Steam Pyrolysis of Oil Sludge for Energy-Valuable Products
Abstract
:1. Introduction
2. Materials and Methods
2.1. Oil Sludge
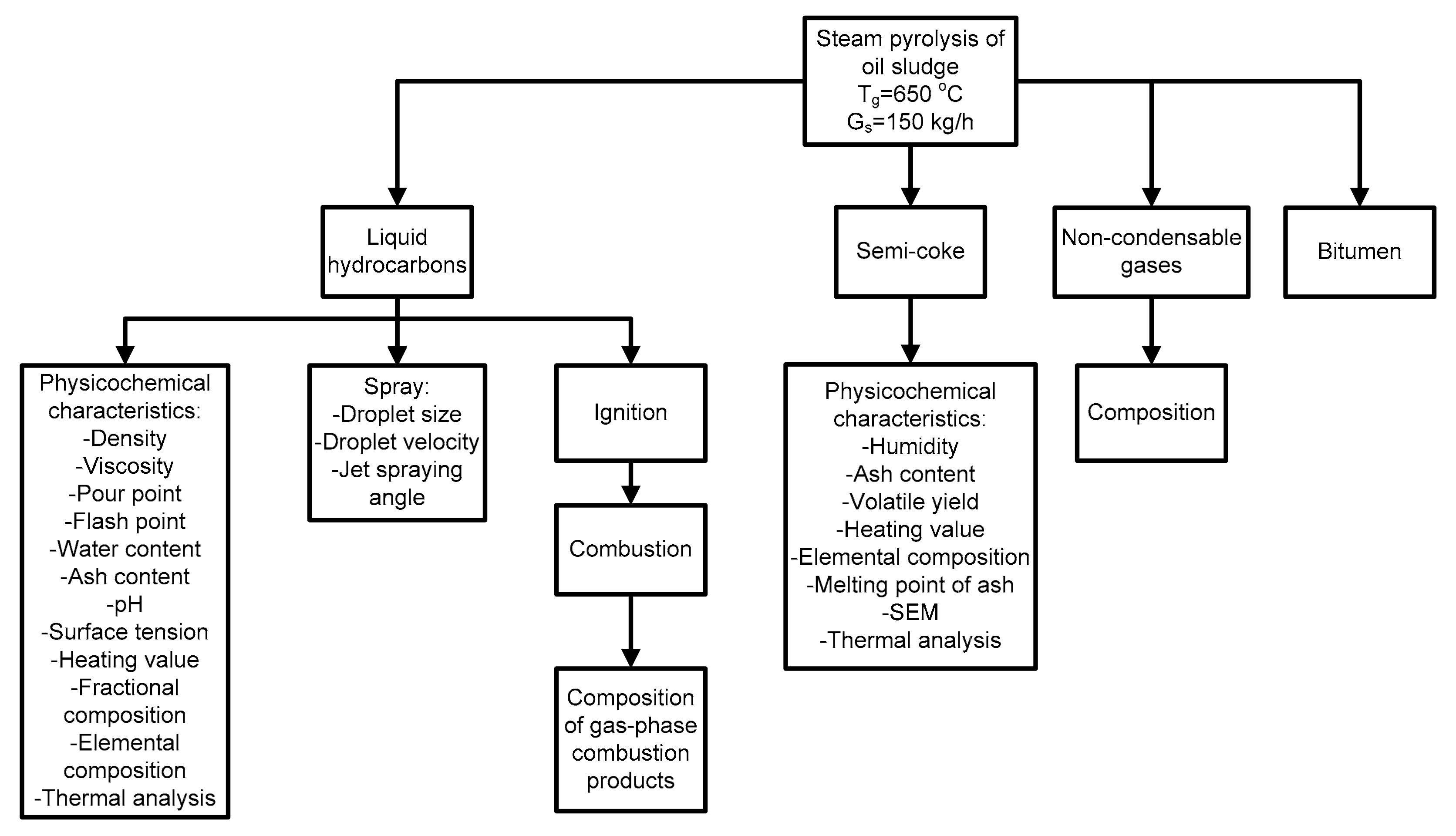
2.2. Methods Structure
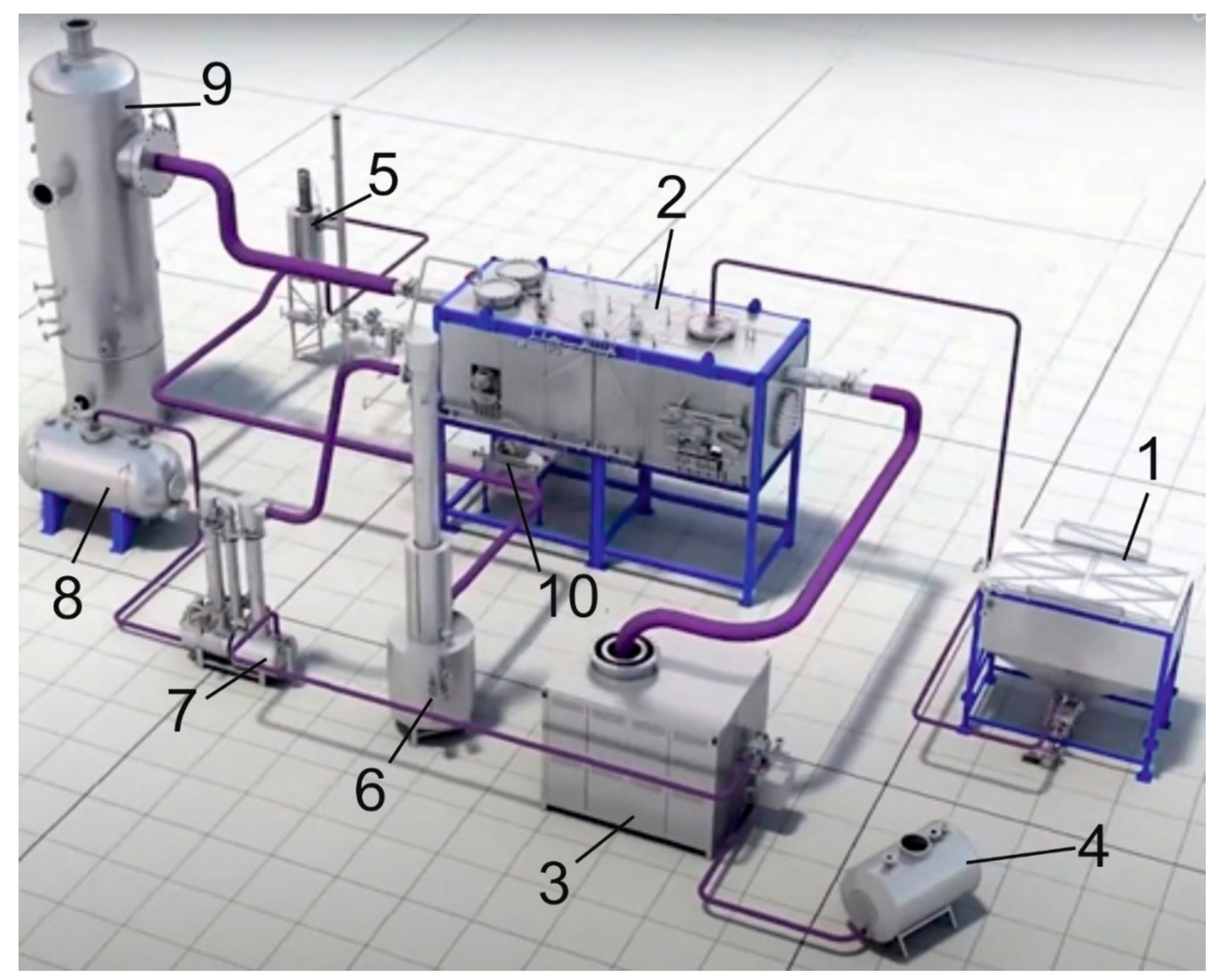
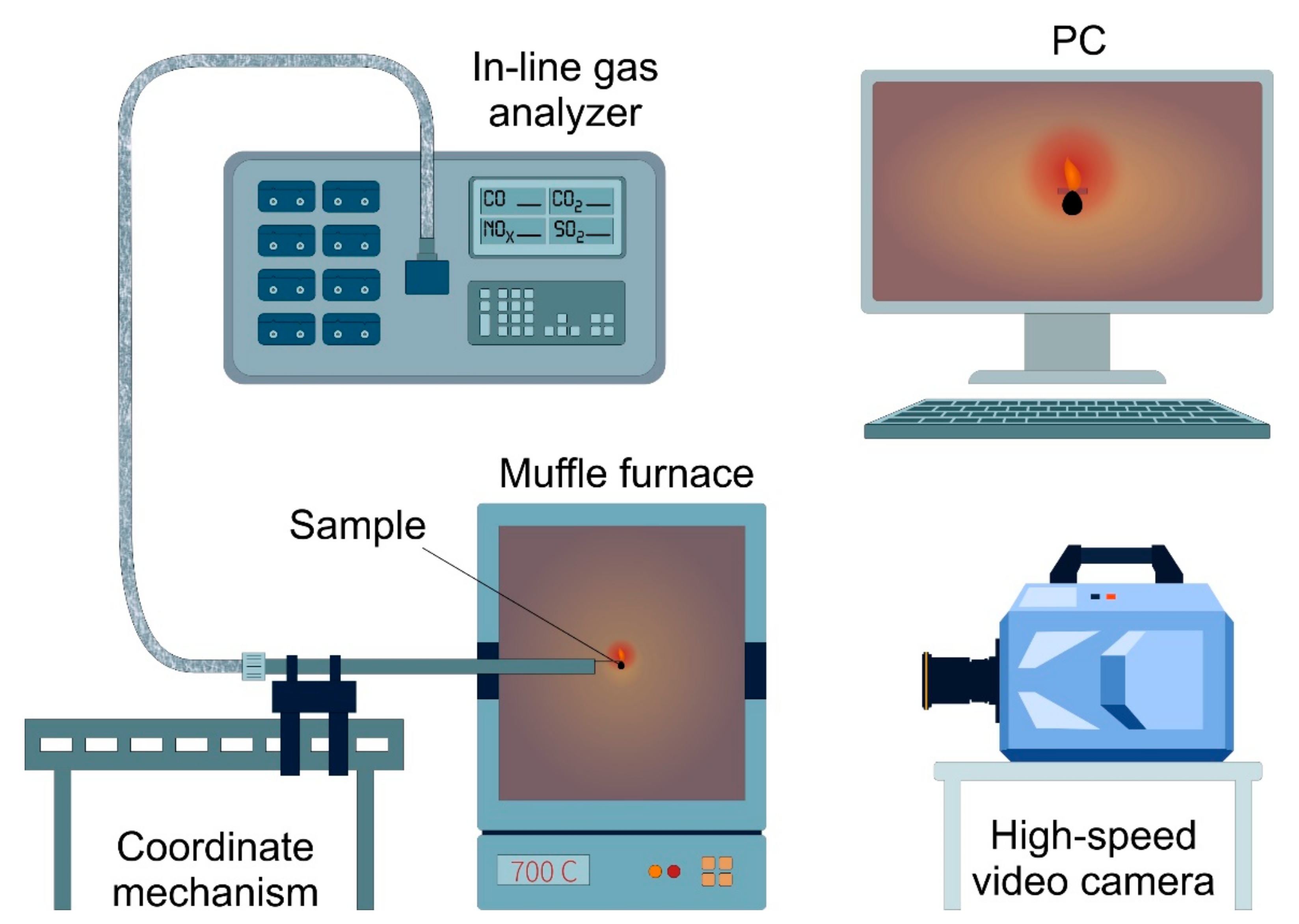
2.3. Setup for Steam Gasification of OS
2.4. Study of Semi-Coke Characteristics
2.5. Study of Liquid Hydrocarbon Characteristics
2.5.1. Characterization of Materials
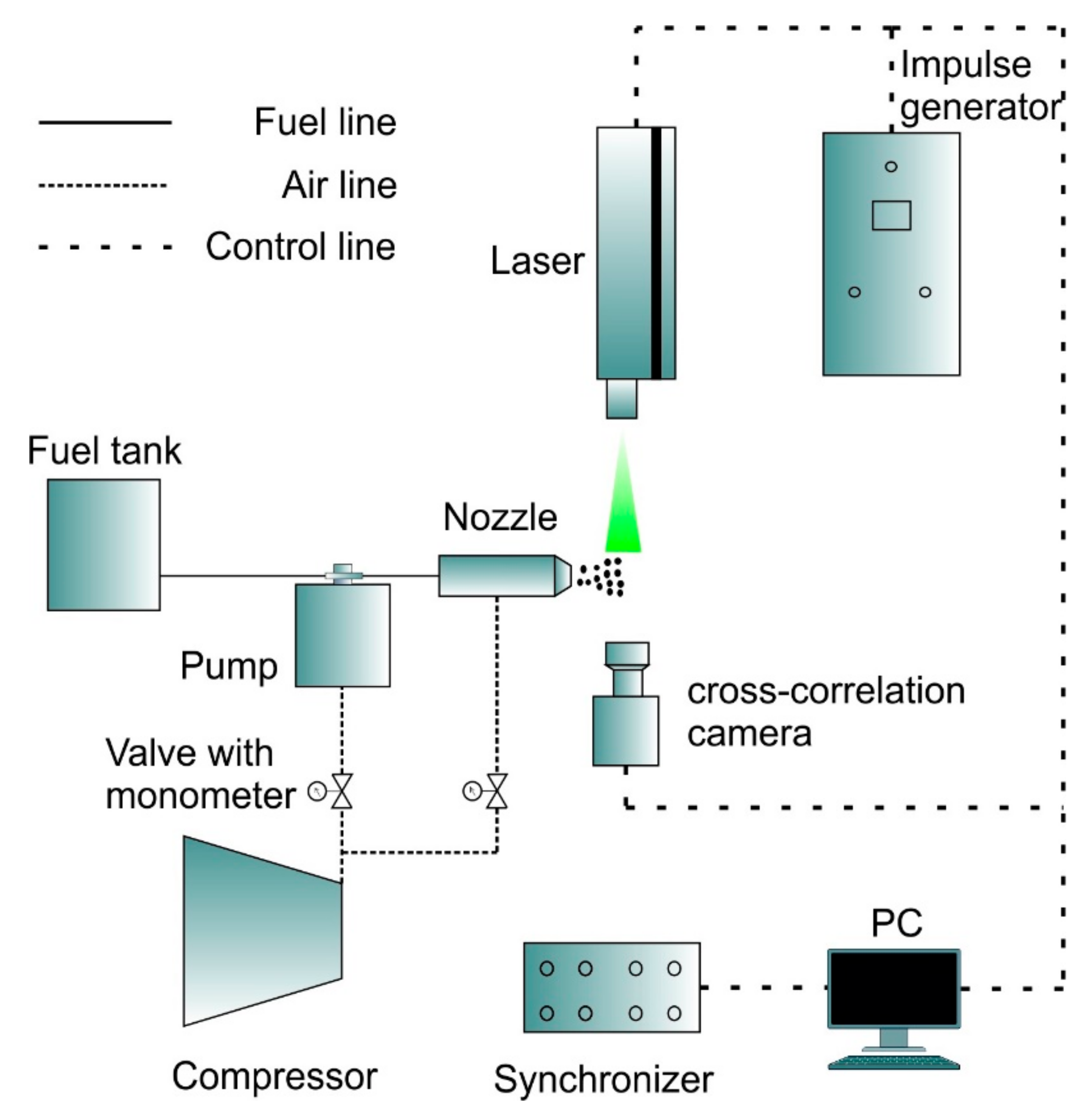
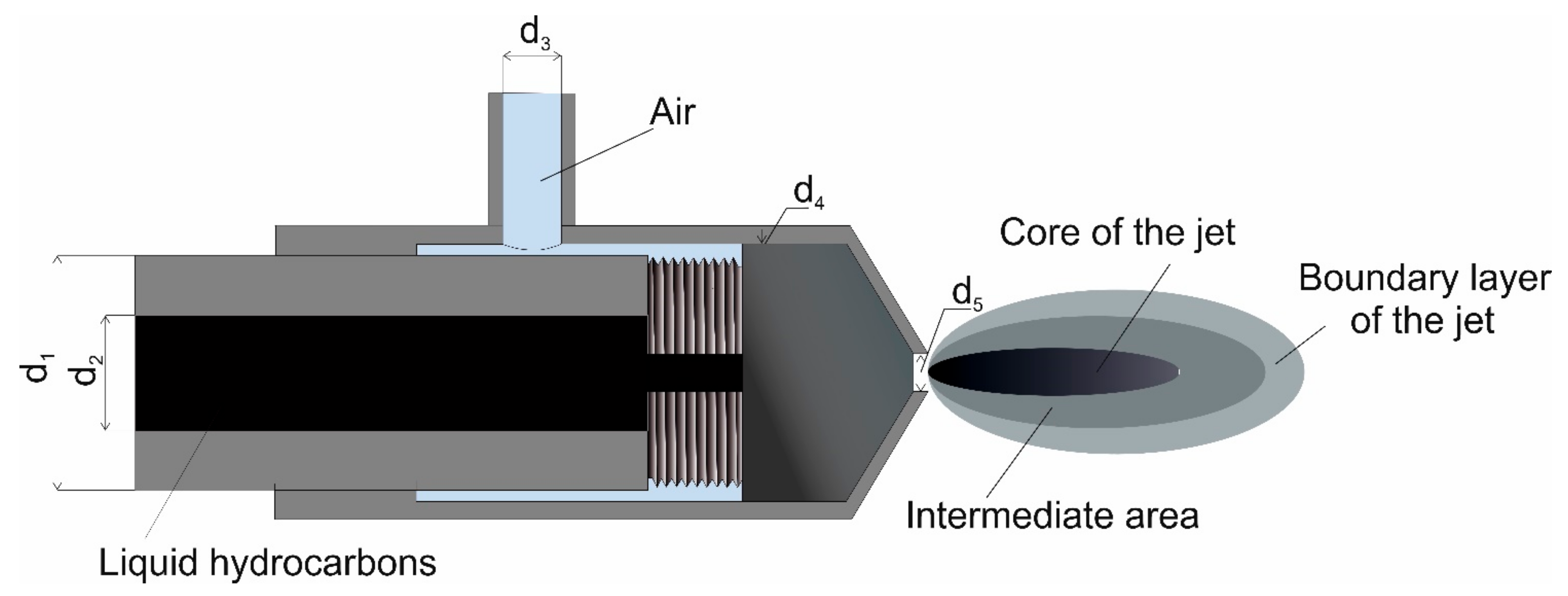
2.5.2. Experimental Study on Atomization Characteristics of Pyrolysis Oil Jet
2.5.3. Ignition and Combustion
3. Results
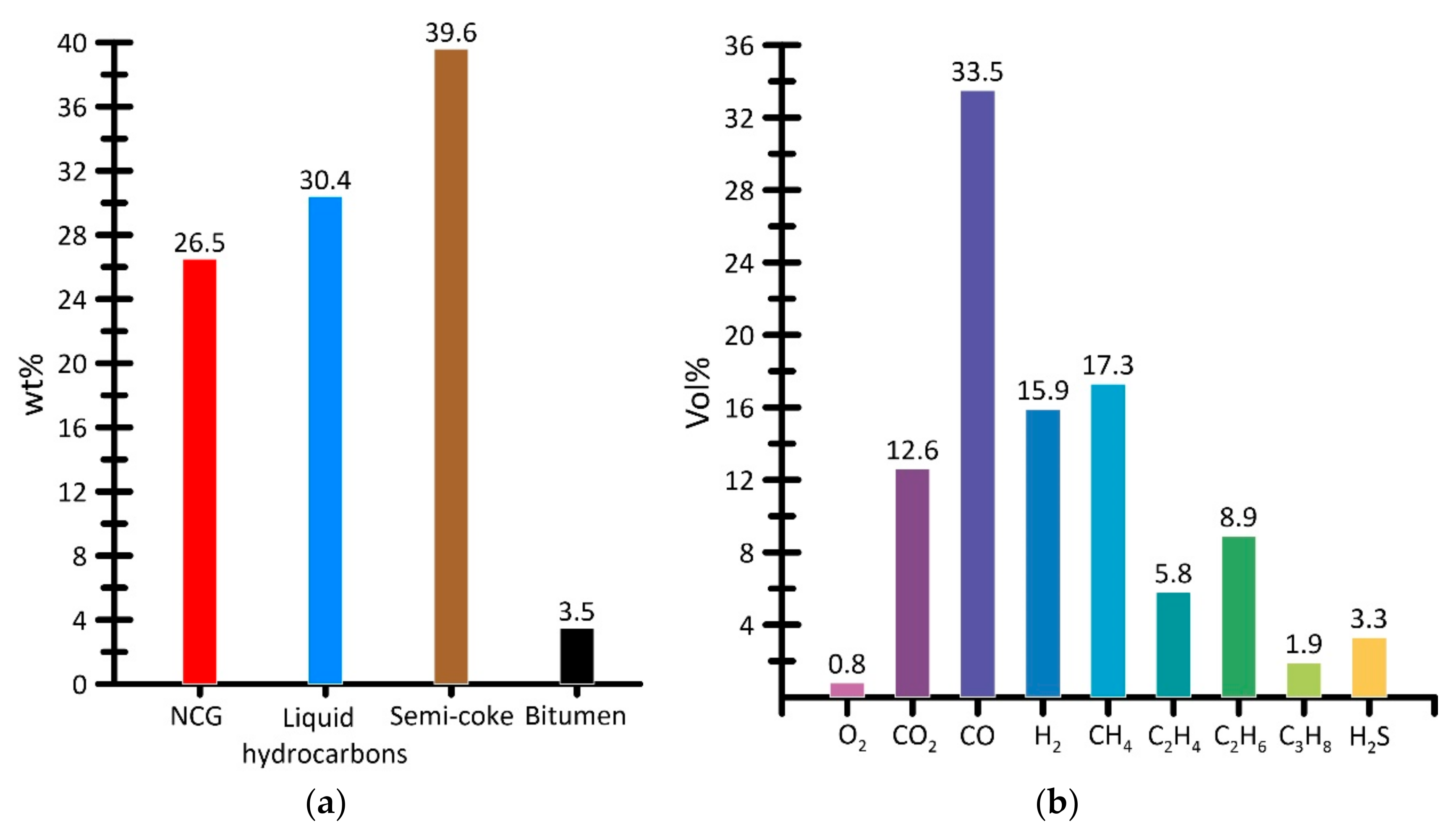
3.1. Material Balance of the Obtained Products of Steam Pyrolysis
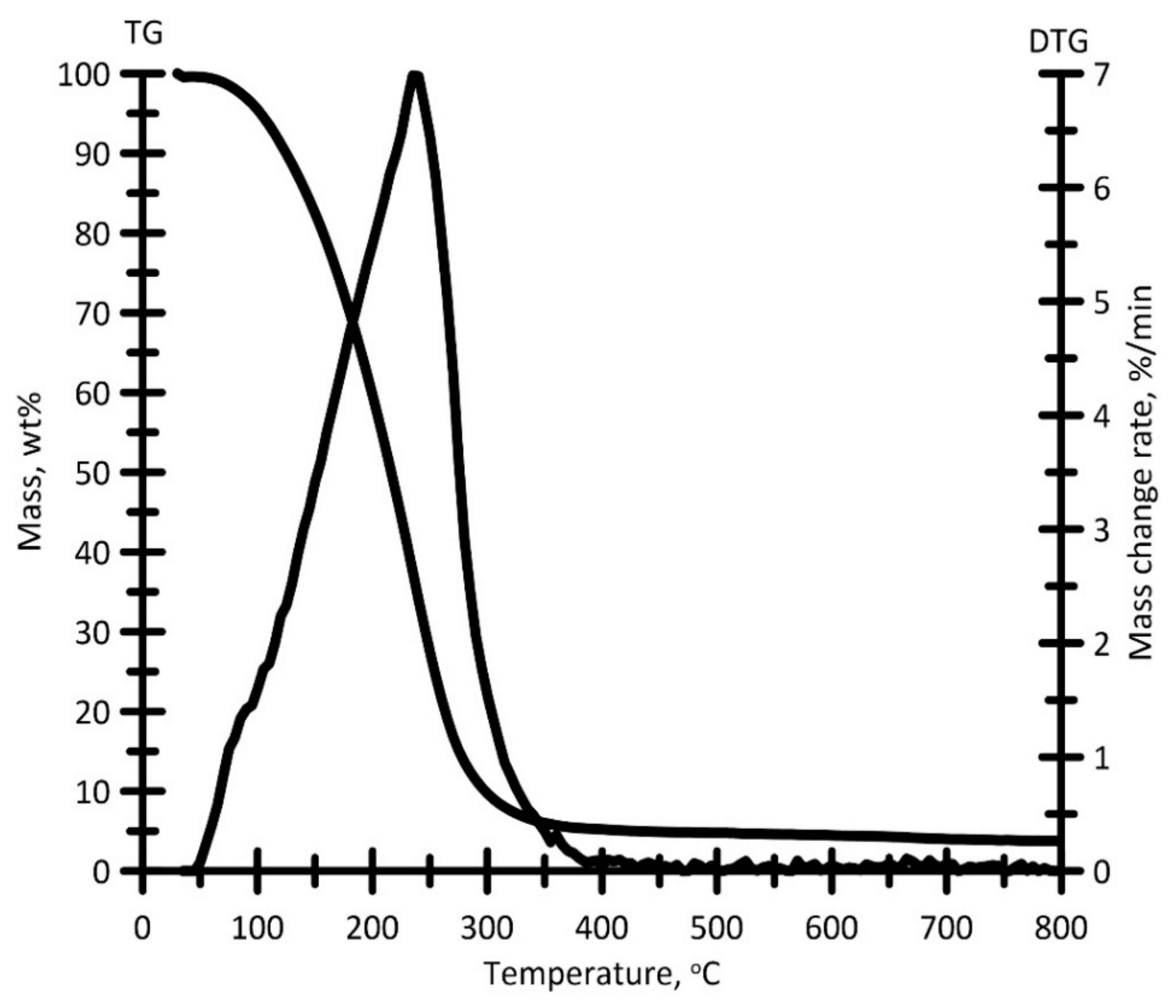
3.2. Characteristics of Semi-Coke
3.3. Liquid Hydrocarbons
3.3.1. Physical and Chemical Characteristics
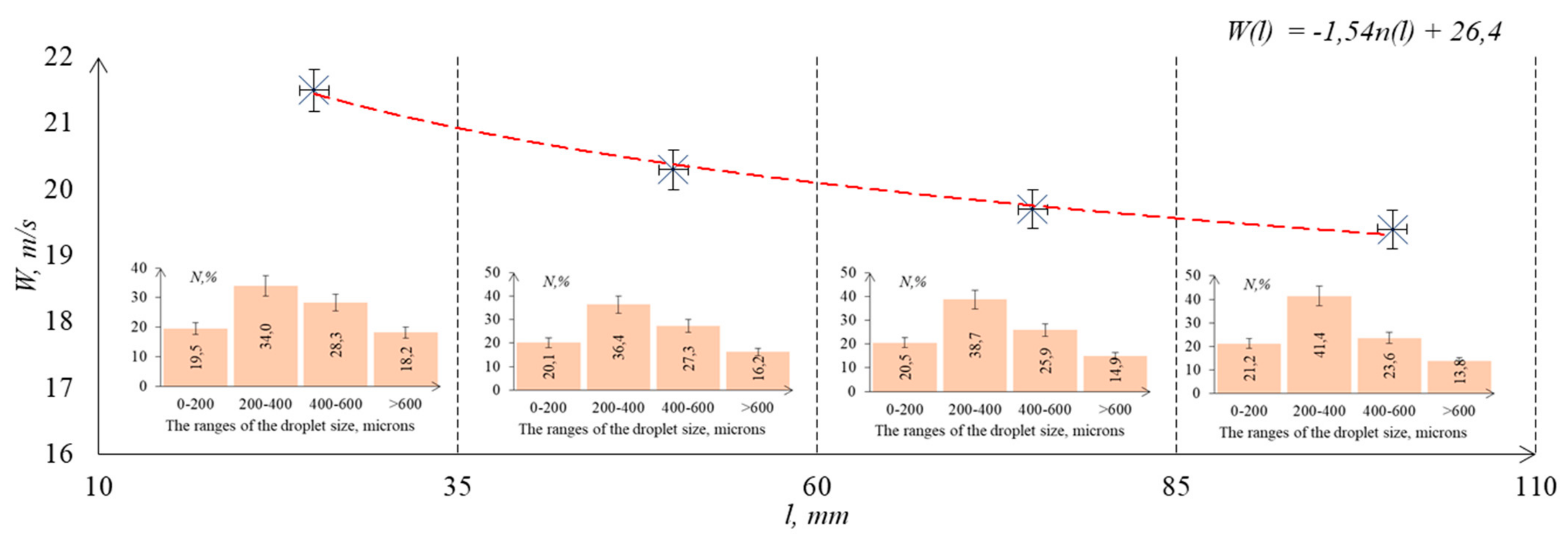
3.3.2. Spray
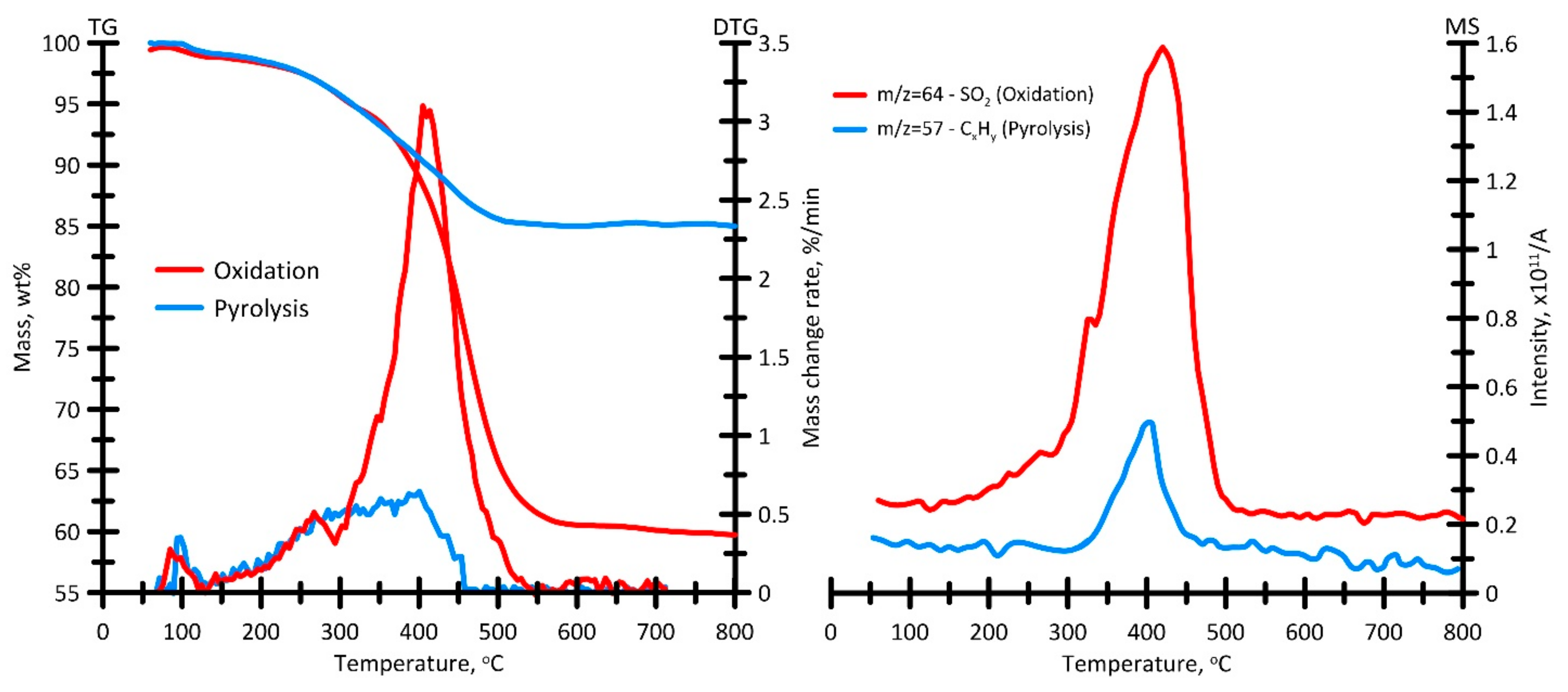
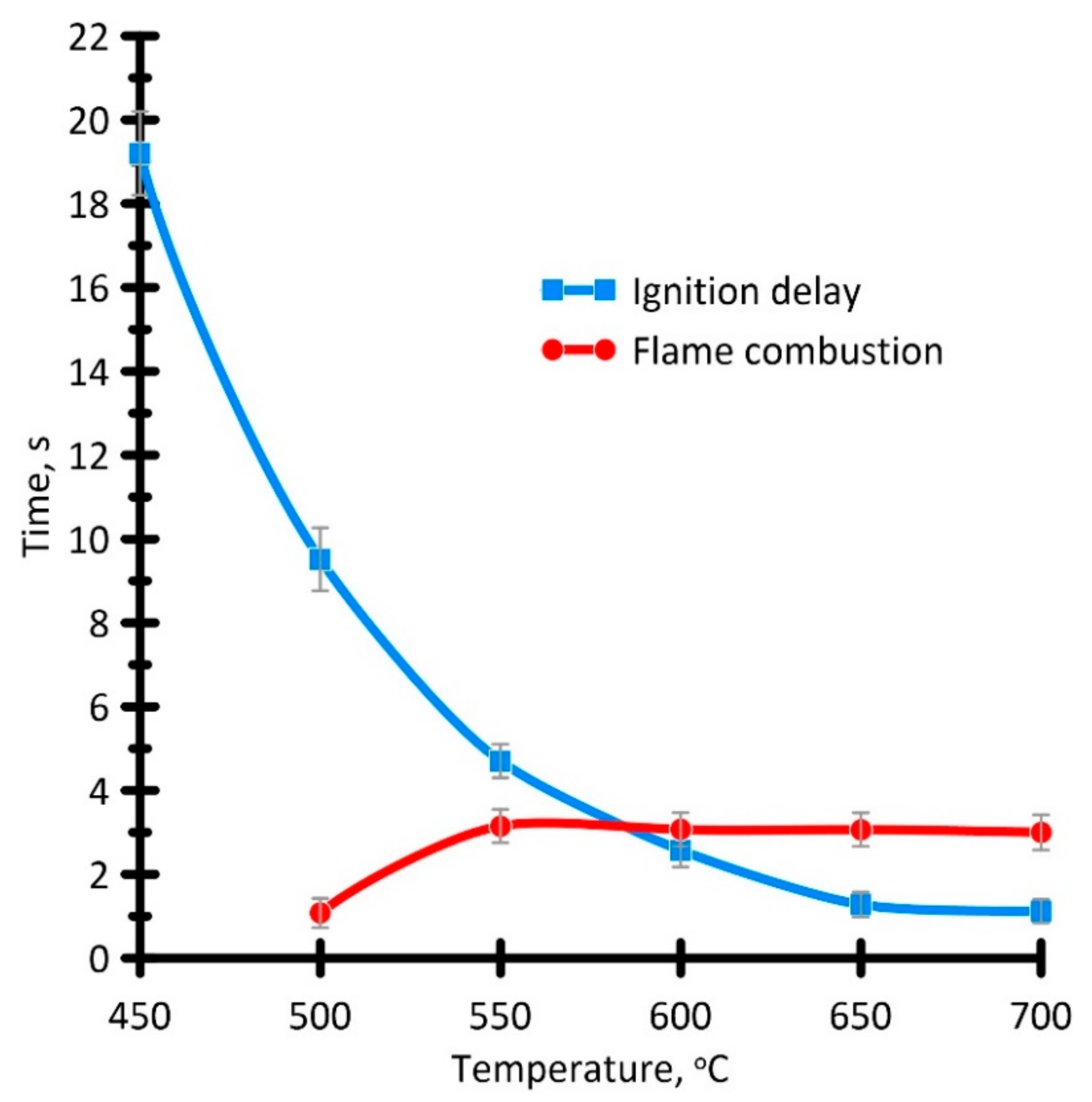
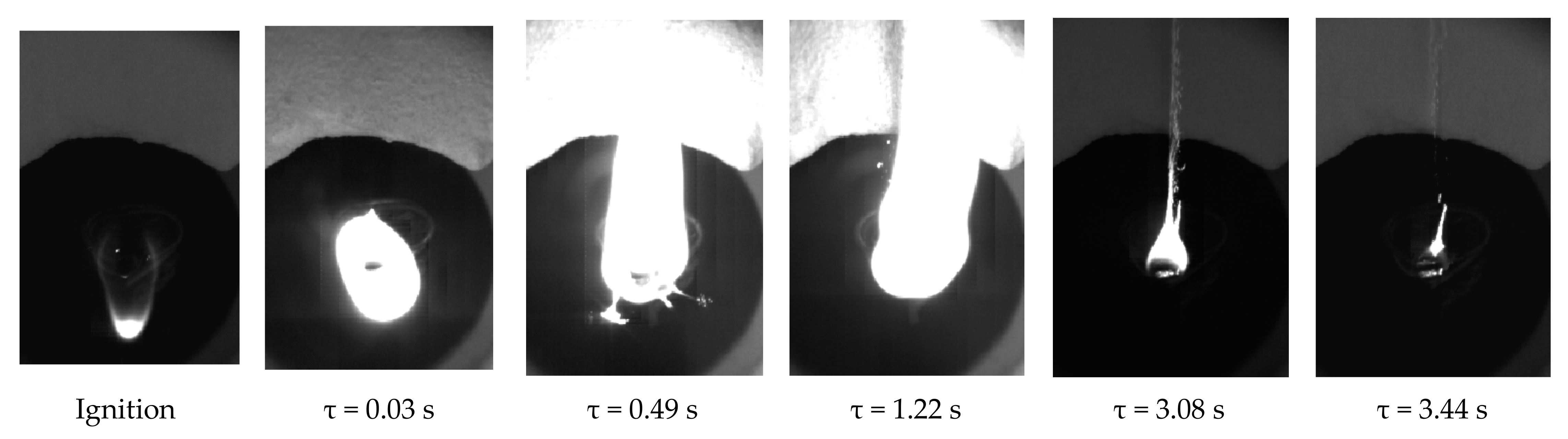
3.3.3. Ignition and Combustion
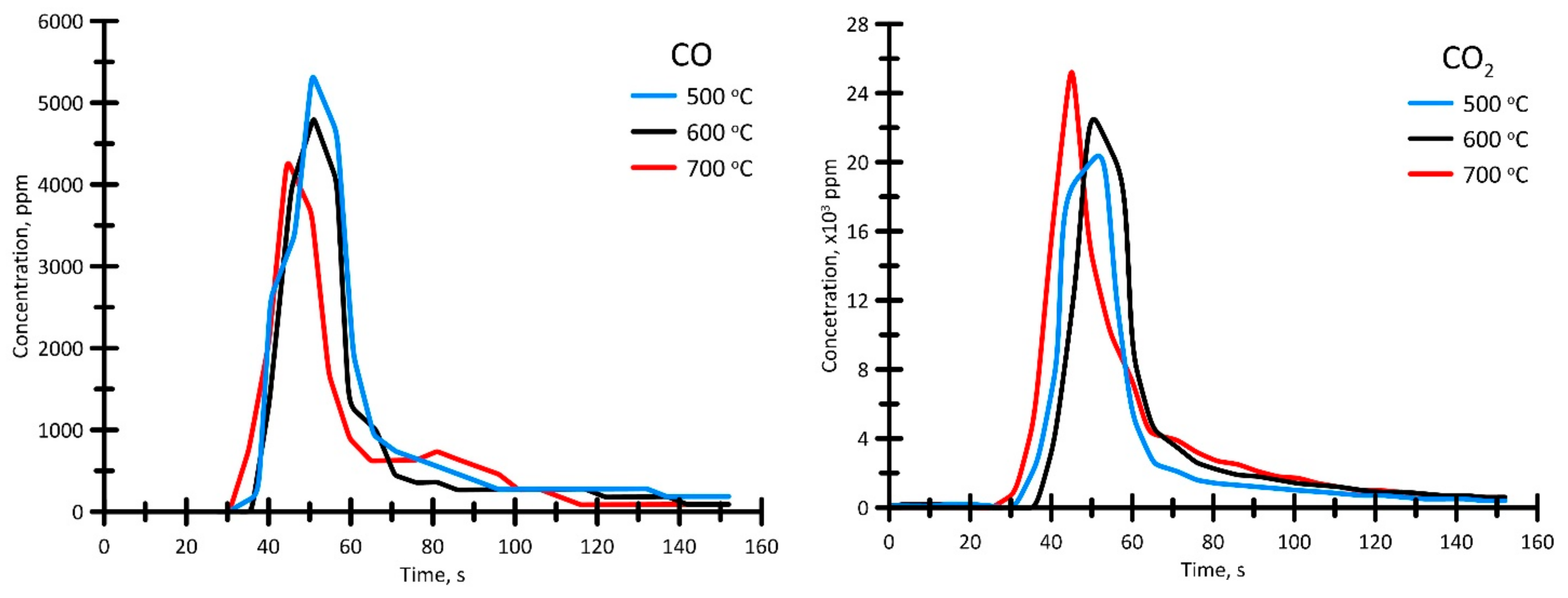
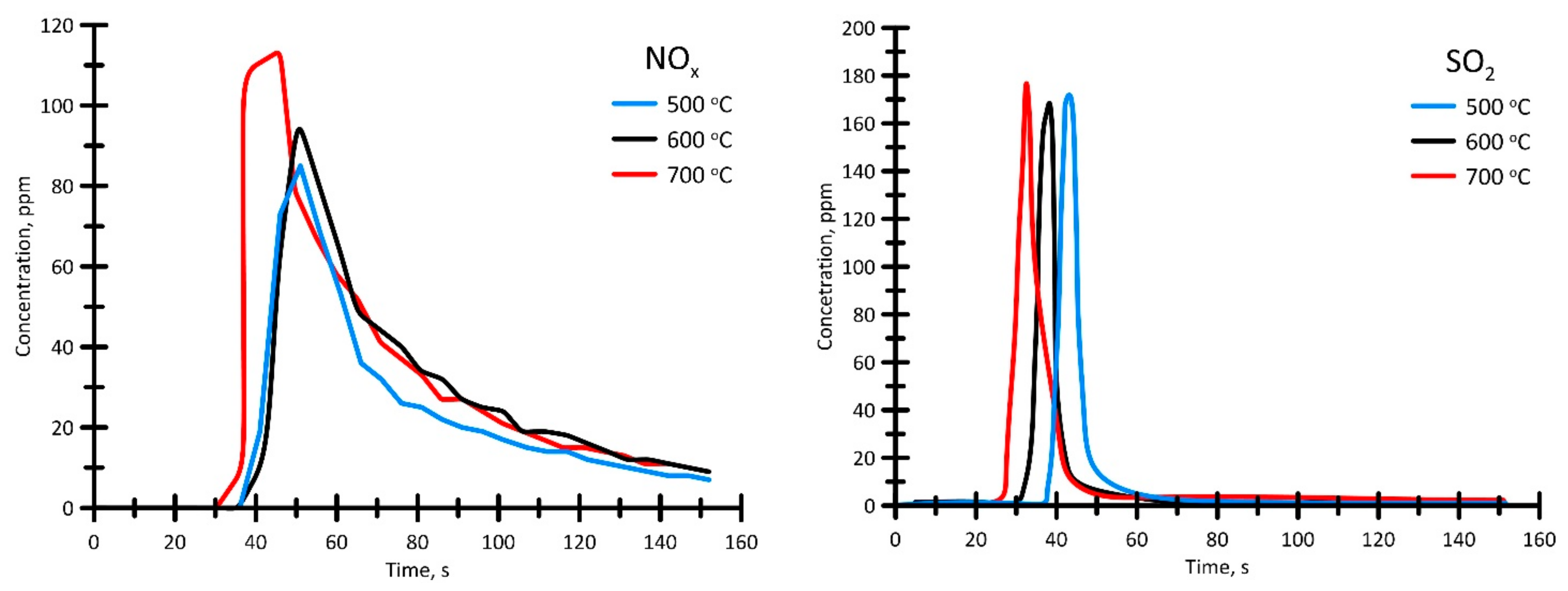
3.3.4. Analysis of Gas-Phase Combustion Products
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
Nomenclature
| OS | oil sludge |
| Tp | pour point |
| Tf | flash point |
| ti | ignition delay time |
| TG | thermogravimetry |
| DTG | derivative thermogravimetry |
| Pf | fuel pressure |
| Pa | air pressure |
| PC | personal computer |
| NCG | non-condensable gas-phase |
| Wa | humidity |
| Ad | ash content |
| Vdaf | volatile yield |
| LHV | lower heating value |
| tc | ash melting point |
| Ti | initial temperature |
| Te | final temperature |
| wmax | maximum reaction rate |
| Tmax | maximum temperature |
| MS | mass spectrometry |
| Tg | air temperature |
References
- IEA. Electricity. Available online: https://www.iea.org/fuels-and-technologies/electricity (accessed on 3 December 2021).
- Li, J.; Lin, F.; Xiang, L.; Zheng, F.; Che, L.; Tian, W.; Guo, X.; Yan, B.; Song, Y.; Chen, G. Hazardous elements flow during pyrolysis of oily sludge. J. Hazard. Mater. 2021, 409, 124986. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.; Li, Y.; Gan, Z.; Nie, M. Method of smoldering combustion for refinery oil sludge treatment. J. Hazard. Mater. 2021, 409, 124995. [Google Scholar] [CrossRef]
