Correlation of Bone Textural Parameters with Age in the Context of Orthopedic X-ray Studies
Abstract
1. Introduction
2. Materials and Methods
2.1. Dataset
2.2. Data Acquisition
2.3. ROI Annotation
2.4. Textural Analysis
2.5. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Downey, P.A.; Siegel, M.I. Bone Biology and the Clinical Implications for Osteoporosis. Phys. Ther. 2006, 86, 77–91. [Google Scholar] [CrossRef] [PubMed]
- Holguin, N.; Brodt, M.D.; Sanchez, M.E.; Silva, M.J. Aging Diminishes Lamellar and Woven Bone Formation Induced by Tibial Compression in Adult C57BL/6. Bone 2014, 65, 83–91. [Google Scholar] [CrossRef]
- Hadjidakis, D.J.; Androulakis, I.I. Bone Remodeling. Ann. N. Y. Acad. Sci. 2006, 1092, 385–396. [Google Scholar] [CrossRef] [PubMed]
- Gartner, L.P.; Hiatt, J.L. Cartilage and Bone. In Concise Histology; Elsevier: Amsterdam, The Netherlands, 2011; pp. 74–93. ISBN 978-0-7020-3114-4. [Google Scholar]
- Rho, J.-Y.; Kuhn-Spearing, L.; Zioupos, P. Mechanical Properties and the Hierarchical Structure of Bone. Med. Eng. Phys. 1998, 20, 92–102. [Google Scholar] [CrossRef] [PubMed]
- McNally, E.A.; Schwarcz, H.P.; Botton, G.A.; Arsenault, A.L. A Model for the Ultrastructure of Bone Based on Electron Microscopy of Ion-Milled Sections. PLoS ONE 2012, 7, e29258. [Google Scholar] [CrossRef]
- Huda, W.; Abrahams, R.B. X-ray-Based Medical Imaging and Resolution. Am. J. Roentgenol. 2015, 204, W393–W397. [Google Scholar] [CrossRef]
- Bonewald, L.F. The Amazing Osteocyte. J. Bone Miner. Res. 2011, 26, 229–238. [Google Scholar] [CrossRef]
- Parfitt, A.M. Osteonal and Hemi-Osteonal Remodeling: The Spatial and Temporal Framework for Signal Traffic in Adult Human Bone. J. Cell. Biochem. 1994, 55, 273–286. [Google Scholar] [CrossRef]
- Di Girolamo, D.J.; Clemens, T.L.; Kousteni, S. The Skeleton as an Endocrine Organ. Nat. Rev. Rheumatol. 2012, 8, 674–683. [Google Scholar] [CrossRef]
- Noirrit-Esclassan, E.; Valera, M.-C.; Tremollieres, F.; Arnal, J.-F.; Lenfant, F.; Fontaine, C.; Vinel, A. Critical Role of Estrogens on Bone Homeostasis in Both Male and Female: From Physiology to Medical Implications. Int. J. Mol. Sci. 2021, 22, 1568. [Google Scholar] [CrossRef]
- Boskey, A.L.; Coleman, R. Aging and Bone. J. Dent. Res. 2010, 89, 1333–1348. [Google Scholar] [CrossRef] [PubMed]
- Florencio-Silva, R.; da Silva Sasso, G.R.; Sasso-Cerri, E.; Simões, M.J.; Cerri, P.S. Biology of Bone Tissue: Structure, Function, and Factors That Influence Bone Cells. BioMed Res. Int. 2015, 2015, 421746. [Google Scholar] [CrossRef] [PubMed]
- Kerr, D.; Morton, A.; Dick, I.; Prince, R. Exercise Effects on Bone Mass in Postmenopausal Women Are Site-Specific and Load-Dependent. J. Bone Miner. Res. 2009, 11, 218–225. [Google Scholar] [CrossRef]
