Influence of Hydrothermal Carbonization on Catalytic Fast Pyrolysis of Agricultural Biomass
Abstract
Featured Application
Abstract
1. Introduction
2. Materials and Methods
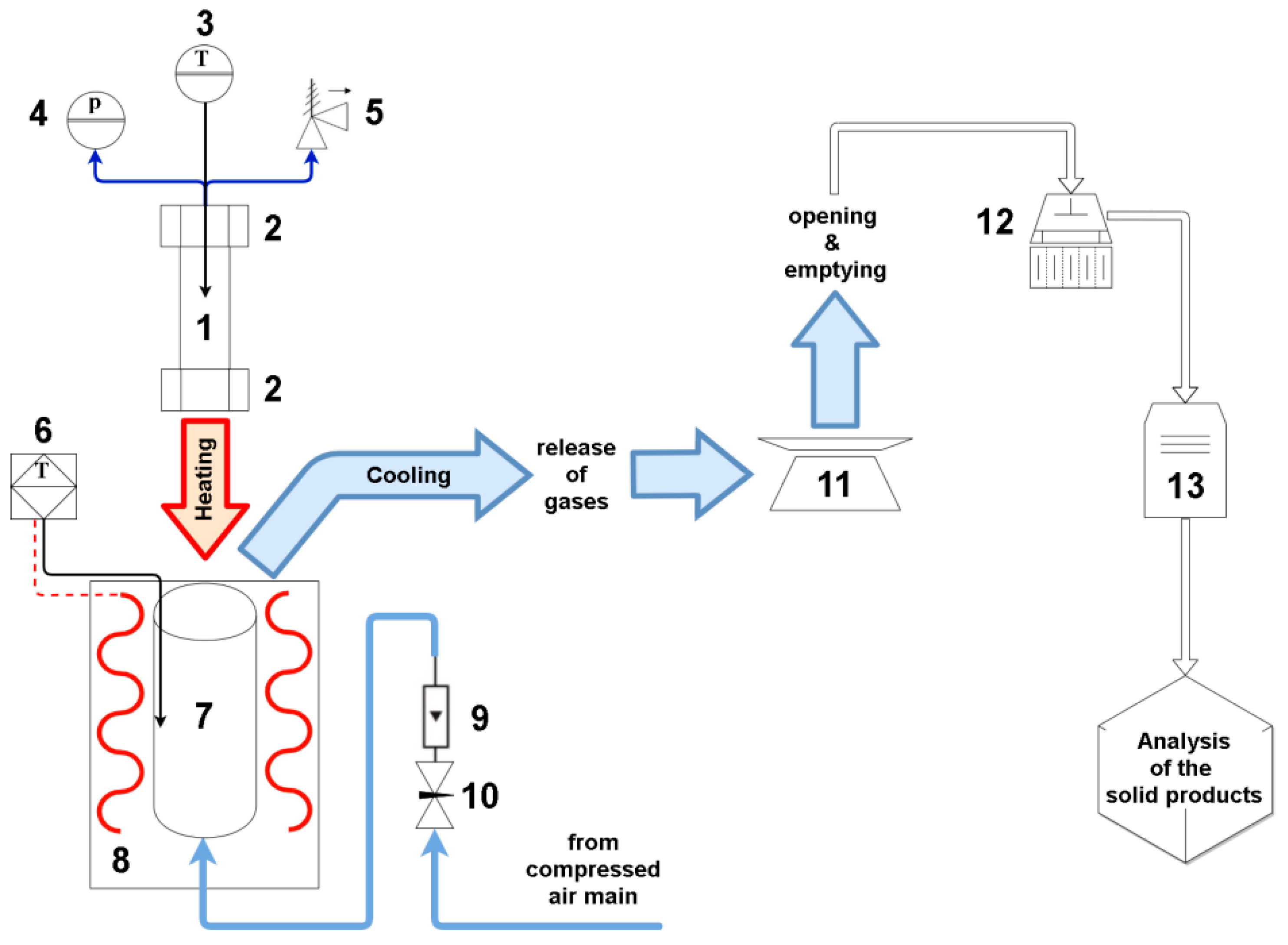
2.1. Feedstock and Hydrothermal Carbonization Experiments
2.2. Cyclonic TGA
2.3. Characterization of Feedstock and Products
2.4. Analyses and Calculations
3. Results and Discussion
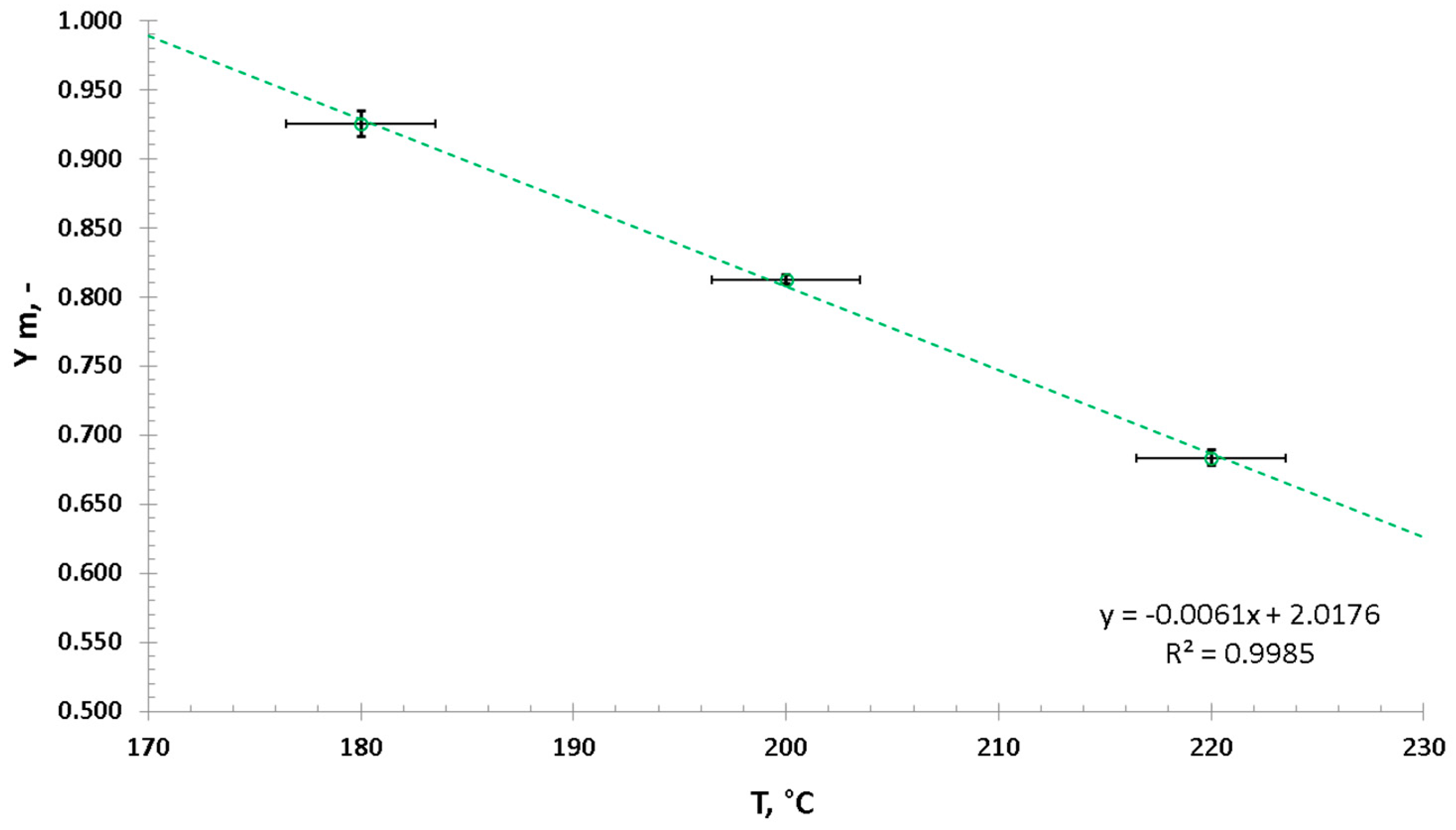
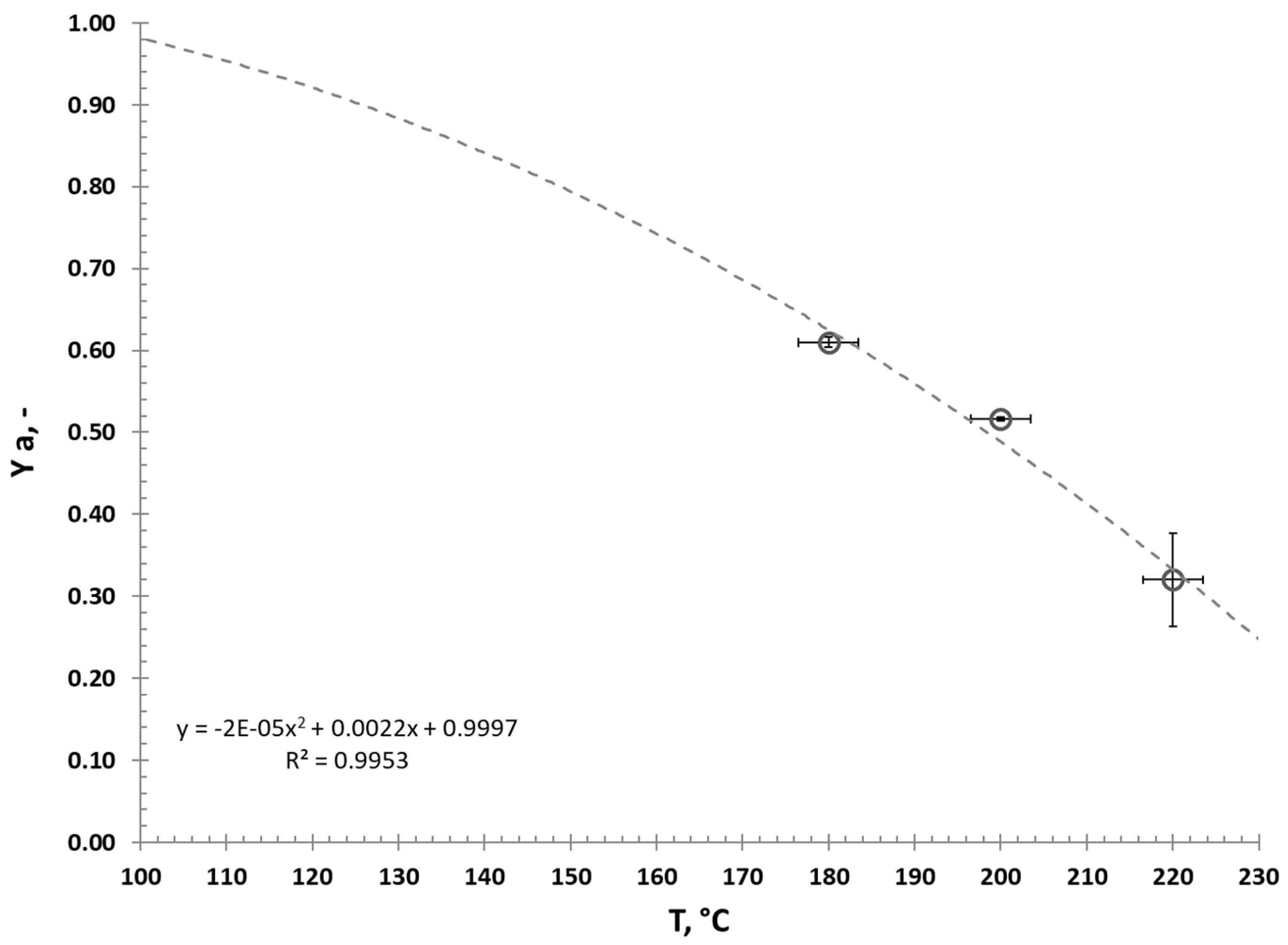
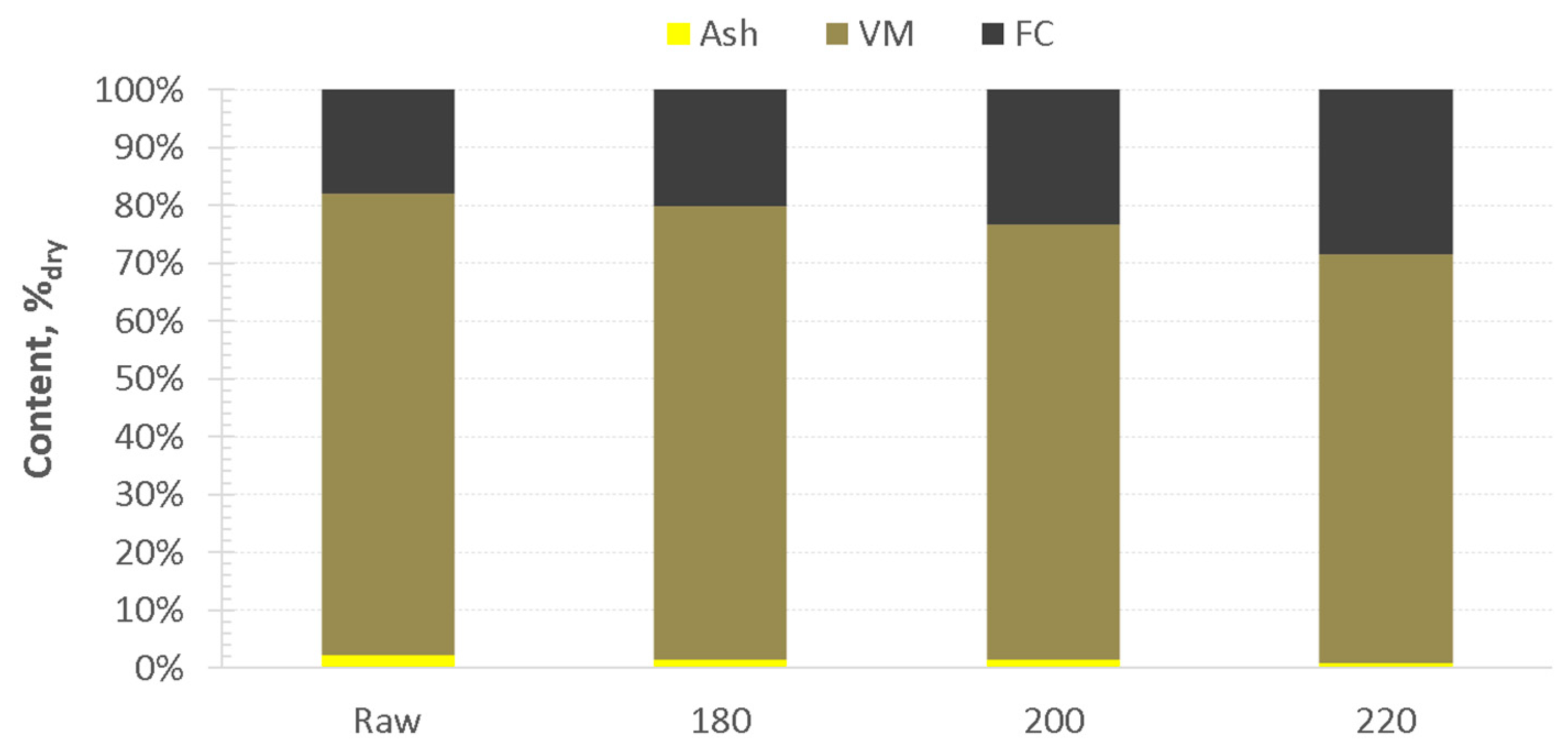
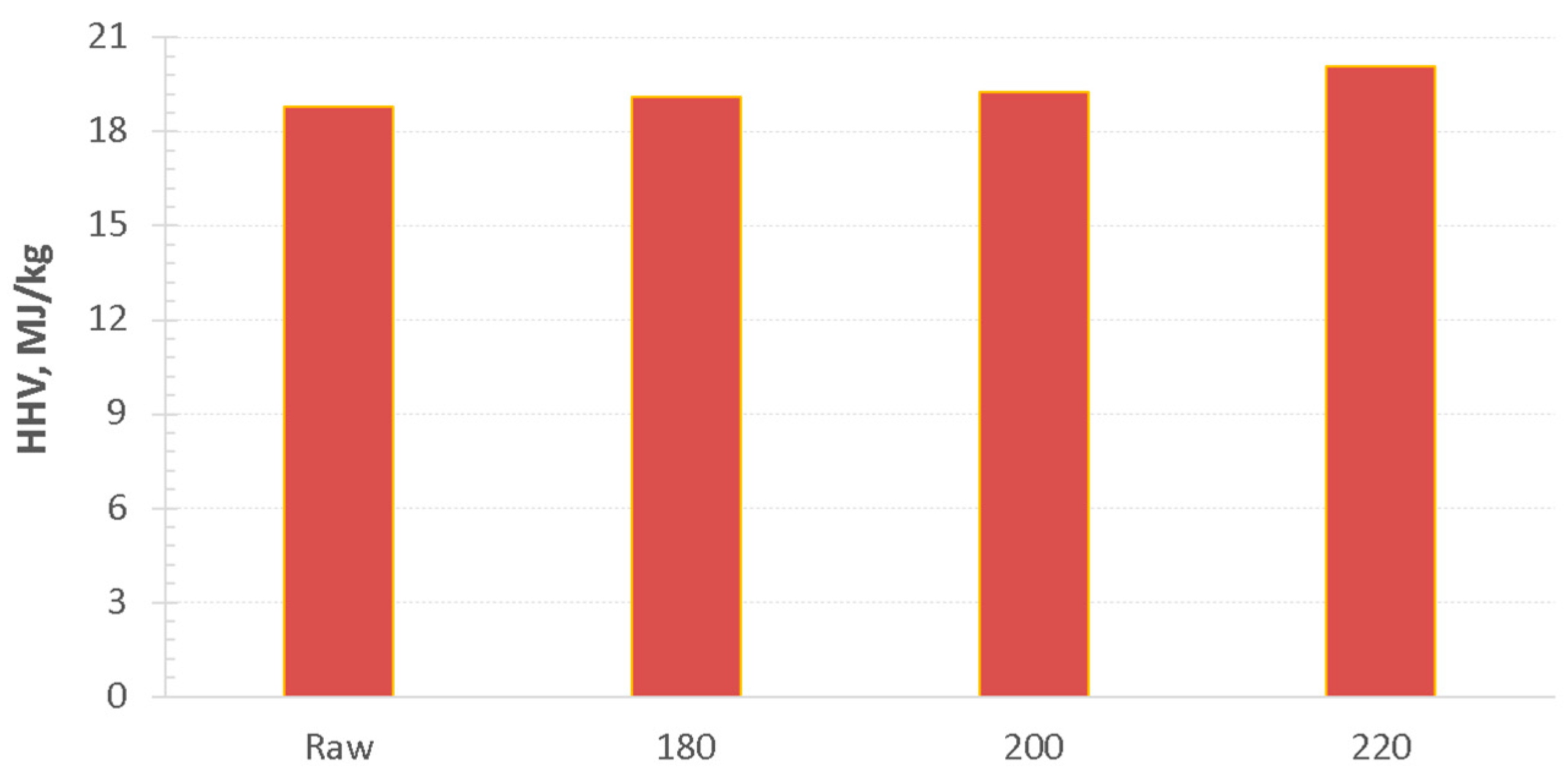
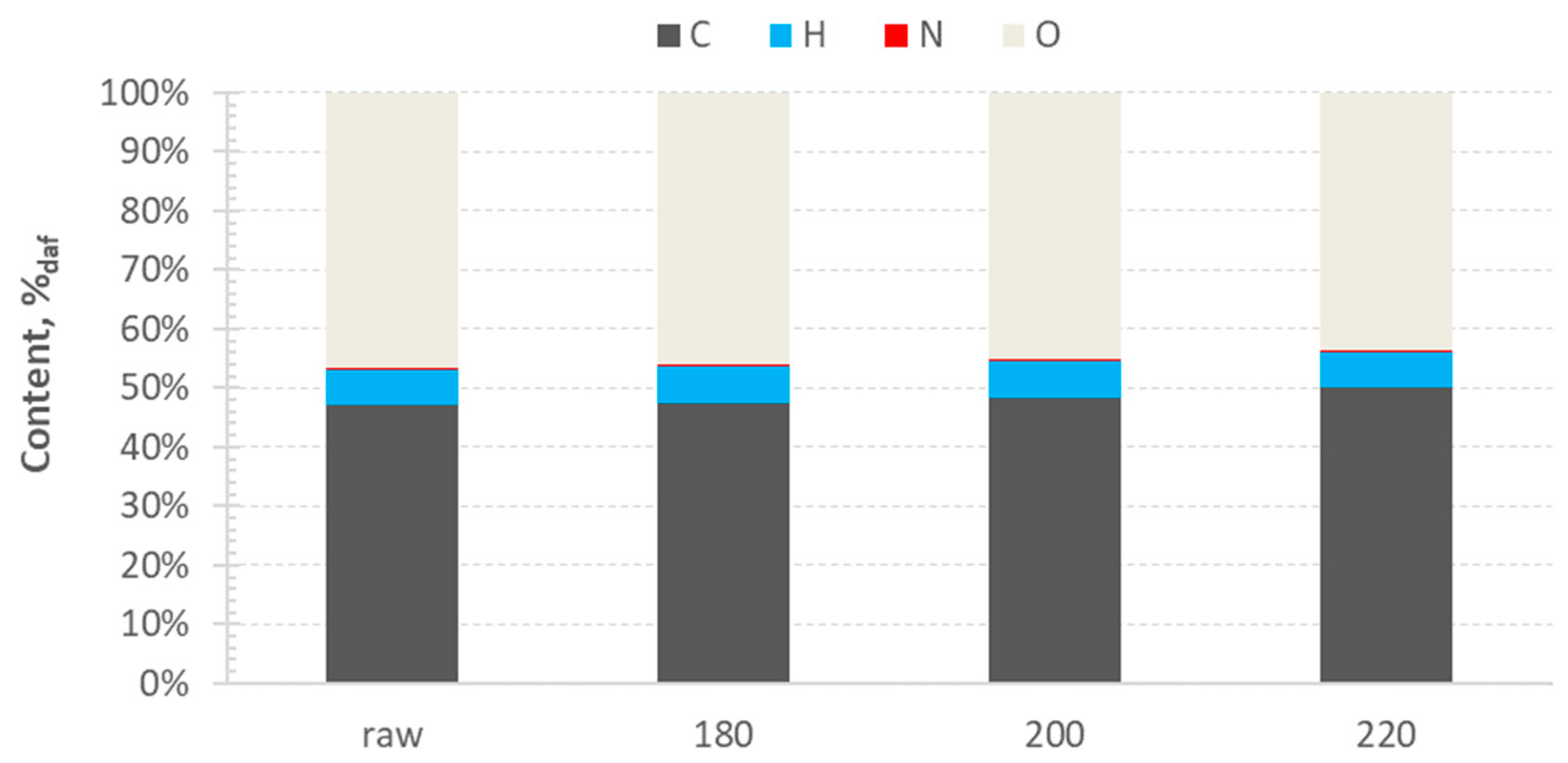
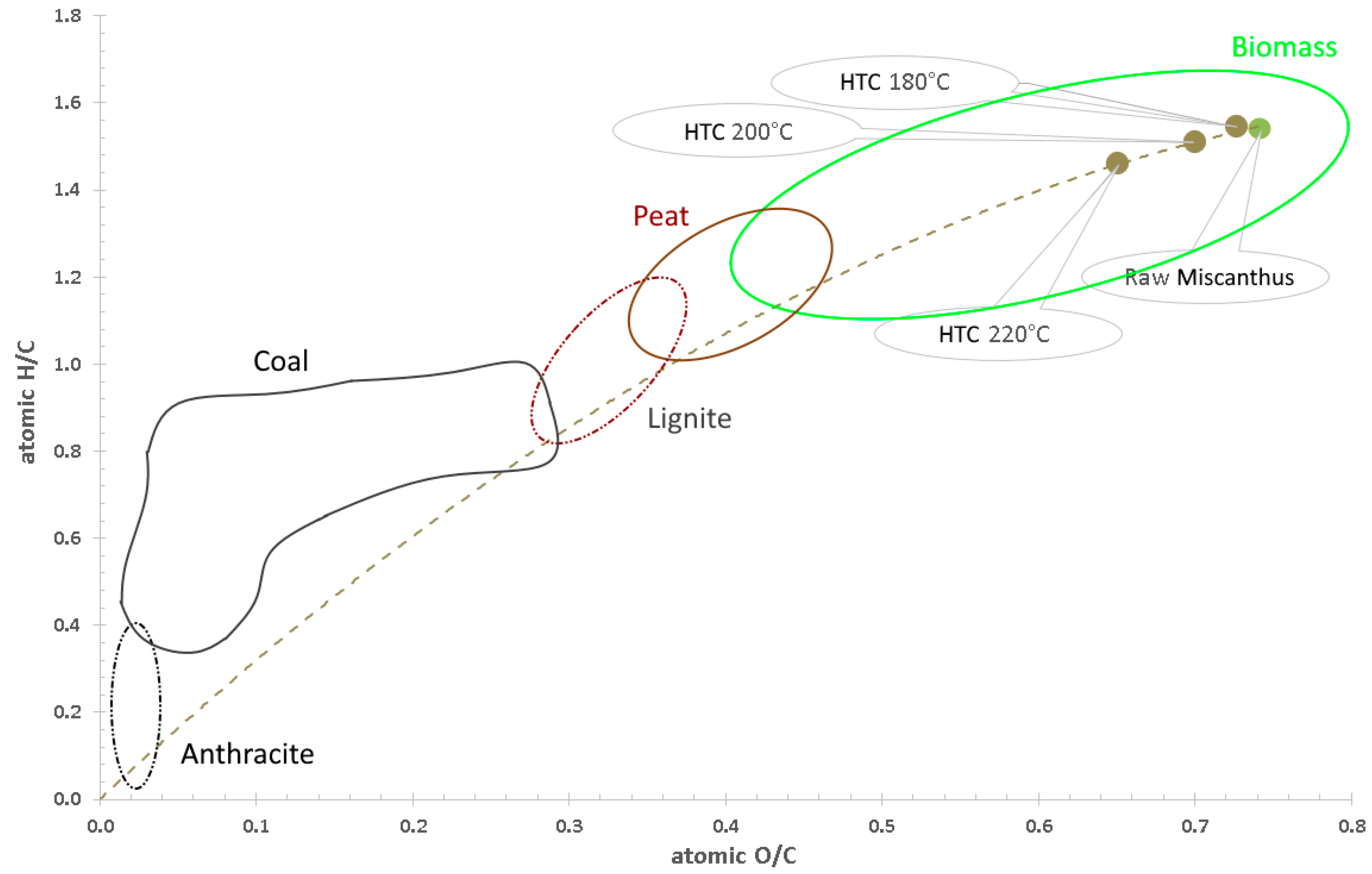
3.1. Hydrothermal Carbonization—Process Performance and Effect of HTC Treatment on Valorized Miscanthus
3.2. Fast Pyrolysis Kinetics—Influcence of HTC and CaO Addition
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Tzelepi, V.; Zeneli, M.; Kourkoumpas, D.S.; Karampinis, E.; Gypakis, A.; Nikolopoulos, N.; Grammelis, P. Biomass Availability in Europe as an Alternative Fuel for Full Conversion of Lignite Power Plants: A Critical Review. Energies 2020, 13, 3390. [Google Scholar] [CrossRef]
- Knapczyk, A.; Francik, S.; Jewiarz, M.; Zawiślak, A.; Francik, R. Thermal Treatment of Biomass: A Bibliometric Analysis—The Torrefaction Case. Energies 2020, 14, 162. [Google Scholar] [CrossRef]
- Szufa, S.; Piersa, P.; Junga, R.; Błaszczuk, A.; Modliński, N.; Sobek, S.; Marczak-Grzesik, M.; Adrian, Ł.; Dzikuć, M. Numerical Modeling of the Co-Firing Process of an in Situ Steam-Torrefied Biomass with Coal in a 230 MW Industrial-Scale Boiler. Energy 2023, 263, 125918. [Google Scholar] [CrossRef]
- Kopczyński, M.; Lasek, J.A.; Iluk, A.; Zuwała, J. The Co-Combustion of Hard Coal with Raw and Torrefied Biomasses (Willow (Salix Viminalis), Olive Oil Residue and Waste Wood from Furniture Manufacturing). Energy 2017, 140, 1316–1325. [Google Scholar] [CrossRef]
- Jian, J.; Lu, Z.; Yao, S.; Li, Y.; Liu, Z.; Lang, B.; Chen, Z. Effects of Thermal Conditions on Char Yield and Char Reactivity of Woody Biomass in Stepwise Pyrolysis. J. Anal. Appl. Pyrolysis 2019, 138, 211–217. [Google Scholar] [CrossRef]
- Luo, H.; Lu, Z.; Jensen, P.A.; Glarborg, P.; Lin, W.; Dam-Johansen, K.; Wu, H. Experimental and Modelling Study on the Influence of Wood Type, Density, Water Content, and Temperature on Wood Devolatilization. Fuel 2020, 260, 116410. [Google Scholar] [CrossRef]
- Wang, S.; Mandfloen, P.; Jönsson, P.; Yang, W. Synergistic Effects in the Copyrolysis of Municipal Sewage Sludge Digestate and Salix: Reaction Mechanism, Product Characterization and Char Stability. Appl. Energy 2021, 289, 116687. [Google Scholar] [CrossRef]
- Zaini, I.N.; Wen, Y.; Mousa, E.; Jönsson, P.G.; Yang, W. Primary Fragmentation Behavior of Refuse Derived Fuel Pellets during Rapid Pyrolysis. Fuel Process. Technol. 2021, 216, 106796. [Google Scholar] [CrossRef]
- Kantorek, M.; Jesionek, K.; Polesek-Karczewska, S.; Ziółkowski, P.; Stajnke, M.; Badur, J. Pilot Installation for Thermal Utilization of Meat-and-Bone Meal Using the Rotary Kiln Pyrolyzer and the Fluidised Bed Boiler. E3S Web Conf. 2019, 137, 01013. [Google Scholar] [CrossRef]
- Kantorek, M.; Jesionek, K.; Polesek-Karczewska, S.; Ziółkowski, P.; Stajnke, M.; Badur, J. Thermal Utilization of Meat-and-Bone Meal Using the Rotary Kiln Pyrolyzer and the Fluidized Bed Boiler—The Performance of Pilot-Scale Installation. Renew. Energy 2021, 164, 1447–1456. [Google Scholar] [CrossRef]
- Čespiva, J.; Jadlovec, M.; Výtisk, J.; Serenčíšová, J.; Tadeáš, O.; Honus, S. Softwood and Solid Recovered Fuel Gasification Residual Chars as Sorbents for Flue Gas Mercury Capture. Environ. Technol. Innov. 2023, 29, 102970. [Google Scholar] [CrossRef]
