Physical, Thermal, and Mechanical Characterization of PMMA Foils Fabricated by Solution Casting
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods of Fabrication of Cast Foils and Characterization
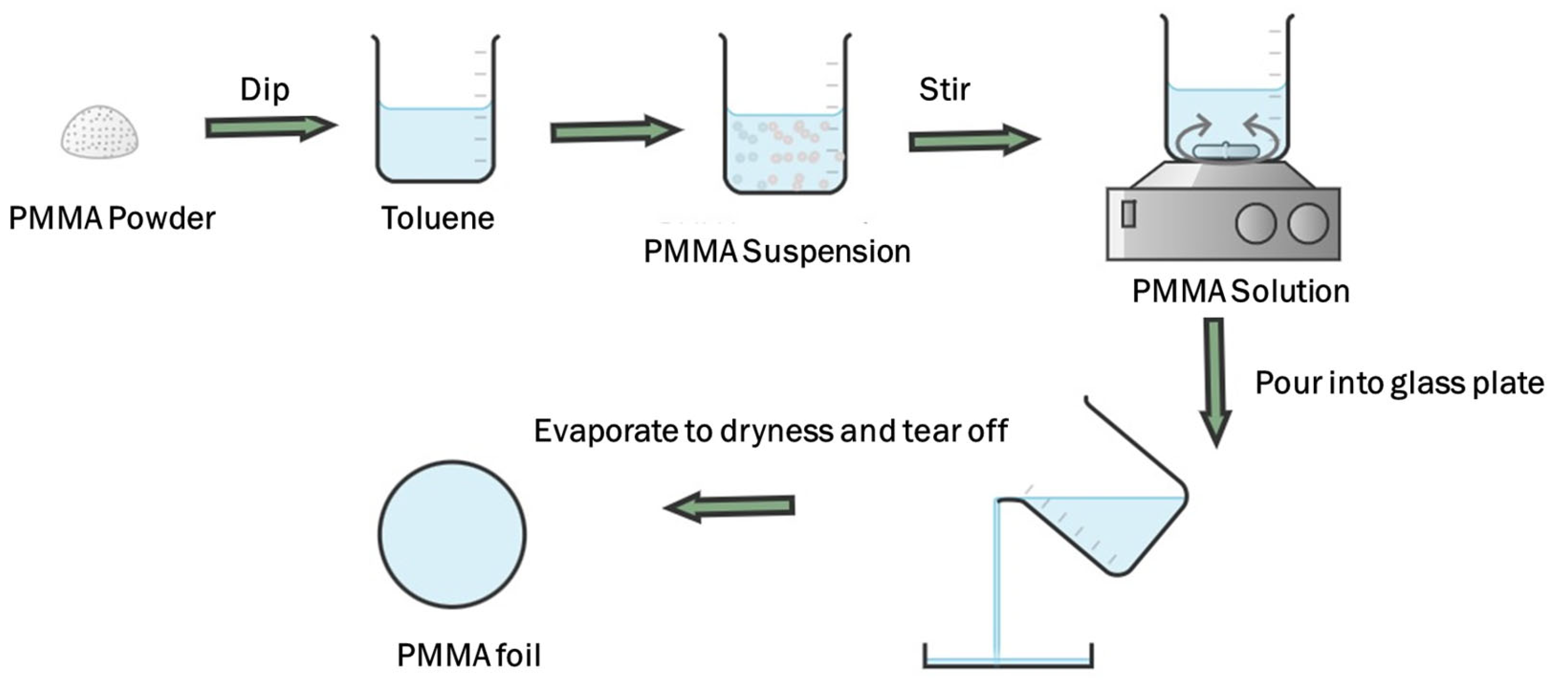
2.2.1. Sample Preparation Using the Casting Method
2.2.2. Characterization Techniques
2.2.3. Fourier-Transform Infrared Spectroscopy (FTIR-ATR)
2.2.4. Mechanical Characterization
3. Results
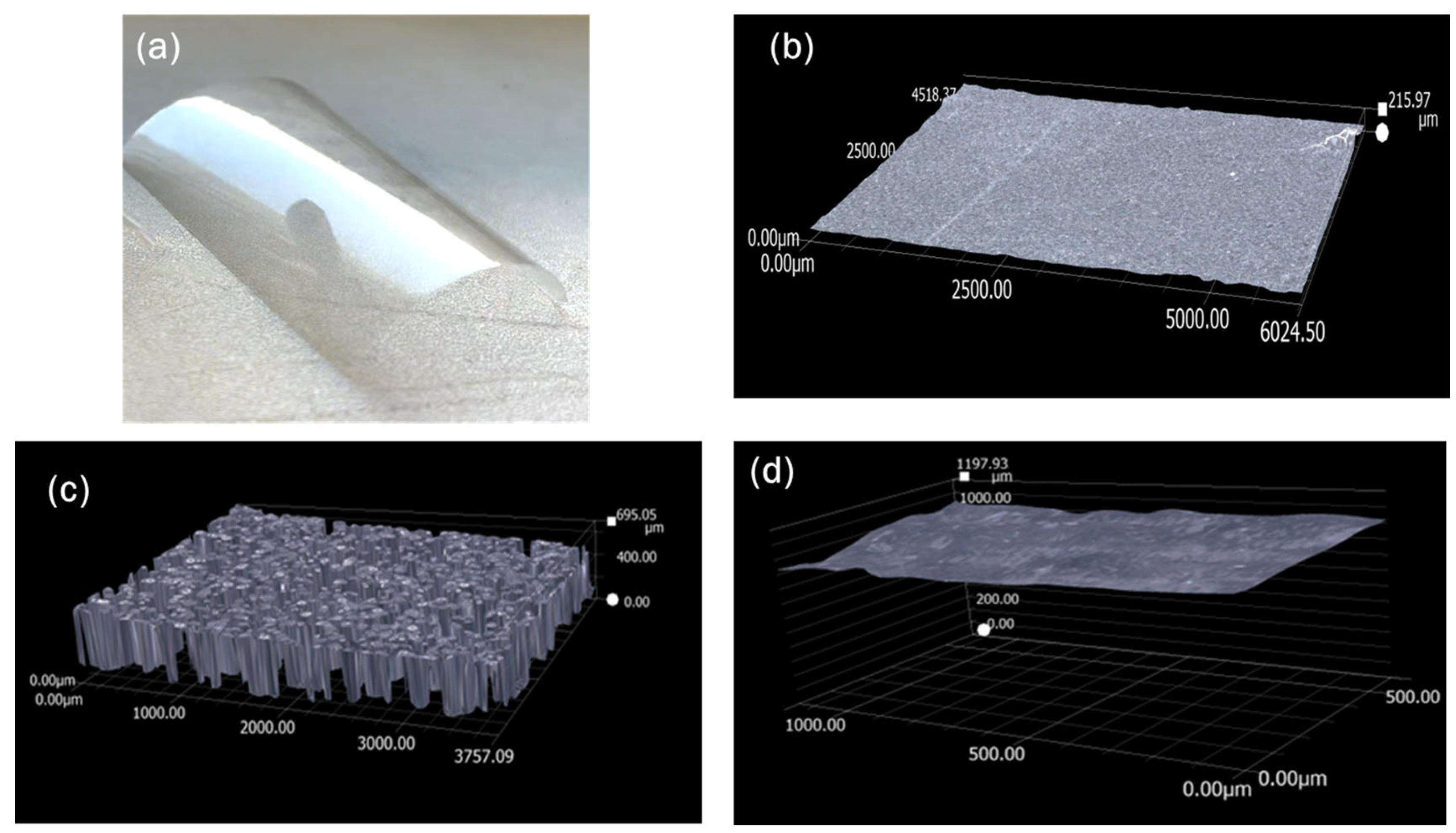
3.1. Surface Features of PMMA Powder, Foils, and As-Cast Foils
3.2. Thermal Characterization of PMMA Powder, Commercial Foil, and Prepared As-Cast Foils
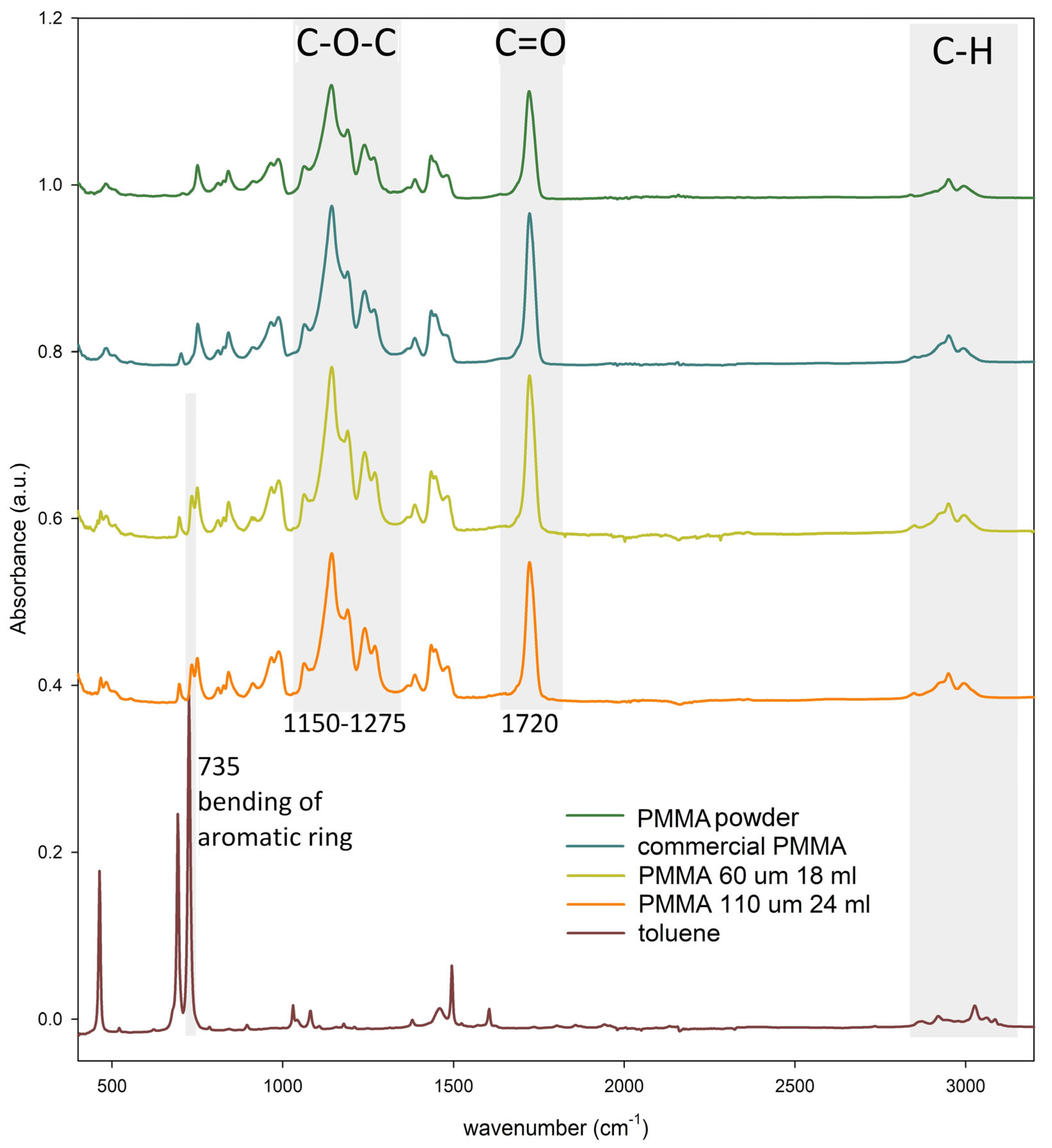
3.3. Structural Characterization of PMMA Foils
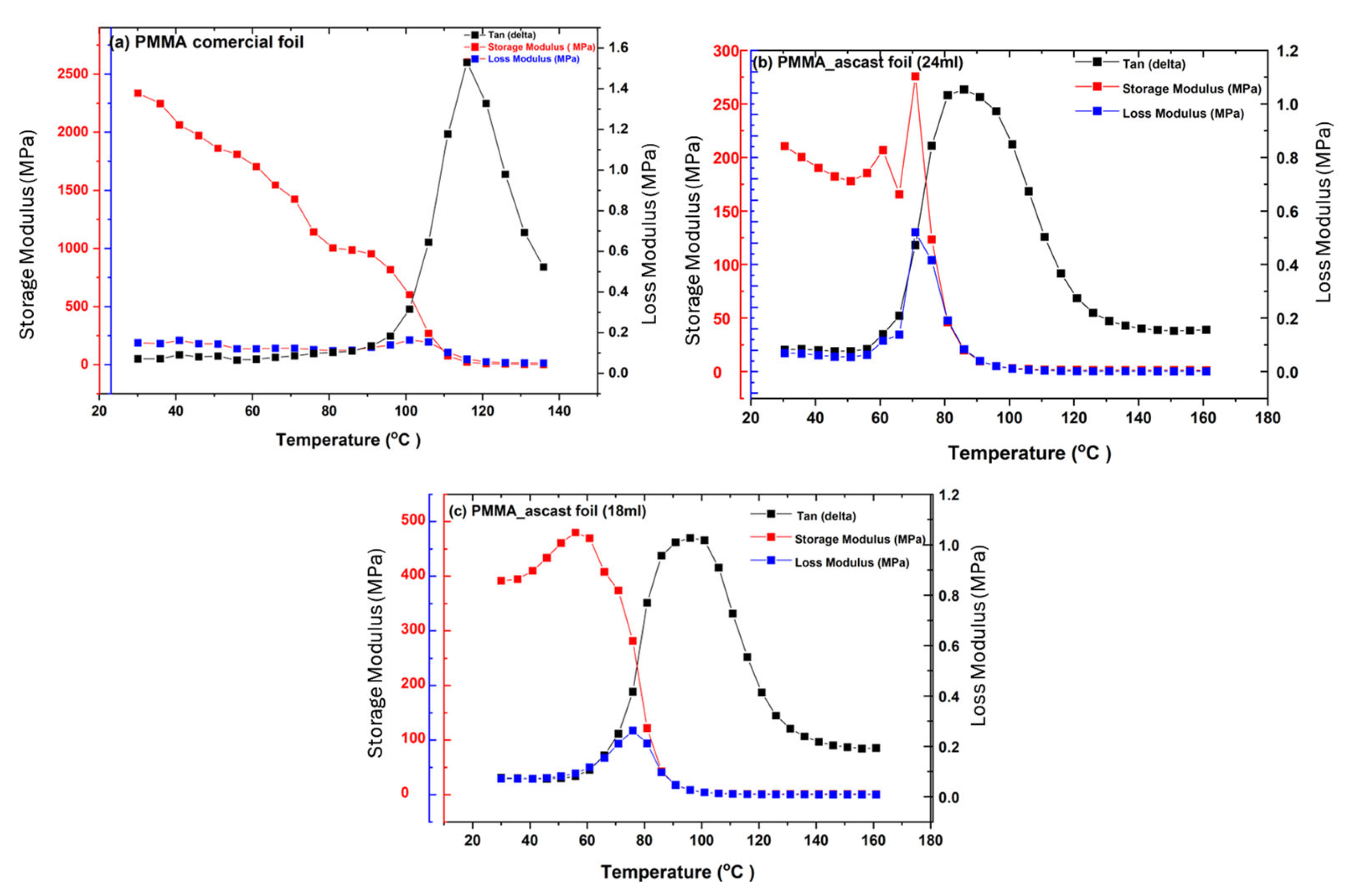
3.4. Dynamical Mechanical Characterization of Commercial PMMA Foil and Cast Foils
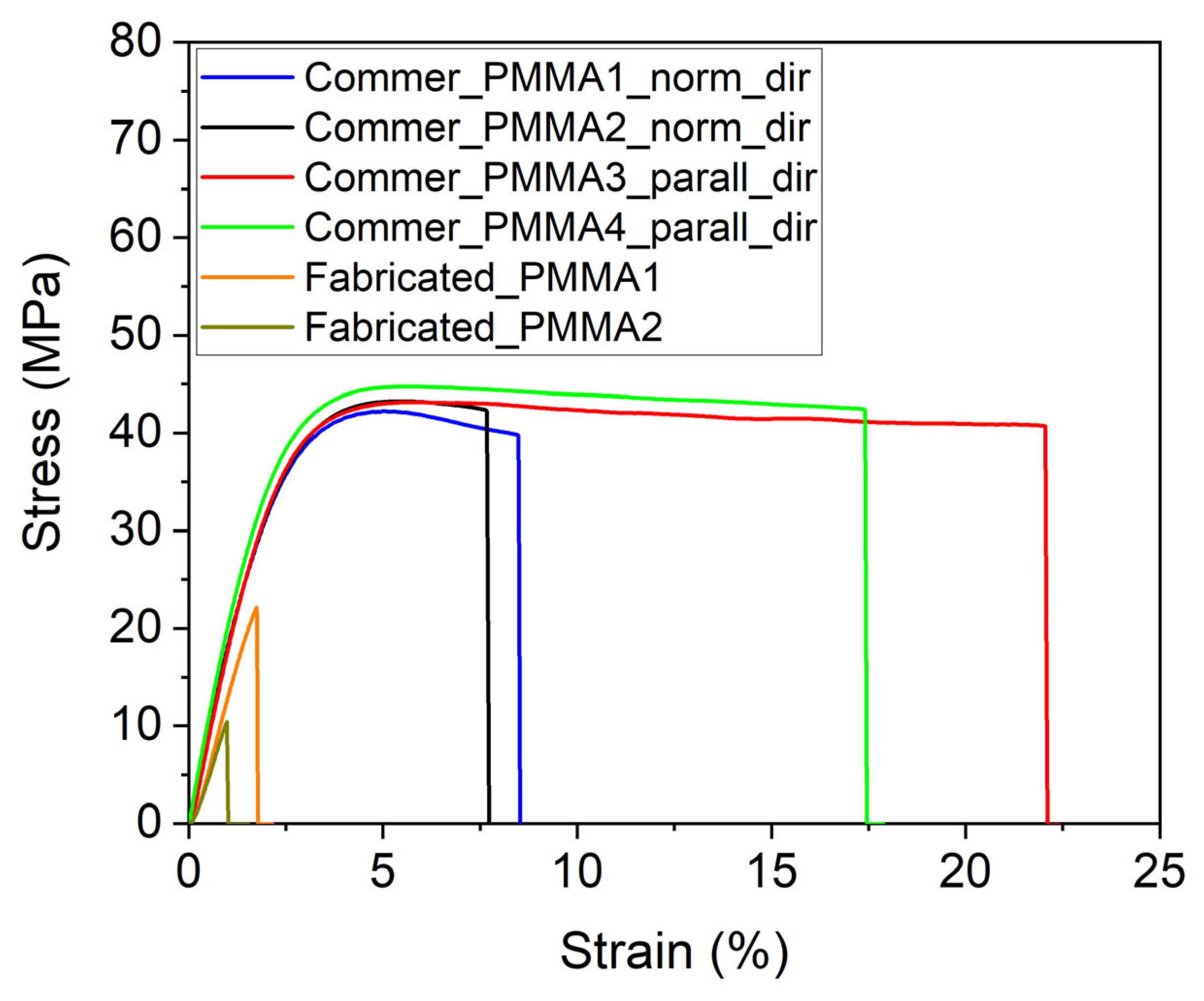
3.5. Tensile Test of PMMA Foils
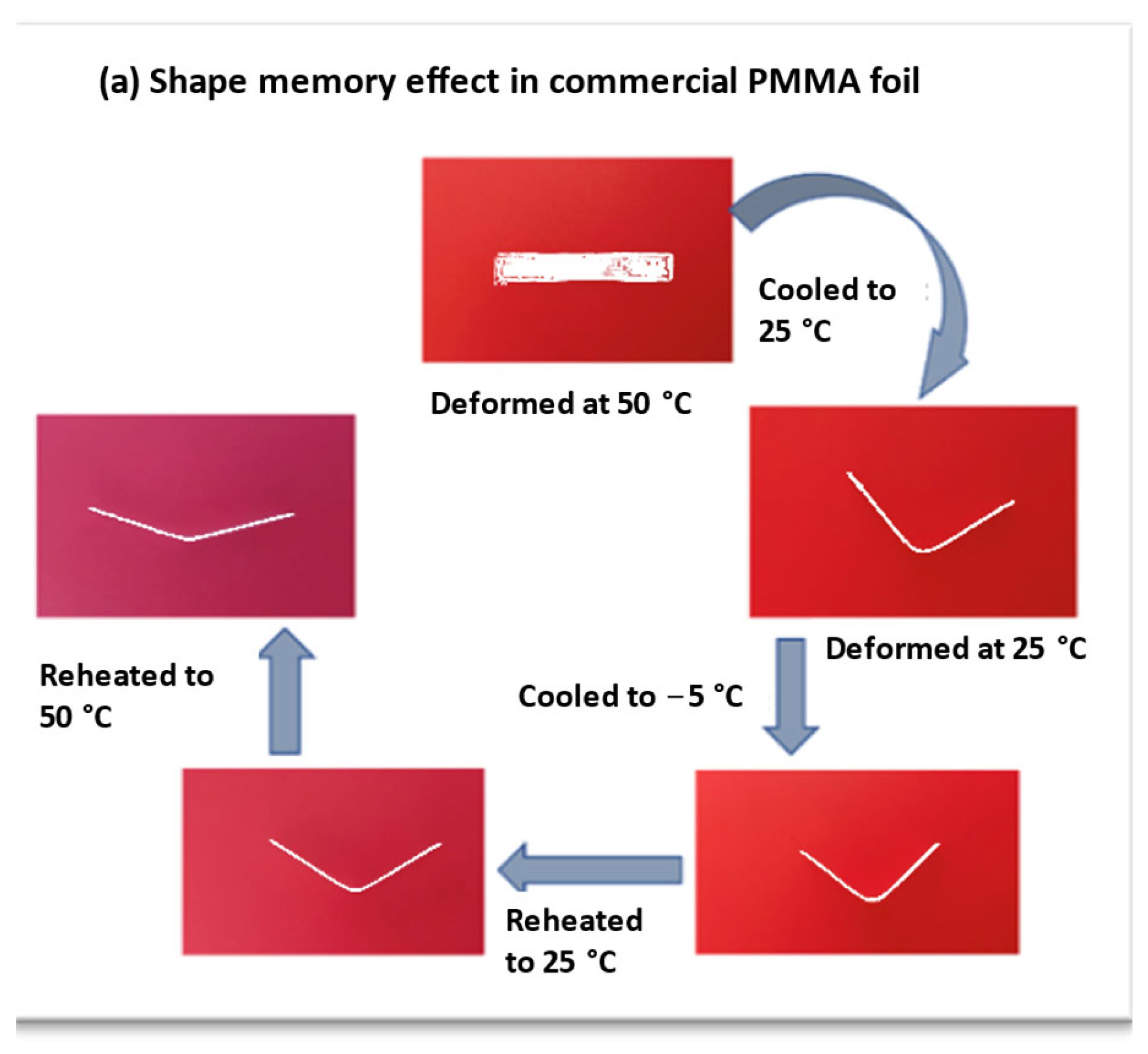
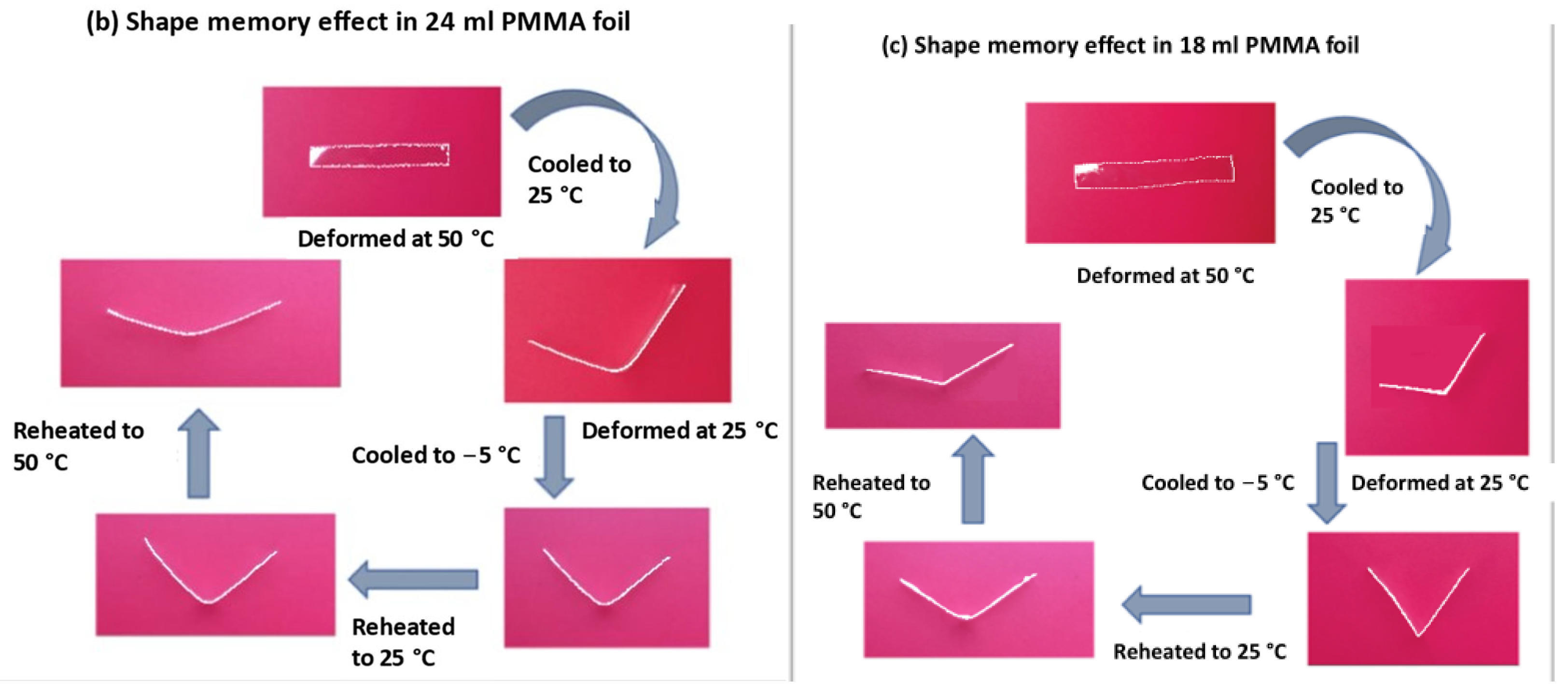
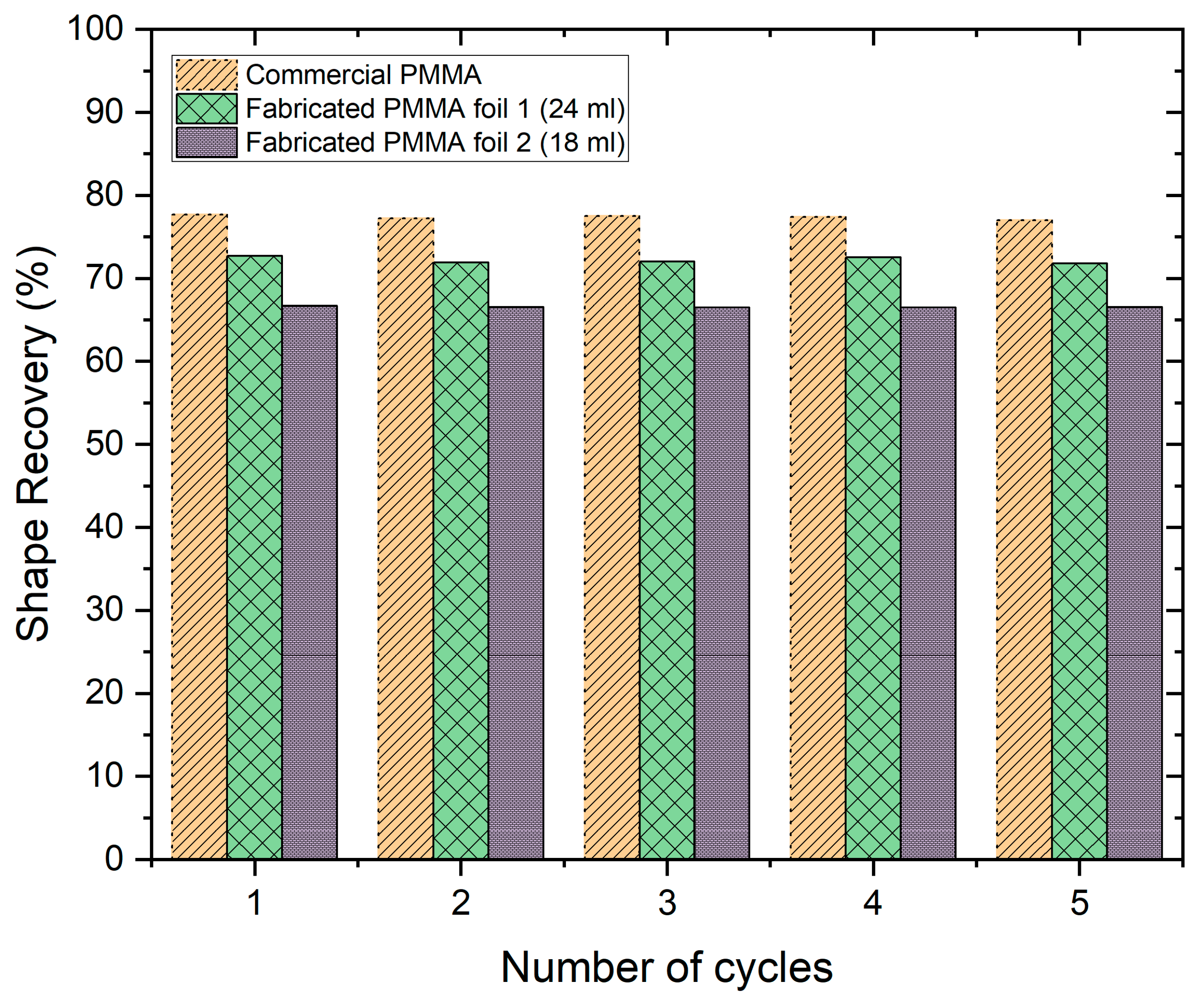
3.6. Shape Memory Effect of Commercial PMMA Foil and Cast Foils
