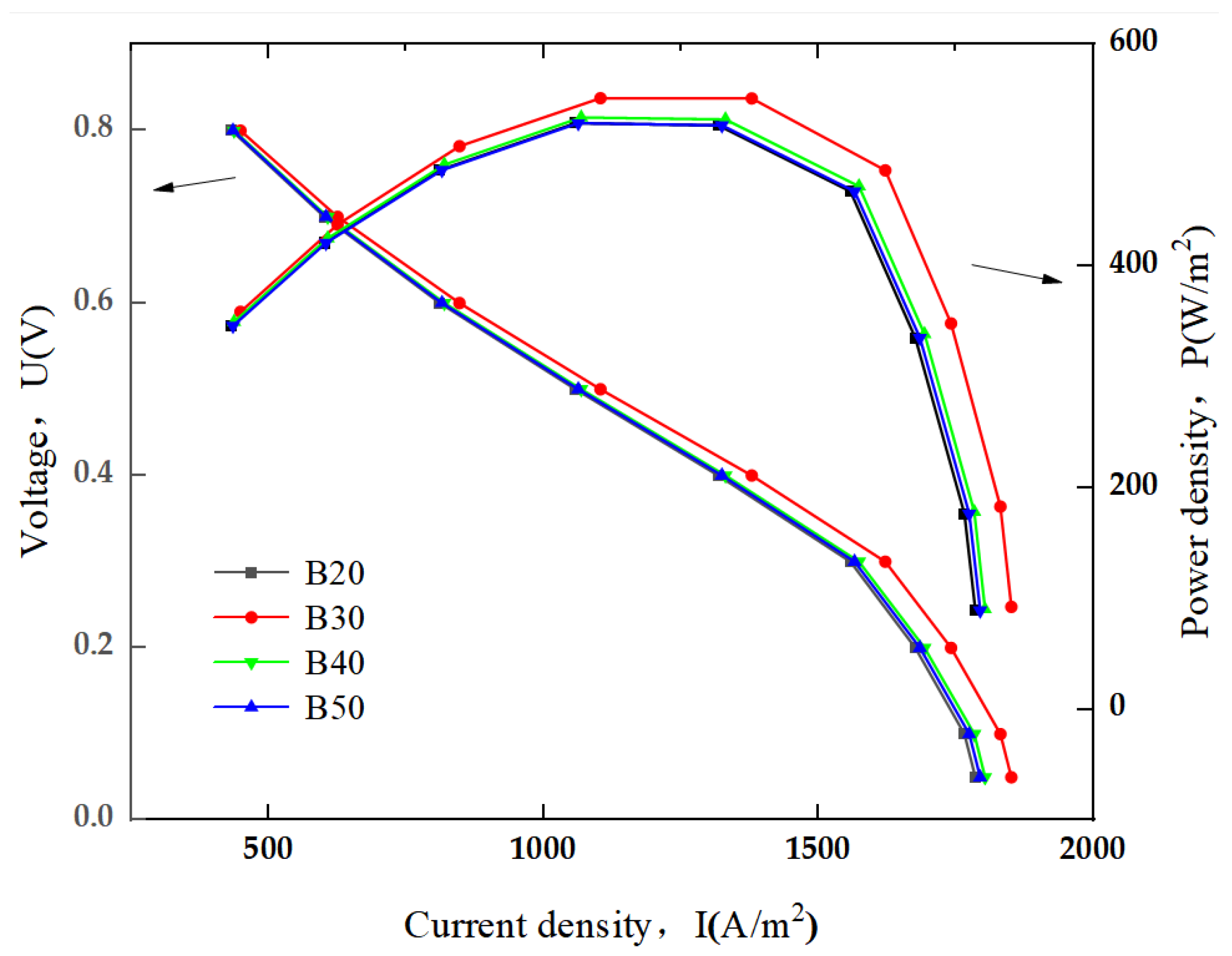
3.3.2. SOFC Polarization Curve and Power Density
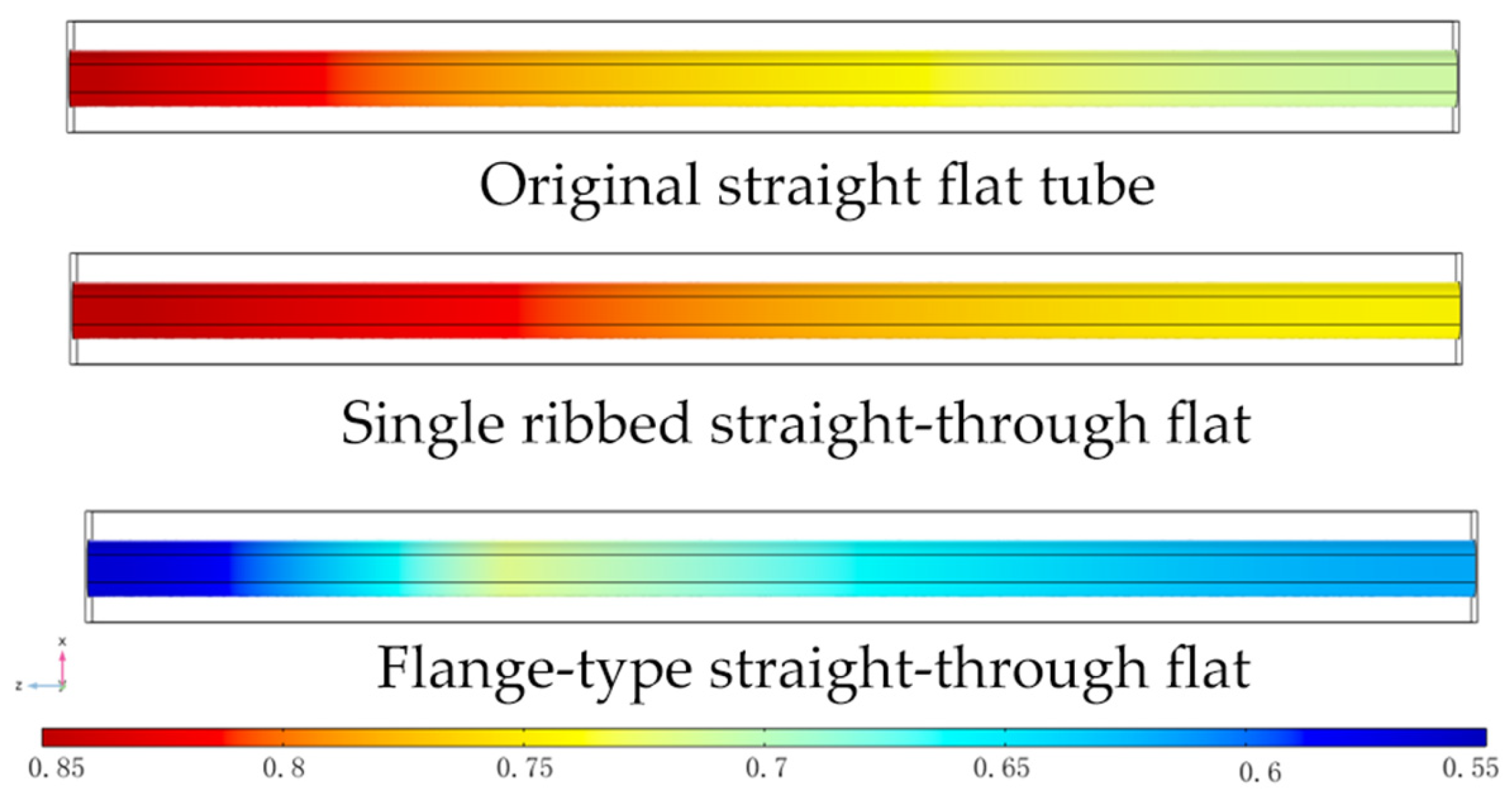
3.4. The Effects of the Presence or Absence of Connectors
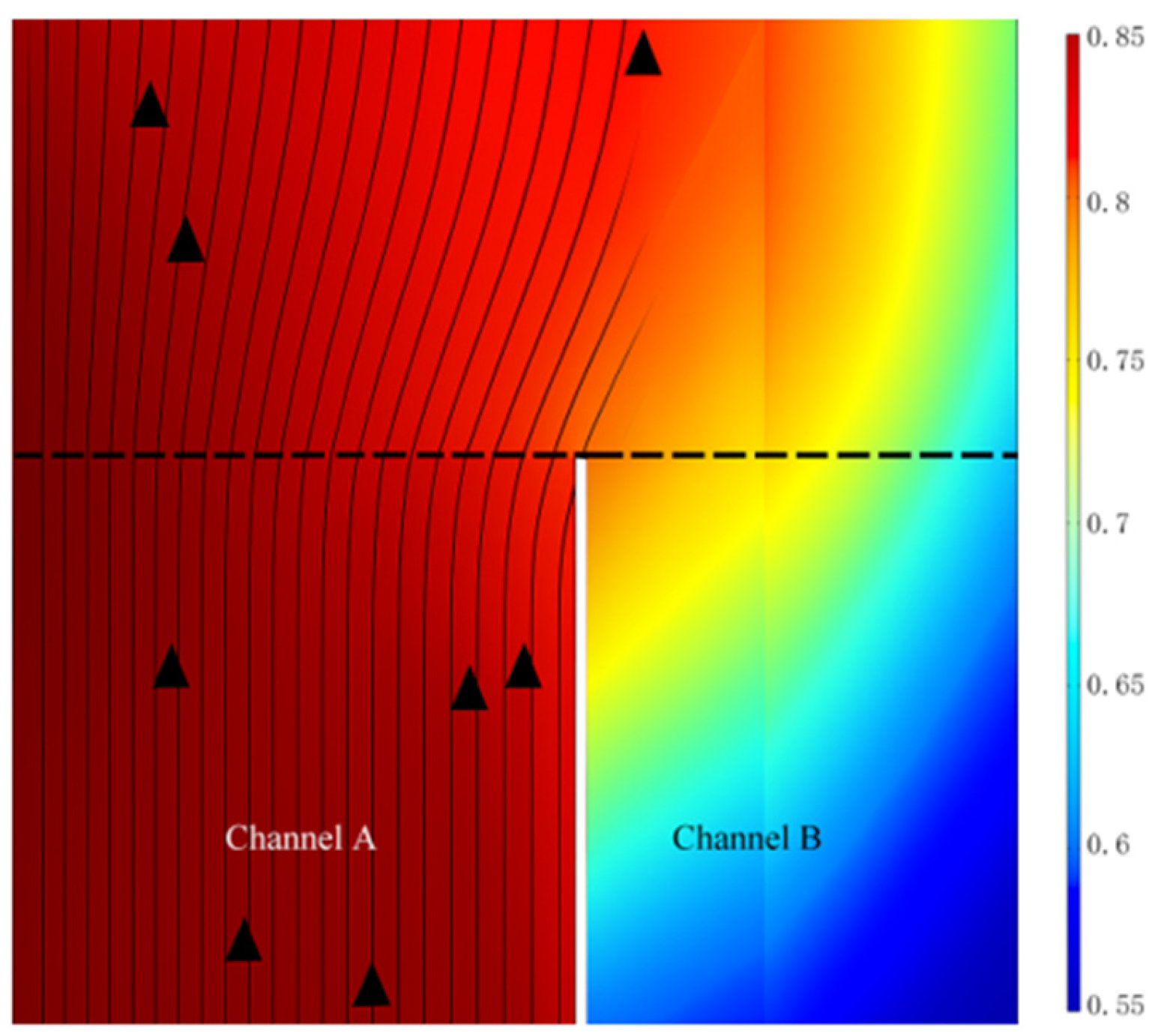
3.4.1. Distribution of Gas Molar Fractions