- Bonjour, J.-P.; Theintz, G.; Law, F.; Slosman, D.; Rizzoli, R. Peak Bone Mass. Osteoporos. Int. 1994, 4, S7–S13. [Google Scholar] [CrossRef]
- Lips, P. Vitamin D Deficiency and Secondary Hyperparathyroidism in the Elderly: Consequences for Bone Loss and Fractures and Therapeutic Implications. Endocr. Rev. 2001, 22, 477–501. [Google Scholar] [CrossRef] [PubMed]
- Khosla, S.; Oursler, M.J.; Monroe, D.G. Estrogen and the Skeleton. Trends Endocrinol. Metab. 2012, 23, 576–581. [Google Scholar] [CrossRef]
- Szczypiński, P.M.; Strzelecki, M.; Materka, A.; Klepaczko, A. MaZda—The software package for textural analysis of biomedical images. In Computers in Medical Activity; Advances in Intelligent and Soft Computing; Springer: Berlin/Heidelberg, Germany, 2009; Volume 65, pp. 73–84. [Google Scholar]
- Pociask, E.; Nurzynska, K.; Obuchowicz, R.; Bałon, P.; Uryga, D.; Strzelecki, M.; Izworski, A.; Piórkowski, A. Differential Diagnosis of Cysts and Granulomas Supported by Texture Analysis of Intraoral Radiographs. Sensors 2021, 21, 7481. [Google Scholar] [CrossRef]
- Shapiro, S.S.; Wilk, M.B. An Analysis of Variance Test for Normality (Complete Samples). Biometrika 1965, 52, 591–611. [Google Scholar] [CrossRef]
- Haralick, R.M.; Shanmugam, K.; Dinstein, I. Textural Features for Image Classification. IEEE Trans. Syst. Man Cybern. 1973, SMC-3, 610–621. [Google Scholar] [CrossRef]
- Zhang, H.; Li, Q.; Liu, J.; Shang, J.; Du, X.; McNairn, H.; Champagne, C.; Dong, T.; Liu, M. Image Classification Using RapidEye Data: Integration of Spectral and Textual Features in a Random Forest Classifier. IEEE J. Sel. Top. Appl. Earth Obs. Remote Sens. 2017, 10, 5334–5349. [Google Scholar] [CrossRef]
- Yang, X.; Tridandapani, S.; Beitler, J.J.; Yu, D.S.; Yoshida, E.J.; Curran, W.J.; Liu, T. Ultrasound GLCM Texture Analysis of Radiation-Induced Parotid-Gland Injury in Head-and-Neck Cancer Radiotherapy: An in Vivo Study of Late Toxicity: Ultrasound Assessment of Post-RT Parotid Gland. Med. Phys. 2012, 39, 5732–5739. [Google Scholar] [CrossRef] [PubMed]
- Julesz, B. Experiments in the Visual Perception of Texture. Sci. Am. 1975, 232, 34–43. [Google Scholar] [CrossRef]
- Priolo, F.; Cerase, A. The Current Role of Radiography in the Assessment of Skeletal Tumors and Tumor-like Lesions. Eur. J. Radiol. 1998, 27, S77–S85. [Google Scholar] [CrossRef]
- Fritscher, K.; Grunerbl, A.; Hanni, M.; Suhm, N.; Hengg, C.; Schubert, R. Trabecular Bone Analysis in CT and X-ray Images of the Proximal Femur for the Assessment of Local Bone Quality. IEEE Trans. Med. Imaging 2009, 28, 1560–1575. [Google Scholar] [CrossRef] [PubMed]
- Akkus, O.; Polyakova-Akkus, A.; Adar, F.; Schaffler, M.B. Aging of Microstructural Compartments in Human Compact Bone. J. Bone Miner. Res. 2003, 18, 1012–1019. [Google Scholar] [CrossRef]