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rosler, J.; Harders, H.; Baker, M. Mechanical behavior of polymers. In Mechanical Behaviour of Engineering Materials, Metals, Ceramics, Polymers, and Composites; Rosler, J., Harders, H., Bäker, M., Eds.; Springer: Berlin/Heidelberg, Germany, 2007; ISBN 3540734481, 9783540734482. [Google Scholar]
- Sugiyama, S.; Amaya, S.; Dao, D.V. Development of polymer MEMS process technology as an approach to a sustainable production system. Adv. Nat. Sci: Nanosci. Nanotechnol. 2012, 3, 015009. [Google Scholar] [CrossRef]
- Zhao, Y.; Cui, T. Fabrication of high-aspect-ratio polymer-based electrostatic comb drives using the hot embossing technique. J. Micromech. Microeng. 2003, 13, 430. [Google Scholar] [CrossRef]
- Haller, I.; Hatzakis, M.; Srinivasan, R. High-Resolution Positive Resists for Electron-Beam Exposure. IBM J. Res. Dev. 1968, 12, 251–256. [Google Scholar] [CrossRef]
- Borges, A.M.G.; Benetoli, L.O.; Licinio, M.A.; Zoldan, V.C.; Santos-Silva, M.C.; Assreuy, J.; Pasa, A.A.; Debacher, N.A.; Soldi, V. Polymers films with surfaces unmodified and modified by non-thermal plasma as new substrates for cell adhesion. Mater. Sci. Eng. C 2013, 33, 1315–1324. [Google Scholar] [CrossRef] [PubMed]