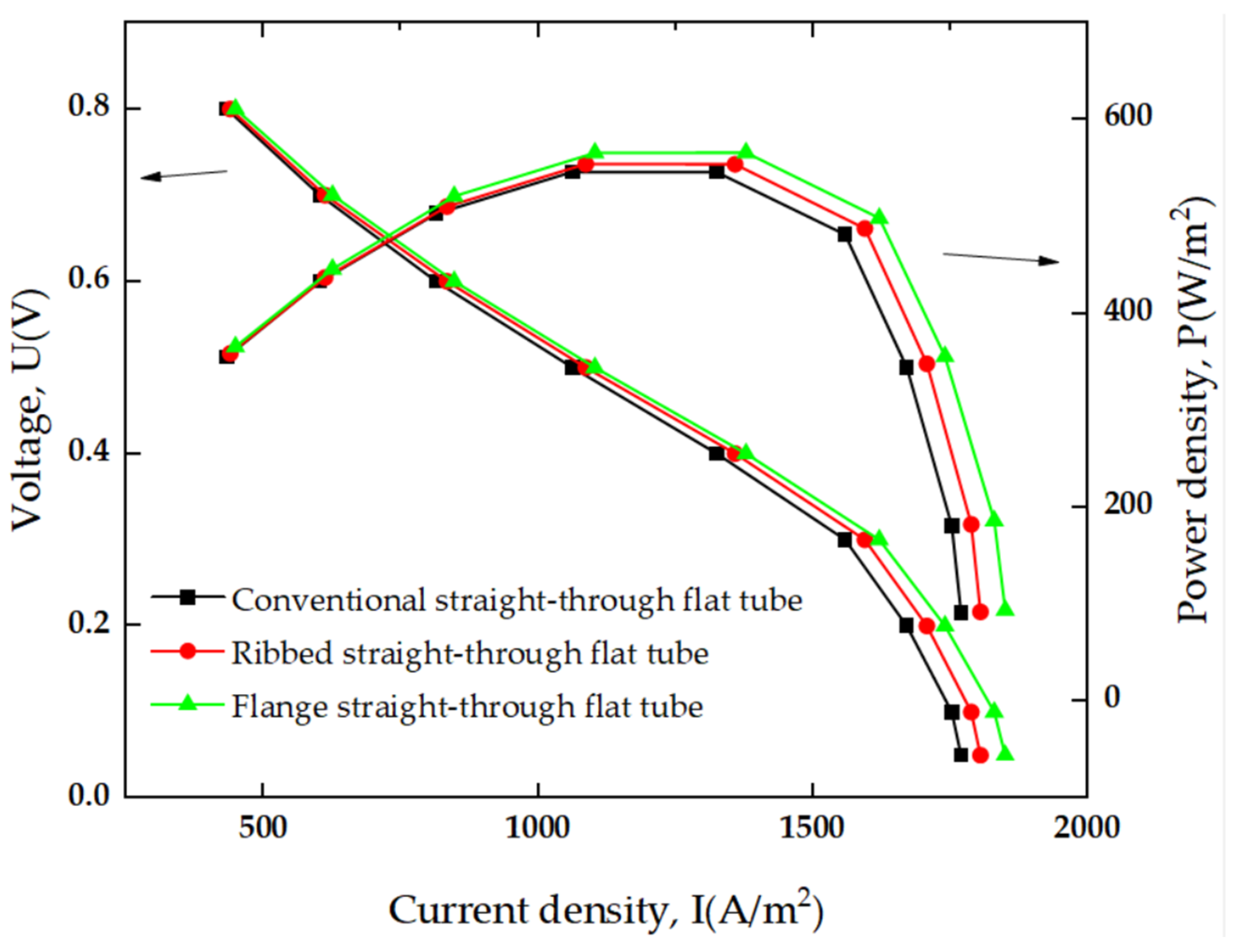
3.4.2. SOFC Polarization Curve and Power Density
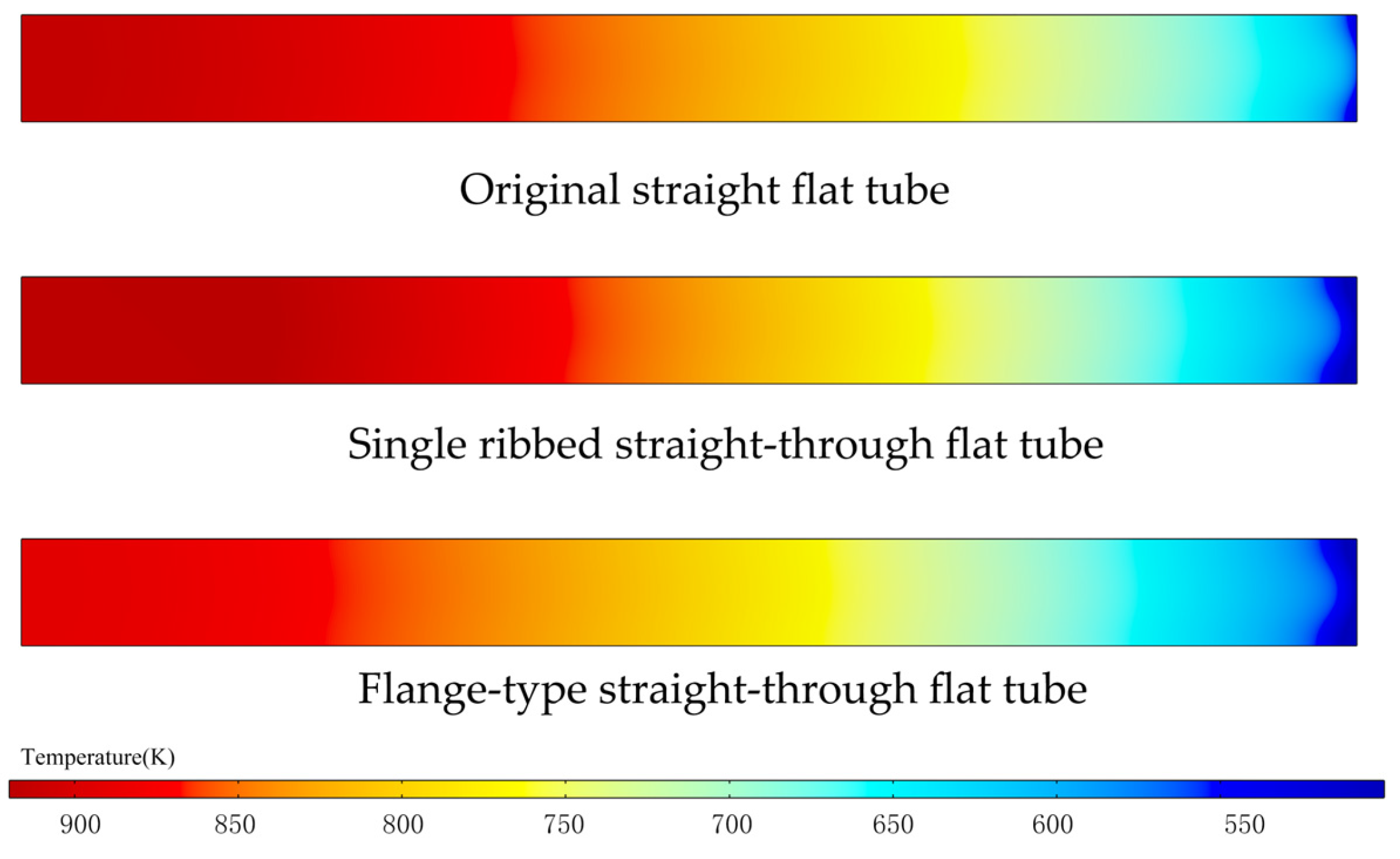
3.4.3. Temperature Distribution of a Single Cell
4. Conclusions
- (1)
- Compared to the conventional and ribbed flat-tube structures, the flange-type connector structure SOFC shows higher current density and power density under the same operating conditions. At a working temperature of 800 K, the flange-type connector structure SOFC demonstrates the highest performance improvement, with the maximum power density increasing by 4.13%.
- (2)
- The temperature of the SOFCs with three connector structures gradually increases along the flow direction. Under certain conditions, the ribbed flat tube has the highest temperature rise, followed by the conventional and flange-type. The flange-type flat tube has the smallest temperature rise due to the larger heat dissipation area in the flow direction, and under constant mass flow rate inlet conditions, the reduced inlet diameter and increased flow velocity within the tube allow heat to be rapidly carried away by the gas, minimizing the temperature increase.