- Castillo Santiago, Y.; Martínez González, A.; Venturini, O.J.; Yepes Maya, D.M. Assessment of the energy recovery potential of oil sludge through gasification aiming electricity generation. Energy 2021, 215, 119210. [Google Scholar] [CrossRef]
- Chu, Z.; Gong, Z.; Wang, Z.; Zhang, H.; Wu, J.; Wang, Z.; Guo, Y.; Zhang, J.; Li, G.; Zhang, L.; et al. Experimental study on kinetic characteristics of oil sludge gasification. Asia-Pacific J. Chem. Eng. 2021, 16, e2616. [Google Scholar] [CrossRef]
- Wang, Z.; Gong, Z.; Wang, Z.; Li, X.; Chu, Z. Application and development of pyrolysis technology in petroleum oily sludge treatment. Environ. Eng. Res. 2021, 26, 1–15. [Google Scholar] [CrossRef]
- Shao, Z.; Ma, Y.; Wang, B.; He, L.; Zhang, Z.; Yue, C. Study of pyrolysis and combustion kinetics of oil sludge. Energy Sources Part A Recovery Util. Environ. Eff. 2021. [Google Scholar] [CrossRef]
- Mazlova, E.A.; Meshcheryakov, S.V. Ecological characteristics of oil sludges. Chem. Technol. Fuels Oils 1999, 35, 49–53. [Google Scholar] [CrossRef]
- Da Silva, L.J.; Alves, F.C.; De França, F.P. A review of the technological solutions for the treatment of oily sludges from petroleum refineries. Waste Manag. Res. 2012, 30, 1016–1030. [Google Scholar] [CrossRef]
- Zhou, L.; Jiang, X.; Liu, J. Characteristics of oily sludge combustion in circulating fluidized beds. J. Hazard. Mater. 2009, 170, 175–179. [Google Scholar] [CrossRef]
- Chen, G.-B.; Li, J.-W.; Lin, H.-T.; Wu, F.-H.; Chao, Y.-C. A Study of the Production and Combustion Characteristics of Pyrolytic Oil from Sewage Sludge Using the Taguchi Method. Energies 2018, 11, 2260. [Google Scholar] [CrossRef] [Green Version]
- Lin, B.; Huang, Q.; Ali, M.; Wang, F.; Chi, Y.; Yan, J. Continuous catalytic pyrolysis of oily sludge using U-shape reactor for producing saturates-enriched light oil. Proc. Combust. Inst. 2019, 37, 3101–3108. [Google Scholar] [CrossRef]
- Gong, Z.; Du, A.; Wang, Z.; Fang, P.; Li, X. Experimental Study on Pyrolysis Characteristics of Oil Sludge with a Tube Furnace Reactor. Energy Fuels 2017, 31, 8102–8108. [Google Scholar] [CrossRef]