- Ammann, P.; Rizzoli, R. Bone Strength and Its Determinants. Osteoporos. Int. 2003, 14, 13–18. [Google Scholar] [CrossRef] [PubMed]
- Bailey, A.J.; Sims, T.J.; Ebbesen, E.N.; Mansell, J.P.; Thomsen, J.S.; Mosekilde, L. Age-Related Changes in the Biochemical Properties of Human Cancellous Bone Collagen: Relationship to Bone Strength. Calcif. Tissue Int. 1999, 65, 203–210. [Google Scholar] [CrossRef] [PubMed]
- Karlamangla, A.S.; Burnett-Bowie, S.-A.M.; Crandall, C.J. Bone Health During the Menopause Transition and Beyond. Obstet. Gynecol. Clin. N. Am. 2018, 45, 695–708. [Google Scholar] [CrossRef] [PubMed]
- Cao, J.J.; Wronski, T.J.; Iwaniec, U.; Phleger, L.; Kurimoto, P.; Boudignon, B.; Halloran, B.P. Aging Increases Stromal/Osteoblastic Cell-Induced Osteoclastogenesis and Alters the Osteoclast Precursor Pool in the Mouse. J. Bone Miner. Res. 2005, 20, 1659–1668. [Google Scholar] [CrossRef]
- Chan, G.K.; Duque, G. Age-Related Bone Loss: Old Bone, New Facts. Gerontology 2002, 48, 62–71. [Google Scholar] [CrossRef]
- Hart, N.H.; Nimphius, S.; Rantalainen, T.; Ireland, A.; Siafarikas, A.; Newton, R.U. Mechanical Basis of Bone Strength: Influence of Bone Material, Bone Structure and Muscle Action. J. Musculoskelet. Neuronal Interact. 2017, 17, 114–139. [Google Scholar] [PubMed]
- Kersh, M.E.; Pandy, M.G.; Bui, Q.M.; Jones, A.C.; Arns, C.H.; Knackstedt, M.A.; Seeman, E.; Zebaze, R.M. The Heterogeneity in Femoral Neck Structure and Strength. J. Bone Miner. Res. Off. J. Am. Soc. Bone Miner. Res. 2013, 28, 1022–1028. [Google Scholar] [CrossRef] [PubMed]
- Riis, B.J.; Hansen, M.A.; Jensen, A.M.; Overgaard, K.; Christiansen, C. Low Bone Mass and Fast Rate of Bone Loss at Menopause: Equal Risk Factors for Future Fracture: A 15-Year Follow-up Study. Bone 1996, 19, 9–12. [Google Scholar] [CrossRef] [PubMed]
- Soldati, E.; Roseren, F.; Guenoun, D.; Mancini, L.; Catelli, E.; Prati, S.; Sciutto, G.; Vicente, J.; Iotti, S.; Bendahan, D.; et al. Multiscale Femoral Neck Imaging and Multimodal Trabeculae Quality Characterization in an Osteoporotic Bone Sample. Materials 2022, 15, 8048. [Google Scholar] [CrossRef] [PubMed]
- Carpenter, R.D.; Sigurdsson, S.; Zhao, S.; Lu, Y.; Eiriksdottir, G.; Sigurdsson, G.; Jonsson, B.Y.; Prevrhal, S.; Harris, T.B.; Siggeirsdottir, K.; et al. Effects of Age and Sex on the Strength and Cortical Thickness of the Femoral Neck. Bone 2011, 48, 741–747. [Google Scholar] [CrossRef][Green Version]
- Pajamäki, I.; Sievänen, H.; Kannus, P.; Jokihaara, J.; Vuohelainen, T.; Järvinen, T.L.N. Skeletal Effects of Estrogen and Mechanical Loading Are Structurally Distinct. Bone 2008, 43, 748–757. [Google Scholar] [CrossRef]
- Jeong, H.; Kim, J.; Ishida, T.; Akiyama, M.; Kim, Y. Computerised Analysis of Osteoporotic Bone Patterns Using Texture Parameters Characterising Bone Architecture. Br. J. Radiol. 2013, 86, 20101115. [Google Scholar] [CrossRef]
