1. Introduction
Nature is a rich source of ideas and inspiration in various technological activities of humankind. Mechatronics, as a branch of engineering, has its own subset of activities known as biomechatronics. Within this field, aspects of biology, mechanics, electronics, and informatics are integrated in the design of complex products that imitate biological systems occurring in nature. The development of tools that can improve or restore human bodily function is one of the key objectives of biomechatronics [
1,
2].
The subject of engineering known as biomechatronics is incredibly intriguing and has great potential to help those who are ill or disabled live better lives. Innovative solutions are being developed in this area of study in an effort to assist people overcome physical constraints and increase their independence and mobility [
3]. Healthcare could be transformed through biomechatronics, enhancing, augmenting, or supplementing human performance and overall convenience of life [
4].
The initial implementation of biomechatronics in patient treatment aimed to replace standard rehabilitation therapies. However, recent studies indicate that biomechatronics may serve as a valuable tool in complementing, rather than substituting, rehabilitation. This is particularly significant with regard to behavioural aspects concerning sensory perception, motor adaptation, and neuronal motor skill learning [
5].
One of the types of disability that is becoming increasingly common in developed countries is the lack of a full upper limb. The extent of amputation varies, but the most common causes are accidents, particularly for men, followed by cancer, vascular diseases, and infectious diseases. Amputation disrupts efferent and afferent pathways responsible for motor control and sensory feedback from the limb to the brain [
6]. The complexity of neuronal changes that occur after amputation presents both a challenge and an opportunity for better planning of patient rehabilitation strategies. Answers to questions regarding the necessity of sensory feedback, learning motor control with prostheses, neuroplasticity, and individual patient needs complement each other, creating a deeper and more appropriate knowledge base for effective rehabilitation [
7].
Prosthesis rejection is a well-known problem in amputation rehabilitation. As research indicates, better prosthesis quality, understood as emphasizing individual user needs and requirements, facilitates successful prosthetic fitting, increases prosthesis usage time, and minimizes the risk of prosthesis rejection [
8]. Therefore, the possibility of easy and inexpensive prosthesis customization is an important factor that can significantly improve the well-being of people with disabilities.
Additive manufacturing is a technology that allows for relatively inexpensive production of fully individualized products. This technology has found wide application in practically all fields of engineering, including medicine. Numerous medical devices, tailored to the anatomy of a specific patient, can be made through additive manufacturing techniques [
9]. There are many different methods of additive manufacturing that differ significantly from one another. The most popular and economically accessible method is fused deposition modelling (FDM), also known as fused filament fabrication (FFF), which enables the production of thermoplastic products such as ABS (acrylonitrile–butadiene–styrene) and PLA (polylactic acid) [
10].
There are many examples in the literature of various upper limb prostheses that are wholly or partially produced using additive manufacturing technology. A common feature of these studies is the relatively small amount of data on the strength properties of the prostheses, as well as information related to their actual use by patients [
11]. Another research gap is the area of upper limb prosthetics that could be used by patients for recreational and sports purposes. Such prostheses, equipped with appropriate sensors, could provide data to improve training and increase user performance [
12].
Additive manufacturing requires a digital model of the product. Individualizing the geometry to meet the individual anatomical needs of the patient requires appropriate angle measurement, which becomes the basis for designing the prosthetic socket. When the patient has one healthy arm, measuring its characteristics also provides important information for the designer. Measuring a patient’s anatomical features can be performed, for example, using non-contact methods such as 3D scanning with structured light on dedicated measuring stations [
13,
14], or by freely scanning an unsupported stump in a natural position for the patient or dedicated to the intended product [
15].
Active prostheses are equipped with various sensors that expand their basic functions. On the one hand, sensors enable control of the actuator elements, such as myoelectric control of finger clenching [
16]. On the other hand, prosthesis sensors can provide valuable information about the prosthesis itself or its surrounding environment, such as contact with another object (tactile sensing) [
17]. Sensory feedback systems reduce visual dependence and mental effort, particularly for individuals with double amputation or impaired vision. Therefore, they are of great interest in the field of upper limb prosthesis research and can be developed as additional equipment for already-used passive prostheses [
18]. Research is being conducted, and demonstration sensor systems are being built for upper limb prostheses, which have the primary advantage of being low cost, and therefore more accessible [
19]. However, commercially available solutions are still very expensive.
The use of more advanced and complicated sensory systems may be particularly significant for specialized biomechanical prostheses in recreational or professional sports. As recent research suggests, the use of orientation metrics, such as Euler angles or quaternions, would provide a deeper insight into biomechanical processes, and athletes could improve their skills by analysing gathered information. Miniaturization of sensors and implementation of artificial intelligence for data analysis could allow interaction with a greater number or even new types of sensors, such as oxygen, glucose, and blood pressure levels [
20].
In authors’ opinion, research on sensorised upper limb prostheses dedicated for sports, particularly for cycling, is imperative due to several reasons. It is challenging for individuals with upper limb amputations to participate in sport activities, especially those that require the use of hands and arms. This limitation can result in physical, psychological, and social drawbacks for amputee cyclists. It is noteworthy that sports-oriented prostheses need to be tailored to person’s specific needs and personal preferences, both for amateur and professional athletes.
The use of sensor technologies can provide real-time feedback for athletes to optimize their prosthetic usage and fine-tune their performance. Additionally, accurate sensing of limb movements and biofeedback can help reduce the risk of injuries during training and competition. Performed research has the potential to improve the durability and functionality of prosthetic devices. While conventional prosthetic devices are highly functional, the use of complementing sensors to track muscle signals can enhance prosthetic control and movement precision for amputee athletes. Implementing low-cost 3D printing technologies and low-cost, widely available sensors would make the sport activities more accessible for the wider group of people with missing limbs.
The above-mentioned considerations are the exact motivation for undertaking the studies presented in the paper. There is a noticeable lack of available prosthetic solutions that help adult patients in their sports activities, such as cycling, while being cheap and personalized at the same time. Usually, such devices are either universal devices (not dedicated for cycling) or very expensive ones, or not fully customized to patient’s anatomy and behaviours. That is why these studies are performed to test if it is possible to quickly, economically, and easily manufacture a usable prosthesis for cycling, equipped with sensor technologies.
2. Materials and Methods
2.1. Research Concept and Plan
This paper presents research on a new, proposed concept of biomechatronic hand prosthesis. A modular 3D printed individualized prosthesis is considered, with its adjustment to needs and preferences of an adult patient and converting static mechanical device into a mechatronic prosthesis, equipped with sensors for monitoring the activities performed by the prosthesis user.
The initial concept of the prosthesis was made as part of the project “Automation of design and rapid production of individualized orthopaedic and prosthetic products based on data from anthropometric measurements”, serving the development of the prototype for the AutoMedPrint system [
14], continuing previous long-term studies by the authors.
The prosthesis has been specifically tailored to meet the requirements of an adult patient engaged in rigorous and continuous cycling. The modular mechanical prosthesis was originally made for child patients (and successfully implemented, as described in [
14] and [
21], and shown in
Figure 1).
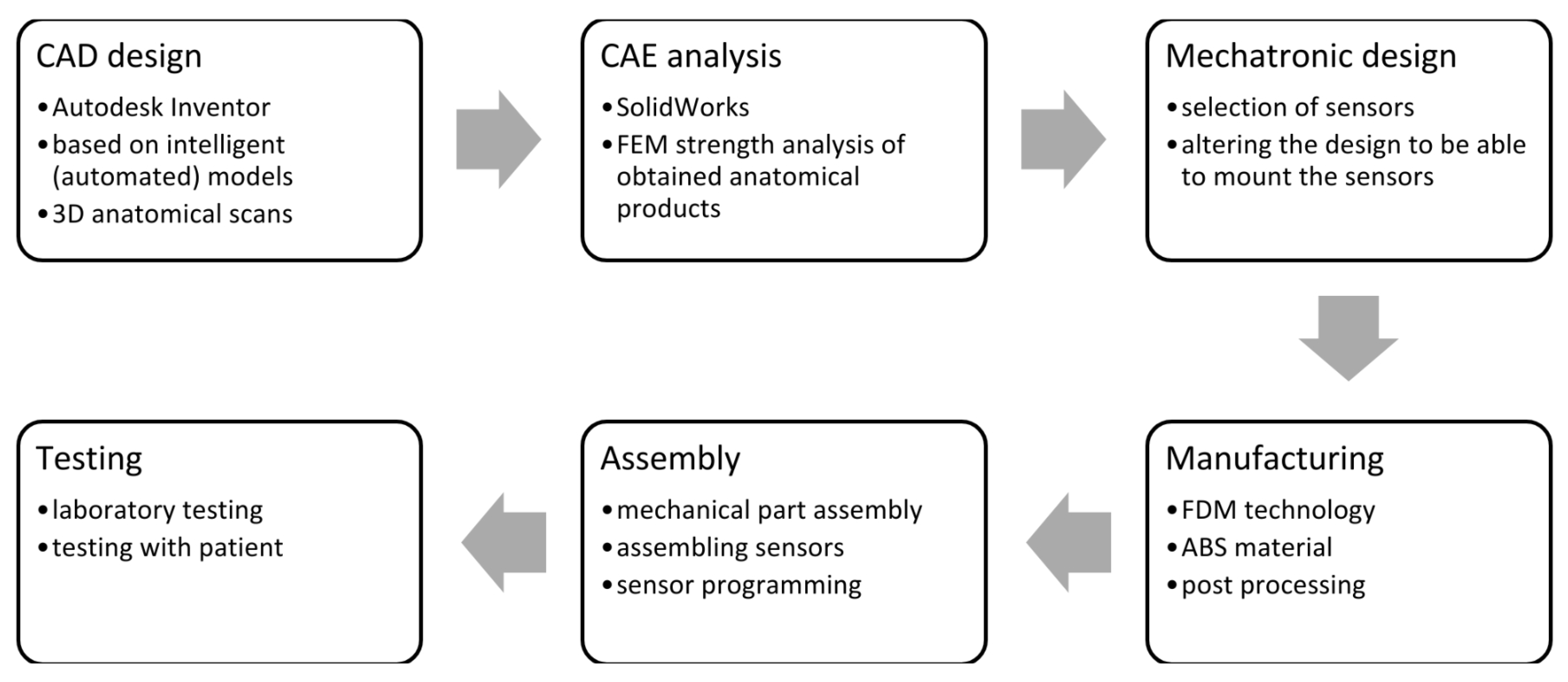
Building upon the foundational concept of the modular prosthesis model, essential modifications were implemented to customize it for adult use. After testing and optimising the device with the bicycle, the adult mechanical prosthesis was transformed into a mechatronic one through a sensorisation process. The designed prosthesis was 3D printed, assembled, and programmed. It was simulated for strength using Finite Element Method, and after assembly, testing with the patient was performed. The course of the studies described in this paper with basic characteristics of each stage is presented in the scheme of
Figure 2.
