Evaluation of Sit-to-Stand Movement Focusing on Kinematics, Kinetics, and Muscle Activity after Modern Total Knee Arthroplasty
Abstract
1. Introduction
2. Materials and Methods
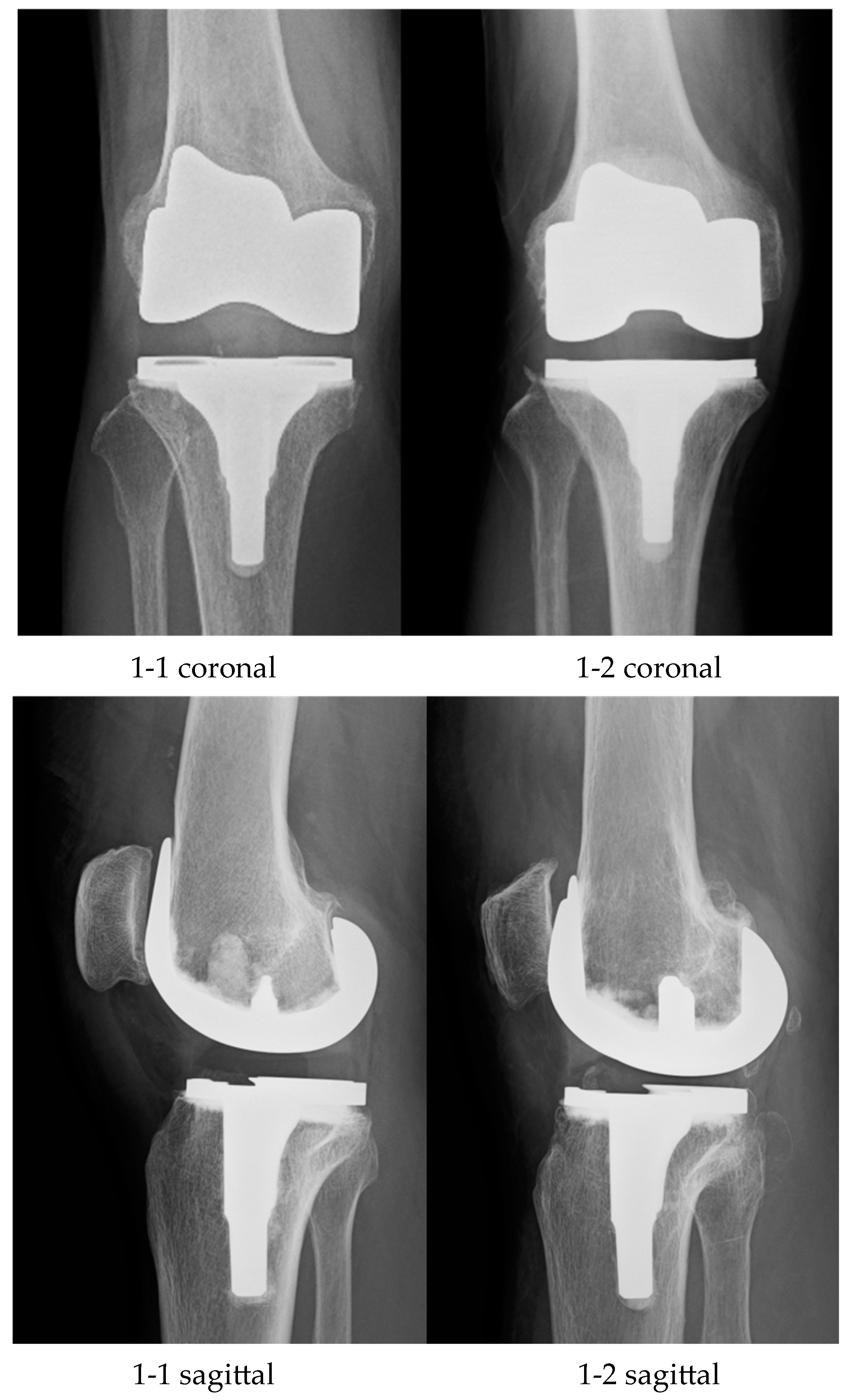
2.1. Surgery and Postoperative Rehabilitation
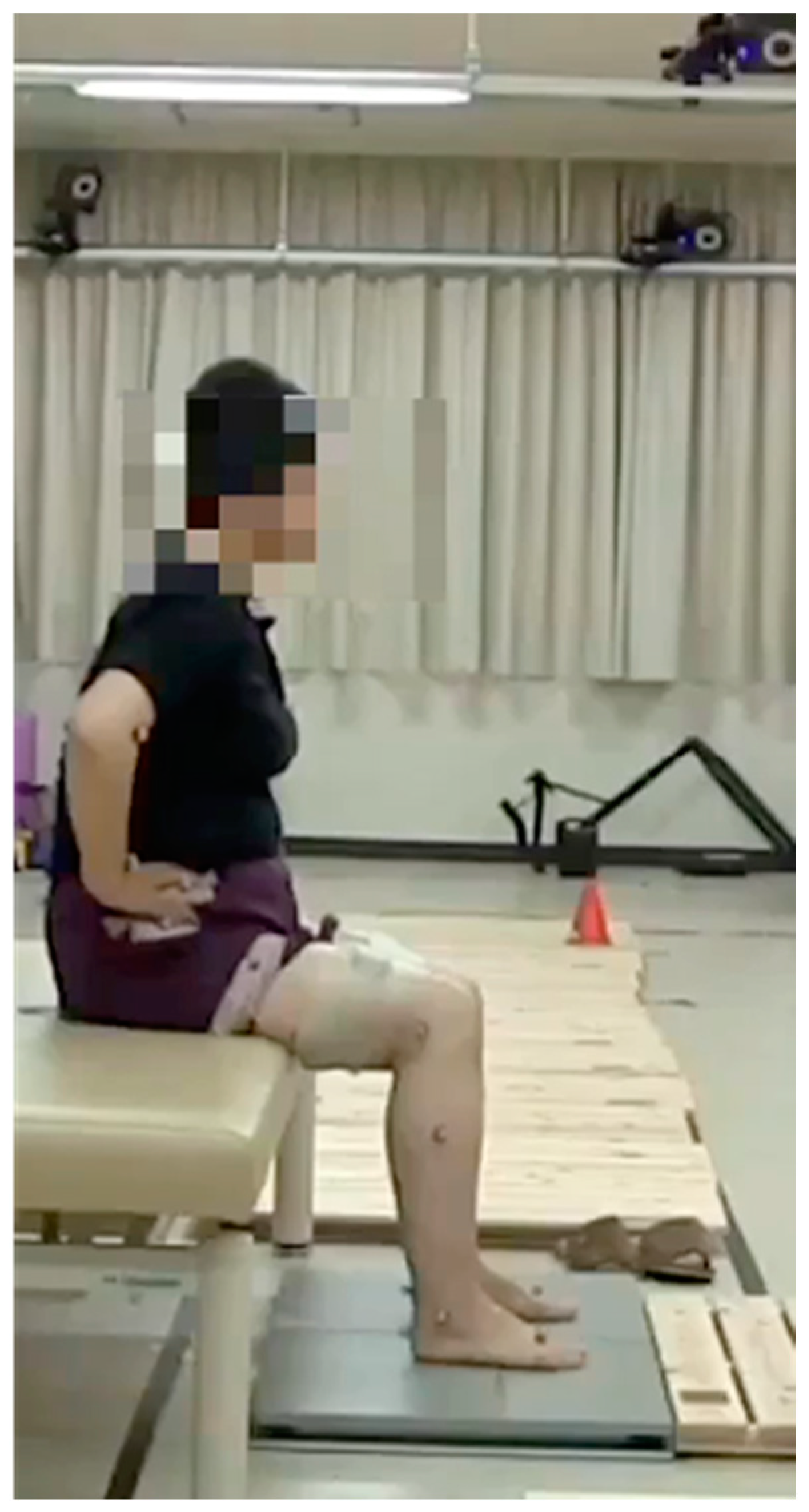
2.2. Evaluation Items and Statistical Analysis
3. Results
3.1. Kinematics
3.2. Kinetics
3.3. Surface Electromyography
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Yoshimura, N.; Muraki, S.; Oka, H.; Mabuchi, A.; En-Yo, Y.; Yoshida, M.; Saika, A.; Yoshida, H.; Suzuki, T.; Yamamoto, S.; et al. Prevalence of knee osteoarthritis, lumbar spondylosis, and osteoporosis in Japanese men and women: The research on osteoarthritis/osteoporosis against disability study. J. Bone Min. Metab. 2009, 27, 620–628. [Google Scholar] [CrossRef] [PubMed]
- Gherghel, R.; Iordan, D.A.; Mocanu, M.D.; Onu, A.; Onu, I. Osteoarthritis is not a disease, but rather an accumulation of predisposing factors. A systematic review. Balneo PRM Res. J. 2021, 12, 218–226. [Google Scholar] [CrossRef]
- Onu, I.; Matei, D.; Sardaru, D.-P.; Cascaval, D.; Onu, A.; Gherghel, R.; Serban, I.L.; Mocanu, G.D.; Iordan, D.A.; Murariu, G.; et al. Rehabilitation of Patients with Moderate Knee Osteoarthritis Using Hyaluronic Acid Viscosupplementation and Physiotherapy. Appl. Sci. 2022, 12, 3165. [Google Scholar] [CrossRef]
- Diduch, D.R.; Insall, J.N.; Scott, W.N.; Scuderi, G.R.; Font-Rodriguez, D. Total knee replacement in young, active patients. Long-term follow-up and functional outcome. J. Bone Jt. Surg. Am. 1997, 79, 575–582. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Tan, Y.; Deng, Y.; Chen, L. Posterior cruciate-retaining versus posterior stabilized total knee arthroplasty: A meta-analysis of randomized controlled trials. Knee Surg. Sports Traumatol. Arthrosc. 2014, 22, 556–564. [Google Scholar] [CrossRef] [PubMed]
- Ritter, M.A.; Keating, E.M.; Sueyoshi, T.; Davis, K.E.; Barrington, J.W.; Emerson, R.H. Twenty-Five-Years and Greater, Results After Nonmodular Cemented Total Knee Arthroplasty. J. Arthroplast. 2016, 31, 2199–2202. [Google Scholar] [CrossRef] [PubMed]
- Grieco, T.F.; Sharma, A.; Dessinger, G.M.; Cates, H.E.; Komistek, R.D. In Vivo Kinematic Comparison of a Bicruciate Stabilized Total Knee Arthroplasty and the Normal Knee Using Fluoroscopy. J. Arthroplast. 2018, 33, 565–571. [Google Scholar] [CrossRef] [PubMed]