- Kjaer, L.; Ring, P.; Thomsen, C.; Henriksen, O. Texture Analysis in Quantitative MR Imaging. Tissue Characterisation of Normal Brain and Intracranial Tumours at 1.5 T. Acta Radiol. 1995, 36, 127–135. [Google Scholar] [CrossRef]
- Kovalev, V.; Kruggel, F. Texture Anisotropy of the Brain’s White Matter as Revealed by Anatomical MRI. IEEE Trans. Med. Imaging 2007, 26, 678–685. [Google Scholar] [CrossRef]
- Mahmoud-Ghoneim, D.; Alkaabi, M.K.; de Certaines, J.D.; Goettsche, F.-M. The Impact of Image Dynamic Range on Texture Classification of Brain White Matter. BMC Med. Imaging 2008, 8, 18. [Google Scholar] [CrossRef]
- Holli, K.; Lääperi, A.-L.; Harrison, L.; Luukkaala, T.; Toivonen, T.; Ryymin, P.; Dastidar, P.; Soimakallio, S.; Eskola, H. Characterization of Breast Cancer Types by Texture Analysis of Magnetic Resonance Images. Acad. Radiol. 2010, 17, 135–141. [Google Scholar] [CrossRef] [PubMed]
- Herlidou, S.; Rolland, Y.; Bansard, J.Y.; Le Rumeur, E.; de Certaines, J.D. Comparison of Automated and Visual Texture Analysis in MRI: Characterization of Normal and Diseased Skeletal Muscle. Magn. Reson. Imaging 1999, 17, 1393–1397. [Google Scholar] [CrossRef] [PubMed]
- Hirvasniemi, J.; Gielis, W.P.; Arbabi, S.; Agricola, R.; van Spil, W.E.; Arbabi, V.; Weinans, H. Bone Texture Analysis for Prediction of Incident Radiographic Hip Osteoarthritis Using Machine Learning: Data from the Cohort Hip and Cohort Knee (CHECK) Study. Osteoarthr. Cartil. 2019, 27, 906–914. [Google Scholar] [CrossRef] [PubMed]
- Mazur, P. The Influence of Bit-Depth Reduction on Correlation of Texture Features with a Patient’s Age. In Progress in Image Processing, Pattern Recognition and Communication Systems; Choraś, M., Choraś, R.S., Kurzyński, M., Trajdos, P., Pejaś, J., Hyla, T., Eds.; Lecture Notes in Networks and Systems; Springer International Publishing: Cham, Switzerland, 2022; Volume 255, pp. 191–198. ISBN 978-3-030-81522-6. [Google Scholar]
- Mazur, P.; Obuchowicz, R.; Piórkowski, A. The Influence of Age on Morphometric and Textural Vertebrae Features in Lateral Cervical Spine Radiographs. In Information Technology in Biomedicine; Pietka, E., Badura, P., Kawa, J., Wieclawek, W., Eds.; Advances in Intelligent Systems and Computing; Springer International Publishing: Cham, Switzerland, 2021; Volume 1186, pp. 71–80. ISBN 978-3-030-49665-4. [Google Scholar]
- Maciel, J.G.; de Araújo, I.M.; Trazzi, L.C.; de Azevedo-Marques, P.M.; Salmon, C.E.G.; Paula, F.J.A.d.; Nogueira-Barbosa, M.H. Association of Bone Mineral Density with Bone Texture Attributes Extracted Using Routine Magnetic Resonance Imaging. Clinics 2020, 75, e1766. [Google Scholar] [CrossRef]
