Investigation and Spatial Distribution of Hard Ticks by Geographical Information System (GIS) in the Region of Istria, Croatia
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Sampling Procedures
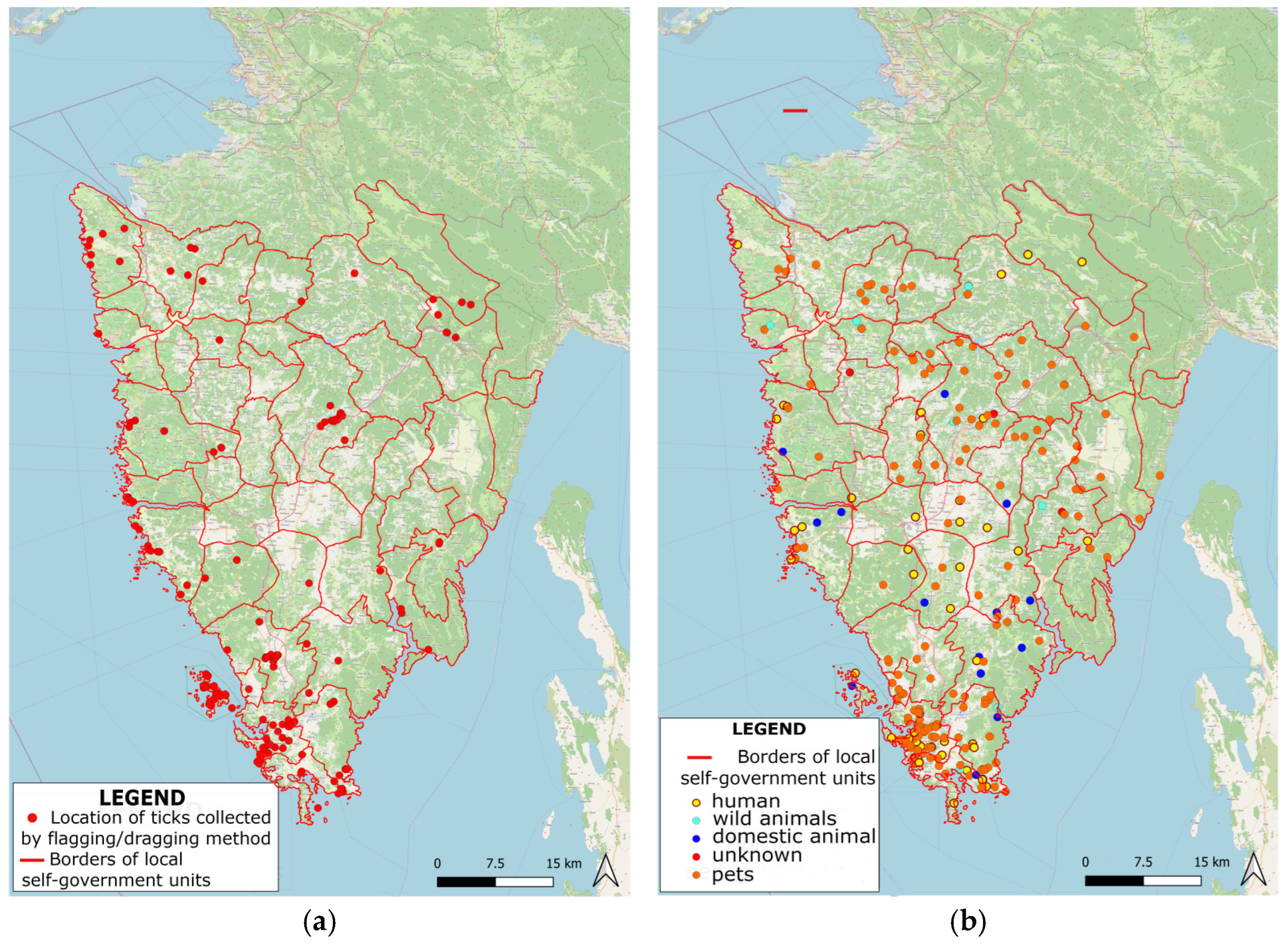
2.2.1. Flagging/Dragging Method
2.2.2. Animal/Human Host Method
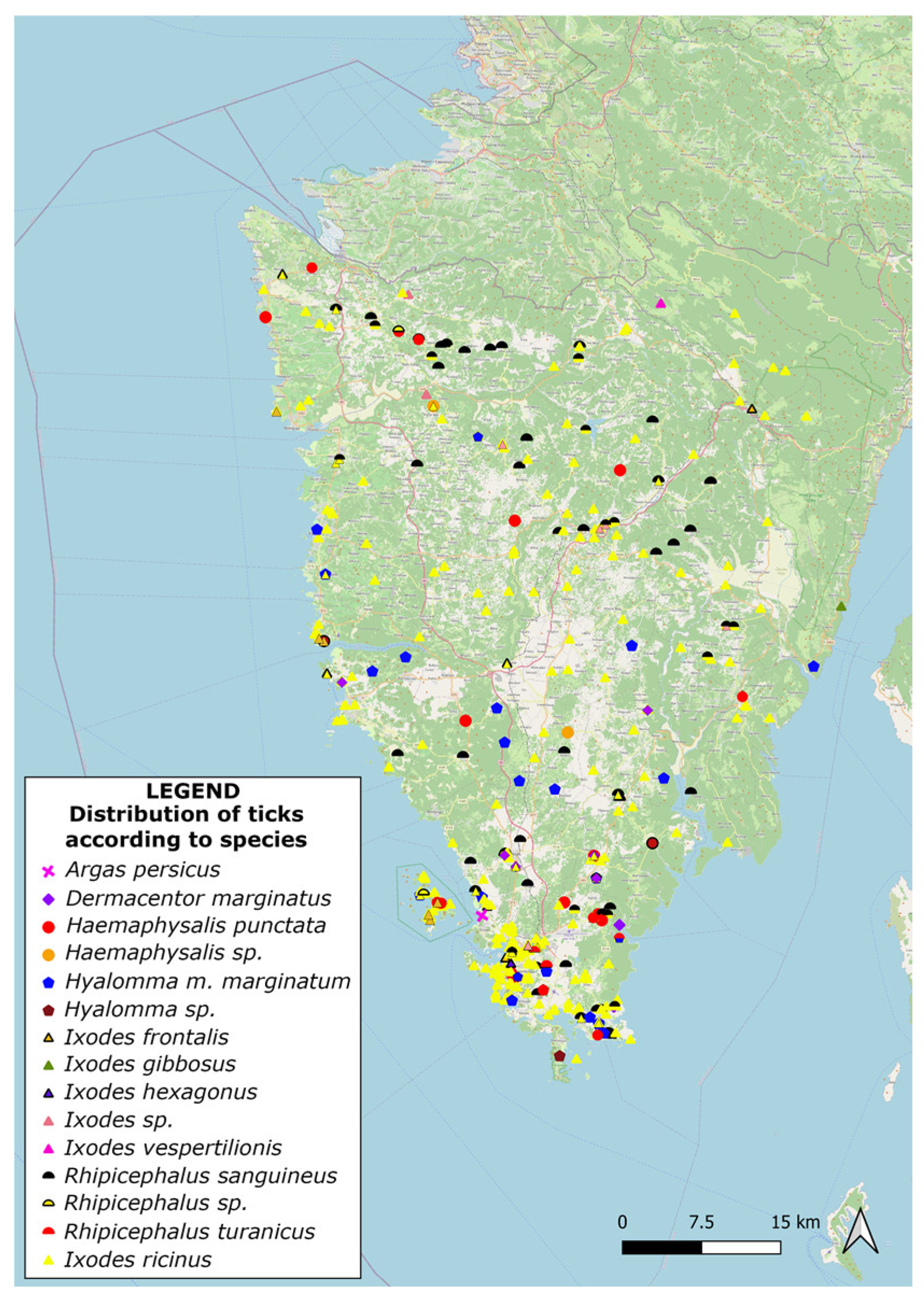
2.2.3. Morphological Determination of Hard Ticks
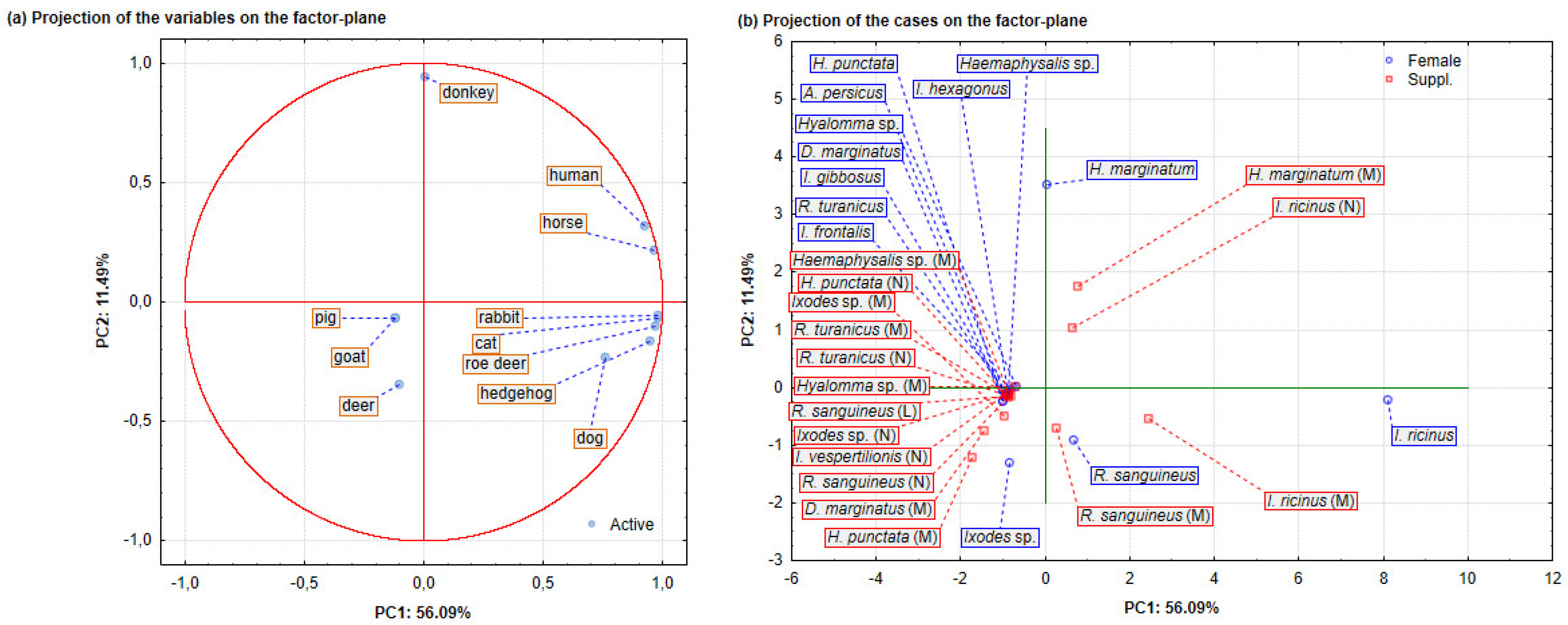
2.3. Data Analysis
3. Results and Discussion
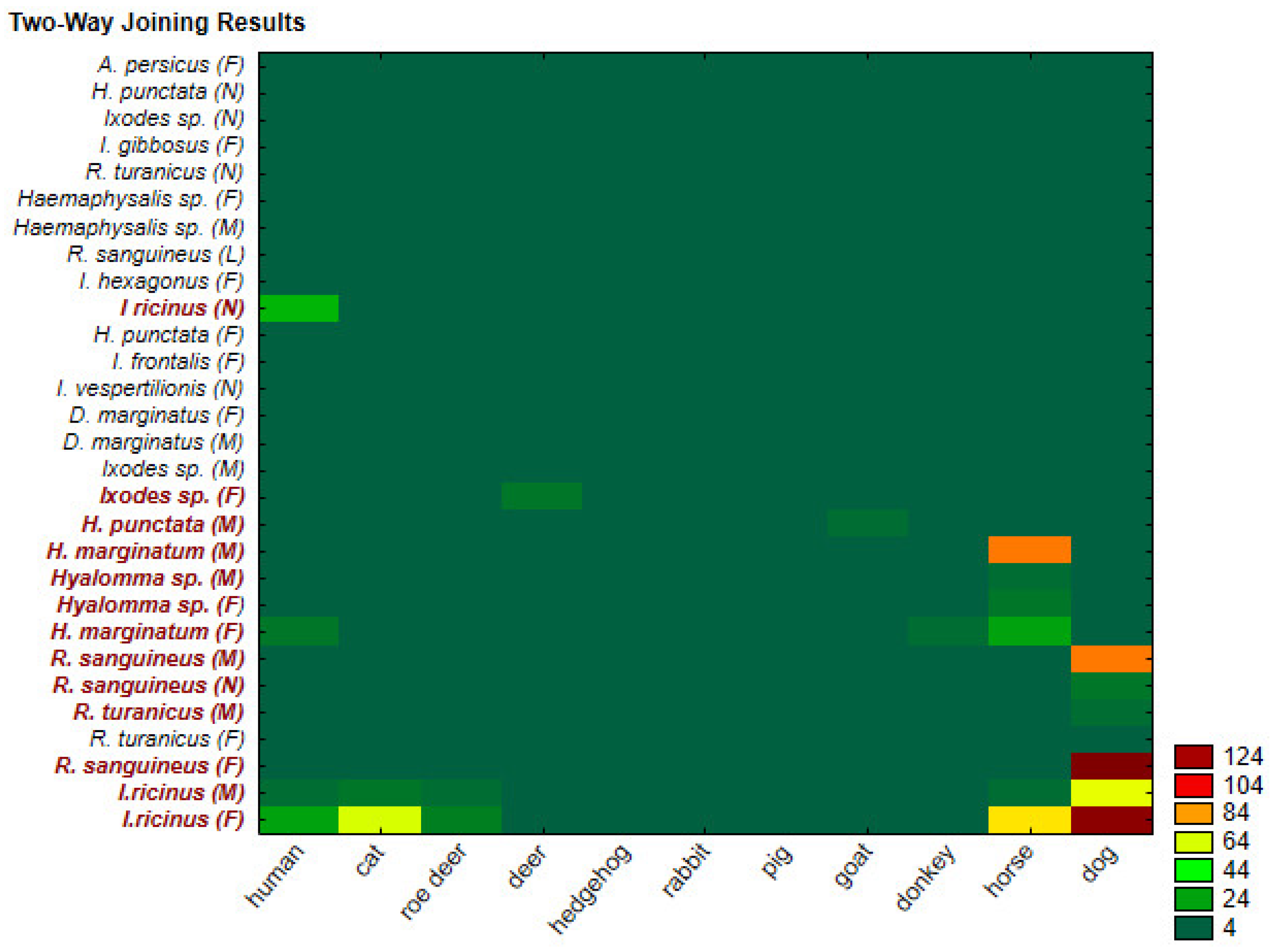
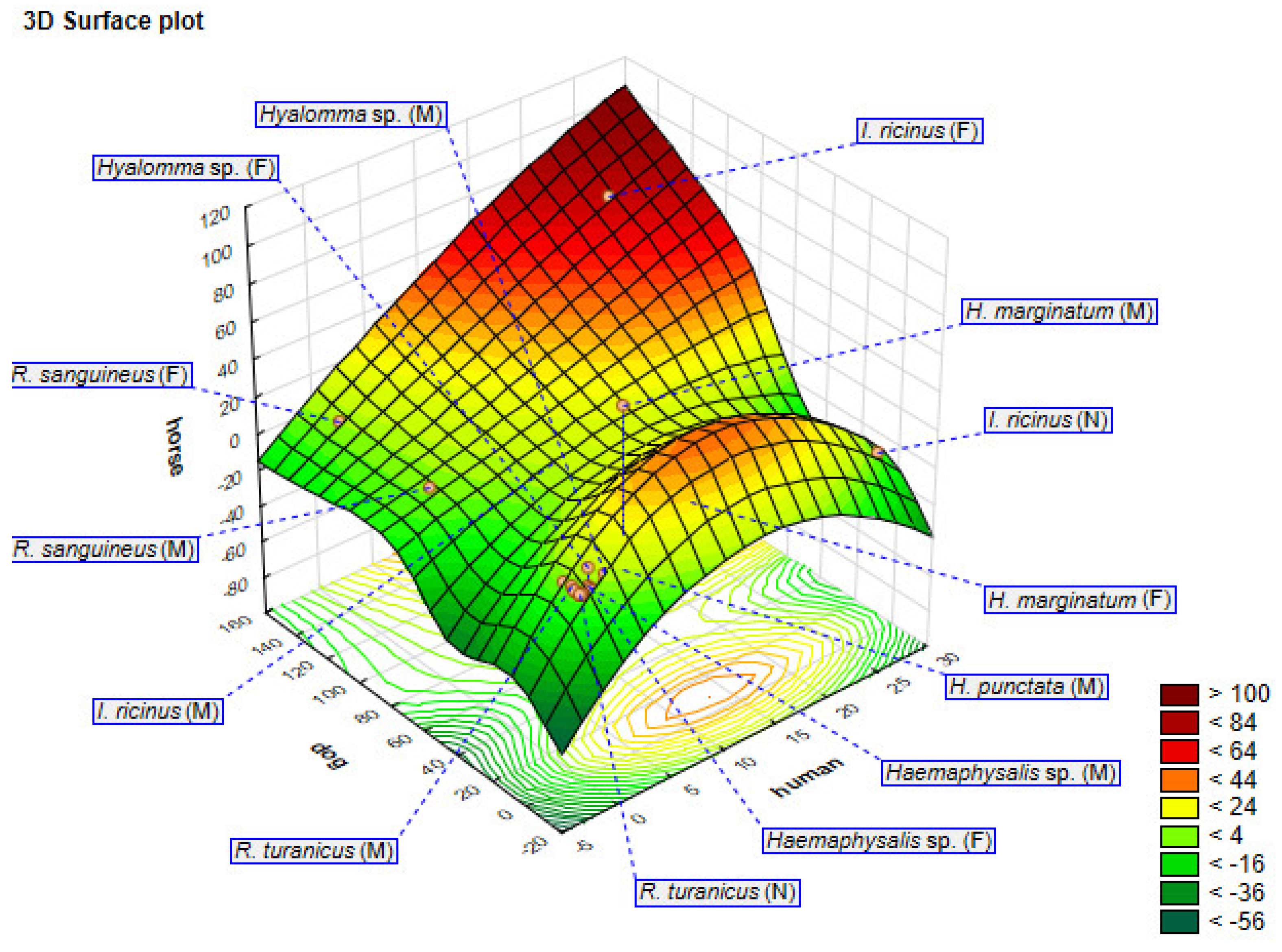
Spatial Distribution and Morphological Identification of Hart Ticks
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mulić, R.; Petković, B.; Klismanić, Z.; Jerončić, I. Tick-borne diseases in the Republic of Croatia. Lijec. Vjesn. 2011, 133, 89–95. [Google Scholar] [PubMed]