- Szul, M.; Iluk, T.; Zuwała, J. Use of CO2 in Pressurized, Fluidized Bed Gasification of Waste Biomasses. Energies 2022, 15, 1395. [Google Scholar] [CrossRef]
- Sieradzka, M.; Mlonka-Mędrala, A.; Magdziarz, A. Comprehensive Investigation of the CO2 Gasification Process of Biomass Wastes Using TG-MS and Lab-Scale Experimental Research. Fuel 2022, 330, 125566. [Google Scholar] [CrossRef]
- Szufa, S.; Dzikuć, M.; Adrian, Ł.; Piersa, P.; Romanowska-Duda, Z.; Lewandowska, W.; Marcza, M.; Błaszczuk, A.; Piwowar, A. Torrefaction of Oat Straw to Use as Solid Biofuel, an Additive to Organic Fertilizers for Agriculture Purposes and Activated Carbon—TGA Analysis, Kinetics. E3S Web Conf. 2020, 154, 02004. [Google Scholar] [CrossRef]
- Kerdsuwan, S.; Laohalidanond, K.; Gupta Ashwani, K. Upgrading Refuse-Derived Fuel Properties from Reclaimed Landfill Using Torrefaction. J. Energy Resour. Technol. 2021, 143, 021302. [Google Scholar] [CrossRef]
- Piersa, P.; Szufa, S.; Czerwińska, J.; Ünyay, H.; Adrian, Ł.; Wielgosinski, G.; Obraniak, A.; Lewandowska, W.; Marczak-Grzesik, M.; Dzikuć, M.; et al. Pine Wood and Sewage Sludge Torrefaction Process for Production Renewable Solid Biofuels and Biochar as Carbon Carrier for Fertilizers. Energies 2021, 14, 8176. [Google Scholar] [CrossRef]
- Lasek, J.A.; Kopczyński, M.; Janusz, M.; Iluk, A.; Zuwała, J. Combustion Properties of Torrefied Biomass Obtained from Flue Gas-Enhanced Reactor. Energy 2017, 119, 362–368. [Google Scholar] [CrossRef]
- Piersa, P.; Unyay, H.; Szufa, S.; Lewandowska, W.; Modrzewski, R.; Ślężak, R.; Ledakowicz, S. An Extensive Review and Comparison of Modern Biomass Torrefaction Reactors vs. Biomass Pyrolysis—Part 1. Energies 2022, 15, 2227. [Google Scholar] [CrossRef]
- Wen, Y.; Wang, S.; Shi, Z.; Nuran Zaini, I.; Niedzwiecki, L.; Aragon-Briceno, C.; Tang, C.; Pawlak-Kruczek, H.; Jönsson, P.G.; Yang, W. H2-Rich Syngas Production from Pyrolysis of Agricultural Waste Digestate Coupled with the Hydrothermal Carbonization Process. Energy Convers. Manag. 2022, 269, 116101. [Google Scholar] [CrossRef]
- Wang, S.; Wen, Y.; Shi, Z.; Niedzwiecki, L.; Baranowski, M.; Czerep, M.; Mu, W.; Kruczek, H.P.; Jönsson, P.G.; Yang, W. Effect of Hydrothermal Carbonization Pretreatment on the Pyrolysis Behavior of the Digestate of Agricultural Waste: A View on Kinetics and Thermodynamics. Chem. Eng. J. 2022, 431, 133881. [Google Scholar] [CrossRef]
- Jahirul, M.I.; Rasul, M.G.; Chowdhury, A.A.; Ashwath, N. Biofuels Production through Biomass Pyrolysis—A Technological Review. Energies 2012, 5, 4952–5001. [Google Scholar] [CrossRef]
- Mysore Prabhakara, H.; Bramer, E.A.; Brem, G. Role of Dolomite as an In-Situ CO2 Sorbent and Deoxygenation Catalyst in Fast Pyrolysis of Beechwood in a Bench Scale Fluidized Bed Reactor. Fuel Process. Technol. 2021, 224, 107029. [Google Scholar] [CrossRef]
- Bridgwater, A.V. Review of Fast Pyrolysis of Biomass and Product Upgrading. Biomass Bioenergy 2012, 38, 68–94. [Google Scholar] [CrossRef]
- Ryšavý, J.; Horák, J.; Hopan, F.; Kuboňová, L.; Krpec, K.; Molchanov, O.; Garba, M.; Ochodek, T. Influence of Flue Gas Parameters on Conversion Rates of Honeycomb Catalysts. Sep. Purif. Technol. 2021, 278, 119491. [Google Scholar] [CrossRef]
- Čespiva, J.; Skřínský, J.; Vereš, J.; Borovec, K.; Wnukowski, M. Solid-Recovered Fuel to Liquid Conversion Using Fixed Bed Gasification Technology and a Fischer–Tropsch Synthesis Unit—Case Study. Int. J. Energy Prod. Manag. 2020, 5, 212–222. [Google Scholar] [CrossRef]
- Li, H.; Wang, Y.; Zhou, N.; Dai, L.; Deng, W.; Liu, C.; Cheng, Y.; Liu, Y.; Cobb, K.; Chen, P.; et al. Applications of Calcium Oxide–Based Catalysts in Biomass Pyrolysis/Gasification—A Review. J. Clean. Prod. 2021, 291, 125826. [Google Scholar] [CrossRef]