- Murakami, K.; Hamai, S.; Okazaki, K.; Gondo, H.; Wang, Y.; Ikebe, S.; Higaki, H.; Shimoto, T.; Mizu-Uchi, H.; Akasaki, Y.; et al. Knee kinematics in bi-cruciate stabilized total knee arthroplasty during squatting and stair-climbing activities. J. Orthop. 2018, 15, 650–654. [Google Scholar] [CrossRef]
- Bercik, M.J.; Joshi, A.; Parvizi, J. Posterior cruciate-retaining versus posterior-stabilized total knee arthroplasty: A meta-analysis. J. Arthroplast. 2013, 28, 439–444. [Google Scholar] [CrossRef]
- Kim, Y.H.; Choi, Y.; Kwon, O.R.; Kim, J.S. Functional outcome and range of motion of high-flexion posterior cruciate-retaining and high-flexion posterior cruciate-substituting total knee prostheses: A prospective, randomized study. J. Bone Jt. Surg. Am. 2009, 91, 753–760. [Google Scholar] [CrossRef]
- de Beer, J.; Petruccelli, D.; Adili, A.; Piccirillo, L.; Wismer, D.; Winemaker, M. Patient perspective survey of total hip vs total knee arthroplasty surgery. J. Arthroplast. 2012, 27, 865–869.e5. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, N.; Takeuchi, R.; Sawaguchi, T.; Ishikawa, H.; Saito, T.; Goldhahn, S. Cross-cultural adaptation and validation of the Japanese Knee Injury and Osteoarthritis Outcome Score (KOOS). J. Orthop. Sci. 2011, 16, 516–523. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, H.; Hanyu, T.; Sledge, C.B.; Lingard, E.A. Validation of a Japanese patient-derived outcome scale for assessing total knee arthroplasty: Comparison with Western Ontario and McMaster Universities osteoarthritis index (WOMAC). J. Orthop. Sci. 2003, 8, 288–293. [Google Scholar] [CrossRef] [PubMed]
- Noble, P.C.; Conditt, M.A.; Cook, K.F.; Mathis, K.B. The John Insall Award: Patient expectations affect satisfaction with total knee arthroplasty. Clin. Orthop. Relat. Res. 2006, 452, 35–43. [Google Scholar] [CrossRef] [PubMed]
- Nakahara, H.; Okazaki, K.; Mizu-Uchi, H.; Hamai, S.; Tashiro, Y.; Matsuda, S.; Iwamoto, Y. Correlations between patient satisfaction and ability to perform daily activities after total knee arthroplasty: Why aren’t patients satisfied? J. Orthop. Sci. 2015, 20, 87–92. [Google Scholar] [CrossRef] [PubMed]
- Nishio, Y.; Onodera, T.; Kasahara, Y.; Takahashi, D.; Iwasaki, N.; Majima, T. Intraoperative medial pivot affects deep knee flexion angle and patient-reported outcomes after total knee arthroplasty. J. Arthroplast. 2014, 29, 702–706. [Google Scholar] [CrossRef] [PubMed]