- Shirvaikar, M.; Huang, N.; Dong, X.N. The Measurement of Bone Quality Using Gray Level Co-Occurrence Matrix Textural Features. J. Med. Imaging Health Inform. 2016, 6, 1357–1362. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Paalvast, O.; Nauta, M.; Koelle, M.; Geerdink, J.; Vijlbrief, O.; Hegeman, J.H.; Seifert, C. Radiology Report Generation for Proximal Femur Fractures Using Deep Classification and Language Generation Models. Artif. Intell. Med. 2022, 128, 102281. [Google Scholar] [CrossRef]
- Meena, T.; Roy, S. Bone Fracture Detection Using Deep Supervised Learning from Radiological Images: A Paradigm Shift. Diagnostics 2022, 12, 2420. [Google Scholar] [CrossRef]
- Urakawa, T.; Tanaka, Y.; Goto, S.; Matsuzawa, H.; Watanabe, K.; Endo, N. Detecting Intertrochanteric Hip Fractures with Orthopedist-Level Accuracy Using a Deep Convolutional Neural Network. Skelet. Radiol. 2019, 48, 239–244. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Xu, Z.; Tong, Y.; Xia, L.; Jie, B.; Ding, P.; Bai, H.; Zhang, Y.; He, Y. Detection and Classification of Mandibular Fracture on CT Scan Using Deep Convolutional Neural Network. Clin. Oral Investig. 2022, 26, 4593–4601. [Google Scholar] [CrossRef] [PubMed]
- Secgin, Y.; Oner, Z.; Turan, M.; Oner, S. Gender Prediction with the Parameters Obtained from Pelvis Computed Tomography Images and Machine Learning Algorithms. J. Anat. Soc. India 2022, 71, 204. [Google Scholar] [CrossRef]
- Zhou, J.; Li, Z.; Zhi, W.; Liang, B.; Moses, D.; Dawes, L. Using Convolutional Neural Networks and Transfer Learning for Bone Age Classification. In Proceedings of the 2017 International Conference on Digital Image Computing: Techniques and Applications (DICTA), Sydney, Australia, 29 November–1 December 2017; pp. 1–6. [Google Scholar]
- Cernazanu-Glavan, C.; Holban, S. Segmentation of Bone Structure in X-ray Images Using Convolutional Neural Network. Adv. Electr. Comput. Eng. 2013, 13, 87–94. [Google Scholar] [CrossRef]
- Liu, X.; Han, C.; Wang, H.; Wu, J.; Cui, Y.; Zhang, X.; Wang, X. Fully Automated Pelvic Bone Segmentation in Multiparameteric MRI Using a 3D Convolutional Neural Network. Insights Imaging 2021, 12, 93. [Google Scholar] [CrossRef]
- Eweje, F.R.; Bao, B.; Wu, J.; Dalal, D.; Liao, W.; He, Y.; Luo, Y.; Lu, S.; Zhang, P.; Peng, X.; et al. Deep Learning for Classification of Bone Lesions on Routine MRI. eBioMedicine 2021, 68, 103402. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Pan, I.; Bao, B.; Halsey, K.; Chang, M.; Liu, H.; Peng, S.; Sebro, R.A.; Guan, J.; Yi, T.; et al. Deep Learning-Based Classification of Primary Bone Tumors on Radiographs: A Preliminary Study. eBioMedicine 2020, 62, 103121. [Google Scholar] [CrossRef]
- Kozakiewicz, M. Measures of Corticalization. J. Clin. Med. 2022, 11, 5463. [Google Scholar] [CrossRef]