- Yang, S.; Zhang, X.; Chen, L.; Sun, L.; Xie, X.; Zhao, B. Production of Syngas from Pyrolysis of Biomass Using Fe/CaO Catalysts: Effect of Operating Conditions on the Process. J. Anal. Appl. Pyrolysis 2017, 125, 1–8. [Google Scholar] [CrossRef]
- Sun, L.; Zhang, X.; Chen, L.; Xie, X.; Yang, S.; Zhao, B.; Si, H. Effect of Preparation Method on Structure Characteristics and Fast Pyrolysis of Biomass with Fe/CaO Catalysts. J. Anal. Appl. Pyrolysis 2015, 116, 183–189. [Google Scholar] [CrossRef]
- Vichaphund, S.; Sricharoenchaikul, V.; Atong, D. Industrial Waste Derived CaO-Based Catalysts for Upgrading Volatiles during Pyrolysis of Jatropha Residues. J. Anal. Appl. Pyrolysis 2017, 124, 568–575. [Google Scholar] [CrossRef]
- Echresh Zadeh, Z.; Abdulkhani, A.; Saha, B. A Comparative Production and Characterisation of Fast Pyrolysis Bio-Oil from Populus and Spruce Woods. Energy 2021, 214, 118930. [Google Scholar] [CrossRef]
- Funke, A.; Ziegler, F. Hydrothermal Carbonisation of Biomass: A Summary and Discussion of Chemical Mechanisms for Process Engineering. Biofuels Bioprod. Biorefining 2010, 4, 160–177. [Google Scholar] [CrossRef]
- Moscicki, K.J.; Niedzwiecki, L.; Owczarek, P.; Wnukowski, M. Commoditization of Wet and High Ash Biomass: Wet Torrefaction—A Review. J. Power Technol. 2017, 97, 354–369. [Google Scholar]
- Jackowski, M.; Semba, D.; Trusek, A.; Wnukowski, M.; Niedzwiecki, L.; Baranowski, M.; Krochmalny, K.; Pawlak-Kruczek, H. Hydrothermal Carbonization of Brewery’s Spent Grains for the Production of Solid Biofuels. Beverages 2019, 5, 12. [Google Scholar] [CrossRef]
- Peterson, A.A.; Vogel, F.; Lachance, R.P.; Fröling, M.; Antal, M.J.; Tester, J.W. Thermochemical Biofuel Production in Hydrothermal Media: A Review of Sub- and Supercritical Water Technologies. Energy Environ. Sci. 2008, 1, 32–65. [Google Scholar] [CrossRef]
- Reza, M.T.; Andert, J.; Wirth, B.; Busch, D.; Pielert, J.; Lynam, J.G.; Mumme, J. Hydrothermal Carbonization of Biomass for Energy and Crop Production. Appl. Bioenergy 2014, 1, 11–29. [Google Scholar] [CrossRef]
- Picone, A.; Volpe, M.; Messineo, A. Process Water Recirculation during Hydrothermal Carbonization of Waste Biomass: Current Knowledge and Challenges. Energies 2021, 14, 2962. [Google Scholar] [CrossRef]
- Calucci, L.; Forte, C. Influence of Process Parameters on the Hydrothermal Carbonization of Olive Tree Trimmings: A 13C Solid-State NMR Study. Appl. Sci. 2023, 13, 1515. [Google Scholar] [CrossRef]
- Satira, A.; Paone, E.; Bressi, V.; Iannazzo, D.; Marra, F.; Calabrò, P.S.; Mauriello, F.; Espro, C. Hydrothermal Carbonization as Sustainable Process for the Complete Upgrading of Orange Peel Waste into Value-Added Chemicals and Bio-Carbon Materials. Appl. Sci. 2021, 11, 10983. [Google Scholar] [CrossRef]
- González, R.; Ellacuriaga, M.; Aguilar-Pesantes, A.; Carrillo-Peña, D.; García-Cascallana, J.; Smith, R.; Gómez, X. Feasibility of Coupling Anaerobic Digestion and Hydrothermal Carbonization: Analyzing Thermal Demand. Appl. Sci. 2021, 11, 11660. [Google Scholar] [CrossRef]
- Louwes, A.C.; Basile, L.; Yukananto, R.; Bhagwandas, J.C.; Bramer, E.A.; Brem, G. Torrefied Biomass as Feed for Fast Pyrolysis: An Experimental Study and Chain Analysis. Biomass Bioenergy 2017, 105, 116–126. [Google Scholar] [CrossRef]
- Sieradzka, M.; Gao, N.; Quan, C.; Mlonka-Mędrala, A.; Magdziarz, A. Biomass Thermochemical Conversion via Pyrolysis with Integrated CO2 Capture. Energies 2020, 13, 1050. [Google Scholar] [CrossRef]
- Kantorek, M.; Jesionek, K.; Polesek-Karczewska, S.; Ziółkowski, P.; Badur, J. Thermal Utilization of Meat and Bone Meals. Performance Analysis in Terms of Drying Process, Pyrolysis and Kinetics of Volatiles Combustion. Fuel 2019, 254, 115548. [Google Scholar] [CrossRef]