- Matsuda, S.; Kawahara, S.; Okazaki, K.; Tashiro, Y.; Iwamoto, Y. Postoperative alignment and ROM affect patient satisfaction after TKA. Clin. Orthop. Relat. Res. 2013, 471, 127–133. [Google Scholar] [CrossRef]
- Azukizawa, M.; Kuriyama, S.; Nakamura, S.; Nishitani, K.; Lyman, S.; Morita, Y.; Furu, M.; Ito, H.; Matsuda, S. Intraoperative medial joint laxity in flexion decreases patient satisfaction after total knee arthroplasty. Arch. Orthop. Trauma. Surg. 2018, 138, 1143–1150. [Google Scholar] [CrossRef]
- Van Onsem, S.; Verstraete, M.; Van Eenoo, W.; Van Der Straeten, C.; Victor, J. Are TKA Kinematics During Closed Kinetic Chain Exercises Associated with Patient-reported Outcomes? A Preliminary Analysis. Clin. Orthop. Relat. Res. 2020, 478, 255–263. [Google Scholar] [CrossRef]
- Brander, V.A.; Stulberg, S.D.; Adams, A.D.; Harden, R.N.; Bruehl, S.; Stanos, S.P.; Houle, T. Predicting total knee replacement pain: A prospective, observational study. Clin. Orthop. Relat. Res. 2003, 416, 27–36. [Google Scholar] [CrossRef]
- Koga, H.; Nakamae, A.; Shima, Y.; Iwasa, J.; Myklebust, G.; Engebretsen, L.; Bahr, R.; Krosshaug, T. Mechanisms for noncontact anterior cruciate ligament injuries: Knee joint kinematics in 10 injury situations from female team handball and basketball. Am. J. Sports Med. 2010, 38, 2218–2225. [Google Scholar] [CrossRef] [PubMed]
- Komistek, R.D.; Mahfouz, M.R.; Bertin, K.C.; Rosenberg, A.; Kennedy, W. In vivo determination of total knee arthroplasty kinematics: A multicenter analysis of an asymmetrical posterior cruciate retaining total knee arthroplasty. J. Arthroplast. 2008, 23, 41–50. [Google Scholar] [CrossRef] [PubMed]
- Hoff, W.A.; Komistek, R.D.; Dennis, D.A.; Gabriel, S.M.; Walker, S.A. Three-dimensional determination of femoral-tibial contact positions under in vivo conditions using fluoroscopy. Clin. Biomech. 1998, 13, 455–472. [Google Scholar] [CrossRef] [PubMed]
- Khan, H.; Walker, P.S.; Zuckerman, J.D.; Slover, J.; Jaffe, F.; Karia, R.J.; Kim, J.H. The potential of accelerometers in the evaluation of stability of total knee arthroplasty. J. Arthroplast. 2013, 28, 459–462. [Google Scholar] [CrossRef] [PubMed]
- Kadaba, M.P.; Ramakrishnan, H.K.; Wootten, M.E. Measurement of lower extremity kinematics during level walking. J. Orthop. Res. 1990, 8, 383–392. [Google Scholar] [CrossRef]
