Characterization of Flexor Digitorum Superficialis Muscle Stiffness Using Ultrasound Shear Wave Elastography and MyotonPRO: A Cross-Sectional Study Investigating the Correlation between Different Approaches
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Sample Size
2.3. Participants and Setting
2.4. Measurements
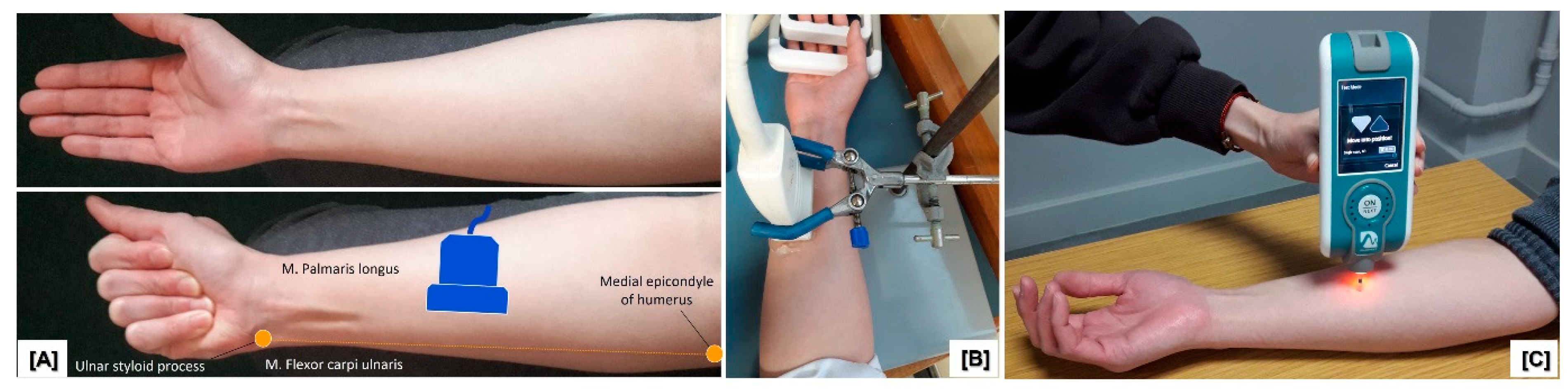
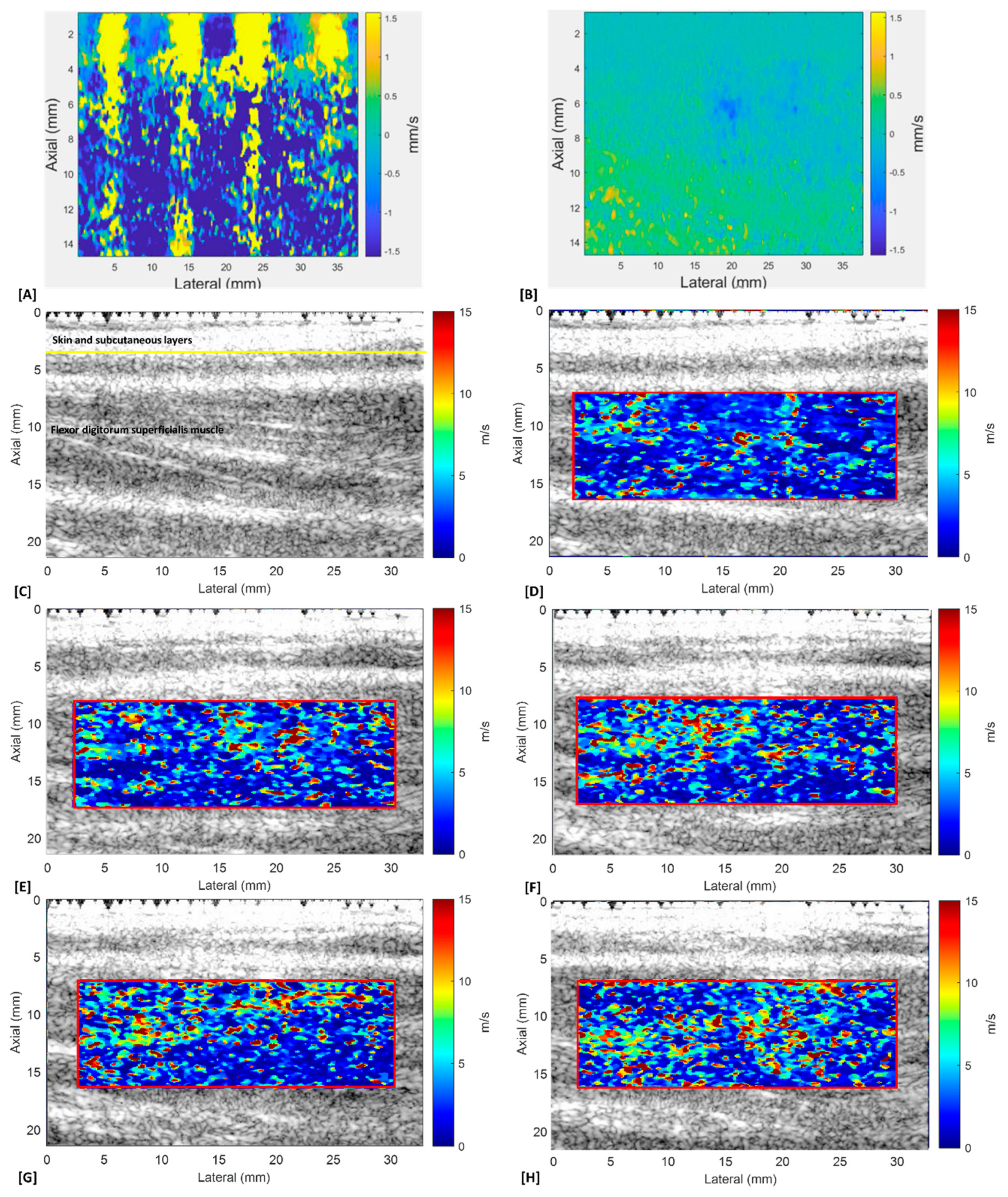
2.4.1. Ultrasound Shear Wave Elastography (SWE) Measurement
2.4.2. MyotonPRO Measurement
2.5. Procedures
2.6. Data Analysis
2.6.1. Young’s Modulus Estimation
2.6.2. Dynamic Stiffness Calculation
2.7. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bilston, L.E.; Tan, K. Measurement of passive skeletal muscle mechanical properties in vivo: Recent progress, clinical applications, and remaining challenges. Ann. Biomed. Eng. 2015, 43, 261–273. [Google Scholar] [CrossRef] [PubMed]
- Needle, A.R.; Baumeister, J.; Kaminski, T.W.; Higginson, J.S.; Farquhar, W.B.; Swanik, C.B. Neuromechanical coupling in the regulation of muscle tone and joint stiffness. Scand. J. Med. Sci. Sports 2014, 24, 737–748. [Google Scholar] [CrossRef] [PubMed]
- Nordin, M.; Frankel, V.H. (Eds.) Basic Biomechanics of the Musculoskeletal System; Lippincott Williams & Wilkins: New York, NY, USA, 2001. [Google Scholar]
- Oatis, C.A. Kinesiology: The Mechanics and Pathomechanics of Human Movement; Lippincott Williams & Wilkins: Baltimore, MD, USA, 2009. [Google Scholar]
- Ferreira-Sánchez, M.D.R.; Moreno-Verdú, M.; Cano-de-La-Cuerda, R. Quantitative measurement of rigidity in Parkinson’s disease: A systematic review. Sensors 2020, 20, 880. [Google Scholar] [CrossRef] [PubMed]
- Faturi, F.M.; Santos, G.L.; Ocamoto, G.N.; Russo, T.L. Structural muscular adaptations in upper limb after stroke: A systematic review. Top. Stroke Rehabil. 2019, 26, 73–79. [Google Scholar] [CrossRef] [PubMed]
- Kenis-Coskun, O.; Giray, E.; Gencer-Atalay, Z.K.; Yagci, I.; Karadag-Saygi, E. Reliability of quantitative ultrasound measurement of flexor digitorum superficialis and profundus muscles in stroke. J. Comp. Eff. Res. 2020, 9, 1293–1300. [Google Scholar] [CrossRef] [PubMed]
- Simon, A.M.; Turner, K.L.; Miller, L.A.; Dumanian, G.A.; Potter, B.K.; Beachler, M.D.; Hargrove, L.J.; Kuiken, T.A. Myoelectric prosthesis hand grasp control following targeted muscle reinnervation in individuals with transradial amputation. PLoS ONE 2023, 18, e0280210. [Google Scholar] [CrossRef]
- Vu, P.P.; Vaskov, A.K.; Irwin, Z.T.; Henning, P.T.; Lueders, D.R.; Laidlaw, A.T.; Davis, A.J.; Nu, C.S.; Gates, D.H.; Cederna, P.S. A regenerative peripheral nerve interface allows real-time control of an artificial hand in upper limb amputees. Sci. Transl. Med. 2020, 12, eaay2857. [Google Scholar] [CrossRef]
