Design of the CIEMAT Corrosion Loop for Liquid Metal Experiments
Abstract
1. Introduction
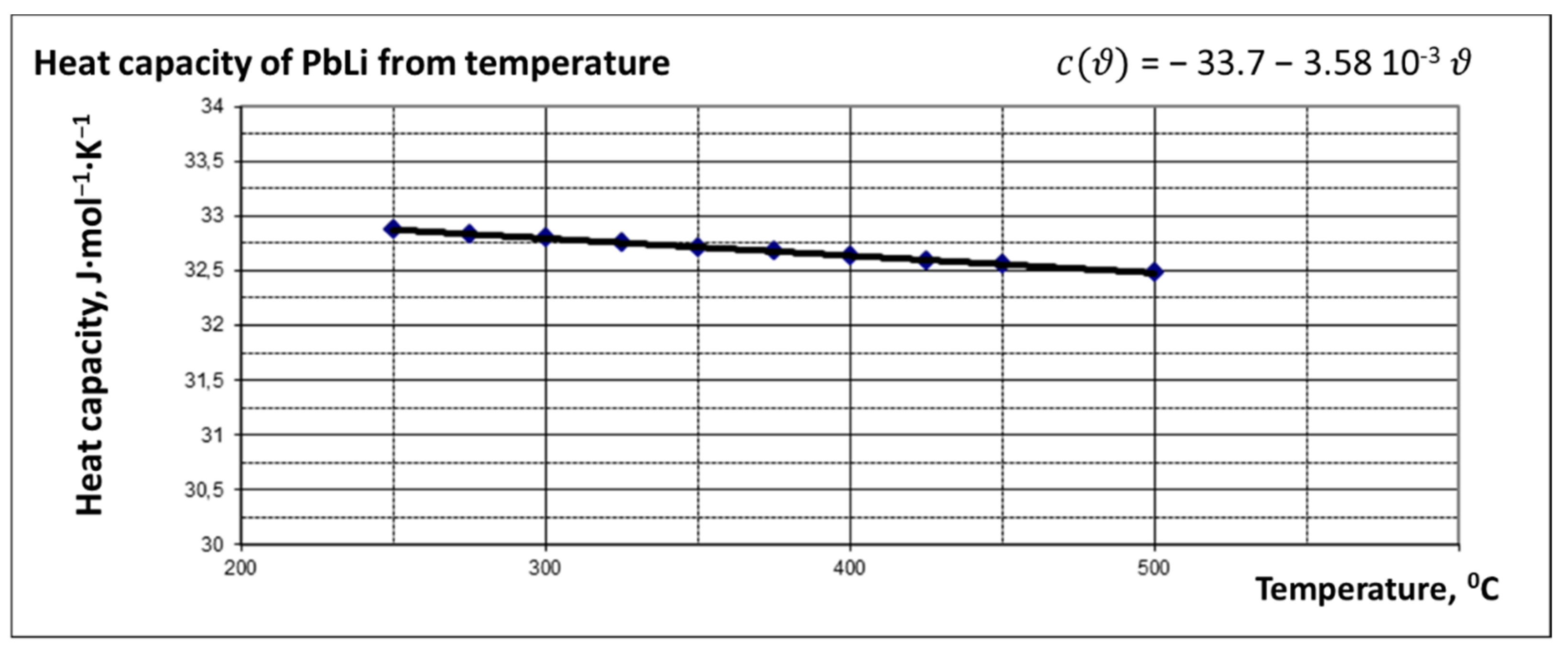
2. Corrosion Issue in a Lithium Lead Loop
- the melt itself (the impurity content and the composition of the impurities as well as the lithium content of the liquid metal alloy);
- the microstructure of the material in contact with lithium lead;
- the exposure time [33];
- the flow velocity and profile;
- the difference in temperature across the loop (ΔT in some specific part of the loop, such as cold traps or pump channels);
- the PbLi temperature profile.