- Andriacchi, T.P.; Dyrby, C.O.; Johnson, T.S. The use of functional analysis in evaluating knee kinematics. Clin. Orthop. Relat. Res. 2003, 410, 44–53. [Google Scholar] [CrossRef]
- Hamada, D.; Wada, K.; Takasago, T.; Goto, T.; Nitta, A.; Higashino, K.; Fukui, Y.; Sairyo, K. Native rotational knee kinematics are lost in bicruciate-retaining total knee arthroplasty when the tibial component is replaced. Knee Surg. Sports Traumatol. Arthrosc. 2018, 26, 3249–3256. [Google Scholar] [CrossRef]
- Bellemans, J.; Banks, S.; Victor, J.; Vandenneucker, H.; Moemans, A. Fluoroscopic analysis of the kinematics of deep flexion in total knee arthroplasty: Influence of posterior condylar offset. J. Bone Jt. Surg. Br. 2002, 84, 50–53. [Google Scholar] [CrossRef]
- Dennis, D.A.; Komistek, R.D.; Mahfouz, M.R.; Haas, B.D.; Stiehl, J.B. Multicenter determination of in vivo kinematics after total knee arthroplasty. Clin. Orthop. Relat. Res. 2003, 416, 37–57. [Google Scholar] [CrossRef]
- Schache, A.G.; Blanch, P.D.; Rath, D.A.; Wrigley, T.V.; Starr, R.; Bennell, K.L. A comparison of overground and treadmill running for measuring the three-dimensional kinematics of the lumbo-pelvic-hip complex. Clin. Biomech. 2001, 16, 667–680. [Google Scholar] [CrossRef]
- Holden, J.P.; Selbie, W.S.; Stanhope, S.J. A proposed test to support the clinical movement analysis laboratory accreditation process. Gait Posture 2003, 17, 205–213. [Google Scholar] [CrossRef] [PubMed]
- Zeni, J.A., Jr.; Higginson, J.S. Differences in gait parameters between healthy subjects and persons with moderate and severe knee osteoarthritis: A result of altered walking speed? Clin. Biomech. 2009, 24, 372–378. [Google Scholar] [CrossRef] [PubMed]
- Kim, W.S.; Kim, E.Y. Comparing self-selected speed walking of the elderly with self-selected slow, moderate, and fast speed walking of young adults. Ann. Rehabil. Med. 2014, 38, 101–108. [Google Scholar] [CrossRef] [PubMed]
- Bolink, S.A.; Naisas, H.; Senden, R.; Essers, H.; Heyligers, I.C.; Meijer, K.; Grimm, B. Validity of an inertial measurement unit to assess pelvic orientation angles during gait, sit-stand transfers and step-up transfers: Comparison with an optoelectronic motion capture system. Med. Eng. Phys. 2016, 38, 225–231. [Google Scholar] [CrossRef] [PubMed]
- Nagura, T.; Otani, T.; Suda, Y.; Matsumoto, H.; Toyama, Y. Is high flexion following total knee arthroplasty safe?: Evaluation of knee joint loads in the patients during maximal flexion. J. Arthroplast. 2005, 20, 647–651. [Google Scholar] [CrossRef] [PubMed]