- Chen, P.-Y.; Yang, T.-H.; Kuo, L.-C.; Shih, C.-C.; Huang, C.-C. Characterization of Hand Tendons through High-Frequency Ultrasound Elastography. IEEE Trans. Ultrason. Ferroelectr. Freq. Control 2019, 67, 37–48. [Google Scholar] [CrossRef]
- Maslow, J.I.; Posey, S.L.; Habet, N.; Duemmler, M.; Odum, S.; Gaston, R.G. Central Slip Reconstruction with a Distally Based Flexor Digitorum Superficialis Slip: A Biomechanical Study. J. Hand Surg. 2022, 47, 145–150. [Google Scholar] [CrossRef]
- Kocur, P.; Piwińska, I.; Goliwąs, M.; Adamczewska, K. Assessment of myofascial stiffness of flexor digitorum superficialis muscles in rock climbers. Acta Bioeng. Biomech. 2021, 23, 23–31. [Google Scholar] [CrossRef]
- Colomar, J.; Baiget, E.; Corbi, F. Influence of strength, power, and muscular stiffness on stroke velocity in junior tennis players. Front. Physiol. 2020, 11, 196. [Google Scholar] [CrossRef] [PubMed]
- Vinstrup, J.; Calatayud, J.; Jakobsen, M.D.; Sundstrup, E.; Jørgensen, J.R.; Casaña, J.; Andersen, L.L. Hand strengthening exercises in chronic stroke patients: Dose-response evaluation using electromyography. J. Hand Ther. 2018, 31, 111–121. [Google Scholar] [CrossRef] [PubMed]
- Waitayawinyu, T.; Numnate, W.; Boonyasirikool, C.; Niempoog, S. Outcomes of Endoscopic Carpal Tunnel Release with Ring Finger Flexor Digitorum Superficialis Opponensplasty in Severe Carpal Tunnel Syndrome. J. Hand Surg. 2019, 44, 1095.e1–1095.e7. [Google Scholar] [CrossRef] [PubMed]
- Boser, Q.A.; Dawson, M.R.; Schofield, J.S.; Dziwenko, G.Y.; Hebert, J.S. Defining the design requirements for an assistive powered hand exoskeleton: A pilot explorative interview study and case series. Prosthet. Orthot. Int. 2020, 45, 161–169. [Google Scholar] [CrossRef] [PubMed]
- Díez, J.A.; BlancoJosé, A.; Catalán, J.M.; Badesa, F.J.; Lledó, L.D.; García-Aracil, N. Hand exoskeleton for rehabilitation therapies with integrated optical force sensor. Adv. Mech. Eng. 2018, 10, 1687814017753881. [Google Scholar] [CrossRef]
- Brandenburg, J.E.; Eby, S.F.; Song, P.; Zhao, H.; Brault, J.S.; Chen, S.; An, K.-N. Ultrasound Elastography: The New Frontier in Direct Measurement of Muscle Stiffness. Arch. Phys. Med. Rehab. 2014, 95, 2207–2219. [Google Scholar] [CrossRef]
- Creze, M.; Nordez, A.; Soubeyrand, M.; Rocher, L.; Maître, X.; Bellin, M.F. Shear wave sonoelastography of skeletal muscle: Basic principles, biomechanical concepts, clinical applications, and future perspectives. Skelet. Radiol. 2018, 47, 457–471. [Google Scholar] [CrossRef]
- Ryu, J.; Jeong, W.K. Current status of musculoskeletal application of shear wave elastography. Ultrasonography 2017, 36, 185–197. [Google Scholar] [CrossRef]
