Acorns as a Source of Valuable Compounds for Food and Medical Applications: A Review of Quercus Species Diversity and Laboratory Studies
Abstract
1. Introduction
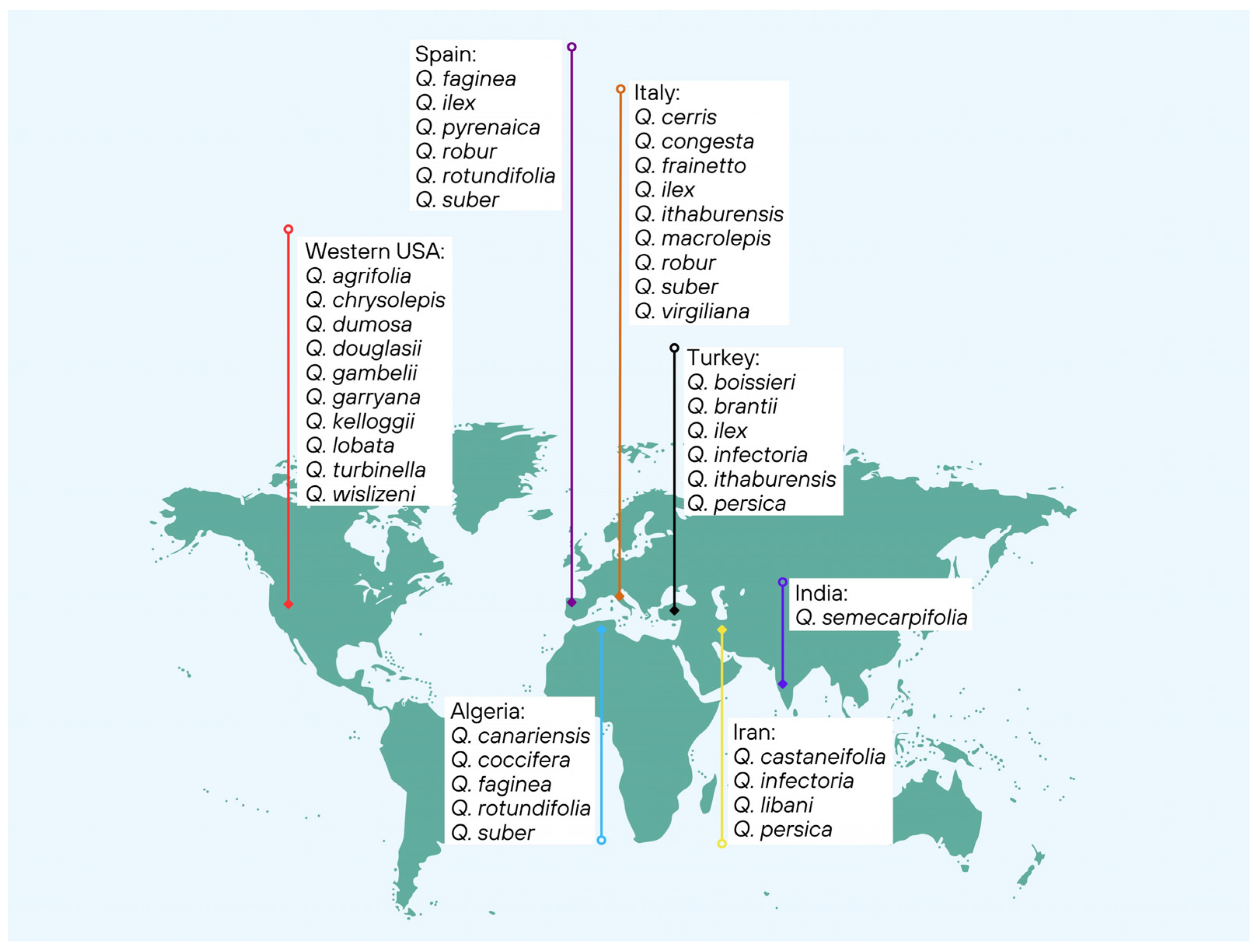
2. Origin and Occurrence of Acorns
3. Impact of Quercus Species Diversity on Acorn Morphological and Chemical Characteristics

| Quercus Species | Chemical Components | References | ||||
|---|---|---|---|---|---|---|
| Fat | Protein | Carbohydrates | Starch | Ash | ||
| Q. brantii | 0.72 | 3.37 | n.d. | 63.00 | 2.09 | [44] |
| Q. calliprinos | 2.31 | 4.94 | 77.86 | n.d. | 1.78 | [45] |
| Q. cerris | 1.05 | 4.3 | n.d. | 64.55 | 1.75 | [44] |
| Q. coccifera | 2.67 | 2.54 | n.d. | 66.2 | 1.81 | [44] |
| Q. ilex | 9.14–14.95 | 3.90–5.94 | 8.95–12.47 * | n.d. | 1.34–2.02 | [46] |
| Q. infectoria | 1.55 | 4.37 | n.d. | 68.15 | 1.95 | [44] |
| Q. ithaburensis | 0.76 | 2.84 | 58.94 | n.d. | 3.21 | [45] |
| Q. rotundifolia | 8.38–13.51 | 3.56–4.34 | n.d. | 47.98–59.95 | 1.94–2.19 | [42] |
| Q. suber | 2.09–8.22 | 6.19–9.38 | n.d. | 22.03–60.00 | 2.12–2.98 | [42,44] |
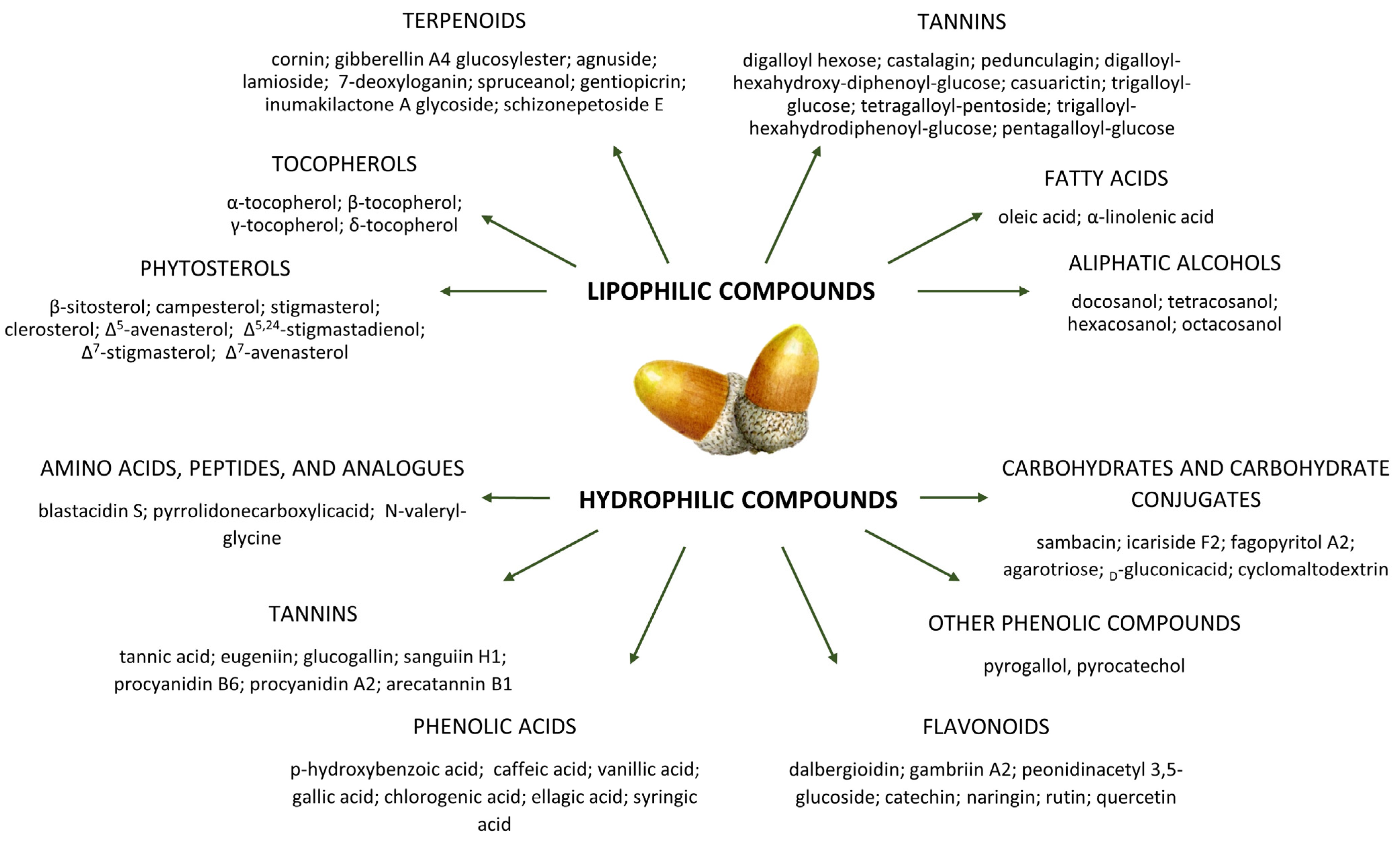
4. Bioactive Compounds in Acorns
| Quercus Species | Chemical Group | Predominant Compound | Content of Predominant Compound [mg/100 g d.m.] | References |
|---|---|---|---|---|
| Q. coccifera | tocopherols | γ-tocopherol | 4.51 | [7,49] |
| sterols * | β-sitosterol | 93.77 | ||
| Q. faginea | tocopherols | γ-tocopherol | 4.03 | [7,14] |
| carotenoids | β-carotene | 131.2 | ||
| lycopene | 18.3 | |||
| chlorophylls | chlorophyll a | 400 | ||
| Q. ilex | tocopherols | γ-tocopherol | 5.22 | [7,14] |
| carotenoids | β-carotene | 47.3 | ||
| lycopene | 14.8 | |||
| chlorophylls | chlorophyll a | 200 | ||
| sterols | β-sitosterol | 91.31 * | ||
| Q. pyreneica | tocopherols | γ-tocopherol | 3.63 | [7] |
| Q. robur | tocopherols | γ-tocopherol | 18.86–42.30 | [74] |
| Q. rotundifolia | tocopherols | γ-tocopherol | 5.75–10.79 | [48] |
| Q. rubra | tocopherols | β-tocopherol | 13.26–22.37 | [74] |
| Q. suber | tocopherols | γ-tocopherol | 3.80 | [7,14] |
| carotenoids | β-carotene | 130.1 | ||
| lycopene | 7.9 | |||
| chlorophylls | chlorophyll a | 300 |
5. Controversial Role of Acorn Tannins
6. Historical and Potential Modern Uses of Acorns in Food
7. Historical and Potential Modern Uses of Acorns in Medicine
8. Other Possibilities for Using Acorns
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Karimian, M.; Najafi, R.; Jaimand, K.; Hatami, F.; Abbasi, N.; Jalali Ghalousangh, A. Extraction and identification of phytochemicals in Iranian oak (Quercus brantii var. persica) collected in Arghavan Valley, Ilam County by HS-SPME and GC-MS. J. Med. Plants By-Prod. 2020, 9, 81–86. [Google Scholar]
- Migaskó, H.; Ecseri, K. Significance of Quercus species in nutrition. Gradus 2020, 7, 6–11. [Google Scholar] [CrossRef]
- Pinna, C. Acorn bread: A traditional food of the past in Sardinia (Italy). J. Cult. Herit. 2013, 14, S71–S74. [Google Scholar]
- Zocchi, D.M.; Bondioli, C.; Hamzeh Hosseini, S.; Miara, M.D.; Musarella, C.M.; Mohammadi, D.; Manduzai, A.K.; Issa, K.D.; Sulaiman, N.; Khatib, C.; et al. Food Security beyond Cereals: A cross-geographical comparative study on acorn bread heritage in the Medi-terranean and the Middle East. Foods 2022, 11, 3898. [Google Scholar] [CrossRef] [PubMed]
- Śmiałek, E.; Gutkowska, K. Kuchnia molekularna i jej możliwości rozwoju w Polsce®. Postępy Tech. Przetw. Spoż. 2017, 1, 89–96. [Google Scholar]
- Vinha, A.F.; Barreira, J.C.; Costa, A.S.; Oliveira, M.B.P. A new age for Quercus spp. fruits: Review on nutritional and phytochemical composition and related biological activities of acorns. Compr. Rev. Food Sci. Food Saf. 2016, 15, 947–981. [Google Scholar] [CrossRef] [PubMed]
