1. Introduction
The discovery of natural zeolites has opened a significant chapter in the field of mineralogy, owing to their intriguing surface and structural properties that have been exploited in various sectors, including agriculture, industrial technology, livestock, cosmetics, and the biotechnological industry [
1].
Zeolites form a large family of minerals and are among the most important in terms of microporous materials. The term “zeolite” refers to a polymorph of crystalline silica or aluminosilicate based on various tetrahedral TO
4 units that share corners (T = typically silicon and aluminum), forming a three-dimensional structure with regularly sized molecular dimension pores [
1,
2]. More than 50 different species have been identified within this mineral group, with more yet to be discovered. Zeolites have been classified based on their morphological features, crystalline structure, chemical composition, effective pore diameter, natural occurrence, and other criteria [
3,
4]. The Si/Al ratio is an important characteristic of zeolites. The charge imbalance resulting from the presence of aluminum in the zeolite structure determines the ionic exchange characteristics of zeolites and is expected to induce potential acidic sites. The Si/Al ratio is inversely proportional to the cation content but directly proportional to thermal stability [
3]. Among natural zeolites, clinoptilolite [
5] stands out, commonly used in agricultural practices as a soil amendment and to promote nitrogen retention in soils [
1,
6,
7].
On the other hand, water is one of the essential substances for development, especially in arid and semi-arid regions of the world where renewable water resources are scarce [
2]. Among the various sectors that consume freshwater, agricultural farms are considered the primary consumers, using more than two-thirds of renewable water resources in these activities [
8]. Additionally, population growth demands greater food quantities, making efficient cultivation crucial to sustain urban development. While high-efficiency agricultural activities are needed to meet current food demands, several issues have emerged in achieving this goal, primarily related to irrigation and, importantly, the quality of agricultural waters.
The quality of water used for crop irrigation is a crucial factor that can have a significant impact on agricultural productivity and soil health [
9,
10]. It is due to its importance that the FAO has been conducting the review and updating of its “Water Quality for Agriculture” report since 1976 [
11]. This document serves as a field guide for assessing the suitability of water for irrigation and addresses issues such as the following:
Water Salinity: Salinity is a critical factor. If the water used for irrigation has a high salt content, it can accumulate in the soil as water evaporates, affecting the plant’s ability to absorb water and nutrients. This can lead to soil salinization and reduce crop yields.
Infiltration and Drainage: Water quality also affects the rate of water infiltration into the soil and drainage. If the water is of low quality and contains sodium, it can affect soil structure, decrease infiltration, and cause drainage problems. This can result in compacted soil and waterlogging, negatively affecting plant growth.
Toxicity of Specific Ions: Some ions present in water, such as sodium, chloride, and boron, can become toxic to plants at high concentrations. This can affect plant development and reduce yields.
Effects on Soil Health: Poor water quality can contribute to long-term soil degradation. The accumulation of salts, loss of soil structure, and reduced biological activity can impact overall soil health.
Sustainability of Irrigation: Sustainable water management is essential for long-term agriculture. The use of poor-quality water without proper management can deplete water resources and have negative environmental consequences.
Impact on Crop Quality: Water quality can also influence the quality of agricultural products. High concentrations of salts or toxic ions can affect the taste and appearance of crops, which are critical in fresh produce markets.
Among the elements posing significant challenges for removal or reduction in agricultural waters, two stand out: sodium and boron. Sodium can harm crops by creating short-term osmotic stress or causing long-term direct toxicity. As the osmotic pressure decreases in saline soils, plant turgor pressure is affected and tissues dehydrate, slowing down photosynthesis and leading to inhibited growth, biomass loss, a significant reduction in production, and, ultimately, death [
12]. On the other hand, although boron is a necessary micronutrient for the growth and development of plants in very small amounts, exceeding these levels, either through continuous input in irrigation, soil, etc., or through bioaccumulation in the plant organism, can result in various toxic effects, depending on the crops, such as leaf chlorosis and necrosis, root weakness, and death [
13,
14,
15].
In this context, the present study aimed to investigate the sorption of boron and sodium found in natural agricultural waters on commercial clinoptilolite-type zeolite, commonly used as a soil conditioner. The goal was to identify the zeolite’s retention potential for these two phytotoxic cations in crops, providing a complementary and/or secondary benefit to soil improvement, such as water retention, increased oxygen, and nitrogen mobilization. The study of the process and the identification of the optimal experimental conditions were conducted through factorial planning. Additionally, a kinetic study was carried out to investigate the optimal contact time for the sorbent/cation and the sorption mechanisms that best describe the process.