- Milhé, M.; van de Steene, L.; Haube, M.; Commandré, J.-M.; Fassinou, W.-F.; Flamant, G. Autothermal and allothermal pyrolysis in a continuous fixed bed reactor. J. Anal. Appl. Pyrolysis 2013, 103, 102–111. [Google Scholar] [CrossRef]
- Kankia, M.U.; Baloo, L.; Danlami, N.; Samahani, W.N.; Mohammed, B.S.; Haruna, S.; Jagaba, A.H.; Abubakar, M.; Ishak, E.A.; Sayed, K.; et al. Optimization of Cement-Based Mortar Containing Oily Sludge Ash by Response Surface Methodology. Materials 2021, 14, 6308. [Google Scholar] [CrossRef] [PubMed]
- Kankia, M.U.; Baloo, L.; Mohammed, B.S.; Suhaimi, B.H.; Ishak, E.A.; Zango, U.Z. Review of petroleum sludge thermal treatment and utilization of ash as a construction material, a way to environmental sustainability. Int. J. Adv. Appl. Sci. 2020, 7, 68–81. [Google Scholar] [CrossRef]
- Moulin, L.; Da Silva, S.; Bounaceur, A.; Herbolt, M.; Soudais, Y. Assessment of Recovered Carbon Black Obtained by Waste Tires Steam Water Thermolysis: An Industrial Application. Waste Biomass Valor. 2017, 8, 2757–2770. [Google Scholar] [CrossRef] [Green Version]
- Li, Q.; Li, F.; Meng, A.; Tan, Z.; Zhang, Y. Thermolysis of scrap tire and rubber in sub/super-critical water. Waste Manag. 2018, 71, 311–319. [Google Scholar] [CrossRef] [PubMed]
- Larionov, K.B.; Slyusarskiy, K.V.; Kirgina, M.V.; Gvozdyakov, D.V.; Bogdanov, I.A.; Zenkov, A.V.; Yankovsky, S.A.; Gubin, V.E. Liquid Hydrocarbons Production by the Steam Pyrolysis of Used Tires: Energy Characteristics and Environmental Sustainability. Waste Biomass Valorization 2021. [Google Scholar] [CrossRef]
- Mei, Z.; Chen, D.; Zhang, J.; Yin, L.; Huang, Z.; Xin, Q. Sewage sludge pyrolysis coupled with self-supplied steam reforming for high quality syngas production and the influence of initial moisture content. Waste Manag. 2020, 106, 77–87. [Google Scholar] [CrossRef]
- Häring, H.-W. Hydrogen and Carbon Monoxide: Synthesis Gases. In Industrial Gases Processing; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2008; pp. 135–184. [Google Scholar]