- Akcan, T.; Gökçe, R.; Asensio, M.; Estévez, M.; Morcuende, D. Acorn (Quercus spp.) as a novel source of oleic acid and tocopherols for livestock and humans: Discrimination of selected species from Mediterranean forest. J. Food Sci. Technol. 2017, 54, 3050–3057. [Google Scholar] [CrossRef] [PubMed]
- Papoti, W.T.; Kizaki, N.; Skaltsi, A.; Karayannakidis, P.D.; Papageorgiou, M. The phytochemical rich potential of acorn (Quercus aegilops) products and by products. Food Sci. Biotechnol. 2018, 27, 819–828. [Google Scholar] [CrossRef]
- Lei, Y.; Huang, Y.; Wang, Y.; He, X. Potential anti-neuroinflammatory triterpenoids isolated from Chinese acorns (Quercus serrata var. brevipetiolata). J. Funct. Foods 2018, 50, 18–25. [Google Scholar] [CrossRef]
- Burlacu, E.; Nisca, A.; Tanase, C. A comprehensive review of phytochemistry and biological activities of Quercus species. Forests 2020, 11, 904. [Google Scholar] [CrossRef]
- Szabłowska, E.; Tańska, M. Acorn flour properties depending on the production method and laboratory baking test results: A review. Compr. Rev. Food Sci. Food Saf. 2021, 20, 980–1008. [Google Scholar] [CrossRef] [PubMed]
- Makhlouf, F.; Squeo, G.; Barkat, M.; Trani, A.; Caponio, F. Antioxidant activity, tocopherols and polyphenols of acorn oil obtained from Quercus species grown in Algeria. Food Res. Int. 2018, 114, 208–213. [Google Scholar] [CrossRef] [PubMed]
- Taib, M.; Bouyazza, L.; Lyoussi, B. Acorn oil: Chemistry and functionality. J. Food Qual. 2020, 2020, 8898370. [Google Scholar] [CrossRef]
- Vinha, A.F.; Costa, A.S.G.; Barreira, J.C.; Pacheco, R.; Oliveira, M.B.P. Chemical and antioxidant profiles of acorn tissues from Quercus spp.: Potential as new industrial raw materials. Ind. Crops Prod. 2016, 94, 143–151. [Google Scholar] [CrossRef]
- Eaton, E.G.S.D.J.; Caudullo, G.; Oliveira, S.; De Rigo, D. Quercus robur and Quercus petraea in Europe: Distribution, habitat, usage and threats. In European Atlas of Forest Tree Species; San-Miguel-Ayanz, J., de Rigo, D., Caudullo, G., Eds.; Publications Office of the European Union: Luxembourg, 2016; pp. 160–163. [Google Scholar]
- Tantray, Y.R.; Wani, M.S.; Hussain, A. Genus Quercus: An overview. Int. J. Adv. Eng. Res. Sci. 2017, 6, 1880–1886. [Google Scholar]
- Ballesteros, D.; Pritchard, H.W. The cryobiotechnology of oaks: An integration of approaches for the long-term ex situ conservation of Quercus species. Forests 2020, 11, 1281. [Google Scholar] [CrossRef]
- Sacchelli, S.; Cavuta, T.; Borghi, C.; Cipollaro, M.; Fratini, R.; Bernetti, I. Financial analysis of acorns chain for food production. Forests 2021, 12, 784. [Google Scholar] [CrossRef]
- Denk, T.; Grimm, G.W.; Manos, P.S.; Deng, M.; Hipp, A.L. An updated infrageneric classification of the oaks: Review of previous taxonomic schemes and synthesis of evolutionary patterns. In Oaks Physiological Ecology. Exploring the Functional Diversity of Genus Quercus L. Tree Physiology; Gil-Pelegrín, E., Peguero-Pina, J., Sancho-Knapik, D., Eds.; Springer: Cham, Switzerland, 2017; pp. 13–38. [Google Scholar]
- Wang, Y.; Xu, C.; Wang, Q.; Jiang, Y.; Qin, L. Germplasm resources of oaks (Quercus L.) in China: Utilization and prospects. Biology 2023, 12, 76. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Zhou, T.; Duan, D.; Yang, J.; Feng, L.; Zhao, G. Comparative analysis of the complete chloroplast genomes of five Quercus species. Front. Plant Sci. 2016, 7, 959. [Google Scholar] [CrossRef]
- Saha, S.; Kuehne, C.; Bauhus, J. Lessons learned from oak cluster planting trials in central Europe. Can. J. For. Res. 2017, 47, 139–148. [Google Scholar] [CrossRef]
- Forest Data Bank. Available online: https://www.bdl.lasy.gov.pl/portal/lasy-na-swiecie (accessed on 4 September 2023).
