De Novo Transcriptome Analysis of the Lizard Fish (Saurida elongata): Novel Insights into Genes Related to Sex Differentiation
Abstract
Featured Application
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Collection
2.2. Transcriptome Sequencing
2.3. De Novo Assembly and Functional Annotation
2.4. Candidate Sex-Related Genes Analysis
2.5. Potential Simple Sequence Repeat (SSR) Marker Detection
2.6. Quantitative Real-Time PCR (qRT-PCR) Validation
3. Results
3.1. Transcriptome Sequencing and De Novo Assembly
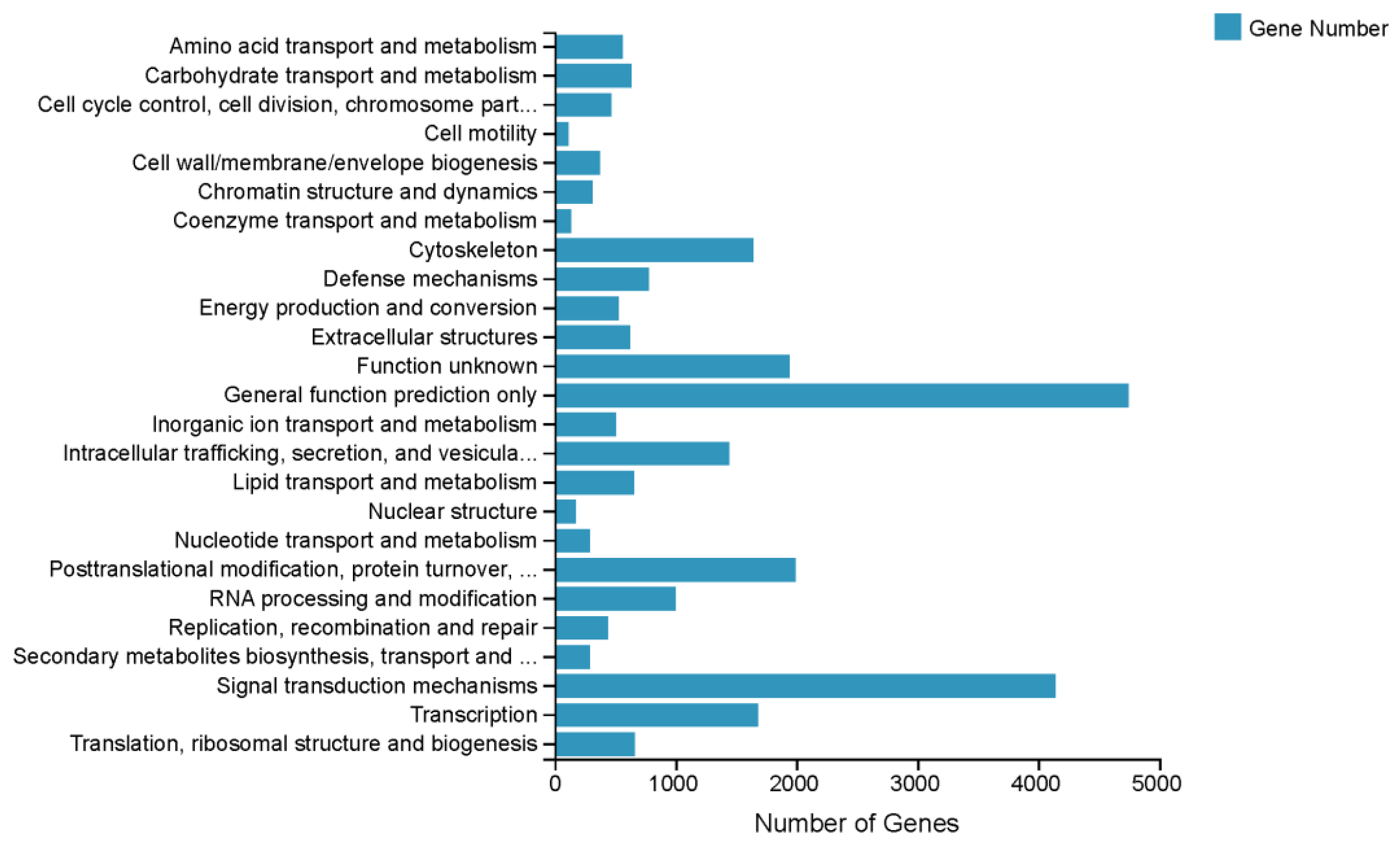
3.2. Functional Annotation of the S. elongata Transcriptome
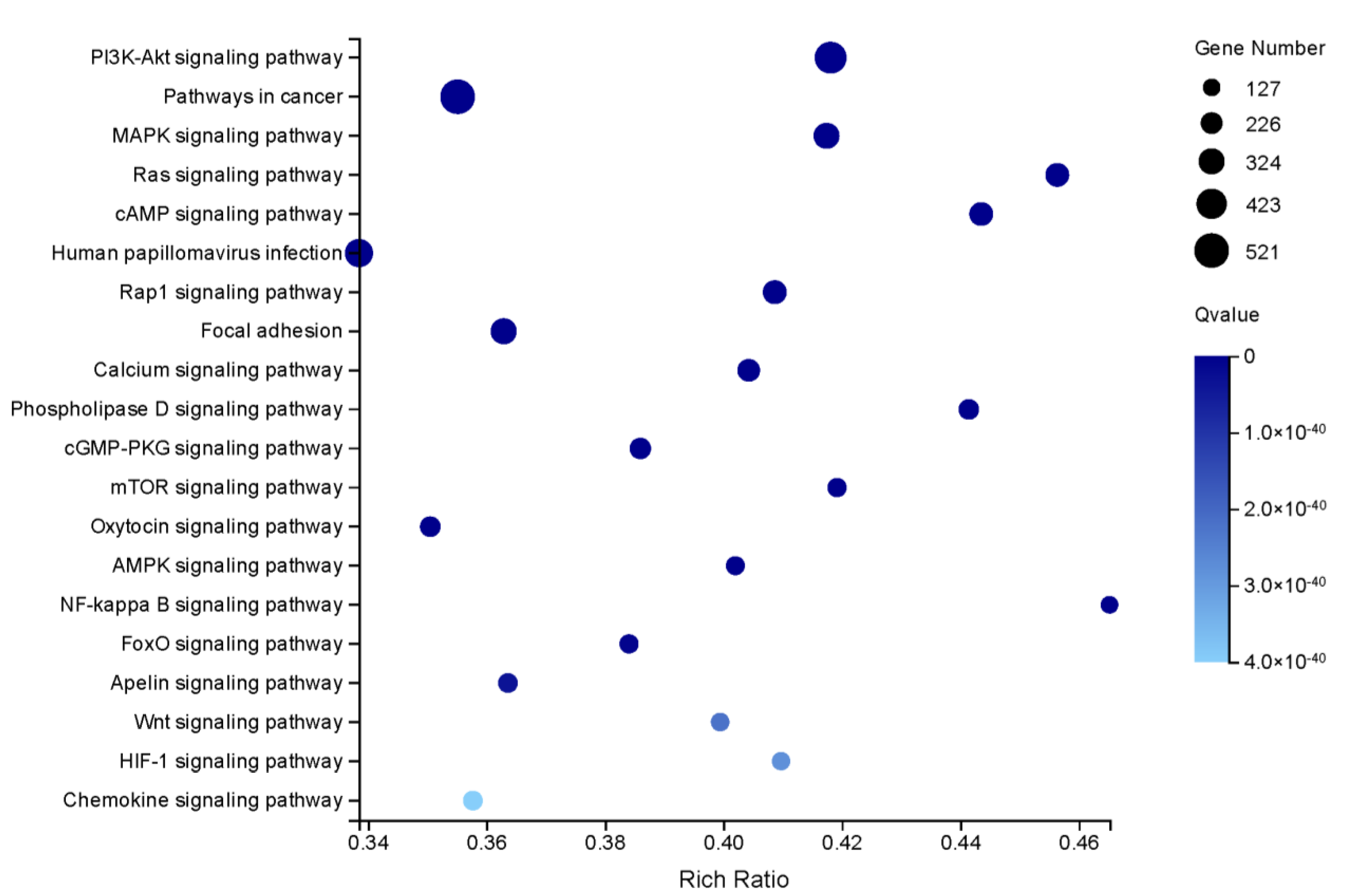
3.3. Candidate Sex-Related Genes and Functional Enrichment Analysis Results
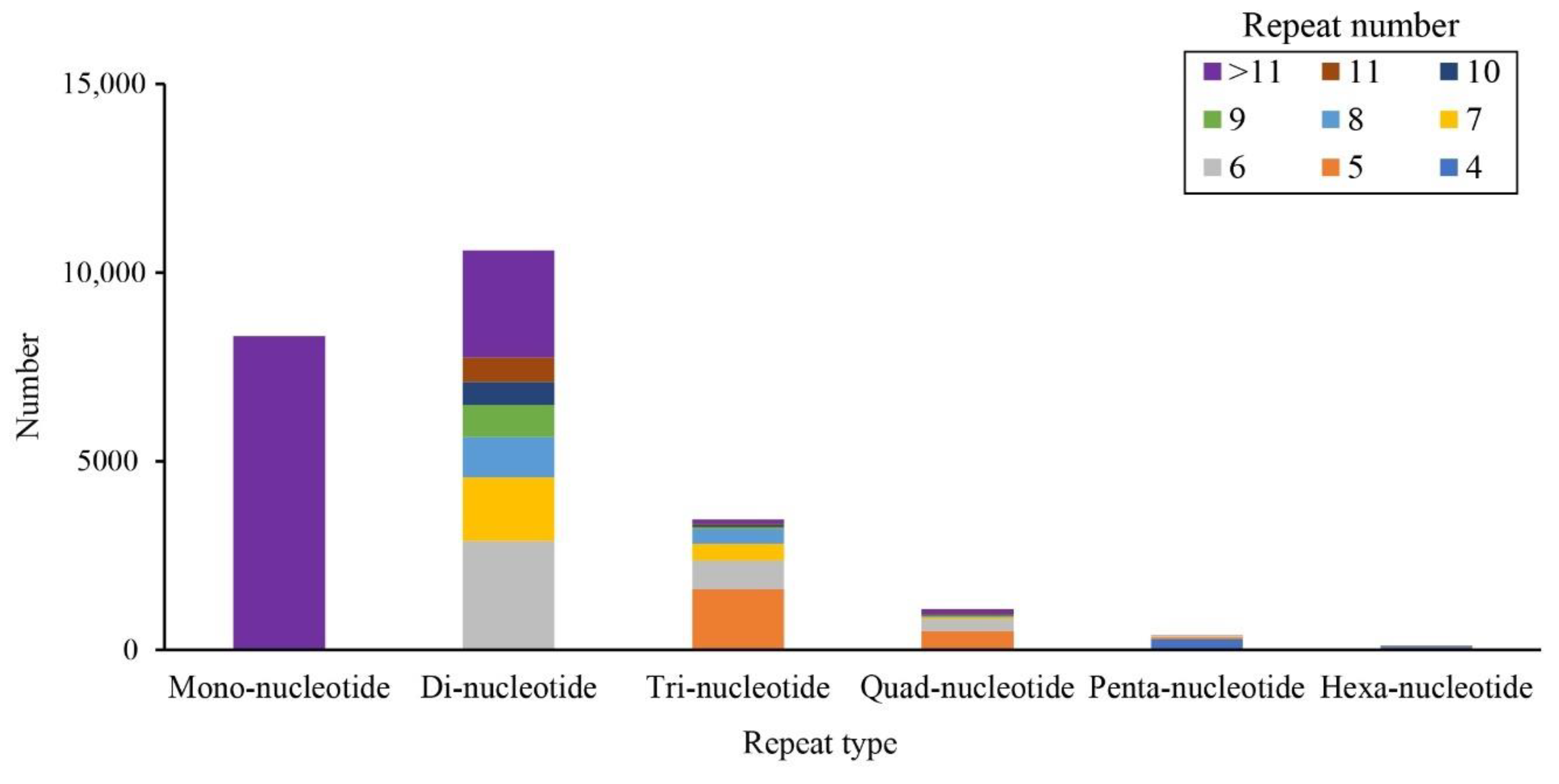
3.4. SSR Markers Detection
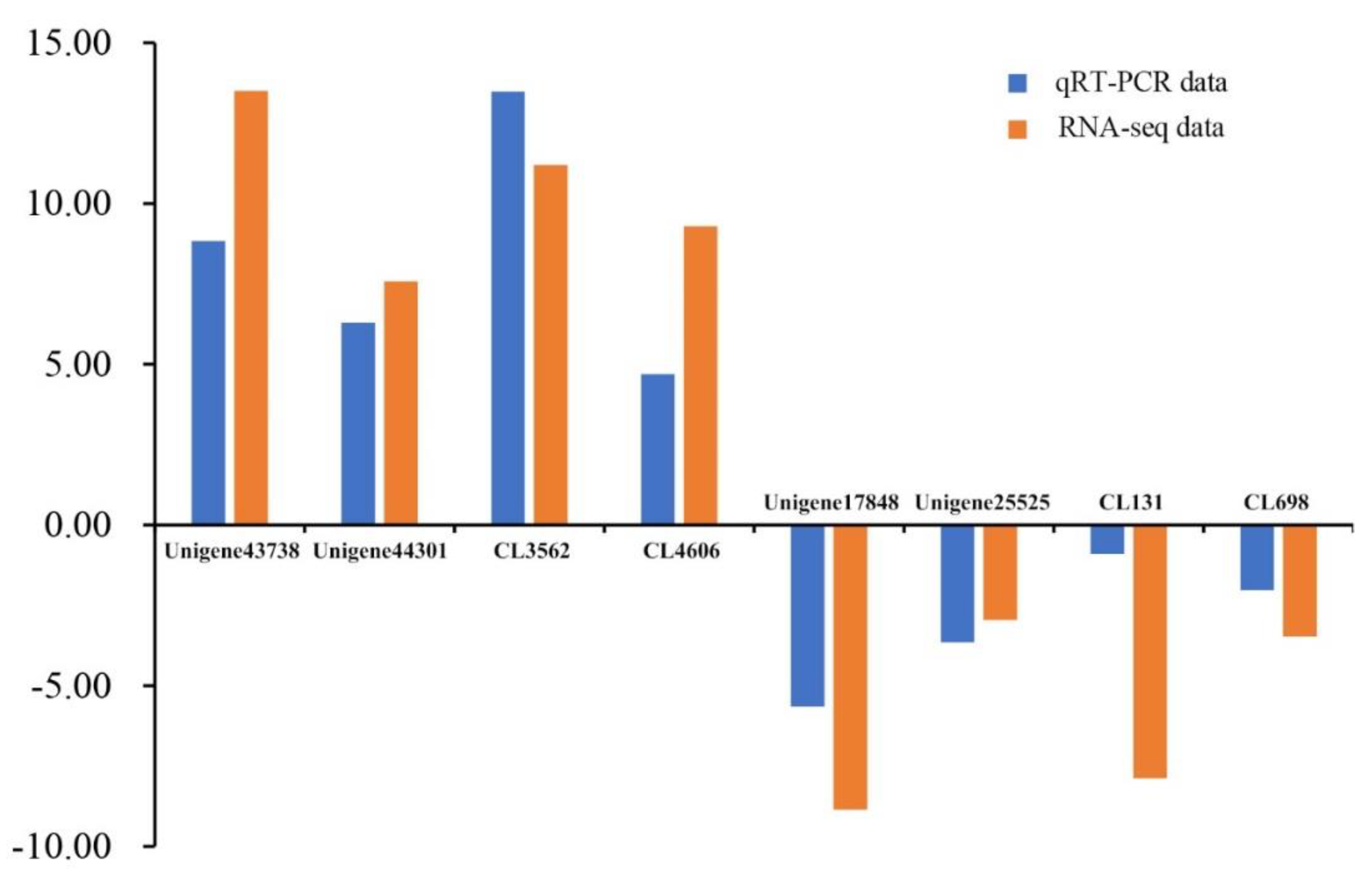
3.5. Transcriptome Data Validation
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ottolenghi, C.; Pelosi, E.; Tran, J.; Colombino, M.; Douglass, E.; Nedorezov, T.; Cao, A.; Forabosco, A.; Schlessinger, D. Loss of Wnt4 and Foxl2 leads to female-to-male sex reversal extending to germ cells. Hum. Mol. Genet. 2007, 16, 2795–2804. [Google Scholar] [CrossRef]
- Smith, C.A.; Roeszler, K.N.; Ohnesorg, T.; Cummins, D.M.; Farlie, P.G.; Doran, T.J.; Sinclair, A.H. The avian Z-linked gene DMRT1 is required for male sex determination in the chicken. Nature 2009, 461, 267–271. [Google Scholar] [CrossRef] [PubMed]
- Lavery, R.; Chassot, A.A.; Pauper, E.; Gregoire, E.P.; Klopfenstein, M.; de Rooij, D.G.; Mark, M.; Schedl, A.; Ghyselinck, N.B.; Chaboissier, M.C. Testicular differentiation occurs in absence of R-spondin1 and Sox9 in mouse sex reversals. PLoS Genet. 2012, 8, e1003170. [Google Scholar] [CrossRef]
- Chandler, J.C.; Elizur, A.; Ventura, T.J.H. The decapod researcher’s guide to the galaxy of sex determination. Hydrobiologia 2018, 825, 61–80. [Google Scholar] [CrossRef]
- Tao, W.J.; Chen, J.L.; Tan, D.J.; Yang, J.; Sun, L.N.; Wei, J.; Conte, M.A.; Kocher, T.D.; Wang, D.S. Transcriptome display during tilapia sex determination and differentiation as revealed by RNA-Seq analysis. BMC Genomics 2018, 19, 363. [Google Scholar] [CrossRef] [PubMed]
- Barske, L.A.; Capel, B. Blurring the edges in vertebrate sex determination. Curr. Opin. Genet. Dev. 2008, 18, 499–505. [Google Scholar] [CrossRef] [PubMed]