2.2. Background—Prosthesis Design and Patient Case Description
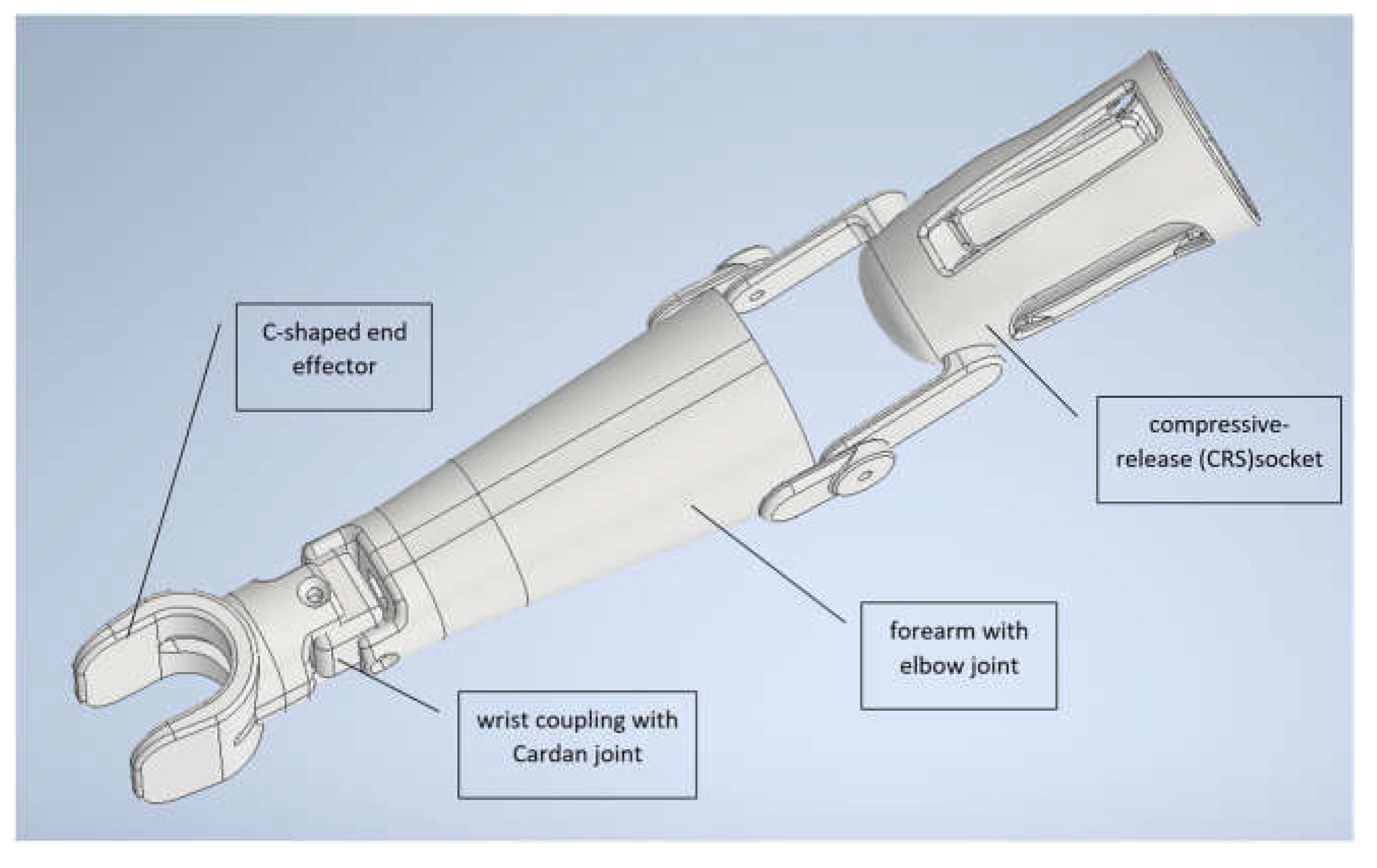
The design was commenced with a project of an intelligent model of the upper limb prosthesis, which is characterized by a modular structure. The device represents a cohesive entity comprising multiple interconnected elements. The model is loaded with anthropometric and configuration data directly from an external Excel file [
22], enabling both the generation of anatomically matched prosthesis components and the manipulation of its variants to create any combination of all parts. Therefore, the model allows for the quick and fully automated production of many configurations of individualized prostheses for the same or different patients. It is presented in
Figure 3 and is its basic variant, containing a compressive-release socket (CRS [
23]), a forearm with elbow joint, and C-shaped end effector with Cardan joint at the wrist.
The prosthesis model was adapted to an adult patient. A case of 40-year-old male patient was selected for the studies. The patient was born without his right forearm (no functioning elbow joint). Both of the patient’s upper limbs were 3D scanned (
Figure 4), and the resulting data were automatically processed, using the AutoMedPrint system hardware and software capabilities (as described in earlier works [
14,
22]).
In the preliminary studies, the initial version of prosthesis was manufactured and tested by the patient. After minor corrections (for strength improvement), the prototype was tested in real conditions by the patient riding the bicycle (
Figure 5). These tests were positively concluded, and the patient was given the prosthesis for longer use, to obtain feedback regarding further construction and use issues.
In the course of numerous tests and subsequent design iterations (considering patient’s comments and usage reports, as well as authors’ observations), the following modifications have been introduced in the mechanical part of the prosthesis:
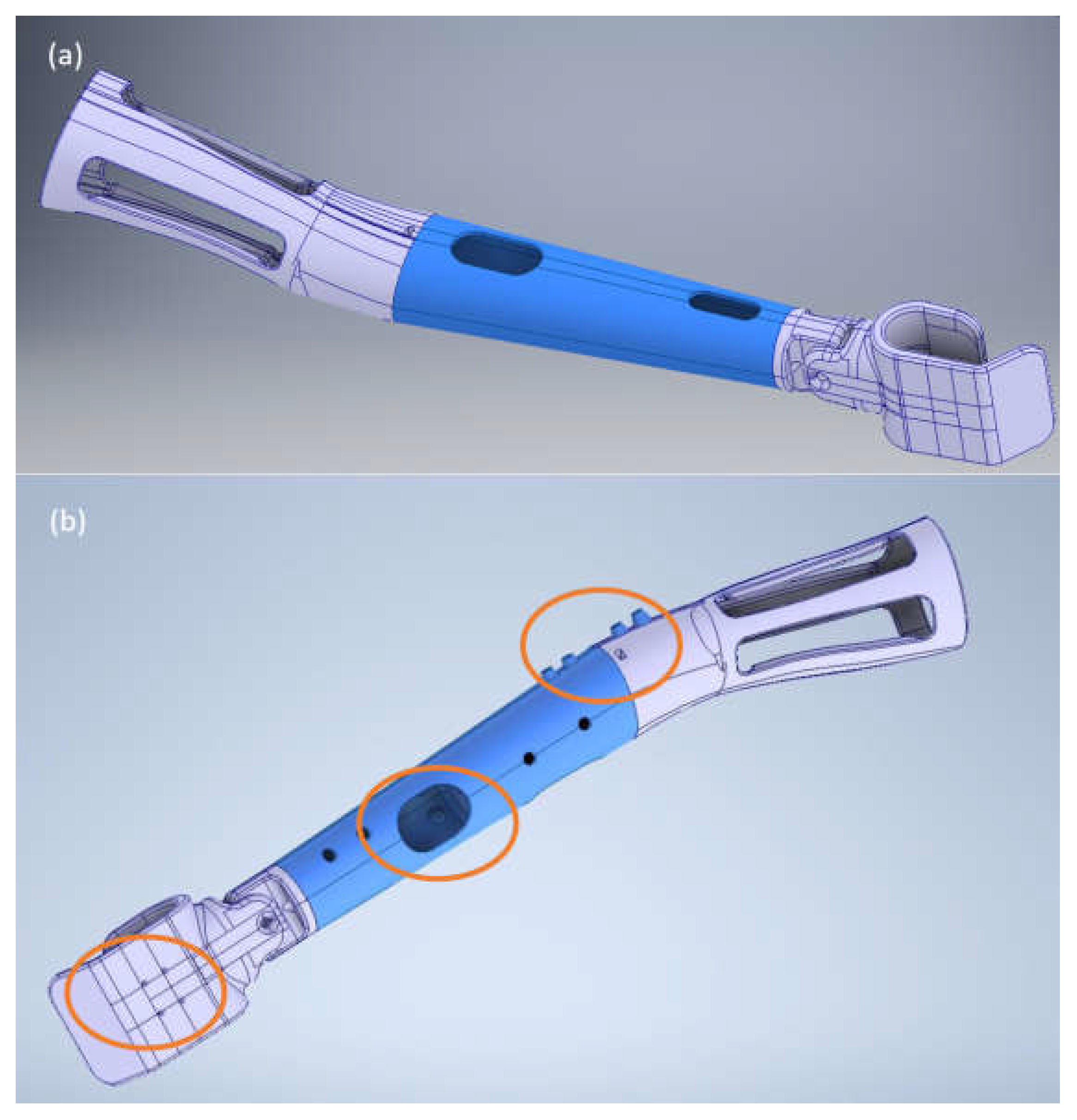
The CRS socket was modified as a result of removing the movable connection in the elbow joint. It was connected to the forearm rigidly due to too much load in the case of an adult The mounting plane was moved away from the tip of the socket to a properly selected distance, mounting hardware was added to allow connection to the forearm (
Figure 6a).
