From Neuroimaging to Computational Modeling of Burnout: The Traditional versus the Fuzzy Approach—A Review
Abstract
Featured Application
Abstract
1. Introduction
1.1. Types of Neuroimaging Used to Study Brain Structure and Activity
1.1.1. Electroencephalogram
1.1.2. Positron Emission Tomography
1.1.3. Magnetic Resonance Imaging and Functional Magnetic Resonance Imaging
1.2. Types of Computational Systems and Models Used to Study Brain Structures and Activity
- Data analysis, including biomedical signals (from simple sequences of laboratory data to EEG analysis, still and moving images, gait, or speech analysis as a biomedical signal);
- Pattern recognition and classification, and machine learning (ML), including traditional and deep artificial neural networks (ANN), support vector machine (SVM), naïveBayesian classifier, kNN, regressions, decision trees, and random forests, etc.;
- Swarm intelligence, including ant colony optimization (ACO) and particle swarm optimization (PSO);
- Fuzzy systems, including ordered fuzzy numbers and trend analysis;
- Multidimensional scaling (MDS) and similar tools for attractor analysis;
- Multifractal analysis, including the analysis of the trend change, e.g., based on changes in the Hurst index;
- Artificial intelligence optimization of the production processes of medical devices (prostheses, orthoses, exoskeletons, implants, drugs, etc.) and therapeutic processes (e.g., optimization of rehabilitation robot algorithms);
- Second opinion systems;
- Predictive systems;
- Modeling of central nervous system processes on biologically faithful neural networks (Emergent, Genesis, NEURON);
- Modeling the processes of the peripheral nervous system based on the patterns of the signal theory;
- Dedicated data science systems in Matlab, R (Python), or other data analysis environments for predictive medicine purposes.
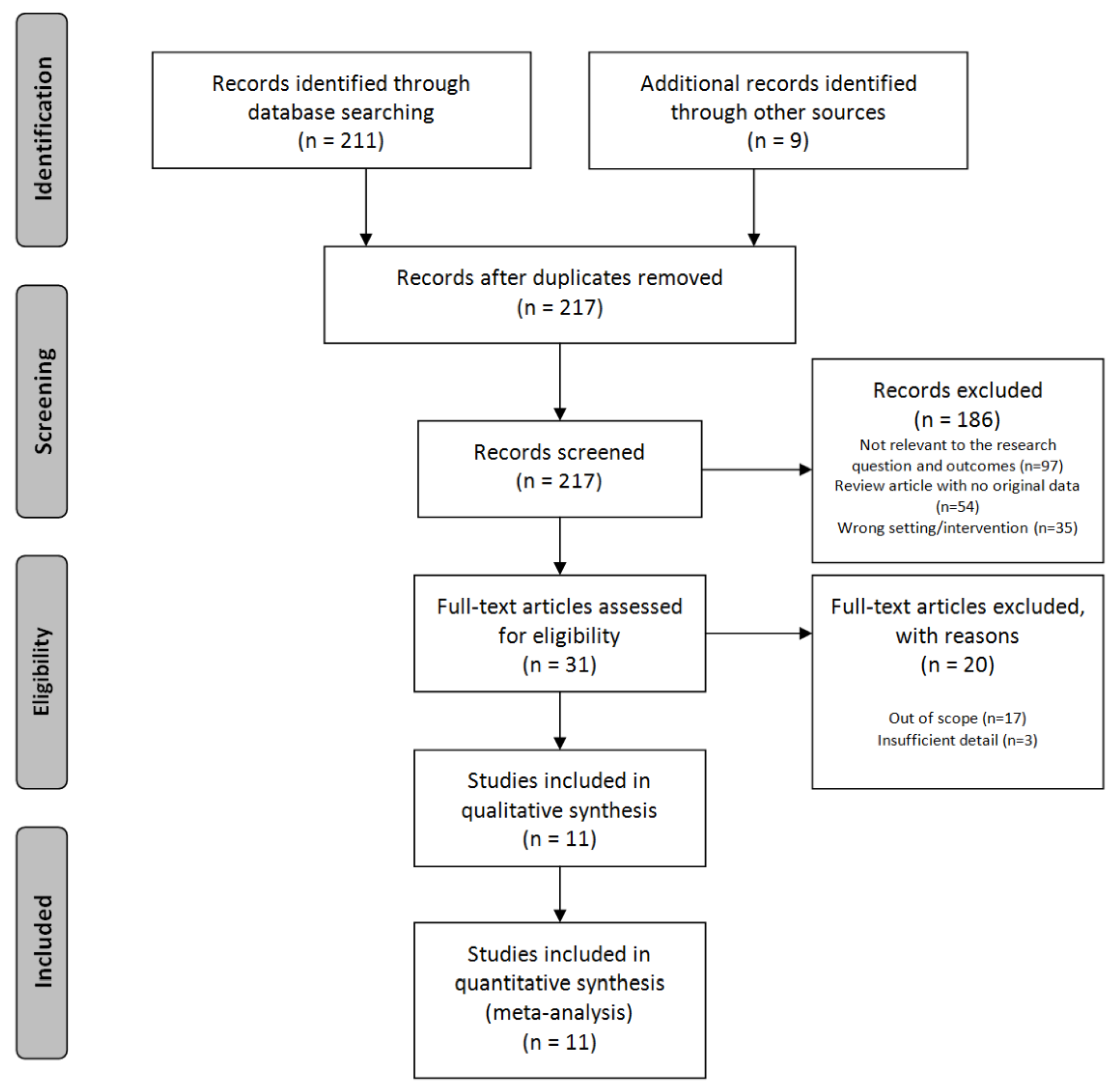
2. Materials and Methods
2.1. Literature Review on Neuroimaging in Burnout
- “work stress”= “occupational stress”;
- “professional burnout syndrome”= “professional burnout”;
- “mental stress” = “stress” = “stress exhaustion syndrome.