- De la Fuente, J.; Estrada-Pena, A.; Venzal, J.M.; Kocan, K.M.; Sonenshine, D.E. Overview: Ticks as vectors of pathogens that cause disease in humans and animals. Front. Biosci. 2008, 13, 6938–6946. [Google Scholar] [CrossRef] [PubMed]
- Boulanger, N.; Boyer, P.; Talagrand-Reboul, E.; Hansmann, Y. Ticks and tick-borne diseases. Med. Mal. Infect. 2019, 49, 87–97. [Google Scholar] [CrossRef] [PubMed]
- Ogden, N. Changing geographic ranges of ticks and tick-borne pathogens: Drivers, mechanisms and consequences for pathogen diversity. Front. Cell. Infect. Microbiol. 2013, 3, 46. [Google Scholar] [CrossRef] [PubMed]
- Estrada-Pena, A.; Ayllón, N.; Fuente, J. Impact of climate trends on tick-borne pathogen transmission. Front. Physiol. 2012, 3, 64. [Google Scholar] [CrossRef] [PubMed]
- Randolph, S.E.; Rogers, D.J. Fragile transmission cycles of tick-borne encephalitis virus may be disrupted by predicted climate change. Proc. R. Soc. B Biol. Sci. 2000, 267, 1741–1744. [Google Scholar] [CrossRef] [PubMed]
- Gebreyes, W.A.; Dupouy-Camet, J.; Newport, M.J.; Oliveira, C.J.; Schlesinger, L.S.; Saif, Y.M.; Kariuki, S.; Saif, L.J.; Saville, W.; Wittum, T.; et al. The global One Health paradigm: Challenges and opportunities for tackling infectious diseases at the human, animal, and environment interface in low-resource settings. PLoS Negl. Trop. Dis. 2014, 8, e3257. [Google Scholar] [CrossRef]
- Romanović, M.; Mulić, R.; Ropac, D. Doprinos poznavanju medicinski važnih člankonožaca na otocima i priobalju Republike Hrvatske. Entomol. Croat. 1999, 4, 71–80. [Google Scholar]
- Nava, S.; Gugliemone, A.A.; Mangold, A.J. An overview of systematic and evolution of ticks. Front. Biosci. 2009, 14, 2857–2877. [Google Scholar] [CrossRef]