The forearm model was changed as a result of abandoning the movable joint in the elbow joint. It was a decision of the patient as he uses bicycle in one, stable position, the presence of elbow joint in the prosthesis (that he does not have in his stump) would not be ergonomic and could be potentially destabilizing the body position while riding. In other words, eliminating the elbow joint helped with safety and rigidity, while introducing no practical function impairment. The space of elbow was filled in with material for better strength. Five mounting holes and two access holes were added (
Figure 6b).
The C-handle was modified by extending the jaws and narrowing the distance between them. These changes were introduced after patient’s feedback as they increased the stability and comfort of cycling. In addition, the connection used in the wrist was abandoned as the lack of a joint resulted in an increase in stiffness with the simultaneous impossibility of rotation. This was possible after adjusting the angles for a given patient and his bicycle (
Figure 6c).
The improved prosthesis in its mechanical version was well proven in use. It must be emphasized that the patient was using the first version (as shown in
Figure 5) for one full bicycle season and the second version (modified and visible in
Figure 6) for another full season. The prostheses did not sustain any major damage and were successfully used for bicycle riding in normal, road conditions (no rough terrain use). As such, it was assumed that the design was safe, ergonomic and proven for practical use. This assumption was a base for the actual research on sensor-equipped prosthesis, described in the further parts of this paper.
2.3. Strength Analysis
The final design of the prosthesis was subjected to strength analysis prior to equipping it with sensors. The ISO 22523 standard was used here, and the recommended scheme of loading was taken from the standard [
24]. The objective of this test was to evaluate the strength characteristics of the prosthesis by simulating a distal tensile test with the SolidWorks Simulation finite element analysis (FEA) module included in the SolidWorks CAD package. The principle of the test is shown in
Figure 7. The prosthesis model was subjected to a distal traction load after being firmly attached to a rigid support that fits inner surfaces of the upper arm. The traction load gradually increases from 0 (zero) to 750 N in five steps. The main purpose of the simulation was to make sure that the prosthesis is safe for the user and detect possible locations of fractures. If the FEM detected any higher stress ratio than the safe rates for the given material, changes in the design would have been introduced.
The following hypotheses were adopted when preparing the finite element model of the tensile test:
The displacement (deflection), force, and stress quantities manipulated by the FEA model are expressed using the following measurement units: displacement (deflection)in millimeter (mm); force in Newton (N); and stress in megapascal (MPa) (1 MPa = 1 N/mm2). The FEA test was prepared using the following assumptions:
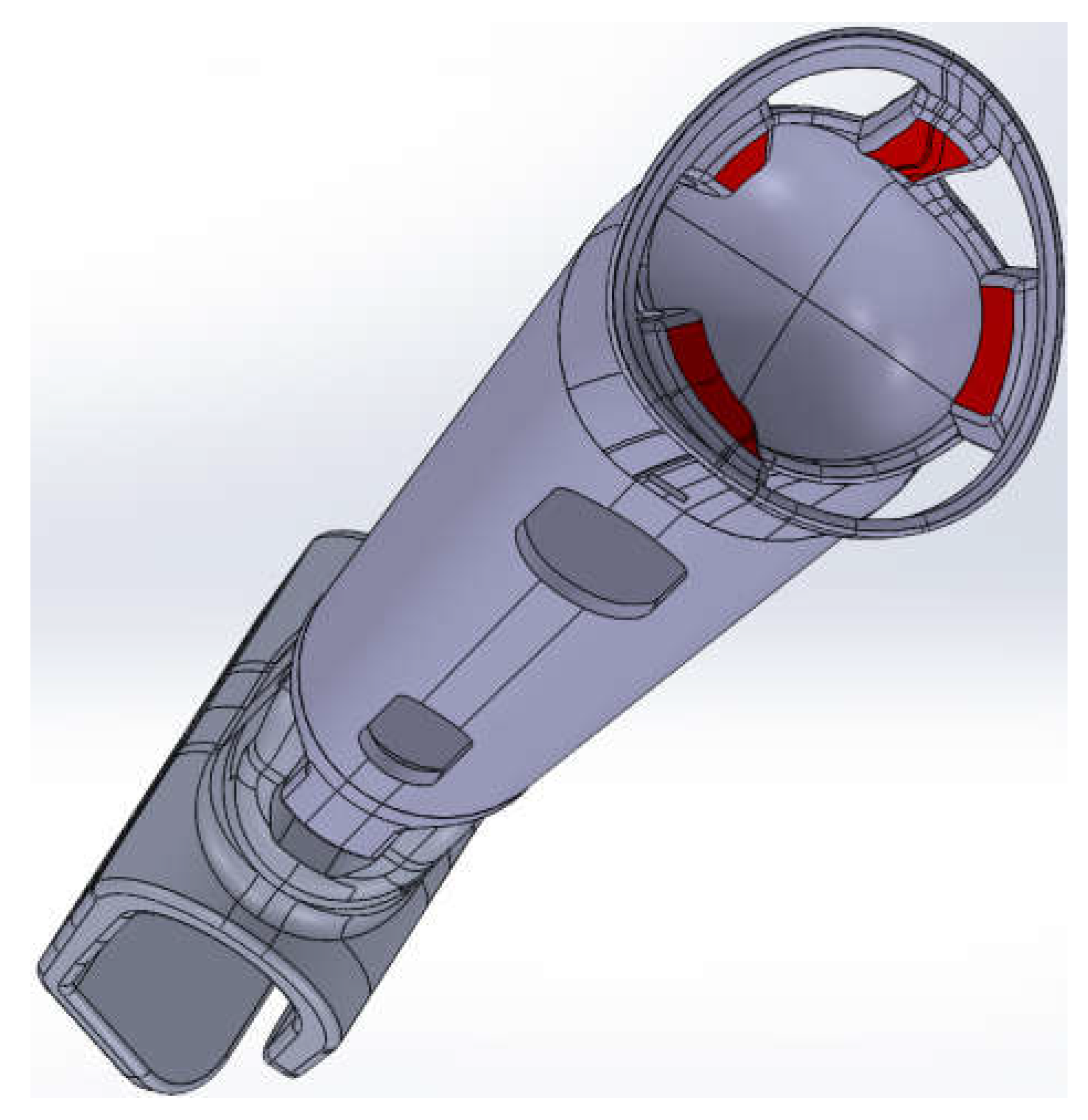
The locking boundary on inner surfaces of the compressive release prosthetic socket (
Figure 8) is compatible with places where the socket firmly sits on patient’s arm;
Five cases of loads (150 N, 300 N, 450 N, 600 N, and 750 N) set as a distributed force located in the inner lower surface of the end effector (as visible in
Figure 7) are considered;
A tetrahedral mesh is generated using “Fine” setting of the SolidWorks Simulation module, resulting in small size of finite elements for better resolution of results (
Figure 9).
The simulation was conducted using the above-mentioned settings. As a method of determining simulated stresses in the prosthesis, von Mises equivalent stress was selected. Values of the stress were checked all over the volume of prosthesis, and an evaluation was performed to find out if the prosthesis is safe for use in the real conditions.
2.4. Concept and Design of Electronic Part
The aim of this work was to modify the hand prosthesis in order to create a biomechatronic device, used by human, with monitoring of activities performed in the prosthesis (mostly cycling or similar activities). The main concept was placing an electronic measuring system in the prosthesis, thanks to which it would be possible to determine its operating properties. The detailed purposes of the built electronic system were as follows:
Enable measurement of the orientation of the upper limb prosthesis in space;
Enable measurement of the force exerted in the prosthesis while riding the bicycle;
Enable storing data on an SD card.