It is emphasized that, despite the abundance of available studies focused on the sorption capacity of zeolite [
16,
17,
18,
19], to date, no research has been found that works with real irrigation waters, which are considered complex due to the diversity of chemical and biological elements they contain. Furthermore, none of these studies concurrently focused on the two chemical species that are most “problematic” in agriculture, sodium and boron, given their negative impact on plant growth and, consequently, on production.
2. Materials and Methods
2.1. Adsorbent Characterization
For this study, commercially available natural zeolite of the clinoptilolite type, specifically FERTCEL zeolite (Zeocel Ltda., Águeda, Portugal), was employed. Widely utilized in agriculture to regulate soil fertility, maximize nutrient utilization, and qualitatively and quantitatively improve crop yields, this zeolite boasts a high cation-exchange capacity (CTC) exceeding 150 mEq/100 g, temporarily retaining ions and releasing them as required by plants. According to the manufacturer’s data, it reduces nitrogen losses in the soil, prevents phosphorus fixation by iron, aluminum, and manganese in acidic soils, enhances potassium efficiency, and mitigates soil acidity. Additionally, it can absorb up to 40% of its weight in water. The physicochemical and mineralogical characterization of clinoptilolite zeolite (hydrated aluminosilicate) was conducted in an independent laboratory specializing in chemical–agricultural analyses, accredited by the National Accreditation Entity (ENAC, Madrid, Spain), ensuring the objectivity of the results.
In this context, the crystalline phases of the zeolite were assessed through X-ray diffraction (XRD, Bruker-AXR D8 Advance) under the following conditions: Δ2θ = 5–65°, 2° increments, integration time = 0.05 s, Cu Kα1 radiation, λ = 1.540598 Å [
20]. The obtained diffractograms were analyzed and compared with the Crystallography Open Database (COD, available at
http://www.crystallography.net/ (accessed on 27 December 2023) using the specialized software Match! version 3.16. The chemical composition was determined through Fourier-transform infrared spectroscopy (FTIR-4700 type A, FTIR spectrometer JASCO Quick Start System with ATR Pro One as an accessory). Spectra were collected with a resolution of 4 cm⁻
1 in the range of 400 cm⁻
1 to 1400 cm⁻
1 and analyzed using OriginPro 9.5 Software (OriginLab Corporation, Northampton, USA) [
21,
22]. Furthermore, laser diffraction techniques (Master Sizer 2000, APA2000 model from Malvern Instruments Ltd., Cambridge, UK) in the range of 0.01 to 3500 µm were employed [
23].
2.2. Irrigation Water Characterization
In pursuit of elucidating the retention and/or immobilization capacity of boron and sodium—elements commonly prevalent in irrigation waters and notorious for their adverse effects on agriculture, particularly in citrus cultivation—samples were drawn from an irrigation reservoir situated on a commercial citric farm in Ojós municipality, Murcia, Spain. The preference for these authentic natural waters rested on their compositional intricacies, posing a significant challenge to replicate accurately in laboratory conditions using synthetic counterparts. The presence or absence of diverse compounds and contaminants in these waters can distinctly shape, modify, or entirely transform the reactions and interactions of dissolved species [
24].
The irrigation water samples, obtained in non-sterile 15 L polypropylene containers from accessible points in the reservoir, were immediately transported and stored at a constant temperature of 4 °C until their characterization and use on the same collection day. A rigorous characterization of all irrigation water samples was carried out before and after the experiments using inductively coupled plasma mass spectrometry (ICP-MS Model 2030, Shimadzu, Kyoto, Japan), selected for its precision and well-established recognition in the characterization of liquid samples. Additionally, continuous real-time monitoring and characterization of all agronomically significant ions (sodium, potassium, pH, electrical conductivity, chloride, nitrate, magnesium, calcium, and ammonium, among others) present in the effluents were meticulously carried out throughout the experiments. This detailed monitoring was conducted using the Imacimus Multi Ion 10 portable multi-ion nutrient analyzer (Imacimus NT Sensors, El Catllar, Spain), which provides readings of the agronomic parameters of interest every 2 min. It is noteworthy that this probe is specialized for field measurements and is widely utilized in the agricultural estates of the region [
25,
26].