- Richter, B. Medicinska Parazitologija; A.B.D. Merkur: Zagreb, Croatia, 2002; pp. 145–151. [Google Scholar]
- Golubić, D.; Rijepkema, S.; Tkalec-Malkovec, N.; Ružić, E. Epidemiologic, ecologic and clinical characteristics of Lyme borreliosis in Northwest Croatia. Acta Med. Croat. 1998, 1, 7–13. [Google Scholar]
- Eisen, L.; Lane, R.S. Vectors of Borelia burgdorferi sensu lato. In Lyme Borreliosis Biology, Epidemiology and Control; Gray, J., Kahl, O., Lane, R.S., Stanek, G., Eds.; Cabi Publishing: Wallingford, UK, 2002; p. 91. [Google Scholar]
- Krčmar, S. Hard ticks (Acari, Ixodidae) of Croatia. Zookeys 2012, 19–57. [Google Scholar] [CrossRef] [PubMed]
- Borčić, B.; Kaić, B.; Kralj, V. Some epidemiological data on TBE and Lyme borreliosis in Croatia. Zentralbl. Bakteriol. 1999, 289, 540–547. [Google Scholar] [CrossRef] [PubMed]
- Léger, E.; Vourc’h, G.; Vial, L.; Chevillon, C.; McCoy, K.D. Changing distributions of ticks: Causes and consequences. Exp. Appl. Acarol. 2013, 59, 219–244. [Google Scholar] [CrossRef] [PubMed]
- European Centre for Disease Prevention and Control; European Food Safety Authority. Field Sampling Methods for Mosquitoes, Sandflies, Biting Midges and Ticks—VectorNet Project 2014–2018; ECDC: Stockholm, Sweden; EFSA: Parma, Italy, 2018. [Google Scholar]
- Daniel, M.; Kolár, J.; Zeman, P. GIS tools for tick and tick-borne disease occurrence. Parasitology 2004, 129, S329–S352. [Google Scholar] [CrossRef] [PubMed]
- Croatian Bureau of Statistics. First Digital Census of Population 2021. Available online: https://dzs.gov.hr/en (accessed on 15 May 2023).