- Avbenake, O.P.; Yakasai, F.I.; Jibril, B.Y. Sustainability Effect of Water Gas Shift Reaction (Syngas) in Catalytic Upgrading of Heavy Crude Oil and Bitumen. In Sustainable Alternative Syngas Fuel; IntechOpen Limited: London, UK, 2019. [Google Scholar]
- Chimpae, S.; Wongsakulphasatch, S.; Vivanpatarakij, S.; Glinrun, T.; Wiwatwongwana, F.; Maneeprakorn, W.; Assabumrungrat, S. Syngas Production from Combined Steam Gasification of Biochar and a Sorption-Enhanced Water–Gas Shift Reaction with the Utilization of CO2. Processes 2019, 7, 349. [Google Scholar] [CrossRef] [Green Version]
- Efika, C.E.; Wu, C.; Williams, P.T. Syngas production from pyrolysis–catalytic steam reforming of waste biomass in a continuous screw kiln reactor. J. Anal. Appl. Pyrolysis 2012, 95, 87–94. [Google Scholar] [CrossRef]
- Motlagh, A.H.; Klyuev, S.V.; Surendar, A.; Ibatova, A.Z.; Maseleno, A. Catalytic gasification of oil sludge with calcined dolomite. Pet. Sci. Technol. 2018, 36, 1998–2002. [Google Scholar] [CrossRef]
- Gao, N.; Kamran, K.; Quan, C.; Williams, P.T. Thermochemical conversion of sewage sludge: A critical review. Prog. Energy Combust. Sci. 2020, 79, 100843. [Google Scholar] [CrossRef]
- Anufriev, I.S.; Shadrin, E.Y.; Kopyev, E.P.; Alekseenko, S.V.; Sharypov, O.V. Study of liquid hydrocarbons atomization by supersonic air or steam jet. Appl. Therm. Eng. 2019, 163, 114400. [Google Scholar] [CrossRef]
- Volkov, R.S.; Zabelin, M.V.; Strizhak, P.A. On The Laws of Liquid Drop Deformation in Gas Flows. Chem. Pet. Eng. 2016, 52, 85–89. [Google Scholar] [CrossRef]
- Mahmoudi, A.H.; Pozarlik, A.K.; van der Weide, E.; Kersten, S.R.A.; Luding, S.; Brem, G. Effect of char on the combustion process of multicomponent bio-fuel. Chem. Eng. Sci. 2018, 175, 286–295. [Google Scholar] [CrossRef]
- Akinbomi, J.G.; Asifat, S.O.; Ajao, A.; Oladeji, O. Asphalt Making Potential of Pyrolytic Bitumen from Waste Rubber Tyres: An Adaptive Measure to Climate Change. In Handbook of Climate Change Resilience; Springer International Publishing: Berlin/Heidelberg, Germany, 2019; pp. 1–15. [Google Scholar]
- Gao, N.; Jia, X.; Gao, G.; Ma, Z.; Quan, C.; Naqvi, S.R. Modeling and simulation of coupled pyrolysis and gasification of oily sludge in a rotary kiln. Fuel 2020, 279, 118152. [Google Scholar] [CrossRef]