- Wang, S.; Yang, H.; Shi, Z.; Zaini, I.N.; Wen, Y.; Jiang, J.; Jönsson, P.G.; Yang, W. Renewable Hydrogen Production from the Organic Fraction of Municipal Solid Waste through a Novel Carbon-Negative Process Concept. Energy 2022, 252, 124056. [Google Scholar] [CrossRef]
- Magdziarz, A.; Wilk, M.; Wądrzyk, M. Pyrolysis of Hydrochar Derived from Biomass—Experimental Investigation. Fuel 2020, 267, 117246. [Google Scholar] [CrossRef]
- Duan, Y.; Ning, Y.; Gao, N.; Quan, C.; Grammelis, P.; Boutikos, P. Effect of Hydrothermal Process on the Pyrolysis of Oily Sludge: Characterization and Analysis of Pyrolysis Products. Fuel 2023, 338, 127347. [Google Scholar] [CrossRef]
- Acharjee, T.C.; Coronella, C.J.; Vasquez, V.R. Effect of Thermal Pretreatment on Equilibrium Moisture Content of Lignocellulosic Biomass. Bioresour. Technol. 2011, 102, 4849–4854. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, M.; Andreottola, G.; Elagroudy, S.; Negm, M.S.; Fiori, L. Coupling Hydrothermal Carbonization and Anaerobic Digestion for Sewage Digestate Management: Influence of Hydrothermal Treatment Time on Dewaterability and Bio-Methane Production. J. Environ. Manag. 2021, 281, 111910. [Google Scholar] [CrossRef] [PubMed]
- Gao, N.; Li, Z.; Quan, C.; Miskolczi, N.; Egedy, A. A New Method Combining Hydrothermal Carbonization and Mechanical Compression In-Situ for Sewage Sludge Dewatering: Bench-Scale Verification. J. Anal. Appl. Pyrolysis 2019, 139, 187–195. [Google Scholar] [CrossRef]
- Wang, L.-F.; Qian, C.; Jiang, J.-K.; Ye, X.-D.; Yu, H.-Q. Response of Extracellular Polymeric Substances to Thermal Treatment in Sludge Dewatering Process. Environ. Pollut. 2017, 231, 1388–1392. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Zhang, L.; Li, A. Hydrothermal Treatment Coupled with Mechanical Expression at Increased Temperature for Excess Sludge Dewatering: Influence of Operating Conditions and the Process Energetics. Water Res. 2014, 65, 85–97. [Google Scholar] [CrossRef]
- Ruiz, H.A.; Rodríguez-Jasso, R.M.; Fernandes, B.D.; Vicente, A.A.; Teixeira, J.A. Hydrothermal Processing, as an Alternative for Upgrading Agriculture Residues and Marine Biomass according to the Biorefinery Concept: A Review. Renew. Sustain. Energy Rev. 2013, 21, 35–51. [Google Scholar] [CrossRef]
- Sobek, S.; Tran, Q.-K.; Junga, R.; Werle, S. Hydrothermal Carbonization of the Waste Straw: A Study of the Biomass Transient Heating Behavior and Solid Products Combustion Kinetics. Fuel 2021, 314, 122725. [Google Scholar] [CrossRef]
- Czerwińska, K.; Śliz, M.; Wilk, M. Hydrothermal Carbonization Process: Fundamentals, Main Parameter Characteristics and Possible Applications Including an Effective Method of SARS-CoV-2 Mitigation in Sewage Sludge. A Review. Renew. Sustain. Energy Rev. 2022, 154, 111873. [Google Scholar] [CrossRef]
- Hansen, L.J.; Fendt, S.; Spliethoff, H. Impact of Hydrothermal Carbonization on Combustion Properties of Residual Biomass. Biomass Convers. Biorefinery 2022, 12, 2541–2552. [Google Scholar] [CrossRef]
- Smith, A.M.; Singh, S.; Ross, A.B. Fate of Inorganic Material during Hydrothermal Carbonisation of Biomass: Influence of Feedstock on Combustion Behaviour of Hydrochar. Fuel 2016, 169, 135–145. [Google Scholar] [CrossRef]
- Śliz, M.; Tuci, F.; Czerwińska, K.; Fabrizi, S.; Lombardi, L.; Wilk, M. Hydrothermal Carbonization of the Wet Fraction from Mixed Municipal Solid Waste: Hydrochar Characteristics and Energy Balance. Waste Manag. 2022, 151, 39–48. [Google Scholar] [CrossRef] [PubMed]
- Śliz, M.; Czerwińska, K.; Magdziarz, A.; Lombardi, L.; Wilk, M. Hydrothermal Carbonization of the Wet Fraction from Mixed Municipal Solid Waste: A Fuel and Structural Analysis of Hydrochars. Energies 2022, 15, 6708. [Google Scholar] [CrossRef]