- Pan, Q.W.; Anderson, J.; Bertho, S.; Herpin, A.; Wilson, C.; Postlethwait, J.H.; Schartl, M.; Guiguen, Y. Vertebrate sex-determining genes play musical chairs. C. R. Biol. 2016, 339, 258–262. [Google Scholar] [CrossRef]
- Kobayashi, Y.; Nagahama, Y.; Nakamura, M.J.S.D. Diversity and plasticity of sex determination and differentiation in fishes. Sex. Dev. 2013, 7, 115–125. [Google Scholar] [CrossRef]
- Casas, L.; Saborido-Rey, F.; Ryu, T.; Michell, C.; Ravasi, T.; Irigoien, X. Sex Change in Clownfish: Molecular insights from transcriptome analysis. Sci. Rep. 2016, 6, 35461. [Google Scholar] [CrossRef]
- Saillant, E.; Fostier, A.; Menu, B.; Haffray, P.; Chatain, B. Sexual growth dimorphism in sea bass Dicentrarchus labrax. Aquaculture 2001, 202, 371–387. [Google Scholar] [CrossRef]
- Dutney, L.; Elizur, A.; Lee, P. Analysis of sexually dimorphic growth in captive reared cobia (Rachycentron canadum) and the occurrence of intersex individuals. Aquaculture 2017, 468, 348–355. [Google Scholar] [CrossRef]
- Wang, N.; Wang, R.K.; Wang, R.Q.; Chen, S.L. Transcriptomics analysis revealing candidate networks and genes for the body size sexual dimorphism of Chinese tongue sole (Cynoglossus semilaevis). Funct. Integr. Genomic. 2018, 18, 327–339. [Google Scholar] [CrossRef] [PubMed]
- Cribbin, K.M.; Quackenbush, C.R.; Taylor, K.; Arias-Rodriguez, L.; Kelley, J.L. Sex-specific differences in transcriptome profiles of brain and muscle tissue of the tropical gar. BMC Genom. 2017, 18, 283. [Google Scholar] [CrossRef] [PubMed]
- Yoneda, A.; Sakai, T.; Tokimura, A.; Horikawa, H.; Matsuyama, M. Age and growth of the lizardfish Saurida sp. 1 in the East China Sea using otolith ring marks. Fish. Res. 2002, 55, 231–238. [Google Scholar] [CrossRef]
- Sakai, T.; Yoneda, M.; Shiraishi, T.; Tokimura, M.; Horikawa, H.; Matsuyama, M. Age and growth of the lizardfish Saurida elongata from the Tsushima/Korea Strait. Fish. Sci. 2009, 75, 895–902. [Google Scholar] [CrossRef]
- Shimizu, Y.; Wendakoon, C.N. Agriculture Effects of maturation and spawning on the gel-forming ability of lizardfish (Saurida elongata) muscle tissues. J. Sci. Food Agr. 1990, 52, 331–338. [Google Scholar] [CrossRef]
- Wu, S.G.; Sun, J.H.; Tong, Z.F.; Lan, X.D.; Zhao, Z.X.; Liao, D.K. Optimization of hydrolysis conditions for the production of angiotensin-I converting enzyme-inhibitory peptides and isolation of a novel peptide from lizard fish (Saurida elongata) muscle protein hydrolysate. Mar. Drugs 2012, 10, 1066–1080. [Google Scholar] [CrossRef]