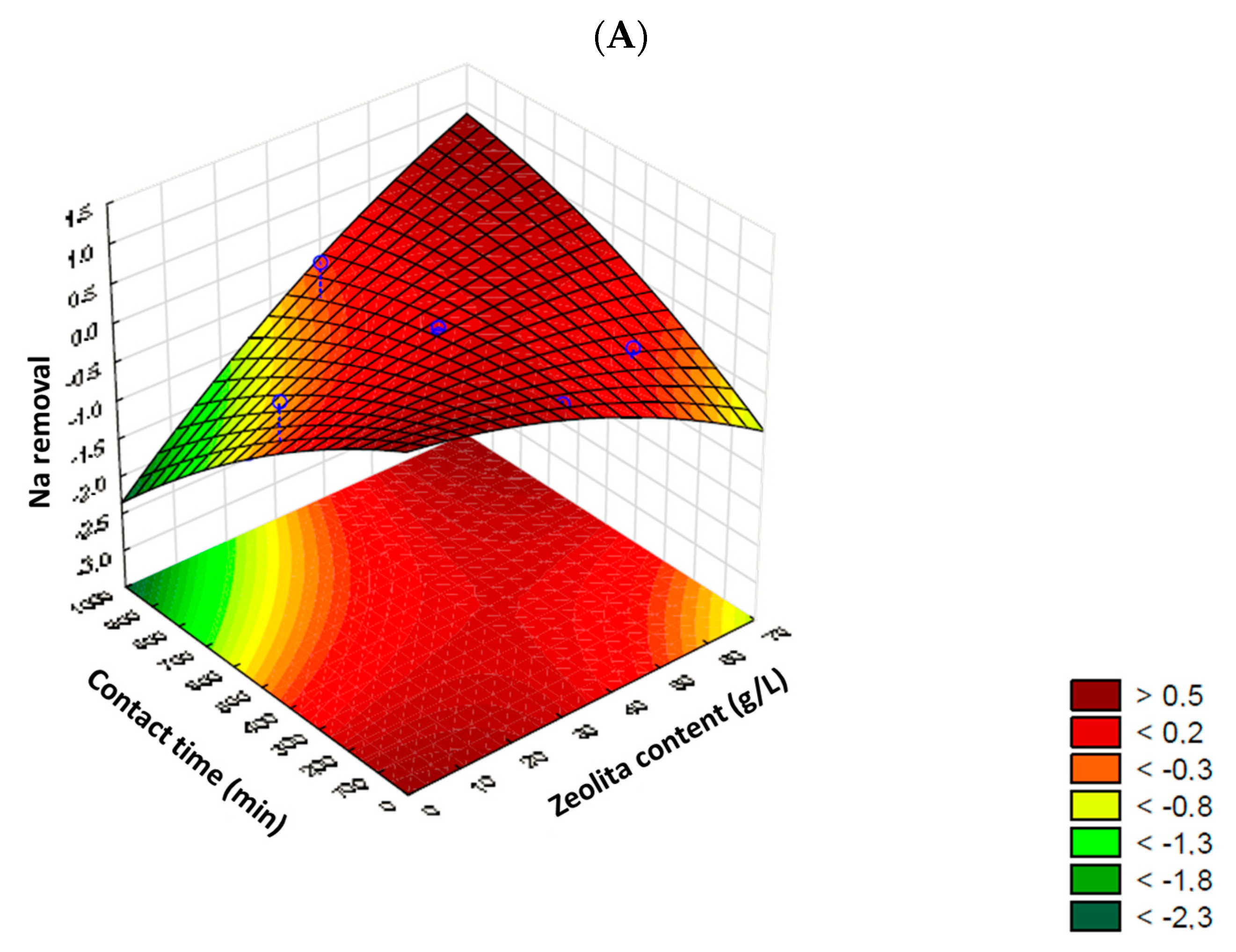
2.3. Statistical Factorial Design
Aiming to determine the optimal system configuration while minimizing the total number of experiments required, a preliminary factorial design was conducted. This design identified factors with significant effects on response and elucidated how a factor’s impact varies, considering the levels of other factors. The exploration of interactions between factors necessitates the application of statistical experimental designs, as optimizing the system cannot be achieved by varying one factor at a time while keeping other factors constant [
27,
28]. In this sense, and aiming to optimize the process, a central composite rotatable design (CCRD) with two factorial variables (2
2) was employed, where the zeolite content (g/L) and contact time (min) served as independent variables, while sodium (Na) and boron (B) concentrations constituted the dependent or response variables. A total of 12 batch experiments were conducted by the statistical design matrix and the studied factorial levels, as presented in
Table 1. To minimize experimental error, the study included a quadruplicate repetition of the central point. Initial levels for the factorial setup were determined based on the literature [
29,
30,
31,
32,
33]. Once the effective range of removal variables was identified through a bibliographic review, preliminary removal assays were conducted by the same scientific team (unpublished data). Building upon the outcomes of these preliminary assays, the minimum and maximum adsorbent content for use in this study were subsequently established. The values for the axial points (−1.4142 and +1.4142) were determined through interpolation by the model’s methodology [
27,
34].