- Giacon, V.M.; Padilha, G.S.; Bartoli, J.R. Fabrication and characterization of polymeric optical by plasma fluorination process. Optik 2015, 126, 74–76. [Google Scholar] [CrossRef]
- Meyerhofer, D. Characteristics of resist films produced by spinning. J. Appl. Phys. 1978, 49, 3993–3997. [Google Scholar] [CrossRef]
- Schubert, D.W. Spin coating as a method for polymer molecular weight determination. Polym. Bull. 1997, 38, 177–184. [Google Scholar] [CrossRef]
- Samal, S.; Kosjakova, O. Surface feature of PMMA films on NiTi alloy substrate by the spin coating method. Ceram. Int. 2022, in press. [Google Scholar] [CrossRef]
- Samal, S. Interface failure and delamination resistance of fiber-reinforced geopolymer composite by simulation and experimental method. Cement Concr. Compos. 2022, 128, 104420. [Google Scholar] [CrossRef]
- Liu, Z.; Zhao, J.-H.; Liu, P.; He, J.-H. Tunable surface morphology of electrospun PMMA fiber using binary solvent. Appl. Surf. Sci. 2016, 364, 516–521. [Google Scholar] [CrossRef]
- Briscoe, B.; Akram, A.; Adams, M.; Johnson, S.; Gorman, D. The influence of solvent quality on the mechanical properties of thin cast isotactic poly(methyl methacrylate) coatings. J. Mat. Sci. 2002, 37, 4929–4936. [Google Scholar] [CrossRef]