- (3)
- Compared to the conventional straight-through SOFC, the flange-type connector structure has the smallest hydrogen gas unevenness, which enhances the hydrogen transport in the anode layer. It helps achieve a uniform fuel gas distribution and output current density distribution, thereby improving the performance of the fuel cell.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hagen, A.; Langnickel, H.; Sun, X.F. Operation of solid oxide fuel cells with alternative hydrogen carriers. Int. J. Hydrogen Energy 2019, 44, 18382–18392. [Google Scholar] [CrossRef]
- Peng, J.X.; Huang, J.; Wu, X.L.; Xu, Y.W.; Chen, H.C.; Li, X. Solid oxide fuel cell (SOFC) performance evaluation, fault diagnosis and health control: A review. J. Power Sources 2021, 505, 230058. [Google Scholar] [CrossRef]
- Al-Khori, K.; Bicer, Y.; Boulfrad, S.; Koç, M. Techno-economic and environmental assessment of integrating SOFC with a conventional steam and power system in a natural gas processing plant. Int. J. Hydrogen Energy 2019, 44, 29604–29617. [Google Scholar] [CrossRef]
- Lee, S.; Park, M.; Kim, H.; Yoon, K.J.; Son, J.-W.; Lee, J.-H.; Kim, B.-K.; Choi, W.; Hong, J. Thermal conditions and heat transfer characteristics of high-temperature solid oxide fuel cells investigated by three-dimensional numerical simulations. Energy 2017, 120, 293–305. [Google Scholar] [CrossRef]
- Tseronis, K.; Fragkopoulos, I.; Bonis, I.; Theodoropoulos, C. Detailed multi-dimensional modeling of direct internal reforming solid oxide fuel cells. Fuel Cells 2016, 16, 294–312. [Google Scholar] [CrossRef]
- Kong, W.; Gao, X.; Liu, S.; Su, S.; Chen, D. Optimization of the interconnect ribs for a cathode-supported solid oxide fuel cell. Energies 2014, 7, 295–313. [Google Scholar] [CrossRef]
- Zeng, S.M.; Zhang, X.Q.; Chen, J.S.; Li, T.S.; Andersson, M. Modeling of solid oxide fuel cells with optimized interconnect designs. Int. J. Heat Mass Transf. 2018, 125, 506–514. [Google Scholar] [CrossRef]
- Blum, L.; Meulenberg, W.A.; Nabielek, H.; Steinberger-Wilckens, R. Worldwide SOFC technology overview and benchmark. Int. J. Appl. Ceram. Technol. 2005, 2, 482–492. [Google Scholar] [CrossRef]
- Chen, R.Y.; Gao, J.T.; Gao, Y.; Zhang, H.Y.; Li, C.X. Advances in Tubular Solid Oxide Fuel Cell Technology. J. Chin. Ceram. Soc. 2024, 52, 1900–1916. [Google Scholar]
- Zhang, Q.Y.; Yang, Q.G.; Xu, H.T.; Mao, Y.J.; Wang, K.; Liu, S. Simulation study on the influence of different flow field structures on the performance of PEMFC. Acta Energiac Solaris Sin. 2023, 44, 62–67. [Google Scholar]
- Wu, S.W.; Fu, L.R.; Liu, W.F.; Zhang, X.S.; Liu, J.Y.; Gong, P.H. Simulation and performance study of mass transfer in a new expanded flow path PEMFC. Acta Energic Solaris Sin. 2023, 44, 74–79. [Google Scholar]
- Zhan, R.B.; Wang, Y.; Ni, M.; Zhang, G.B.; Du, Q.; Jiao, K. Three-dimensional simulation of solid oxide fuel cell with metal foam as cathode flow distributor. Int. J. Hydrogen Energy 2020, 45, 6897–6911. [Google Scholar] [CrossRef]
- Chen, Q.Y.; Wang, Q.W.; Zhang, J.; Yuan, J.L. Effect of bi-layer interconnector design on mass transfer performance in porous anode of solid oxide fuel cells. Int. J. Heat Mass Transf. 2011, 54, 1994–2003. [Google Scholar] [CrossRef]
- Liu, Y.; Hashimoto, S.I.; Nishino, H.; Takei, K.; Mori, M.; Suzuki, T.; Funahashi, Y. Fabrication and characterization of micro-tubular cathode-supported SOFC for intermediate temperature operation. J. Power Sources 2007, 174, 95–102. [Google Scholar] [CrossRef]