- Zhao, R.; Qin, J.; Chen, T.; Wang, L.; Wu, J. Experimental study on co-combustion of low rank coal semicoke and oil sludge by TG-FTIR. Waste Manag. 2020, 116, 91–99. [Google Scholar] [CrossRef] [PubMed]
- Upadhyay, D.S.; Khosla, A.; Chaudhary, A.; Patel, R.N. Effect of catalyst to lignite ratio on the performance of a pilot scale fixed bed gasifier. Energy 2019, 189, 116229. [Google Scholar] [CrossRef]
- Larionov, K.B.; Yankovsky, S.A.; Gubin, V.E.; Slyusarskiy, K.V.; Ulko, A.A.; Gasparian, G.D. Production of Briquetted Semicoke from Wood Waste by Multistep Low-Temperature Pyrolysis. Coke Chem. 2020, 63, 592–598. [Google Scholar] [CrossRef]
- Kuznetsov, G.V.; Yankovskii, S.A.; Tolokol’nikov, A.A.; Cherednik, I.V. Mechanism of the Suppression of Sulfur Oxides in the Oxidative Thermolysis Products of Coals upon Their Combustion in a Mixture with Dispersed Wood. Solid Fuel Chem. 2020, 54, 311–317. [Google Scholar] [CrossRef]
- Čepić, Z.; Mihajlović, V.; Đurić, S.; Milotić, M.; Stošić, M.; Stepanov, B.; Ilić Mićunović, M. Experimental Analysis of Temperature Influence on Waste Tire Pyrolysis. Energies 2021, 14, 5403. [Google Scholar] [CrossRef]
- Singh, R.K.; Ruj, B.; Sadhukhan, A.K.; Gupta, P.; Tigga, V.P. Waste plastic to pyrolytic oil and its utilization in CI engine: Performance analysis and combustion characteristics. Fuel 2020, 262, 116539. [Google Scholar] [CrossRef]
- Williams, P.T. Pyrolysis of waste tyres: A review. Waste Manag. 2013, 33, 1714–1728. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Osayi, J.I.; Iyuke, S.; Daramola, M.O.; Osifo, P.; Van Der Walt, I.J.; Ogbeide, S.E. Evaluation of pyrolytic oil from used tires and natural rubber (Hevea brasiliensis). Chem. Eng. Commun. 2018, 205, 805–821. [Google Scholar] [CrossRef]
- Larionov, K.B.; Slyusarskiy, K.V.; Tsibulskiy, S.A.; Kaltaev, A.Z.; Berezikov, N.I.; Gorshkov, A.S.; Lavrinenko, S.V.; Gubin, V.E. Activation of Anthracite Combustion Using Pyrolysis Oil from Thermal Conversion of Waste Car Tires. ACS Omega 2021, 6, 19731–19739. [Google Scholar] [CrossRef] [PubMed]
- Hou, S.-S.; Rizal, F.M.; Lin, T.-H.; Yang, T.-Y.; Wan, H.-P. Microexplosion and ignition of droplets of fuel oil/bio-oil (derived from lauan wood) blends. Fuel 2013, 113, 31–42. [Google Scholar] [CrossRef]