- Lan, X.D.; Sun, L.X.; Muhammad, Y.; Wang, Z.F.; Liu, H.B.; Sun, J.H.; Zhou, L.Q.; Feng, X.Z.; Liao, D.K.; Wang, S.F. Studies on the interaction between angiotensin-converting enzyme (ACE) and ACE inhibitory peptide from Saurida elongata. J. Agric. Food Chem. 2018, 66, 13414–13422. [Google Scholar] [CrossRef]
- Liu, Y.W.; Zhang, C.L.; Zan, X.X.; Sun, M.; Xu, B.D.; Xue, Y.; Ren, Y.P. Distribution of relative abundance of slender lizardfish and its influencing factors in southern coastal waters of Shandong during autumn. Period. Ocean Univ. China 2020, 50, 45–53. [Google Scholar]
- Tu, Z.; Liu, M.; Wang, Y.P.; Xu, S.Y.; Song, N.; Gao, T.X.; Han, Z.Q. The low mitochondrial diversities in lizardfish Saurida elongata: Recent population expansion and selection. Biochem. Syst. Ecol. 2016, 68, 44–50. [Google Scholar] [CrossRef]
- Lobo, I.K.C.; do Nascimento, A.R.; Yamagishi, M.E.B.; Guiguen, Y.; da Silva, G.F.; Severac, D.; Amaral, A.D.; Reis, V.R.; de Almeida, F.L. Transcriptome of tambaqui Colossoma macropomum during gonad differentiation: Different molecular signals leading to sex identity. Genomics 2020, 112, 2478–2488. [Google Scholar] [CrossRef] [PubMed]
- Chatchaiphan, S.; Srisapoome, P.; Kim, J.H.; Devlin, R.H.; Na-Nakorn, U. De novo transcriptome characterization and growth-related gene expression profiling of diploid and triploid bighead catfish (Clarias macrocephalus Gunther, 1864). Mar. Biotechnol. 2017, 19, 36–48. [Google Scholar] [CrossRef] [PubMed]
- Lou, F.R.; Yang, T.Y.; Han, Z.Q.; Gao, T.X. Transcriptome analysis for identification of candidate genes related to sex determination and growth in Charybdis japonica. Gene 2018, 677, 10–16. [Google Scholar] [CrossRef] [PubMed]
- Ma, D.; Ma, A.; Huang, Z.; Wang, G.N.; Wang, T.; Xia, D.; Ma, B. Transcriptome analysis for identification of genes related to gonad differentiation, growth, immune response and marker discovery in the turbot (Scophthalmus maximus). PLoS ONE 2016, 11, e0149414. [Google Scholar] [CrossRef] [PubMed]
- Tian, C.; Li, Z.; Dong, Z.; Huang, Y.; Du, T.; Chen, H.; Jiang, D.; Deng, S.; Zhang, Y.; Wandia, S.; et al. Transcriptome analysis of male and female mature gonads of silver sillago (Sillago sihama). Genes 2019, 10, 129. [Google Scholar] [CrossRef]
- Shan, B.; Liu, Y.; Yang, C.; Zhao, Y.; Sun, D. Comparative transcriptomic analysis for identification of candidate sex-related genes and pathways in Crimson seabream (Parargyrops edita). Sci. Rep. 2021, 11, 1077. [Google Scholar] [CrossRef]