- e Lima, K.M.M.; Júnior, J.F.S.C.; de Albuquerque Pereira, W.C.; de Oliveira, L.F. Assessment of the mechanical properties of the muscle-tendon unit by supersonic shear wave imaging elastography: A review. Ultrasonography 2018, 37, 3–15. [Google Scholar] [CrossRef]
- Blank, J.; Blomquist, M.; Arant, L.; Cone, S.; Roth, J. Characterizing Musculoskeletal Tissue Mechanics Based on Shear Wave Propagation: A Systematic Review of Current Methods and Reported Measurements. Ann. Biomed. Eng. 2022, 50, 751–768. [Google Scholar] [CrossRef]
- Davis, L.C.; Baumer, T.G.; Bey, M.J.; Van Holsbeeck, M.T. Clinical utilization of shear wave elastography in the musculoskeletal system. Ultrasonography 2019, 38, 2–12. [Google Scholar] [CrossRef] [PubMed]
- Romer, C.; Czupajllo, J.; Zessin, E.; Fischer, T.; Wolfarth, B.; Lerchbaumer, M.H. Stiffness of Muscles and Tendons of the Lower Limb of Professional and Semiprofessional Athletes Using Shear Wave Elastography. J. Ultrasound Med. 2022, 41, 3061–3068. [Google Scholar] [CrossRef] [PubMed]
- Friede, M.C.; Klauser, A.; Fink, C.; Csapo, R. Stiffness of the iliotibial band and associated muscles in runner’s knee: Assessing the effects of physiotherapy through ultrasound shear wave elastography. Phys. Ther. Sport 2020, 45, 126–134. [Google Scholar] [CrossRef]
- Mendes, B.; Firmino, T.; Oliveira, R.; Neto, T.; Infante, J.; Vaz, J.R.; Freitas, S.R. Hamstring stiffness pattern during contraction in healthy individuals: Analysis by ultrasound-based shear wave elastography. Eur. J. Appl. Physiol. 2018, 118, 2403–2415. [Google Scholar] [CrossRef] [PubMed]
- Römer, C.; Czupajllo, J.; Zessin, E.; Fischer, T.; Wolfarth, B.; Lerchbaumer, M.H. Muscle and Tendon Stiffness of the Lower Limb of Professional Adolescent Soccer Athletes Measured Using Shear Wave Elastography. Diagnostics 2022, 12, 2453. [Google Scholar] [CrossRef]
- Zhang, H.; Peng, W.; Qin, C.; Miao, Y.; Zhou, F.; Ma, Y.; Gao, Y. Lower Leg Muscle Stiffness on Two-Dimensional Shear Wave Elastography in Subjects with Medial Tibial Stress Syndrome. J. Ultrasound Med. 2022, 41, 1633–1642. [Google Scholar] [CrossRef]
- Wang, Z.; Lyu, G.; Zhong, H.; Yan, L.; Xu, Z. Shear Wave Elastography for Detecting Calf Muscle Stiffness: An Effective Tool for Assessing Sarcopenia. J. Ultrasound Med. 2023, 42, 891–900. [Google Scholar] [CrossRef]
- Yin, L.; Du, L.; Li, Y.; Xiao, Y.; Zhang, S.; Ma, H.; He, W. Quantitative Evaluation of Gastrocnemius Medialis Stiffness During Passive Stretching Using Shear Wave Elastography in Patients with Parkinson’s Disease: A Prospective Preliminary Study. Korean J. Radiol. 2021, 22, 1841–1849. [Google Scholar] [CrossRef]