2.2. Concept of Computational Model of Burnout
3. Results of the Literature Review
3.1. EEG Studies
- Frontal asymmetry;
- P300 amplitude;
- Alpha peak frequency;
- Alpha and beta power comparison with eyes open and closed.
- Beta potency was significantly reduced in burnout patients compared to the control group;
- There was no difference in alpha potency compared to the control group.
3.2. MRI and fMRI Studies
- In the study group: N = 17 (15 men and 2 women);
- In the control group: N = 17 (10 men and 7 women) [21];
- compassion fatigue—that is, individuals become overly emotionally involved;
- emotional dissonance—that is, a discrepancy between felt and expressed emotions along with decreased emotional regulation.
- severity of burnout was assessed using the MBI;
3.3. PET Studies
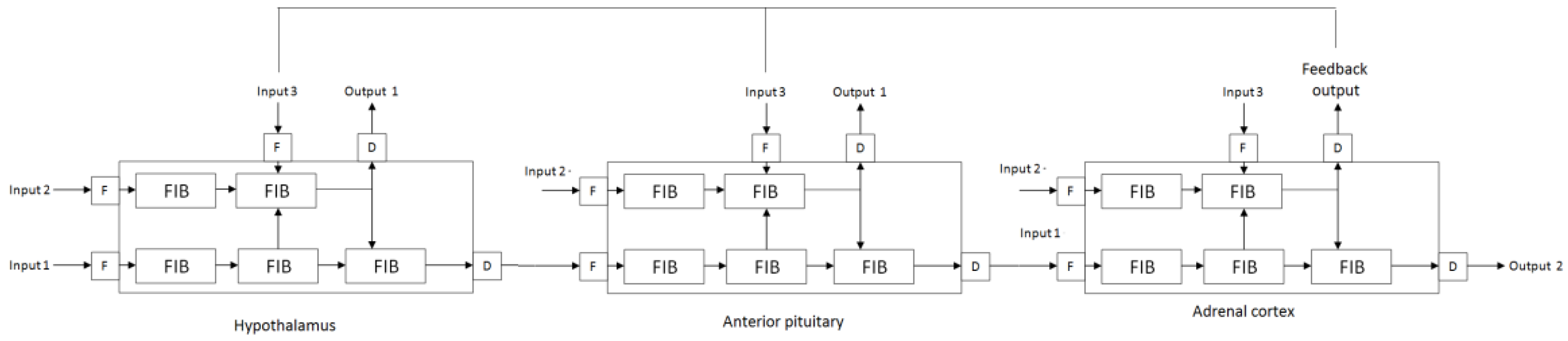
4. Computational Models of Burnout
4.1. Fuzziness of Burnout
4.2. Current Fuzzy Models of Burnout
4.3. Further Computational Studies
5. Discussion
- corticotropin-releasing hormone (CRH);
- adrenocorticotropic hormone (ACTH);
- cortisol;
- prolactin;
- thyroid hormones;
- The influence of the immune system on stress sensitivity [74];
- The role of norepinephrine and serotonin [3];
- The role of selected neuropeptides in the inhibitory mechanism of the impact of stress [75];
- The role of the hypothalamic–pituitary–adrenal axis in influencing stress susceptibility and resistance to stress [76];
- The role of epigenetic modulation of gene expression in influencing stress susceptibility [77];
- Neurobiological mechanisms by which factors such as diet, exercise, and peer support influence stress resistance [78].