- Ocampo-Barrera, R.; Villasenor, R.; Diego-Marin, A. An experimental study of the effect of water content on combustion of heavy fuel oil/water emulsion droplets. Combust. Flame 2001, 126, 1845–1855. [Google Scholar] [CrossRef]
- Mikulski, M.; Ambrosewicz-Walacik, M.; Hunicz, J.; Nitkiewicz, S. Combustion engine applications of waste tyre pyrolytic oil. Prog. Energy Combust. Sci. 2021, 85, 100915. [Google Scholar] [CrossRef]
- Yaqoob, H.; Teoh, Y.H.; Jamil, M.A.; Gulzar, M. Potential of tire pyrolysis oil as an alternate fuel for diesel engines: A review. J. Energy Inst. 2021, 96, 205–221. [Google Scholar] [CrossRef]
- Erdogan, S. Recycling of Waste Plastics into Pyrolytic Fuels and Their Use in IC Engines. In Sustainable Mobility; IntechOpen: London, UK, 2020. [Google Scholar]
- Ramirez-Canon, A.; Muñoz-Camelo, Y.; Singh, P. Decomposition of Used Tyre Rubber by Pyrolysis: Enhancement of the Physical Properties of the Liquid Fraction Using a Hydrogen Stream. Environments 2018, 5, 72. [Google Scholar] [CrossRef] [Green Version]
- Tipler, S.; Mergulhão, C.S.; Vanhove, G.; Van Haute, Q.; Contino, F.; Coussement, A. Ignition Study of an Oxygenated and High-Alkene Light Petroleum Fraction Produced from Automotive Shredder Residues. Energy Fuels 2019, 33, 5664–5672. [Google Scholar] [CrossRef] [Green Version]
- Viswanathan, B. Petroleum. In Energy Sources; Elsevier: Amsterdam, The Netherlands, 2017; pp. 29–57. [Google Scholar]
- Shin, J.; Kim, D.; Seo, J.; Park, S. Effects of the physical properties of fuel on spray characteristics from a gas turbine nozzle. Energy 2020, 205, 118090. [Google Scholar] [CrossRef]
- Shlegel, N.E.; Tkachenko, P.P.; Strizhak, P.A. Influence of viscosity, surface and interfacial tensions on the liquid droplet collisions. Chem. Eng. Sci. 2020, 220, 115639. [Google Scholar] [CrossRef]
- Ju, D.; Sun, X.; Jia, X.; Huang, Z.; Qiao, X.; Han, D.; Huang, Z. Experimental investigation of the atomization behavior of ethanol and kerosene in acoustic fields. Fuel 2017, 202, 613–619. [Google Scholar] [CrossRef]