- Szewczyk, W.; Kuzminski, R. Rola czynników środowiskowych w zamieraniu drzewostanów dębowych. In Zarządzanie Ochroną Przyrody w Lasach; Konnenberg, K., Szramka, H., Eds.; Wyższa Szkoła Zarządzania Środowiskiem w Tucholi: Tuchola, Poland, 2009. [Google Scholar]
- Pourhashemi, M.; Panahi, P.; Zandebasiri, M. Application of visual surveys to estimate acorn production of Brants oak (Quercus brantii Lindl.) in northern Zagros Forests of Iran. Casp. J. Environ. Sci. 2013, 11, 85–95. [Google Scholar]
- Akli, A.; Khellaf, R.; Alvarez, E.T.; Alia, R.; Kheloul, L.; Messaoudene, M. Biometry of acorn of the putative oak from Akfadou Forest in North Africa (Algeria). Forestist 2022, 72, 212–224. [Google Scholar] [CrossRef]
- Chirino, E.; Vilagrosa, A.; Hernández, E.I.; Matos, A.; Vallejo, V.R. Effects of a deep container on morpho-functional characteristics and root colonization in Quercus suber L. seedlings for reforestation in Mediterranean climate. For. Ecol. Manag. 2008, 256, 779–785. [Google Scholar] [CrossRef]
- Pantera, A.; Papadopoulos, A.; Papanastasis, V.P. Valonia oak agroforestry systems in Greece: An overview. Agrofor. Syst. 2018, 92, 921–931. [Google Scholar] [CrossRef]
- Rodríguez-Estévez, V.; Sánchez-Rodríguez, M.; Arce, C.R.; García, A.M.; Perea, J.M.; Gustavo Gómez-Castro, A.G. Consumption of acorns by finishing Iberian pigs and their function in the conservation of the dehesa agroecosystem. In Agroforestry for Biodiversity and Ecosystem Services; Kaonga, M.L., Ed.; IntechOpen: London, UK, 2012. [Google Scholar]
- Stavi, I.; Thevs, N.; Welp, M.; Zdruli, P. Provisioning ecosystem services related with oak (Quercus) systems: A review of challenges and opportunities. Agrofor. Syst. 2012, 96, 293–313. [Google Scholar] [CrossRef]
- Villar-Salvador, P.; Planelles, R.; Enrıquez, E.; Rubira, J.P. Nursery cultivation regimes, plant functional attributes, and field performance relationships in the Mediterranean oak Quercus ilex L. For. Ecol. Manag. 2004, 196, 257–266. [Google Scholar] [CrossRef]
- Koenig, W.D.; Knops, J.M. Environmental correlates of acorn production by four species of Minnesota oaks. Popul. Ecol. 2014, 56, 63–71. [Google Scholar] [CrossRef]
- O’Reilly-Wapstra, J.M.; Miller, A.M.; Hamilton, M.G.; Williams, D.; Glancy-Dean, N.; Potts, B.M. Chemical variation in a dominant tree species: Population divergence, selection and genetic stability across environments. PLoS ONE 2013, 8, e58416. [Google Scholar] [CrossRef]
- Gea-Izquierdo, G.; Cañellas, I.; Montero, G. Acorn production in Spanish holm oak woodlands. For. Syst. 2006, 15, 339–354. [Google Scholar] [CrossRef]
- Wikipedia. Available online: www.wikipedia.pl (accessed on 12 January 2024).
- Oaks of the World. Available online: http://oaks.of.the.world.free.fr/ (accessed on 12 January 2024).
- European Forest Genetic Resources Programme. Available online: https://www.euforgen.org/species/ (accessed on 12 January 2024).
- Flora of Cyprus—A Dynamic Checklist. Available online: https://flora-of-cyprus.eu/ (accessed on 12 January 2024).
- Oak Name Checklist. Available online: http://www.oaknames.org/ (accessed on 12 January 2024).
- My Mediterranean Garden. Available online: https://mymediterraneangarden.com/ (accessed on 12 January 2024).
- Cal Photos. Available online: https://calphotos.berkeley.edu/ (accessed on 12 January 2024).
- Ferraz de Oliveira, M.I.F.; Machado, M.G.; D’Abreu, M.C. Acorn chemical composition depending on shedding date and Quercus species. Options Méditer. A Sém. 2012, 101, 229–234. [Google Scholar]
- Taib, M.; Bouyazza, L. Composition, physicochemical properties, and uses of Acorn starch. J. Chem. 2021, 2021, 9988570. [Google Scholar] [CrossRef]
- Kaya, E.; Kamalak, A. Potential nutritive value and condensed tannin contents of acorns from different oak species. Kafkas Univ. Vet. Fak. Derg. 2012, 18, 1061–1066. [Google Scholar] [CrossRef][Green Version]
- Rababah, T.M.; Ereifej, K.I.; Al-Mahasneh, M.A.; Alhamad, M.N.; Alrababah, M.A.; Muhammad, A.H. The physicochemical composition of acorns for two Mediterranean Quercus species. Jordan J. Agric. Sci. 2008, 4, 131–137. [Google Scholar]
- Valero Galván, J.; Jorrin Novo, J.J.; Cabrera, A.G.; Ariza, D.; García-Olmo, J.; Cerrillo, R.M.N. Population variability based on the morphometry and chemical composition of the acorn in Holm oak (Quercus ilex subsp. ballota [Desf.] Samp.). Eur. J. For. Res. 2012, 131, 893–904. [Google Scholar] [CrossRef]
- Charef, M.; Yousfi, M.; Saidi, M.; Stocker, P. Determination of the fatty acid composition of acorn (Quercus), Pistacia lentiscus seeds growing in Algeria. J. Am. Oil. Chem. Soc. 2008, 85, 921–924. [Google Scholar] [CrossRef]
- Tejerina, D.; García-Torres, S.; de Vaca, M.C.; Vázquez, F.M.; Cava, R. Acorns (Quercus rotundifolia Lam.) and grass as natural sources of antioxidants and fatty acids in the “montanera” feeding of Iberian pig: Intra-and inter-annual variations. Food Chem. 2011, 124, 997–1004. [Google Scholar] [CrossRef]
- Lassoued, R.; Abderrabba, M.; Mejri, J. Comparative chemical composition of two Quercus species seeds growing in Tunisia. S. Afr. J. Bot. 2022, 146, 71–76. [Google Scholar] [CrossRef]
- Özcan, T. Characterization of Turkish Quercus L. taxa based on fatty acid compositions of the acorns. J. Am. Oil. Chem. Soc. 2007, 84, 653–662. [Google Scholar] [CrossRef]
- Özcan, T.; Bayçu, G. Some elemental concentrations in the acorns of Turkish Quercus L. (Fagaceae) taxa. Pak. J. Bot. 2005, 37, 361–371. [Google Scholar]
- Nikolić, N.Č.; Orlovic, S.; Krstic, B.; Kevrešan, Ž. Variability of acorn nutrient concentrations in pedunculate oak (Quercus robur L.) genotypes. J. For. Sci. 2006, 52, 51–60. [Google Scholar] [CrossRef]
- Ahmed, M.; Adil, M.; Haq, I.; Tipu, M.K.; Qasim, M.; Gul, B. RP-HPLC-based phytochemical analysis and diverse pharmacological evaluation of Quercus floribunda Lindl. Ex A. camus nuts extracts. Nat. Prod. Res. 2019, 35, 2257–2262. [Google Scholar] [CrossRef] [PubMed]
- López-Hidalgo, C.; Menéndez, M.; Jorrin-Novo, J.V. Phytochemical composition and variability in Quercus ilex acorn morphotypes as determined by NIRS and MS-based approaches. Food Chem. 2021, 338, 127803. [Google Scholar] [CrossRef] [PubMed]
- Vinha, A.F.; Barreira, J.C.; Ferreira, I.C.; Oliveira, M.B.P. Therapeutic, phytochemistry, and pharmacology of acorns (Quercus nuts): A review. In Bioactive Compounds in Underutilized Fruits and Nuts; Murthy, H.N., Bapat, V.A., Eds.; Springer: Cham, Switzerland, 2020; pp. 273–287. [Google Scholar]
- Kheirandish, F.; Delfan, B.; Mahmoudvand, H.; Moradi, N.; Ezatpour, B.; Ebrahimzadeh, F.; Rashidipour, M. Antileishmanial, antioxidant, and cytotoxic activities of Quercus infectoria Olivier extract. Biomed. Pharmacother. 2016, 82, 208–215. [Google Scholar] [CrossRef] [PubMed]