- Newman, B.C.; Sutton, W.B.; Wang, Y.; Schweitzer, C.J.; Moncayo, A.C.; Miller, B.T. A standardized method for the construction of a tick drag/flag sampling approach and evaluation of sampling efficacy. Exp. Appl. Acarol. 2019, 79, 433–446. [Google Scholar] [CrossRef]
- Estrada-Peña, A.; Bouattour, A.; Camicas, J.L.; Walker, A.R. Ticks of Domestic Animals in the Mediterranean Region. A Guide to Identification of Species; University of Edinburgh: Edinburgh, UK, 2003; revised 2014; ISBN 8-4962141-84 2004. [Google Scholar]
- Cattell, R.B. The Scree Test for the Number of Factors. Multivar. Behav. Res. 1966, 1, 245–276. [Google Scholar] [CrossRef] [PubMed]
- Baldwin, H.; Landesman, W.J.; Borgmann-Winter, B.; Allen, D. A Geographic Information System Approach to Map Tick Exposure Risk at a Scale for Public Health Intervention. J. Med. Entomol. 2022, 59, 162–172. [Google Scholar] [CrossRef]
- Sarani, M.S.; Sedaghat, M.M.; Zakieh, T.; Abdoreza, S.M.; Abbas, Z. Distribution of Hard Ticks Using Geographical Information System (GIS) in Mountainous Areas of East Golestan Province, Iran. In Proceedings of the Global Conference on Entomology, Chiang Mai, Thailand, 5–9 March 2011. [Google Scholar]
- Eisen, L.; Eisen, R.J. Using geographic information systems and decision support systems for the prediction, prevention, and control of vector-borne diseases. Annu. Rev. Entomol. 2011, 56, 41–61. [Google Scholar] [CrossRef]
- Rosà, R.; Pugliese, A. Effects of tick population dynamics and host densities on the persistence of tick-borne infections. Math. Biosci. 2007, 208, 216–240. [Google Scholar] [CrossRef]
- Randolph, S.E.; Green, R.M.; Peacey, M.F.; Rogers, D.J. Seasonal synchrony: The key to tick-borne encephalitis foci identified by satellite data. Parasitology 2000, 121, 15–23. [Google Scholar] [CrossRef]
- Requena-García, F.; Cabrero-Sañudo, F.; Olmeda-García, S.; González, J.; Valcárcel, F. Influence of environmental temperature and humidity on questing ticks in central Spain. Exp. Appl. Acarol. 2017, 71, 277–290. [Google Scholar] [CrossRef] [PubMed]
- Estrada-Peña, A. Climate, niche, ticks, and models: What they are and how we should interpret them. Parasitol. Res. 2008, 103, S87–S95. [Google Scholar] [CrossRef] [PubMed]
- Ostfeld, R.S.; Keesing, F. Biodiversity and the dilution effect in disease ecology. Ecology 2000, 82, 609. [Google Scholar]
- Schmidt, K.A.; Ostfeld, R.S. Biodiversity and disease risk: The case of Lyme disease. Conserv. Biol. 2001, 14, 722. [Google Scholar]
- Giudice, K.L.; Ostfeld, R.S.; Schmidt, K.A.; Keesing, F. The ecology of infectious disease: Effects of host diversity and community composition on Lyme disease risk. Proc. Natl. Acad. Sci. USA 2003, 100, 567. [Google Scholar] [CrossRef]
- Wikel, S.K. Ticks and Tick-Borne Infections: Complex Ecology, Agents, and Host Interactions. Vet. Sci. 2018, 5, 60. [Google Scholar] [CrossRef]
- Pfäffle, M.; Littwin, N.; Muders, S.V.; Petney, T.N. The ecology of tick-borne diseases. Int. J. Parasitol. 2013, 43, 1059–1077. [Google Scholar] [CrossRef]
- Baneth, G. Tick-borne infections of animals and humans: A common ground. Int. J. Parasitol. 2014, 44, 591–596. [Google Scholar] [CrossRef]