- Zhao, Y.; He, X.; Li, M.; Yao, K. Experimental investigation on spray characteristics of aircraft kerosene with an external-mixing atomizer. Fuel Process. Technol. 2020, 209, 106531. [Google Scholar] [CrossRef]
- Li, B.; Wang, Y.; Long, W.; Zhu, J.; Tian, J.; Cui, J.; Wang, Y. Experimental research on the effects of kerosene on the pre-injection spray characteristics and engine performance of dual-direct injection diesel Jet Controlled Compression Ignition mode. Fuel 2020, 281, 118691. [Google Scholar] [CrossRef]
- Kook, S.; Pickett, L.M. Liquid length and vapor penetration of conventional, Fischer–Tropsch, coal-derived, and surrogate fuel sprays at high-temperature and high-pressure ambient conditions. Fuel 2012, 93, 539–548. [Google Scholar] [CrossRef]
- Pinchuk, V.A.; Sharabura, T.A.; Kuzmin, A.V.; Pinchuk, S.A. Engineering equations for determining coal-water fuel combustion stages. J. Energy Inst. 2020, 93, 1924–1933. [Google Scholar] [CrossRef]
- Wang, C.H.; Liu, X.Q.; Law, C.K. Combustion and microexplosion of freely falling multicomponent droplets. Combust. Flame 1984, 56, 175–197. [Google Scholar] [CrossRef]


| Parameter | Value, mm |
|---|---|
| Nozzle outer diameter d1 | 20 |
| Fuel inlet channel diameter d2 | 8 |
| Air inlet channel diameter d3 | 4 |
| Height of the annular gap d4 | 2 |
| Nozzle outlet diameter d5 | 2.5 |
| Parameter | Value |
|---|---|
| Humidity Wa, wt% | 0.8 |
| Ash content Ad, wt% | 63.7 |
| Volatile yield Vdaf, wt% | 62.7 |
| Heating value LHV, MJ/kg | 14.6 |
| Elemental compositiond, wt% | |
| C | 25.3 |
| H | 2.0 |
| N | 0.3 |
| S | 1.7 |
| O | 1.0 |
| Elemental composition of the ash residued, % | |
| SiO2 | 63,4 |
| Al2O3 | 12.1 |
| Fe2O3 | 10.4 |
| SO3 | 2.4 |
| CaO | 4.7 |
| MgO | 2.9 |
| K2O + Na2O | 4.1 |
| Parameter | Value | ||||
|---|---|---|---|---|---|
| Test Sample | Fuel Oil [19] | Pyrolysis Oil from Tires at 500 °C (in Steam Environment) [19] | Classic Pyrolysis Oil from Tires at 500 °C [36] | Pyrolysis Oil from Plastic at 500 °C [37] | |
| Density, kg/m3 | |||||
| At 15 °C | 884.2 | 950.6 | 928.9 | 937 | 734 |
| At 20 °C | 880.7 | - | - | - | - |
| At 40 °C | 866.7 | - | - | - | - |
| Dynamic viscosity, mPa·s | |||||
| At 15 °C | 13.6 | - | - | - | - |
| At 20 °C | 10.1 | - | - | - | - |