- Bistac, S.; Schultz, J. Study of solution-cast films of PMMA by dielectric spectroscopy: Influence of the nature of the solvent on α and β relaxations. Int. J. Adhesion. Adhesives. 1997, 17, 197–201. [Google Scholar] [CrossRef]
- Yang, S.-Y.; Lee, M.-J. Poly (Methyl Methacrylate)-Containing Silver-Phosphate Glass Exhibits Potent Antimicrobial Activity without Deteriorating the Mechanical and Biological Properties of Dental Prostheses. Polymers 2023, 15, 297. [Google Scholar] [CrossRef]
- Pituru, S.M.; Greabu, M.; Totan, A.; Imre, M.; Pantea, M.; Spinu, T.; Tancu, A.M.C.; Popoviciu, N.O.; Stanescu, I.-I.; Ionescu, E. A Review on the Biocompatibility of PMMA-Based Dental Materials for Interim Prosthetic Restorations with a Glimpse into Their Modern Manufacturing Techniques. Materials 2020, 13, 2894. [Google Scholar] [CrossRef]
- Bae, G.; Park, T.; Song, I.-H. Surface Modification of Polymethylmethacrylate (PMMA) by Ultraviolet (UV) Irradiation and IPA Rinsing. Micromachines 2022, 13, 1952. [Google Scholar] [CrossRef]
- Cao, B.; Wu, P.; Zhang, W.; Liu, S.; Zhao, J. The Fabrication of High-Hardness and Transparent PMMA-Based Composites by an Interface Engineering Strategy. Molecules 2023, 28, 304. [Google Scholar] [CrossRef]