- Yan, M.; Fu, P.; Li, X.; Zeng, M.; Wang, Q. Mass transfer enhancement of a spiral-like interconnector for planar solid oxide fuel cells. Appl. Energy 2015, 160, 954–964. [Google Scholar] [CrossRef]
- Moreno-Blanco, J.; Elizalde-Blancas, F.; Riesco-Avila, J.M.; Belman-Flores, J.M.; Gallegos-Muñoz, A. On the effect of gas channels-electrode interface area on SOFCs performance. Int. J. Hydrogen Energy 2019, 44, 446–456. [Google Scholar] [CrossRef]
- Ergürhan, A.A.; Şenel, O.; Yılmaz, E.; Arpapay, B.; Kulakcı, M.; Serincan, U. Effect of tilt angle and drying temperature on the surface coverage rate of polystyrene nanospheres in evaporation-driven self-assembly process. J. Micro/Nanopatterning Mater. Metrol. 2024, 23, 013001. [Google Scholar] [CrossRef]
- Yang, N.T.; Shen, Y.C.; Yan, W.; Meng, X.X.; Tan, X.Y.; Ma, Z.F. Microstructure and Performance of Anode in Micro Tube Solid Oxide Fuel Cells. J. Inorg. Mater. 2014, 29, 1246–1252. [Google Scholar]
- Lin, Z.; Stevenson, J.W.; Khaleel, M.A. The effect of interconnect rib size on the fuel cell concentration polarization in planar SOFCs. J. Power Sources 2003, 117, 92–97. [Google Scholar] [CrossRef]
- Wang, G.L.; Yang, Y.Z.; Zhang, H.O. Three dimensional thermal electrochemical analysis of solid oxide fuel cells. CSEE J. Power Energy Syst. 2007, 27, 99–103. [Google Scholar]
- Canavar, M.; Timurkutluk, B. Design and fabrication of novel anode flow-field for commercial size solid oxide fuel cells. J. Power Sources 2017, 346, 49–55. [Google Scholar] [CrossRef]
- Schluckner, C.; Subotić, V.; Preißl, S.; Hochenauer, C. Numerical analysis of flow configurations and electrical contact positions in SOFC single cells and their impact on local effects. Int. J. Hydrogen Energy 2019, 44, 1877–1895. [Google Scholar] [CrossRef]
- Li, Z.H.; Guo, X.Y. Numerical analysis of the influence of connector structure on the performance of solid oxide fuel cells. Energy Res. Inf. 2023, 39, 128–135. [Google Scholar]
- Qiao, Y.X.; Fan, C.; Sun, K.N. Exploration of the configuration of anode current collecting ribs in flat tube fuel cells. Chin. J. Power Sources 2023, 47, 79–82. [Google Scholar]
- Zhao, F.; Virkar, A.V. Dependence of polarization in anode-supported solid oxide fuel cells on various cell parameters. J. Power Sources 2005, 141, 79–95. [Google Scholar] [CrossRef]
- Mustafa, F.C.; Mustafa, I.; Senay, Y. Effects of physical properties and operating parameters on numerically developed flat-tube solid oxide fuel cell performance. Int. J. Hydrogen Energy 2023, 48, 23136–23145. [Google Scholar]
- Jia, J.X.; Shen, S.Q.; Liu, X.H.; Abdullah, A. Analysis of Ohmic Polarization of Single Tube Solid Oxide Fuel Cells. Acta Energic Solaris Sin. 2004, 25, 457–461. [Google Scholar]
- Wu, Q.Q. Research on Micro Tube Solid Oxide Fuel Cells Using Methane as Fuel. Master’s Thesis, Nanjing University, Nanjing, China, 2017. [Google Scholar]
- Shi, Y.X.; Li, C.; Cai, N.S. Mechanism Model and Performance Analysis of Tubular Solid Oxide Fuel Cells. CIESC J. 2007, 58, 6. [Google Scholar]
- Liu, H.Y.; Ji, X.Y.; Zhou, X.; Yang, Y. Study on the Influence of Fuel Gas Parameters on the Discharge Characteristics of Solid Oxide Fuel Cells. Hunan Electr. Power 2023, 43, 31–36. [Google Scholar]
- COMSOL, Inc., COMSOL Multiphysics Reference Manual. Available online: https://doc.comsol.com/6.3/docserver/#!/com.comsol.help.comsol/html_COMSOL_ReferenceManual.html (accessed on 10 August 2025).