| At 40 °C | 4.9 | 545.7 | 9.7 | - | - |
| Kinematic viscosity, mm2/s | |||||
| At 15 °C | 15.4 | - | - | - | - |
| At 20 °C | 11.5 | - | - | - | - |
| At 40 °C | 5.6 | 583.7 | 10.6 | 4.7 | 2.9 |
| Pour point Tp, °C | 5 | 10 | −52 | −11 | −3 |
| Flash point Tf, °C | 89 | 175 | 82 | 31 | 46 |
| Water content, % | 0.2 | 0.2 | 0.7 | - | - |
| Ash content, % | traces | traces | traces | - | <1.0 |
| pH | 6.8 | 6.8 | 6.0 | - | - |
| Surface tension, mN/m | 31.2 | - | - | - | - |
| Lower heating value LHV, MJ/kg | 42.6 | 43.9 | 43.3 | 42.4 | 41.3 |
| Fractional composition | |||||
| 10 vol.% | 41 °C | - | - | - | - |
| 20 vol.% | 125 °C | - | - | - | - |
| 30 vol.% | 188 °C | - | - | - | - |
| 40 vol.% | 243 °C | - | - | - | - |
| 50 vol.% | 307 °C | - | - | - | - |
| 60 vol.% | 336 °C | - | - | - | - |
| 70 vol.% | 360 °C | - | - | - | - |
| Elemental composition, wt.% | |||||
| C | 83.4 | 87.8 | 86.2 | 85.4 | - |
| H | 11.4 | 9.9 | 11.0 | 10.1 | - |
| N | - | 0.2 | 0.6 | 1.2 | - |
| S | 0.8 | 1.8 | 0.8 | 0.5 | - |
| O | 4.4 | 0.3 | 1.4 | 2.8 | - |
| H/C | 1.64 | 1.4 | 1.5 | 1.4 | - |
| O/C | 0.04 | - | 0.01 | 0.02 | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Larionov, K.; Kaltaev, A.; Slyusarsky, K.; Gvozdyakov, D.; Zenkov, A.; Kirgina, M.; Bogdanov, I.; Gubin, V. Steam Pyrolysis of Oil Sludge for Energy-Valuable Products. Appl. Sci. 2022, 12, 1012. https://doi.org/10.3390/app12031012
Larionov K, Kaltaev A, Slyusarsky K, Gvozdyakov D, Zenkov A, Kirgina M, Bogdanov I, Gubin V. Steam Pyrolysis of Oil Sludge for Energy-Valuable Products. Applied Sciences. 2022; 12(3):1012. https://doi.org/10.3390/app12031012
Chicago/Turabian StyleLarionov, Kirill, Albert Kaltaev, Konstantin Slyusarsky, Dmitriy Gvozdyakov, Andrey Zenkov, Maria Kirgina, Ilya Bogdanov, and Vladimir Gubin. 2022. "Steam Pyrolysis of Oil Sludge for Energy-Valuable Products" Applied Sciences 12, no. 3: 1012. https://doi.org/10.3390/app12031012
APA StyleLarionov, K., Kaltaev, A., Slyusarsky, K., Gvozdyakov, D., Zenkov, A., Kirgina, M., Bogdanov, I., & Gubin, V. (2022). Steam Pyrolysis of Oil Sludge for Energy-Valuable Products. Applied Sciences, 12(3), 1012. https://doi.org/10.3390/app12031012