- Schroeder, A.; Mueller, O.; Stocker, S.; Salowsky, R.; Leiber, M.; Gassmann, M.; Lightfoot, S.; Menzel, W.; Granzow, M.; Ragg, T. The RIN: An RNA integrity number for assigning integrity values to RNA measurements. BMC Mol. Biol. 2006, 7, 3. [Google Scholar] [CrossRef]
- Bolger, A.M.; Lohse, M.; Usadel, B. Trimmomatic: A flexible trimmer for Illumina sequence data. Bioinformatics 2014, 30, 2114–2120. [Google Scholar] [CrossRef]
- Grabherr, M.G.; Haas, B.J.; Yassour, M.; Levin, J.Z.; Thompson, D.A.; Amit, I.; Adiconis, X.; Fan, L.; Raychowdhury, R.; Zeng, Q.J. Trinity: Reconstructing a full-length transcriptome without a genome from RNA-Seq data. Nat. Biotechnol. 2011, 29, 644. [Google Scholar] [CrossRef]
- Simão, F.A.; Waterhouse, R.M.; Ioannidis, P.; Kriventseva, E.V.; Zdobnov, E.M. BUSCO: Assessing genome assembly and annotation completeness with single-copy orthologs. Bioinformatics 2015, 31, 3210–3212. [Google Scholar] [CrossRef]
- Langmead, B.; Salzberg, S.L. Fast gapped-read alignment with Bowtie 2. Nat. Methods 2012, 9, 357. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Dewey, C.N. RSEM: Accurate transcript quantification from RNA-Seq data with or without a reference genome. BMC Bioinform. 2011, 12, 323. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.K.; Feng, Z.X.; Wang, X.; Wang, X.W.; Zhang, X.G. DEGseq: An R package for identifying differentially expressed genes from RNA-seq data. Bioinformatics 2010, 26, 136–138. [Google Scholar] [CrossRef]
- Untergasser, A.; Cutcutache, I.; Koressaar, T.; Ye, J.; Faircloth, B.C.; Remm, M.; Rozen, S.G. Primer3—New capabilities and interfaces. Nucleic Acids Res. 2012, 40, e115. [Google Scholar] [CrossRef]
- Filby, A.L.; Tyler, C.R. Appropriate ’housekeeping’ genes for use in expression profiling the effects of environmental estrogens in fish. BMC Mol. Biol. 2007, 8, 10. [Google Scholar] [CrossRef]
- Martins, R.S.; Pinto, P.I.; Guerreiro, P.M.; Zanuy, S.; Carrillo, M.; Canário, A.V. Novel galanin receptors in teleost fish: Identification, expression and regulation by sex steroids. Gen. Comp. Endocr. 2014, 205, 109–120. [Google Scholar] [CrossRef] [PubMed]
- Domingos, J.A.; Zenger, K.R.; Jerry, D.R. Whole-genome shotgun sequence assembly enables rapid gene characterization in the tropical fish barramundi, Lates calcarifer. Anim. Genet. 2015, 46, 468–469. [Google Scholar] [CrossRef]