- Tayel, A.A.; El-Sedfy, M.A.; Ibrahim, A.I.; Moussa, S.H. Application of Quercus infectoria extract as a natural antimicrobial agent for chicken egg decontamination. Rev. Argent. Microbiol. 2018, 50, 391–397. [Google Scholar] [CrossRef] [PubMed]
- Chokpaisarn, J.; Urao, N.; Voravuthikunchai, S.; Koh, T. Quercus infectoria inhibits Set7/NF-ΚB inflammatory pathway in macrophages exposed to a diabetic environment. Cytokine 2017, 94, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Abdullah, A.R.; Hapidin, H.; Abdullah, H. The role of semipurified fractions isolated from Quercus infectoria on bone metabolism by using HFOB 1.19 human fetal osteoblast cell model. Evid. Based Complement. Alternat. Med. 2018, 2018, 5319528. [Google Scholar] [CrossRef] [PubMed]
- Sati, A.; Sati, S.C.; Sati, N.; Sati, O.P. Chemical composition and antimicrobial activity of fatty acid methyl ester of Quercus leucotrichophora fruits. Nat. Prod. Res. 2017, 31, 713–717. [Google Scholar] [CrossRef]
- Yin, P.; Wang, Y.; Yang, L.; Sui, J.; Liu, Y. Hypoglycemic effects in alloxan-induced diabetic rats of the phenolic extract from mongolian oak cups enriched in ellagic acid, kaempferol and their derivatives. Molecules 2018, 23, 1046. [Google Scholar] [CrossRef] [PubMed]
- Zhou, D.; Liu, Z.-H.; Wang, D.-M.; Li, D.-W.; Yang, L.-N.; Wang, W. Chemical composition, antibacterial activity and related mechanism of valonia and shell from Quercus variabilis blume (Fagaceae) against Salmonella paratyphi a and Staphylococcus aureus. BMC Complement. Altern. Med. 2019, 19, 271. [Google Scholar] [CrossRef]
- Setti, C.; Bouras, A.D.; Meziane, M.; Bouderoua, K.; Setti, H. Antioxidant and antibacterial activities of green oak acorn (Quercus ilex) from Algeria. S. Asian J. Biol. Sci. 2020, 10, 285–291. [Google Scholar] [CrossRef]
- Rakić, S.; Petrowić, S.; Kukić, J.; Jadranin, M.; Tešewić, V.; Povrenović, D.; Šiler-Marinković, S. Influence of thermal treatment on phenolic compounds and antioxidant properties of oak acorns from Serbia. Food Chem. 2007, 104, 830–834. [Google Scholar] [CrossRef]
- Bursal, E.; Boğa, R. Polyphenols analysed by UHPLC-ESI-MS/MS and antioxidant activities of molasses, acorn and leaves of oak (Quercus robur subsp. pedunculiflora). Prog. Nutr. 2018, 20, 167–175. [Google Scholar]
- Cantos, E.; Espín, J.C.; López-Bote, C.; de la Hoz, L.; Ordóñez, J.A.; Tomás-Barberán, F.A. Phenolic compounds and fatty acids from acorns (Quercus spp.), the main dietary constituent of free-ranged Iberian pigs. J. Agric. Food Chem. 2003, 51, 6248–6255. [Google Scholar] [CrossRef]
- Mezni, F.; Stiti, B.; Fkiri, S.; Ayari, F.; Slimane, L.B.; Ksouri, R.; Khaldi, A. Phenolic profile and in vitro anti-diabetic activity of acorn from four African Quercus species (Q. suber, Q. canariensis, Q. coccifera and Q. ilex). S. Afr. J. Bot. 2022, 146, 771–775. [Google Scholar] [CrossRef]
- Molina-García, L.; Martínez-Expósito, R.; Fernández-de Córdova, M.L.; Llorent-Martínez, E.J. Determination of the phenolic profile and antioxidant activity of leaves and fruits of Spanish Quercus coccifera. J. Chem. 2018, 2018, 2573270. [Google Scholar] [CrossRef]
- Rakić, S.; Povrenović, D.; Tešewić, V.; Simić, M.; Maletić, R. Oak acorn, polyphenols and antioxidants activity in functional food. J. Food Eng. 2006, 74, 416–423. [Google Scholar] [CrossRef]
- Pleszczynska, M.; Szczodrak, J. Taniny i ich rozkład enzymatyczny. Biotechnologia 2005, 1, 152–165. [Google Scholar]
- Luczaj, Ł.; Adamczak, A.; Duda, M. Tannin content in acorns (Quercus spp.) from Poland. Dendrobiology 2014, 72, 103–111. [Google Scholar] [CrossRef]
- Al-Rousan, W.M.; Ajo, T.Y.; Al-Ismail, K.M.; Shaker, R.R.; Osaili, T.M. Characterization of acorn fruit oils extracted from selected Mediterranean Quercus Species. Grasas Aceites 2013, 64, 554–560. [Google Scholar] [CrossRef]
- Taib, M.; Rezzak, Y.; Bouyazza, L.; Lyoussi, B. Medicinal uses, phytochemistry, and pharmacological activities of Quercus species. Evid. Based. Complement. Altern. Med. 2020, 2020, 1920683. [Google Scholar] [CrossRef] [PubMed]
- Górnaś, P. Oak Quercus rubra L. and Quercus robur L. acorns as an unconventional source of gamma-and beta-tocopherol. Eur. Food Res. Technol. 2019, 245, 257–261. [Google Scholar] [CrossRef]
- Górnaś, P.; Rudzińska, M.; Grygier, A.; Ying, Q.; Mišina, I.; Urvaka, E.; Rungis, D. Sustainable valorization of oak acorns as a potential source of oil rich in bioactive compounds. Process Saf. Environ. Prot. 2019, 128, 244–250. [Google Scholar] [CrossRef]
- León-Camacho, M.; Viera-Alcaide, I.; Vicario, I.M. Acorn (Quercus spp.) fruit lipids: Saponifiable and unsaponifiable fractions: A detailed study. J. Am. Oil Chem. Soc. 2004, 81, 447–453. [Google Scholar] [CrossRef]
- Das, A.K.; Islam, M.N.; Faruk, M.O.; Ashaduzzaman, M.; Dungani, R. Review on tannins: Extraction processes, applications and possibilities. S. Afr. J. Bot. 2020, 135, 58–70. [Google Scholar] [CrossRef]
- Hassanpour, S.; MaheriSis, N.; Eshratkhah, B. Plants and secondary metabolites (Tannins): A Review. Int. J. For. Soil Eros. 2011, 1, 47–53. [Google Scholar]
- Thakur, A.; Sharma, V.; Thakur, A. An overview of anti-nutritional factors in food. Int. J. Chem. Stud. 2019, 7, 2472–2479. [Google Scholar]
- Sharma, K.; Kumar, V.; Kaur, J.; Tanwar, B.; Goyal, A.; Sharma, R.; Gat, J.; Kumar, A. Health effects, sources, utilization and safety of tannins: A critical review. Toxin Rev. 2021, 40, 432–444. [Google Scholar] [CrossRef]
- Javaid, N.; Shah, M.A.; Rasul, A.; Chauhdary, Z.; Saleem, U.; Khan, H.; Ahmed, N.; Uddin, M.S.; Mathew, B.; Behl, T.; et al. Neuroprotective effects of ellagic acid in Alzheimer’s disease: Focus on underlying molecular mechanisms of therapeutic potential. Curr. Pharm. Des. 2021, 27, 3591–3601. [Google Scholar] [CrossRef]
- Serrano, J.; Puupponen-Pimiä, R.; Dauer, A.; Aura, A.M.; Saura-Calixto, F. Tannins: Current knowledge of food sources, intake, bioavailability and biological effects. Mol. Nutr. Food Res. 2009, 53, S310–S329. [Google Scholar] [CrossRef]
- Smeriglio, A.; Barreca, D.; Bellocco, E.; Trombetta, D. Proanthocyanidins and hydrolysable tannins: Occurrence, dietary intake and pharmacological effects. Br. J. Pharmacol. 2017, 174, 1244–1262. [Google Scholar] [CrossRef]
- Barbehenn, R.V.; Constabel, C.P. Tannins in plant–herbivore interactions. Phytochemistry 2011, 72, 1551–1565. [Google Scholar] [CrossRef]
- Awad, H.M.; Boersma, M.G.; Boeren, S.; van Bladeren, P.J.; Vervoort, J.; Rietjens, I.M. Structure–activity study on the quinone/quinone methide chemistry of flavonoids. Chem. Res. Toxicol. 2001, 14, 398–408. [Google Scholar] [CrossRef]
- Espín, J.C.; García-Conesa, M.T.; Tomás-Barberán, F. Biological significance of urolithins, the gut microbial ellagic acid-derived metabolites: The evidence so far. Evid. Based. Complement. Altern. Med. 2013, 2013, 270418. [Google Scholar] [CrossRef]