- Hong, P.D.; Huang, H.T.; Chou, C.M. Study of the solvent effect on miscibility between poly(vinyl chloride) and poly(methyl methacrylate) in the solution state—Viscometric measurements. Polym. Int. 2000, 49, 407. [Google Scholar] [CrossRef]
- Bubb, D.M.; Papantonakis, M.; Collins, B.; Brookes, E.; Wood, J.; Gurudas, U. The influence of solvent parameters upon the surface roughness of matrix assisted laser deposited thin polymer films. Chem. Phys. Lett. 2007, 448, 194. [Google Scholar] [CrossRef]
- González-Benito, J.; Koenig, J.L. Nature of PMMA dissolution process by mixtures of acetonitrile/alcohol (poor solvent/nonsolvent) monitored by FTIR-imaging. Polymer 2006, 47, 3065–3072. [Google Scholar] [CrossRef]
- Samal, S.; Tyc, O.; Heller, L.; Šittner, P.; Malik, M.; Poddar, P.; Catauro, M.; Blanco, I. Study of Interfacial Adhesion between Nickel-Titanium Shape Memory Alloy and a Polymer Matrix by Laser Surface Pattern. Appl. Sci. 2020, 10, 2172. [Google Scholar] [CrossRef]
- Samal, S.; Kosjakova, O.; Vokoun, D.; Stachiv, I. Shape Memory Behaviour of PMMA-Coated NiTi Alloy under Thermal Cycle. Polymers 2022, 14, 2932. [Google Scholar] [CrossRef] [PubMed]