All experiments were conducted at a constant temperature of 22 ± 2 °C. Each trial involved using 100 mL of liquid sample in non-sterile polypropylene Erlenmeyer flasks with a maximum capacity of 250 mL and a standardized neck (Kartell Labware, Noviglio, Italy). Cellulose acetate membranes of 0.20 µm and 47 mm in diameter (Teknokroma, Barcelona, Spain) were employed for sample water filtration.
The experimental data were analyzed using appropriate statistical methods for the applied experimental design. Mathematical models for each response were assessed through multiple linear regression analysis and were processed using STATISTICA 18 software from StatSoft (TIBCO Software Inc., Palo Alto, CA, USA). Statistically significant terms in the model, whether linear or nonlinear, were determined through analysis of variance (ANOVA) for each response, with a significance level set at
p < 0.05 (95% confidence). Identifying the significant factors in the model allowed for a reliable prediction of performance through interpolation across a range of data, facilitating the construction of response surface graphs (3D). These graphs contribute to a better understanding of the investigated process [
35].
2.4. Batch Kinetic Sorption Tests
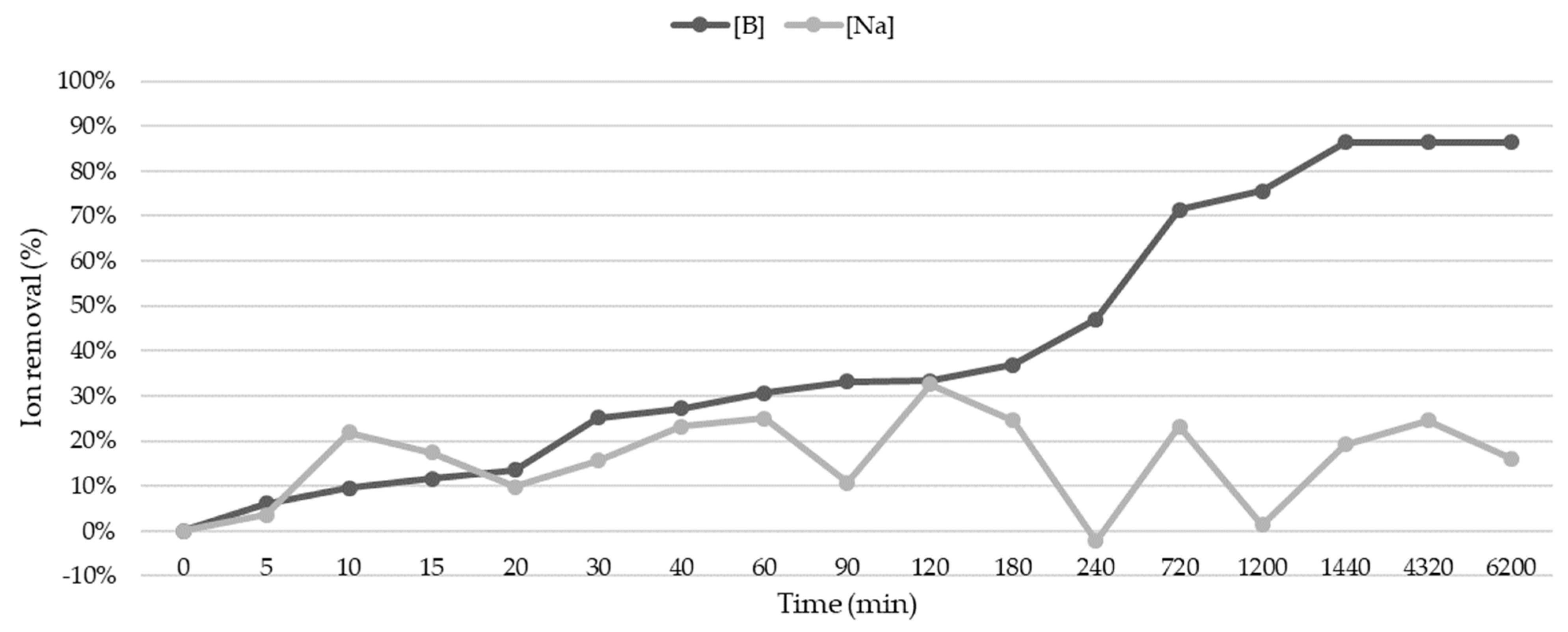
Based on the results obtained from the experimental factorial design, kinetic sorption tests were conducted to identify and define the most appropriate kinetics that describe the process. This knowledge is crucial, as it provides valuable information about the mechanism of the process and indicates the sorption rate, which, in treating an effluent, determines the residence time of the sorbate at the interface between the solution and the sorbent material.