- Purnomo, C.; Castello, D.; Fiori, L. Granular Activated Carbon from Grape Seeds Hydrothermal Char. Appl. Sci. 2018, 8, 331. [Google Scholar] [CrossRef]
- Chung, J.; Edewi, O.; Foppen, J.; Gerner, G.; Krebs, R.; Lens, P. Removal of Escherichia Coli by Intermittent Operation of Saturated Sand Columns Supplemented with Hydrochar Derived from Sewage Sludge. Appl. Sci. 2017, 7, 839. [Google Scholar] [CrossRef]
- Scrinzi, D.; Andreottola, G.; Fiori, L. Composting Hydrochar-OFMSW Digestate Mixtures: Design of Bioreactors and Preliminary Experimental Results. Appl. Sci. 2021, 11, 1496. [Google Scholar] [CrossRef]
- Adjuik, T.; Rodjom, A.M.; Miller, K.E.; Reza, M.T.M.; Davis, S.C. Application of Hydrochar, Digestate, and Synthetic Fertilizer to a Miscanthus X Giganteus Crop: Implications for Biomass and Greenhouse Gas Emissions. Appl. Sci. 2020, 10, 8953. [Google Scholar] [CrossRef]
- Luo, H.; Wang, X.; Liu, X.; Wu, X.; Shi, X.; Xiong, Q. A Review on CFD Simulation of Biomass Pyrolysis in Fluidized Bed Reactors with Emphasis on Particle-Scale Models. J. Anal. Appl. Pyrolysis 2022, 162, 105433. [Google Scholar] [CrossRef]
- Luo, H.; Liu, X.; Niedzwiecki, L.; Wu, X.; Lin, W.; Lu, B.; Wang, W.; Wu, H. Analysis of Model Dimensionality, Particle Shrinkage, Boundary Layer Reactions on Particle-Scale Modelling of Biomass Char Conversion under Pulverized Fuel Combustion Conditions. Proc. Combust. Inst. 2022. [Google Scholar] [CrossRef]
- Luo, H.; Wang, X.; Krochmalny, K.; Niedzwiecki, L.; Czajka, K.; Pawlak-Kruczek, H.; Wu, X.; Liu, X.; Xiong, Q. Assessments and Analysis of Lumped and Detailed Pyrolysis Kinetics for Biomass Torrefaction with Particle-Scale Modeling. Biomass Bioenergy 2022, 166, 106619. [Google Scholar] [CrossRef]
- Wang, S.; Persson, H.; Yang, W.; Jönsson, P.G. Pyrolysis Study of Hydrothermal Carbonization-Treated Digested Sewage Sludge Using a Py-GC/MS and a Bench-Scale Pyrolyzer. Fuel 2020, 262, 116335. [Google Scholar] [CrossRef]
- Liu, Y.; Zhai, Y.; Li, S.; Liu, X.; Liu, X.; Wang, B.; Qiu, Z.; Li, C. Production of Bio-Oil with Low Oxygen and Nitrogen Contents by Combined Hydrothermal Pretreatment and Pyrolysis of Sewage Sludge. Energy 2020, 203, 117829. [Google Scholar] [CrossRef]
- Wang, S.; Wen, Y.; Hammarström, H.; Jönsson, P.G.; Yang, W. Pyrolysis Behaviour, Kinetics and Thermodynamic Data of Hydrothermal Carbonization–Treated Pulp and Paper Mill Sludge. Renew. Energy 2021, 177, 1282–1292. [Google Scholar] [CrossRef]
- Czajka, K.; Kisiela, A.; Moroń, W.; Ferens, W.; Rybak, W. Pyrolysis of Solid Fuels: Thermochemical Behaviour, Kinetics and Compensation Effect. Fuel Process. Technol. 2016, 142, 42–53. [Google Scholar] [CrossRef]
- Czajka, K.M. The Impact of the Thermal Lag on the Interpretation of Cellulose Pyrolysis. Energy 2021, 236, 121497. [Google Scholar] [CrossRef]
- Sobek, S.; Werle, S. Solar Pyrolysis of Waste Biomass: Part 2 Kinetic Modeling and Methodology of the Determination of the Kinetic Parameters for Solar Pyrolysis of Sewage Sludge. Renew. Energy 2020, 153, 962–974. [Google Scholar] [CrossRef]
- Dudziak, M.; Werle, S.; Marszałek, A.; Sobek, S.; Magdziarz, A. Comparative Assessment of the Biomass Solar Pyrolysis Biochars Combustion Behavior and Zinc Zn(II) Adsorption. Energy 2022, 261, 125360. [Google Scholar] [CrossRef]
- Louwes, A.C.; Halfwerk, R.B.; Bramer, E.A.; Brem, G. Experimental Study on Fast Pyrolysis of Raw and Torrefied Woody Biomass. Energy Technol. 2020, 8, 1900799. [Google Scholar] [CrossRef]
- Brem, G.; Bramer, E.A. PyRos: A New Flash Pyrolysis Technology for the Production of Bio-Oil from Biomass Residues. Bioenergy Outlook 2007, 25–27. [Google Scholar]