- McClelland, J.A.; Webster, K.E.; Feller, J.A. Gait analysis of patients following total knee replacement: A systematic review. Knee 2007, 14, 253–263. [Google Scholar] [CrossRef]
- Naili, J.E.; Brostrom, E.W.; Gutierrez-Farewik, E.M.; Schwartz, M.H. The centre of mass trajectory is a sensitive and responsive measure of functional compensations in individuals with knee osteoarthritis performing the five times sit-to-stand test. Gait Posture 2018, 62, 140–145. [Google Scholar] [CrossRef]
- Hubley-Kozey, C.L.; Hatfield, G.L.; Astephen Wilson, J.L.; Dunbar, M.J. Alterations in neuromuscular patterns between pre and one-year post-total knee arthroplasty. Clin. Biomech. 2010, 25, 995–1002. [Google Scholar] [CrossRef]
- Davidson, B.S.; Judd, D.L.; Thomas, A.C.; Mizner, R.L.; Eckhoff, D.G.; Stevens-Lapsley, J.E. Muscle activation and coactivation during five-time-sit-to-stand movement in patients undergoing total knee arthroplasty. J. Electromyogr. Kinesiol. 2013, 23, 1485–1493. [Google Scholar] [CrossRef]
- Lester, D.K.; Shantharam, R.; Zhang, K. Dynamic electromyography after cruciate-retaining total knee arthroplasty revealed a threefold quadriceps demand compared with the contralateral normal knee. J. Arthroplast. 2013, 28, 557–562. [Google Scholar] [CrossRef]
- Kajiwara, M.; Kanamori, A.; Kadone, H.; Endo, Y.; Kobayashi, Y.; Hyodo, K.; Takahashi, T.; Arai, N.; Taniguchi, Y.; Yoshioka, T.; et al. Knee biomechanics changes under dual task during single-leg drop landing. J. Exp. Orthop. 2019, 6, 5. [Google Scholar] [CrossRef] [PubMed]
- Rainoldi, A.; Melchiorri, G.; Caruso, I. A method for positioning electrodes during surface EMG recordings in lower limb muscles. J. Neurosci. Methods 2004, 134, 37–43. [Google Scholar] [CrossRef] [PubMed]
- Salvadore, G.; Meere, P.A.; Verstraete, M.A.; Victor, J.; Walker, P.S. Laxity and contact forces of total knee designed for anatomic motion: A cadaveric study. Knee 2018, 25, 650–656. [Google Scholar] [CrossRef] [PubMed]
- Farquhar, S.J.; Reisman, D.S.; Snyder-Mackler, L. Persistence of altered movement patterns during a sit-to-stand task 1 year following unilateral total knee arthroplasty. Phys. Ther. 2008, 88, 567–579. [Google Scholar] [CrossRef] [PubMed]