- Schmid, R.; Blaxter, M.L. annot8r: GO, EC and KEGG annotation of EST datasets. BMC Bioinform. 2008, 9, 180. [Google Scholar] [CrossRef]
- Kanehisa, M.; Sato, Y.; Morishima, K. BlastKOALA and GhostKOALA: KEGG tools for functional characterization of genome and metagenome sequences. J. Mol. Biol. 2016, 428, 726–731. [Google Scholar] [CrossRef]
- Rozenfeld, C.; Blanca, J.; Gallego, V.; García-Carpintero, V.; Herranz-Jusdado, J.; Pérez, L.; Asturiano, J.F.; Cañizares, J.; Peñaranda, D.S. De novo European eel transcriptome provides insights into the evolutionary history of duplicated genes in teleost lineages. PLoS ONE 2019, 14, e0218085. [Google Scholar] [CrossRef]
- Wassarman, P.M.J.S. The biology and chemistry of fertilization. Science 1987, 235, 553–560. [Google Scholar] [CrossRef] [PubMed]
- Ringuette, M.J.; Chamberlin, M.E.; Baur, A.W.; Sobieski, D.A.; Dean, J.J.D.B. Molecular analysis of cDNA coding for ZP3, a sperm binding protein of the mouse zona pellucida. Dev. Biol. 1988, 127, 287–295. [Google Scholar] [CrossRef]
- Dumont, J.N.; Brummett, A.R.J.O. Egg Envelopes in Vertebrates. In Oogenesis; Springer: Berlin/Heidelberg, Germany, 1985; pp. 235–288. [Google Scholar]
- Litscher, E.S.; Wassarman, P.M. Egg extracellular coat proteins: From fish to mammals. Histol. Histopathol. 2007, 22, 337–347. [Google Scholar] [PubMed]
- Meczekalski, B.; Nawrot, R.; Nowak, W.; Czyzyk, A.; Kedzia, H.; Gozdzicka-Jozefiak, A. Study on the zona pellucida 4 (ZP4) gene sequence and its expression in the ovaries of patients with polycystic ovary syndrome. J. Endocrinol. Investig. 2015, 38, 791–797. [Google Scholar] [CrossRef]
- Kikuchi, K.; Hamaguchi, S. Novel sex-determining genes in fish and sex chromosome evolution. Dev. Dyn. 2013, 242, 339–353. [Google Scholar] [CrossRef]
- Zhang, X.B.; Wang, H.; Li, M.H.; Cheng, Y.Y.; Jiang, D.N.; Sun, L.N.; Tao, W.J.; Zhou, L.Y.; Wang, Z.J.; Wang, D.S. Isolation of doublesex- and mab-3-related transcription factor 6 and its involvement in spermatogenesis in tilapia. Biol. Reprod. 2014, 91, 136. [Google Scholar] [CrossRef]
- Matson, C.K.; Murphy, M.W.; Griswold, M.D.; Yoshida, S.; Bardwell, V.J.; Zarkower, D. The mammalian doublesex homolog DMRT1 is a transcriptional gatekeeper that controls the mitosis versus meiosis decision in male germ cells. Dev. Cell 2010, 19, 612–624. [Google Scholar] [CrossRef]
- Erdman, S.E.; Burtis, K.C. The Drosophila doublesex proteins share a novel zinc finger related DNA binding domain. EMBO J. 1993, 12, 527–535. [Google Scholar] [CrossRef]