5.1. Directions for Future Research
5.2. Limitations of Own Studies
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Luijtelaar, G.V.; Verbraak, M.; Bunt, M.V.; Keijsers, G.; Arns, M. EEG findings in burnout patients. J. Neuropsychiatry Clin. Neurosci. 2010, 22, 208–217. [Google Scholar] [CrossRef] [PubMed]
- Plieger, T.; Melchers, M.; Montag, C.; Meermann, R.; Reuter, M. Life stress as potential risk factor for depression and burnout. Burn. Res. 2015, 2, 19–24. [Google Scholar] [CrossRef]
- Arnsten, A.F.T.; Shanafelt, T. Physician distress and burnout: The neurobiological perspective. Mayo Clin. Proc. 2021, 96, 763–769. [Google Scholar] [CrossRef]
- American Medical Association. International Classification of Diseases. In Clinical Modification(ICD-10-CM) 2022: The Complete Official Codebook with Guidelines, 10th ed.; American Medical Association: Chicago, IL, USA, 2021. [Google Scholar]
- World Health Organization. International Classification of Diseases, 11th ed.; World Health Organization: Geneva, Switzerland, 2022. [Google Scholar]
- American Psychiatric Association. The Diagnostic and Statistical Manual of Mental Disorders, 5th ed.; DSM-5; American Psychiatric Association: Washington, DC, USA, 2013. [Google Scholar]
- Al-Haddad, A.; Al-Omar, F.; Al-Khaleel, A.; Al-Khalaf, A. Prevalence of burnout syndrome and its related risk factors among physicians working in primary health care centers of the Ministry of Health, Al Ahsa Region, Saudi Arabia, 2018–2019. J. Fam. Med. Prim. Care 2020, 9, 571–579. [Google Scholar] [CrossRef] [PubMed]
- Messias, E.; Gathright, M.M.; Freeman, E.S.; Flynn, V.; Atkinson, T.; Thrush, C.R.; Clardy, J.A.; Thapa, P. Differences in burnout prevalence between clinical professionals and biomedical scientists in an academic medical centre: A cross-sectional survey. BMJ Open 2019, 9, e023506. [Google Scholar] [CrossRef] [PubMed]
- Rotenstein, L.S.; Torre, M.; Ramos, M.A.; Rosales, R.C.; Guille, C.; Sen, S.; Mata, D.A. Prevalence of burnout among physicians: A systematic review. JAMA 2018, 320, 1131–1150. [Google Scholar] [CrossRef]
- Roberts, B.L.; Karatsoreos, I.N. Brain-body responses to chronic stress: A brief review. Fac. Rev. 2021, 10, 83. [Google Scholar] [CrossRef]
- Markram, H. Seven challenges for neuroscience. Funct. Neurol. 2013, 28, 145–151. [Google Scholar] [CrossRef]
- Frégnac, Y. How blue is the sky? eNeuro 2021, 8, ENEURO.0130-21.2021. [Google Scholar] [CrossRef]
- Boegl, K.; Adlassnig, K.-P.; Hayashi, Y.; Rothenfluh, T.E.; Leitich, H. Knowledge acquisition in the fuzzy knowledge representation framework of a medical consultation system. Artif. Intell. Med. 2004, 30, 1–26. [Google Scholar] [CrossRef]
- Torres, A.; Nineto, J.J. Fuzzy logic in medicine and bioinformatics. BioMed Res. Int. 2006, 2006, 091908. [Google Scholar] [CrossRef]
- Buckley, J.J.; Feuring, T.; Hayashi, Y. Fuzzy hierarchical analysis revisited. Eur. J. Oper. Res. 2001, 121, 48–64. [Google Scholar] [CrossRef]
- Staszecka, E. Combining uncertainty and imprecision in models of medical diagnosis. Inf. Sci. 2006, 176, 3026–3059. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. The PRISMA Group preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef]
- Tement, S.; Pahor, A.; Jaušovec, N. EEG alpha frequency correlates of burnout and depression: The role of gender. Biol. Psychol. 2016, 114, 1–12. [Google Scholar] [CrossRef]
- Golonka, K.; Gawlowska, M.; Mojsa-Kaja, J.; Marek, T. Psychophysiological characteristics of burnout syndrome: Resting-state EEG analysis. Biomed. Res. Int. 2019, 2019, 3764354. [Google Scholar] [CrossRef] [PubMed]
- Yakovenko, E.A.; Rem, A.V.; Surushkina, S.Y.; Chutko, L.S. Electroencephalographic signs of emotional burnout syndrome. Neurosci.Behav. Phys. 2021, 51, 155–157. [Google Scholar] [CrossRef]
- Durning, S.J.; Costanzo, M.; Artino, A.R.; Dyrbye, L.N.; Beckman, T.J.; Schuwirth, L.; Holmboe, E.; Roy, M.J.; Wittich, C.M.; Lipner, R.S.; et al. Functional neuroimaging correlates of burnout among internal medicine residents and faculty members. Front. Psychiatry 2013, 4, 131. [Google Scholar] [CrossRef] [PubMed]
- Savic, I. Structural changes of the brain in relation to occupational stress. Cereb Cortex 2013, 25, 1554–1564. [Google Scholar] [CrossRef] [PubMed]