- Guo, Y.; Li, X.M.; Zhang, H.; Lu, H.T.; Feng, B.; Wang, Y.Z. Application of Ultrasound Shear Wave Elastography in Rehabilitation Assessment of Triceps Surae and Achilles Tendon after Stroke. Chin. J. Rehabil. Theory Pract. 2020, 12, 753–756. [Google Scholar]
- Ding, C.W.; Song, X.; Fu, X.Y.; Zhang, Y.C.; Mao, P.; Sheng, Y.J.; Yang, M.; Wang, C.S.; Zhang, Y.; Chen, X.F.; et al. Shear wave elastography characteristics of upper limb muscle in rigidity-dominant Parkinson’s disease. Neurol. Sci. 2021, 42, 4155–4162. [Google Scholar] [CrossRef]
- Analan, P.D.; Ozdemir, H. Assessment of Post-Stroke Biceps Brachialis Muscle Stiffness by Shear-Wave Elastography: A Pilot Study. Muscles Ligaments Tendons J. 2020, 10, 531–536. [Google Scholar] [CrossRef]
- Mifune, Y.; Inui, A.; Nishimoto, H.; Kataoka, T.; Kurosawa, T.; Yamaura, K.; Mukohara, S.; Niikura, T.; Kokubu, T.; Akisue, T.; et al. Assessment of posterior shoulder muscle stiffness related to posterior shoulder tightness in college baseball players using shear wave elastography. J. Shoulder Elb. Surg. 2020, 29, 571–577. [Google Scholar] [CrossRef] [PubMed]
- Dieterich, A.V.; Yavuz, U.Ş.; Petzke, F.; Nordez, A.; Falla, D. Neck Muscle Stiffness Measured with Shear Wave Elastography in Women with Chronic Nonspecific Neck Pain. J. Orthop. Sport. Phys. Ther. 2020, 50, 179–188. [Google Scholar] [CrossRef]
- Ateş, F.; Hug, F.; Bouillard, K.; Jubeau, M.; Frappart, T.; Couade, M.; Bercoff, J.; Nordez, A. Muscle shear elastic modulus is linearly related to muscle torque over the entire range of isometric contraction intensity. J. Electromyogr. Kinesiol. 2015, 25, 703–708. [Google Scholar] [CrossRef]
- Watanabe, Y.; Iba, K.; Taniguchi, K.; Aoki, M.; Sonoda, T.; Yamashita, T. Assessment of the Passive Tension of the First Dorsal Interosseous and First Lumbrical Muscles Using Shear Wave Elastography. J. Hand Surg. 2019, 44, 1092.e1–1092.e8. [Google Scholar] [CrossRef]
- Shin, K.J.; Yi, J.; Hahn, S. Shear-wave elastography evaluation of thenar muscle in carpal tunnel syndrome. J. Clin. Ultrasound 2023, 51, 510–517. [Google Scholar] [CrossRef] [PubMed]
- Bernabei, M.; Lee, S.S.M.; Perreault, E.J.; Sandercock, T.G. Shear wave velocity is sensitive to changes in muscle stiffness that occur independently from changes in force. J. Appl. Physiol. 2020, 128, 8–16. [Google Scholar] [CrossRef]
- Herzog, W. The problem with skeletal muscle series elasticity. BMC Biomed. Eng. 2019, 1, 28. [Google Scholar] [CrossRef]
- Wang, A.B.; Perreault, E.J.; Royston, T.J.; Lee, S.S. Changes in shear wave propagation within skeletal muscle during active and passive force generation. J. Biomech. 2019, 94, 115–122. [Google Scholar] [CrossRef]