3. CiCLo-C Loop: Requirements and Design
3.1. Main Components and Functioning
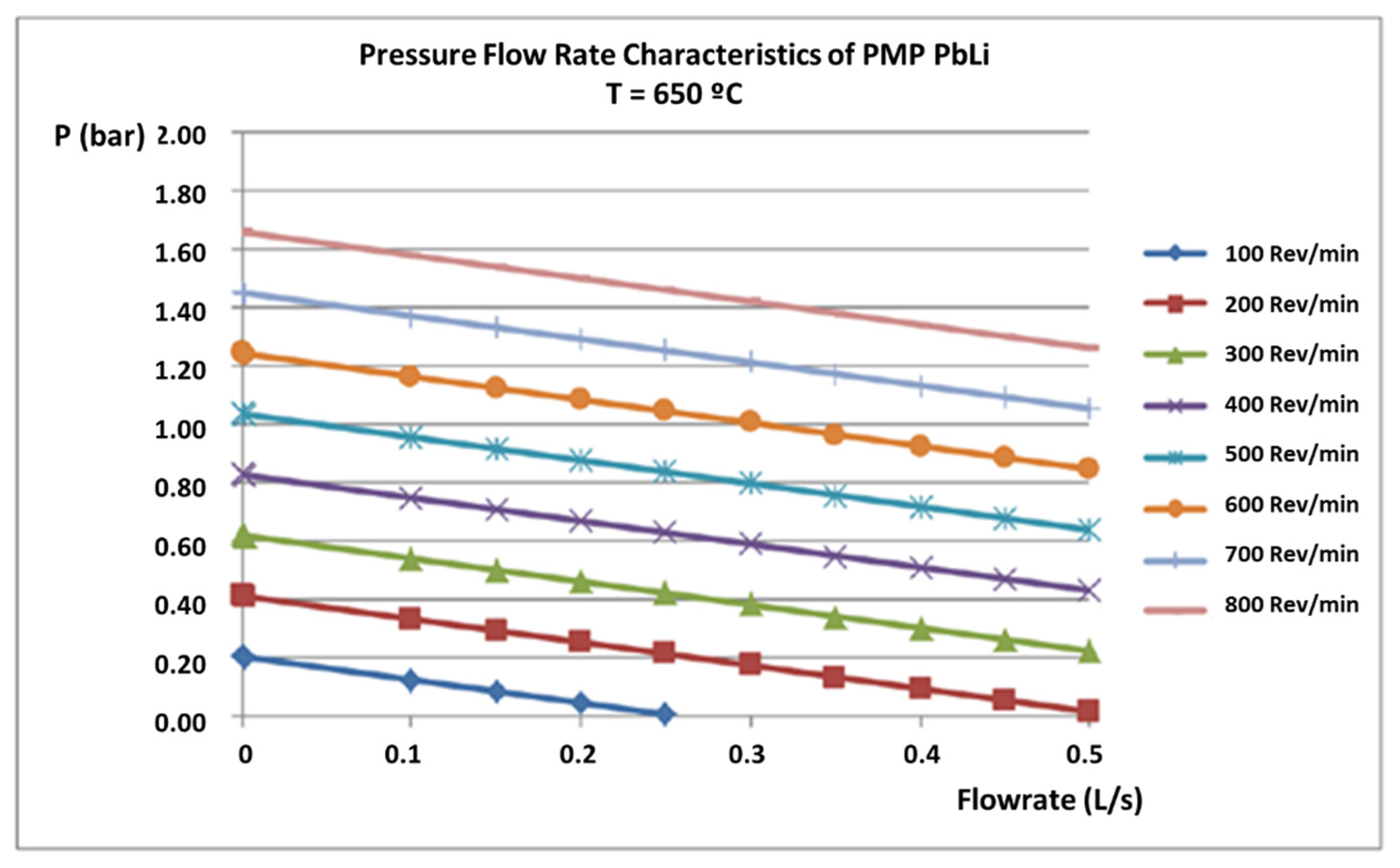
3.2. Flow Meter Calibration
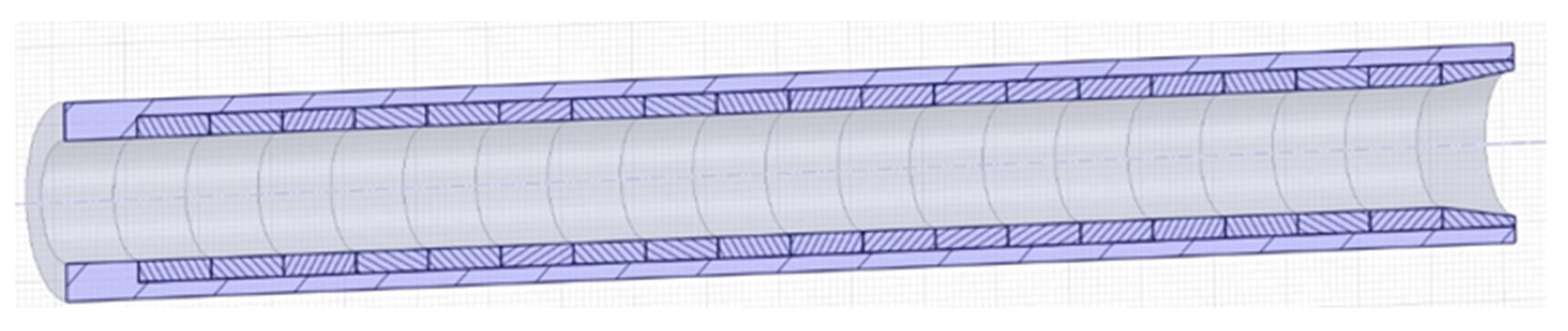
3.3. Test Section
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Lindau, R.; Möslang, A.; Rieth, M.; Klimiankou, M.; Materna-Morris, E.; Alamo, A.; Tavassoli, A.-A.F.; Cayron, C.; Lancha, A.-M.; Fernandez, P.; et al. Present development status of EUROFER and ODS-EUROFER for application in blanket concepts. Fusion Eng. Des. 2005, 75–79, 989–996. [Google Scholar] [CrossRef]
- Boccaccini, L.V.; Aiello, G.; Aubert, J.; Bachmann, C.; Barrett, T.; Del Nevo, A.; Demange, D.; Forest, L.; Hernandez, F.; Norajitra, P.; et al. Objectives and status of EUROfusion DEMO blanket studies. Fusion Eng. Des. 2016, 109–111, 1199–1206. [Google Scholar] [CrossRef]
- Kovari, M.; Coleman, M.; Cristescu, I.; Smith, R. Tritium resources available for fusion reactors. Nucl. Fusion 2017, 58, 026010. [Google Scholar] [CrossRef]