- Blix, E.; Perski, A.; Berglund, H.; Savic, I. Long-term occupational stress is associated with regional reductions in brain tissue volumes. PLoS ONE 2013, 8, e64065. [Google Scholar] [CrossRef]
- Tei, S.; Becker, C.; Kawada, R.; Fujino, J.; Jankowski, K.F.; Sugihara, G.; Murai, T.; Takahashi, H. Can we predict burnout severity from empathy-related brain activity? Transl. Psychiatry 2014, 4, e393. [Google Scholar] [CrossRef]
- Savic, I.; Perski, A.; Osika, W. MRI shows that exhaustion syndrome due to chronic occupational stress is associated with partially reversible cerebral changes. Cereb Cortex 2018, 28, 894–906. [Google Scholar] [CrossRef]
- Gavelin, H.M.; Neely, A.S.; Andersson, M.; Eskilsson, T.; Järvholm, L.S.; Boraxbekk, C.J. Neural activation in stress-related exhaustion: Cross-sectional observations and interventional effects. Psychiatry Res. Neuroimaging 2017, 269, 17–25. [Google Scholar] [CrossRef] [PubMed]
- Jovanovic, H.; Perski, A.; Berglund, H.; Savic, I. Chronic stress is linked to 5-HT1A receptor changes and functional disintegration of the limbic networks. NeuroImage 2011, 55, 1178–1188. [Google Scholar] [CrossRef] [PubMed]
- Maslach, C.; Jackson, S.E. The measurement of experienced burnout. J. Occup. Behav. 1981, 2, 99–113. [Google Scholar] [CrossRef]
- Maslach, C.; Leiter, M.P.; Jackson, S.E. Making a significant difference with burnout interventions: Researcher and practitioner collaboration. J. Organ. Behav. 2012, 33, 296–300. [Google Scholar] [CrossRef]
- Davis, M.H. A multidimensional approach to individual differences in empathy. JSAS Cat. Sel. Doc. Psychol. 1980, 10, 85. [Google Scholar]
- Lundgren-Nilsson, Å.; Jonsdottir, I.H.; Pallant, J. Internal construct validity of the Shirom-Melamed Burnout Questionnaire (SMBQ). BMC Public Health 2012, 12, 1. [Google Scholar] [CrossRef]
- Kirk-Sanchez, N.J.; McGough, E.L. Physical exercise and cognitive performance in the elderly: Current perspectives. Clin. Interv. Aging 2014, 9, 51–62. [Google Scholar] [CrossRef]
- Bherer, L. Cognitive plasticity in older adults: Effects of cognitive training and physical exercise. Ann. N. Y. Acad. Sci. 2015, 1337, 1–6. [Google Scholar] [CrossRef]
- Li, B.Y.; Tang, H.D.; Qiao, Y.; Chen, S.D. Mental training for cognitive improvement in elderly people: What have we learned from clinical and neurophysiologic studies? Curr. Alzheimer Res. 2015, 12, 543–552. [Google Scholar] [CrossRef] [PubMed]
- Dutra, H.S.; Guirardello, E.B.; Li, Y.; Cimiotti, J.P. Nurse burnout revisited: A comparison of computational methods. J. Nurs. Meas. 2019, 27, E17–E33. [Google Scholar] [CrossRef] [PubMed]
- Lou, S.S.; Liu, H.; Warner, B.C.; Harford, D.; Lu, C.; Kannampallil, T. Predicting physician burnout using clinical activity logs: Model performance and lessons learned. J. Biomed. Inform. 2022, 127, 104015. [Google Scholar] [CrossRef] [PubMed]
- Maija, K.; Katri, K. The moral orders of work and health: A case of sick leave due to burnout. Sociol. Health Illn. 2019, 41, 219–233. [Google Scholar] [CrossRef] [PubMed]
- Dong, J.; Yan, S. A multicriteria approach for measuring employee well-being. Front. Psychol. 2022, 13, 795960. [Google Scholar] [CrossRef] [PubMed]
- MacCormack, J.K.; Stein, A.G.; Kang, J.; Giovanello, K.S.; Satpute, A.B.; Lindquist, K.A. Affect in the aging brain: A neuroimaging meta-analysis of older vs. younger adult affective experience and perception. Affect. Sci. 2020, 1, 128–154. [Google Scholar] [CrossRef]
- Prokopowicz, P.; Mikołajewski, D. Fuzzy approach to computational classification of burnout—Preliminary findings. Appl. Sci. 2022, 12, 3767. [Google Scholar] [CrossRef]
- Mikołajewski, D.; Prokopowicz, P. Effect of COVID-19 on selected characteristics of life satisfaction reflected in a fuzzy model. Appl. Sci. 2022, 12, 7376. [Google Scholar] [CrossRef]
- Martínez-Ramón, J.P.; Morales-Rodríguez, F.M.; Pérez-López, S. Burnout, resilience, and COVID-19 among teachers: Predictive capacity of an artificial neural network. Appl. Sci. 2021, 11, 8206. [Google Scholar] [CrossRef]
- Gavelin, H.M.; Neely, A.S.; Dunås, T.; Eskilsson, T.; Järvholm, L.S.; Boraxbekk, C.J. Mental fatigue in stress-related exhaustion disorder: Structural brain correlates, clinical characteristics and relations with cognitive functioning. Neuroimage Clin. 2020, 27, 102337. [Google Scholar] [CrossRef]