For this purpose, 100 mL of irrigation water was placed in non-sterile polypropylene Erlenmeyer flasks with a maximum capacity of 250 mL, containing 4.93 g of zeolite/L, and a total contact time of 6200 min at a constant controlled temperature (22 ± 1 °C). In all cases, the flasks were covered with a transparent biofilm to prevent dust and/or impurities from entering. The quantity of zeolite and the total contact time were predetermined through factorial experimental planning.
At various predefined times, each Erlenmeyer flask was opened for sampling and monitoring of cation concentrations. A total of 17 tests were conducted. Before the characterization analyses, the aqueous samples were filtered with cellulose acetate filters of 0.20 µm and 47 mm in diameter (Teknokroma, Barcelona, Spain). The percentage removal of sodium and boron was determined using Equation (1), where C
0 and C
t represent the initial concentration of cations and the concentration at time t (mg/L), respectively.
The assessment of the kinetic mechanism of the sorption process was conducted by applying the three most commonly used kinetic models to the experimental data. These models are (
i) Lagergren’s pseudo-first-order model, commonly employed for liquid-solid sorption, as it considers the rate of occupation of active sites to be proportional to the number of available active sites in the adsorbent material, also known as physisorption [
36,
37]; (
ii) Ho and McKay’s pseudo-second-order model, based on solid-phase sorption, presupposing it as a chemical sorption process involving the participation of valence forces or electron exchange between sorbate and sorbent, thus describing chemisorption [
38]; and (
iii) Weber and Morris’s intraparticle diffusion model, wherein the limiting stage of the sorption process may be caused by the slowness of the intraparticle diffusion mechanism [
39]. The corresponding equations of the kinetic models are shown in
Table 2.
4. Conclusions
This study addressed the issue of water quality in an irrigation reservoir with high levels of boron (B) and sodium (Na)—9.7 and 0.75 meq/L, respectively—which adversely affect the development of sensitive crops such as citrus. It is crucial to highlight that this study, utilizing zeolite as a sorbent, was conducted in a context where this material was already employed as an agricultural soil amendment in southeastern Spain, an area where irrigation water quality is often compromised.
The results indicated that sodium removal exhibited limited efficiency (10–24%), with desorption observed in some cases, underscoring the challenges of combined removal with boron and various elements present in natural irrigation waters. On the other hand, boron removal demonstrated higher efficiency (up to 100%) under specific conditions, with significant interactions and influences from other chemical species present in water and zeolite composition. Statistical analysis, including analysis of variance (ANOVA), identified significant factors for boron removal (zeolite content and contact time) but not for sodium. Based on statistical significance and practicality, optimal conditions for boron removal were determined as 4.93 g of zeolite/L and 723 min of contact time.
Kinetic studies revealed differential behaviors for sodium and boron, with cyclic sorption/desorption observed for sodium and slow, steady sorption for boron. The pseudo-first-order model was found to be applicable for both cations, indicating a physical or physisorption process, while the pseudo-second-order model suggested possible chemisorption for boron. pH variations during the process indicated a slight decrease, potentially explaining lower sodium adsorption, aligning with literature suggesting higher sodium adsorption at a higher pH.
The study paves the way for future research, particularly in understanding the complex interactions influencing the sorption behavior of sodium and boron on zeolite under dynamic conditions. The findings contribute to the optimization of zeolite-based treatments for agricultural irrigation water, providing insights into challenges and potential solutions for sodium and boron removal.