- Mizner, R.L.; Snyder-Mackler, L. Altered loading during walking and sit-to-stand is affected by quadriceps weakness after total knee arthroplasty. J. Orthop. Res. 2005, 23, 1083–1090. [Google Scholar] [CrossRef]
- Boonstra, M.C.; Schwering, P.J.; De Waal Malefijt, M.C.; Verdonschot, N. Sit-to-stand movement as a performance-based measure for patients with total knee arthroplasty. Phys. Ther. 2010, 90, 149–156. [Google Scholar] [CrossRef]
- Farquhar, S.J.; Kaufman, K.R.; Snyder-Mackler, L. Sit-to-stand 3 months after unilateral total knee arthroplasty: Comparison of self-selected and constrained conditions. Gait Posture 2009, 30, 187–191. [Google Scholar] [CrossRef][Green Version]
- Wang, H.; Simpson, K.J.; Ferrara, M.S.; Chamnongkich, S.; Kinsey, T.; Mahoney, O.M. Biomechanical differences exhibited during sit-to-stand between total knee arthroplasty designs of varying radii. J. Arthroplast. 2006, 21, 1193–1199. [Google Scholar] [CrossRef]
- Evangelista, P.J.; Laster, S.K.; Lenz, N.M.; Sheth, N.P.; Schwarzkopf, R. A Computer Model of Mid-Flexion Instability in a Balanced Total Knee Arthroplasty. J. Arthroplast. 2018, 33, S265–S269. [Google Scholar] [CrossRef]
- Victor, J.; Bellemans, J. Physiologic kinematics as a concept for better flexion in TKA. Clin. Orthop. Relat. Res. 2006, 452, 53–58. [Google Scholar] [CrossRef]
| Group I n = 10 | Group II n = 10 | p-Value | |
|---|---|---|---|
| Age (years) | 69.4 ± 5.3 | 70.3 ± 6.8 | 0.76 |
| Sex (male/female) | 2/8 | 3/7 | |
| Side (right/left) | 5/5 | 6/4 | |
| Height (m) | 1.54 ± 0.06 | 1.56 ± 0.08 | 0.56 |
| Body mass index (kg/m2) | 23.3 ± 3.9 | 25.0 ± 1.8 | 0.26 |
| Postoperative time (months) | 14.1 ± 6.8 | 16.7 ± 5.8 | 0.4 |
| ROM (°) | |||
| Flexion | 124.3 ± 9.7 | 126.5 ± 6.0 | 0.57 |
| Extension | −0.5 ± 1.0 | −0.9 ± 1.1 | 0.44 |
| Preoperative tibiofemoral X-ray axis (°) | 183.2 ± 4.6 | 179 ± 11.6 | 0.33 |
| Tibiofemoral X-ray axis (°) | 176.1 ± 1.6 | 175.3 ± 2.9 | 0.47 |
| KOOS | |||
| Symptoms | 83.9 ± 9.2 | 82.1 ± 8.5 | 0.67 |
| Pain | 86.1 ± 9.8 | 83.9 ± 14.9 | 0.71 |
| ADL | 88.8 ± 8.5 | 87.5 ± 8.5 | 0.74 |
| Sport | 67 ± 22.2 | 50.5 ± 19.9 | 0.11 |
| QOL | 76.3 ± 12.1 | 70.6 ± 14.5 | 0.38 |
| Group I | Group II | p-Value | Control * | |
|---|---|---|---|---|
| Sit-to-stand time (s) | 1.1 ± 0.3 | 1.2 ± 0.2 | 0.32 | 1.9 |
| Maximum Knee-Extension Angular Velocity (°/s) | 126.5 ± 31.7 | 116.4 ± 29.5 | 0.49 | 126.7 |
| Mean Knee-Extension Angular Velocity (°/s) | 66.7 ± 21.3 | 63.8 ± 14.9 | 0.74 | - |