- García-Villalba, R.; Espín, J.C.; Tomás-Barberán, F.A. Chromatographic and spectroscopic characterization of urolithins for their determination in biological samples after the intake of foods containing ellagitannins and ellagic acid. J. Chromatogr. A 2016, 1428, 162–175. [Google Scholar] [CrossRef]
- Giménez-Bastida, J.A.; Ávila-Gálvez, M.Á.; Espín, J.C.; González-Sarrías, A. The gut microbiota metabolite urolithin A, but not other relevant urolithins, induces p53-dependent cellular senescence in human colon cancer cells. Food Chem. Toxicol. 2020, 139, 111260. [Google Scholar] [CrossRef]
- Kang, I.; Buckner, T.; Shay, N.F.; Gu, L.; Chung, S. Improvements in metabolic health with consumption of ellagic acid and subsequent conversion into urolithins: Evidence and mechanisms. Adv. Nutr. 2016, 7, 961–972. [Google Scholar] [CrossRef]
- Silva, S.; Costa, E.M.; Borges, A.; Carvalho, A.P.; Monteiro, M.J.; Pintado, M.M.E. Nutritional characterization of acorn flour (a traditional component of the Mediterranean gastronomical folklore). J. Food Meas. Charact. 2016, 10, 584–588. [Google Scholar] [CrossRef]
- Bainbridge, D.A. The use of acorns for food in California: Past, present, future. In Proceedings of the Symposium on Multiple-Use Management of California’s Hardwood Resources General Technical Report PSW-100; Plumb, T.R., Pillsbury, N.H., Eds.; USDA: Berkeley, CA, USA, 1987; pp. 453–458. [Google Scholar]
- Luczaj, Ł. Dziko rosnące rośliny jadalne użytkowane w Polsce od połowy XIX w. do czasów współczesnych. Etnobiologia Pol. 2011, 1, 57–125. [Google Scholar]
- Pignone, D.; Laghetti, G. On sweet acorn (Quercus spp.) cake tradition in Italian cultural and ethnic islands. Genet. Resour. Crop Evol. 2010, 57, 1261–1266. [Google Scholar] [CrossRef]
- Overstreet, S.; Choi, S.; Park, C.R.; Lee, D.; Gradziel, T. Acorn production and utilization in the Republic of Korea. In Proceedings of the California Oak Symposium: Managing Oak Woodlands in a Dynamic World, Ambler, PA, USA, 3–6 November 2014. [Google Scholar]
- García-Gómez, E.; Pérez-Badia, R.; Pereira, J.; Puri, R.K. The Consumption of Acorns (from Quercus spp.) in the Central West of the Iberian Peninsula in the 20th Century. Econ. Bot. 2017, 71, 256–268. [Google Scholar] [CrossRef]
- Makhlouf, F.Z.; Squeo, G.; Difonzo, G.; Faccia, M.; Pasqualone, A.; Summo, C.; Barkat, M.; Caponio, F. Effects of storage on the oxidative stability of acorn oils extracted from three different Quercus species. J. Sci. Food Agric. 2020, 101, 131–138. [Google Scholar] [CrossRef]
- Ajo, R.Y. Effect of acorn flour substitution on Arabic Bread properties. Pak. J. Agric. Sci. 2018, 55, 913–919. [Google Scholar]
- Molavi, H.; Keramat, J.; Raisee, B. Evaluation of the cake quality made from acorn—Wheat flour blends as a functional food. J. Food Biosci. Technol. 2015, 5, 53–60. [Google Scholar]
- Parsaei, M.; Goli, M.; Abbasi, H. Oak flour as a replacement of wheat and corn flour to improve biscuit antioxidant activity. Food Sci. Nutr. 2018, 6, 253–258. [Google Scholar] [CrossRef]
- Shishehbor, F.; Salimi, Z.; Veissi, M.; Malehi, A.S.; Shiri-Nasab, M.; Helli, B. Effect of oak flour on glycemic index and satiety index of white bread. Iran. Red Crescent Med. J. 2020, 22, e95552. [Google Scholar] [CrossRef]
- Pasqualone, A.; Makhlouf, F.Z.; Barkat, M.; Difonzo, G.; Summo, C.; Squeo, G.; Caponio, F. Effect of acorn flour on the physico-chemical and sensory properties of biscuits. Heliyon 2019, 5, e02242. [Google Scholar] [CrossRef] [PubMed]
- Sekeroglu, N.; Ozkutlu, F.; Kilic, E. Mineral composition of acorn coffees. Indian J. Pharm. Educ. Res. 2017, 51, S504–S507. [Google Scholar] [CrossRef]
- Amina, M.; Djamel, F. Effect of fermentation and germination treatments on physicochemical and sensory properties of enriched biscuits with acorn flour. Ann. Food Sci. Technol. 2018, 19, 667–674. [Google Scholar]
- Coelho, M.; Silva, S.; Rodríguez-Alcalá, L.M.; Oliveira, A.; Costa, E.M.; Borges, A.; Martins, C.; Rodrigues, A.S.; Pintado, M.M.E. Quercus based coffee-like beverage: Effect of roasting process and functional characterization. J. Food Meas. Charact. 2018, 12, 471–479. [Google Scholar] [CrossRef]
- Martins, R.B.; Nunes, M.C.; Ferreira, L.M.M.; Peres, J.A.; Barros, A.I.R.N.A.; Raymundo, A. Impact of acorn flour on gluten-free dough rheology properties. Foods 2020, 9, 560–574. [Google Scholar] [CrossRef]
- Sardão, R.; Alexandre, E.; Amaral, R.; Saraiva, J.A.; Pintado, M. Effect of high pressure processing on a functional acorn beverage. In Proceedings of the XXVI Encontro Nacional da Sociedade Portuguesa de Química, Aveiro, Portugal, 24–26 July 2019. [Google Scholar]
- Zarroug, Y.; Boulares, M.; Mejri, J.; Slimi, B.; Hamdaoui, G.; Djebi, S.; Saidi, F.; Nasri, H.; Sfayhi, D.T.; Kharrat, M. Extraction and characterization of Tunisian Quercus ilex starch and its effect on fermented dairy product quality. Int. J. Anal. Chem. 2020, 2020, 8868673. [Google Scholar] [CrossRef] [PubMed]
- Purabdolah, H.; Sadeghi, A.; Ebrahimi, M.; Kasheninejad, M.; Tabarestani, H.S.; Mohamadzadeh, J. Techno-functional properties of the selected antifungal predominant LAB isolated from fermented acorn (Quercus persica). J. Food Meas. Charact. 2020, 14, 1754–1764. [Google Scholar] [CrossRef]
- Gonzaga, M.; Batista, M.; Guiné, R.P.F.; Correia, P. Development and characterization of wheat breads with acorn flour. In Proceedings of the International Conference on Engineering, Covilhǎ, Portugal, 2–4 December 2015. [Google Scholar]
- Özcan, T. Fatty acid composition in the acorn oil of Quercus rubra L. cultivate in NW Turkey. J. Appl. Biol. Sci. 2008, 2, 51–55. [Google Scholar]
- Korus, J.; Witczak, M.; Ziobro, R.; Juszczak, L. The influence of acorn flour on rheological properties of gluten- free dough and physical characteristics of the bread. Eur. Food Res. Technol. 2015, 240, 1135–1143. [Google Scholar] [CrossRef]
- Amina, M.; Djamel, F.; Djamel, H. Influence of fermentation and germination treatments on physicochemical and functional properties of acorn flour. Bulg. J. Agric. Sci. 2018, 24, 719–726. [Google Scholar]
- Correia, P.R.; Leitão, A.E.; Beirão-da-Costa, M.L. Effect of drying temperatures on chemical and morphological properties of acorn flours. Int. J. Food Sci. Technol. 2009, 44, 1729–1736. [Google Scholar] [CrossRef]
- Correia, P.R.; Beirão-da-Costa, M.L. Effect of drying temperatures on starch-related functional and thermal properties of acorn flours. J. Food Sci. 2011, 76, 196–202. [Google Scholar] [CrossRef] [PubMed]
- Pinto da Fonseca, A.M. A bolota. O Gorgulho. Boletim Informativo sobre Biodiversidade Agrícola. 2008, 8, 11–13. Available online: https://www.aldeia.org/portal/user/documentos/bol_4-8.pdf (accessed on 12 January 2024).