- Carella, E.; Hernández, T.; Moreno, B.; Chinarro, E. Electrical behaviour of ceramic breeder blankets in pebble form after γ-radiation. Nucl. Mater. Energy 2015, 3–4, 1–5. [Google Scholar] [CrossRef]
- Palermo, I.; Fernández, I.; Rapisarda, D.; Ibarra, A. Neutronic analyses of the preliminary design of a DCLL blanket for the EUROfusion DEMO power plant. Fusion Eng. Des. 2016, 109–111, 13–19. [Google Scholar] [CrossRef]
- Casini, G.; Sannier, J. Research and development on liquid Pb-17Li breeder in Europe. J. Nucl. Mater. 1991, 179–181, 47–52. [Google Scholar] [CrossRef]
- Coen, V. Lithium-lead eutectic as breeding material in fusion reactors. J. Nucl. Mater. 1985, 133–134, 46–51. [Google Scholar] [CrossRef]
- Fernandez, P.; Lancha, A.M.; Lapena, J.; Serrano, M.; Hernandez-Mayoral, M. Reduced Activation Ferritic/Martensitic Steel Eurofer 97 as Possible Structural Material for Fusion Devices. Metallurgical Characterization on As-Received Condition and after Simulated Services Conditions; Centro de Investigaciones Energeticas: Madrid, Spain, 2004; Available online: http://inis.iaea.org/Search/search.aspx?orig_q=RN:36026402 (accessed on 20 January 2021).