- Pilch, J.; Šittner, P. A Method of Heat Treatment And/or Inspection of Functional Mechanical Properties, Particularly Transformation Strain And/or Strength, of Shape Memory Alloy Wires and Apparatus for the Application of This Method. US Patent 20120018413A1, 26 January 2012. [Google Scholar]
- Agrawal, S.; Patidar, D.; Dixit, M.; Sharma, K.; Saxena, N.S. Investigation of Thermo-mechanical Properties of PMMA. AIP Conf. Proc. 2010, 1249, 79. [Google Scholar] [CrossRef]
- Patra, N.; Salerno, M.; Diaspro, A.; Athanassiou, A. Effect of solvents on the dynamic viscoelastic behavior of poly(methyl methacrylate) film prepared by solvent casting. J. Mater. Sci. 2011, 46, 5044–5049. [Google Scholar] [CrossRef]
- Patra, N.; Barone, A.; Salerno, M. Solvent effects on the thermal and mechanical properties of poly(methylmethacrylate) casted from concentrated solutions. Adv. Polym. Technol. 2011, 30, 12–20. [Google Scholar] [CrossRef]
- Zhou, Y.; Ren, L.; Zang, J.; Zhang, Z. The Shape Memory Properties and Actuation Performances of 4D Printing Poly (Ether-Ether-Ketone). Polymers 2022, 14, 3800. [Google Scholar] [CrossRef]