- Adams, J.D.; Garcia, C.; Lien, E.J. A comparison of Chinese and American Indian (Chumash) medicine. Evid. Based. Complement. Altern. Med. 2010, 7, 219–225. [Google Scholar] [CrossRef]
- Kaynar, P. Antimicrobial activity of Quercus robur L. (Acorn). Fresenius Environ. Bull. 2017, 26, 6992–6995. [Google Scholar]
- Satıl, F.; Akan, H.; Karaaslan, M.; Balos, M.M.; Başyiğit, B. Ethnobotanical and chemical studies on gezo molasses from Quercus brantii Lindl. Acorns in Turkey. Acta Soc. Bot. Pol. 2021, 90, 9011. [Google Scholar] [CrossRef]
- Morales, D. Oak trees (Quercus spp.) as a source of extracts with biological activities: A narrative review. Trends Food Sci. Technol. 2021, 109, 116–125. [Google Scholar] [CrossRef]
- Custódio, L.; Patarra, J.; Albericio, F.; Neng, N.R.; Nogueira, J.M.F.; Romano, A. Phenolic composition, antioxidant potential and in vitro inhibitory activity of leaves and acorns of Quercus suber on key enzymes relevant for hyperglycemia and Alzheimer’s disease. Ind. Crops Prod. 2015, 64, 45–51. [Google Scholar] [CrossRef]
- Dogan, A.; Celik, I.; Kaya, M.S. Antidiabetic properties of lyophilized extract of acorn (Quercus brantii Lindl.) on experimentally STZ-induced diabetic rats. J. Ethnopharmacol. 2015, 176, 243–251. [Google Scholar] [CrossRef] [PubMed]
- Nogueira, A.; Soares, C.; Grosso, C.; Ramalhosa, M.J. Acorns as a functional food for cardiovascular disease prevention: Chemical characterization and bioactivity. Biol. Life Sci. Forum 2022, 18, 59. [Google Scholar] [CrossRef]
- Şenol, F.S.; Şekeroğlu, N.; Gezici, S.; Kilic, E.; Orhan, İ.E. Neuroprotective potential of the fruit (acorn) from Quercus coccifera L. Turk. J. Agric. For. 2018, 42, 82–87. [Google Scholar]
- Uyar, A.; Jhangir, G.M.; Keleş, Ö.F.; Yener, Z. The effects of Quercus (Oak) acorn on cutaneous wound healing in rats. J. Plant Based Pharm. 2023, 3, 148–155. [Google Scholar] [CrossRef]
- Aslani, A.; Ghannadi, A.; Najafi, H. Design, formulation and evaluation of a mucoadhesive gel from Quercus brantii L. and Coriandrum sativum L. as periodontal drug delivery. Adv. Biomed. Res. 2013, 2, 21. [Google Scholar] [CrossRef] [PubMed]
- Moon, H.R.; Chung, M.J.; Park, J.W.; Cho, S.M.; Choi, D.J.; Kim, S.M.; Chun, M.H.; Park, Y.I. Antiasthma effects through anti-inflammatory action of acorn (Quercus acutissima carr.) in vitro and in vivo. J. Food Biochem. 2013, 37, 108–118. [Google Scholar] [CrossRef]
- Faramarzian, M.; Bahramikia, S. The effects of Quercus brantii acorn extract on hen egg-white lysozyme amyloid formation and disassemble amyloid aggregates. J. Food Process. Preserv. 2020, 44, e14499. [Google Scholar] [CrossRef]
- Sasani, N.; Kazemi, A.; Babajafari, S.; Amiri-Ardekani, E.; Rezaiyan, M.; Barati-Boldaji, R.; Mazloomi, S.E.; Clark, C.C.T.; Ashrafi-Dehkordi, E. The effect of acorn muffin consumption on glycemic indices and lipid profile in type 2 diabetic patients: A randomized double-blind placebo-controlled clinical trial. Food Sci. Nutr. 2023, 11, 883–891. [Google Scholar] [CrossRef] [PubMed]
- Zare, A.; Moshfeghy, Z.; Zarshenas, M.M.; Jahromi, B.N.; Akbarzadeh, M.; Sayadi, M. Quercus brantii Lindl. Vaginal cream versus placebo on Bacterial Vaginosis: A randomized clinical trial. J. Herb. Med. 2019, 16, 100247. [Google Scholar] [CrossRef]
- Bahador, N.; Baserisalehi, M. The effect of Quercus castaneifolia extract on pathogenic enteric bacteria. Anaerobe 2011, 17, 358–360. [Google Scholar] [CrossRef] [PubMed]
- Gezici, S.; Sekeroglu, N. Neuroprotective potential and phytochemical composition of acorn fruits. Ind. Crops Prod. 2019, 128, 13–17. [Google Scholar] [CrossRef]
- Amessis-Ouchemoukh, N.; Ouchemoukh, S.; Meziant, N.; Idiri, Y.; Hernanz, D.; Stinco, C.M.; Rodríguez-Pulido, F.J.; Heredia, F.J.; Madani, K.; Luis, J. Bioactive metabolites involved in the antioxidant, anticancer and anticalpain activities of Ficus carica L., Ceratonia siliqua L. and Quercus ilex L. extracts. Ind. Crops Prod. 2017, 95, 6–17. [Google Scholar] [CrossRef]
- Xu, J.; Wang, X.; Su, G.; Yue, J.; Sun, Y.; Cao, J.; Zhang, X.; Zhao, Y. The antioxidant and anti-hepatic fibrosis activities of acorns (Quercus liaotungensis) and their natural galloyl triterpenes. J. Funct. Foods 2018, 46, 567–578. [Google Scholar] [CrossRef]
- Mai, Y.; Wang, Z.; Wang, Y.; Xu, J.; He, X. Anti-neuroinflammatory triterpenoids from the seeds of Quercus serrata Thunb. Fitoterapia 2020, 142, 104523. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Xu, J.; Wang, Y.; Lei, Y.; Mai, Y.; He, X. Q43, a new triterpenoid extracted from Chinese acorn, exhibits pronounced anti-neuroinflammatory activity through the MAPK and NF-κB pathways. J. Funct. Foods 2021, 83, 104566. [Google Scholar] [CrossRef]
- Wu, M.; Yang, Q.; Wu, Y.; Ouyang, J. Inhibitory effects of acorn (Quercus variabilis Blume) kernel-derived polyphenols on the activities of α-amylase, α-glucosidase, and dipeptidyl peptidase IV. Food Biosci. 2021, 43, 101224. [Google Scholar] [CrossRef]
- Custódio, L.; Patarra, J.; Albericio, F.; Neng, N.R.; Nogueira, J.M.F.; Romano, A. Extracts from Quercus sp. acorns exhibit in vitro neuroprotective features through inhibition of cholinesterase and protection of the human dopaminergic cell line SH-SY5Y from hydrogen peroxide-induced cytotoxicity. Ind. Crops Prod. 2013, 45, 114–120. [Google Scholar] [CrossRef]
- Jiang, Q.; Im, S.; Wagner, J.G.; Hernandez, M.L.; Peden, D.B. Gamma-tocopherol, a major form of vitamin E in diets: Insights into antioxidant and anti-inflammatory effects, mechanisms, and roles in disease management. Free Radic. Biol. Med. 2022, 178, 347–359. [Google Scholar] [CrossRef] [PubMed]
- Pahrudin Arrozi, A.; Shukri, S.N.S.; Wan Ngah, W.Z.; Mohd Yusof, Y.A.; Ahmad Damanhuri, M.H.; Jaafar, F.; Makpol, S. Comparative effects of alpha- and gamma-tocopherol on mitochondrial functions in Alzheimer’s disease in vitro model. Sci. Rep. 2020, 10, 8962. [Google Scholar] [CrossRef] [PubMed]
- Khan, U.M.; Sevindik, M.; Zarrabi, A.; Nami, M.; Ozdemir, B.; Kaplan, D.N.; Selamoglu, Z.; Hasan, M.; Kumar, M.; Alshehri, M.M.; et al. Lycopene: Food sources, biological activities, and human health benefits. Oxid. Med. Cell. Longev. 2021, 2021, 2713511. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Chen, H.; Su, Y.; Liu, H.; Hu, J.; Hong, K. Increased blood alpha-carotene, all-trans-Beta-carotene and lycopene levels are associated with beneficial changes in heart rate variability: A CVD-stratified analysis in an adult population-based study. Nutr. J. 2021, 20, 43. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Tawwab, M.; Abdulrahman, N.M.; Ahmad, V.M.; Ramzi, D.O.; Hassan, B.R. Effects of dietary oak (Quercus aegilops L.) acorn on growth performance, somatic indices, and hemato-biochemical responses of common carp, Cyprinus carpio L., at different stocking densities. J. Appl. Aquac. 2022, 34, 877–893. [Google Scholar] [CrossRef]