- Tortorelli, P.F.; Chopra, O.K. Corrosion and compatibility considerations of liquid metals for fusion reactor applications. J. Nucl. Mater. 1981, 103, 621–632. [Google Scholar] [CrossRef]
- Chopra, O.K.; Tortorelli, P.F. Compatibility of materials for use in liquid-metal blankets of fusion reactors. J. Nucl. Mater. 1984, 123, 1201–1212. [Google Scholar] [CrossRef][Green Version]
- Smolentsev, S.; Li, F.-C.; Morley, N.; Ueki, Y.; Abdou, M.; Sketchley, T. Construction and initial operation of MHD PbLi facility at UCLA. Fusion Eng. Des. 2013, 88, 317–326. [Google Scholar] [CrossRef]
- Martelli, D.; Barone, G.; Tarantino, M.; Utili, M. Design of a new experimental loop and of a coolant purifying system for corrosion experiments on EUROFER samples in flowing Pb-Li environment. Fusion Eng. Design. 2017, 124, 1144–1149. [Google Scholar] [CrossRef]
- Huang, Q.; Zhang, M.; Zhu, Z.; Gao, S.; Wu, Y.; Li, Y.; Song, Y.; Li, C.; Kong, M. Corrosion experiment in the first liquid metal LiPb loop of China. Fusion Eng. Des. 2007, 82, 2655–2659. [Google Scholar] [CrossRef]
- Karditsas, P.; Caloutsis, A. Corrosion and activation in fusion cooling loops—TRACT. Fusion Eng. Des. 2007, 82, 2729–2733. [Google Scholar] [CrossRef]
- Kumar, M.; Patel, A.; Jaiswal, A.; Ranjan, A.; Mohanta, D.; Sahu, S.; Saraswat, A.; Rao, P.; Rao, T.S.; Mehta, V.; et al. Engineering design and development of lead lithium loop for thermo-fluid MHD studies. Fusion Eng. Des. 2019, 138, 1–5. [Google Scholar] [CrossRef]
- Zhu, Z.; Huang, Q.; Gao, S.; Song, Y.; Li, C.; Peng, L.; Chen, H.; Liu, S.; Ling, X.; Chen, Y.; et al. Design analysis of DRAGON-IV LiPb loop. Fusion Eng. Des. 2011, 86, 2666–2669. [Google Scholar] [CrossRef]
- Venturini, A.; Utili, M.; Gabriele, A.; Ricapito, I.; Malavasi, A.; Forgione, N. Experimental and RELAP5-3D results on IELLLO (Integrated European Lead Lithium LOop) operation. Fusion Eng. Des. 2017, 123, 143–147. [Google Scholar] [CrossRef]
- Zhang, M. Corrosion experiment for CLAM and SS316L in liquid LiPb loop of China. Ann. Nucl. Energy 2015, 80, 203–206. [Google Scholar] [CrossRef]
- Konys, J.; Krauss, W. Corrosion and precipitation effects in a forced-convection Pb–15.7Li loop. J. Nucl. Mater. 2013, 442, S576–S579. [Google Scholar] [CrossRef]
- Bucenieks, I.; Krishbergs, R.; Platacis, E.; Lipsbergs, G.; Shishko, A.; Zik, A.; Muktup, F. Investigation of corrosion effects of eurofer steel in Pb-17Li stationary flow in the magnetic field. Magnetohydrodynamics 2006, 42, 237–251. [Google Scholar]
- Rapisarda, D.; Fernandez, I.; Palermo, I.; Urgorri, F.R.; Maqueda, L.; Alonso, D.; Melichar, T.; Frýbort, O.; Vála, L.; Gonzalez, M.; et al. Status of the engineering activities carried out on the European DCLL. Fusion Eng. Des. 2017, 124, 876–881. [Google Scholar] [CrossRef]
- Gazquez, M.C.; Bassini, S.; Hernandez, T.; Utili, M. Al2O3 coating as barrier against corrosion in Pb-17Li. Fusion Eng. Des. 2017, 124, 837–840. [Google Scholar] [CrossRef]
- Hernández, T.; Sánchez, F.J.; Moroño, A.; Aristu, M.; Marco, J.F. Effect of irradiation on the stability of the corrosion layer produced in EUROFER by contact with lithium ceramics. J. Nucl. Mater. 2021, 545, 152614. [Google Scholar] [CrossRef]
- Konys, J.; Krauss, W.; Steiner, H.; Novotny, J.; Skrypnik, A. Flow rate dependent corrosion behavior of Eurofer steel in Pb–15.7Li. J. Nucl. Mater. 2011, 417, 1191–1194. [Google Scholar] [CrossRef]
- Garcinuño, B.; Rapisarda, D.; Fernández, I.; Moreno, C.; Palermo, I.; Ibarra, Á. Design of a permeator against vacuum for tritium extraction from eutectic lithium-lead in a DCLL DEMO. Fusion Eng. Des. 2017, 117, 226–231. [Google Scholar] [CrossRef]
- Garcinuño, B.; Rapisarda, D.; Moreno, C.; Sanz, J.; Ibarra, Á. Design of a System for Hydrogen isotopes Injection into Lead-Lithium. Fusion Eng. Des. 2018, 137, 427–434. [Google Scholar] [CrossRef]
- Smolentsev, S.; Saedi, S.; Malang, S.; Abdou, M. Numerical study of corrosion of ferritic/martensitic steels in the flowing PbLi with and without a magnetic field. J. Nucl. Mater. 2013, 432, 294–304. [Google Scholar] [CrossRef]
- Precipitation Phenomena during Corrosion Testing in Forced-Convection Pb-15.7Li Loop PICOLO | Elsevier Enhanced Reader. Available online: https://www.sciencedirect.com/science/article/abs/pii/S0920379619303564 (accessed on 2 February 2022).