- Prokopowicz, P.; Mikołajewski, D. Fuzzy-based computational simulations of brain functions—Preliminary concept. Bio-Algorithms Med. Syst. 2016, 12, 99–104. [Google Scholar] [CrossRef]
- Bellucci, G.; Camilleri, J.A.; Eickhoff, S.B.; Krueger, F. Neural signatures of prosocial behaviors. Neurosci. Biobehav. Rev. 2020, 118, 186–195. [Google Scholar] [CrossRef] [PubMed]
- Mercer Lindsay, N.; Chen, C.; Gilam, G.; Mackey, S.; Scherrer, G. Brain circuits for pain and its treatment. Sci. Transl. Med. 2021, 13, eabj7360. [Google Scholar] [CrossRef] [PubMed]
- De Hert, S. Burnout in healthcare workers: Prevalence, impact and preventative strategies. Local Reg. Anesthesia 2020, 13, 171–183. [Google Scholar] [CrossRef] [PubMed]
- Weisbaum, E.; Chadi, N. Applied mindfulness for physician wellbeing: A prospective qualitative study protocol. Front. Public Health 2022, 10, 807792. [Google Scholar] [CrossRef]
- Laughey, W.F.; Atkinson, J.; Craig, A.M.; Douglas, L.; Brown, M.E.; Scott, J.L.; Alberti, H.; Finn, G.M. Empathy in medical education: Its nature and nurture—A qualitative study of the views of students and tutors. Med. Sci. Educ. 2021, 31, 1941–1950. [Google Scholar] [CrossRef]
- Paiva-Salisbury, M.L.; Schwanz, K.A. Building compassion fatigue resilience: Awareness, prevention, and intervention for pre-professionals and current practitioners. J. Health Serv. Psychol. 2022, 3, 39–46. [Google Scholar] [CrossRef]
- Lu, L.; Ko, Y.M.; Chen, H.Y.; Chueh, J.W.; Chen, P.Y.; Cooper, C.L. Patient safety and staff well-being: Organizational culture as a resource. Int. J. Environ. Res. Public Health 2022, 19, 3722. [Google Scholar] [CrossRef]
- Krebs, C.; Akesson, E.J.; Weinberg, J. Lippincott’s Illustrated Review of Neuroscience; Wolters Kluwer/Lippincott Williams & Wilkins Health: Amsterdam, The Netherlands, 2012. [Google Scholar]
- De Koninck, B.P.; Guay, S.; Blais, H.; De Beaumont, L. Parametric study of transcranial alternating current stimulation for brain alpha power modulation. Brain Commun. 2021, 3, fcab010. [Google Scholar] [CrossRef]
- Rosenzopf, H.; Wiesen, D.; Basilakos, A.; Yourganov, G.; Bonilha, L.; Rorden, C.; Fridriksson, J.; Karnath, H.O.; Sperber, C. Mapping the human praxis network: An investigation of white matter disconnection in limb apraxia of gesture production. Brain Commun. 2022, 4, fcac004. [Google Scholar] [CrossRef]
- Kelly, R.J.; Hearld, L.R. Burnout and leadership style in behavioral health care: A literature review. J. Behav. Health Serv. Res. 2020, 47, 581–600. [Google Scholar] [CrossRef] [PubMed]
- Lamm, C.; Majdandžić, J. The role of shared neural activations, mirror neurons, and morality in empathy—A critical comment. Neurosci. Res. 2015, 90, 15–24. [Google Scholar] [CrossRef] [PubMed]
- Nishitani, N.; Nagamine, T.; Fujiwara, N.; Yazawa, S.; Shibasaki, H. Cortical-hippocampal auditory processing identified by magnetoencephalography. J. Cogn. Neurosci. 1998, 10, 231–247. [Google Scholar] [CrossRef]
- Montoro, C.I.; Winterholler, C.; Terrasa, J.L.; Montoya, P. Somatosensory gating is modulated by anodal transcranial direct current stimulation. Front. Neurosci. 2021, 15, 651253. [Google Scholar] [CrossRef] [PubMed]
- Rosenblum, Y.; Shiner, T.; Bregman, N.; Fahoum, F.; Giladi, N.; Maidan, I.; Mirelman, A. Event-related oscillations differentiate between cognitive, motor and visual impairments. J. Neurol. 2022, 269, 3529–3540. [Google Scholar] [CrossRef] [PubMed]
- Duma, G.M.; Di Bono, M.G.; Mento, G. Grounding adaptive cognitive control in the intrinsic, functional brain organization: An HD-EEG resting state investigation. Brain Sci. 2021, 11, 1513. [Google Scholar] [CrossRef]
- Cannard, C.; Wahbeh, H.; Delorme, A. Electroencephalography correlates of well-being using a low-cost wearable system. Front. Hum. Neurosci. 2021, 15, 745135. [Google Scholar] [CrossRef]
- Millard, S.K.; Bokelmann, K.; Schalbroeck, R.; van der Wee, N.J.A.; van Loey, N.E.E.; van Laarhoven, A.I.M. No indications for altered EEG oscillatory activity in patients with chronic post-burn itch compared to healthy controls. Sci. Rep. 2022, 12, 5184. [Google Scholar] [CrossRef]