- Meier, D.; Van De Beld, B.; Bridgwater, A.V.; Elliott, D.C.; Oasmaa, A.; Preto, F. State-of-the-Art of Fast Pyrolysis in IEA Bioenergy Member Countries. Renew. Sustain. Energy Rev. 2013, 20, 619–641. [Google Scholar] [CrossRef]
- Plis, A.; Lasek, J.; Skawińska, A.; Zuwała, J. Thermochemical and Kinetic Analysis of the Pyrolysis Process in Cladophora Glomerata Algae. J. Anal. Appl. Pyrolysis 2015, 115, 166–174. [Google Scholar] [CrossRef]
- Pawlak-Kruczek, H.; Krochmalny, K.; Mościcki, K.; Zgóra, J.; Czerep, M.; Ostrycharczyk, M.; Niedźwiecki, Ł. Torrefaction of Various Types of Biomass in Laboratory Scale, Batch-Wise Isothermal Rotary Reactor and Pilot Scale, Continuous Multi-Stage Tape Reactor. Eng. Prot. Environ. 2017, 20, 457–472. [Google Scholar] [CrossRef]
- Pawlak-Kruczek, H.; Krochmalny, K.; Wnukowski, M.; Niedzwiecki, L. Slow Pyrolysis of the Sewage Sludge with Additives: Calcium Oxide and Lignite. J. Energy Resour. Technol. 2018, 140, 062206. [Google Scholar] [CrossRef]
- Moscicki, K.J.; Niedzwiecki, L.; Owczarek, P.; Wnukowski, M. Commoditization of Biomass: Dry Torrefaction and Pelletization—A Review. J. Power Technol. 2014, 94, 233–249. [Google Scholar]
- Poudel, J.; Karki, S.; Gu, J.H.; Lim, Y.; Oh, S.C. Effect of Co-Torrefaction on the Properties of Sewage Sludge and Waste Wood to Enhance Solid Fuel Qualities. J. Residuals Sci. Technol. 2017, 14, 23–36. [Google Scholar] [CrossRef]
- Pulka, J.; Wiśniewski, D.; Gołaszewski, J.; Białowiec, A. Is the Biochar Produced from Sewage Sludge a Good Quality Solid Fuel? Arch. Environ. Prot. 2016, 42, 125–134. [Google Scholar] [CrossRef]
- Mihajlović, M.; Petrović, J.; Maletić, S.; Isakovski, M.K.; Stojanović, M.; Lopičić, Z.; Trifunović, S. Hydrothermal Carbonization of Miscanthus × giganteus: Structural and Fuel Properties of Hydrochars and Organic Profile with the Ecotoxicological Assessment of the Liquid Phase. Energy Convers. Manag. 2018, 159, 254–263. [Google Scholar] [CrossRef]
- Wilk, M.; Magdziarz, A. Hydrothermal Carbonization, Torrefaction and Slow Pyrolysis of Miscanthus Giganteus. Energy 2017, 140, 1292–1304. [Google Scholar] [CrossRef]
- Kambo, H.S.; Dutta, A. Comparative Evaluation of Torrefaction and Hydrothermal Carbonization of Lignocellulosic Biomass for the Production of Solid Biofuel. Energy Convers. Manag. 2015, 105, 746–755. [Google Scholar] [CrossRef]
- Reza, M.T.; Lynam, J.G.; Uddin, M.H.; Coronella, C.J. Hydrothermal Carbonization: Fate of Inorganics. Biomass Bioenergy 2013, 49, 86–94. [Google Scholar] [CrossRef]
- Aragón-Briceño, C.I.; Ross, A.B.; Camargo-Valero, M.A. Mass and Energy Integration Study of Hydrothermal Carbonization with Anaerobic Digestion of Sewage Sludge. Renew. Energy 2021, 167, 473–483. [Google Scholar] [CrossRef]
- Aragón-Briceño, C.I.; Grasham, O.; Ross, A.B.; Dupont, V.; Camargo-Valero, M.A. Hydrothermal Carbonization of Sewage Digestate at Wastewater Treatment Works: Influence of Solid Loading on Characteristics of Hydrochar, Process Water and Plant Energetics. Renew. Energy 2020, 157, 959–973. [Google Scholar] [CrossRef]
- Wilk, M.; Magdziarz, A.; Kalemba-Rec, I.; Szymańska-Chargot, M. Upgrading of Green Waste into Carbon-Rich Solid Biofuel by Hydrothermal Carbonization: The Effect of Process Parameters on Hydrochar Derived from Acacia. Energy 2020, 202, 117717. [Google Scholar] [CrossRef]
- Wilk, M.; Magdziarz, A.; Jayaraman, K.; Szymańska-Chargot, M.; Gökalp, I. Hydrothermal Carbonization Characteristics of Sewage Sludge and Lignocellulosic Biomass. A Comparative Study. Biomass Bioenergy 2019, 120, 166–175. [Google Scholar] [CrossRef]
- Kumar, N.; Weldon, R.; Lynam, J.G. Hydrothermal Carbonization of Coffee Silverskins. Biocatal. Agric. Biotechnol. 2021, 36, 102145. [Google Scholar] [CrossRef]