| Maximum Hip-Flexion Angle (°) | 80.2 ± 11.1 | 81.7 ± 7.5 | 0.74 | 76.2 |
| Maximum Knee-Flexion Angle (°) | 69.3 ± 6.7 | 72.8 ± 7.6 | 0.32 | 88.7 |
| Maximum Ankle-Flexion Angle (°) | 10.8 ± 4.3 | 11.1 ± 4.9 | 0.86 | - |
| Group I | Group II | p-Value | Control * | |
|---|---|---|---|---|
| Maximum knee-extension moment (Nm/kg) | 0.37 ± 0.11 | 0.34 ± 0.12 | 0.59 | 0.42 |
| Maximum knee-valgus moment (Nm/kg) | 0.23 ± 0.18 | 0.16 ± 0.14 | 0.34 | - |
| Maximum knee-varus moment (Nm/kg) | 0.03 ± 0.06 | 0.04 ± 0.03 | 0.54 | - |
| Maximum hip-extension moment (Nm/kg) | 0.47 ± 0.18 | 0.67 ± 0.16 | 0.02 | 0.33 |
| Maximum vGRF (N/kg) | 5.5 ± 0.5 | 5.93 ± 0.4 | 0.06 | 5.95 |
| Mean vGRF (N/kg) | 4.06 ± 0.5 | 4.68 ± 0.4 | 0.01 | - |
| Group I | Group II | p-Value | Control * | |
|---|---|---|---|---|
| Vastus medialis | 56.8 ± 17.9 | 71.9 ± 26.0 | 0.17 | 84.9 |
| Rectus femoris | 27.2 ± 14.9 | 31.9 ± 9.8 | 0.44 | 76.4 |
| Vastus lateralis | 70.0 ± 25.3 | 77.4 ± 21.8 | 0.52 | 70.1 |
| Semitendinosus | 29.3 ± 18.0 | 24.2 ± 13.4 | 0.51 | 24.8 |
| Biceps femoris | 39.0 ± 21.7 | 32.9 ± 13.0 | 0.48 | 37.3 |
| Gluteus medius | 12.4 ± 8.6 | 12.9 ± 8.4 | 0.89 | - |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hyodo, K.; Kanamori, A.; Kadone, H.; Kajiwara, M.; Okuno, K.; Kikuchi, N.; Yamazaki, M. Evaluation of Sit-to-Stand Movement Focusing on Kinematics, Kinetics, and Muscle Activity after Modern Total Knee Arthroplasty. Appl. Sci. 2024, 14, 360. https://doi.org/10.3390/app14010360
Hyodo K, Kanamori A, Kadone H, Kajiwara M, Okuno K, Kikuchi N, Yamazaki M. Evaluation of Sit-to-Stand Movement Focusing on Kinematics, Kinetics, and Muscle Activity after Modern Total Knee Arthroplasty. Applied Sciences. 2024; 14(1):360. https://doi.org/10.3390/app14010360
Chicago/Turabian StyleHyodo, Kojiro, Akihiro Kanamori, Hideki Kadone, Masaya Kajiwara, Kosuke Okuno, Naoya Kikuchi, and Masashi Yamazaki. 2024. "Evaluation of Sit-to-Stand Movement Focusing on Kinematics, Kinetics, and Muscle Activity after Modern Total Knee Arthroplasty" Applied Sciences 14, no. 1: 360. https://doi.org/10.3390/app14010360
APA StyleHyodo, K., Kanamori, A., Kadone, H., Kajiwara, M., Okuno, K., Kikuchi, N., & Yamazaki, M. (2024). Evaluation of Sit-to-Stand Movement Focusing on Kinematics, Kinetics, and Muscle Activity after Modern Total Knee Arthroplasty. Applied Sciences, 14(1), 360. https://doi.org/10.3390/app14010360