- Maksoud, R.; Eaton-Fitch, N.; Matula, M.; Cabanas, H.; Staines, D.; Marshall-Gradisnik, S. Systematic review of sleep characteristics in myalgic encephalomyelitis/chronic fatigue syndrome. Healthcare 2021, 9, 568. [Google Scholar] [CrossRef]
- Hong, Q.N.; Coutu, M.F.; Berbiche, D. Evaluating the validity of the Work Role Functioning Questionnaire (Canadian French version) using classical test theory and item response theory. Work 2017, 57, 501–515. [Google Scholar] [CrossRef]
- Guo, F.; Wang, T.; Ning, Z. Subjective measures of work-related fatigue in automobile factory employees. Work 2017, 58, 233–240. [Google Scholar] [CrossRef] [PubMed]
- Norstedt, M. The (im)possibilities of returning to work after a stroke. Work 2017, 56, 637–647. [Google Scholar] [CrossRef] [PubMed]
- Lexell, E.M.; Langdell, I.; Lexell, J. Vocational situation and experiences from the work environment among individuals with neuromuscular diseases. Work 2017, 56, 519–530. [Google Scholar] [CrossRef]
- Golkar, A.; Johansson, E.; Kasahara, M.; Osika, W.; Perski, A.; Savic, I. The influence of work-related chronic stress on the regulation of emotion and on functional connectivity in the brain. PLoS ONE 2014, 9, e104550. [Google Scholar] [CrossRef] [PubMed]
- Ciobanu, A.M.; Damian, A.C.; Neagu, C. Association between burnout and immunological and endocrine alterations. ROM J. Morphol. Embryol. 2021, 62, 13–18. [Google Scholar] [CrossRef]
- Buselli, R.; Veltri, A.; Baldanzi, S.; Marino, R.; Bonotti, A.; Chiumiento, M.; Girardi, M.; Pellegrini, L.; Guglielmi, G.; Dell’Osso, L.; et al. Plasma Brain-Derived Neurotrophic Factor (BDNF) and serum cortisol levels in a sample of workers exposed to occupational stress and suffering from adjustment disorders. Brain Behav. 2019, 9, e01298. [Google Scholar] [CrossRef]
- de Beer, L.T. Is there utility in specifying professional efficacy as an outcome of burnout in the employee health impairment process. Int. J. Environ. Res. Public Health 2021, 18, 6255. [Google Scholar] [CrossRef]
- Hoogduin, C.A.L.; Schaap, C.P.D.R.; Methorst, G.J. Burnout: Klinisch Beeld en Diagnostiek. In Behandelingsstrategieën in Bij Burnout, 2nd ed.; Hoogduin, C.A.L., Schaap, C.P.D.R., Kladler, A.J., Eds.; Houten/Diegem, Bohn Stafleu van Loghum: Houten, The Netherlands, 2001. [Google Scholar]
- Rajkumar, R.P. Harnessing the neurobiology of resilience to protect the mental well-being of healthcare workers during the COVID-19 pandemic. Front. Psychol. 2021, 12, 621853. [Google Scholar] [CrossRef]
- Verhaeghe, J.; Van Den Eede, F.; Van Den Ameele, H.; Sabbe, B.G. Neuro-endocrine correlates of burnout. Tijdschr. Psychiatry 2012, 54, 517–526. [Google Scholar]
- Lehmann, M.J.; Lormes, W.; Opitz-Gress, A.; Steinacker, J.M.; Netzer, N.; Foster, C.; Gastmann, U. Training and overtraining: An overview and experimental results in endurance sports. J. Sports Med. Phys. Fit. 1997, 37, 7–17. [Google Scholar]
- Leistner, C.; Menke, A. Hypothalamic-pituitary-adrenal axis and stress. Handb. Clin. Neurol. 2020, 175, 55–64. [Google Scholar] [CrossRef] [PubMed]
- Weisel, F.; Shlomchik, M. Memory B cells of mice and humans. Annu. Rev. Immunol. 2017, 35, 255–284. [Google Scholar] [CrossRef] [PubMed]
- Romito, B.T.; Okoro, E.N.; Ringqvist, J.R.B.; Goff, K.L. Burnout and wellness: The anesthesiologist’s perspective. Am. J. Lifestyle Med. 2020, 15, 118–125. [Google Scholar] [CrossRef]
- Wojcik, G.M.; Masiak, J.; Kawiak, A.; Kwasniewicz, L.; Schneider, P.; Postepski, F.; Gajos-Balinska, A. Analysis of decision-making process using methods of quantitative electroencephalography and machine learning tools. Front. Neuroinform. 2019, 13, 73. [Google Scholar] [CrossRef] [PubMed]
- Wojcik, G.M.; Masiak, J.; Kawiak, A.; Kwasniewicz, L.; Schneider, P.; Polak, N.; Gajos-Balinska, A. Mapping the human brain in frequency band analysis of brain cortex electroencephalographic activity for selected psychiatric disorders. Front. Neuroinform. 2018, 12, 73. [Google Scholar] [CrossRef] [PubMed]
- Chow, Y.; Masiak, J.; Mikołajewska, E.; Mikołajewski, D.; Wójcik, G.M.; Wallace, B.; Eugene, A.; Olajossy, M. Limbic brain structures and burnout—A systematic review. Adv. Med. Sci. 2018, 63, 192–198. [Google Scholar] [CrossRef] [PubMed]
- Koutsimani, P.; Montgomery, A.; Masoura, E.; Panagopoulou, E. Burnout and cognitive performance. Int. J. Environ. Res. Public Health 2021, 18, 2145. [Google Scholar] [CrossRef] [PubMed]