- Krentz, A.D.; Murphy, M.W.; Sarver, A.L.; Griswold, M.D.; Bardwell, V.J.; Zarkower, D. DMRT1 promotes oogenesis by transcriptional activation of Stra8 in the mammalian fetal ovary. Dev. Biol. 2011, 356, 63–70. [Google Scholar] [CrossRef]
- Veith, A.M.; Klattig, J.; Dettai, A.; Schmidt, C.; Englert, C.; Volff, J.N. Male-biased expression of X-chromosomal DM domain-less Dmrt8 genes in the mouse. Genomics 2006, 88, 185–195. [Google Scholar] [CrossRef]
- Rather, M.A.; Dhandare, B.C.J.B.R. Genome-Wide identification of doublesex and Mab-3-Related transcription factor (DMRT) genes in nile tilapia (Oreochromis niloticus). Biotechnol. Rep. 2019, 24, e00398. [Google Scholar]
- Johnsen, H.; Andersen, O. Sex dimorphic expression of five dmrt genes identified in the Atlantic cod genome. The fish-specific dmrt2b diverged from dmrt2a before the fish whole-genome duplication. Gene 2012, 505, 221–232. [Google Scholar] [CrossRef]
- Zhou, X.; Li, Q.; Lu, H.; Chen, H.; Guo, Y.Q.; Cheng, H.H.; Zhou, R.J. Fish specific duplication of Dmrt2: Characterization of zebrafish Dmrt2b. Biochimie 2008, 90, 878–887. [Google Scholar] [CrossRef]
- Yamaguchi, A.; Lee, K.H.; Fujimoto, H.; Kadomura, K.; Yasumoto, S.; Matsuyama, M. Expression of the DMRT gene and its roles in early gonadal development of the Japanese pufferfish Takifugu rubripes. Comp. Biochem. Phys. D 2006, 1, 59–68. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Cui, Z.K.; Yang, Y.M.; Xu, W.T.; Shao, C.W.; Fu, X.Q.; Li, Y.Z.; Chen, S.L. Expression analysis and characterization of dmrt2 in Chinese tongue sole (Cynoglossus semilaevis). Theriogenology 2019, 138, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Lourenco, R.; Lopes, S.S.; Saude, L. Left-right function of dmrt2 genes is not conserved between zebrafish and mouse. PLoS ONE 2010, 5, e14438. [Google Scholar] [CrossRef]
- Yoshizawa, A.; Nakahara, Y.; Izawa, T.; Ishitani, T.; Tsutsumi, M.; Kuroiwa, A.; Itoh, M.; Kikuchi, Y. Zebrafish Dmrta2 regulates neurogenesis in the telencephalon. Genes Cells 2011, 16, 1097–1109. [Google Scholar] [CrossRef]
- Guan, G.J.; Kobayashi, T.; Nagahama, Y. Sexually dimorphic expression of two types of DM (Doublesex/Mab-3)-domain genes in a teleost fish, the Tilapia (Oreochromis niloticus). Biochem. Biophys. Res. Commun. 2000, 272, 662–666. [Google Scholar] [CrossRef]