- Samal, S.; de Prado, E.; Tyc, O.; Sittner, P. Shape Setting in super-elastic NiTi ribbon. IOP Conf. Ser. Mater. Sci. Eng. 2018, 461, 012075. [Google Scholar] [CrossRef]
- Behl, M.; Razzaq, M.Y.; Lendlein, A. Multifunctional shape-memory polymers. Adv. Mater. 2010, 22, 3388–3410. [Google Scholar] [CrossRef]
- Smith, N.A.; Antoun, G.G.; Ellis, A.B.; Crone, W.C. Improved adhesion between nickel–titanium shape memory alloy and a polymer matrix via silane coupling agents. Compos. Part A Appl. Sci. Manuf. 2004, 35, 1307–1312. [Google Scholar] [CrossRef]
- Hebali, H.; Chikh, A.; Bousahla, A.A.; Bourada, F.; Tounsi, A.; Benrahou, K.H.; Muzamal, H.; Tounsi, A. Effect of the variable visco-Pasternak foundations on the bending and dynamic behaviors of FG plates using integral HSDT model. Geomech. Eng. 2022, 28, 49–64. [Google Scholar] [CrossRef]
- Van Vinh, P.; Van Chinh, N.; Tounsi, A. Static bending and buckling analysis of bi-directional functionally graded porous plates using an improved first-order shear deformation theory and FEM. Eur. J. Mech. A Solids 2022, 96, v104743. [Google Scholar] [CrossRef]


| Sample | PMMA/Toluene Ratio (g/mL) | Thickness (mm) | Glass Transition Temperature (Tg) |
|---|---|---|---|
| PMMA powder | - | Powder | 117 °C |