- Obuchowicz, R.; Nurzynska, K.; Pierzchala, M.; Piorkowski, A.; Strzelecki, M. Texture Analysis for the Bone Age Assessment from MRI Images of Adolescent Wrists in Boys. J. Clin. Med. 2023, 12, 2762. [Google Scholar] [CrossRef]
- Dieckmeyer, M.; Sollmann, N.; El Husseini, M.; Sekuboyina, A.; Löffler, M.T.; Zimmer, C.; Kirschke, J.S.; Subburaj, K.; Baum, T. Gender-, Age- and Region-Specific Characterization of Vertebral Bone Microstructure Through Automated Segmenta-tion and 3D Texture Analysis of Routine Abdominal CT. Front. Endocrinol. 2022, 12, 792760. [Google Scholar] [CrossRef]
- Lespessailles, E.; Gadois, C.; Kousignian, I.; Neveu, J.P.; Fardellone, P.; Kolta, S.; Roux, C.; Do-Huu, J.P.; Benhamou, C.L. Clinical interest of bone texture analysis in osteoporosis: A case control multicenter study. Osteoporos. Int. 2008, 19, 1019–1028. [Google Scholar] [CrossRef]
- Zheng, K.; Makrogiannis, S. Bone texture characterization for osteoporosis diagnosis using digital radiography. In Proceedings of the 38th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Orlando, FL, USA, 16–20 August 2016; pp. 1034–1037. [Google Scholar] [CrossRef]
- Khojastepour, L.; Hasani, M.; Ghasemi, M.; Mehdizadeh, A.R.; Tajeripour, F. Mandibular Trabecular Bone Analysis Using Local Binary Pattern for Osteoporosis Diagnosis. J. Biomed. Phys. Eng. 2019, 9, 81–88. [Google Scholar] [CrossRef] [PubMed]
- Kawashima, Y.; Fujita, A.; Buch, K.; Li, B.; Qureshi, M.M.; Chapman, M.N.; Sakai, O. Using texture analysis of head CT images to differentiate osteoporosis from normal bone density. Eur. J. Radiol. 2019, 116, 212–218. [Google Scholar] [CrossRef]
- Khider, M.; Taleb-Ahmed, A.; Dubois, P.; Haddad, B. Classification of trabecular bone texture from MRI and CT scan images by multi resolution analysis. In Proceedings of the 29th Annual International Conference of the IEEE Engineering in Medicine and Biology Society, Lyon, France, 22–26 August 2007; pp. 5589–5592. [Google Scholar] [CrossRef]
- Castellanos, N.P.; Martínez, E.; Gutierrez, J. Improving osteoporosis diagnosis in children using image texture analysis. In Proceedings of the 2011 Annual International Conference of the IEEE Engineering in Medicine and Biology Society, Boston, MA, USA, 30 August–3 September 2011. [Google Scholar] [CrossRef]
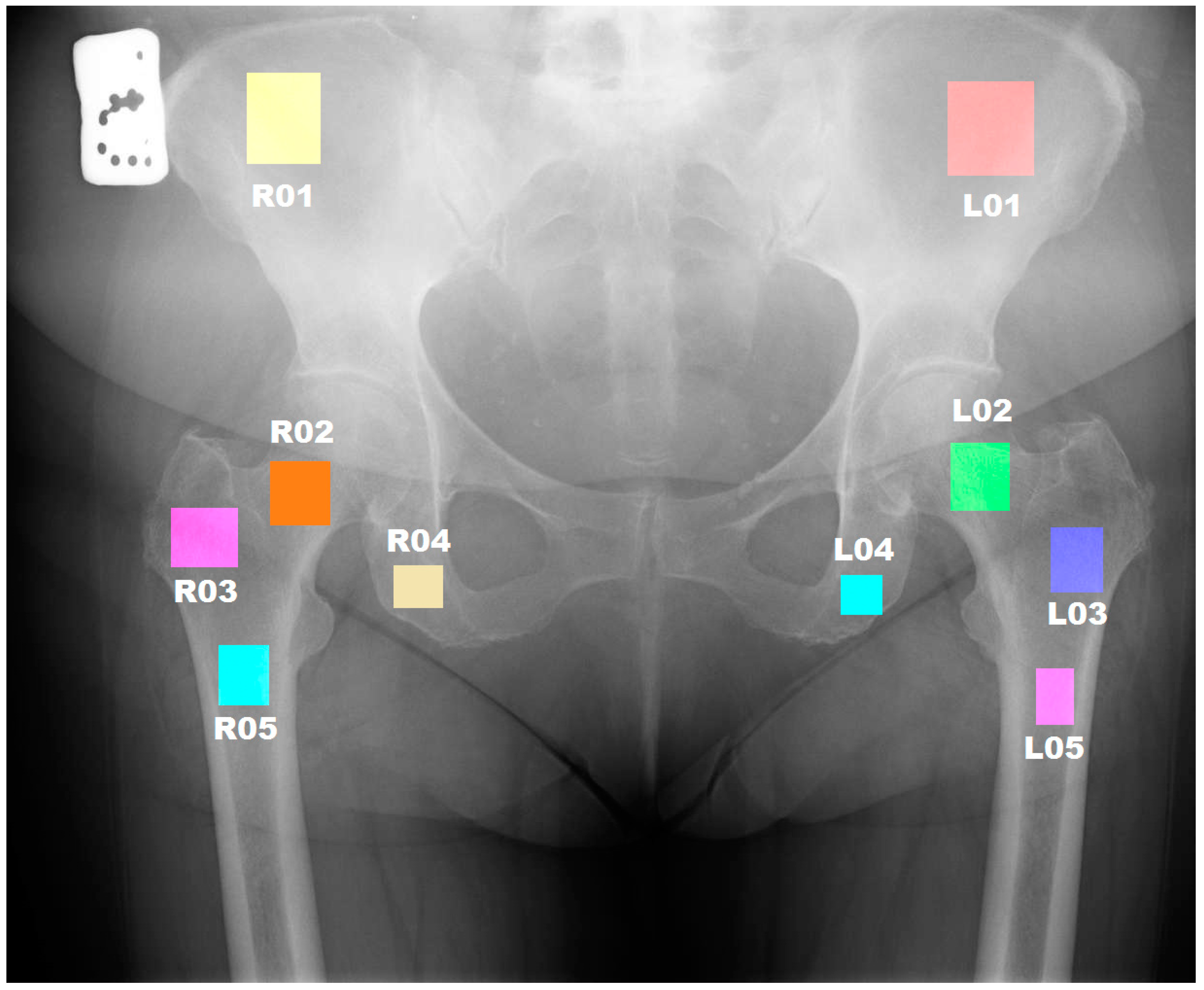



| ROI | Anatomical Structure |
|---|---|
| L 01 | Wing of ilium—left side |
| L 02 | Neck of femur—left side |
| L 03 | Greater trochanter—left side |
| L 04 | Ischium—left side |
| L 05 | Shaft of femur—upper left side |
| R 01 | Wing of ilium—right side |
| R 02 | Neck of femur—right side |
| R 03 | Greater trochanter—right side |
| R 04 | Ischium—right side |
| R 05 | Shaft of femur—upper right side |
| Model | Variable | Estimate (β Coefficients) | SE | p-Value | Adjusted R2 | AIC |