- Kozinc, Ž.; Šarabon, N. Shear-wave elastography for assessment of trapezius muscle stiffness: Reliability and association with low-level muscle activity. PLoS ONE 2020, 15, e0234359. [Google Scholar] [CrossRef]
- Wang, X.; Zhu, J.; Gao, J.; Hu, Y.; Liu, Y.; Li, W.; Chen, S.; Liu, F. Assessment of ultrasound shear wave elastography within muscles using different region of interest sizes, manufacturers, probes and acquisition angles: An ex vivo study. Quant. Imaging Med. Surg. 2022, 12, 3227–3237. [Google Scholar] [CrossRef] [PubMed]
- Romano, A.; Staber, D.; Grimm, A.; Kronlage, C.; Marquetand, J. Limitations of Muscle Ultrasound Shear Wave Elastography for Clinical Routine—Positioning and Muscle Selection. Sensors 2021, 21, 8490. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.-P.; Liu, C.-L.; Zhang, Z.-J. Feasibility of Using a Portable MyotonPRO Device to Quantify the Elastic Properties of Skeletal Muscle. Experiment 2022, 28, e934121-1. [Google Scholar] [CrossRef] [PubMed]
- Milerská, I.; Lhotská, L.; Macaš, M. Biomechanical Parameters of Muscles, Objective Assessment Using MyotonPRO. In Proceedings of the 2018 IEEE International Conference on Bioinformatics and Biomedicine (BIBM), Madrid, Spain, 3–6 December 2018; pp. 1522–1525. [Google Scholar]
- Bailey, L.; Samuel, D.; Warner, M.B.; Stokes, M. Parameters representing muscle tone, elasticity and stiffness of biceps brachii in healthy older males: Symmetry and within-session reliability using the MyotonPRO. J. Neurol. Disord. 2013, 1, 1–7. [Google Scholar] [CrossRef]
- Schneider, S.; Peipsi, A.; Stokes, M.; Knicker, A.; Abeln, V. Feasibility of monitoring muscle health in microgravity environments using Myoton technology. Med. Biol. Eng. Comput. 2015, 53, 57–66. [Google Scholar] [CrossRef]
- Liu, C.; Feng, Y.; Zhang, H.; Li, Y.; Zhu, Y.; Zhang, Z. Assessing the viscoelastic properties of upper trapezius muscle: Intra- and inter-tester reliability and the effect of shoulder elevation. J. Electromyogr. Kinesiol. 2018, 43, 226–229. [Google Scholar] [CrossRef]
- Liu, C.L.; Li, Y.P.; Wang, X.Q.; Zhang, Z.J. Quantifying the Stiffness of Achilles Tendon: Intra- and Inter-Operator Reliability and the Effect of Ankle Joint Motion. Experiment 2018, 24, 4876–4881. [Google Scholar] [CrossRef]
- Feng, Y.N.; Li, Y.P.; Liu, C.L.; Zhang, Z.J. Assessing the elastic properties of skeletal muscle and tendon using shear-wave ultrasound elastography and MyotonPRO. Sci. Rep. 2018, 8, 17064. [Google Scholar] [CrossRef]
- Kelly, J.P.; Koppenhaver, S.L.; Michener, L.A.; Proulx, L.; Bisagni, F.; Cleland, J.A. Characterization of tissue stiffness of the infraspinatus, erector spinae, and gastrocnemius muscle using ultrasound shear wave elastography and superficial mechanical deformation. J. Electromyogr. Kinesiol. 2018, 38, 73–80. [Google Scholar] [CrossRef]