| Commercial sample | unknown | 0.25 | 110 °C |
| Sample 1 | 1:18 | 0.21 | 94 °C |
| Sample 2 | 1:24 | 0.37 | 90 °C |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Samal, S.; Svomova, B.; Spasovová, M.; Tyc, O.; Vokoun, D.; Stachiv, I. Physical, Thermal, and Mechanical Characterization of PMMA Foils Fabricated by Solution Casting. Appl. Sci. 2023, 13, 1016. https://doi.org/10.3390/app13021016
Samal S, Svomova B, Spasovová M, Tyc O, Vokoun D, Stachiv I. Physical, Thermal, and Mechanical Characterization of PMMA Foils Fabricated by Solution Casting. Applied Sciences. 2023; 13(2):1016. https://doi.org/10.3390/app13021016
Chicago/Turabian StyleSamal, Sneha, Barbora Svomova, Monika Spasovová, Ondřej Tyc, David Vokoun, and Ivo Stachiv. 2023. "Physical, Thermal, and Mechanical Characterization of PMMA Foils Fabricated by Solution Casting" Applied Sciences 13, no. 2: 1016. https://doi.org/10.3390/app13021016
APA StyleSamal, S., Svomova, B., Spasovová, M., Tyc, O., Vokoun, D., & Stachiv, I. (2023). Physical, Thermal, and Mechanical Characterization of PMMA Foils Fabricated by Solution Casting. Applied Sciences, 13(2), 1016. https://doi.org/10.3390/app13021016