- Cao, J.L.; Chen, J.J.; Wu, T.T.; Gan, X.; Luo, Y.J. Molecular cloning and sexually dimorphic expression of DMRT4 gene in Oreochromis aureus. Mol. Biol. Rep. 2010, 37, 2781–2788. [Google Scholar] [CrossRef]
- Wen, A.Y.; You, F.; Tan, X.G.; Sun, P.; Ni, J.; Zhang, Y.Q.; Xu, D.D.; Wu, Z.H.; Xu, Y.L.; Zhang, P.J. Expression pattern of dmrt4 from olive flounder (Paralichthys olivaceus) in adult gonads and during embryogenesis. Fish Physiol. Biochem. 2009, 35, 421–433. [Google Scholar] [CrossRef]
- Nakamoto, M.; Matsuda, M.; Wang, D.S.; Nagahama, Y.; Shibata, N. Molecular cloning and analysis of gonadal expression of Foxl2 in the medaka, Oryzias latipes. Biochem. Biophys. Res. Commun. 2006, 344, 353–361. [Google Scholar] [CrossRef] [PubMed]
- Fan, Z.F.; Zou, Y.X.; Liang, D.D.; Tan, X.G.; Jiao, S.; Wu, Z.H.; Li, J.; Zhang, P.J.; You, F. Roles of forkhead box protein L2 (foxl2) during gonad differentiation and maintenance in a fish, the olive flounder (Paralichthys olivaceus). Reprod. Fert. Develop. 2019, 31, 1742–1752. [Google Scholar] [CrossRef]
- Li, C.G.; Wang, H.; Chen, H.J.; Zhao, Y.; Fu, P.S.; Ji, X.S. Differential expression analysis of genes involved in high-temperature induced sex differentiation in Nile tilapia. Comp. Biochem. Phys. B 2014, 177, 36–45. [Google Scholar] [CrossRef] [PubMed]
- Shi, H.J.; Gao, T.; Liu, Z.L.; Sun, L.N.; Jiang, X.L.; Chen, L.L.; Wang, D.S. Blockage of androgen and administration of estrogen induce transdifferentiation of testis into ovary. J. Endocrinol. 2017, 233, 65–80. [Google Scholar] [CrossRef] [PubMed]
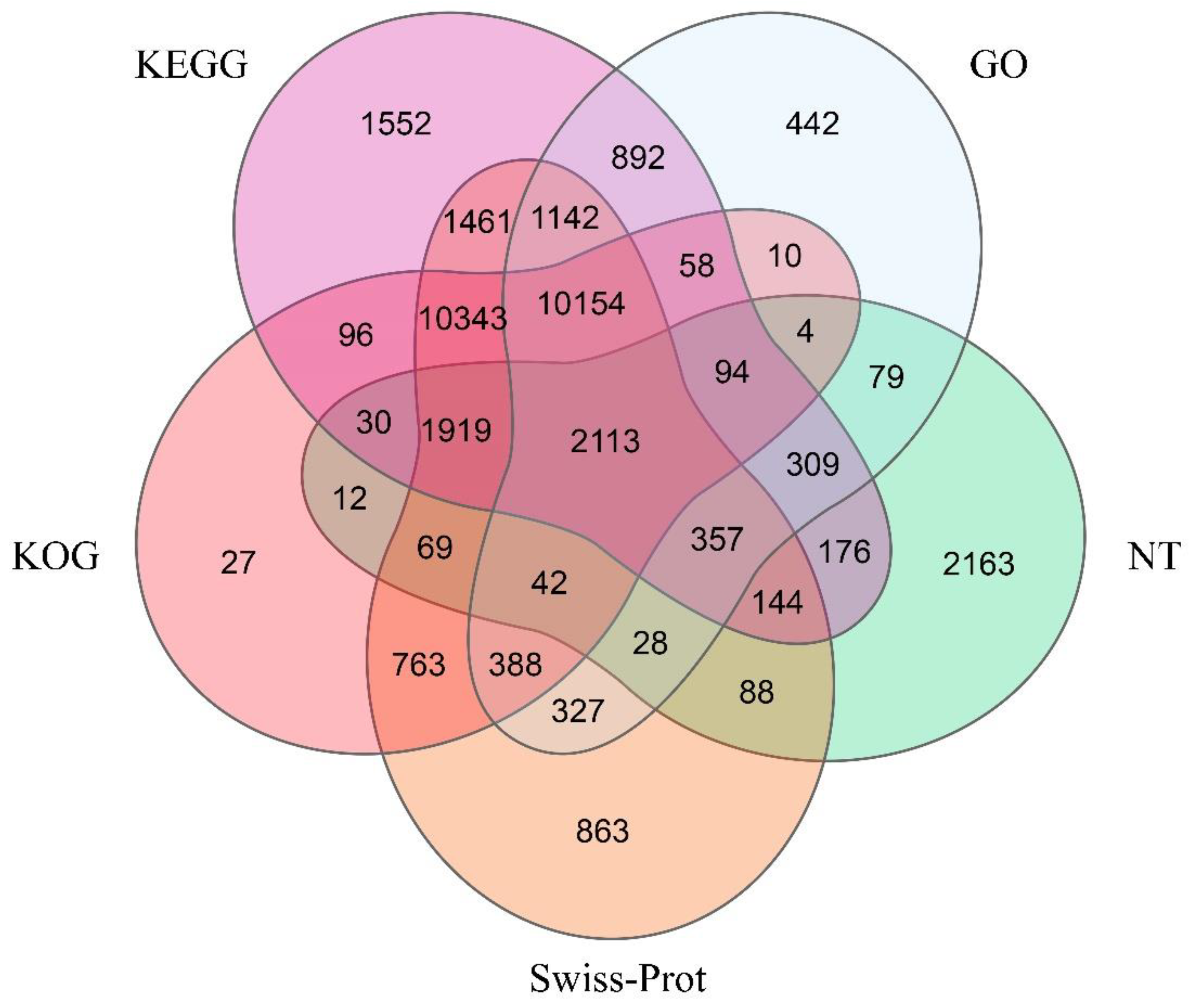


| Database | Number of Unigenes | Percentage (%) |
|---|---|---|
| NR | 35,479 | 59.23% |
| Swiss-Prot | 30,201 | 50.42% |
| KEGG | 30,840 | 51.48% |
| KOG | 26,122 | 43.61% |
| GO | 16,439 | 27.44% |
| Unigene | Annotation | log2 (♂/♀) | p-Value |
|---|---|---|---|
| CL1525.Contig1_All | Zona pellucida sperm-binding protein 1 | −17.663 | 0 |
| Unigene35458_All | Zona pellucida sperm-binding protein 2 | −9.959 | 0 |
| CL1340.Contig1_All | Zona pellucida sperm-binding protein 3 | −8.554 | 8.109 × 10−101 |
| CL4606.Contig2_All | Zona pellucida sperm-binding protein 4 | −8.602 | 3.971 × 10−224 |
| Unigene16088_All | StAR-related lipid transfer protein 4 | 2.207 | 4.498 × 10−28 |
| Unigene42268_All | StAR-related lipid transfer protein 5 | −3.659 | 9.132 × 10−09 |
| Unigene3434_All | StAR-related lipid transfer protein 7 | −2.968 | 2.820 × 10−74 |