- Lucantonio, S.; Di Giuliano, A.; Gallucci, K. Influences of the Pretreatments of Residual Biomass on Gasification Processes: Experimental Devolatilizations Study in a Fluidized Bed. Appl. Sci. 2021, 11, 5722. [Google Scholar] [CrossRef]
- Hansen, L.J.; Fendt, S.; Spliethoff, H. Comparison of Fuels and Effluents Originating from Washing and Hydrothermal Carbonisation of Residual Biomass. Waste Biomass Valorization 2022, 13, 2321–2333. [Google Scholar] [CrossRef]
- Gao, L.; Volpe, M.; Lucian, M.; Fiori, L.; Goldfarb, J.L. Does Hydrothermal Carbonization as a Biomass Pretreatment Reduce Fuel Segregation of Coal-Biomass Blends during Oxidation? Energy Convers. Manag. 2019, 181, 93–104. [Google Scholar] [CrossRef]
- Ferrentino, R.; Ceccato, R.; Marchetti, V.; Andreottola, G.; Fiori, L. Sewage Sludge Hydrochar: An Option for Removal of Methylene Blue from Wastewater. Appl. Sci. 2020, 10, 3445. [Google Scholar] [CrossRef]
- Gallucci, K.; Taglieri, L.; Papa, A.A.; Di Lauro, F.; Ahmad, Z.; Gallifuoco, A. Non-Energy Valorization of Residual Biomasses via HTC: CO2 Capture onto Activated Hydrochars. Appl. Sci. 2020, 10, 1879. [Google Scholar] [CrossRef]
- Channiwala, S.A.; Parikh, P.P. A Unified Correlation for Estimating HHV of Solid, Liquid and Gaseous Fuels. Fuel 2002, 81, 1051–1063. [Google Scholar] [CrossRef]
- Friedl, A.; Padouvas, E.; Rotter, H.; Varmuza, K. Prediction of Heating Values of Biomass Fuel from Elemental Composition. Anal. Chim. Acta 2005, 544, 191–198. [Google Scholar] [CrossRef]
- Sallevelt, J.L.H.P.; Gudde, J.E.P.; Pozarlik, A.K.; Brem, G. The Impact of Spray Quality on the Combustion of a Viscous Biofuel in a Micro Gas Turbine. Appl. Energy 2014, 132, 575–585. [Google Scholar] [CrossRef]
- Sallevelt, J.L.H.P.; Pozarlik, A.K.; Brem, G. Characterization of Viscous Biofuel Sprays Using Digital Imaging in the Near Field Region. Appl. Energy 2015, 147, 161–175. [Google Scholar] [CrossRef]
- Bhakta Sharma, H.; Panigrahi, S.; Dubey, B.K. Food Waste Hydrothermal Carbonization: Study on the Effects of Reaction Severities, Pelletization and Framework Development Using Approaches of the Circular Economy. Bioresour. Technol. 2021, 333, 125187. [Google Scholar] [CrossRef]
- Romanowska-Duda, Z.; Szufa, S.; Grzesik, M.; Piotrowski, K.; Janas, R. The Promotive Effect of Cyanobacteria and Chlorella sp. Foliar Biofertilization on Growth and Metabolic Activities of Willow (Salix viminalis L.) Plants as Feedstock Production, Solid Biofuel and Biochar as C Carrier for Fertilizers via Torrefaction Proce. Energies 2021, 14, 5262. [Google Scholar] [CrossRef]
- Szufa, S.; Piersa, P.; Adrian, Ł.; Czerwińska, J.; Lewandowski, A.; Lewandowska, W.; Sielski, J.; Dzikuć, M.; Wróbel, M.; Jewiarz, M.; et al. Sustainable Drying and Torrefaction Processes of Miscanthus for Use as a Pelletized Solid Biofuel and Biocarbon-Carrier for Fertilizers. Molecules 2021, 26, 1014. [Google Scholar] [CrossRef]
- Szufa, S.; Piersa, P.; Adrian, Ł.; Sielski, J.; Grzesik, M.; Romanowska-Duda, Z.; Piotrowski, K.; Lewandowska, W. Acquisition of Torrefied Biomass from Jerusalem Artichoke Grown in a Closed Circular System Using Biogas Plant Waste. Molecules 2020, 25, 3862. [Google Scholar] [CrossRef]
- Brachi, P.; Miccio, F.; Miccio, M.; Ruoppolo, G. Torrefaction of Tomato Peel Residues in a Fluidized Bed of Inert Particles and a Fixed-Bed Reactor. Energy Fuels 2016, 30, 4858–4868. [Google Scholar] [CrossRef]
- Gucho, E.; Shahzad, K.; Bramer, E.; Akhtar, N.; Brem, G. Experimental Study on Dry Torrefaction of Beech Wood and Miscanthus. Energies 2015, 8, 3903–3923. [Google Scholar] [CrossRef]
- Ribeiro, J.M.C.; Godina, R.; Matias, J.C.d.O.; Nunes, L.J.R. Future Perspectives of Biomass Torrefaction: Review of the Current State-of-the-Art and Research Development. Sustainability 2018, 10, 2323. [Google Scholar] [CrossRef]