- Madarshahian, S.; Latash, M.L. Effects of hand muscle function and dominance on intra-muscle synergies. Hum. Mov. Sci. 2022, 82, 102936. [Google Scholar] [CrossRef]
- Höppner, H.; Große-Dunker, M.; Stillfried, G.; Bayer, J.; van der Smagt, P. Key insights into hand biomechanics: Human grip stiffness can be decoupled from force by co-contraction and predicted from electromyography. Front. Neurorobotics 2017, 11, 17. [Google Scholar] [CrossRef] [PubMed]
- Martin, J.A.; Ramsay, J.; Hughes, C.; Peters, D.M.; Edwards, M.G. Age and grip strength predict hand dexterity in adults. PLoS ONE 2015, 10, e0117598. [Google Scholar] [CrossRef] [PubMed]
- Brahee, D.D.; Ogedegbe, C.; Hassler, C.; Nyirenda, T.; Hazelwood, V.; Morchel, H.; Patel, R.S.; Feldman, J. Body mass index and abdominal ultrasound image quality: A pilot survey of sonographers. J. Diagn. Med. Sonogr. 2013, 29, 66–72. [Google Scholar] [CrossRef]
- Oldfield, R.C. The assessment and analysis of handedness: The Edinburgh inventory. Neuropsychologia 1971, 9, 97–113. [Google Scholar] [CrossRef] [PubMed]
- Song, P.; Chen, S. Comb-push Ultrasound Shear Elastography. In Ultrasound Elastography for Biomedical Applications and Medicine; Wiley: Hoboken, NJ, USA, 2018; pp. 388–397. [Google Scholar]
- Song, P.; Urban, M.W.; Manduca, A.; Zhao, H.; Greenleaf, J.F.; Chen, S. Comb-push Ultrasound Shear Elastography (CUSE): A novel and fast technique for shear elasticity imaging. In Proceedings of the IEEE International Ultrasonics Symposium, Dresden, Germany, 7–10 October 2012; pp. 1842–1845. [Google Scholar]
- Song, P.; Urban, M.W.; Manduca, A.; Zhao, H.; Greenleaf, J.F.; Chen, S. Comb-Push Ultrasound Shear Elastography (CUSE) with Various Ultrasound Push Beams. IEEE Trans. Med. Imaging 2013, 32, 1435–1447. [Google Scholar] [CrossRef] [PubMed]
- Song, P.; Zhao, H.; Manduca, A.; Urban, M.W.; Greenleaf, J.F.; Chen, S. Comb-Push Ultrasound Shear Elastography (CUSE): A Novel Method for Two-Dimensional Shear Elasticity Imaging of Soft Tissues. IEEE Trans. Med. Imaging 2012, 31, 1821–1832. [Google Scholar] [CrossRef]
- Khowailed, I.A.; Lee, Y.; Lee, H. Assessing the differences in muscle stiffness measured with shear wave elastography and myotonometer during the menstrual cycle in young women. Clin. Physiol. Funct. Imaging 2022, 42, 320–326. [Google Scholar] [CrossRef] [PubMed]
- Maksuti, E.; Widman, E.; Larsson, D.; Urban, M.W.; Larsson, M.; Bjällmark, A. Arterial Stiffness Estimation by Shear Wave Elastography: Validation in Phantoms with Mechanical Testing. Ultrasound Med. Biol. 2016, 42, 308–321. [Google Scholar] [CrossRef]