- Mekki, I.; Smeti, S.; Hajji, H.; Yagoubi, Y.; Mahouachi, M.; Atti, N. Effect of oak acorn (Quercus ilex) intake during suckling and fattening of Barbarine lambs on growth, meat quality and fatty acid profile. J. Anim. Feed Sci. 2019, 28, 22–30. [Google Scholar] [CrossRef]
- Pugliese, C.; Sirtori, F.; Ruiz, J.; Martin, D.; Parenti, S.; Franci, O. Effect of pasture on chestnut or acorn on fatty acid composition and aromatic profile of fat of Cinta Senese dry-cured ham. Grasas Aceites 2009, 60, 271–276. [Google Scholar] [CrossRef]
- Belkova, J.; Rozkot, M.; Weisbauerova, E.; Truneckova, J. Influence of feed substitution with acorns on fattening performance, carcass characteristics, and meat composition of Prestice Black-Pied pigs finished in a conventional indoor system. Cogent Food Agric. 2023, 9, 2220182. [Google Scholar] [CrossRef]
- Szyndler-Nędza, M.; Świątkiewicz, M.; Migdał, Ł.; Migdał, W. The quality and health-promoting value of meat from pigs of the native breed as the effect of extensive feeding with acorns. Animals 2021, 11, 789. [Google Scholar] [CrossRef]
- Stiti, B.; Khalfaoui, M.; Bahri, S.; Khaldi, A. Towards optimizing acorn use as animal feed in Tunisia: Evaluation and impact on natural regeneration. BOIS For. Trop. 2021, 348, 17–26. [Google Scholar] [CrossRef]
- Huang, W.; Niu, H.; Li, Z.; Lin, W.; Gong, G.; Wang, W. Effect of ellagitannin acyl hydrolase, xylanase and cellulase on ellagic acid production from cups extract of valonia acorns. Process Biochem. 2007, 42, 1291–1295. [Google Scholar] [CrossRef]
- Mota, S.; Pinto, C.; Cravo, S.; Rocha e Silva, J.; Afonso, C.; Sousa Lobo, J.M.; Tiritan, M.E.; Cidade, H.; Almeida, I.F. Quercus suber: A Promising Sustainable Raw Material for Cosmetic Application. Appl. Sci. 2022, 12, 4604. [Google Scholar] [CrossRef]
- Kyriakidou, K.; Mourtzinos, I.; Biliaderis, C.G.; Makris, D.P. Optimization of a green extraction/inclusion complex formation process to recover antioxidant polyphenols from oak acorn husks (Quercus robur) using aqueous 2-hydroxypropyl-β-cyclodextrin/glycerol mixtures. Environments 2016, 3, 3. [Google Scholar] [CrossRef]
- Karabaş, H. Biodiesel production from crude acorn (Quercus frainetto L.) kernel oil: An optimisation process using the Taguchi method. Renew. Energ. 2013, 53, 384–388. [Google Scholar] [CrossRef]
- Karabaş, H. Acorn (Quercus frainetto L.) kernel oil as an alternative feedstock for biodiesel production in Turkey. J. Energy Resour. Technol. 2013, 135, 01120. [Google Scholar] [CrossRef]
- Karabaş, H. The optimum production parameters of methyl ester from acorn kernel oil. Environ. Prog. Sustain. Energy 2014, 33, 625–628. [Google Scholar] [CrossRef]
- Nourmoradi, H.; Moghadam, K.F.; Jafari, A.; Kamarehie, B. Removal of acetaminophen and ibuprofen from aqueous solutions by activated carbon derived from Quercus brantii (Oak) acorn as a low-cost biosorbent. J. Environ. Chem. Eng. 2018, 6, 6807–6815. [Google Scholar] [CrossRef]


| Quercus Species | Chemical Group | Subgroup | Content | References |
|---|---|---|---|---|
| Q. coccifera | tannins [mg/100 g d.m.] | condensed | 2670 | [44] |
| Q. ilex | TPC [mg GAE/100 g d.m.] | - | 1100.17–5692 | [14,63] |
| TFC [mg GAE/100 g d.m.] | - | 47.3 | ||
| tannins [mg CE/100 g d.m.] | - | 903 | ||
| Q. robur | TPC [mg GAE/100 g d.m.] | - | 22,300 | [64] |
| tannins [mg GAE/100 g d.m.] | - | 20,400 | ||
| Q. rotundifolia | TPC [mg GAE/100 g d.m.] | - | 840–9163 | [42,48] |
| tannins [mg CE/100 g d.m.] | condensed | 50–140 | ||
| hydrolysable | 950–1150 | |||
| gallotannins | 740–860 | |||
| ellagitannins | 210–290 | |||
| Q. suber | TPC [mg GAE/100 g d.m.] | - | 13,290–233,350 | [14,42,44] |
| tannins [mg/100 g d.m.] | condensed | 2090 |
| Quercus Species | Acorn Form/Amount in Product | Food Product Type | Specified Nutritional Advantage | References |
|---|---|---|---|---|
| Q. aegilops | acorn flour/5–15% substitution of wheat flour | Arabic bread | fiber content increase | [97] |
| acorn oil | edible oil | domination of oleic acid, high content of tocopherols (with γ-tocopherol domination), sterols (predominantly β-sitosterol), and phenols | [72] | |
| Q. brantii | acorn flour/5–30% substitution of wheat flour | sponge-fat cake | n.a. | [98] |
| acorn flour/15–45% substitution of wheat flour | wheat biscuits | peroxide value decrease; antioxidant activity increase | [99] | |
| acorn flour/15–45% substitution of corn flour | gluten-free biscuits | peroxide value decrease; antioxidant activity increase | [99] | |
| acorn flour/25–50% substitution of wheat flour | white yeast bread | satiety index increase; feeling of satiety increase | [100] | |
| Q. calliprinos | acorn oil | edible oil | domination of oleic acid, high content of tocopherols (predominantly γ-tocopherol), sterols (predominantly β-sitosterol), and phenols | [72] |
| Q. coccifera | acorn oil | edible oil | high content of tocopherols (predominantly β + γ-tocopherols) and phenols (predominantly hydrolyzable tannins); high antioxidant activity | [12] |
| acorn flour/30–60% substitution of wheat flour | biscuits | total phenolic content increase | [101] | |
| grounded acorn kernels | coffee substitute | relatively high content of minerals (K, Mg, P, Fe, Cu, and Zn) | [102] | |
| Q. ilex | acorn flour/10% substitution of wheat flour | biscuits | ash content increase; starch content decrease | [103] |
| acorn cotyledons | coffee substitute | high phenol content (predominantly ellagic acid) and antioxidant activity | [104] | |
| acorn oil | edible oil | high content of tocopherols (predominantly β + γ-tocopherols) and phenols (predominantly hydrolyzable tannins); high antioxidant activity | [12] | |
| acorn flour/50–75% substitution of barley flour | gluten-free bread | total and insoluble fiber content increase | [105] | |
| acorn flour/30–60% substitution of wheat flour | biscuits | total phenolic content increase | [101] | |
| acorn kernels as raw material | HPP beverage | low thrombogenicity and atherogenicity indices; high content of phenols (predominantly gallic acid) | [106] | |
| acorn kernels as raw material | flour | high fiber and lipids (predominantly unsaturated fatty acids, especially oleic acid) content | [90] | |