- Corrosion Properties | Tantaline® Surface Treatment. Tantaline®. Available online: https://tantaline.com/technology/corrosion-properties/ (accessed on 2 February 2022).
- Muñoz, P.; Malo, M.; Moroño, A.; García-Cortés, I.; Cabrera, S. RIPER: An irradiation facility to test Radiation Induced Permeation and release of deuterium for fusion blanket materials. Fusion Eng. Des. 2019, 145, 66–71. [Google Scholar] [CrossRef]
- Electron Accelerator | Laboratorio Nacional de Fusión. Available online: http://www.fusion.ciemat.es/competitive-access-to-facilities/electron-accelerator/ (accessed on 2 February 2022).
- González de Vicente, S.M.; Boutard, J.-L.; Zinkle, S.J.; Tanigawa, H. Materials testing facilities and programmes for fission and ion implantation damage. Nucl. Fusion 2017, 57, 092011. [Google Scholar] [CrossRef]
- Konys, J.; Krauss, W.; Zhu, Z.; Huang, Q. Comparison of corrosion behavior of EUROFER and CLAM steels in flowing Pb–15.7Li. J. Nucl. Mater. 2014, 455, 491–495. [Google Scholar] [CrossRef]
- Abánades, A.; García, A.; Casal, N.; Perlado, J.M.; Ibarra, A. Conceptual design of the liquid metal laboratory of the TECHNOFUSION facility. Fusion Eng. Des. 2012, 87, 161–166. [Google Scholar] [CrossRef][Green Version]
- Garcinuño, B.; Rapisarda, D.; Fernández-Berceruelo, I.; Carella, E.; Sanz, J. The CIEMAT LiPb Loop Permeation Experiment. Fusion Eng. Des. 2019, 146, 1228–1232. [Google Scholar] [CrossRef]
- Lenk, S. Theory of Flywheel V7 2019. Available online: https://saasonline.de/en/measure/ (accessed on 2 February 2022).


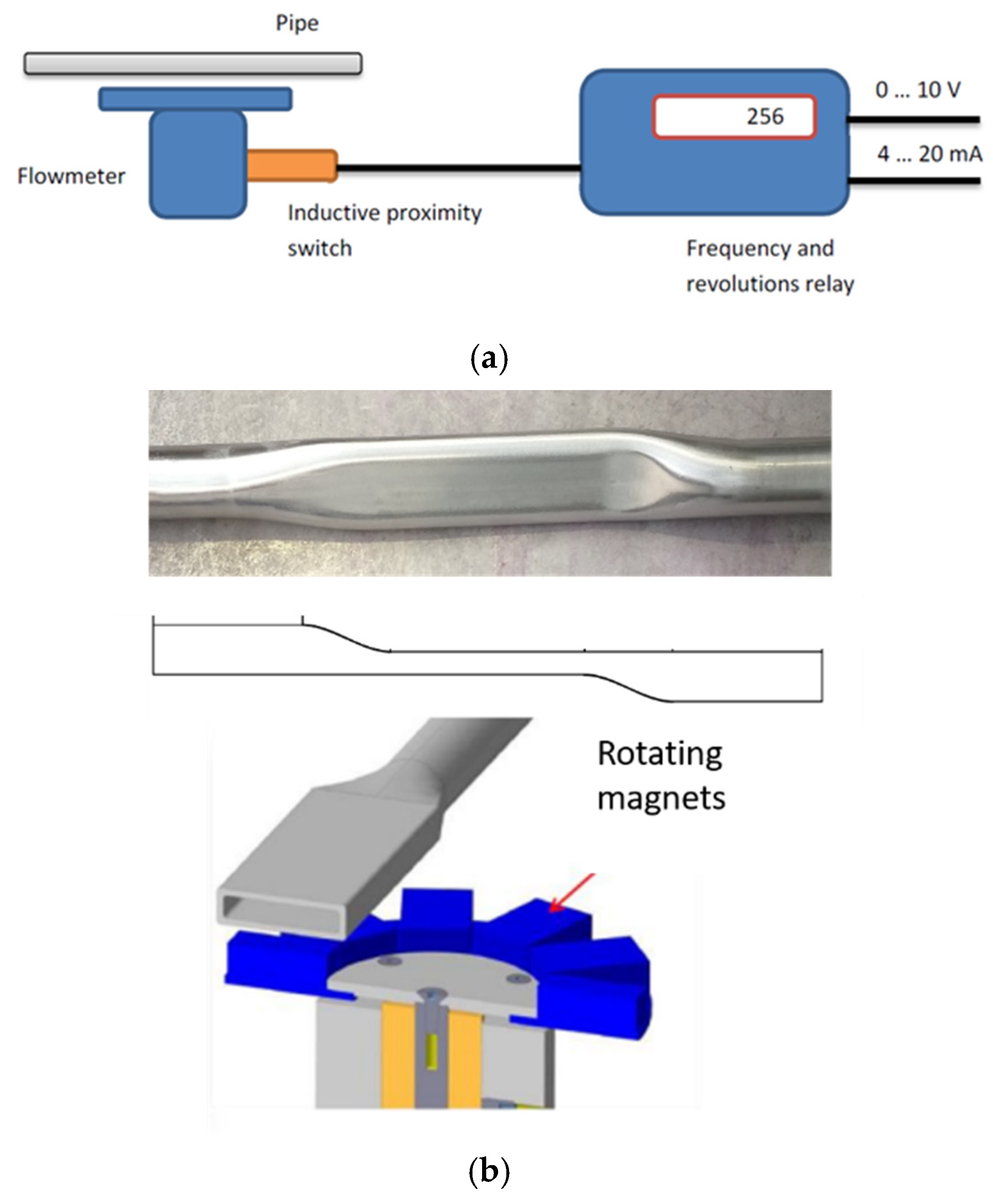




| Main Components | |
|---|---|
| 1 | Fluid temperature measurement |
| 2 | Connection to level probes |
| 3 | Connection to argon supply |
| 4 | Expansion tank S-001 |
| 5 | Loop pipes |
| 6 | Test section (TS) |
| 7 | Heat exchanger air outlet |
| 8 | Pneumatic ball valve |
| 9 | System filling line |
| 10 | System draining line |
| 11 | Draining tank S-002 |