- van Dijk, D.M.; van Rhenen, W.; Murre, J.M.J.; Verwijk, E. Cognitive functioning, sleep quality, and work performance in non-clinical burnout: The role of working memory. PLoS ONE 2020, 15, e0231906. [Google Scholar] [CrossRef]
- Guggisberg, A.G.; Koch, P.J.; Hummel, F.C.; Buetefisch, C.M. Brain networks and their relevance for stroke rehabilitation. Clin. Neurophysiol. 2019, 130, 1098–1124. [Google Scholar] [CrossRef]
- Miyawaki, A. Brain clearing for connectomics. Microscopy 2015, 64, 5–8. [Google Scholar] [CrossRef]
- Middlebrooks, E.H.; Grewal, S.S. Brain connectomics. Neuroimaging Clin. N. Am. 2022, 32, 543–552. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, M.S.; Ahmad, I.; Bin, L.; Khan, S.; Rodrigues, J.J.P.C. Deep learning recognition of diseased and normal cell representation. Trans. Emerg. Telecommun. Technol. 2020, 42, e4017. [Google Scholar] [CrossRef]
- Iqbal, M.S.; El-Ashram, S.; Hussain, S.; Khan, T.; Huang, S.; Mehmood, R.; Luo, B. Efficient cell classification of mitochondrial images by using deep learning. J. Opt. 2019, 48, 113–122. [Google Scholar] [CrossRef]

| Study (Year) | Method/Design | Characteristics of Samples Studied | Main Result(s) Statistical Significance |
|---|---|---|---|
| Luijtelaar et al. (2010) [1] | Examined brain function in burnout patients by analyzing EEG and neuropsychological outcomes. | 13 burnout subjects (mean age 48.2, range 26–55). 13 healthy controls 4 females, 9 males in each group. | Reduced P300 potency, peak alpha (F = 4.40,df = 1. 24, p < 0.05)and beta (F = 5.86, df = 1.24, p < 0.05) in burnout patients. |
| Tement et al. (2016) [18] | Examined the relationship between potential biomarkers in the alpha frequency band and student self-reports of occupational burnout and the role of gender. | 117 subjects. 75 females, 42 males, aged between 19 and 29 (females mean = 22.41, SD = 1.81; males: mean22.26, SD = 2.34). | IAF is associated with depression and power is associated with burnout (t(115) = 3.02, p = 0.003); gender differences were observed only for burnout questionnaire scores: males had a significantly higher score. |
| Golonka et al. (2019) [19] | Examined differences in brain activity were investigated by analyzing EEG power versus resting frequency in burnout and control patients. | 46 burnout employees (incl. 19 males). 46 heathy controls (incl. 19 males), mean age: 36.14 years, SD = 7.89. | Reduced alpha power in burnout patients indicatedcortical hyperactivity associated with greater mental effort and the development of compensatory mechanisms.Alpha power significantly correlated with burnout only in the males: anterior area: r = −0.37, p = 0.021;central area: r = −0.37, p = 0.023; posterior area: r = −0.35, p = 0.032. |
| Yakovenko et al. (2021) [20] | Quantitative EEG analysis of different stages of burnout syndrome (tension, resistance, exhaustion). | 131 burnout employees (middle school teachers and medical workers). 38 males and 93 females, aged 25–45 (mean 34.2 ± 7.3) years, 143 healthy controls. | At various stages of burnout, an increase in dysfunction of the brain’s regulatory systems was observed, correlating with an increase in the severity of clinical symptoms (p < 0.05). |
| Durning et al. (2013) [21] | Examined how burnout modulates brain activity during clinical reasoning in physicians. | 17 internal medicine residents aged 29.6 ± 2 (range 28–35) years, 15 males and 2 females, 17 board-certified internists aged 39.5 ± 7 (range = 32–51) years, 10 males and 7 females. | Depersonalization was related to less BOLD in DLPFC and MFG (p = 0.033), residents mean = 2.33 (SD 2), while internists mean = 0.8 (SD 1.08). Exhaustion of emotions was related with more BOLD in MFG and CC (p = 0.011), residents mean = 3 (SD 2.12) while internists mean = 1.67 (SD 1.29). |
| Savic (2013) [22] | Investigated structural changes in the brain in connection with occupational stress in the MRI study (cortical thickness (Cth) and subcortical volumes). | 40 burnout subjects (38 ± 6 years, range 19–46 years, 15 males, 25 females). 40 healthy controls (36 ± 6 years, range 15–45 years, 15 females, 25 males). | Reduced volume of the amygdala, caudate nucleus, and impaired motor function in burnout patients (r = 0.44, p = 0.04; r = 0.43, p = 0.04). |