- Dantas-Torres, F.; Chomel, B.B.; Otranto, D. Ticks and tick-borne diseases: A One Health perspective. Trends Parasitol. 2012, 28, 437–446. [Google Scholar] [CrossRef]
- Wikel, S.K. Ticks and tick-borne pathogens at the cutaneous interface: Host defences, tick countermeasures, and a suitable environment for pathogen establishment. Front. Microbiol. 2013, 4, 337. [Google Scholar] [CrossRef]
- Labuda, M.; Jones, L.D.; Williams, T.; Danielova, V.; Nuttal, P.A. Efficient transmission of tick-borne encephalitis virus between cofeeding ticks. J. Med. Entomol. 1993, 30, 295. [Google Scholar] [CrossRef]
- Randolph, S.E.; Gern, L.; Nuttal, P.A. Co-feeding ticks: Epidemiological significance for tick-borne pathogen transmission. Parasitol. Today 1996, 12, 472. [Google Scholar] [CrossRef]
- Zannou, O.M.; Ouedraogo, A.S.; Biguezoton, A.S.; Abatih, E.; Coral-Almeida, M.; Farougou, S.; Yao, K.P.; Lempereur, L.; Saegerman, C. Models for Studying the Distribution of Ticks and Tick-Borne Diseases in Animals: A Systematic Review and a Meta-Analysis with a Focus on Africa. Pathogens 2021, 10, 893. [Google Scholar] [CrossRef]
| Sampling Method | ||||||||
|---|---|---|---|---|---|---|---|---|
| Animal Host | Human Host | Flagging/Dragging Method | ||||||
| Domestic animals | Pets | Wild animals | Unknown | 90 | 1289 | |||
| horses | 211 | dogs | 470 | wild boars | 2 | 4 | ||
| pigs | 5 | rabbits | 3 | |||||
| donkeys | 11 | cats | 80 | hedgehogs | 5 | |||
| goats | 7 | roe deer | 25 | |||||
| deer | 16 | |||||||
| 234 | 550 | 51 | 4 | 90 | 1289 | |||
| Total: 2218 | ||||||||
| Species | Gender/Development Stage | No. |
|---|---|---|
| Dermacentor marginatus | M | 2 |
| F | 1 | |
| Haemaphysalis punctata | M | 16 |
| F | 8 | |
| N | 14 | |
| Haemaphysalis sp. | N | 1 |
| Haemaphysalis sulcata | F | 1 |
| Hyalomma m. marginatum | F | 3 |
| Ixodes frontalis | F | 4 |
| N | 26 | |
| Ixodes ricinus | M | 41 |
| F | 148 | |
| N | 800 | |
| L | 148 | |
| Ixodes sp. | M | 2 |
| F | 2 | |
| N | 23 | |
| Ixodes vespertilionis | F | 1 |
| Rhiphicephalus sanguineus | M | 6 |
| F | 3 | |
| N | 20 | |
| Rhiphicephalus sp. | N | 19 |
| M—male; F—female; N—nymph; L—larva | 1289 | |
| Variable | Eigenvectors Spreadsheet | Variable Contributions | ||||||
|---|---|---|---|---|---|---|---|---|
| PC1 | PC2 | PC3 | PC4 | PC1 | PC2 | PC3 | PC4 | |
| human | 0.37 | 0.28 | −0.01 | −0.00 | 0.14 | 0.08 | 0.00 | 0.00 |
| dog | 0.31 | −0.21 | −0.05 | −0.00 | 0.09 | 0.04 | 0.00 | 0.00 |
| cat | 0.42 | −0.06 | −0.01 | −0.00 | 0.16 | 0.00 | 0.00 | 0.00 |
| hedgehog | 0.38 | −0.15 | −0.05 | −0.00 | 0.14 | 0.02 | 0.00 | 0.00 |
| rabbit | 0.39 | −0.05 | −0.02 | −0.00 | 0.16 | 0.00 | 0.00 | 0.00 |
| roe deer | 0.39 | −0.09 | 0.08 | 0.00 | 0.15 | 0.01 | 0.01 | 0.00 |
| deer | −0.04 | −0.31 | 0.76 | 0.00 | 0.00 | 0.10 | 0.58 | 0.00 |
| goat | −0.05 | −0.06 | −0.43 | 0.71 | 0.00 | 0.00 | 0.18 | 0.50 |
| horse | 0.44 | 0.19 | 0.03 | 0.00 | 0.15 | 0.04 | 0.00 | 0.00 |
| pig | −0.05 | −0.06 | −0.43 | −0.71 | 0.00 | 0.00 | 0.18 | 0.50 |
| donkey | 0.00 | 0.84 | 0.20 | 0.00 | 0.00 | 0.70 | 0.04 | 0.00 |
| Tick Species | Gender/Development | PC1 | PC2 | PC3 | PC4 |
|---|---|---|---|---|---|