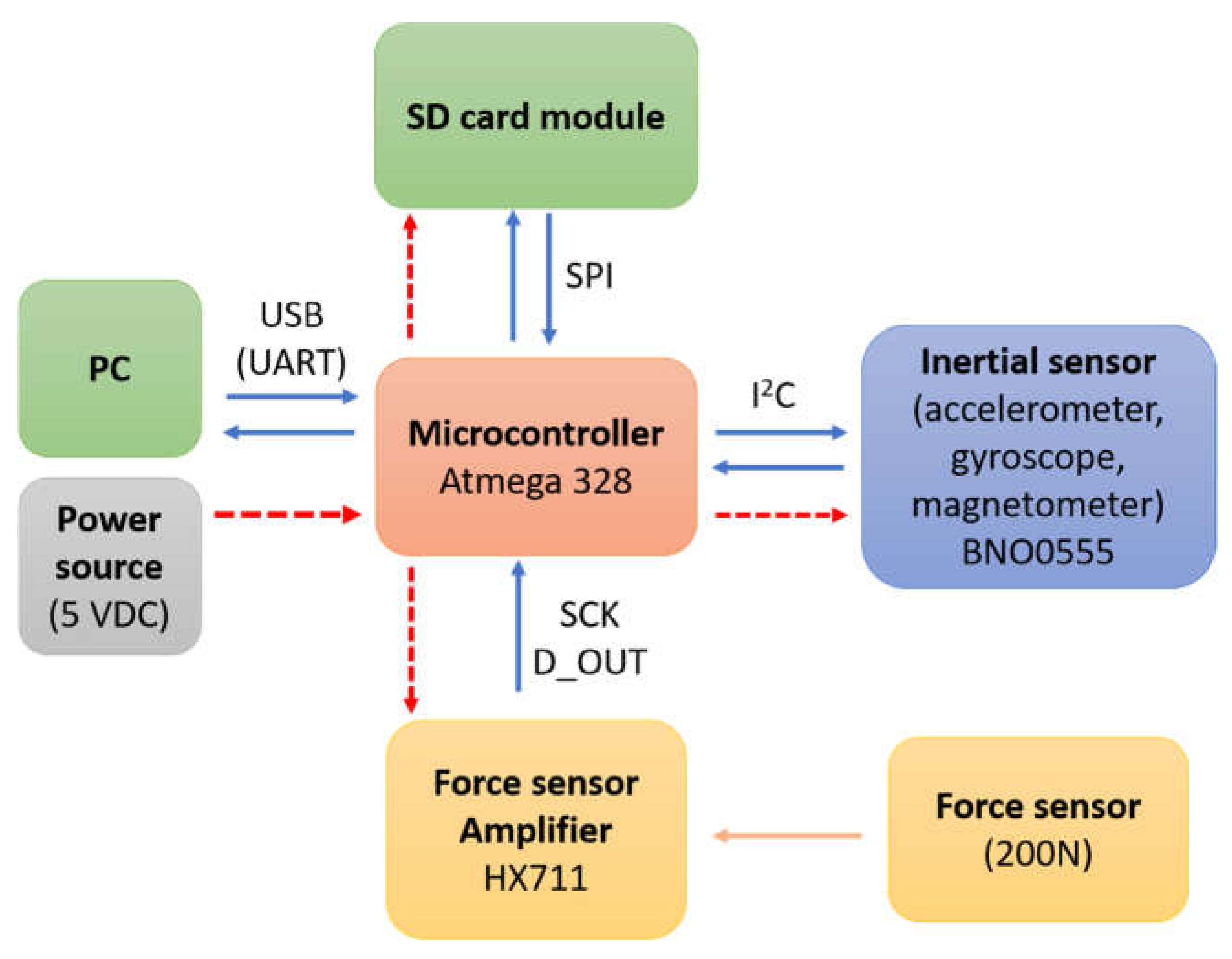
The device consisted of the following components:
A microcontroller module (Arduino NANO) [
25];
A force sensor measuring amplifier module (HX711 with force sensor up to 200N);
An inertial sensor module (BOSCH BNO055);
An Arduino SD card module;
Power source (a USB connected power bank).
The components were selected to fulfil their role in the simplest possible manner, ensuring robust operation and steady and stable communication and maintaining as low as possible price (as the whole prosthesis is a low-cost project, as mentioned in the research concept). The schematic diagram of the designed device is shown in
Figure 10.
The final mechanical version of the prosthesis CAD model was modified, to be able to fit the electronics of the prosthesis inside it, in a manner allowing steady riding on a bicycle or similar device, without risk of disconnecting or otherwise damaging the components, as well as not making them disturb the patient during the activities. The following main changes have been introduced to the design:
The forearm was modified to enable mounting of the microcontroller, inertial sensor, and SD card module inside cavities of the forearm, and insets were created with holes, for self-tapping purposes (
Figure 11);
At the joint of the forearm and socket, insets were added for mounting of the force sensor (beam), and the place (elbow) was selected to easily detect the torques and forces during the bicycle ride;
A number of assembly holes and cable feedthroughs were added to enable unproblematic assembly of the electronic part inside the prosthesis.
The final version of the CAD model of the prosthesis before and after adding features for electronics assembly is shown in
Figure 12.
2.5. Manufacturing
The designed prosthesis parts in their final version were manufactured using a Zortrax M300 Dual machine (Zortrax, Olsztyn, Poland). The M300 Dual is an upgraded version of the original Zortrax M300, offering dual extrusion capabilities. Key features of the Zortrax M300 Dual include dual extrusion, large build volume (265 × 265 × 300 mm, with a 0.4 mm diameter nozzle, resolution between 100–300 microns, max. temperature of the Table is 100 °C, and max. temperature of the heating block is 380 °C), and compatibility with variable materials, including Zortrax’s proprietary filaments as well as third-party filaments that meet the required specifications. This machine can be classified as a semi-professional 3D printer, with an approximate purchase cost of USD 4490 (as of 2023).
For manufacturing of mechanical parts of the prosthesis, ABS material was used (Spectrum Group, Pęcice, Poland), in the form of 1.75 mm-diameter filament. Material processing characteristics (based on the material supplier data) are presented in
Table 2.
Constant material parameters were kept during manufacturing of all parts, i.e., layer thickness was 0.2 mm, and the internal filling was 30%. The temperatures and extrusion speeds were selected on the basis of the most suitable values recommended by the producer, slightly modified by authors’ previous experiences (bringing the most stable process, without layer disjoint, under extrusion or other typical errors in the FDM process). The values of other manufacturing parameters (not shown in
Table 1) were assumed standard as recommended by machine and material manufacturers.
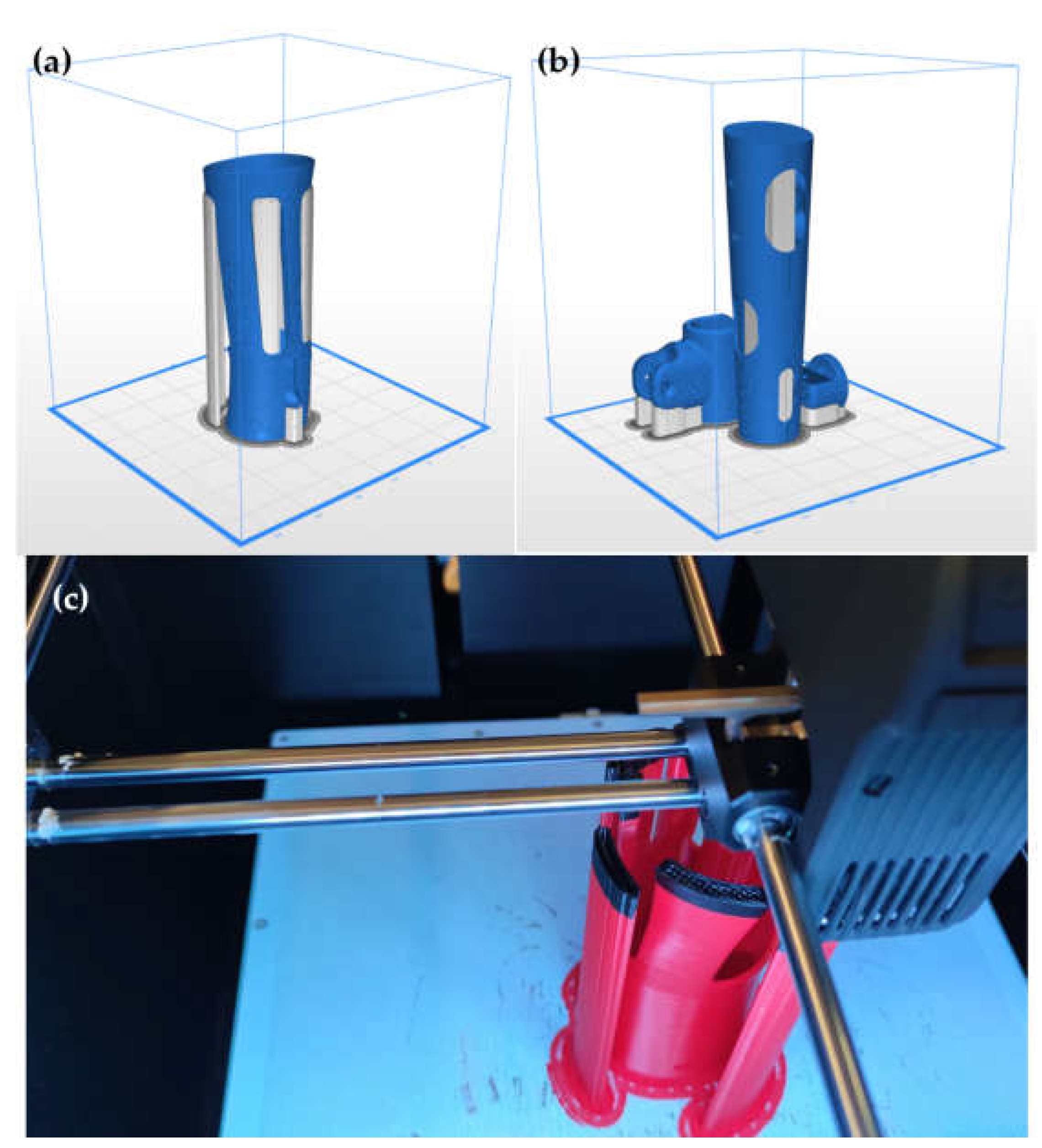
The printing of the prosthesis was divided into two batches, i.e., first the prosthetic socket was printed alone, and then the other elements. The programs were prepared in the Z-Suite program that was dedicated to work with Zortrax printers. The prosthetic socket was manufactured in the appropriate orientation so as to obtain the best possible quality of the inner surface, which is ultimately in direct contact with the residual limb. The expected print time was 21 h, and the estimated material consumption was 210 g. The remaining parts of the prosthesis (forearm with holes for electronics, C-handle, and Cardan) were printed simultaneously, which significantly reduced the production time. The expected print time was approx. 30 h, and the material consumption was 333 g.