- Marlevi, D.; Maksuti, E.; Urban, M.W.; Winter, R.; Larsson, M. Plaque characterization using shear wave elastography—Evaluation of differentiability and accuracy using a combined ex vivo and in vitro setup. Phys. Med. Biol. 2018, 63, 235008. [Google Scholar] [CrossRef]
- Garra, B.S. Elastography: History, principles, and technique comparison. Abdom. Imaging 2015, 40, 680–697. [Google Scholar] [CrossRef]
- Lee, Y.; Kim, M.; Lee, H. The Measurement of Stiffness for Major Muscles with Shear Wave Elastography and Myoton: A Quantitative Analysis Study. Diagnostics 2021, 11, 524. [Google Scholar] [PubMed]
- Okafor, L.; Varacallo, M. Anatomy, Shoulder and Upper Limb, Hand Flexor Digitorum Superficialis Muscle. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2021. [Google Scholar]
- Baumer, T.G.; Davis, L.; Dischler, J.; Siegal, D.S.; van Holsbeeck, M.; Moutzouros, V.; Bey, M.J. Shear wave elastography of the supraspinatus muscle and tendon: Repeatability and preliminary findings. J. Biomech. 2017, 53, 201–204. [Google Scholar] [CrossRef] [PubMed]
- Dieterich, A.V.; Andrade, R.J.; Le Sant, G.; Falla, D.; Petzke, F.; Hug, F.; Nordez, A. Shear wave elastography reveals different degrees of passive and active stiffness of the neck extensor muscles. Eur. J. Appl. Physiol. 2017, 117, 171–178. [Google Scholar] [PubMed]
- Gennisson, J.-L.; Deffieux, T.; Macé, E.; Montaldo, G.; Fink, M.; Tanter, M. Viscoelastic and Anisotropic Mechanical Properties of in vivo Muscle Tissue Assessed by Supersonic Shear Imaging. Ultrasound Med. Biol. 2010, 36, 789–801. [Google Scholar] [CrossRef]
- Kuo, P.-H.; Deshpande, A.D. Muscle-tendon units provide limited contributions to the passive stiffness of the index finger metacarpophalangeal joint. J. Biomech. 2012, 45, 2531–2538. [Google Scholar] [CrossRef]
- Roberts, T.J. Contribution of elastic tissues to the mechanics and energetics of muscle function during movement. J. Exp. Biol. 2016, 219, 266–275. [Google Scholar] [CrossRef]
- Bouillard, K.; Nordez, A.; Hug, F. Estimation of individual muscle force using elastography. PLoS ONE 2021, 6, e29261. [Google Scholar] [CrossRef]
- Hug, F.; Tucker, K.; Gennisson, J.L.; Tanter, M.; Nordez, A. Elastography for muscle biomechanics: Toward the estimation of individual muscle force. Exerc. Sport Sci. Rev. 2015, 43, 125–133. [Google Scholar] [CrossRef]
| Total (n = 25) | Males (n = 12) | Females (n = 13) | p-Value | |
|---|---|---|---|---|
| Age (years) | 29.60 ± 9.81 | 27.92 ± 5.53 | 31.15 ± 12.73 | 0.017 * |
| Weight (kg) | 65.85 ± 14.76 | 73.67 ± 14.62 | 58.63 ± 11.04 | 0.915 |
| Height (m) | 1.69 ± 0.10 | 1.76 ± 0.07 | 1.62 ± 0.06 | 0.101 |