- Tonekabonimoghadam, S.; Akikur, R.; Hussain, M.; Hajimolana, S.; Saidur, R.; Ping, H.; Chakrabarti, M.; Brandon, N.; Aravind, P.; Nayagar, J. Mathematical modelling and experimental validation of an anode-supported tubular solid oxide fuel cell for heat and power generation. Energy 2015, 90, 1759–1768. [Google Scholar] [CrossRef]


| SOFC Component Parameters/Units | Value |
|---|---|
| Cathode thickness/mm | 0.05 |
| Anode thickness/mm | 0.56 |
| Electrolyte thickness/mm | 0.01 |
| Flow channel length/mm | 100.00 |
| Parameter Name/Unit | Value |
|---|---|
| Pressure/Pa | 101,325 |
| Voltage/V | 0.8 |
| Cathode density/(kg·m−3) | 8750 |
| Anode density/(kg·m−3) | 8640 |
| Electrolyte density/(kg·m−3) | 2000 |
| Connector density/(kg·m−3) | 8700 |
| Cathode specific heat capacity/[J·(kg·K)−1] | 385 |
| Anode specific heat capacity/[J·(kg·K)−1] | 380 |
| Electrolyte specific heat capacity/[J·(kg·K)−1] | 300 |
| Connector specific heat capacity/[J·(kg·K)−1] | 396 |
| Connection Type | Number of Cases | Usage |
|---|---|---|
| Traditional straight flat tube | 1 | To use as a base case to compare with the other cases with structural modification. |
| Ribbed straight-through flat tube | 4 | To calculate the flat tube models with 1, 2, 3, and 4 ribs, respectively, and to investigate the influence of multiple ribs on the performance of a single cell. |
| 6 | To calculate the flat tube models with six different rib distances to find out the influence of the rib distance on the performance of a single cell. | |
| Flange straight-through flat tube | 4 | To calculate the models of flange-type connectors with various inner tube length of 20 mm, 30 mm, 40 mm, and 50 mm, and to investigate the influence of the inner tube length on the performance of a single cell. |
| Parameter Name/Unit | Value |
|---|---|
| Flat tube length/mm | 25 |
| Flat tube width/mm | 6 |
| The radius of a semicircle/mm | 3 |
| Rib width/mm | 0.5 |
| Distance between the ribs/mm | 4 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yao, J.; Guo, X. Numerical Analyses of the Influences of Connector Structures on the Performance of Flat-Tube SOFC. Appl. Sci. 2025, 15, 9251. https://doi.org/10.3390/app15179251
Yao J, Guo X. Numerical Analyses of the Influences of Connector Structures on the Performance of Flat-Tube SOFC. Applied Sciences. 2025; 15(17):9251. https://doi.org/10.3390/app15179251
Chicago/Turabian StyleYao, Jian, and Xueyan Guo. 2025. "Numerical Analyses of the Influences of Connector Structures on the Performance of Flat-Tube SOFC" Applied Sciences 15, no. 17: 9251. https://doi.org/10.3390/app15179251
APA StyleYao, J., & Guo, X. (2025). Numerical Analyses of the Influences of Connector Structures on the Performance of Flat-Tube SOFC. Applied Sciences, 15(17), 9251. https://doi.org/10.3390/app15179251