Figure 13 shows both prints as simulated in the Z-Suite software and also printer during the process.
The manual post-processing of the obtained products was limited to the simplest activities: support removal, basic manual grinding, and thermal removal of excess strings of material. The produced parts were visually assessed during and after the process to check for major defects that would make them unusable and minor defects that would require certain processing in order to make the prosthesis functional or more comfortable. They were further used for assembly of complete prosthesis.
2.6. Assembly and Programming
The most important components of the mechanical part of the construction were produced using additive manufacturing methods and then assembled into the final product using standardized connecting elements, such as screws and nuts. During the assembly, only basic workshop tools, such as metric wrenches, pliers, and screwdrivers were used.
The assembly of the electronic part was more complex and time-consuming. Due to the significant criterion of the prosthesis cost, generally available and inexpensive electronic components were used. The terminal outputs were soldered into the microcontroller module. The same was conducted with the BNO055 module accelerometer, gyroscope, and magnetometer (Bosch Sensortec Company, Reutlingen, Germany). In order to ensure better connection and reduce disturbances resulting from the assembly method, the force sensor was directly soldered into the HX711 amplifier module (
Figure 14). This transducer can operate at a frequency of up to 80 Hz and provides a measurement resolution of 24 bits. It is equipped with a low-noise preamplifier.
Finally, the electronic components were connected to each other according to the diagram shown in
Figure 15. Components ready for connection and basic testing are presented in
Figure 16.
The electronic components were connected to the mechanical part of the prosthesis using screws and adhesive connections. The major volume of parts of the electronic system was able to be housed within the cavities of the forearm of the prosthesis. The force sensor had an initial pre-tension due to the small variations in mounting hole spacing between the CAD project and additively manufactured parts, which was then taken into account by the data collection method.
Powering the electronic system requires two different voltages. The SD card reader operates at a voltage of 3.3 V, while all other components require a voltage supply of 5 V. The battery power is connected to the Arduino module via the USB connector, which converts them to the appropriate values for each of the connected modules.
The programming of the control code for the prosthetic electronic component was accomplished using the Arduino programming environment version 1.8. The BNO055 inertial sensor module had to be initialized before the working loop. The BNO055 sensor used in the measurement system is based on a 32-bit Cortex M0+ microcontroller. It is equipped with an external 23.768 kHz crystal for greater accuracy. It is equipped with two I2C and UART interfaces. The second of these interfaces is used in the device. Its main advantage is the ability to directly convert quaternion or Euler angle values from the sensor without the need for microcontroller filtering. Determination of the absolute value in Euler angles is carried out on the sensor with a frequency of 100 Hz.
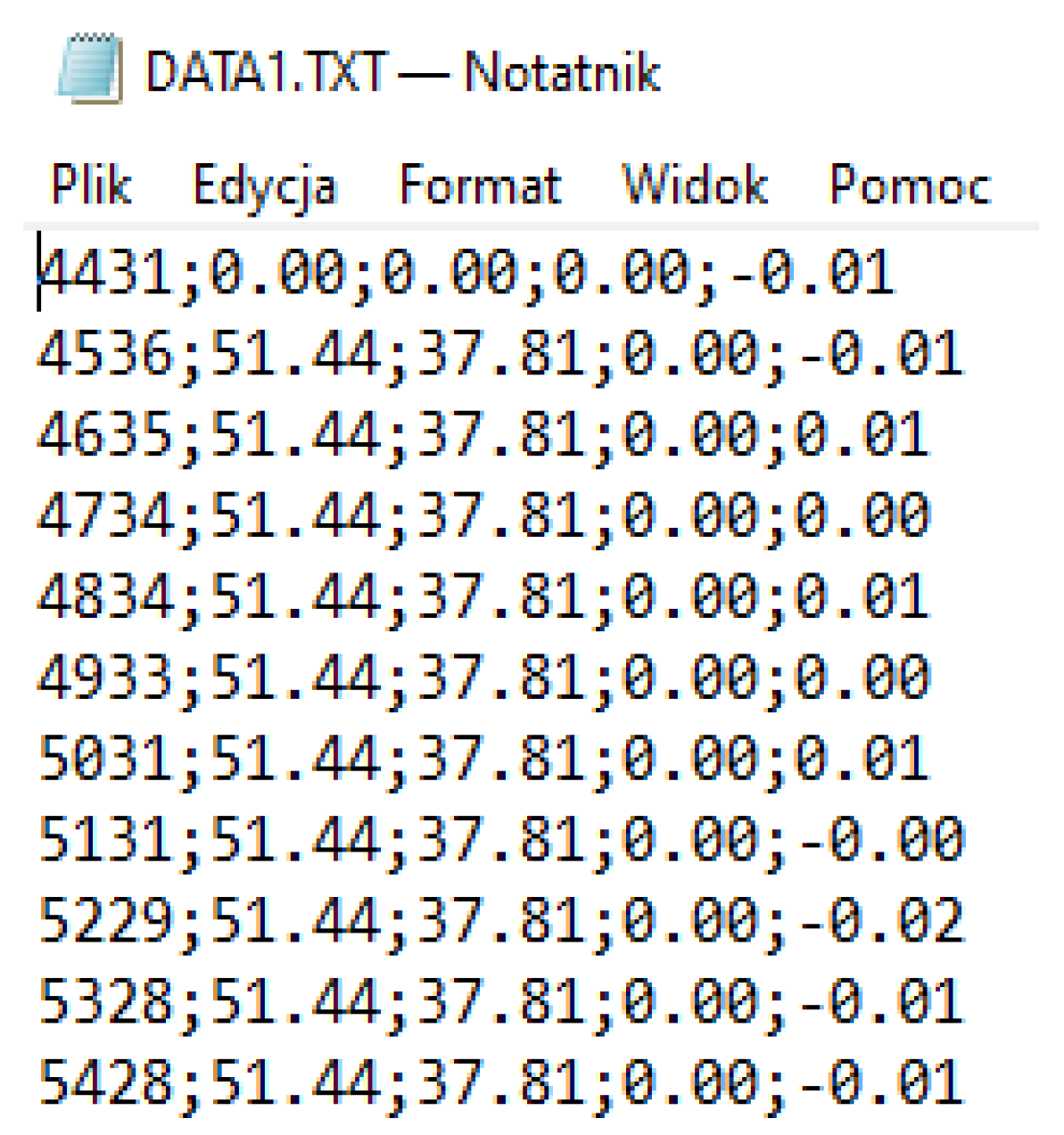
All measurement data are read by the microcontroller program and sent serially to be eventually stored on the SD card. Communication with the SD card module was performed via the SPI interface. The datafile consisted of lines. Each line started with a timestamp, followed with three recorded Euler angles, and followed with a recorded force value (
Figure 17). The data were saved on an SD card, which made the measurements independent of communication with external devices, and also allowed tests to be performed without the need to run specialized software. For this, the measurement was not limited by the distance of the transmitter from the receiver.
Here, it must be mentioned that the SD card storage solution is preliminary and temporary, for experimental purposes only, where the sensor data are analysed post factum (after getting off the bicycle). The assumption at this stage was that if the device reaches further stages of development, the sensor data would need to be sent wirelessly to an external software, in real time, for the patient to see them and react properly if, e.g., forces or accelerations exceed certain recommended “safe” values. This will be explored in further studies.
2.7. Testing Procedure
The first phase of testing was a laboratory procedure, realized without the patient. Initially, the prosthesis was connected to the power source—a power bank—via USB cable (
Figure 18).
During that phase of tests, sample data were recorded. The prosthesis was tested by the researchers, by moving it around in various random patterns and applying various forces, simulating resting on handlebar, static loading (slight bending and compression), and dynamic loading (slight hitting). To generate the visualization of the data, MATLAB software was used. After confirming proper recording of sensor data, further tests were planned and realized.
The second phase of testing involved working with the patient, who volunteered to take part in the experiments (his informed consent was obtained, and the ethical committee approval was gained prior to the studies). This testing stage consisted of fitting tests, where the prosthesis was tested by the patient for comfort, weight, and general fit. It was also compared to a purely mechanical prosthesis, to check if adding the electronic components influenced the use comfort and easiness.
In the next stage of patient testing, the patient was invited to perform tests on the actual device. An electric scooter was used as a reference vehicle. In the first attempt, the patient wore the prosthesis and tried it with the vehicle, to check if everything is correct mechanically. Then, after slight modifications were introduced to the angular position of the end effector (to adjust the prosthesis for the specific vehicle, used by the patient for the first time), the patient performed actual testing with data recording. It consisted of three separate tests:
During both static and dynamic tests, data sets were recorded by the prosthesis sensors. These data sets, similarly, as in the case of laboratory tests, were visualized using MATLAB and compared, to check if the recordings match the actual course of patient’s movement.
It is important to mention here, that these tests were focused on proper gathering of diversified data from the prosthesis sensors. The functions of the prosthesis and various aspects of bicycle riding, such as turning, braking, etc., were already positively verified in the previous season, while patient was using prosthesis of the same design, but purely mechanical (as mentioned in
Section 2.2). As such, turning and braking was not validated here, especially due to technical reasons that patient was unable to bring his own bicycle. However, these functions were properly realized using a standard bicycle, with the front brake simply not used due to lack of grasping capabilities in the prosthesis end effector, and turning was perfectly possible using the C-shaped end effector.
3. Results
3.1. Strength Simulation Results
The main results of FEM calculations are presented in
Figure 21 (fifth, highest case of loading—750 N) and
Table 3. The table presents the maximum values of the von Mises equivalent stress
σeq,max, the maximum deflections
dmax, and the traction forces
F corresponding to different load cases.