| BMI (kg/m2) | 22.95 ± 3.53 | 23.61 ± 3.35 | 22.34 ± 3.71 | 0.899 |
| 0%MVC | 20%MVC | 40%MVC | 60%MVC | 80%MVC | |
|---|---|---|---|---|---|
| Young’s modulus (kPa): | |||||
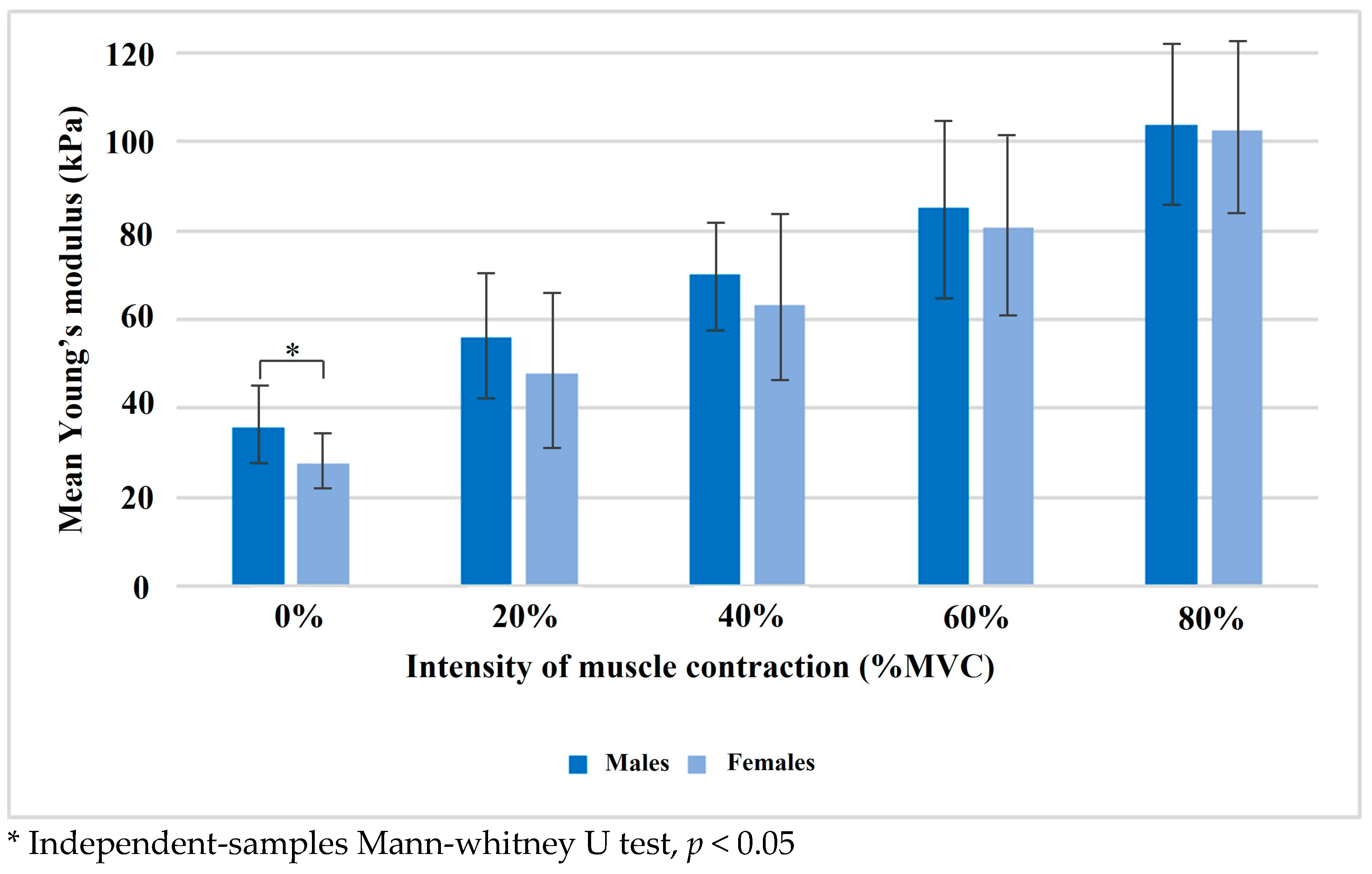
| Male | 35.86 ± 8.74 | 55.92 ± 14.73 | 70.00 ± 11.81 | 85.15 ± 18.95 | 103.79 ± 17.76 |
| Female | 27.47 ± 5.59 | 47.91 ± 16.76 | 63.16 ± 19.26 | 80.82 ± 19.67 | 102.39 ± 20.36 |
| p-value | 0.010 * | 0.168 | 0.247 | 0.769 | 0.728 |
| Dynamic muscle stiffness (N/m): | |||||
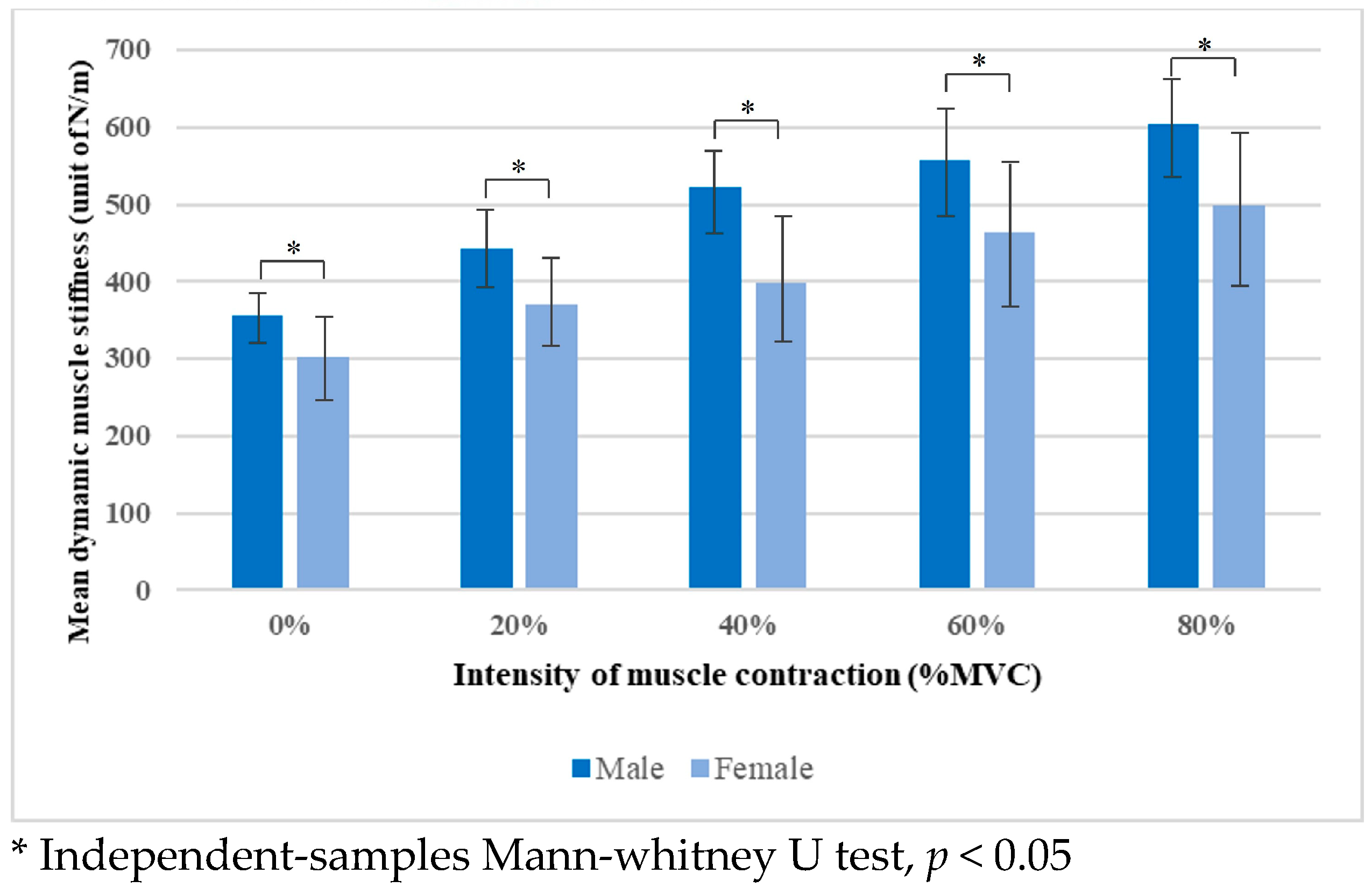
| Male | 355.46 ± 26.07 | 442.96 ± 50.12 | 522.58 ± 53.04 | 556.63 ± 62.74 | 603.63 ± 70.70 |
| Female | 303.54 ± 45.66 | 371.12 ± 57.99 | 398.81 ± 88.15 | 464.69 ± 90.46 | 497.73 ± 98.25 |
| p-value | 0.005 * | 0.005 * | <0.001 * | 0.002 * | 0.007 * |
| Young’s Modulus (kPa): | Dynamic Muscle Stiffness (N/m): | R | p-Value | |
|---|---|---|---|---|
| 0%MVC | 31.50 ± 8.30 | 328.46 ± 45.33 | 0.489 | 0.013 * |
| 20%MVC | 51.76 ± 16.02 | 405.60 ± 64.61 | 0.366 | 0.072 |
| 40%MVC | 66.44 ± 16.17 | 458.22 ± 121.72 | 0.479 | 0.015 * |
| 60%MVC | 82.90 ± 19.05 | 508.82 ± 89.96 | 0.330 | 0.107 |
| 80%MVC | 103.07 ± 18.77 | 548.56 ± 100.17 | 0.243 | 0.242 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tantipoon, P.; Praditpod, N.; Pakleppa, M.; Li, C.; Huang, Z. Characterization of Flexor Digitorum Superficialis Muscle Stiffness Using Ultrasound Shear Wave Elastography and MyotonPRO: A Cross-Sectional Study Investigating the Correlation between Different Approaches. Appl. Sci. 2023, 13, 6384. https://doi.org/10.3390/app13116384
Tantipoon P, Praditpod N, Pakleppa M, Li C, Huang Z. Characterization of Flexor Digitorum Superficialis Muscle Stiffness Using Ultrasound Shear Wave Elastography and MyotonPRO: A Cross-Sectional Study Investigating the Correlation between Different Approaches. Applied Sciences. 2023; 13(11):6384. https://doi.org/10.3390/app13116384
Chicago/Turabian StyleTantipoon, Phongpan, Nuttaporn Praditpod, Markus Pakleppa, Chunhui Li, and Zhihong Huang. 2023. "Characterization of Flexor Digitorum Superficialis Muscle Stiffness Using Ultrasound Shear Wave Elastography and MyotonPRO: A Cross-Sectional Study Investigating the Correlation between Different Approaches" Applied Sciences 13, no. 11: 6384. https://doi.org/10.3390/app13116384
APA StyleTantipoon, P., Praditpod, N., Pakleppa, M., Li, C., & Huang, Z. (2023). Characterization of Flexor Digitorum Superficialis Muscle Stiffness Using Ultrasound Shear Wave Elastography and MyotonPRO: A Cross-Sectional Study Investigating the Correlation between Different Approaches. Applied Sciences, 13(11), 6384. https://doi.org/10.3390/app13116384