| Unigene7294_All | StAR-related lipid transfer protein 13 | 1.237 | 3.248 × 10−08 |
| Unigene10706_All | Spermatogenesis-associated protein 20 | −1.184 | 3.857 × 10−05 |
| CL3615.Contig1_All | Spermatogenesis-associated protein 5-like protein 1 | −8.618 | 1.846 × 10−26 |
| Unigene15846_All | Spermatogenesis-associated protein 7 | −1.838 | 4.243 × 10−26 |
| Unigene3310_All | Spermatogenesis-associated protein 2 | −1.208 | 1.846 × 10−30 |
| Unigene9109_All | Spermatogenesis-associated protein 13 | 1.496 | 6.667 × 10−19 |
| Unigene44347_All | Zteroidogenic acute regulatory protein | −8.039 | 5.755 × 10−05 |
| Unigene12505_All | Meiotic nuclear division protein 1 homolog | −1.587 | 3.245 × 10−26 |
| CL3550.Contig1_All | Wee1-like protein kinase | −8.342 | 0 |
| Unigene21067_All | Doublesex- and mab-3-related transcription factor 2 | 8.150 | 1.289 × 10−04 |
| Unigene40645_All | Doublesex- and mab-3-related transcription factor 4 | −9.348 | 1.183 × 10−204 |
| Unigene39523_All | Forkhead box protein L2 | −8.071 | 2.528 × 10−11 |
| Unigene44534_All | P43 5S RNA-binding protein-like | −10.204 | 0 |
| Unigene8224_All | Sex hormone-binding globulin | 1.450 | 5.326 × 10−34 |
| Unigene43332_All | Follicle stimulating hormone receptor | −8.492 | 1.461 × 10−15 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shan, B.; Wang, L.; Liu, Y.; Yang, C.; Liu, M.; Sun, D.; Huang, P. De Novo Transcriptome Analysis of the Lizard Fish (Saurida elongata): Novel Insights into Genes Related to Sex Differentiation. Appl. Sci. 2022, 12, 11319. https://doi.org/10.3390/app122211319
Shan B, Wang L, Liu Y, Yang C, Liu M, Sun D, Huang P. De Novo Transcriptome Analysis of the Lizard Fish (Saurida elongata): Novel Insights into Genes Related to Sex Differentiation. Applied Sciences. 2022; 12(22):11319. https://doi.org/10.3390/app122211319
Chicago/Turabian StyleShan, Binbin, Liangming Wang, Yan Liu, Changping Yang, Manting Liu, Dianrong Sun, and Pujiang Huang. 2022. "De Novo Transcriptome Analysis of the Lizard Fish (Saurida elongata): Novel Insights into Genes Related to Sex Differentiation" Applied Sciences 12, no. 22: 11319. https://doi.org/10.3390/app122211319
APA StyleShan, B., Wang, L., Liu, Y., Yang, C., Liu, M., Sun, D., & Huang, P. (2022). De Novo Transcriptome Analysis of the Lizard Fish (Saurida elongata): Novel Insights into Genes Related to Sex Differentiation. Applied Sciences, 12(22), 11319. https://doi.org/10.3390/app122211319