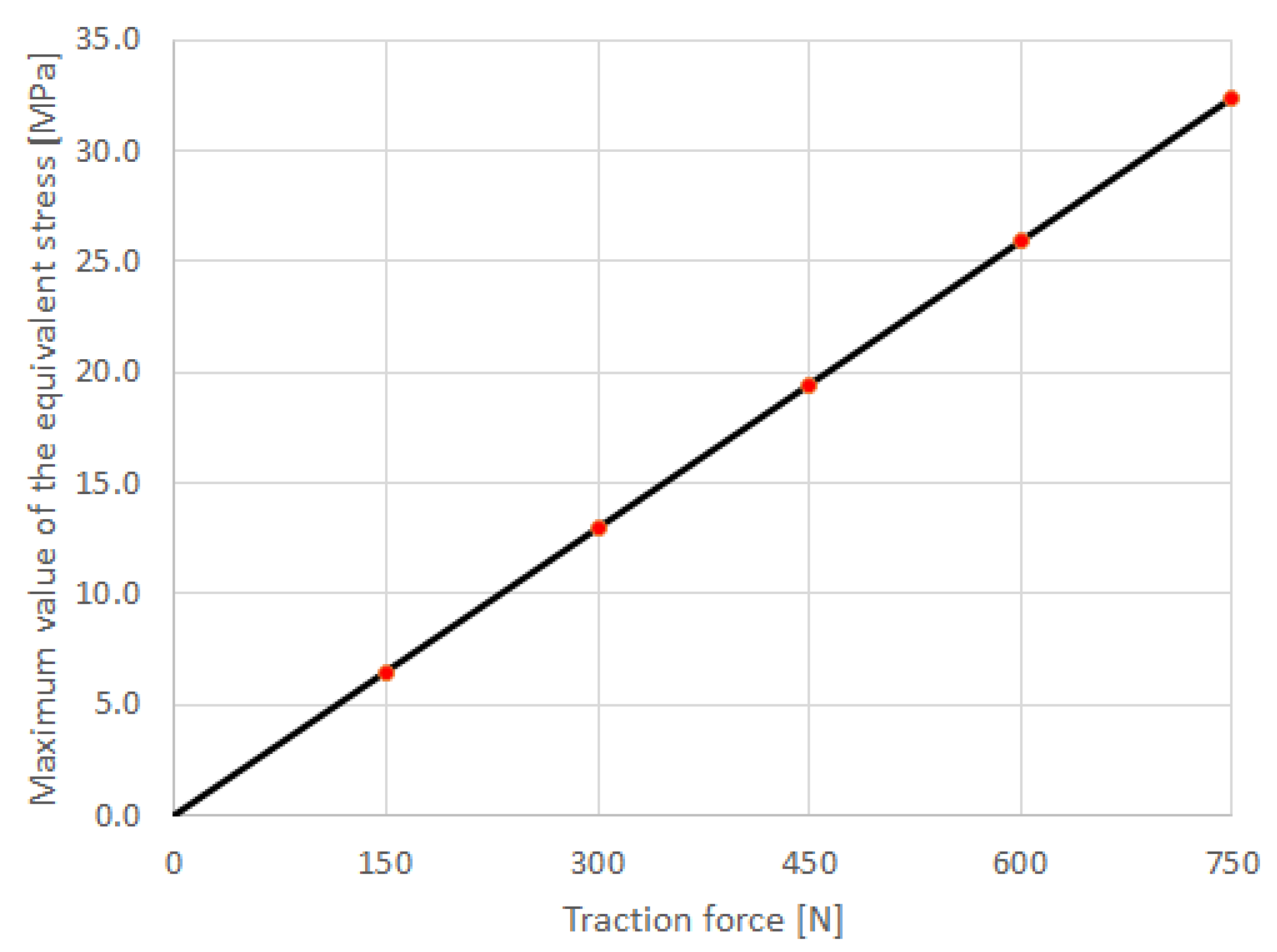
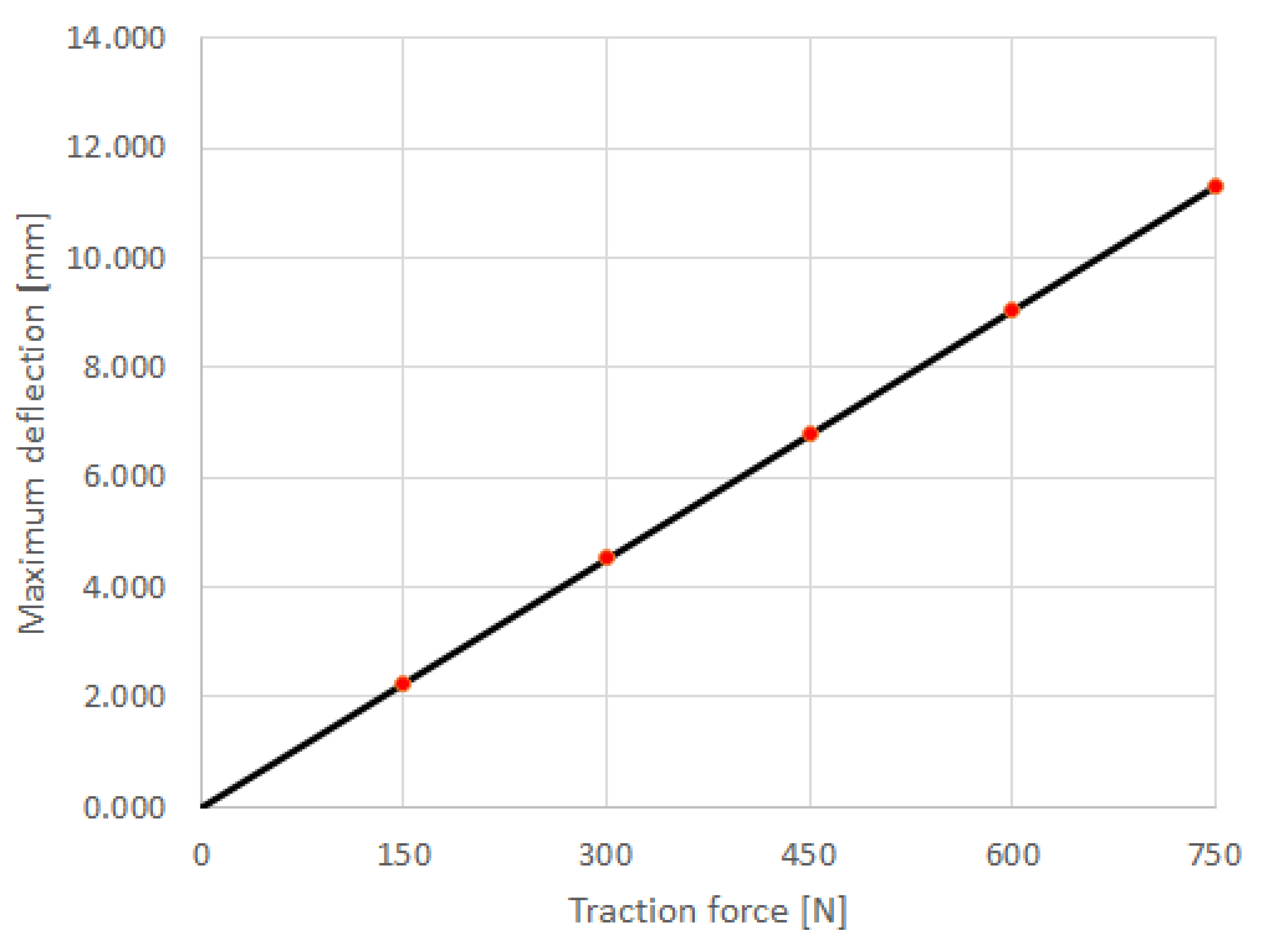
The plots in
Figure 22 and
Figure 23 show the dependencies
σeq,max vs.
F and
dmax vs.
F, respectively. Both diagrams allow the observation that the mechanical response of the prosthesis is linear. In fact, the dependencies
σeq,max vs.
F and
dmax vs.
F are well approximated by the regressions
and
respectively (see the black lines in
Figure 22 and
Figure 23).
It can be easily seen in
Table 2 and
Figure 22 that
σeq,max equals the tensile strength of the ABS material (30 MPa—see
Table 1) for a traction force 600 N <
Fcr < 750 N. This critical load results from Equation (1) as soon as the replacement
σeq,max = 30 MPa and is as follows:
When interpreting results, two main considerations need to be taken into account:
The load of 750 N is impractical and will probably never occur while using the prosthesis as intended. It would require an equivalent mass of ~75 kg to hang off the prosthesis end effector, which would be impossible to bear for the patient, and the socket would probably slide off the stump first before the prosthesis is actually damaged;
The simulation does not consider dependence of properties of ABS material manufactured using FDM technology on layered deposition process parameters, such as build orientation or layer thickness and many others, noted in many publications [
26,
27]. The real tensile strength of ABS (as well as impact and bending strengths) would be therefore significantly lower than the declared values by the producer, depending mostly on build orientation and also other process parameters.
Also, it is important to notice that only one loading scenario was considered in the analysis, with other load types and locations, and different areas of prosthesis could be in danger of breaking. Special attention should be paid to the wrist joint area, as the nut and bolt connection could be also a weak point when loading the prosthesis.
As such, effective recommended load, in authors’ opinion, should not exceed the third load case (450 N), and the equivalent stress in prosthesis mechanical parts made using FDM technology should not exceed 20 MPa, to avoid risk of breaking or otherwise damaging the prosthesis mid-activity. The force sensor should be able to answer what are the real loads during the prosthesis use.