| Argas persicus | F | −0.37 | −0.11 | −0.01 | −0.00 |
| Haemaphysalis punctata | M | −0.70 | −1.07 | −8.82 | 14.70 |
| Haemaphysalis punctata | F | −0.40 | −0.22 | −1.48 | 2.45 |
| Haemaphysalis punctata | N | −0.37 | −0.11 | −0.01 | −0.00 |
| Haemaphysalis sp. | M | −0.35 | −0.08 | −0.01 | −0.00 |
| Haemaphysalis sp. | F | −0.35 | −0.07 | −0.01 | −0.00 |
| Hyalomma marginatum | M | 0.31 | 1.57 | −1.09 | −2.45 |
| Hyalomma marginatum | F | 0.02 | 3.13 | 0.71 | 0.00 |
| Hyalomma sp. | M | −0.29 | 0.01 | −0.01 | −0.00 |
| Hyalomma sp. | F | −0.27 | 0.01 | 0.00 | −0.00 |
| Ixodes ricinus | M | 0.98 | −0.49 | −0.05 | −0.00 |
| Ixodes ricinus | F | 3.26 | −0.18 | −0.09 | −0.00 |
| Ixodes ricinus | N | 0.26 | 0.93 | −0.06 | −0.00 |
| Ixodes sp. | M | −0.40 | −0.44 | 0.86 | 0.00 |
| Ixodes sp. | F | −0.33 | −1.16 | 2.65 | 0.00 |
| Ixodes sp. | N | −0.37 | −0.11 | −0.01 | −0.00 |
| Ixodes gibbosus | F | −0.37 | −0.11 | −0.01 | −0.00 |
| Ixodes hexagonus | F | −0.35 | −0.11 | −0.01 | −0.00 |
| Ixodes frontalis | F | −0.33 | −0.03 | −0.01 | −0.00 |
| Ixodes vespertilionis | N | −0.35 | −0.07 | −0.01 | −0.00 |
| Rhiphycephalus sanguineus | M | 0.11 | −0.62 | −0.18 | −0.00 |
| Rhiphycephalus sanguineus | F | 0.27 | −0.81 | −0.23 | −0.00 |
| Rhiphycephalus sanguineus | N | −0.35 | −0.14 | −0.02 | −0.00 |
| Rhiphycephalus sanguineus | L | −0.33 | −0.13 | 0.01 | −0.00 |
| Dermacentor marginatus | M | −0.59 | −0.66 | −4.41 | −7.35 |
| Dermacentor marginatus | F | −0.40 | −0.22 | −1.48 | −2.45 |
| Rhiphycephalus turanicus | M | −0.36 | −0.12 | −0.01 | −0.00 |
| Rhiphycephalus turanicus | F | −0.37 | −0.12 | −0.02 | −0.00 |
| Rhiphycephalus turanicus | N | −0.37 | −0.11 | −0.01 | −0.00 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cvek, M.; Broznić, D.; Puškadija, D.; Blagonić, B.; Kirin, I.; Pustijanac, E.; Landeka, N.; Stojanović, A.; Ožanič Bučar, M.; Tomić Linšak, D. Investigation and Spatial Distribution of Hard Ticks by Geographical Information System (GIS) in the Region of Istria, Croatia. Appl. Sci. 2023, 13, 9483. https://doi.org/10.3390/app13169483
Cvek M, Broznić D, Puškadija D, Blagonić B, Kirin I, Pustijanac E, Landeka N, Stojanović A, Ožanič Bučar M, Tomić Linšak D. Investigation and Spatial Distribution of Hard Ticks by Geographical Information System (GIS) in the Region of Istria, Croatia. Applied Sciences. 2023; 13(16):9483. https://doi.org/10.3390/app13169483
Chicago/Turabian StyleCvek, Maja, Dalibor Broznić, Darja Puškadija, Boris Blagonić, Ivana Kirin, Emina Pustijanac, Nediljko Landeka, Aleksandar Stojanović, Mateja Ožanič Bučar, and Dijana Tomić Linšak. 2023. "Investigation and Spatial Distribution of Hard Ticks by Geographical Information System (GIS) in the Region of Istria, Croatia" Applied Sciences 13, no. 16: 9483. https://doi.org/10.3390/app13169483
APA StyleCvek, M., Broznić, D., Puškadija, D., Blagonić, B., Kirin, I., Pustijanac, E., Landeka, N., Stojanović, A., Ožanič Bučar, M., & Tomić Linšak, D. (2023). Investigation and Spatial Distribution of Hard Ticks by Geographical Information System (GIS) in the Region of Istria, Croatia. Applied Sciences, 13(16), 9483. https://doi.org/10.3390/app13169483