|---|---|---|---|---|---|---|
| MODEL_L02_YM6HogO8b4 fixed_regression | AGE | −0.009 | 0.001 | 2.4 × 10−14 | 0.271 | −161.1173 |
| MODEL_L02_YM4GlcmH4DifEntrp Fixed_regression | AGE | −0.002 | 0.0003 | 1.55 × 10−13 | 0.2565 | −602.232 |
| Model | Variable | Estimate (β Coefficients) | SE | T | p-Value | Lower_2.5 | Upper_97.5 | AIC |
|---|---|---|---|---|---|---|---|---|
| MODEL_L02_YM6HogO8b4 mixed_regression | AGE | −0.008 | 0.001 | −5.444 | 0 | −0.011 | −0.005 | −159.9161 |
| MODEL_L02_YM4GlcmH4DifEntrp mixed_regression | AGE | −0.002 | 0.0004 | −4.998 | 0 | −0.003 | −0.001 | −588.468 |
| ROI | Parameter | rho Spearman | p-Value |
|---|---|---|---|
| L 01 | YD4DwtHaarS4HH | 0.35 | 1.38 × 10−9 |
| L 02 | YM6HogO8b4 | −0.52 | 4.95 × 10−14 |
| L 03 | YS4Gab24H12Mag | −0.27 | 0.24 × 10−3 |
| L 04 | YM5Gab8Z4Mag | −0.14 | 0.4 × 10−2 |
| L 05 | YS5GrlmHMGLevNonUn | 0.32 | 1.60 × 10−5 |
| R 01 | YS6GlcmZ5SumAverg | 0.26 | 1.60 × 10−4 |
| R 02 | YLbpCs8n5 | 0.31 | 7.54 × 10−5 |
| R 03 | YN8DwtHaarS1HH | 0.37 | 4.62 × 10−6 |
| R 04 | YD4GrlmHRLNonUni | 0.17 | 0.53 × 10−4 |
| R 05 | YN4Gab24Z12Mag | −0.27 | 0.90 × 10−3 |
| Parameter | rho Spearman | p-Value |
|---|---|---|
| YM6HogO8b4 | −0.5194 | 4.95 × 10−14 |
| YM5HogO8b4 | −0.5143 | 9.55 × 10−14 |
| YM8HogO8b4 | −0.5139 | 1.00 × 10−13 |
| YM7HogO8b4 | −0.5130 | 1.12 × 10−13 |
| YM4GlcmH4DifEntrp | −0.5100 | 1.65 × 10−13 |
| YM4HogO8b4 | −0.5100 | 1.70 × 10−13 |
| YM4GlcmH4InvDfMom | 0.5088 | 1.93 × 10−13 |
| YM4GlcmH4Contrast | −0.5085 | 1.99 × 10−13 |
| YM4HogO8b5 | −0.5078 | 2.19 × 10−13 |
| YM5GlcmH4Contrast | −0.5071 | 2.38 × 10−13 |
| YM7GlcmH4Contrast | −0.5070 | 2.42 × 10−13 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kamiński, P.; Obuchowicz, R.; Stępień, A.; Lasek, J.; Pociask, E.; Piórkowski, A. Correlation of Bone Textural Parameters with Age in the Context of Orthopedic X-ray Studies. Appl. Sci. 2023, 13, 6618. https://doi.org/10.3390/app13116618
Kamiński P, Obuchowicz R, Stępień A, Lasek J, Pociask E, Piórkowski A. Correlation of Bone Textural Parameters with Age in the Context of Orthopedic X-ray Studies. Applied Sciences. 2023; 13(11):6618. https://doi.org/10.3390/app13116618
Chicago/Turabian StyleKamiński, Paweł, Rafał Obuchowicz, Aleksandra Stępień, Julia Lasek, Elżbieta Pociask, and Adam Piórkowski. 2023. "Correlation of Bone Textural Parameters with Age in the Context of Orthopedic X-ray Studies" Applied Sciences 13, no. 11: 6618. https://doi.org/10.3390/app13116618
APA StyleKamiński, P., Obuchowicz, R., Stępień, A., Lasek, J., Pociask, E., & Piórkowski, A. (2023). Correlation of Bone Textural Parameters with Age in the Context of Orthopedic X-ray Studies. Applied Sciences, 13(11), 6618. https://doi.org/10.3390/app13116618