| acorn starch | food additives | n.a. | [107] | |
| Q. infectoria | acorn oil | edible oil | domination of oleic acid, high content of tocopherols (predominantly γ-tocopherol), sterols (predominantly β-sitosterol), and phenols | [72] |
| Q. persica | acorn flour/10% substitution of wheat flour | bread | antioxidant activity increase; inhibition of surface moldiness | [108] |
| Q. rotundifolia | acorn flour/10–15% substitution of wheat flour | bread | n.a. | [109] |
| acorn kernels as raw material | flour | high fiber and lipids (predominantly unsaturated fatty acids, especially oleic acid) content | [90] | |
| Q. rubra | acorn oil | edible oil | domination of unsaturated fatty acids, especially oleic acid | [110] |
| Q. suber L. | acorn cotyledons | coffee substitute | high phenols content (predominantly ellagic acid) and antioxidant activity | [104] |
| acorn oil | edible oil | high content of tocopherols (predominantly β + γ-tocopherols) and phenols (predominantly hydrolyzable tannins); high antioxidant activity | [12] | |
| acorn flour/30–60% substitution of wheat flour | biscuits | total phenolic content increase | [101] |
| Quercus Species | Acorn Form | Application Form | Specified Biological Activity | References |
|---|---|---|---|---|
| Q. acutissima | extract from the acorn kernels with 80% ethanol | in vitro experiment and study on mice—orally administered at a dose of 100 or 200 mg/kg body weight/day | anti-inflammatory activity—inhibition of tumor necrosis factor-a and T-helper 2 (Th2) type cytokines (IL-4 and IL-13)—anti-asthma potential | [126] |
| Q. brantii | extract from the seed hull with 70% ethanol | mucoadhesive gel for the treatment of periodontitis | antioxidant and antibacterial activity of polyphenols | [125] |
| water extract from acorns | intraperitoneally administered at a single dose of 50 mg/kg—study on rats | inhibition of hepatic enzymes (ALT, AST, ALP, and LDH)—prevents liver damage | [121] | |
| hydroalcoholic extract from acorn internal fruit | in vitro experiment | inhibition of HEWL amyloid formation | [127] | |
| acorn flour | acorn muffins—oral administration in diet (10 g of acorn flour per day) | reduction of glycated hemoglobin (HbA1c)—anti-diabetic activity of polyphenols | [128] | |
| extract from the acorns’ inner stratum with 80% ethanol | O/W vaginal cream | antibacterial activity of acorn tannins | [129] | |
| Q. castaneifolia | alcoholic and water extracts from acorns | in vitro experiment | anti-microbial effect of alcoholic extract on Escherichia coli, Salmonella thyphimurium, Shigella dysentriae, and Yersinia enterocolitic | [130] |
| Q. coccifera | extract from the acorn kernels with 70% ethanol | in vitro experiment | inhibition of AChE and BChE—neuroprotective potential | [131] |
| extract from the acorn kernels and acorn coffee substitute with 80% ethanol | in vitro experiment | inhibition of AChE and BChE—neuroprotective potential | [123] | |
| Q. faginea | extract from acorn whole fruit, kernel, and pericarp with ethanol-water (1:1, v/v) | in vitro experiment | antioxidant and antibacterial activity of polyphenols, carotenoids, chlorophylls, and tocopherols | [14] |
| Q. ilex | extract from the acorn kernels with ethanol | in vitro experiment | inhibition of xanthine oxidase, high anti-proliferative activity on glioblastoma—antioxidant activity of polyphenols, anti-cancer potential cells | [132] |
| extract from acorn whole fruit, kernel, and pericarp with ethanol-water (1:1, v/v) | in vitro experiment | antioxidant and antibacterial activity of polyphenols, carotenoids, chlorophylls, and tocopherols | [14] | |
| Q. infectoria subsp. boissieri | extract from acorns with methanol | 10% cream with glycerin—study on rats | increase in collagen synthesis and fibroblast and myofibroblast activation | [124] |
| Q. liaotungensis | extract from dried acorn kernels with 75% ethanol and microporous resins | in vitro experiment | antioxidant and anti-fibrotic activity of galloyl triterpenes | [133] |
| Q. nigra | extract from acorn whole fruit, kernel, and pericarp with ethanol-water (1:1, v/v) | in vitro experiment | antioxidant and antibacterial activity of polyphenols, carotenoids, chlorophylls, and tocopherols | [14] |
| Q. robur | extract from powdered acorn kernels with potassium phosphate buffer solution 10 mM | in vitro experiment | inhibition of α-glucosidase—anti-diabetic potential | [122] |
| Q. serrata | extract from acorns with 95% ethanol | in vitro experiment | inhibition of NO production by triterpenoids—antineuroinflammatory potential | [134] |
| Q. serrata var. brevipetiolata | Q43 terpenoid isolated from ethanol (70%) acorn extract | in vitro experiment | inhibition of NO synthase and pro-inflammatory cytokines—antineuroinflammatory potential | [135] |
| Q. suber | extract from the acorns and leaves with hexane, methanol, and water | in vitro experiment | inhibition of AChE and BChE—neuroprotective potential | [120] |
| extract from acorn whole fruit, kernel, and pericarp with ethanol-water (1:1, v/v) | in vitro experiment | antioxidant and antibacterial activity of polyphenols, carotenoids, chlorophylls, and tocopherols | [14] | |
| Q. variabilis | extract from defatted acorn kernels with 70% ethanol (free polyphenols) and sodium hydroxide hydrolysis (bound polyphenols) | in vitro experiment | inhibition of α-amylase, α-glucosidase, and dipeptidyl peptidase IV—polyphenol activity | [136] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Szabłowska, E.; Tańska, M. Acorns as a Source of Valuable Compounds for Food and Medical Applications: A Review of Quercus Species Diversity and Laboratory Studies. Appl. Sci. 2024, 14, 2799. https://doi.org/10.3390/app14072799
Szabłowska E, Tańska M. Acorns as a Source of Valuable Compounds for Food and Medical Applications: A Review of Quercus Species Diversity and Laboratory Studies. Applied Sciences. 2024; 14(7):2799. https://doi.org/10.3390/app14072799
Chicago/Turabian StyleSzabłowska, Emilia, and Małgorzata Tańska. 2024. "Acorns as a Source of Valuable Compounds for Food and Medical Applications: A Review of Quercus Species Diversity and Laboratory Studies" Applied Sciences 14, no. 7: 2799. https://doi.org/10.3390/app14072799
APA StyleSzabłowska, E., & Tańska, M. (2024). Acorns as a Source of Valuable Compounds for Food and Medical Applications: A Review of Quercus Species Diversity and Laboratory Studies. Applied Sciences, 14(7), 2799. https://doi.org/10.3390/app14072799