- Szwaja, S.; Magdziarz, A.; Zajemska, M.; Poskart, A. A Torrefaction of Sida Hermaphrodita to Improve Fuel Properties. Advanced Analysis of Torrefied Products. Renew. Energy 2019, 141, 894–902. [Google Scholar] [CrossRef]
- Jaworski, T.J.; Kajda-Szcześniak, M. Study on the Similarity of the Parameters of Biomass and Solid Waste Fuel Combustion for the Needs of Thermal Power Engineering. Sustainability 2020, 12, 7894. [Google Scholar] [CrossRef]
- Botelho, T.; Costa, M.; Wilk, M.; Magdziarz, A. Evaluation of the Combustion Characteristics of Raw and Torrefied Grape Pomace in a Thermogravimetric Analyzer and in a Drop Tube Furnace. Fuel 2018, 212, 95–100. [Google Scholar] [CrossRef]
- Mlonka-Mędrala, A.; Magdziarz, A.; Dziok, T.; Sieradzka, M.; Nowak, W. Laboratory Studies on the Influence of Biomass Particle Size on Pyrolysis and Combustion Using TG GC/MS. Fuel 2019, 252, 635–645. [Google Scholar] [CrossRef]
- Kumar, A.; Monika; Mishra, R.K.; Jaglan, S. Pyrolysis of Low-Value Waste Miscanthus Grass: Physicochemical Characterization, Pyrolysis Kinetics, and Characterization of Pyrolytic End Products. Process Saf. Environ. Prot. 2022, 163, 68–81. [Google Scholar] [CrossRef]
- Matusiak, M.; Slęzak, R.; Ledakowicz, S. Thermogravimetric Kinetics of Selected Energy Crops Pyrolysis. Energies 2020, 13, 3977. [Google Scholar] [CrossRef]
- Cortés, A.M.; Bridgwater, A.V. Kinetic Study of the Pyrolysis of Miscanthus and Its Acid Hydrolysis Residue by Thermogravimetric Analysis. Fuel Process. Technol. 2015, 138, 184–193. [Google Scholar] [CrossRef]
- Asadullah, M. Biomass Gasification Gas Cleaning for Downstream Applications: A Comparative Critical Review. Renew. Sustain. Energy Rev. 2014, 40, 118–132. [Google Scholar] [CrossRef]


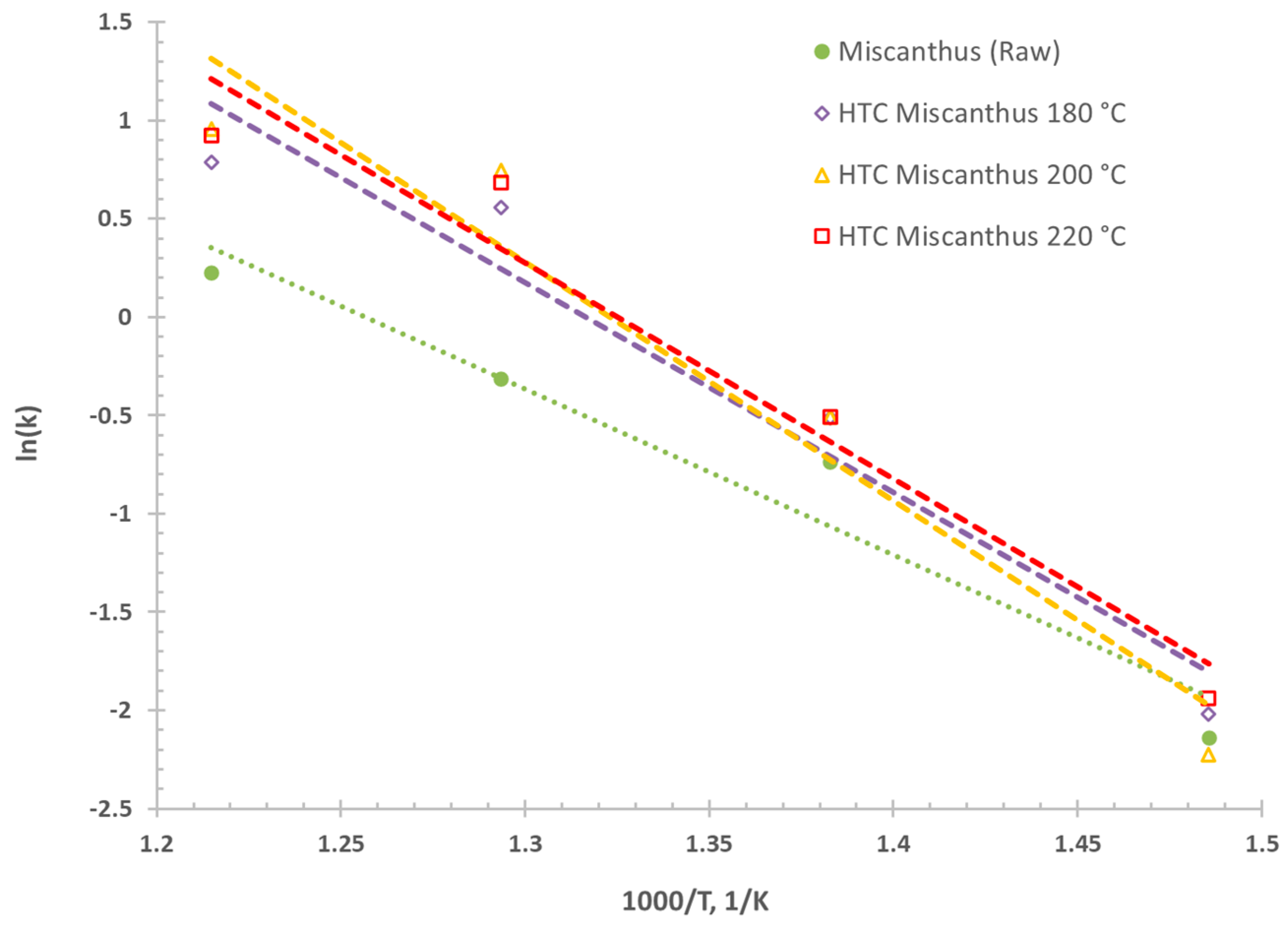

| R2 | Ea | ln A | A | ||
|---|---|---|---|---|---|
| kJ/mol | 1/s | 1/s | 1/min | ||
| Miscanthus (Raw) | 0.9454 | 70.25 | 10.619 | 4.09 × 104 | 6.82 × 102 |
| Miscanthus + 10% CaO | 0.9909 | 86.75 | 13.594 | 8.01 × 105 | 1.34 × 104 |
| Miscanthus + 20% CaO | 0.9717 | 74.48 | 11.774 | 1.30 × 105 | 2.16 × 103 |
| HTC Miscanthus 180 °C | 0.9452 | 88.83 | 14.064 | 1.28 × 106 | 2.14 × 104 |
| HTC Miscanthus 200 °C | 0.9399 | 101.10 | 16.087 | 9.69 × 106 | 1.62 × 105 |
| HTC Miscanthus 220 °C | 0.9530 | 91.33 | 14.556 | 2.10 × 106 | 3.49 × 104 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Niedzwiecki, L.; Moscicki, K.; Bijl, A.; Owczarek, P.; Arora, A.; Wnukowski, M.; Aragon-Briceno, C.; Vishwajeet; Pawlak-Kruczek, H.; Bramer, E.; et al. Influence of Hydrothermal Carbonization on Catalytic Fast Pyrolysis of Agricultural Biomass. Appl. Sci. 2023, 13, 4190. https://doi.org/10.3390/app13074190
Niedzwiecki L, Moscicki K, Bijl A, Owczarek P, Arora A, Wnukowski M, Aragon-Briceno C, Vishwajeet, Pawlak-Kruczek H, Bramer E, et al. Influence of Hydrothermal Carbonization on Catalytic Fast Pyrolysis of Agricultural Biomass. Applied Sciences. 2023; 13(7):4190. https://doi.org/10.3390/app13074190
Chicago/Turabian StyleNiedzwiecki, Lukasz, Krzysztof Moscicki, Anton Bijl, Pawel Owczarek, Amit Arora, Mateusz Wnukowski, Christian Aragon-Briceno, Vishwajeet, Halina Pawlak-Kruczek, Eddy Bramer, and et al. 2023. "Influence of Hydrothermal Carbonization on Catalytic Fast Pyrolysis of Agricultural Biomass" Applied Sciences 13, no. 7: 4190. https://doi.org/10.3390/app13074190
APA StyleNiedzwiecki, L., Moscicki, K., Bijl, A., Owczarek, P., Arora, A., Wnukowski, M., Aragon-Briceno, C., Vishwajeet, Pawlak-Kruczek, H., Bramer, E., Brem, G., & Pozarlik, A. (2023). Influence of Hydrothermal Carbonization on Catalytic Fast Pyrolysis of Agricultural Biomass. Applied Sciences, 13(7), 4190. https://doi.org/10.3390/app13074190