| Blix et al. (2013) [23] | MRI study where cerebral gray matter and white matter volume were compared between patients with chronic work-related stress and healthy subjects. | 30 burnout subjects (23 females and 7 males, age 41.3 ± 6.6, range 36–55 years). 68 healthy controls (53 females and 15 males, age 37.5 ± 7.2 years, range 27–51 years). | Reduced volumes of GM, ACC, and DLPFC in burnout patients (4.2 ± 1.1 vs. 2.2 ± 0.5; p <0.0001; F = 100.6, df = 1) vs. below 3.0 in the control group. |
| Tei et al. (2014) [24] | Investigated the relationship between self-reported burnout severity scores and psychological measures of empathic disposition. | 25 nursesin active service with less than 11 years of experience (20 females, aged 22–34, mean = 26.0, SD = 3.14 years). | The severity of burnout among physicians wasexplained by “decreased” brain activity related to empathy. AI/IFG and TPJ activity negatively correlated with emotional exhaustion on the MBI: r = −0.590, p = 0.002, and r = −0.550, p = 0.004, respectively, |
| Savic et al. (2018) [25] | Study of the cerebral effects of chronic occupational stress and their possible reversibility. | 48 patients with occupational exhaustion syndrome (29 females), aged 38 ± 6 (26–46) years. 80 healthy controls(47 females), aged 32 ± 7 (20–45) years. After 1–2 years: 25 patients with occupational exhaustion syndrome. 19 healthy controls. | Chronic work-related stress was associated with partially reversible structural abnormalities in key stress-processing regions, correlated with the degree of perceived stress. Sustained attention as well as verbal memory were impaired only among females (F = 1.40, p = 0.79, df = 1). |
| Gavelin et al. (2017) [26] | A study of the relationship between burnout and neuron activation in working memory processing in patients with stress-induced exhaustion and the neural effects of cognitive training in post-stress rehabilitation. | 55 patients with a clinical diagnosis of exhaustion syndrome, including 10 patients completed a 12-week cognitive training intervention in addition to stress rehabilitation. 46 females, 9 males, aged 42.85 (SD = 8.97), range 22–60 years. The control group was treated traditionally. | There was no correlation between the level of burnout and working memory performance. The striatal frontal nerve responses related to working memory were modulated by the severity of burnout. Levels of burnout decreased following rehabilitation: from mean = 4.86 (SD = 0.93) to mean = 4.08 (SD = 1.05). p < 0.001. |
| Jovanovic et al. (2011) [27] | Limbic function test with PET on chronic stress subjects. | 16 stress subjects: 11 females, 5 males;mean age 38 ± 5, range 28–47 years. 16 healthy controls. | Functional disconnection between amygdala and ACC/MPFC in chronically stressed subjects. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mikołajewska, E.; Prokopowicz, P.; Chow, Y.; Masiak, J.; Mikołajewski, D.; Wójcik, G.M.; Wallace, B.; Eugene, A.R.; Olajossy, M. From Neuroimaging to Computational Modeling of Burnout: The Traditional versus the Fuzzy Approach—A Review. Appl. Sci. 2022, 12, 11524. https://doi.org/10.3390/app122211524
Mikołajewska E, Prokopowicz P, Chow Y, Masiak J, Mikołajewski D, Wójcik GM, Wallace B, Eugene AR, Olajossy M. From Neuroimaging to Computational Modeling of Burnout: The Traditional versus the Fuzzy Approach—A Review. Applied Sciences. 2022; 12(22):11524. https://doi.org/10.3390/app122211524
Chicago/Turabian StyleMikołajewska, Emilia, Piotr Prokopowicz, YeeKong Chow, Jolanta Masiak, Dariusz Mikołajewski, Grzegorz Marcin Wójcik, Brian Wallace, Andy R. Eugene, and Marcin Olajossy. 2022. "From Neuroimaging to Computational Modeling of Burnout: The Traditional versus the Fuzzy Approach—A Review" Applied Sciences 12, no. 22: 11524. https://doi.org/10.3390/app122211524
APA StyleMikołajewska, E., Prokopowicz, P., Chow, Y., Masiak, J., Mikołajewski, D., Wójcik, G. M., Wallace, B., Eugene, A. R., & Olajossy, M. (2022). From Neuroimaging to Computational Modeling of Burnout: The Traditional versus the Fuzzy Approach—A Review. Applied Sciences, 12(22), 11524. https://doi.org/10.3390/app122211524