3.2. Manufacturing and Assembly Results
All the parts were manufactured successfully at first attempt. As expected, the printing time was about 21 h for the socket and about 33 h for the other elements, with the material consumption also being generally compatible with the initial estimation. The obtained model required additional post-processing consisting of removal of the supports produced during printing. All the parts were manufactured in a stable manner, without any large errors or deformations. Minor problems occurred at one end of the forearm (contact surface with the socket part), and it was slightly deformed and needed some manual post-processing for better fitting and strength.
The assembly process started with putting together mechanical parts of the prosthesis, starting from the end effector (modified C-shape), with a simplified wrist joint connecting it to the forearm part. The assembled module is presented in
Figure 24.
The second step was putting together the electronic system and placing it inside cavities of the forearm. At this step, some adjustments were made to assure better fitting and access to the mounting elements, so that disassembly of mechanical components of the prosthesis is possible without removing the electronic part (easy access with tools). Also, cable feedthroughs had to be made slightly wider, to accommodate for the cable connections between modules. These efforts are presented in
Figure 25.
In the three cavities in the prosthesis forearm, the electronic components placed were as follows:
The first narrow hole near the wrist was used for SD card reader and output of USB cable for power source (
Figure 26a);
The second, central hole at the forearm was used as a hosting place for the main microcontroller (
Figure 26b);
The third, widest opening near the connection to the socket was used to mount the force sensor controller and the IMU device (
Figure 26c).
The assembly required using standard manual and power tools. Also, some electronic connections needed to be reinforced by soldering.
In the final stage of the assembly, the socket was assembled with the other parts. This was made mechanically, with screws. After joining the parts together, the force sensor was screwed to both the parts. As there were slight discrepancies in mounting hole distances between project and printout, the force sensor had an initial pre-tension, which was then accounted for in the data gathering algorithm. The complete prosthesis is shown in
Figure 27.
Table 4 contains a summary of working times of the subsequent stages of prosthesis development as assumed for a single patient, excluding the design phase.
3.3. Testing Results and Discussion
The data obtained from laboratory testing (two exemplary courses) are presented in
Figure 28 and
Figure 29. It is visible that all the events were registered correctly, both in terms of position and force change. It is noteworthy that the registered forces with simulated usage are very low (not exceeding 4 N). However, no dynamic scenarios (such as hitting against hard objects or lifting a heavy weight) were considered at this point, so that was to be expected.
The tests with the patient went without any disturbances, and the prosthesis worked as expected, and the patient was comfortable with it and was able to operate the vehicle without any problems. However, as compared with the mechanical version, it was observed by the patient and the researchers that the electronic parts (especially the force sensor beam and SD card reader) stick outside the prosthesis main shell and could interfere with the clothing (long sleeves). Also, proper covering would be needed to prevent damage to the electronics via unfavourable atmospheric conditions (such as rain or snow). Another issue might be the need to use and attach a power bank properly (e.g., at patient’s belt or in pocket), which might pose problems with ergonomics. These design considerations will be taken into account in the next version of the prosthesis.
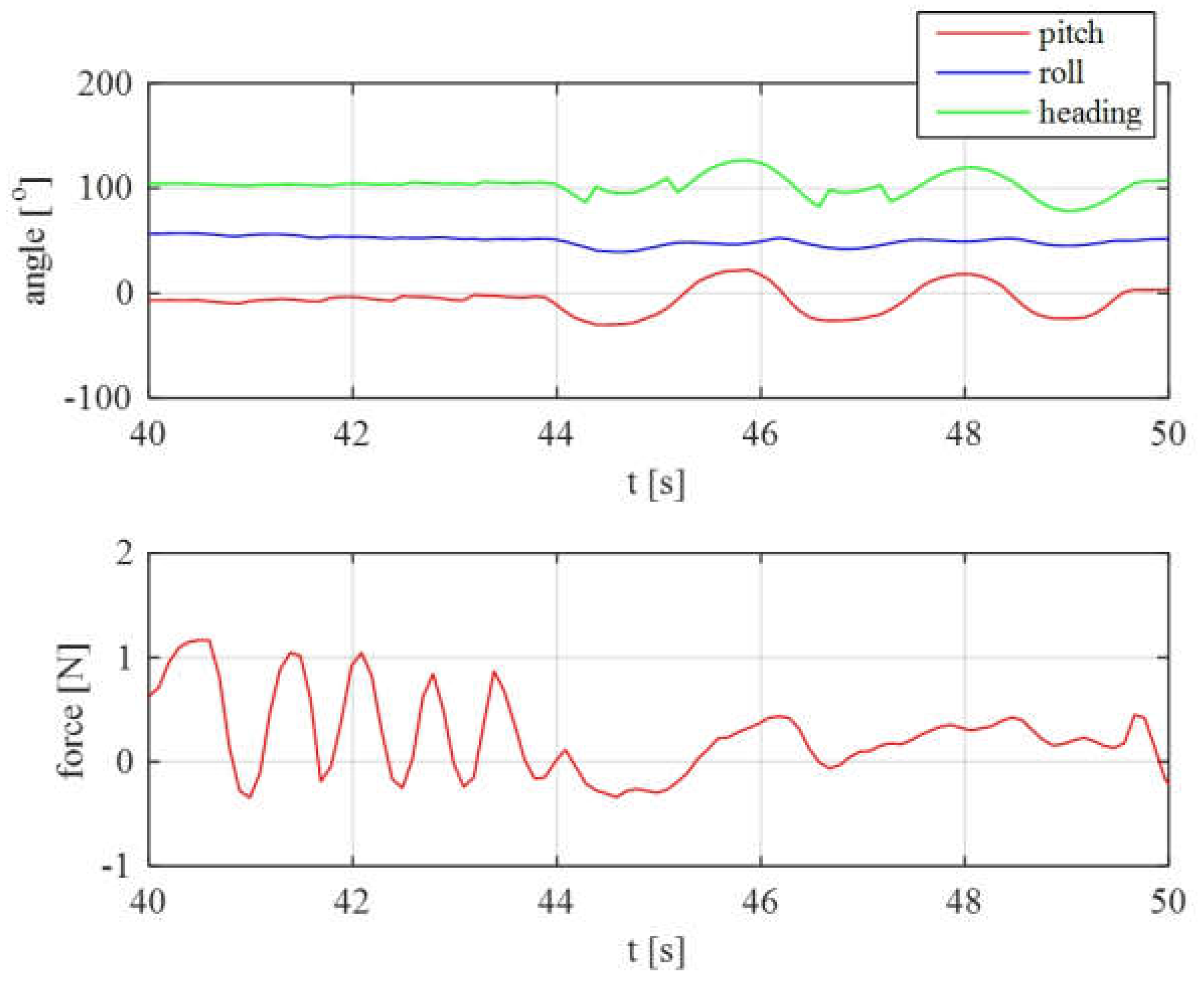
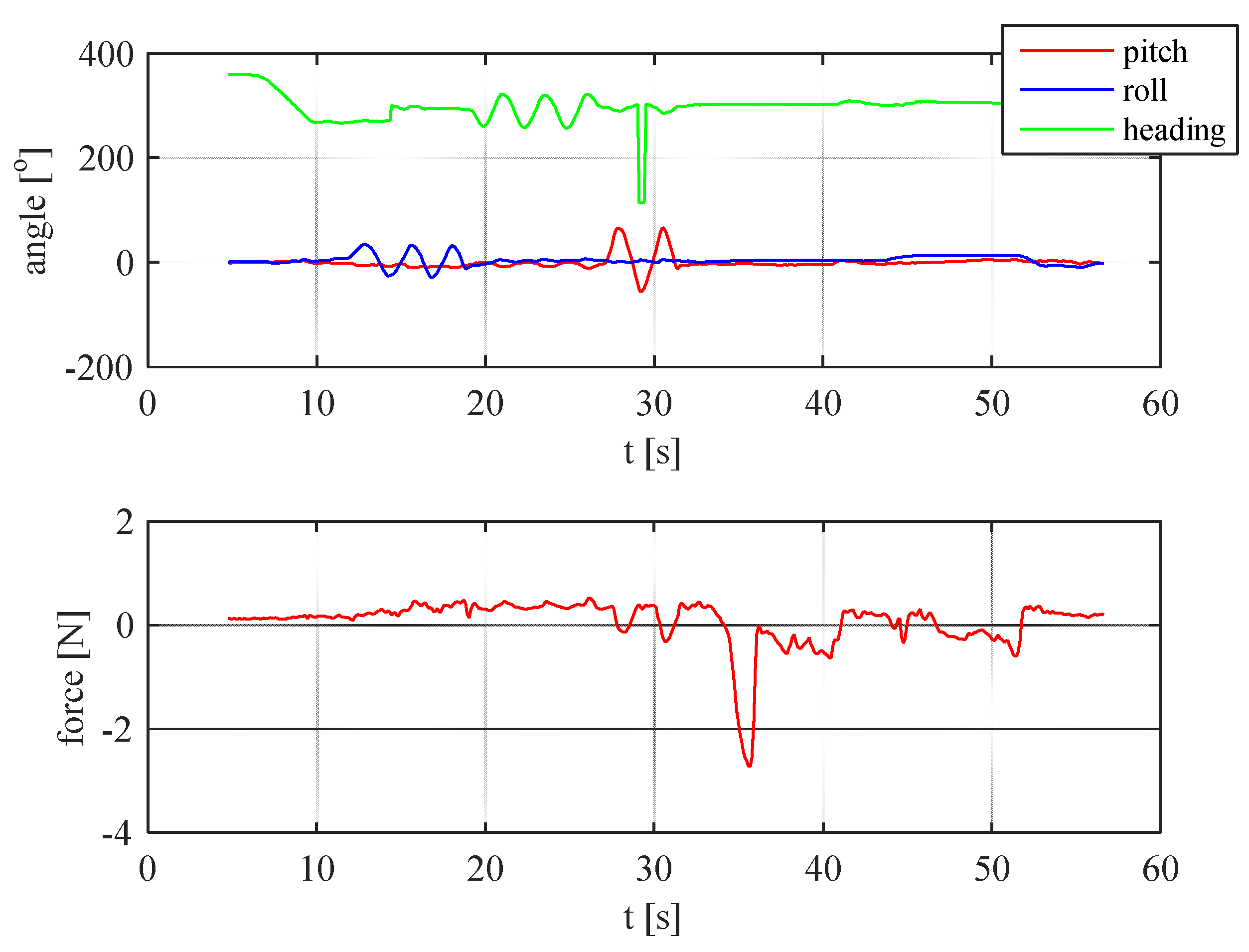
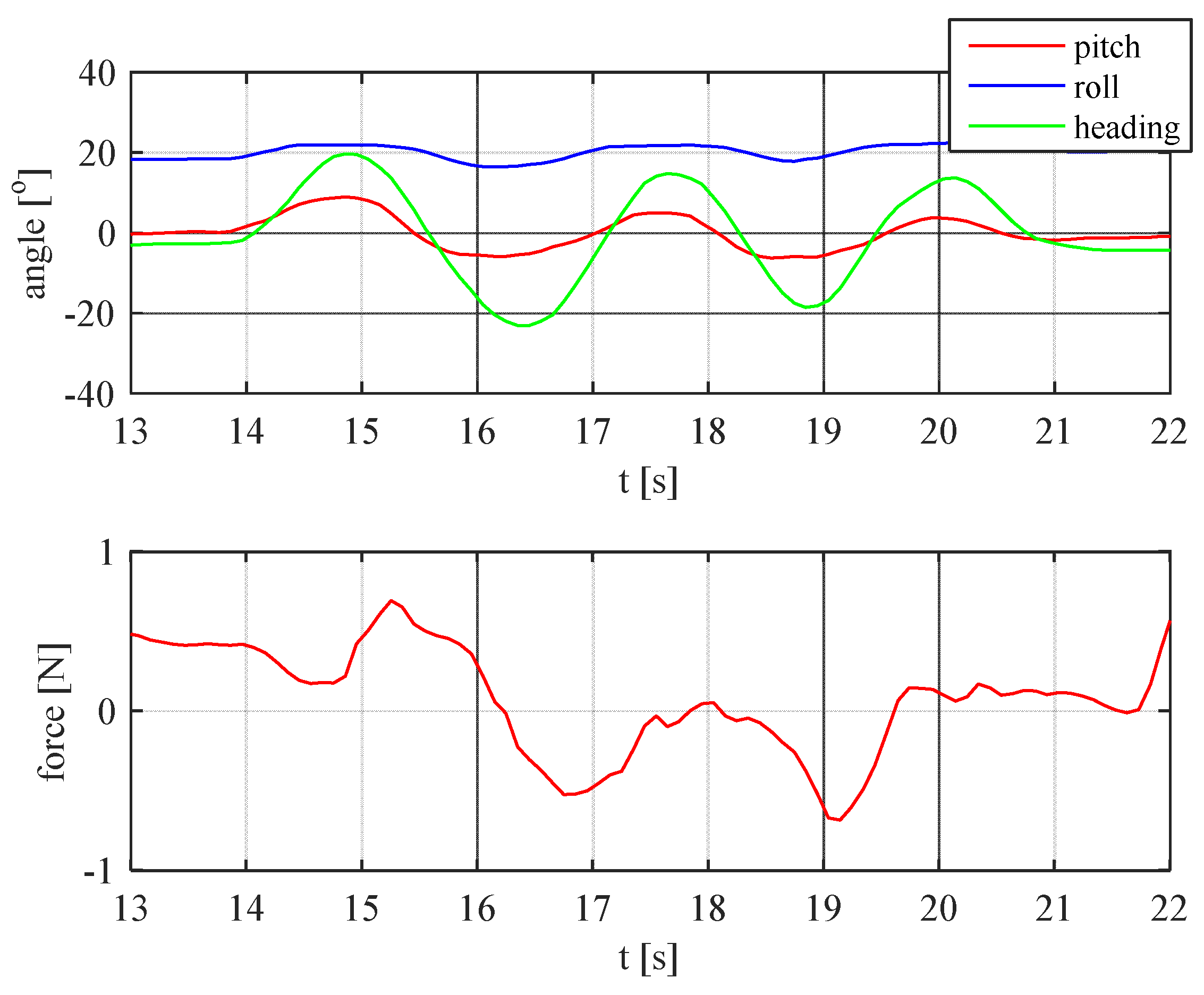
The data were gathered properly, without any breaks and improper data points. The data plots were recorded from two static tests, and the dynamic test (ride) are presented in
Figure 32,
Figure 33 and
Figure 34.
In
Figure 32 and
Figure 33, it is clearly visible that the nature of realized movements was different, by analysing the IMU data, and they correspond closely with real observed movements. Also, the force sensor readings are compatible with the IMU data, i.e., moments when the patient encountered resistance of the handlebar and changed the direction of the movement are clearly identifiable.
In
Figure 34, the nature of the patient’s movement can be also easily guessed from the Euler angles patterns recorded in the plot. The force changes also correspond closely with the movement. A single visible dip was a threshold that patient encountered in the middle of the test drive path.
It can be observed that the registered force during the test drive is considerably higher than in any other test. However, it is still very low (not exceeding 10 N). As such, the use of prosthesis in that manner is completely safe. However, the conditions in the test were also “safe” asthe patient did not drive very fast (no faster than 10 km/h) and did not encounter any serious terrain obstacles or height differences that might have caused the higher forces to occur. Further tests and more results are needed, and the patient has been given a prosthesis, and the results will be gathered and used in other experiments by the authors.