| 12 | Heat exchanger air inlet |
| 13 | Rack |
| 14 | Pneumatic horizontal drive for flow meter |
| 15 | Flow meter |
| 16 | Permanent magnetic pump (PMP) |
| Parameters | Values |
|---|---|
| Temperature of the liquid PbLi | Max 600 °C |
| Volume flow of the loop | Max 0.5 L/s |
| Static pressure | Max 3 bar |
| Capacity PbLi | 7 L |
| Gase value | Pressurized air 10 bar/argon 3 bar |
| Dimensions | 2360 × 2790 × 900 (height × length × width) |
| Total weight | app. 700 kg |
| Inner diameter | 32 mm |
| Outer diameter | 38 mm |
| Parameters | Inside Medium PbLi | Outside Medium Air |
|---|---|---|
| Mass flow rate (kg/h) | 6535 | 324.5 |
| Volume flow rate (m3/h) | 0.72 | 278.7 |
| Normal flow rate (m3/h) | - | 251.1 |
| Pressure inlet (bar) | 2 | 1.03 |
| Inlet temperature °C | 550 | 20 |
| Outlet temperature with cooler °C | 532 | 85 |
| Power transmission W | −1253 | 1253 |
| Operative Parameters | Test Section | Rest of the Loop |
|---|---|---|
| Design temperature (°C) | 550 | 400–550 |
| Min. temperature of PbLi (°C) | 475 | 400 |
| Max. temperature of PbLi (°C) | 550 | 475 |
| Max. PbLi mass flow rate (kg/s) | 2.72 | 2.77 |
| Max. PbLi velocity (m/s) | 1 | 0.25 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Carella, E.; Rapisarda, D.; Lenk, S. Design of the CIEMAT Corrosion Loop for Liquid Metal Experiments. Appl. Sci. 2022, 12, 3104. https://doi.org/10.3390/app12063104
Carella E, Rapisarda D, Lenk S. Design of the CIEMAT Corrosion Loop for Liquid Metal Experiments. Applied Sciences. 2022; 12(6):3104. https://doi.org/10.3390/app12063104
Chicago/Turabian StyleCarella, Elisabetta, David Rapisarda, and Stephan Lenk. 2022. "Design of the CIEMAT Corrosion Loop for Liquid Metal Experiments" Applied Sciences 12, no. 6: 3104. https://doi.org/10.3390/app12063104
APA StyleCarella, E., Rapisarda, D., & Lenk, S. (2022). Design of the CIEMAT Corrosion Loop for Liquid Metal Experiments. Applied Sciences, 12(6), 3104. https://doi.org/10.3390/app12063104