To sum up the results, it should be stated that the obtained sensor data represented the measured activity in an expected manner. The prosthesis has been evaluated successfully by obtaining positive feedback from the patient himself, also recording proper data with installed low-cost sensors, compliant with the observations. As such, its concept was approved, and initial prototype was assumed as ready for further implementation work. The prosthesis with its sensors can be considered as a successful prototype as the idea of low-cost personalized 3D printed bicycle prosthesis equipped with sensors for registering the course of driving was proven to be viable and will be continuously developed.
4. Conclusions
In the paper, a full process of development of a sensorised, specialized, low-cost customized prosthesis for an adult patient was presented. A customized design, for long-term use, was achieved, and the prosthesis was also converted into a mechatronic device, enabling to read sensor signals and monitor its use, albeit not yet in real time. Such designs are not common in the available literature. The authors realized it by a direct demand from the patients as all-purpose prostheses for adult patients with transhumeral upper limb amputations (or birth defects) are not well suited for sports activities, especially advanced cycling. After realizing a standard, modular design approach and extensive testing, the patient’s feedback was further incorporated, to create a fully customized, but still low-cost device that allows him to undertake more advanced activities.
The prosthesis (in its mechanical version, stripped of electronics) was fully accepted for use and the patient is currently using it for regular cycling activities. It is very difficult to compare our approach with existing solutions, as nothing similar (in terms of customization level, low cost, and induced accessibility) is commercially available. However, in patient’s own words, no producer was able to support him with a prosthesis of this capability during more than 40 years of his lifetime. That suggests the high level of acceptance, confirmed by another ongoing studies on a larger group of child and adult patients (some results are partially mentioned in [
21]). Results of these studies will be the topic of numerous future publications).
The presented sensorised device must, however, still be considered a prototype that needs further refining and improvement. Electronic components need to be hidden inside the prosthesis and protected from the atmospheric conditions. A suitable location must be found for the battery pack (so far it was carried by the patient in his pocket). The sensors arrangement must be also thought of more carefully—possibly the force sensor could be moved to the wrist–forearm joint, or another sensor could be added, to gather force data from various directions. In the software part, live streaming and visualization of results could be obtained by adding a Bluetooth or Wi-Fi module and connecting the prosthesis with a dedicated mobile application, e.g., to feed data back to the patient in time to avoid possible accidents or excessive wear of the prosthesis. All these improvements are considered and will be pursued in further iterations of the device.
In terms of accessibility and easiness of production, the concept of low-cost sensor-equipped prosthesis was successfully confirmed. Very simple and accessible electronic components were used, and they fulfilled their role. Time of assembly and programming did not exceed one working day. The longest (and most costly) part of the process is 3D printing of the parts, that took a total of 54 h. It could be performed in a considerably shorter time by using parallel manufacturing with several machines; however, single large parts such as the socket still take more than 20 h to produce, and that time would be difficult to reduce significantly. It is safe to assume that after automating the design (as performed by authors in the previous research cases), it would be possible to deliver working prostheses in one week, which is still a very short time when compared to commercial personalized specialized prosthetics.
It can be assumed, basing on patient’s positive feedback and authors’ experiences to date, that the personalized design of low-cost 3D printed prosthesis with sensors could be an invention that could change approach to amateur sports (especially cycling) by disabled people lacking one of their upper limbs, especially when they are also missing the elbow joint. Such prostheses could be automatically designed and produced cheaply, allowing wide groups of patients to regain capabilities of undertaking sports and other activities safely and comfortably. That is why this direction of research is promising and must be continued.
In the future, the integration of the prosthesis with a modular robotic gripper can be considered [
28,
29]. Fused deposition modelling, Stereolitography, or PolyJet can be thought of as suitable solutions for realizing low-cost prosthetics and orthotics using 3D printing [
30,
31,
32].